Abstract
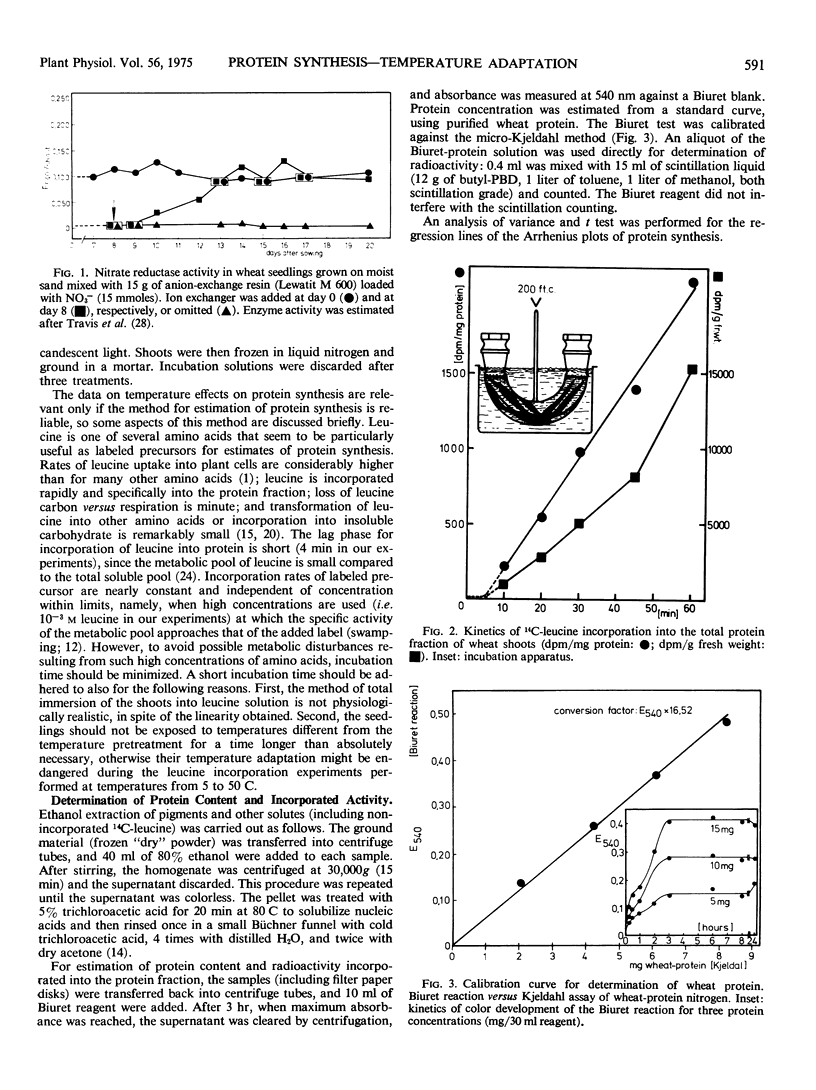
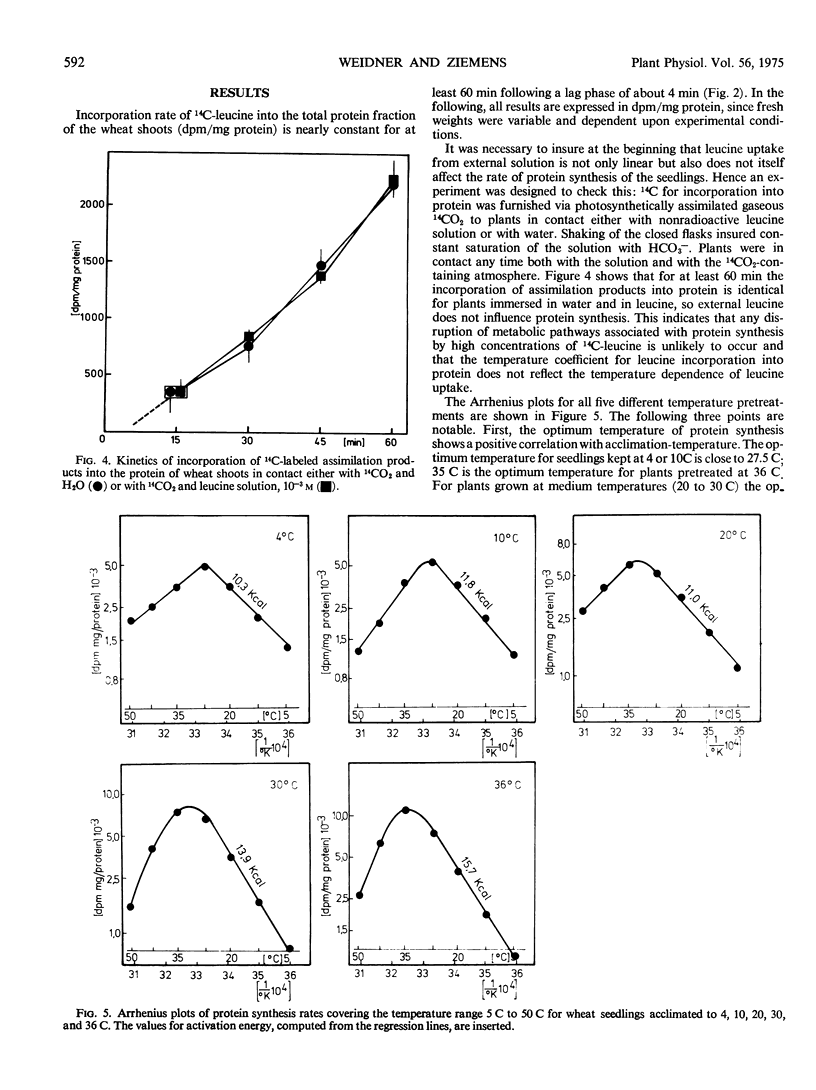
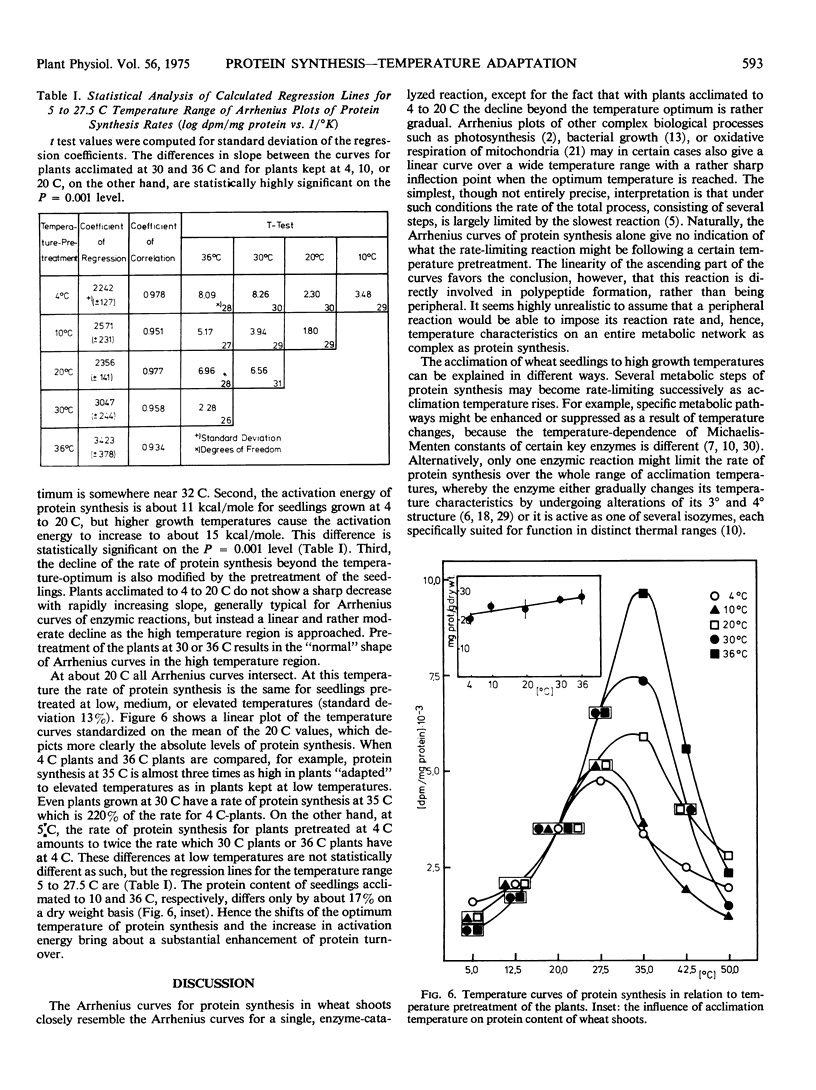
The optimum temperature of protein synthesis in wheat seedlings (Triticum aestivum L.), measured as 14C-leucine incorporation, depends on the growing temperature. Plants grown at reduced temperature (4 C) reach their optimum at 27.5 C, whereas plants kept at 36 C have the highest rate of protein synthesis at 35 C. The transition is gradual. The activation energy of protein synthesis for seedlings grown at medium or reduced temperature is lower (about 11 kcal/mole), than for plants grown at higher temperatures (15 keal/mole). The decline of the rate of protein synthesis beyond the temperature optimum is also affected by the growth temperature; only plants kept at 30 or 36 C show a sharp decrease with increasing slope; plants kept at 4, 10, and 20 C exhibit a linear and comparatively moderate decline.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BIRT L. M., HIRD F. J. Kinetic aspects of the uptake of amino acids by carrot tissue. Biochem J. 1958 Oct;70(2):286–292. doi: 10.1042/bj0700286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GIBSON K. D. True and apparent activation energies of enzymic reactions. Biochim Biophys Acta. 1953 Feb;10(2):221–229. doi: 10.1016/0006-3002(53)90246-7. [DOI] [PubMed] [Google Scholar]
- INGRAHAM J. L. Growth of psychrophilic bacteria. J Bacteriol. 1958 Jul;76(1):75–80. doi: 10.1128/jb.76.1.75-80.1958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KAUZMANN W. Some factors in the interpretation of protein denaturation. Adv Protein Chem. 1959;14:1–63. doi: 10.1016/s0065-3233(08)60608-7. [DOI] [PubMed] [Google Scholar]
- Larson L. A., Beevers H. Amino Acid Metabolism in Young Pea Seedlings. Plant Physiol. 1965 May;40(3):424–432. doi: 10.1104/pp.40.3.424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyons J. M., Raison J. K. Oxidative activity of mitochondria isolated from plant tissues sensitive and resistant to chilling injury. Plant Physiol. 1970 Apr;45(4):386–389. doi: 10.1104/pp.45.4.386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNaughton S. J. Thermal Inactivation Properties of Enzymes from Typha latifolia L. Ecotypes. Plant Physiol. 1966 Dec;41(10):1736–1738. doi: 10.1104/pp.41.10.1736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oaks A. The soluble leucine pool in maize root tips. Plant Physiol. 1965 Jan;40(1):142–149. doi: 10.1104/pp.40.1.142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pace B., Campbell L. L. Correlation of maximal growth temperature and ribosome heat stability. Proc Natl Acad Sci U S A. 1967 Apr;57(4):1110–1116. doi: 10.1073/pnas.57.4.1110. [DOI] [PMC free article] [PubMed] [Google Scholar]


