Abstract
The composition and function of platelet-derived Extracellular Vesicles (EVs) in health and in disease are a major topic of investigation in biomedical research. However, efforts to delineate specific molecular repertoires and roles for different types of EVs in the circulation are limited not only by the lack of flow cytometers capable of analyzing submicron- and nano-materials across the full size spectrum of plasma EVs, but also by the lack of standardized methods and reference materials that would permit inter-laboratory reproducibility for these analyses. In this review, we summarize the flow cytometry of EVs, with a focus on platelet vesicles in plasma. In addition to delineating the basic principles that govern what precautions must be considered when using flow cytometry for the analysis of platelet vesicles, we provide an overview for how to standardize, control, annotate, and report EV flow cytometry data reproducibly, while looking forward to a next generation of high sensitivity instruments for the analysis of EVs and other submicron biomaterials in the circulation.
Keywords: Extracellular vesicles, exosomes, microvesicles, flow cytometry, fluorescence
Introduction
While it is now appreciated that all cells release small membrane vesicles, variously referred to as exosomes, microvesicles or extracellular vesicles (EVs), the roles of these vesicles in the circulatory system, in wound healing, and in other biological processes were unrecognized until relatively recently (1–3). One of the earliest accounts of this phenomenon was in blood, where Wolf observed that “platelet dust” had potent pro-coagulant activity(4). Fifty years later, there is a large body of evidence pointing towards the physiological importance of platelet-derived vesicles, but the mechanisms regulating the formation and release of platelet EVs, as well as the molecular bases of their functional effects remain obscure. A primary reason for this is a lack of sensitive and specific methods for the quantitative analysis of these small, heterogeneous membrane particles.
Flow cytometry is a powerful approach to the quantitative analysis of individual particles. The technique is widely used for the analysis of cells, including large (>20 μm) neurons and alveolar macrophages, lymphocytes (~10 μm), and platelets (2–3 μm) (5). Flow cytometry can provide low resolution morphological information about individual cells, including information about relative size (diameter) and internal structure (“granularity”). This information allows the discrimination of small cells with relatively simple structures (such as lymphocytes) and larger cells with relatively more complex internal structures (such as granulocytes or adipocytes). The use of fluorescence probes, in particular fluorescence-labelled antibodies, can provide information on the abundance of specific molecular components of cells and their functional physiology. Platelets represent the smallest “cellular” material that most flow cytometers were generally designed to detect, and multicolor flow cytometry is a useful tool for the characterization of platelets and their receptor repertoires (6).
Flow cytometry has also been used for the analysis of submicron (< 1 μm) EVs. In general, flow cytometry analysis of EVs has followed the same paradigms as lymphocyte analysis, with light scatter being used to detect individual EVs and fluorescence to detect specific molecular components or physiology-related features. However, most flow cytometers were designed for studying cells rather than EVs, which are one to two orders of magnitude smaller than cells. In addition, there is wide variation in the limits of detection for commercially available flow cytometers. Due to this variation, and the fact that signals from EVs challenge the sensitivity of conventional instruments, flow cytometry of EVs has not yet gained the same wide acceptance as flow cytometry of cells. Moreover, there is significant variation in the details of the sample preparation and measurement approach, little agreement on standardization, calibration, and experimental design principles, and a lack of uniformity in data reporting and archiving. This has resulted in many publications in which estimated EV concentrations of similar samples vary by orders of magnitude and in which fluorescence measurements are largely uncalibrated. Without shared calibration methods and metrics, it is impossible to compare data between laboratories, despite widespread recognition of the need to improve reproducibility in preclinical research (7,8).
Meanwhile, the limitations of conventional flow cytometry have become apparent (9), and education regarding basic principles of flow cytometer operation and calibration, as well as sound experimental design principles, have begun to focus on EV analysis. Consequently, consensus is building regarding several key issues, and prospects are brighter for the development of robust and reproducible quantitative analytical methods for the study of EVs from platelets and other cells. In this review we will highlight some of the practical issues regarding flow cytometry of EVs and the implications for quantitative analysis of platelet vesicles.
Practical considerations
A key conceptual step in applying flow cytometry technology to the measurement of EVs is the realization that EVs are not lymphocytes. The current generation of commercial flow cytometers are highly complex, sensitive instruments, that are optimized for the measurement of lymphocytes and other similarly sized cells. The immunophenotyping of immune cells in blood has revolutionized our understanding of the immune system, largely thanks to the large numbers of parameters (“colors”) that can be measured simultaneously on single cells(10–12). Advances in fluorescent probe development, multicolor panel and experimental design principles, and automated data analysis tools have enabled biologists to perform sophisticated measurements with little need for a detailed understanding of the operational details of the flow cytometer. However, a simple extrapolation of approaches that are robust for lymphocyte analysis to the analysis of EVs can result in highly variable and nonsensical data. These artifactual data are not at all apparent to users who do not understand the design and operational limitations of their instruments. To better recognize and avoid these artifacts, and to produce data that is accurate and reproducible, it is instructive to consider the differences between lymphocytes, platelets, and EVs (Table 1) and the implications of these differences for flow cytometry-based measurements.
Table 1.
Differences between cells and extracellular vesicles (EVs)
| Diameter (μm) | Surface area (μm2) | Volume (fL) | Surface antigens | |
|---|---|---|---|---|
| Cell | 10 | 314 | 524 | 103–105 |
| Platelet | 2–3 | 8–12 | 1–2 | 102–104 |
| EV | 0.1 | 0.04 | 0.0005 | 1–103 |
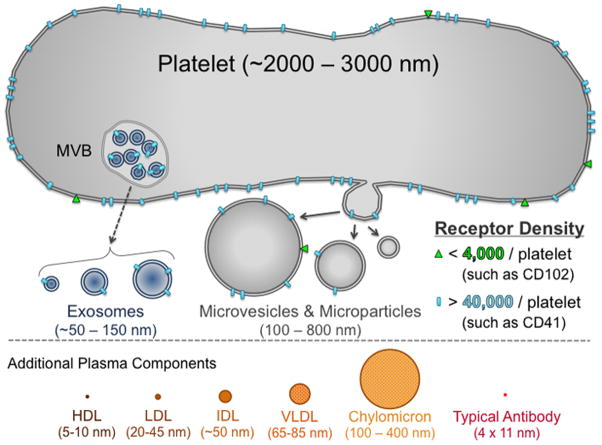
The most obvious difference between platelets and EVs is size (Figure 1). For purposes of illustration, lymphocytes are on the order of 10 μm in diameter while most EVs are on the order of 0.1 μm in diameter. Assuming that both lymphocytes and EVs are spherical, this 100-fold difference in diameter results in a 10,000-fold difference in surface area and a 1,000,000-fold difference in volume between the two particle types. Platelets are discoid rather than spherical, a feature which affects some of the following arguments, but not fundamentally. This difference in size impacts almost every aspect of the flow cytometry measurement.
Figure 1.
A comparison of sizes of platelets, platelet-derived extracellular vesicles, and other lipo-protein particles commonly found in the circulation. Extracellular vesicles, from platelets or cells are typically produced either as exosomes, released from multivesicular bodies (MVB), or as microvesicles or microparticles, shed from the cell surface. As major regulators of hemostasis and wound healing, platelet-derived EVs are commonly studied in human biofluids, such as plasma, where additional submicron particles also are present and must be considered and accounted for (or removed during sample preparation) for any flow cytometric analysis of bio-fluid derived EVs. Figure drawn with objects to approximate relative scales, and high-versus low-receptor densities, for molecules such as CD41 and CD102, respectively, are shown as blue and green labels, drawn to indicate that the low abundance epitopes on platelet-derived may be detectable on the largest of the shed microparticles, but are likely not detectable with conventional flow cytometers.
First, small particles scatter much less light than large particles of the same material. Thus, while individual lymphocytes can be readily discriminated from various sources of background using flow cytometry, EVs cannot. For particles that are smaller than the wavelength of the illuminating laser, light scatter decreases drastically (as the 4th–6th power of the radius) meaning that as EVs get smaller, they may scatter much less light.
Second, assuming a similar surface density of surface receptors and antigens, EVs will bear ~10,000-times fewer surface molecules compared to their cell of origin. Thus, for high abundance cell surface molecules, such as CD41 (also known as integrin α2, GPIIb), which is present at ~40,000 molecules per platelet (13), the expected abundance on a 120 nm platelet EV would be ~200 molecules, which is near the detection limit of the most sensitive commercial flow cytometers. For a low abundance surface molecule, such as CD102 (also known as αLβ2 integrin or Intracellular Adhesion Molecule-2, ICAM-2) and α2β1 (or GPIa-IIa), the estimated number of molecules is ~2000–4000 per platelet(14,15). Consequently, the number of molecules on each platelet EV might be less than 10, which is far below the detection limits of conventional commercial flow cytometers. In addition to biologically active surface receptors, EVs also carry functional cargo, including microRNA and other nucleotides. RNA-specific dyes (such as SytoRNA-Select and Syto-84) have been used to label and quantitate RNA cargo of EVs bound to beads or in other bulk assays. However, attempts to label and detect RNA in individual EVs by flow cytometry have generally failed, likely due to the low abundance of RNA per EV (16) and the short length of RNA. Consequently, the small number of RNA-associated labels result in RNA-labelling levels per EV that typically fall below the limits of detection of commercially available flow cytometers.
Third, assuming a similar concentration of cytoplasmic contents in cells and EVs, the cytoplasmic contents of an EV will be ~1/1,000,000th that of the cell or origin. This means that the fluorescence from fluorescent probes that stain contents in the intra-vesicular volume will be orders of magnitude less bright for EVs compared to the cell of origin. These small signals will also challenge the fluorescence sensitivity of conventional flow cytometers. On the other hand, the small volume of EVs also means that the number of auto-fluorescent compounds will be low. In contrast to the situation for cells, where the predominant source of background is cellular auto-fluorescence (often equaling the intensity of several thousand exogenous fluorophores), the auto-fluorescence of EVs is low. For EV, the fluorescence sensitivity is expected to be limited by other sources of optical and electronic background.
Beyond these factors impacting light scatter and fluorescence sensitivity and resolution, other aspects of flow cytometry differ between EV and cell analysis. Because the size of cells is comparable to the dimensions of the focused laser beam, they can be readily distinguished from cells that are closely spaced or coincident as they pass through the laser. For example, coincidence detection of cells can be identified by the signal pulse width or the relationship between pulse height and area. By contrast, because EVs are much smaller than the focused laser spot, pulse width is not an effective means to identify coincident particles, and artifacts from coincidence (aka “swarm” in the EV analysis field) are pervasive in the literature(17). Thus, conventional methods for “doublet detection” that are used for analysis of cells, need to be replaced in EV analytical protocols with alternate quality control practices. Coincidence detection of EV can be identified by performing serial sample dilutions (18) and ensuring that measured intensities are not dependent on particle concentration.
In addition, platelets and platelet-derived EVs are part of a circulatory system that is comprised of a highly heterogeneous mixture of cells, EVs, lipoproteins, and other soluble materials, which partly overlap in size (Figure 1). Therefore, the analysis of EVs in the circulation requires differentiation between EVs and similar-sized particles. To this end, whereas immunologists commonly report results in terms of percentages of populations (e.g. regulatory T cell expansion is commonly documented by reporting an increase in the percentage of foxp3-positive cells among total CD4+ T cells), extreme caution must be used when selecting units for reporting EV flow cytometric results, due to overlapping detection of lipoproteins and due to differences in limits of detection of different instruments (even the same model, at different locations, or on different days). If 10 laboratories test the same sample and each report the number of EVs with a specific marker per mL, the results are more likely to be consistent than if the laboratories report the percent of EVs with that marker. Volumetric EV enumeration is discussed in more detail below.
When analyzing EVs on instruments designed to measure cells, many of the contradictory and irreproducible results on EV concentration and surface marker expression could have been prevented by considering the differences between cells and EVs. Recognizing these differences, understanding how they impact the measurement of EVs, and understanding the limits of the instrument being used, allows one to avoid the most common and misleading artifacts and obtain reproducible results with conventional instruments. Meanwhile, the field awaits the further development and availability of a next generation of higher sensitivity instruments (19), which demonstrate the potential capabilities and promise of flow cytometers that have been redesigned to provide the fluorescence and light scatter sensitivity and resolution required for robust analysis of EVs and other nanomaterials.
Flow cytometry of platelet vesicles
With the differences between cells and EVs in mind, we turn to the essential components of robust and reproducible EV flow cytometry, which include: sample collection and processing, EV detection, standardization and calibration of EV light scatter and fluorescence measurements, experimental design including necessary controls, and data analysis and reporting. While many of these components will depend upon the specific aims and samples being studied, and the instrumentation with which they are being analyzed, we outline the following general principles that should be considered in the experimental design.
Sample collection and processing
The manner in which a blood sample is collected, processed and stored can have a significant effect on the downstream flow cytometric analysis. Pre-analytic variables have been the subject of numerous studies using various conventional flow cytometry approaches(20–23). Because these conventional approaches have significant limitations, definitive studies have yet to be conducted. However, some general lessons have emerged. Several studies have examined the effect of different anti-coagulants. There is evidence that heparin can induce post-collection release of platelet EVs, thereby altering the sample, while citrate or EDTA does not introduce this artifact. It is generally recommended to process the blood, to remove platelets and cells and produce cell free plasma, as soon as possible after collection. The ISTH-promulgated protocol of centrifuging blood twice for 15′ at 2500xg is followed in a large fraction of published reports (24–26). Several studies have examined the effect of freezing on EV numbers and size and come to different conclusions as to whether this affects the final results. As a practical matter, most studies snap freeze cell free plasma for later analysis. Many groups advocate the addition of thrombin inhibitors (hirudin, PPAK, etc) to prevent spontaneous clotting in vesicle-containing plasma, which would be expected to impact EV analysis results.
The EV staining and washing procedure can also alter the sample and introduce uncertainty into the measurements. No sample processing step enables 100% recovery, and centrifugation steps are especially prone to sample loss. Therefore, it is important to record sample volumes and particle concentrations (if possible) at each step, so that these numbers can be factored in to EV concentration measurements. It should be noted that ultracentrifugation, which is a popular way to wash and concentrate EVs, can induce the formation of EV and protein aggregates that can impact size and concentration measurements (27). The above considerations are relevant to any post-collection EV-related analysis. Because the final word has yet to be written on the most appropriate pre-analysis sample collection and processing, it is important to fully report all details and to monitor future work to determine the optimal conditions for a particular EV analysis.
EV detection
Detection of particles using flow cytometry requires selecting a “triggering” channel (or channels) to alert the data acquisition system that a particle is present in the laser beam. For cells light scatter is the most common trigger channel, as scatter intensities are much larger than background. For small EVs, light scatter is generally below background levels, and light scatter-based triggering either detects only the very largest EVs (>1 μm) and/or also collects data from “noise” events. With regards to platelets, light scatter is commonly used to trigger detection of EVs (particularly “microparticles” or “microvesicles”) that are shed upon activation. Polystyrene beads are often used as reference particles to establish “gates” to identify putative EVs, though the unfortunate myth that bead light scatter can be used to calibrate EV particle size persists among novice users. In fact, particle light scatter is a complex function of not only size, but also of refractive index, illumination wavelength, laser polarization, and angle of detection. The education of users that estimation of particle size from light scatter intensity requires sophisticated analysis is an ongoing effort.
With these caveats about light scatter-based EV detection in mind, extensive efforts to standardize platelet EV measurements using conventional light scatter-based detection have conducted and reported over the past several years, primarily under the auspices of the International Society on Thrombosis and Haemostasis (24–26). In general, these approaches employ uniform polymer beads to standardize a “window of analysis” in the light scatter parameters to facilitate comparison of results over time and across instruments. Combined with fluorescence staining for annexin and CD41, this approach can produce platelet microparticle (PMP) concentration estimates of 2–4 ×103/μL (in a pool of 10 donors) that are reasonably precise (inter-instrument CVs of ~19–46%). Nevertheless, this approach likely detects only the largest and brightest particles (the “tip of the iceberg”) in plasma. Standardization efforts such as these will be essential to the development of rigorous and reproducible PMP measurement methods that can produce data that can be shared and compared among labs.
EV detection can also be triggered by fluorescence, either from specific fluorescent ligands (eg. antibodies or annexin V) (28,29), which can label selected molecules on EVs, or from membrane probes which can label EVs and other membranous particles more generally (30,31). The fluorescence background can be much lower than the light scatter background, resulting in higher detection efficiencies with lower levels of “noise” events. The limit of detection for fluorescence triggering will be determined by the number of fluorophores that label the EV and the ability of the instrument to resolve dim signals above background.
With regard to platelet EVs in plasma, especially notable is the work of Brisson et al. (29), who performed a comprehensive analysis of the relationship between EV size and surface marker expression as measured by immuno cryo-EM and flow cytometry using light scatter and fluorescence triggering. Brisson et al. clearly showed the advantages for detecting EVs bearing specific surface molecules, such as CD41 on PMPs. This approach reported ~2 ×104 PMPs/μL, ten times more than observed for light scatter triggering, but still possibly an underestimate as the lower limit of detection was estimated at ~1000 molecules of CD41. Also notable is the approach of Stoner et al (30), who used a membrane-specific probe to detect and size EVs as small as ~70 nm, and to identify subsets of EVs expressing as few as ~200 ligand-conjugated fluorophores. Such labelling of EV membranes with lipophilic dyes have generally required a wash step to remove free dye that caused unacceptable background. Nevertheless, this approach demonstrates that the careful choice of membrane probe and staining conditions enable to detect membranous particles, including platelet EVs, directly in diluted plasma.
The volume of EVs can also be labeled. For example, fluorogenic substrates, such as carboxyfluorescein diacetate succinimydal ester (CFSE), can be activated and trapped inside EVs. When using CFSE or similar probes for general protein labeling of the EV volume, or lipid-intercalating dyes for labeling the EV surface, the fluorescence signal from EV-associated labels must exceed the signal from various sources of background in order to be detected. Because the background in the fluorescence channels are generally lower than the scatter channels, fluorescence offers distinct advantages over light scattering for triggering the detection of EVs in flow cytometry, especially with instruments for which the scattered light intensity from the EVs is insufficient to fully resolve the EV population from background.
Whether light scatter or fluorescence signals are used to trigger detection, the user is still faced with the decision of how to set the trigger threshold (also called the “discriminator” on some systems). In practice, buffer (or buffer plus staining reagents) can be used to set a threshold level that results in an acceptable level of background events. The number of background events can be used to define a particle number limit of detection (LOD), which, as an example, could be designated as three standard deviations above the mean of the background counts (99% confidence limit). The value of the trigger detection threshold can be expressed in arbitrary units of channel number, or as a calibrated value, such as number of photons or mean equivalent soluble fluorophores (MESF), as is discussed below. Details about the trigger threshold are important to report, because individual flow cytometers and sample preparation methods have different levels of background, thereby affecting the optimal settings and the amount of particles being detected. In colloquial terms, this is the problem of reporting how much of the “iceberg” is being detected.
Experimental design
The differences between cells and EVs also necessitate some differences in experimental design when measuring EVs. The issue of detection specificity is a challenge as it can be difficult to distinguish the small, dim signals from EVs, from other small, dim signals originating from various sources of background from the instrument, reagents or sample. The appropriate control experiments (Table 2) can help to determine the contribution of these background signals and guide efforts to reduce them.
Table 2.
Common artifacts in EV analysis and corresponding control experiments
| Artifact | Cause | Control | Remedy |
|---|---|---|---|
| False events | Background particles | Buffer only | Filtration, ultracentrifugation, UV treatment |
| False events | Fluorescent label aggregates | Buffer plus staining reagents | Centrifugation, filtration, SEC |
| False events | Non-EV particulates, including proteins and insoluble lipoproteins | Sample plus detergent | |
| Coincidence | High particle concentration | Serial dilution of sample to show decrease in event rate but no change in intensity | Dilute sample |
First, all fluids, including sheath fluids and dilution and wash buffers, contain entities that fluoresce and/or scatter light. In high pressure systems (such as commercially available high sensitivity jet-in-air sorters), abrupt depressurization of the sheath fluid can result in degassing of the buffer and generation of micro(nano)-bubbles, which may be detected by scattered light. Buffer-only controls are therefore commonly used to set detection trigger thresholds and should be part of any results presentation. Additional background sources that might be introduced during staining include fluorescent antibody aggregates (32) and calcium phosphate micro-precipitates (33), which can be mistaken for EVs. Staining reagent-only controls (i.e. controls with staining reagents and sample buffer, without any sample) can identify these background sources and evaluate the effectiveness of filtration or centrifugation in reducing them. Even after extensive filtering or centrifugation, background sources can remain. A useful control sample to discriminate non-EV background from real EVs is treatment with detergent (e.g. 0.05% Triton X-100), which will solubilize EVs, leaving only non-membranous particles(34). When measuring EVs directly in a biofluid, such as plasma, one must also be cognizant of the presences of other similarly-sized particles in blood, such as lipoproteins (including chylomicrons) (35). Lipoproteins are partially detergent soluble, whereas immunoglobulin and protein complexes(36) are not. While EVs may be distinguished from these other particles by the presence of specific surface markers, it is probably best to use plasma from fasted subjects where possible(37), as is often done for other blood chemistry analysis. The recognition of these artifacts and steps to minimize or eliminate them have been described in the literature, and it is the responsibility of authors and reviewers to ensure that future published work does not repeat past mistakes.
Coincidence is a phenomenon that results from the occurrence of multiple particles in the measurement probe volume at the same time. Coincidence is a purely statistical phenomenon that depends on the particle concentration in the sample and instrument-specific attributes (such as laser spot size and signal processing attributes). When particles are large compared to the height of the focused laser beam, it is a trivial matter to identify coincident events and exclude them from analysis. When particles are small relative to the laser beam spot, as is the case for EVs, it is much more difficult to distinguish events triggered by coincident particles from those triggered by single particles. The resulting artifact, sometimes referred to as “swarm” detection, is a pervasive in the literature and results in the underestimation of the true particle count and an over estimation of particle brightness. The only way to minimize the frequency of coincidence is to adjust the concentration of the particles being measured. When the particle concentration is not known, as is typical in EV measurement applications, it is necessary to perform serial dilutions on samples. As is expected for single particle measurements, serial dilutions should demonstrate that the event detection rate decreases in response to dilution, but that the measured optical intensities (fluorescence and/or light scatter) of those events do not change.
Standardization, calibration, and data reporting
As the perfect EV measurement approach has yet to be developed, and the literature is replete with “novel” methods for the analysis of EVs, standardization in the reporting of key EV properties is essential. For both research and clinical applications, these properties may include (Table 3) the number (and concentration) of EVs detected, the size distribution of those EVs, the numbers of EVs detected bearing a particular target molecule, the number of target molecules detected, and the limit of detection of the analysis method. The standardization and calibration of each of these measurements involves different considerations, but can be readily adapted from established protocols for cell analysis, with appropriate modifications to account for the differences between cells and vesicles.
Table 3.
Reportable quantities in EV analysis and relevant calibrators and standards
| Measurement | Derived from | Calibrator or standards | Reportable Values |
|---|---|---|---|
| EV concentration | Number of events detected per volume of fluid | Beads of known concentration to calibrate sample flow rate | EVs/uL |
| EV size | Signal intensity | EV preps of known size (determined by orthogonal method, such as NTA or TRPS) or by calculation using standards of known size and refractive index. | Diameter, surface area, or volume of detected EVs |
| EV fluorescence positive concentration | Number of events detected above a threshold per volume of fluid | Beads of known concentration to calibrate sample flow rate plus negative control sample to set threshold for positivity | Positive EVs/uL above a threshold |
| EV fluorescence brightness/antibody number | Signal intensity | Standard particles with calibrated intensity or antibody capture capacity | Mean fluorescence intensity (MESF) or antibodies bound per EV |
Particle number and concentration
The number of detected EVs can be derived from the number of detected events after accounting for non-EV events resulting from other particles or noise sources. The number and source of background events can be estimated from buffer-only and reagent-only controls. For EVs, which are detergent soluble, detergent treatment of samples can provide further confirmation of the number of EV-specific events detected, and is a valuable control.
The EV concentration is estimated using the volume of sample measured and considering any concentration or dilution steps introduced during sample preparation. Some instruments feature calibrated volumetric sample rates, which enable calculation of the volume measured from the time of sample analysis. There are several approaches to estimating the sample volumetric flow rate, including the measurement of beads at a known concentration for a fixed amount of time, which is simple to implement. With knowledge of the volume measured and the number of EV-specific events detected, the concentration of EVs in the processed sample can be readily calculated. Dilutions and concentration of EVs during sample preparation will depend on the sample preparation approach, and should be recorded and reported.
Particle size
One of the distinguishing features of EVs is their small size. Much has been made of the difference in size between “exosomes” and “microvesicles,” in spite of the fact the particles isolated by methods purported to enrich for these two classes of EVs have size distributions that substantially overlap (38,39). With regard to flow cytometry of EVs, the published literature is littered with examples of ad hoc and invalid “size calibration” of light scatter using polymer beads as size standards to estimate EV size. In recent years, several publications have demonstrated that, while polymer beads may be useful to establish a standardized “window of analysis” between similar instruments, estimating particle size from light scatter intensity requires consideration of particle refractive index, illumination wavelength, and light collection angle. Published examples have illustrated the use of light scattering models that use these considerations to estimate particle size from light scatter measurements down to the limits of light scatter detection of EVs, which is on the order of 500 nm for most commonly used flow cytometers. More sensitive detection of even smaller particles, 100–200 nm for certain commercially available instruments with optimized light scatter sensitivity, and 30–50 nm for next-generation instruments (19), have been demonstrated for beads and purified EVs in purified buffers, raising the prospect that these approaches might be applied to real world samples such as the measurement of platelet EVs in plasma.
An alternative approach has been described based on fluorescence, in which a membrane-specific probe stains EVs in proportion to their surface area, and where the use of synthetic membrane EVs as size standards enable the measurement of size of EVs as small as ~70 nm (30,38) It is valuable to augment flow cytometry-based size estimates with independent determinations using orthogonal single particle sizing methods, such as nanoparticle tracking analysis (NTA)(40,41), resistive pulse sensing (RPS)(42), or electron microscopy(43–45).
Particle brightness
The biggest reason to use flow cytometry to analyze EVs is the ability to use fluorescence-labelled antibodies or other ligands to detect EV components and cargo. Here, the small size and antigen numbers challenge the sensitivity of conventional lymphocyte analyzers, so that the number of antibody-positive EVs measured in a sample on one instrument may differ from that measured on another instrument. Thus the calibration of fluorescence measurements and the standardization of reporting is essential to allow comparisons of results obtained on different instruments.
Fortunately, the tools for calibration of flow cytometry fluorescence intensity measurements are well established, and include commercially-available calibrated intensity standard particles and software that make the conversion of arbitrary intensities (channel numbers) into absolute intensities with units of photoelectrons or Mean Equivalent Soluble Fluorochromes (MESF) (5,46–48). For biological interpretation, expressing fluorescence intensity in units of MESF is the most useful, since this can be related to the number of antibodies bound per EV in a fairly straightforward manner. Beads with intensities calibrated in MESF units are available for several popular fluorophores, and provide a way to compare measurements made in different laboratories and using different instruments. For fluorophores for which there are no commercially available intensity standards, multi-intensity hard-dyed beads can serve as useful reference particles to standardize brightness measurements across labs. A third approach involves using antibody-capture beads with calibrated binding capacities that can, in principle, be used for antibodies labeled with any fluorophore. Although these commercially available beads were designed to calibrate measurements of cells, and the availability of beads designed to calibrate EV measurements are an unmet need, any of these approaches is preferable to reporting results as uncalibrated mean fluorescence intensities, or MFIs.
Marker positivity
In flow cytometry of lymphocytes or other cells, immunofluorescence staining results are often expressed as the “percent positive,” which is useful and appropriate when all of the cells can be detected and there is little uncertainty about the denominator. However, when there is uncertainty about how many of the particles of interest are actually being detected, as is the case with EV detection by flow cytometry, the percent positive will depend on both the number positive and on the number detected. Consequently, expressing results as percentages has little use and may actually be misleading. For this reason, it is important to report the number (or better still, concentration) of positive EVs. Moreover, because the number of positive EVs will depend on the sensitivity of the instrument, the brightness of the reagents, and background signals from various sources, the number (or concentration) of positive events should be presented in terms of a threshold brightness for positivity, ideally expressed in units of MESF as described above.
Status and prospects
While it is clear that EVs from platelets and cells are involved in many aspects of health and disease, our ability to understand the mechanistic roles they play and to exploit this to develop better diagnostics and treatments is still fairly limited. A key to improving our understanding of the functions of EVs and their use in clinical practice is improved methods for their quantitative analysis. Flow cytometry is a powerful single particle analysis tool, but flow cytometry instruments and methods developed for cell analysis are not optimal for the analysis of small, dim EVs. These limitations, combined with differences in sample preparation, experimental design, detection and gating strategies, and data reporting have produced plasma PMP concentration estimates in the peer-reviewed literature that vary by orders of magnitude. The application of appropriate experimental design, controls, standards and calibration are essential to reduce this variability and harness the strengths of flow cytometry for single EV analysis in a way that allows rigorous and reproducible measurements that can be compared among labs.
Fortunately, the field is increasingly aware of these limitations, and an improved appreciation of the importance of standardization in measurement approaches, instrument calibration and data reporting will enable the routine comparison of results across labs. To facilitate this, the International Society for Extracellular Vesicles (ISEV), together with the International Society for Advancement of Cytometry (ISAC) and the international Society on Thrombosis and Haemostastis (ISTH), is leading a working group on EV flow cytometry to standardize fluorescence calibration and data reporting for EV analysis. Such standardization will accelerate the adaptation and validation of new, high sensitivity instruments for the analysis of EVs in biofluids, including platelets EVs in plasma.
Footnotes
Declaration of interest: The authors report no declarations of interest.
Contributor Information
John P Nolan, Scintillon Institute, San Diego, CA 92121.
Jennifer C Jones, 9000 Rockville Pike, 10-CRC-B2-3500, National Institutes of Health, Bethesda, MD 20892.
Literature Cited
- 1.Kim HK, Song KS, Chung JH, Lee KR, Lee SN. Platelet microparticles induce angiogenesis in vitro. Br J Haematol. 2004;124:376–84. doi: 10.1046/j.1365-2141.2003.04773.x. [DOI] [PubMed] [Google Scholar]
- 2.Boilard E, Nigrovic PA, Larabee K, Watts GF, Coblyn JS, Weinblatt ME, Massarotti EM, Remold-O’Donnell E, Farndale RW, Ware J, et al. Platelets amplify inflammation in arthritis via collagen-dependent microparticle production. Science. 2010;327:580–3. doi: 10.1126/science.1181928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brill A, Dashevsky O, Rivo J, Gozal Y, Varon D. Platelet-derived microparticles induce angiogenesis and stimulate post-ischemic revascularization. Cardiovasc Res. 2005;67:30–8. doi: 10.1016/j.cardiores.2005.04.007. [DOI] [PubMed] [Google Scholar]
- 4.Wolf P. The nature and significance of platelet products in human plasma. British journal of haematology. 1967;13:269–288. doi: 10.1111/j.1365-2141.1967.tb08741.x. [DOI] [PubMed] [Google Scholar]
- 5.Shapiro HM. Practical Flow Cytometry. John Wiley & Sons; 2005. [Google Scholar]
- 6.Hedley BD, Llewellyn-Smith N, Lang S, Hsia CC, MacNamara N, Rosenfeld D, Keeney M. Combined accurate platelet enumeration and reticulated platelet determination by flow cytometry. Cytometry B Clin Cytom. 2015;88:330–7. doi: 10.1002/cyto.b.21245. [DOI] [PubMed] [Google Scholar]
- 7.Collins FS, Tabak LA. Policy: NIH plans to enhance reproducibility. Nature. 2014;505:612–3. doi: 10.1038/505612a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Baker M. 1,500 scientists lift the lid on reproducibility. Nature. 2016;533:452–4. doi: 10.1038/533452a. [DOI] [PubMed] [Google Scholar]
- 9.Erdbrugger U, Lannigan J. Analytical challenges of extracellular vesicle detection: A comparison of different techniques. Cytometry A. 2016;89:123–34. doi: 10.1002/cyto.a.22795. [DOI] [PubMed] [Google Scholar]
- 10.Chattopadhyay PK, Hogerkorp CM, Roederer M. A chromatic explosion: the development and future of multiparameter flow cytometry. Immunology. 2008;125:441–449. doi: 10.1111/j.1365-2567.2008.02989.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.De Rosa SC, Herzenberg LA, Roederer M. 11-color, 13-parameter flow cytometry: identification of human naive T cells by phenotype, function, and T-cell receptor diversity. Nat Med. 2001;7:245–248. doi: 10.1038/84701. [DOI] [PubMed] [Google Scholar]
- 12.Perfetto SP, Chattopadhyay PK, Roederer M. Seventeen-colour flow cytometry: unravelling the immune system. Nat Rev Immunol. 2004;4:648–655. doi: 10.1038/nri1416. [DOI] [PubMed] [Google Scholar]
- 13.Bennett JS. The molecular biology of platelet membrane proteins. Semin Hematol. 1990;27:186–204. [PubMed] [Google Scholar]
- 14.Clemetson KJ, Clemetson JM. Platelet GPIb complex as a target for anti-thrombotic drug development. Thromb Haemost. 2008;99:473–9. doi: 10.1160/TH07-12-0718. [DOI] [PubMed] [Google Scholar]
- 15.Diacovo TG, deFougerolles AR, Bainton DF, Springer TA. A functional integrin ligand on the surface of platelets: intercellular adhesion molecule-2. J Clin Invest. 1994;94:1243–51. doi: 10.1172/JCI117442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chevillet JR, Kang Q, Ruf IK, Briggs HA, Vojtech LN, Hughes SM, Cheng HH, Arroyo JD, Meredith EK, Gallichotte EN, et al. Quantitative and stoichiometric analysis of the microRNA content of exosomes. Proc Natl Acad Sci U S A. 2014;111:14888–93. doi: 10.1073/pnas.1408301111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.van der Pol E, Boing AN, Harrison P, Sturk A, Nieuwland R. Classification, Functions, and Clinical Relevance of Extracellular Vesicles. Pharmacological Reviews. 2012;64:676–705. doi: 10.1124/pr.112.005983. [DOI] [PubMed] [Google Scholar]
- 18.Nolan JP, Stoner SA. A trigger channel threshold artifact in nanoparticle analysis. Cytometry Part A. 2013;83A:301–305. doi: 10.1002/cyto.a.22255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhu S, Ma L, Wang S, Chen C, Zhang W, Yang L, Hang W, Nolan JP, Wu L, Yan X. Light-scattering detection below the level of single fluorescent molecules for high-resolution characterization of functional nanoparticles. ACS Nano. 2014;8:10998–1006. doi: 10.1021/nn505162u. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chandler WL. Measurement of microvesicle levels in human blood using flow cytometry. Cytometry Part B: Clinical Cytometry. 2016 doi: 10.1002/cyto.b.21343. [DOI] [PubMed] [Google Scholar]
- 21.Lacroix R, Judicone C, Poncelet P, Robert S, Arnaud L, Sampol J, DIGNAT-GEORGE F. Impact of pre-analytical parameters on the measurement of circulating microparticles: towards standardization of protocol. Journal of Thrombosis and Haemostasis. 2012;10:437–446. doi: 10.1111/j.1538-7836.2011.04610.x. [DOI] [PubMed] [Google Scholar]
- 22.Yuana Y, Bertina RM, Osanto S. Pre-analytical and analytical issues in the analysis of blood microparticles. Thrombosis and Haemostasis. 2011;105:396. doi: 10.1160/TH10-09-0595. [DOI] [PubMed] [Google Scholar]
- 23.Yuana Y, Böing AN, Grootemaat AE, van der Pol E, Hau CM, Cizmar P, Buhr E, Sturk A, Nieuwland R. Handling and storage of human body fluids for analysis of extracellular vesicles. Journal of extracellular vesicles. 2015;4 doi: 10.3402/jev.v4.29260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cointe S, Judicone C, Robert S, Mooberry M, Poncelet P, Wauben M, Nieuwland R, Key N, Dignat-George F, Lacroix R. Standardization of microparticle enumeration across different flow cytometry platforms: results of a multicenter collaborative workshop. Journal of Thrombosis and Haemostasis. 2016 doi: 10.1111/jth.13514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lacroix R, Judicone C, Mooberry M, Boucekine M, Key NS, Dignat-George F the ISSCW. Standardization of pre-analytical variables in plasma microparticle determination: results of the International Society on Thrombosis and Haemostasis SSC Collaborative workshop. Journal of Thrombosis and Haemostasis. 2013;11:1190–1193. doi: 10.1111/jth.12207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lacroix R, Robert S, Poncelet P, Kasthuri R, Key N, Dignat-George F. Standardization of platelet-derived microparticle enumeration by flow cytometry with calibrated beads: results of the International Society on Thrombosis and Haemostasis SSC Collaborative workshop. Journal of Thrombosis and Haemostasis. 2010;8:2571–2574. doi: 10.1111/j.1538-7836.2010.04047.x. [DOI] [PubMed] [Google Scholar]
- 27.Linares R, Tan S, Gounou C, Arraud N, Brisson AR. High-speed centrifugation induces aggregation of extracellular vesicles. Journal of extracellular vesicles. 2015:4. doi: 10.3402/jev.v4.29509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Arraud N, Gounou C, Linares R, Brisson AR. A Simple Flow Cytometry Method Improves the Detection of Phosphatidylserine-Exposing Extracellular Vesicles. Journal of Thrombosis and Haemostasis. 2014;13:237–247. doi: 10.1111/jth.12767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Arraud N, Gounou C, Turpin D, Brisson AR. Fluorescence triggering: A general strategy for enumerating and phenotyping extracellular vesicles by flow cytometry. Cytometry Part A. 2016;89:184–195. doi: 10.1002/cyto.a.22669. [DOI] [PubMed] [Google Scholar]
- 30.Stoner SA, Duggan E, Condello D, Guerrero A, Turk JR, Narayanan PK, Nolan JP. High sensitivity flow cytometry of membrane vesicles. Cytometry Part A. 2016;89:196–206. doi: 10.1002/cyto.a.22787. [DOI] [PubMed] [Google Scholar]
- 31.van der Vlist EJ, Nolte EN, Stoorvogel W, Arkesteijn GJ, Wauben MH. Fluorescent labeling of nano-sized vesicles released by cells and subsequent quantitative and qualitative analysis by high-resolution flow cytometry. nature protocols. 2012;7:1311–1326. doi: 10.1038/nprot.2012.065. [DOI] [PubMed] [Google Scholar]
- 32.Aass HCD, Øvstebø R, Trøseid AMS, Kierulf P, Berg JP, Henriksson CE. Fluorescent particles in the antibody solution result in false TF-and CD14-positive microparticles in flow cytometric analysis. Cytometry Part A. 2011;79:990–999. doi: 10.1002/cyto.a.21147. [DOI] [PubMed] [Google Scholar]
- 33.Larson MC, Luthi MR, Hogg N, Hillery CA. Calcium-phosphate microprecipitates mimic microparticles when examined with flow cytometry. Cytometry Part A. 2013;83:242–250. doi: 10.1002/cyto.a.22222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Osteikoetxea X, Sodar B, Nemeth A, Szabo-Taylor K, Paloczi K, Vukman KV, Tamasi V, Balogh A, Kittel A, Pallinger E, et al. Differential detergent sensitivity of extracellular vesicle subpopulations. Org Biomol Chem. 2015;13:9775–82. doi: 10.1039/c5ob01451d. [DOI] [PubMed] [Google Scholar]
- 35.Sódar BW, Kittel Á, Pálóczi K, Vukman KV, Osteikoetxea X, Szabó-Taylor K, Németh A, Sperlágh B, Baranyai T, Giricz Z, et al. Low-density lipoprotein mimics blood plasma-derived exosomes and microvesicles during isolation and detection. Scientific Reports. 2016;6:24316. doi: 10.1038/srep24316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.György B, Módos K, Pállinger É, Pálóczi K, Pásztói M, Misják P, Deli MA, Sipos Á, Szalai A, Voszka I, et al. Detection and isolation of cell-derived microparticles are compromised by protein complexes resulting from shared biophysical parameters. Blood. 2011;117:e39–e48. doi: 10.1182/blood-2010-09-307595. [DOI] [PubMed] [Google Scholar]
- 37.Witwer KW, Buzás EI, Bemis LT, Bora A, Lässer C, Lötvall J, Nolte-’t Hoen EN, Piper MG, Sivaraman S, Skog J, et al. Standardization of sample collection, isolation and analysis methods in extracellular vesicle research. Journal of Extracellular Vesicles. 2013;2 doi: 10.3402/jev.v2i0.20360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Akers JC, Ramakrishnan V, Nolan JP, Duggan E, Fu C-C, Hochberg FH, Chen CC, Carter BS. Comparative Analysis of Technologies for Quantifying Extracellular Vesicles (EVs) in Clinical Cerebrospinal Fluids (CSF) PloS one. 2016;11:e0149866. doi: 10.1371/journal.pone.0149866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kanada M, Bachmann MH, Hardy JW, Frimannson DO, Bronsart L, Wang A, Sylvester MD, Schmidt TL, Kaspar RL, Butte MJ. Differential fates of biomolecules delivered to target cells via extracellular vesicles. Proceedings of the National Academy of Sciences. 2015;112:E1433–E1442. doi: 10.1073/pnas.1418401112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dragovic RA, Gardiner C, Brooks AS, Tannetta DS, Ferguson DJ, Hole P, Carr B, Redman CW, Harris AL, Dobson PJ. Sizing and phenotyping of cellular vesicles using Nanoparticle Tracking Analysis. Nanomedicine: Nanotechnology, Biology and Medicine. 2011;7:780–788. doi: 10.1016/j.nano.2011.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Gardiner C, Ferreira YJ, Dragovic RA, Redman CW, Sargent IL. Extracellular vesicle sizing and enumeration by nanoparticle tracking analysis. Journal of Extracellular Vesicles. 2013;2:19671. doi: 10.3402/jev.v2i0.19671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Coumans FAW, van der Pol E, Böing AN, Hajji N, Sturk G, van Leeuwen TG, Nieuwland R. Reproducible extracellular vesicle size and concentration determination with tunable resistive pulse sensing. Journal of Extracellular Vesicles. 2014;3:25922. doi: 10.3402/jev.v3.25922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Arraud N, Linares R, Tan S, Gounou C, Pasquet JM, Mornet S, Brisson AR. Extracellular Vesicles from Blood Plasma: Determination of their morphology, size, phenotype and concentration. Journal of Thrombosis and Haemostasis. 2014;12:614–627. doi: 10.1111/jth.12554. [DOI] [PubMed] [Google Scholar]
- 44.van der Pol E, Coumans FAW, Grootemaat AE, Gardiner C, Sargent IL, Harrison P, Sturk A, van Leeuwen TG, Nieuwland R. Particle size distribution of exosomes and microvesicles determined by transmission electron microscopy, flow cytometry, nanoparticle tracking analysis, and resistive pulse sensing. Journal of Thrombosis and Haemostasis. 2014;12:1182–1192. doi: 10.1111/jth.12602. [DOI] [PubMed] [Google Scholar]
- 45.Yuana Y, Koning RI, Kuil ME, Rensen PC, Koster AJ, Bertina RM, Osanto S. Cryo-electron microscopy of extracellular vesicles in fresh plasma. Journal of Extracellular Vesicles. 2013;2:21494. doi: 10.3402/jev.v2i0.21494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Schwartz A, Gaigalas AK, Wang L, Marti GE, Vogt RF, Fernandez-Repollet E. Formalization of the MESF unit of fluorescence intensity. Cytometry Part B: Clinical Cytometry. 2004;57:1–6. doi: 10.1002/cyto.b.10066. [DOI] [PubMed] [Google Scholar]
- 47.Schwartz A, Wang L, Early E, Gaigalas A, Zhang Y-z, Marti GE, Vogt RF. Quantitating fluorescence intensity from fluorophore: The definition of MESF assignment. Journal of Research-National Institute of Standards and Technology. 2002;107:83–92. doi: 10.6028/jres.107.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang L, Gaigalas AK, Abbasi F, Marti GE, Vogt RF, Schwartz A. Quantitating fluorescence intensity from fluorophores: practical use of MESF values. JOURNAL OF RESEARCH-NATIONAL INSTITUTE OF STANDARDS AND TECHNOLOGY. 2002;107:339–354. doi: 10.6028/jres.107.027. [DOI] [PMC free article] [PubMed] [Google Scholar]