Abstract
Purpose
Up to 50% of postmenopausal breast cancer survivors taking aromatase inhibitors (AIs) experience AI-associated arthralgias, or joint pain, which causes many to stop taking AIs and may inhibit exercise, despite known health benefits. We thus evaluated exercise adherence and factors associated with better exercise adherence in breast cancer survivors experiencing AI-induced arthralgia in the Hormones and Physical Exercise (HOPE) year-long randomized controlled trial.
Methods
We included the 61 HOPE women randomized to exercise (150 min/wk of moderate-intensity aerobic exercise and twice-weekly supervised strength training). Our main outcomes were aerobic exercise measured with daily activity logs, attendance at supervised exercise sessions, and changes in cardiorespiratory fitness, measured maximal oxygen consumption (VO2max). We examined means and standard deviations (SD) for exercise adherence by demographic and medical characteristics and used the t-test for mean differences. We also examined predictors of adherence using linear regression.
Results
On average, at the end of the year-long trial, women reported 119 (SD 78) min/wk of moderate-intensity aerobic exercise and participated in 70% of supervised exercise training sessions. After adjustment for other factors that influence adherence, at 6-months post-randomization only baseline VO2max was associated with higher aerobic exercise levels and at 12-months only older age predicted better supervised exercise training attendance.
Conclusions
Breast cancer survivors taking AIs and experiencing arthralgia are able to initiate and maintain a yearlong exercise program, regardless of other factors that influence activity levels.
Implications
Breast cancer survivors can exercise at levels that have been shown to improve AI-associated arthralgia.
Keywords: physical activity, exercise, breast cancer, aromatase inhibitors, arthralgia
Introduction
There are an estimated 2.8 million breast cancer survivors in the U.S. [2]. In 2004 aromatase inhibitors (AIs) became standard of care for postmenopausal women with hormone receptor positive breast cancer [7], but recent studies have found that approximately 20% of these women discontinue AI therapy within the first year of use and up to 50% do not take AIs regularly, primarily because of side effects [10, 18]. The most common AI side effect is arthralgia, defined as pain or stiffness in the joints [10]. Both non-adherence to and early discontinuation of AIs have been shown to be independent predictors of mortality [12], and there are few established effective treatments for AI-induced arthralgia [8]. Thus, it is essential to identify effective interventions that alleviate arthralgia, which may in turn improve AI adherence.
The American College of Sports Medicine, the American Cancer Society, and the US Department of Health and Human Services issued exercise guidelines recommending 150 minutes of moderate-intensity or 75 minutes of vigorous-intensity aerobic physical activity weekly based on findings that activity is associated with lower risks of all-cause and cancer-specific mortality, health-related fitness, patient-reported outcomes, lymphedema and comorbid conditions [1, 3, 21]. Physical activity in the breast cancer survivor population, and specifically among women on AIs, is paramount to improving health outcomes in this population. However, few breast cancer survivors engage in recommended levels of physical activity [5, 14]. Objective measures of cardiorespiratory fitness such as VO2max have also been associated with lower risk of cardiovascular disease mortality [11], cancer mortality [22], and in cross-sectional studies of cardiovascular function. At diagnosis, cancer survivors showed an approximately 30% lower VO2max, compared with age- and sex-matched sedentary controls without a history of cancer [15, 16].
The recently completed Hormones and Physical Exercise (HOPE) Study examined the effect of a yearlong exercise intervention vs. usual care on severity of AI-induced arthralgia in 121 women taking AIs and experiencing arthralgia [13]. At 12-months, AI-induced arthralgia, assessed via the Brief Pain Inventory worst joint pain score, decreased by 29% among breast cancer survivors randomized to exercise vs. a 3% increase among breast cancer survivors randomized to usual care (p < .0001).
The HOPE study was the first randomized trial of exercise in breast cancer survivors experiencing AI-induced arthralgia, making it an ideal population for describing exercise adherence, fitness and exercise predictors. Thus, the aims of this paper were to: 1) describe the HOPE exercise intervention conducted in breast cancer survivors taking AIs and experiencing AI-induced arthralgia; 2) report 6- and 12-month HOPE exercise adherence and cardiorespiratory fitness changes; and 3) identify predictors of exercise adherence.
Methods
Eligibility and Recruitment
The details of this trial have been previously described [13]. In short, breast cancer survivors were recruited between June 1, 2010 and December 30, 2012 from 5 hospitals in Connecticut (CT) through the Rapid Case Ascertainment Shared Resource of the Yale Cancer Center (RCA), a field arm of the CT Tumor Registry.
Eligible participants were physically inactive (< 90 min/wk of physical activity in the past six months and no strength training in the past year), postmenopausal women, diagnosed 0.5-4.0 years prior to enrollment with hormone receptor positive Stage I to III breast cancer, and taking an AI for at least six months. Participants had to be experiencing arthralgias for at least two months that were at least mild in severity (i.e., a score of at least 3 out of 10 on the worst pain item of the Brief Pain Inventory (BPI)) [6]. Women were eligible if their arthralgias started after initiation of an AI or if they had pre-existing joint pain that was exacerbated by AI use.
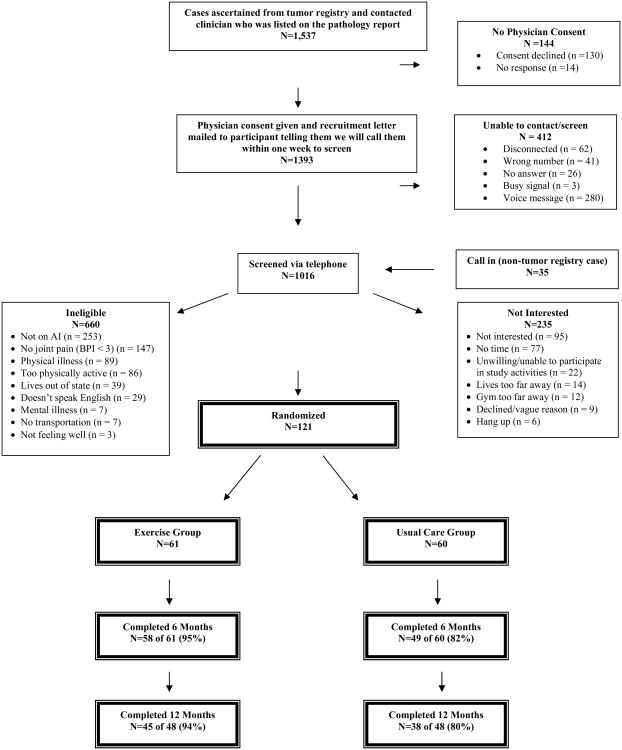
A total of 121 women were randomized, with 61 randomized to exercise and 60 randomized to usual care. However, given funding cuts in the final year, the last 25 women (13 exercisers and 12 usual care) of the 121 women recruited were enrolled into a 6-month rather than a 12-month trial. Thus, their study compliance is based on 6-month data (see Figure 1).
Figure 1. Flow of participants through the HOPE Study.
All study procedures, including written informed consent, were approved by the Yale School of Medicine Human Investigation Committee and Connecticut Department of Public Health Human Investigation Committee.
Exercise Intervention
The yearlong exercise intervention was a combination of a twice-weekly supervised aerobic and resistance training program (under the supervision of an American College of Sports Medicine certified cancer exercise trainer) at a local health club and a home-based aerobic exercise program [23]. Participants wore heart rate monitors during each workout. Following each exercise session, participants recorded the type, duration, and average heart rate during exercise, in daily physical activity logs, as a measure of exercise adherence [4]. Participants returned logs to the exercise trainers at the end of each week. Exercise trainers recorded attendance (yes/no) to the supervised sessions.
The aerobic exercise intervention consisted primarily of brisk walking (treadmill or outside), although participants could choose other aerobic exercise such as stationary bicycling, and was performed during the twice-weekly supervised exercise sessions as well as at home. The goal was to participate in at least 150 min/wk of aerobic exercise, in accordance with current exercise recommendations for cancer survivors. Exercise started at 50% of maximal heart rate (determined from VO2max testing) and increased over the first month to 60-80% of maximal heart rate for the study duration. The strength training protocol consisted of six exercises (e.g., bench press, latissimus pull down, seated row, leg press, leg extension, and leg curl) performed for 8-12 repetitions for three sets. Participants progressed up to three sets per exercise over the first month. After two sessions during which a participant lifted the same weight 12 times during each set, the weight was then increased by the smallest possible increment. There were no adverse events associated with the exercise program.
Outcome Measures
Exercise Adherence
Our primary outcome measures were 6- and 12-month adherence to the exercise intervention goal of 150 min/wk of aerobic exercise measured via the daily activity logs and participation in the twice-weekly supervised exercise sessions.
Cardiorespiratory Fitness
Change in cardiorespiratory fitness from baseline to end of study was examined as a secondary outcome measure. Cardiorespiratory fitness was measured at baseline and end of study with a standard VO2 max treadmill test (including 12-lead electrocardiogram). Expired gases were analyzed using a metabolic measurement cart (CPX-D; Medical Graphics, St Paul, MN). Peak oxygen consumption was determined by taking the highest values during a 15-second period. We used a modified “Branching Treadmill Protocol” whereby participants began at a 3.0 mph walking speed and 0% grade. After two minutes the speed was increased to 3.5 mph with no change in grade. Thereafter, the grade was increased by 3% every two min. Oxygen consumption (VO2), carbon dioxide production (VCO2), and flow rate were measured continuously. Efforts were made to have participants safely exercise to maximal levels (e.g., Respiratory exchange ratio (RER) > 1.0). The test was ended when the participant indicated that she could not continue any further, or if the supervising physician recommended halting the exercise for medical concerns. Maximal heart rate and reasons for stopping the test were recorded, and cardiorespiratory measures (VO2max, VCO2max, RER, and MET) were recorded.
Covariate Measures
Demographics and Medical History
Medical record reviews and an interviewer-administered questionnaire were used to determine disease stage, surgery, adjuvant therapy, endocrine therapy and comorbidities. To determine eligibility based on baseline physical activity levels, an interviewer-administered questionnaire assessing the duration, frequency and intensity of 20 recreational activities over the prior 6 months was completed [13].
Height and Weight
Height (stadiometer) and weight (digital scale, no shoes) were measured at baseline, 6-, and 12-months. All measurements were taken twice and averaged. Body mass index (BMI) was calculated as kg/m2.
Brief Pain Inventory (BPI)
The BPI is a 14-item questionnaire, developed for use in cancer patients, that assesses pain over the past week, reported on a 0-10 scale [6]. Worst pain is categorized as mild pain (score of 3-4), moderate pain (score of 5-7) or severe pain (score of 8-10). The BPI is the most common, valid and reliable measure to assess pain in cancer survivors (Cronbach alpha and test-retest reliability scores both greater than 0.80) [6]. The BPI was modified to capture joint pain and stiffness by adding the term “joint pain/stiffness” rather than just the word “pain” throughout the questionnaire.
Statistical analysis
This analysis included the 61 women randomized to exercise, with 6-month intent-to-treat exercise adherence reported for 61 women, and 12-month intent-to-treat exercise adherence data reported for the 48 women who were randomized to the 12-month trial. We calculated means and standard deviations (SDs) of aerobic exercise min/wk and % of supervised strength training sessions attended overall. We also stratified by demographic and medical characteristics, dividing continuous variables into dichotomous categories split on median values. In univariate analyses we used t-tests for dichotomous categories and an F-test for multi-level categories to generate p-values for mean differences in the outcome of interest. We then used generalized linear models, adjusting means for covariates previously significant at a p<0.1 level, to generate adjusted means for univariate modifiers of adherence. We used SAS 9.3 (Cary, NC) in all analyses.
Results
Participant Characteristics
The average age of study participants was 62 years (Table 1). The majority of participants were white (87%) and diagnosed with Stage I breast cancer (59%). Average time between diagnosis and enrollment was 2.5 years, and average time on AI therapy was 1.9 (SD 1.9) years. Women had an average BMI of 30.0 kg/m2, and a worst pain severity score of 5.7 (SD 1.9) as reported on the BPI. Study follow-up was high, with 58 of 61 exercisers (95%) completing 6-month follow-up visits, and 45 of 48 exercisers (94%) completing 12-month follow-up visits.
Table 1. Baseline characteristics of women randomized to the exercise intervention in the HOPE Study (n=61).
| Characteristic | Mean (SD) or N (%) |
|---|---|
| Age (years) | 62.0 (7.0) |
| Time since diagnosis (years) | 2.7 (3.1) |
| Time on AIs (years) | 1.9 (1.9) |
| Ethnicity | |
| Non-Hispanic White | 53 (86.9%) |
| African American | 6 (9.8%) |
| Asian/Pacific Islander | 1 (1.6%) |
| Other | 1 (1.6%) |
| Cancer Stage | |
| Stage 0 | 1 (1.6%) |
| Stage I | 36 (59.0%) |
| Stage II | 18 (29.5%) |
| Stage III | 6 (9.8%) |
| Type of surgery | |
| Lumpectomy or partial mastectomy | 43 (70.5%) |
| Radical Mastectomy | 1 (1.6%) |
| Simple Mastectomy | 9 (14.8%) |
| Double radical mastectomy | 1 (1.6%) |
| Simple radical mastectomy | 7 (11.5%) |
| Treatment | |
| None | 7 (11.5%) |
| Radiation only | 22 (36.1%) |
| Chemotherapy only | 4 (6.6%) |
| Radiation and chemotherapy | 28 (45.9%) |
| Weight (kg) | 78.4 (18.0) |
| BMI (kg/m2) | 30.0 (6.8) |
| Mean min/week of exercise | 55.1 (92.9) |
| Worst pain severity reported on BPI | 5.7 (1.9) |
Exercise Adherence
Women randomized to exercise reported an average 115 min/wk and 119 min/wk aerobic exercise at 6- and 12-months, respectively (Table 2). Approximately 50% of women reported ≥120 min/wk (80% of aerobic exercise goal) at both 6 and 12 months post-randomization and over 30% of women reached the goal of 150 min/wk. Participants attended an average of 72% and 70% of strength training sessions at 6- and 12-months, respectively.
Table 2. Adherence to aerobic exercise, attendance to supervised strength and aerobic training sessions and VO2max at 6 and 12 months post-randomization in the HOPE Study.
| 6 months (n=61) | 12 months (n=48) | |
|---|---|---|
| Aerobic exercise | ||
| Min/wk of aerobic exercise, mean (sd) | 115 (73) | 119 (78) |
| Percent of women meeting specified cutoffs of aerobic exercise | ||
| ≥ 150 min/wk (100% of goal) | 33% | 33% |
| ≥ 120 min/wk (80% of goal) | 48% | 48% |
| ≥ 90 min/wk (60% of goal) | 61% | 71% |
| ≥ 60 min/wk (40% of goal) | 79% | 79% |
| ≥ 30 min/wk (20% of goal) | 84% | 81% |
| Strength training | ||
| % sessions attended | 72% | 70% |
| Cardiorespiratory fitness | ||
| VO2max, ml/kg/min, mean (SD)b | n/a | 24.6 (5.9) |
| Change in VO2max from baseline, ml/kg/min | 1.2 | |
| Change in VO2max from baseline, % | 5.1% |
Aerobic exercise was calculated as min/wk from 7-day daily activity logs averaged over the trial duration.
56 women had VO2 maximum measured at 6 months; 42 women had VO2 maximum measured at 12 months.
There were five women in the exercise intervention who reported 0 min/wk of aerobic exercise and attended 0 strength training sessions; yet their results of 0 min/wk of aerobic exercise and 0% participation in strength training were included in the exercise adherence results described above. These non-adherers were between 43-61years old, had an average BMI of 36.6 kg/m2, and had been taking AIs for an average of 1.1 years. Two of the women who did not exercise had stage I breast cancer, two had stage II breast cancer and the third had a stage III tumor.
Cardiorespiratory Fitness (VO2max)
At baseline, mean VO2 max was 23.4 ml/kg/min (SD 5.3); at follow-up, the mean VO2 max increased to 24.6 ml/kg/min (SD 5.9), for a 5.1% increase (Table 2).
Factors Associated with Exercise Adherence
In unadjusted analyses, we observed higher mean min/wk of aerobic exercise measured at 6- and 12-months among women who were more educated, and had a higher baseline VO2max and BMI <30 kg/m2. At 6-months we found higher aerobic exercise levels among women who had been taking an AI for ≥ 1.9 years (Table 3). Participation in supervised strength training sessions was higher among women who were older, more educated, had a BMI < 30 kg/m2, reported higher baseline physical activity levels, had been on an AI for ≥ 1.9 years, received radiation therapy (only significant for 6 month exercise adherence), and among women who were not married (only significant for 12 month exercise adherence). Disease stage, adjuvant treatment, and AI-associated arthralgia assessed by the BPI were not associated with exercise adherence. We also looked at change in BPI and adherence and found no significant associations for predicting aerobic or strength training at either time point.
Table 3. Unadjusted mean adherence levels for aerobic exercise and strength training at six and 12 months by demographics and tumor characteristics.
| Aerobic exercise, mean (SD) min/wka | Strength training, % attendance | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | 6 months | p-valueb | N | 12 months | p-valueb | 6 months | p-valueb | 12 months | p-valueb | |
| Age (years) | 0.152 | 0.081 | 0.066 | 0.017 | ||||||
| <62.0 | 30 | 101.7 (77.4) | 23 | 98.3 (74.4) | 64.9 | 59.4 | ||||
| ≥62.0 | 31 | 128.6 (67.0) | 25 | 137.3 (77.2) | 78.5 | 79.7 | ||||
| Race | 0.146 | 0.099 | 0.569 | 0.653 | ||||||
| White | 53 | 120.7 (73.9) | 42 | 125.6 (76.5) | 72.6 | 70.7 | ||||
| Other | 8 | 80.3 (58.7) | 6 | 69.7 (73.0) | 66.4 | 65.0 | ||||
| Education | 0.022 | 0.003 | 0.069 | 0.017 | ||||||
| High school/some college | 26 | 91.6 (77.5) | 22 | 83.7 (75.8) | 62.2 | 58.4 | ||||
| College degree | 16 | 111.2 (49.6) | 10 | 117.6 (50.5) | 77.0 | 72.3 | ||||
| Graduate degree | 19 | 151.4 (71.8) | 16 | 167.3 (70.5) | 80.6 | 84.4 | ||||
| Chemotherapy | 0.584 | 0.496 | 0.745 | 0.734 | ||||||
| No | 29 | 120.8 (68.4) | 23 | 126.7 (66.4) | 73.1 | 71.4 | ||||
| Yes | 32 | 110.4 (77.7) | 25 | 111.2 (87.3) | 70.7 | 68.6 | ||||
| Disease Stage | 0.938 | 0.736 | 0.810 | 0.503 | ||||||
| Stage 0 | 1 | 154.0 (0.0) | 1 | 162.0 (0.0) | 88.0 | 87.0 | ||||
| Stage I | 36 | 117.1 (76.7) | 27 | 127.1 (79.7) | 71.2 | 71.9 | ||||
| Stage II | 18 | 109.3 (66.1) | 16 | 107.2 (71.5) | 74.8 | 70.5 | ||||
| Stage III | 6 | 116.7 (86.4) | 4 | 96.0 (105.0) | 63.8 | 50.3 | ||||
| Radiation | 0.462 | 0.606 | 0.038 | 0.195 | ||||||
| No | 11 | 100.5 (98.2) | 8 | 130.2 (80.6) | 55.6 | 58.0 | ||||
| Yes | 50 | 118.6 (67.0) | 40 | 106.0 (73.8) | 75.4 | 72.4 | ||||
| Married | 0.211 | 0.283 | 0.135 | 0.085 | ||||||
| No | 33 | 126.2 (68.8) | 25 | 130.2 (80.6) | 77.0 | 76.9 | ||||
| Yes | 28 | 102.6 (76.9) | 23 | 106.0 (73.8) | 65.6 | 62.4 | ||||
| VO2 max (ml/kg/min) | 0.004 | 0.017 | 0.432 | 0.581 | ||||||
| <23.4 | 32 | 91.2 (58.0) | 25 | 93.4 (64.8) | 69.1 | 67.8 | ||||
| ≥23.4 | 28 | 143.8 (79.3) | 23 | 146.0 (82.3) | 75.0 | 72.3 | ||||
| BMI (kg/m2) | <0.001 | <0.001 | 0.060 | 0.005 | ||||||
| <30.0 | 33 | 144.8 (72.6) | 25 | 160.1 (68.7) | 78.5 | 81.2 | ||||
| ≥30.0 | 28 | 80.7 (57.4) | 23 | 73.5 (60.2) | 63.9 | 57.7 | ||||
| Baseline MVPA | 0.294 | 0.267 | 0.004 | 0.016 | ||||||
| min/week | ||||||||||
| <54.8 | 42 | 108.7 (72.9) | 33 | 110.2 (70.9) | 66.4 | 64.8 | ||||
| ≥54.8 | 19 | 130.1 (72.9) | 15 | 137.2 (90.3) | 83.7 | 81.3 | ||||
| Diabetes | 0.477 | 0.443 | 0.288 | 0.355 | ||||||
| No | 57 | 117.1 (71.2) | 44 | 121.2 (75.8) | 73.9 | 72.3 | ||||
| Yes | 4 | 90 (104.5) | 4 | 89.8 (103.7) | 42.3 | 44.3 | ||||
| Depression | 0.797 | 0.503 | 0.535 | 0.136 | ||||||
| No | 48 | 116.6 (68.6) | 38 | 122.5 (72.1) | 73.0 | 73.1 | ||||
| Yes | 13 | 110.7 (90.5) | 10 | 103.8 (98.8) | 67.4 | 58.0 | ||||
| Worst pain reported on BPI | 0.983 | 0.523 | 0.330 | 0.434 | ||||||
| <5.7 | 30 | 115.6 (57.4) | 25 | 111.6 (57.8) | 75.5 | 73.1 | ||||
| ≥5.7 | 31 | 115.2 (86.4) | 23 | 126.3 (96.0) | 68.3 | 66.5 | ||||
| Years on AIs | 0.079 | 0.267 | <0.001 | 0.009 | ||||||
| <1.9 | 40 | 105.4 (83.3) | 28 | 108.8 (90.7) | 66.1 | 61.9 | ||||
| ≥1.9 | 21 | 134.5 (43.4) | 20 | 132.3 (53.5) | 82.8 | 81.2 | ||||
| Years since diagnosis | 0.527 | 0.999 | 0.199 | 0.541 | ||||||
| <2.5 | 36 | 110.7 (83.5) | 24 | 118.6 (92.2) | 67.9 | 67.4 | ||||
| ≥2.5 | 25 | 122.0 (55.4) | 24 | 118.6 (61.6) | 77.5 | 72.5 | ||||
Baseline moderate to vigorous-intensity physical activity (MVPA) was measured by an interviewer-administered questionnaire assessing the duration, frequency and intensity of 20 recreational activities over the prior 6 months.
p-values for a difference in means were generated using the t-test for two categories or ANOVA for multiple categories.
We further adjusted mean exercise levels for those factors that were significant at a p<0.1 level in unadjusted analyses (Table 4). In aerobic exercise models we thus included age, race, education, VO2max, BMI and years on AIs. Fully adjusted models showed that participants with a higher baseline VO2max performed more aerobic exercise at 6 months, and more educated and women with a BMI < 30 kg/m2 performed more aerobic exercise at 12 months. Only VO2max was statistically significant at a more stringent p<0.05 level in the fully adjusted models. For supervised strength training attendance we included age, education, BMI, years on AIs, baseline physical activity and marital status in models. At 12 months, age ≥ 62 years was associated with better attendance at supervised exercise training sessions; higher educational level also suggested better attendance, although was not statistically significant at a p<0.05 threshold.
Table 4. Adjusted predictors of aerobic exercise and strength training attendance levels at 6 and 12 months post-randomizationa.
| 6 months | p-valueb | 12 months | p-valueb | |
|---|---|---|---|---|
| Aerobic exercise (mean min/wk) | ||||
| Age (years) | 0.189 | 0.115 | ||
| <62.0 | 103.4 | 98.6 | ||
| ≥62.0 | 128.9 | 133.4 | ||
| Race | 0.844 | 0.430 | ||
| White | 116.0 | 119.4 | ||
| Other | 110.1 | 93.2 | ||
| Education | 0.262 | 0.094 | ||
| High school/some college | 98.0 | 89.2 | ||
| College degree | 116.1 | 122.7 | ||
| Graduate degree | 136.7 | 145.3 | ||
| VO2 max (ml/kg/min) | 0.033 | 0.331 | ||
| <23.4 | 91.6 | 105.5 | ||
| ≥23.4 | 143.4 | 132.9 | ||
| BMI (kg/m2) | 0.255 | 0.087 | ||
| <30.0 | 128.6 | 140.5 | ||
| ≥30.0 | 100.9 | 91.4 | ||
| Years on AIs | 0.402 | 0.974 | ||
| <1.9 | 109.1 | 115.7 | ||
| ≥1.9 | 126.3 | 116.4 | ||
|
| ||||
| Strength Training (% attendance) | ||||
| Age (years) | 0.137 | 0.025 | ||
| <62.0 | 66.3 | 61.1 | ||
| ≥62.0 | 77.1 | 78.1 | ||
| Education | 0.213 | 0.089 | ||
| High school/some college | 64.5 | 61.1 | ||
| College degree | 78.1 | 74.6 | ||
| Graduate degree | 76.4 | 79.3 | ||
| BMI (kg/m2) | 0.240 | 0.134 | ||
| <30.0 | 75.7 | 75.6 | ||
| ≥30.0 | 67.2 | 63.8 | ||
| Years on AIs | 0.191 | 0.168 | ||
| <1.9 | 68.4 | 65.5 | ||
| ≥1.9 | 78.3 | 76.2 | ||
| Baseline MVPA min/week | 0.369 | 0.632 | ||
| <54.8 | 69.4 | 68.7 | ||
| ≥54.8 | 77.0 | 72.8 | ||
| Married | 0.261 | 0.138 | ||
| No | 75.5 | 75.2 | ||
| Yes | 67.5 | 64.3 | ||
Means were adjusted all variables that were significantly different at a p<0.1 level in unadjusted analyses.
Discussion
Among physically inactive breast cancer survivors with aromatase inhibitor-associated arthralgias, women randomized to exercise performed 119 (SD 78) min/wk of aerobic exercise and attended 70% of strength training sessions. Although all women had AI-associated arthralgia at baseline, this arthralgia, as measured by worst pain score on the BPI, did not influence exercise adherence. From baseline to end of study, VO2max increased by an average 5.1%. These findings demonstrate that approximately 120 min/wk of aerobic exercise and twice-weekly strength training is feasible among this population of sedentary breast cancer survivors with AI-induced arthralgia.
Although only 33% of women met the U.S. Physical Activity Guidelines of 150 minutes of moderate-intensity aerobic activity weekly, our observed levels of weekly aerobic exercise and strength training attendance were similar to previously observed exercise trials in breast cancer survivors [9, 17, 20]. The increase in activity levels that we observed have still been associated with significant health benefits [23]. Previous studies have also reported BMI [17], baseline physical activity [17], and aerobic fitness [9] as predictors of exercise adherence, similar to our univariate, or unadjusted findings. However, our finding that at 12 months post-randomization only older age predicted strength training adherence in multivariate adjusted analyses suggests that physical activity interventions may need to be adapted to meet the needs of younger breast cancer survivors.
Previous studies have shown a worse CVD risk profile among breast cancer patients compared to healthy controls [15] and CVD is a leading cause of death among breast cancer survivors. At baseline in HOPE, the average VO2max among participants was 23.4 ml/kg/min, indicating relatively low-normal fitness levels. Our observed average 1.2 ml/kg/min improvement in VO2max is encouraging in regard to improving survival in this group of inactive breast cancer survivors, as it has been reported that a 1 ml/kg/min improvement in VO2max is associated with a 12% lower mortality risk [19].
Strengths of our study include the population-based recruitment strategy, detailed measurement of physical activity and adherence, gold standard assessment of VO2max, the yearlong study duration, and a focus on women experiencing arthralgias due to AI use. Study limitations include that our participants were predominantly non-Hispanic white and highly educated, limiting generalizability of study results to other populations.
Our study builds on previous literature supporting exercise as feasible in breast cancer survivors, and uniquely supports the feasibility of an exercise program in breast cancer survivors with aromatase inhibitor-induced arthralgia, a population who might suffer additional barriers to exercise and additional cardiovascular disease risk. Furthermore, our findings help identify predictors of exercise adherence, which could inform future exercise trials using personalized intervention strategies. Our findings thus should encourage referral of breast cancer survivors to community-based exercise programs. Community-based exercise programs are becoming increasingly available, such as the LIVESTRONG® at the YMCA program, which offers free exercise programs to cancer survivors at various YMCAs across the United States.
In summary, as new breast cancer therapies are developed and breast cancer survivorship rates improve, physical activity behaviors and cardiovascular health will be increasingly important to maintain health among this population. As we transition into the era of “personalized medicine” in oncology, it will be critical to identify exercise prescriptions that are suitable for the clinical and treatment characteristics of patient subgroups. Our findings of good exercise adherence among women with AI-induced arthralgia will inform and encourage future effective, targeted exercise plans among breast cancer survivors.
Acknowledgments
We thank Marianna Rothbard, Dan Root, Willie Moore, Liz Fraser, Michelle Baglia, Adrienne Viola, Yanchang Zhang, Bridget Winterhalter, Norbert Hootsmans, Celeste Wong, and Meghan Hughes for their assistance. We thank Rajni Mehta and the Rapid Case Ascertainment of Yale Cancer Center, as well as Smilow Cancer Hospital at Yale-New Haven, St. Raphael's Hospital, St. Vincent's Medical Center, Bridgeport Hospital and Greenwich Hospital and all the clinicians who consented or referred their patients to our study. Most importantly, we are indebted to the participants for their dedication and time to the HOPE study.
Funding: HA is supported by the intramural research program at the National Cancer Institute. HOPE was supported by NCI R01 CA132931 and in part by a grant from the Breast Cancer Research Foundation. Also supported in part by the Yale Cancer Center Support Grant (CCSG) P30 CA016359 and the CTSA Grant Number UL1 TR000142 from the National Center for Advancing Translational Science (NCATS), a component of the National Institutes of Health (NIH). Certain data used in this study were obtained from the Connecticut Tumor Registry located in the Connecticut Department of Public Health. The authors assume full responsibility for analyses and interpretation of these data.
Cary P. Gross: Research funding from 21st Century Oncology, Medtronic, Johnson & Johnson Tara Sanft: Consulting or Advisory Role at bioTheranostics; Research funding from bioTheranostics.
Footnotes
Compliance with Ethical Standards: Conflicts of interest: All other authors report no conflicts of interest.
Informed consent: Informed consent was obtained from all individual participants included in the study.
References
- 1.Continuous Update Project Report. World Cancer Research Fund International/American Institute for Cancer Research; 2014. Diet, Nutrition, Physical Activity, and Breast Cancer Survivors. [Google Scholar]
- 2.Society AC, editor. What are the key statistics about breast cancer? American Cancer Society; Atlanta, GA: 2013. [Google Scholar]
- 3.Ballard-Barbash R, Friedenreich CM, Courneya KS, Siddiqi SM, McTiernan A, Alfano CM. Physical Activity, Biomarkers, and Disease Outcomes in Cancer Survivors: A Systematic Review. Journal of the National Cancer Institute. 2012;104:815–840. doi: 10.1093/jnci/djs207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Blair SN, Haskell WL, Ho P, Paffenbarger RS, Vranizan KM, Farquhar JW, Wood PD. Assessment of habitual physical activity by a sevenday recall in a community survey and controlled experiments. American Journal of Epidemiology. 1985;122:794–804. doi: 10.1093/oxfordjournals.aje.a114163. [DOI] [PubMed] [Google Scholar]
- 5.Blanchard CM, Courneya KS, Stein K. Cancer Survivors' Adherence to Lifestyle Behavior Recommendations and Associations With Health-Related Quality of Life: Results From the American Cancer Society's SCS-II. Journal of Clinical Oncology. 2008;26:2198–2204. doi: 10.1200/jco.2007.14.6217. [DOI] [PubMed] [Google Scholar]
- 6.Cleeland C, Ryan K. Pain assessment: global use of the Brief Pain Inventory. Annals of the Academy of Medicine, Singapore. 1994;23:129–138. [PubMed] [Google Scholar]
- 7.Clemons M, Coleman RE, Verma S. Aromatase inhibitors in the adjuvant setting: bringing the gold to a standard? Cancer Treatment Reviews. 2004;30:325–332. doi: 10.1016/j.ctrv.2004.03.004. doi: http://dx.doi.org/10.1016/j.ctrv.2004.03.004. [DOI] [PubMed] [Google Scholar]
- 8.Coleman RE, Bolten WW, Lansdown M, Dale S, Jackisch C, Merkel D, Maass N, Hadji P. Aromatase inhibitor-induced arthralgia: Clinical experience and treatment recommendations. Cancer Treatment Reviews. 2008;34:275–282. doi: 10.1016/j.ctrv.2007.10.004. doi: http://dx.doi.org/10.1016/j.ctrv.2007.10.004. [DOI] [PubMed] [Google Scholar]
- 9.Courneya KS, Segal RJ, Gelmon K, Reid RD, Mackey JR, Friedenreich CM, Proulx C, Lane K, Ladha AB, Vallance JK, McKenzie DC. Predictors of supervised exercise adherence during breast cancer chemotherapy. Medicine and science in sports and exercise. 2008;40:1180–1187. doi: 10.1249/MSS.0b013e318168da45. [DOI] [PubMed] [Google Scholar]
- 10.Crew KD, Greenlee H, Capodice J, Raptis G, Brafman L, Fuentes D, Sierra A, Hershman DL. Prevalence of Joint Symptoms in Postmenopausal Women Taking Aromatase Inhibitors for Early-Stage Breast Cancer. Journal of Clinical Oncology. 2007;25:3877–3883. doi: 10.1200/jco.2007.10.7573. [DOI] [PubMed] [Google Scholar]
- 11.Haskell WL, Lee IM, Pate RR, Powell KE, Blair SN, Franklin BA, Macera CA, Heath GW, Thompson PD, Bauman A. Physical activity and public health: updated recommendation for adults from the American College of Sports Medicine and the American Heart Association. Circulation. 2007;116:1081. doi: 10.1161/CIRCULATIONAHA.107.185649. [DOI] [PubMed] [Google Scholar]
- 12.Hershman DL, Shao T, Kushi LH, Buono D, Tsai WY, Fehrenbacher L, Kwan M, Gomez SL, Neugut AI. Early discontinuation and non-adherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer. Breast cancer research and treatment. 2011;126:529–537. doi: 10.1007/s10549-010-1132-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Irwin ML, Cartmel B, Gross CP, Ercolano E, Li F, Yao X, Fiellin M, Capozza S, Rothbard M, Zhou Y. Randomized Exercise Trial of Aromatase Inhibitor–Induced Arthralgia in Breast Cancer Survivors. Journal of Clinical Oncology:JCO. 2014;2057:1547. doi: 10.1200/JCO.2014.57.1547. 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Irwin ML, McTiernan A, Bernstein L, Gilliland FD, Baumgartner R, Baumgartner K, Ballard-Barbash R. Physical activity levels among breast cancer survivors. Medicine and science in sports and exercise. 2004;36:1484. [PMC free article] [PubMed] [Google Scholar]
- 15.Jones LW, Haykowsky M, Pituskin EN, Jendzjowsky NG, Tomczak CR, Haennel RG, Mackey JR. Cardiovascular reserve and risk profile of postmenopausal women after chemoendocrine therapy for hormone receptor–positive operable breast cancer. The Oncologist. 2007;12:1156–1164. doi: 10.1634/theoncologist.12-10-1156. [DOI] [PubMed] [Google Scholar]
- 16.Jones LW, Liang Y, Pituskin EN, Battaglini CL, Scott JM, Hornsby WE, Haykowsky M. Effect of exercise training on peak oxygen consumption in patients with cancer: a meta-analysis. The oncologist. 2011;16:112–120. doi: 10.1634/theoncologist.2010-0197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Latka RN, Alvarez-Reeves M, Cadmus L, Irwin ML. Adherence to a randomized controlled trial of aerobic exercise in breast cancer survivors: the Yale exercise and survivorship study. J Cancer Surviv. 2009;3:148–157. doi: 10.1007/s11764-009-0088-z. [DOI] [PubMed] [Google Scholar]
- 18.Murphy CC, Bartholomew LK, Carpentier MY, Bluethmann SM, Vernon SW. Adherence to adjuvant hormonal therapy among breast cancer survivors in clinical practice: a systematic review. Breast cancer research and treatment. 2012;134:459–478. doi: 10.1007/s10549-012-2114-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Myers J, Prakash M, Froelicher V, Do D, Partington S, Atwood JE. Exercise capacity and mortality among men referred for exercise testing. New England Journal of Medicine. 2002;346:793–801. doi: 10.1056/NEJMoa011858. [DOI] [PubMed] [Google Scholar]
- 20.Pinto BM, Rabin C, Dunsiger S. Home-based exercise among cancer survivors: adherence and its predictors. Psycho-Oncology. 2009;18:369–376. doi: 10.1002/pon.1465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rock CL, Doyle C, Demark-Wahnefried W, Meyerhardt J, Courneya KS, Schwartz AL, Bandera EV, Hamilton KK, Grant B, McCullough M. Nutrition and physical activity guidelines for cancer survivors. CA: a cancer journal for clinicians. 2012;62:242–274. doi: 10.3322/caac.21142. [DOI] [PubMed] [Google Scholar]
- 22.Schmid D, Leitzmann M. Cardiorespiratory fitness as predictor of cancer mortality: a systematic review and meta-analysis. Annals of Oncology. 2014 doi: 10.1093/annonc/mdu250. mdu250. [DOI] [PubMed] [Google Scholar]
- 23.Schmitz KH, Courneya KS, Matthews C, Demark-Wahnefried W, Galvao DA, Pinto BM, Irwin ML, Wolin KY, Segal RJ, Lucia A, Schneider CM, von Gruenigen VE, Schwartz AL. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Medicine and science in sports and exercise. 2010;42:1409–1426. doi: 10.1249/MSS.0b013e3181e0c112. [DOI] [PubMed] [Google Scholar]