Abstract
Background
Endophthalmitis refers to severe infection within the eye that involves the aqueous humor or vitreous humor, or both, and threatens vision. Most cases of endophthalmitis are exogenous (i.e. due to inoculation of organisms from an outside source), and most exogenous endophthalmitis is acute and occurs after an intraocular procedure. The mainstay of treatment is emergent administration of broad‐spectrum intravitreous antibiotics. Due to their anti‐inflammatory effects, steroids in conjunction with antibiotics have been proposed to be beneficial in endophthalmitis management.
Objectives
To assess the effects of antibiotics combined with steroids versus antibiotics alone for the treatment of acute endophthalmitis following intraocular surgery or intravitreous injection.
Search methods
We searched the Cochrane Central Register of Controlled Trials (CENTRAL) (which contains the Cochrane Eyes and Vision Trials Register) (2016, Issue 11), MEDLINE Ovid (1946 to 8 December 2016), Embase Ovid (1980 to 8 December 2016), LILACS (Latin American and Caribbean Health Sciences Literature Database) (1982 to 8 December 2016), the ISRCTN registry (www.isrctn.com/editAdvancedSearch); searched 8 December 2016, ClinicalTrials.gov (www.clinicaltrials.gov); searched 8 December 2016, and the WHO International Clinical Trials Registry Platform (ICTRP) (www.who.int/ictrp/search/en); searched 8 December 2016. We did not use any date or language restrictions in the electronic searches for trials.
Selection criteria
We included randomized controlled trials comparing the effectiveness of adjunctive steroids with antibiotics alone in the management of acute, clinically diagnosed endophthalmitis following intraocular surgery or intravitreous injection. We excluded trials with participants with endogenous endophthalmitis unless outcomes were reported by source of infection. We imposed no restrictions on the method or order of administration, dose, frequency, or duration of antibiotics and steroids.
Data collection and analysis
Two review authors independently screened the search results, assessed risk of bias, and extracted data using methods expected by Cochrane. We contacted study authors to try to obtain missing information or information to clarify risk of bias. We conducted a meta‐analysis for any outcomes that were reported by at least two studies. Outcomes reported from single studies were summarized in the text. We assessed the certainty of the evidence using GRADE.
Main results
We included three trials with a total of 95 randomized participants in this review and identified one ongoing trial. The studies were conducted in South Africa, India, and the Netherlands. Out of the 92 analyzed participants, 91 participants were diagnosed with endophthalmitis following cataract surgery. In the remaining participant, endophthalmitis was attributable to penetrating keratoplasty. All studies used intravitreous dexamethasone for adjunctive steroid therapy and a combination of two intravitreous antibiotics that provided gram‐positive and gram‐negative coverage for the antibiotic therapy. We judged one trial to be at overall low risk of bias and two studies to be at overall unclear risk of bias due to lack of reporting of study methods. None of the three trials had been registered in a clinical trial register.
While none of the included studies reported the primary outcome of complete resolution of endophthalmitis as defined in our protocol, one study reported combined anatomical and functional success (i.e. proportion of participants with intraocular pressure of at least 5 mmHg and visual acuity of at least 6/120). Very low‐certainty evidence suggested no difference in combined success when comparing adjunctive steroid antibiotics alone (risk ratio (RR) 1.08, 95% confidence interval (CI) 0.80 to 1.45; 32 participants). Low‐certainty evidence from two studies showed that a higher proportion of participants who received adjunctive dexamethasone had a good visual outcome (Snellen visual acuity 6/6 to 6/18) at three months compared with those in the antibiotics‐alone group (RR 1.95, 95% CI 1.05 to 3.60; 60 participants). Similarly, low‐certainty evidence from one study suggested that more participants in the dexamethasone group had a good visual outcome at 12 months compared to those who did not receive dexamethasone (RR 2.00, 95% CI 0.98 to 4.08; 28 participants). Investigators of one study reported improvement in visual acuity, but we could not estimate the effect of adjunctive steroid therapy because the study investigators did not provide standard deviations or standard errors. Two studies reported adverse events (retinal detachment, hypotony, proliferative vitreoretinopathy, and seclusion of pupil). The total numbers of adverse events were 8 out of 30 (26.7%) for those who received dexamethasone versus 6 out of 30 (20.0%) for those who did not. We could only perform a pooled analysis for the occurrence of retinal detachment; any difference between the two treatment groups was uncertain (RR 1.57, 95% CI 0.50 to 4.90; 60 participants) (very low‐certainty evidence). No study reported intraocular pressure or cost outcomes.
Authors' conclusions
Current evidence on the effectiveness of adjunctive steroid therapy versus antibiotics alone in the management of acute endophthalmitis after intraocular surgery is inadequate. We found no studies that had enrolled cases of acute endophthalmitis following intravitreous injection. A combined analysis of two studies suggests adjunctive steroids may provide a higher probability of having a good visual outcome at three months than not using adjunctive steroids. However, considering that most of the confidence intervals crossed the null and that this review was limited in scope and applicability to clinical practice, it is not possible to conclude whether the use adjunctive steroids is effective at this time. Any future trials should examine whether adjunctive steroids may be useful in certain clinical settings such as type of causative organism or etiology. These studies should include outcomes that take patient's symptoms and clinical examination into account, report outcomes in a uniform and consistent manner, and follow up at short‐ and long‐term intervals.
Plain language summary
Steroids plus antibiotics versus antibiotics alone for treatment of acute endophthalmitis after eye surgery or injections into the eye
What is the aim of this review? The aim of this Cochrane review was to find out whether using steroids in addition to antibiotics works better than using antibiotics alone for acute endophthalmitis (infection inside the eyeball that can cause vision loss) after eye surgery or injections into the eye. Cochrane researchers looked for all relevant studies to answer this question and found three studies.
Key messages It is uncertain whether using steroids in addition to antibiotics is helpful or harmful compared with using antibiotics alone to treat acute endophthalmitis after eye surgery or injections into the eye.
What was studied in the review? Although endophthalmitis is rare, it is important for people undergoing surgery or injections to the eye to be aware of the risk and for their doctors to know how best to treat it because it can result in vision loss. It is most commonly caused by entry of bacteria into the eye during, or a few days after, surgery or injection. As soon as endophthalmitis is suspected, a sample of the fluid inside the eye is usually obtained (and the fluid drained in severe cases) and antibiotics that cover most types of bacteria are injected into the eye. Although the use of antibiotics is widely accepted, the use of adjunctive steroids to treat endophthalmitis is debatable. Steroids may help to decrease the inflammation inside the eye in people with endophthalmitis. In this review, Cochrane researchers studied whether giving steroids in addition to antibiotics has any effect on patient outcome.
What are the main results of the review? Cochrane researchers found three relevant studies from South Africa, India, and the Netherlands. Almost all the people that took part in these studies had endophthalmitis after cataract surgery. All three studies compared injecting dexamethasone (a steroid) plus two antibiotics into the eye versus injecting only the antibiotics into the eye. Low‐certainty evidence showed that more participants in the group receiving dexamethasone had a good visual outcome three and 12 months after treatment than in the group that did not receive dexamethasone. However, the effects of using steroids on resolution of endophthalmitis and harms were uncertain. Due to the uncertainty of evidence for most outcomes, it is not clear whether doctors should use steroids with antibiotics to treat endophthalmitis after a procedure in the eye.
How up‐to‐date is this review? Cochrane researchers searched for studies that had been published up to 8 December 2016.
Summary of findings
Summary of findings for the main comparison. Combined steroid and antibiotic therapy compared with antibiotics alone for acute endophthalmitis after intraocular procedure.
| Combined steroid and antibiotic therapy compared with antibiotics alone for acute endophthalmitis after intraocular procedure | ||||||
|
Population: eyes with acute endophthalmitis following an intraocular procedure (e.g. surgery, intravitreous injection) Settings: ophthalmology clinic or hospital Intervention: steroids plus antibiotics Comparison: antibiotics alone | ||||||
| Outcomes* | Illustrative comparative risks** (95% CI) | Relative effect (95% CI) | No of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Antibiotics alone | Combined steroid and antibiotic therapy | |||||
|
Complete resolution of endophthalmitis at 3 months' follow‐up |
813 per 1000 | 878 per 1000 (650 to 1000) | RR 1.08 (0.80 to 1.45) | 32 (1 study) | ⊕⊝⊝⊝ very low1,2,3 | Assessed as combined anatomical and functional success, where anatomical success was defined as IOP of at least 5 mmHg, and functional success was defined as visual acuity of at least 6/120. |
|
Visual acuity 6/6 to 6/18 at 3 months' follow‐up |
300 per 1000 | 585 per 1000 (315 to 1000) | RR 1.95 (1.05 to 3.60) | 60 (2 studies) | ⊕⊕⊝⊝ low1,2 | At 12 months' follow‐up, the RR was 2.00 (95% CI 0.98 to 4.08; n = 28; 1 study; low‐certainty evidence).1,2 |
|
Improvement in visual acuity at 3 months' follow‐up |
See comment | Investigators of 1 study reported improvement in visual acuity, but the effect of adjunctive steroid therapy could not be estimated because the study investigators did not provide standard deviations or standard errors. | ||||
|
Intraocular pressure at 3 months' follow‐up |
Not reported | |||||
|
Adverse events at 3 months' follow‐up |
133 per 1000 | 209 per 1000 (67 to 653) | RR 1.57 (0.50 to 4.90) | 60 (2 studies) | ⊕⊝⊝⊝ very low1,4 | Data are for retinal detachment. Other reported adverse events included hypotony (2/29 participants), proliferative vitreoretinopathy (1/29 participants), and seclusion of pupil (1/29 participants). |
|
Costs associated with the interventions at 3 months' follow‐up |
Not reported | |||||
| *The primary follow‐up time point for the review was one month; however, none of the included trials reported outcomes at this follow‐up time. Data are presented for the follow‐up time for which data were available. **The basis for the assumed risk was the mean control group risk across studies. The corresponding risk (and its 95% CI) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; IOP: intraocular pressure; RR: risk ratio | ||||||
| GRADE Working Group grades of evidence High‐certainty: Further research is very unlikely to change our confidence in the estimate of effect. Moderate‐certainty: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low‐certainty: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low‐certainty: We are very uncertain about the estimate. | ||||||
1Downgraded (‐1) for unclear risk of bias in the trials. 2Downgraded (‐1) for imprecision (wide confidence interval). 3Downgraded (‐1) for indirectness of the outcome definition. 4Downgraded (‐2) for imprecision (very wide confidence interval).
Background
Description of the condition
Endophthalmitis is a rare but potentially visually devastating condition that is defined as infection in the intraocular cavities (i.e. aqueous or vitreous humor, or both) (Durand 2013). A variety of pathogens, including bacteria, viruses, fungi, or parasites, can cause the infection; the culprit pathogens differ somewhat by the mechanism of infection (Keynan 2012). Most cases of endophthalmitis are exogenous; possible routes of infection include surgery, intravitreous injection, trauma, corneal infection, and glaucoma‐filtering bleb infection (Sadaka 2012). Postoperative endophthalmitis is the most common type of exogenous endophthalmitis (Keynan 2012). While endophthalmitis is a possible complication of any intraocular surgery, 90% of postoperative endophthalmitis is due to cataract surgery, as it is the most frequent intraocular surgery (Lemley 2007). Endogenous endophthalmitis, which makes up 2% to 15% of endophthalmitis cases, occurs when organisms spread hematogenously from a distant infection site to the eye (Keynan 2012). The most common primary sites of infection are the liver, lung, and endocardium (Jackson 2014).
Epidemiology
The reported incidence of postoperative endophthalmitis varies widely, but available data indicate that the rate of endophthalmitis has been very low in recent years. Analyses of Medicare claims data in the USA showed that while the annual rate of post‐cataract surgery endophthalmitis increased from 1.79 (95% confidence interval (CI) 1.46 to 2.18) to 2.62 (95% CI 2.22 to 3.07) cases per thousand surgeries between 1994 and 2000 (West 2005), it decreased from 1.32 (95% CI 1.27 to 1.38) to 1.11 (95% CI 1.06 to 1.16) per thousand surgeries between 2003 and 2004 (Keay 2012), and was stable between 2006 and 2011 (Du 2014). More recently, the Intelligent Research in Sight Registry found that the rate of endophthalmitis after cataract surgery was 0.08% (0.8 per 1000 surgeries) for 2013 and 2014 combined (Coleman 2015). Also, there were no cases of endophthalmitis following 21,501 office‐based cataract surgeries performed at Kaiser Permanente Colorado from 2011 to 2014 (Ianchulev 2016). Reported incidences for other types of exogenous endophthalmitis are between 0.006% and 0.16% for intravitreous injection, between 0.3% and 0.7% for trabeculectomy, 2.0% for glaucoma drainage surgery, and between 0.9% and 17% for traumatic injury to the globe (Sadaka 2012; Stein 2008). Risk factors for exogenous endophthalmitis include host factors and factors associated with the procedure. Some preoperative risk factors are male gender, older age, black and Native American race, immunocompromised states (e.g. diabetes, chronic steroid use), and recent history of periocular infections (e.g. blepharitis, conjunctivitis) (Hatch 2009; Keay 2012; Keynan 2012). The main intraoperative risk factor is increased intraocular exposure to the patient’s adnexal and ocular surface flora, which increases with surgical complexity and complications such as posterior capsule rupture and vitreous loss (Hatch 2009; Mamalis 2002). Therefore, possible reasons for the decline in the early 2000s include improved surgical techniques, sterility, and prophylactic antibiotics (Barry 2013). A multicenter study across nine European countries showed that the use of antibiotic prophylaxis with intracameral cefuroxime at the end of cataract surgery decreased the rate of endophthalmitis from 0.35% to 0.05% (ESCRS Study 2007). Higher rates of endophthalmitis are also associated with surgeries performed by surgeons with lower annual volume and fewer years of experience (Keay 2012).
Clinical presentation and diagnosis
Postoperative endophthalmitis usually presents within one to two weeks after surgery (acute type), but can also present a few weeks or months after surgery (chronic type) (Keynan 2012). On average, endophthalmitis presents five days after intravitreous injection (Lyall 2012; Simunovic 2012). While endogenous endophthalmitis occasionally affects both eyes, exogenous endophthalmitis affects only the eye that was exposed to the insult (Jackson 2003; Keynan 2012). The most common symptoms, regardless of the mechanism, are eye pain, red eye, and decreased vision. People with exogenous endophthalmitis usually feel well otherwise and are afebrile (Durand 2013). On physical examination, endophthalmitis presents with eyelid and conjunctival swelling, injected conjunctiva, corneal edema, and poor view of the fundus due to inflammation in the aqueous or vitreous humor, or both (Durand 2013; Keynan 2012). Hypopyon, the accumulation of white blood cells in the anterior chamber, is found in over 80% of cases (Lalwani 2008). The main concern following endophthalmitis is persistent vision loss despite treatment. Visual outcome is highly dependent on the causative organism, with any streptococci causing the worst outcomes, while coagulase‐negative staphylococci cause milder cases (Durand 2013). Overall, approximately half of eyes affected by postcataract endophthalmitis do not regain a visual acuity of 20/40 or better, and 10% will lose useful vision (5/200 or worse) (EVS Group 1995). As streptococci cause a higher rate of postinjection endophthalmitis than postcataract endophthalmitis, postinjection endophthalmitis is associated with poorer visual outcomes (Simunovic 2012). The diagnosis of endophthalmitis is made clinically, but it is confirmed by a positive aqueous or vitreous culture. A vitreous specimen is preferable because it has a higher detection rate than an aqueous specimen (54.9% versus 22.5%) (Barza 1997). Nevertheless, a negative culture, which occurs in about 30% of cases, does not exclude the diagnosis (Durand 2013).
Specimen collection is performed by either needle aspirate (an office procedure) or by vitrectomy (performed in the operating room) (Durand 2013). Vitrectomy is considered a useful diagnostic as well as therapeutic method, as it is the fastest way of removing an infection from the vitreous humor. It is favored in patients with severe vision loss (i.e. worse than hand motion) and rapidly worsening vision, or those who are likely to have endophthalmitis caused by more virulent strains (Barza 1997; Durand 2013).
Description of the intervention
Following specimen collection, endophthalmitis is treated immediately with antibiotics. Intravitreous administration of antibiotics is recommended, as other routes (e.g. topical, subconjunctival, intravenous) have been shown to be less effective (Durand 2013; Packer 2011). While the antibiotic regimen ideally targets the antibiotic sensitivities of the causative organism, it may not be possible to distinguish between different organisms from the presenting symptoms and signs alone. Broad‐spectrum antibiotics (i.e. vancomycin plus ceftazidime or amikacin) are therefore typically used first. If there is no improvement in 48 hours, the culture results dictate whether to give the patient another injection of vancomycin or ceftazidime (Durand 2013).
The use of steroids as adjunctive therapy for endophthalmitis remains controversial. In the 2004 American Society of Retina Specialists Preference and Trend Survey, 43% of respondents replied that they routinely use intravitreous steroids with or without systemic steroids in addition to intravitreous antibiotics for postcataract endophthalmitis (Pollack 2004). The most commonly used and widely studied steroid is dexamethasone (Bui 2014).
How the intervention might work
Gram‐positive bacteria cause the vast majority of cases of postoperative and postinjection endophthalmitis. Most of these cases are caused by coagulase‐negative staphylococci, which are commonly found in the normal flora of the ocular surface (Callegan 2002; Keynan 2012). All gram‐positive pathogens, except vancomycin‐resistant Enterococcus, are susceptible to vancomycin. Most gram‐negative pathogens that are responsible for bacterial endophthalmitis can be treated with an oxyimino‐cephalosporin (e.g. ceftazidime) or an aminoglycoside (e.g. amikacin) (Han 1996; Kunimoto 1999). These antibiotics are bacteriocidal (i.e. they destroy bacteria directly), in contrast to bacteriostatic antibiotics, which work by inhibiting bacterial growth and replication.
It has been hypothesized that steroids may be beneficial in the treatment of endophthalmitis due to their anti‐inflammatory effect. Experimental studies suggest that intraocular inflammation is incited by the growth of organisms and the antibiotic‐induced release of bacterial cell walls and cell wall components. While an inflammatory response is crucial to the clearance of the infecting organism, this response may result in irreversible damage of the sensitive photoreceptor cells and other secondary damage (Callegan 2002; Callegan 2006). Steroids decrease inflammation by inhibiting migration of macrophages, disruption of vascular membranes, and production of inflammatory mediators (Sadaka 2012). In a rat model of endophthalmitis, a higher level of expression of inflammatory mediators TNF‐α, IL‐1β, and IFN‐γ was associated with worse clinical presentation (Petropoulos 2006). Targeting both the infection and subsequent inflammatory response could have synergistic effects and lead to more rapid resolution of endophthalmitis and improvement in vision.
One argument against using steroids as adjunctive therapy is that they may worsen infection control, especially with fungal infections, because of the immunosuppressive properties of steroids. However, a retrospective review of 20 people with postcataract or post‐traumatic fungal endophthalmitis showed no difference in the visual outcomes of those who received intravitreous dexamethasone versus those who did not (Majji 1999). Furthermore, there was no statistically significant difference in mean vitreous vancomycin concentration among people with presumed postoperative bacterial endophthalmitis, with versus without intravitreous dexamethasone (Gan 2005).
Why it is important to do this review
Although endophthalmitis is a rare complication of intraocular surgery and intravitreous injection, its treatment is an important clinical issue due to the large number of people who undergo these procedures and the poor visual outcomes after endophthalmitis. Antibiotics are well established as the mainstay of treatment, but the role of steroids remains unclear. Most literature on the use of steroids as adjunctive therapy are reports of preclinical or retrospective case studies; randomized controlled studies on this topic tend to have small sample sizes. By consolidating results across multiple studies in this review, we aimed to determine whether combination therapy with antibiotics and steroids has an obvious benefit over monotherapy with antibiotics. In this review, we also planned to address the question of whether there are certain clinical settings in which adjunctive therapy with steroids may be particularly useful in treating endophthalmitis.
Objectives
To assess the effects of antibiotics combined with steroids versus antibiotics alone for the treatment of acute endophthalmitis following intraocular surgery or intravitreous injection.
Methods
Criteria for considering studies for this review
Types of studies
We conducted this review according to our published Cochrane protocol (Kim 2016). We included randomized controlled trials in our primary analyses. We also searched for comparative, non‐randomized studies (e.g. cohort studies) for the purposes of discussion only; we described and summarized the results of such studies qualitatively.
Types of participants
We included trials with participants who had clinically diagnosed endophthalmitis within three months of undergoing any intraocular surgery or intravitreous injection (acute endophthalmitis). There was no restriction regarding the result of the vitreous or aqueous culture. We excluded trials with participants with endogenous endophthalmitis, unless outcomes were reported separately by source of infection.
Types of interventions
We included trials that compared antibiotics and steroids versus antibiotics alone for the management of acute postprocedure endophthalmitis. There was no restriction on the method of administration, dose, frequency, or duration of antibiotics or steroids. In participants who received both antibiotics and steroids, the two treatments could have been administered simultaneously or one type of treatment could have been administered before the other treatment.
Types of outcome measures
Primary outcomes
The primary outcome was complete resolution of endophthalmitis one month after the initiation of therapy. We considered complete resolution as resolution of associated symptoms or hypopyon, or both, but we also accepted definitions used by the investigators of the included trials. We based our primary analysis on one‐month outcomes to evaluate whether using steroids as adjunctive therapy provides any benefit in the short term. We also compared outcomes at three, six, and 12 months whenever data were available.
Secondary outcomes
Proportion with best‐corrected visual acuity (BCVA) 20/40 (LogMAR 0.30) or better one month after the initiation of therapy.
Improvement in BCVA, defined as a proportion with a gain of 2 lines or more from baseline on a LogMAR chart or equivalent, at one month.
Mean change in intraocular pressure (IOP) from baseline at one month. When the mean one‐month change in IOP was not reported, we used the mean IOP at one month as long as the baseline IOP was similar between intervention groups.
Proportion with IOP less than 21 mmHg at one month.
Adverse events
We evaluated ocular and systemic adverse effects relating to antibiotic or steroid use that were reported in the included trials.
Economic data
We did not conduct formal cost‐effectiveness analyses, but we compared the costs associated with the interventions when data were available.
Search methods for identification of studies
Electronic searches
The Cochrane Eyes and Vision Information Specialist conducted systematic searches in the following databases. There were no study design, language or publication year restrictions. The date of the search was 8 December 2016.
Cochrane Central Register of Controlled Trials (CENTRAL; 2016, Issue 11) (which contains the Cochrane Eyes and Vision Trials Register) in the Cochrane Library (searched 8 December 2016) (Appendix 1)
MEDLINE Ovid (1946 to 8 December 2016) (Appendix 2)
Embase Ovid (1980 to 8 December 2016) (Appendix 3)
LILACS (Latin American and Caribbean Health Science Information Database (1982 to 8 December 2016) (Appendix 4)
ISRCTN registry (www.isrctn.com/editAdvancedSearch; searched 8 December 2016) (Appendix 5)
US National Institutes of Health Ongoing Trials Register ClinicalTrials.gov (www.clinicaltrials.gov; searched 8 December 2016) (Appendix 6)
World Health Organization International Clinical Trials Registry Platform (www.who.int/ictrp; searched 8 December 2016) (Appendix 7)
Searching other resources
We searched the reference lists of included trials to identify additional relevant trials. We did not search conference proceedings for the specific purposes of this review because they are searched annually by Cochrane Eyes and Vision and all reports from trials identified are included in CENTRAL.
Data collection and analysis
Selection of studies
Two review authors independently assessed the titles and abstracts of all records identified from the literature searches. We categorized each record as 'definitely relevant,' 'possibly relevant,' or 'definitely not relevant.' For records assessed as 'definitely relevant' or 'possibly relevant', we obtained the full‐text report and grouped them into studies. Two review authors independently reviewed each study and classified it as ‘include,’ ‘exclude,’ or ‘unsure.’ A third review author resolved any discrepancies between the two reviewing authors at each stage of selection. We documented studies that were excluded following review of the full‐text reports along with the reasons for exclusion in the Characteristics of excluded studies table. For studies assessed as ‘unsure’ after review of the full‐text reports, we contacted the study authors for clarification. In cases where we received no response after two weeks, we classified the study based on available information.
Data extraction and management
We extracted and recorded data‐related study methods, participant characteristics, and outcomes from the selected trials onto standard paper data collection forms. We pilot tested the forms with one study before using them for all included studies.
Two review authors independently extracted the data and resolved any discrepancies by discussion. One review author entered the data into Review Manager 5 (Review Manager 5 2014), and a second review author checked the data entry.
Assessment of risk of bias in included studies
Two review authors independently assessed each included study for risk of bias according to Chapter 8 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). We considered the following 'Risk of bias' domains.
Selection bias: random sequence generation, allocation concealment before randomization
Performance bias: masking of study participants and personnel
Detection bias: masking of outcome assessors
Attrition bias: loss to follow‐up
Reporting bias: selective outcome reporting
Other potential sources of bias (e.g. funding source, sponsor's involvement in trial)
We evaluated each domain for each trial as low, high, or unclear risk. Any discrepancies between the two review authors were resolved by discussion.
Measures of treatment effect
We measured treatment effects based on the types of data as described in Chapter 9 of the Cochrane Handbook for Systematic Reviews of Interventions (Deeks 2011), which included the following.
Dichotomous data
We reported the proportions of participants with complete resolution of endophthalmitis, improvement of BCVA, IOP less than 21 mmHg, and adverse effects. We calculated risk ratios (RRs) with 95% confidence intervals (CIs) to estimate treatment effects.
Continuous data
Outcomes with continuous data included mean IOP and mean change in IOP. We calculated mean differences with 95% CIs to estimate treatment effects.
Qualitative data
We described types of adverse effects and economic data qualitatively whenever quantitative description was not possible.
Unit of analysis issues
The individual (one study eye per participant) was the unit of analysis. There were no unit of analysis issues because each person was randomized to a treatment, and only one eye was affected.
Dealing with missing data
We contacted study investigators to request unpublished data, clarify unclearly reported data, and/or to provide reasons for missing data or loss to follow‐up. When we did not receive a response within two weeks, we used the information available in the published reports. We did not impute data for the purposes of this review. When we were unable to assume data were missing at random, we documented the outcomes with missing data and commented on potential implications in the Discussion of the review. We referred to Chapter 16 of the Cochrane Handbook for Systematic Reviews of Interventions for guidelines on how to interpret missing data (Higgins 2011).
Assessment of heterogeneity
We assessed clinical and methodological heterogeneity by examining variations in study characteristics (e.g. methodology, participant characteristics, interventions compared, follow‐up duration). We used the I2 value to assess the degree of statistical heterogeneity across studies. When the I2 value was greater than 50%, we considered the impact of heterogeneity to be substantial. We also performed a visual assessment of the forest plot to assess heterogeneity, with poor overlap of study estimates and confidence intervals indicating heterogeneity.
Assessment of reporting biases
Had 10 or more studies been included in a meta‐analysis, we would have created funnel plots in Review Manager 5 to assess publication bias. We evaluated selective outcome reporting as part of the ‘Risk of bias’ assessment. We planned to compare outcomes reported in included studies with outcomes reported in study protocols, published design and methods papers, or clinical trial registry records, when these were available.
Data synthesis
We presented the results in a narrative summary when there was substantial clinical or methodological heterogeneity, or when statistical heterogeneity and assessment of the forest plots indicated that meta‐analysis was not appropriate. In the absence of substantial heterogeneity, we combined the results of included studies and estimated treatment effects using meta‐analysis with a random‐effects model, or with a fixed‐effect model when the outcome was available from fewer than three studies.
Subgroup analysis and investigation of heterogeneity
We planned to perform subgroup analysis and investigate heterogeneity by type of intraocular procedure (e.g. cataract, glaucoma, retina), mode of steroid delivery, mode of antibiotic delivery, class of antibiotic used, and use of pars plana vitrectomy, when sufficient data were available.
Sensitivity analysis
We planned to conduct sensitivity analyses to evaluate the impact of excluding studies at high risk of bias in one or more domains, studies with only unpublished outcome data, and industry‐funded studies, when sufficient data were available.
Summary of findings
A 'Summary of findings' table provides the key findings regarding the certainty of evidence, the magnitude of effect of the interventions examined, and the sum of available data on all primary and secondary outcomes. Two review authors independently assessed the certainty of evidence for each outcome using the GRADE approach (GRADEpro 2014). Any discrepancies were resolved by discussion.
Results
Description of studies
Results of the search
We retrieved 1540 records from the electronic database search as of 8 December 2016 (Figure 1). We did not identify any additional records from reviewing the reference lists of included studies. The Cochrane Information Specialist removed 210 duplicate records. Of the remaining 1330 records, we selected 11 records for full‐text review. These 11 full‐text reports represented eight studies; we included three studies (six reports), identified one ongoing study (Lindstedt 2014), and excluded four studies (four reports).
1.
Study flow diagram.
Included studies
This review included three randomized controlled trials (Albrecht 2011; Das 1999; Gan 2005). In all trials, one eye per participant was randomized. Two trials stratified participants based on etiology groups: the groups for Albrecht 2011 were "post cataract," "bleb‐related," and "other" (trauma related, endogenous, post‐pars plana vitrectomy), while the groups for Das 1999 were "postoperative" and "post‐traumatic."
Types of participants
We included a total of 95 participants in this review after exclusion of 13 participants from the "bleb‐related" group and 17 participants from the "other" group, in Albrecht 2011, and 34 participants from the "post‐traumatic" group (Das 1999). We excluded the "bleb‐related" group because bleb‐related endophthalmitis usually occurs months to years after glaucoma surgery. All included studies enrolled participants with a similar clinical diagnosis of suspected bacterial endophthalmitis. Except for one participant in the "postoperative group" in Das 1999 who had a penetrating keratoplasty prior to being diagnosed with endophthalmitis, all included participants had postcataract endophthalmitis. The inclusion criteria for Gan 2005 stated that the cataract surgery must have been performed within six weeks of the onset of endophthalmitis and with the expectation that the postoperative visual acuity would be 20/100 or better. Albrecht 2011 and Das 1999 did not report a maximum time period between the date of surgery and onset of endophthalmitis. However, Albrecht 2011 reported the mean delay of presentation as 20.25 days when including three chronic cases and 8.6 days when excluding chronic cases. It was not possible to exclude the participants with chronic cases from this review. All included studies excluded any person with suspected fungal endophthalmitis. The studies were conducted in the following countries: South Africa (Albrecht 2011), India (Das 1999), and the Netherlands (Gan 2005). Albrecht 2011 and Das 1999 enrolled participants who were similar in age (mean age of about 60), while Gan 2005 enrolled an older patient population (mean age of about 80). Both males and females were included in all studies. Baseline characteristics were similar between intervention groups in Albrecht 2011. The other two studies did not report statistics comparing baseline characteristics between intervention groups. The overall positive culture rates were 52.5% and 69% for Albrecht 2011 and Gan 2005, respectively. The positive culture rate for the postoperative group in Das 1999 was 56.25%.
Types of interventions
All trials used intravitreous dexamethasone with intravitreous antibiotics in the steroid group. The choice of antibiotics differed by study: vancomycin and ceftazidime in Albrecht 2011, vancomycin and amikacin in Das 1999, and vancomycin and gentamicin in Gan 2005. In each trial, the control group received the same intravitreous antibiotics as the steroid group. Participants in the control group for Albrecht 2011 and Gan 2005 received antibiotics in a placebo solution, while participants in the control group in Das 1999 received intravitreous antibiotics without a placebo solution. No participants in Albrecht 2011 had a vitrectomy, while all participants in Das 1999 had vitrectomies. All participants with a baseline visual acuity at light perception only and some participants with complications in Gan 2005 had vitrectomies. The trials also differed on whether another injection of intravitreous dexamethasone and antibiotics was provided. Participants in Albrecht 2011 received a second injection of dexamethasone and antibiotics after 48 to 72 hours if necessary. Some participants in Das 1999 received another injection of intravitreous antibiotics, but no participants received additional intravitreous dexamethasone. In Gan 2005, the injection of antibiotics and dexamethasone or placebo was repeated once in all participants three to four days later. Other additional interventions are detailed in the Characteristics of included studies tables.
Types of outcomes
Primary outcome
No studies reported on our primary outcome, complete resolution of endophthalmitis. However, Das 1999 reported a somewhat similar outcome called "combined anatomical and functional success." This outcome was defined as the total percentage of participants with an IOP of at least 5 mmHg (i.e. anatomical success) and a BCVA of at least 6/120 (i.e. functional success). The time point for this outcome was unclear, but Das 1999 reported that visual acuity was measured at baseline as well as one, four, and 12 weeks after surgery. Other outcomes reported by Das 1999 involved quantitative inflammation scoring based on the clinical appearance of the cornea, anterior chamber, iris, and the vitreous. However, these outcomes were reported for postoperative and post‐traumatic participants as a whole (while combined anatomical and functional success were reported for each group separately), and were not included in the evidence for this review.
Secondary outcomes
No study investigators reported proportion of participants with BCVA of 20/40 or better as proposed in the Methods section. However, Albrecht 2011 reported the percentage of participants in each of the following Snellen visual acuity groups at three months: group 1 (6/6 to 6/18), group 2 (6/24 to 6/60), group 3 (worse than 6/60). Gan 2005 reported number of participants in each of the following Snellen visual acuity groups at three and 12 months: light perception (LP) to hand motion (HM), counting fingers (CF), 0.1 to 0.25, 0.4 to 1.0. We combined the three‐month Snellen visual acuity data by converting the percentage of participants into number of participants for each visual acuity group for Albrecht 2011 and re‐grouping the visual acuities for Gan 2005 using the cutoffs reported in Albrecht 2011. The "post cataract" group from Albrecht 2011 and all participants from Gan 2005 were included in the meta‐analysis. We also categorized 12‐month visual acuities from Gan 2005 into the groups used for the three‐month visual acuities (i.e. 6/6 to 6/18, 6/24 to 6/60, worse than 6/60). For both the three‐ and 12‐month visual acuity outcomes, we compared only the proportion of participants in group 1 (6/6 to 6/18) with those not in group 1 because this comparison was the most clinically useful. Of note, neither Albrecht 2011 nor Gan 2005 specified whether the visual acuities that they reported were 'best‐corrected.'
While no studies reported improvement in BCVA as a proportion with a gain of 2 lines or more, Albrecht 2011 reported mean number of lines of improvement on a Snellen visual acuity chart for the "post cataract" group. For each participant, the number of lines of improvement was determined by comparing the visual acuity at three months with the visual acuity at admission. While the Snellen visual acuities at baseline, three months, and 12 months were provided for all participants in Gan 2005, it was not possible to calculate the number of lines of improvement. Some of the participants had a baseline visual acuity of light perception, which does not have an equivalent visual acuity on the Snellen chart.
Except for what was included in the "combined anatomical and functional success" outcome, no outcomes related to visual acuity or IOP were reported by Das 1999. The other two studies did not report any IOP outcomes.
Adverse events
Two studies reported adverse events in participants (Albrecht 2011; Gan 2005). Das 1999 did not report any adverse event.
Economic data
No studies reported costs associated with the interventions.
Excluded studies
We excluded four studies after our review of full‐text reports. None of the studies was a randomized controlled trial. We have provided the reasons for exclusion in the Characteristics of excluded studies table.
Risk of bias in included studies
A summary of 'Risk of bias' assessment is shown in Figure 2.
2.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
We assessed two trials as at low risk of bias for reporting random sequence generation procedures (Albrecht 2011; Das 1999), and one trial at low risk for allocation concealment before randomization (Albrecht 2011). In Das 1999, randomly generated assignments were placed in sealed envelopes and opened by circulating nurses just before the preparation of the intraocular solutions. However, the risk of allocation concealment was unclear because the article does not mention whether the envelopes were sequentially numbered or opaque or whether they were opened sequentially. No information on random sequence generation and/or allocation concealment was provided in Gan 2005, so we assessed the risk of selection bias as unclear.
Masking (performance bias and detection bias)
We assessed Albrecht 2011 as at low risk of performance bias because a label hid the injection assignment from the participants and surgeon. After the trial author clarified over email that visual acuities were measured by nursing staff without any knowledge of the trial, we also assessed Albrecht 2011 as at low risk of detection bias. We classified the risk of performance and detection bias as unclear for the other two trials because the masking of participants and personnel administering the injection as well as outcome assessors was not described.
Incomplete outcome data
Das 1999 reported no losses to follow‐up or adverse events, so we assessed the risk of attrition bias as low. We also assessed Gan 2005 as having a low risk of attrition bias because only one participant was lost to follow‐up at 12 months, and the numbers of adverse events were similar between the steroid and placebo groups. We assessed the risk of attrition bias as unclear for Albrecht 2011 because four out of 62 participants in the placebo group were lost to follow‐up, and the reasons for losses to follow‐up were not reported (and unknown to trial author when asked by email). A modified intention‐to‐treat analysis was performed in which participants were analyzed by assigned treatment groups, but those without outcome information at three months' follow‐up were removed.
Selective reporting
We assessed all three trials at unclear risk of reporting bias because no trial protocols or clinical trial registry records were available to compare planned outcomes with reported outcomes. Furthermore, Das 1999 reported visual acuity outcomes differently than described in the Methods section of the study report. Instead of reporting visual acuity measurements at one, four, and 12 weeks postoperatively, Das 1999 reported a combined rate of functional success (visual acuity of at least 6/120) and anatomical success (IOP of at least 5 mmHg). It was not possible to verify whether the rate of functional and anatomical success was a planned outcome because we did not have access to the original study protocol.
Other potential sources of bias
We assessed the risk of bias as unclear for two studies because demographic characteristics were presented in a table, but no statistics were reported assessing whether baseline characteristics were equal between intervention groups (Das 1999; Gan 2005). We identified no other potential risk of bias in Albrecht 2011. None of the included studies reported industry funding or other funding sources that could have introduced a conflict of interest.
Effects of interventions
See: Table 1
Of a total of 95 randomized participants, 92 participants were analyzed. Forty‐six participants received intravitreous steroids in addition to intravitreous antibiotics and 46 participants received only intravitreous antibiotics.
Resolution of endophthalmitis
While none of the included studies reported on complete resolution of endophthalmitis, Das 1999 reported on the rate of combined anatomical and functional success. Anatomical success was defined as IOP of at least 5 mmHg and functional success was defined as visual acuity of at least 6/120. Among the participants with postoperative endophthalmitis, 87.5% (14 out of 16) of those who received intravitreous dexamethasone and intravitreous antibiotics achieved anatomical and functional success compared with 81.5% (13 out of 16) of the intravitreous antibiotics‐alone group (RR 1.08, 95% CI 0.80 to 1.45; Analysis 1.1). For an unknown reason, the percentage of participants who achieved combined success in the dexamethasone group was slightly higher (i.e. 93.8%; 15 out of 16) if calculated from Das 1999's Table 3 compared to the percentage reported in the study's Results text. We chose to include the percentage from the Results text in this review. We could not perform a pooled analysis for this outcome because the other two studies did not report any similar outcomes. We graded the certainty of the evidence as very low (‐1 for risk of bias, ‐1 for indirectness, ‐1 for imprecision).
1.1. Analysis.

Comparison 1 Adjunctive intravitreal dexamethasone versus antibiotics alone, Outcome 1 Combined anatomical and functional success rate.
Visual acuity
Albrecht 2011 and Gan 2005 assessed visual acuity outcomes in people with endophthalmitis after cataract surgery. At three months, more participants who received intravitreous dexamethasone and antibiotics had a good visual outcome (6/6 to 6/18) compared with those in the antibiotics‐alone group (RR 1.95, 95% CI 1.05 to 3.60; Analysis 1.2; Figure 3). We assessed the evidence as of low‐certainty (‐1 for risk of bias, ‐1 for imprecision). Only Gan 2005 reported visual acuity at 12 months following treatment. At 12 months, the risk ratio showed more participants in the dexamethasone group had a good visual outcome (6/6 to 6/18) compared to the antibiotics‐alone group (RR 2.00, 95% CI 0.98 to 4.08). We graded the evidence for the 12‐month visual acuity outcomes as low‐certainty (‐1 for risk of bias, ‐1 for imprecision).
1.2. Analysis.
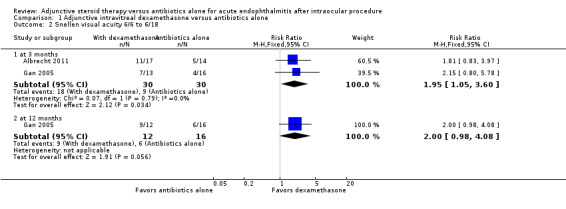
Comparison 1 Adjunctive intravitreal dexamethasone versus antibiotics alone, Outcome 2 Snellen visual acuity 6/6 to 6/18.
3.
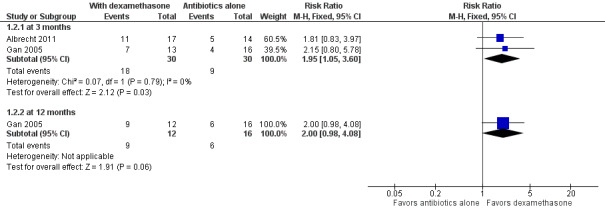
Forest plot of comparison: 1 Adjunctive intravitreal dexamethasone versus antibiotics alone, outcome: 1.2 Snellen visual acuity 6/6 to 6/18.
Albrecht 2011 was the only study that reported an outcome related to improvement in visual acuity. For participants in the postcataract group, there was no statistically significant difference in the mean number of Snellen lines of improvement at three months between those who received intravitreous dexamethasone and those who received placebo (4.1 versus 2.7, P = 0.33). Since Albrecht 2011 did not provide standard deviations or standard errors, we could not calculate estimated mean difference and 95% CI. We graded the certainty of the evidence as moderate (‐1 for publication bias); we could not assess imprecision due to the absence of CIs.
Das 1999 reported no visual acuity outcomes except for what was included in the combined anatomical and functional success outcome.
Intraocular pressure
Except for what was included in the combined anatomical and functional success outcome in Das 1999, no outcomes related to IOP were reported by any included study.
Adverse events
Albrecht 2011 and Gan 2005 reported occurrences of adverse events as described in Table 2. The total number of adverse events was 8 out of 30 (26.7%) for those who received dexamethasone versus 6 out of 30 (20.0%) for those who received antibiotics only. All three retinal detachments (RDs) reported by Albrecht 2011 followed complicated cataract surgery and the use of intravitreous steroids. Since RDs were the only adverse events shared by both studies, we performed a pooled analysis for this outcome with the postcataract group from Albrecht 2011 and all participants in Gan 2005 (Figure 4). The difference in the occurrence of retinal detachment between those who received dexamethasone and those who did not was uncertain (RR 1.57, 95% CI 0.50 to 4.90; Analysis 1.3); we judged the evidence for this outcome to be of very low‐certainty (‐1 for risk of bias, ‐2 for imprecision).
1. Adverse events.
| Adverse event | Study ID | With dexamethasone | Antibiotics alone |
| Retinal detachment | Albrecht 2011a | 3/17 (17.6%) | 0/14 (0%) |
| Gan 2005 | 3/13 (23.1%) | 4/16 (25.0%) | |
| Hypotony | Gan 2005 | 1/13 (7.7%) | 1/16 (6.3%) |
| Proliferative vitreoretinopathy | Gan 2005 | 1/13 (7.7%) | 0/16 (0%) |
| Seclusion of pupil | Gan 2005 | 0/13 (0%) | 1/16 (6.3%) |
aIncludes participants from the "post cataract" endophthalmitis group only.
4.
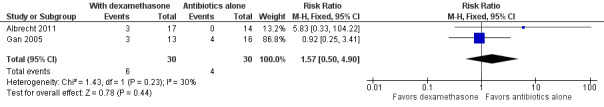
Forest plot of comparison: 1 Adjunctive intravitreal dexamethasone versus antibiotics alone, outcome: 1.3 Retinal detachment.
1.3. Analysis.
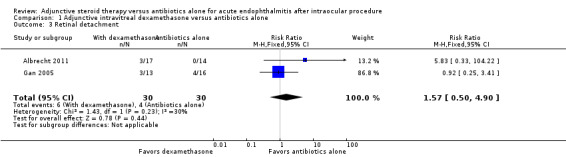
Comparison 1 Adjunctive intravitreal dexamethasone versus antibiotics alone, Outcome 3 Retinal detachment.
Gan 2005 also reported the number of participants who underwent a secondary procedure in each intervention group. Three participants in the dexamethasone group underwent a secondary procedure (one vitrectomy and gas for RD, one vitrectomy and silicone oil for persistent inflammation and proliferative vitreoretinopathy, one vitrectomy and silicone oil for RD) compared to four participants in the placebo group (three vitrectomy and silicone oil for RD, one vitrectomy for hypotony).
Das 1999 did not report any adverse events.
Economic data
No trial reported costs associated with the interventions.
Discussion
Summary of main results
In this review, we have presented the results of three randomized controlled trials that compared the effects of adjunctive steroid therapy versus antibiotics alone in people with suspected bacterial endophthalmitis after an intraocular procedure. All studies used intravitreous dexamethasone for adjunctive steroid therapy and a combination of intravitreous antibiotics with gram‐positive and gram‐negative bacterial coverage for the antibiotic therapy. Overall, the included trials provided insufficient evidence to either support or refute the use of adjunctive steroids for treatment of postoperative endophthalmitis (Table 1). One study found the effect of adjunctive steroid therapy on the rate of combined functional and anatomical success (as defined by visual acuity and IOP parameters) to be uncertain compared with antibiotics alone (Das 1999). A pooled analysis of two studies showed a higher proportion of participants with a good visual outcome (i.e. Snellen visual acuity of 6/6 to 6/18) in the steroid group (Albrecht 2011; Gan 2005). At 12 months, a higher proportion of participants who received dexamethasone had a good visual outcome versus those who did not (Gan 2005). One study reported no difference in visual acuity improvement from baseline between the participants treated with versus without steroids (Albrecht 2011). Only two studies reported adverse events, and the only adverse event shared between the two studies was retinal detachment (Albrecht 2011; Gan 2005). The effect of adjunctive steroid therapy on the occurrence of retinal detachment was uncertain.
Overall completeness and applicability of evidence
By expanding the inclusion criteria of this review to include participants with acute endophthalmitis after any type of intraocular procedure, we hoped that we would find trials that examined the treatment of endophthalmitis following a variety of intraocular procedures. However, almost all of the included trials reported outcomes for participants who were diagnosed with endophthalmitis after cataract surgery. Also, none of the studies examined the effect of adjunctive steroid therapy by causative organism. We were therefore unable to address whether adjunctive steroids are useful in certain clinical settings (e.g. specific procedures or causative organisms).
It should be noted that there were some differences in the treatment regimens between the different studies as well as what is considered current practice, which may limit the applicability of the evidence. Currently, a vitrectomy is recommended as soon as endophthalmitis is suspected and prior to intravitreous antibiotics for patients with a presenting visual acuity of light perception only (Barry 2013). If the patient does not improve within the first 24 to 48 hours, a vitrectomy would usually be considered (if not performed already), or a partial vitrectomy would be expanded to a full vitrectomy (Barry 2013). However, a vitrectomy was performed in the appropriate set of participants in Gan 2005 only. All participants had a vitrectomy prior to intravitreous injection in Das 1999, and no participants had a vitrectomy in Albrecht 2011. There were also differences in the choice of intravitreous antibiotics. All studies used vancomycin, but the second antibiotic used was ceftazidime in Albrecht 2011, amikacin in Das 1999, and gentamicin for Gan 2005. Current guidelines recommend using vancomycin and ceftazidime or amikacin. Although gentamicin also provides gram‐negative (including Pseudomonas) coverage, the use of gentamicin could make this study less applicable to current practice than the other two studies. Also, the variability in the treatment regimen following the additional injection is less than ideal. There are no guidelines on whether to give a second steroid injection, but it is recommended that a second injection of intravitreous antibiotics (antibiotic choice dependent on culture results) be considered in 24 to 48 hours if there is no improvement (Barry 2013). A repeat intravitreous antibiotic injection was only given when necessary in Albrecht 2011 and Das 1999, but all participants had a repeat injection three to four days later in Gan 2005.
The inconsistency in the types of outcomes reported and the follow‐up intervals made it difficult to combine the studies. Unfortunately, there is no standard way of quantifying resolution or successful management of endophthalmitis. Das 1999's measure of combined anatomical and functional success may be appropriate in a research setting, but may not be useful for determining whether treatment is successful in a clinical setting. Other potential ways of quantifying success are by clinical examination and patient‐reported eye pain (or other quality of life measures). The only pooled analysis performed on visual acuity outcomes was the proportion of participants in each of the Snellen visual acuity groups (as defined by Albrecht 2011) at three months. The evidence regarding visual acuity would have been more complete if visual acuity outcomes at additional time intervals were reported and shared among studies. Had we been able to examine how visual acuity changed over time between the two intervention groups, we would have a better understanding of whether adjunctive steroid therapy is harmful or beneficial at certain points in the recovery process.
Quality of the evidence
We found one study at overall low risk of bias, Albrecht 2011, and two studies with some methodological limitations (Das 1999; Gan 2005). We considered the evidence for the effect of adjunctive steroid therapy on combined anatomical and functional success to be of very low‐certainty; we downgraded for unclear risk of bias on most domains, indirectness of outcome, and imprecision of results with uncertainty in the direction of the true effect. We graded the certainty of the evidence as low for the proportion of participants with visual acuity of 6/6 to 6/18 at three months and 12 months due to unclear risk of bias and imprecision. We graded the certainty of evidence for mean lines of improvement as moderate due to potential publication bias since Albrecht 2011 assessed the outcome, but provided insufficient information to analyze the effect of adjunctive steroid therapy. Also, the absence of data needed to calculate confidence intervals prevented us from assessing precision for this outcome. Lastly, we graded the certainty of evidence for occurrence of retinal detachment as low due to unclear risk of bias and imprecision of results.
Potential biases in the review process
We did not identify any specific biases. It is likely that all relevant studies have been included in this review, as we used a highly sensitive strategy to search bibliographic databases, clinical trial databases, and reference lists of included studies. Also, two review authors performed major steps of the review process independently to minimize bias and errors.
Agreements and disagreements with other studies or reviews
While only three randomized controlled trials have compared adjunctive steroid therapy with antibiotics alone for the treatment of acute postoperative endophthalmitis, several non‐randomized studies have studied this comparison. Most studies found that use of intravitreous steroids had no significant effect on final visual outcome (Dev 2005; Eifrig 2003; Hall 2008; Miller 2004; Pijl 2010). Due to the retrospective nature of these studies, there was a wide range in the follow‐up periods within and among studies, ranging from months to years. Three studies included only participants with postcataract endophthalmitis (Dev 2005; Hall 2008; Pijl 2010) while the other two studies examined participants with endophthalmitis caused by a specific organism regardless of etiology, Streptococcus pneumoniae in Miller 2004 and Pseudomonas aeruginosa in Eifrig 2003. The one study that showed a harmful effect of adjunctive steroid therapy was Shah 2000, which found that administering adjunctive steroids in participants with postcataract endophthalmitis resulted in a significant worsening of visual acuity. This negative effect was seen when comparing mean visual acuities at one, three, and six months, as well as comparing the percentage of participants with a three‐line improvement by one and three months. The study investigators of Shah 2000 proposed steroid‐induced toxicity or blunting of immune response, or both as possible reasons for their findings. In contrast, one small retrospective case series on culture‐positive Staphylococcus aureus endophthalmitis (note: 26% of participants had non‐acute postoperative endophthalmitis) showed that addition of intravitreous dexamethasone to intravitreous antibiotics had a significantly beneficial effect on final visual outcome (Mao 1993). Similar to our study, a significant effect was found when examining the proportion with good visual outcome (i.e. visual acuity of 20/50 or better) at last follow‐up time point (mean: 13 months, range: one month to 4.5 years). Another study that observed a beneficial effect of adjunctive steroids examined the use of systemic and topical steroids, unlike this review (Koul 1989). This retrospective, multicenter study in Sweden reported that participants treated with a combination of topical and systemic steroids in conjunction with intravitreous antibiotics had better final visual acuities than those treated with no steroids or only topical steroids. Only a few studies compared the occurrence of adverse events. Dev 2005 reported no difference in the need for second procedure to manage endophthalmitis and occurrence of late postoperative complications, while Eifrig 2003 reported no difference in the need for enucleation or evisceration.
Considering the paucity of data and heterogeneity of the results, it is not possible to conclude whether intravitreous dexamethasone is beneficial or harmful in the treatment of acute endophthalmitis after intraocular procedure from these studies. A recent review concluded that since most of the current literature does not show an effect of adjunctive steroid therapy, and Shah 2000 showed a detrimental effect, the routine use of adjunctive intravitreous steroids for acute endophthalmitis is not supported at this time (Bui 2014). We agree with Bui 2014 that randomized controlled trials at low risk of bias and with adequate power should be completed to clarify whether adjunctive steroid therapy may be helpful in certain clinical settings and should be avoided in others.
Authors' conclusions
Implications for practice.
Evidence for the effectiveness of using steroids in conjunction with intravitreous antibiotics for the treatment of acute endophthalmitis following intraocular procedure remains too meager to provide support either for or against their usage. One trial indirectly measured treatment success by determining whether participants achieved a level visual acuity and intraocular pressure, but the effect of adjunctive steroid therapy on this outcome was uncertain. We found low‐certainty evidence to suggest a higher probability of having a good visual outcome at three and 12 months in the adjunctive steroid group. It also has not been established whether using adjunctive steroids affects the rate of complications (e.g. retinal detachment). Although this review showed a possible beneficial effect on visual acuity with adjunctive steroid use, most of the current evidence on this topic is inconsistent and has major limitations. It is therefore not possible to conclude whether or not to use adjunctive steroid therapy in the treatment of acute postprocedure endophthalmitis at this time.
Implications for research.
This review highlights the limited amount of randomized controlled trial data available for assessing the effect of adjunctive steroids for postprocedure endophthalmitis treatment as well as the potential ways to expand the current literature. Most of the randomized controlled trials and comparative, non‐randomized studies that have been conducted have focused on the treatment of endophthalmitis after cataract surgery. This is understandable considering that the large majority of endophthalmitis occurs after cataract surgery, but we believe that studying the effect of adjunctive steroids on other intraocular surgeries, as well as intraocular injections, is also important. Furthermore, we found that two of the three included studies combined patients of different etiologies (e.g. post‐traumatic) for some of the outcomes, which could make the patient population more heterogeneous than desired. Also, a larger randomized controlled trial may allow for the effect of adjunctive steroid therapy to be examined by type of causative organism. Studying the comparison of adjunctive steroid therapy and antibiotics alone in more specific patient populations may not only help us understand whether we should administer steroids for endophthalmitis, but also for which cases of endophthalmitis.
One of the major limitations of this review was the inconsistency of the outcomes reported by the trials and the time intervals at which these outcomes were collected. Since the reporting of most outcomes were not similar across studies, few meta‐analyses were possible. Any future trials should therefore report outcomes in ways that facilitate comparing findings with previous research. We also believe that it is important to formulate an outcome that considers the patient's symptoms (e.g. pain, discharge) and clinical examination (e.g. inflammatory changes) to measure the success of a treatment option. Ideally, a standardized measure would be created and adopted by multiple trials. An example of such an outcome is the inflammation score in Das 1999. Unfortunately, since Das 1999 reported this outcome for postoperative and post‐traumatic patients as a whole, we decided not to include this outcome in the review. Any future studies should also have earlier (i.e. a month or earlier) and later follow‐up intervals (i.e. one to several years) to determine whether adjunctive steroid therapy is beneficial or harmful throughout the post‐treatment period.
To maximize the applicability of future trials, we recommend employing the treatment regimen that is considered standard practice with regard to use of vitrectomy as well as antibiotic and steroid choice, dosage, and frequency. Also, researchers should conduct trials that have sufficient power to detect important clinical differences and implement methods to minimize bias. This requires that researchers not only to use methods that reduce the risk of bias (e.g. random sequence generation, allocation concealment, masking of participants, personnel, and outcome assessors, accounting for missing participants), but also report how they performed these methods.
Acknowledgements
We are grateful to Kristina Lindsley, Project Director of the US Cochrane Eyes and Vision, for her advice and support throughout the development and writing of this review. We thank Iris Gordon, Information Specialist, for creating and executing the electronic searches. We also acknowledge Richard Mills, Elizabeth Clearfield, Barbara Hawkins, and Sonal Singh for their comments on the protocol and Sami Kamjoo and Barbara Hawkins for their comments on the review. We thank Eric Albrecht for addressing our questions regarding a few of the 'Risk of bias' domains for his trial (Albrecht 2011).
Appendices
Appendix 1. CENTRAL search strategy
#1 MeSH descriptor: [Endophthalmitis] explode all trees #2 endophthalmiti* #3 ophthalmia #4 #1 or #2 or #3 #5 MeSH descriptor: [Anti‐Bacterial Agents] explode all trees #6 antibiotic* #7 MeSH descriptor: [Anti‐Infective Agents] explode all trees #8 chloramphenicol* #9 ciprofloxacin* #10 gentamicin* #11 levofloxacin* #12 neomycin* #13 ofloxacin* #14 polymyxin* #15 cefazolin* #16 cefuroxime* #17 moxifloxacin* #18 norfloxacin* #19 vancomycin* #20 cephtazidime* #21 amikacin* #22 tobramycin* #23 gatifloxacin* #24 #5 or #6 or #7 or #8 or #9 or #10 or #11 or #12 or #13 or #14 or #15 or #16 or #17 or #18 or #19 or #20 or #21 or #22 or #23 #25 MeSH descriptor: [Adrenal Cortex Hormones] explode all trees #26 steroid* #27 glucocorticoid* #28 dexamethasone* #29 betamethasone* #30 triamcinolone* #31 prednisolone* #32 fluorometholone* #33 #25 or #27 or #28 or #29 or #30 or #31 or #32 #34 #4 and #24 and #33
Appendix 2. MEDLINE Ovid search strategy
1. exp endophthalmitis/ 2. endophthalmiti$.tw. 3. ophthalmia.tw. 4. or/1‐3 5. exp anti bacterial agents/ 6. antibiotic$.tw. 7. exp Anti‐Infective Agents/ 8. chloramphenicol$.tw. 9. ciprofloxacin.tw. 10. gentamicin$.tw. 11. levofloxacin$.tw. 12. neomycin$.tw. 13. ofloxacin$.tw. 14. polymyxin$.tw. 15. cefazolin$.tw. 16. cefuroxime$.tw. 17. moxifloxacin$.tw. 18. norfloxacin$.tw. 19. vancomycin$.tw. 20. cephtazidime$.tw. 21. amikacin$.tw. 22. tobramycin$.tw. 23. gatifloxacin$.tw. 24. or/5‐23 25. exp Adrenal Cortex Hormones/ 26. steroid$.tw. 27. glucocorticoid$.tw. 28. dexamethasone$.tw. 29. betamethasone$.tw. 30. triamcinolone$.tw. 31. prednisolone$.tw. 32. fluorometholone$.tw. 33. or/25‐32 34. 4 and 24 and 33
Appendix 3. Embase Ovid search strategy
1. exp endophthalmitis/ 2. endophthalmiti$.tw. 3. ophthalmia.tw. 4. or/1‐3 5. exp antibiotic agent/ 6. antibiotic$.tw. 7. chloramphenicol$.tw. 8. ciprofloxacin.tw. 9. gentamicin$.tw. 10. levofloxacin$.tw. 11. neomycin$.tw. 12. ofloxacin$.tw. 13. polymyxin$.tw. 14. cefazolin$.tw. 15. cefuroxime$.tw. 16. moxifloxacin$.tw. 17. norfloxacin$.tw. 18. vancomycin$.tw. 19. cephtazidime$.tw. 20. amikacin$.tw. 21. tobramycin$.tw. 22. gatifloxacin$.tw. 23. or/5‐22 24. exp Steroids/ 25. steroid$.tw. 26. glucocorticoid$.tw. 27. dexamethasone$.tw. 28. betamethasone$.tw. 29. triamcinolone$.tw. 30. prednisolone$.tw. 31. fluorometholone$.tw. 32. or/24‐31 33. 4 and 23 and 32
Appendix 4. LILACS search strategy
endophthalmitis and antibiotic OR chloramphenicol OR ciprofloxacin OR gentamicin OR levofloxacin OR neomycin OR ofloxacin OR polymyxin cefazolin OR cefuroxime OR moxifloxacin OR norfloxacin OR vancomycin OR cephtazidime OR amikacin OR tobramycin OR gatifloxacin and steroid OR glucocorticoid OR dexamethasone OR betamethasone OR triamcinolone OR prednisolone OR fluorometholone
Appendix 5. ISRCTN search strategy
endophthalmitis AND (antibiotic OR chloramphenicol OR ciprofloxacin OR gentamicin OR levofloxacin OR neomycin OR ofloxacin OR polymyxin cefazolin OR cefuroxime OR moxifloxacin OR norfloxacin OR vancomycin OR cephtazidime OR amikacin OR tobramycin OR gatifloxacin) AND (steroid OR glucocorticoid OR dexamethasone OR betamethasone OR triamcinolone OR prednisolone OR fluorometholone)
Appendix 6. ClinicalTrials.gov search strategy
Interventional Studies | endophthalmitis | antibiotic OR chloramphenicol OR ciprofloxacin OR gentamicin OR levofloxacin OR neomycin OR ofloxacin OR polymyxin OR cefazolin OR cefuroxime OR moxifloxacin OR norfloxacin OR vancomycin OR amikacin OR tobramycin OR gatifloxacin OR cephtazidime
Appendix 7. WHO ICTRP search strategy
endophthalmitis = Condition AND antibiotic OR chloramphenicol OR ciprofloxacin OR gentamicin OR levofloxacin OR neomycin OR ofloxacin OR polymyxin OR cefazolin OR cefuroxime OR moxifloxacin OR norfloxacin OR vancomycin OR cephtazidime OR amikacin OR tobramycin OR gatifloxacin = Intervention
Data and analyses
Comparison 1. Adjunctive intravitreal dexamethasone versus antibiotics alone.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Combined anatomical and functional success rate | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 2 Snellen visual acuity 6/6 to 6/18 | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 2.1 at 3 months | 2 | 60 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.95 [1.05, 3.60] |
| 2.2 at 12 months | 1 | 28 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.0 [0.98, 4.08] |
| 3 Retinal detachment | 2 | 60 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.57 [0.50, 4.90] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Albrecht 2011.
| Methods |
Study design: randomized controlled trial Number randomized: 62 participants in total, 32 in postcataract endophthalmitis group (17 steroid, 15 placebo), 13 in bleb‐related endophthalmitis group (4 steroid, 9 placebo), 17 in other‐endophthalmitis group (9 steroid, 8 placebo) Exclusions after randomization: admission visual acuity was not recorded for 1 participant in other‐endophthalmitis group who received steroids Losses to follow‐up: 1 participant in postcataract group placebo group, 2 participants in bleb‐related placebo group, 1 participant in other placebo group Number analyzed: 57 participants in total, 31 in postcataract group (17 steroid, 14 placebo), 11 in bleb‐related group (4 steroid, 7 placebo), 15 in other group (8 steroid, 7 placebo) Unit of analysis: participant (1 eye per participant) Notes: other‐endophthalmitis group included 8 trauma‐related (4 steroid, 4 placebo), 3 endogenous (1 steroid, 2 placebo), and 5 post‐pars plana vitrectomy endophthalmitis (4 steroid, 2 placebo) participants |
|
| Participants |
Country: South Africa Setting: Groote Schuur Hospital (Cape Town) Study period: January 2001 to December 2005 Mean age (years): 59 (steroid), 61 (placebo) Gender: 11 men and 19 women (steroid); 18 men and 14 women (placebo) Inclusion criteria: all people with presumed bacterial endophthalmitis Exclusion criteria:
Equivalence of baseline characteristics: yes |
|
| Interventions |
Intervention (n = 30): intravitreal dexamethasone 0.4 mg/1 mL with intravitreal vancomycin 1 mg/0.1 mL and ceftazidime 2.225 mg/0.1 mL (replaced with amikacin 0.4 mg/1 mL for participants allergic to penicillin) Comparator (n = 32): intravitreal placebo 0.1 mL balanced salt solution with intravitreal vancomycin 1 mg/0.1 mL and ceftazidime 2.225 mg/0.1 mL Additional interventions (all participants): "Vitreous and aqueous samples were sent for microbiological analysis. A subconjunctival injection of vancomycin (25 mg/0.5 ml), ceftazidime (50 mg/0.5 ml) and betamethasone (1.5 mg/0.5 ml) was also administered at the end of the procedure. Post injection, patients received topical ofloxacin and topical dexamethasone. Patients were re‐injected after 48‐72 h if needed." Length of follow‐up: 2 to 4 months |
|
| Outcomes |
Outcomes:
Other findings reported: % positive culture rate overall, most common organism cultured, mean delay in presentation of endophthalmitis Adverse events reported: rhegmatogenous retinal detachment Intervals at which outcomes were assessed: baseline, 3 months |
|
| Notes |
References to other relevant studies: none Trial registration: none reported Funding source: none reported Declarations of interest: none reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | "The pharmacy randomised the patients within the three groups using standard computer generated randomisation tables." |
| Allocation concealment (selection bias) | Low risk | Allocation was performed by the pharmacy. |
| Masking of participants and personnel (performance bias) | Low risk | "A double‐blinding label (dexamethasone/placebo) masked the dexamethasone/placebo injection to both surgeon and patient." |
| Masking of outcome assessment (detection bias) | Low risk | Visual acuities were measured by nursing staff who had no knowledge of the trial, so they were unaware of the participants' intervention assignments. Note: this information was provided by the study author via email. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Out of the total 62 participants, 4 participants in the placebo group were lost to follow‐up and the admission visual acuity was not recorded for a participant in the steroid group. The reasons for losses to follow‐up were not reported (and unknown to the study author when asked by email). These participants were excluded from the final analysis (after randomization). |
| Selective reporting (reporting bias) | Unclear risk | We did not have access to the original study protocol to compare planned versus reported outcomes. |
| Other bias | Low risk | None identified. |
Das 1999.
| Methods |
Study design: randomized controlled trial Number randomized: 68 participants, 34 in postoperative endophthalmitis group, 34 in post‐traumatic endophthalmitis group Exclusions after randomization: 5 participants in total (because initial vitreous smear or final vitreous culture was positive for fungus), 2 in postoperative group, 3 in post‐traumatic group Losses to follow‐up: none reported Number analyzed: 63 participants in total, 32 in postoperative group (16 with steroid, 16 without steroid), 31 in post‐traumatic group (13 with steroid, 18 without steroid) Unit of analysis: participant (1 eye per participant) |
|
| Participants |
Country: India Setting: Retina Vitreous Services, L V Prasad Eye Institute (Hyderabad) Study period: January 1993 to December 1994 Mean age (years): 55.1 (postoperative/with steroid), 64.5 (postoperative/without steroid), 20.8 (post‐traumatic/with steroid), 12.2 (post‐traumatic/without steroid) Gender: 11 men/boys and 5 women/girls (postoperative/with steroid); 11 men/boys and 5 women/girls (postoperative/without steroid); 10 men/boys and 3 women/girls (post‐traumatic/with steroid); 10 men/boys and 8 women/girls (post‐traumatic/without steroid) Inclusion criteria: all people with suspected bacterial endophthalmitis Exclusion criteria:
Equivalence of baseline characteristics: not reported |
|
| Interventions |
Intervention (n = 29): intravitreal dexamethasone 0.4 mg/1 mL with vitrectomy and intravitreal vancomycin 1 mg and amikacin 0.4 mg Comparator (n = 34): vitrectomy and intravitreal vancomycin 1 mg and amikacin 0.4 mg Additional interventions (all participants): "Both intravitreal antibiotics and dexamethasone were injected only after obtaining the vitreous biopsy microscopy report from the microbiology laboratory. The usual time taken for the microscopy report was 10–15 minutes ... . All the eyes received subconjunctival antibiotics (same as the intravitreal antibiotics) at the end of the surgery; subconjunctival steroid was not given to any of the eyes in the study. All the patients also received intravenous antibiotics (gentamicin and cefazolin) for a period of 7 days ... . Four patients in the postoperative group and six patients in the post‐trauma group received a second injection of intravitreal antibiotics after the culture report was available. This was usually after 72 hours, and was culture adjusted. Intravitreal dexamethasone was not repeated." Length of follow‐up: 3 months |
|
| Outcomes |
Outcomes:
Other findings reported: % positive culture rate overall and per group Adverse events reported: none reported Intervals at which outcomes were assessed: baseline, 1, 4, and 12 weeks |
|
| Notes |
References to other relevant studies: none Trial registration: none reported Funding source: grants from the Hyderabad Eye Resarch Foundation (Hyderabad, India) Declarations of interest: none reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | "The randomisation was done using the standard randomisation table." |
| Allocation concealment (selection bias) | Unclear risk | "The sealed envelopes were opened by the circulating nurse just before the preparation of the IOABs [intraocular antibiotics]." It is unclear if the envelopes were sequentially numbered or opaque and if the envelopes were opened sequentially. |
| Masking of participants and personnel (performance bias) | Unclear risk | Masking of participants and personnel was not described. |
| Masking of outcome assessment (detection bias) | Unclear risk | "The inflammation score (IS) was done by two independent observers — one ophthalmologist and one senior optometrist, experienced with eye examination by slit lamp and indirect ophthalmoscope ... . The IS [at the follow‐up visits] was done by same two independent observers to quantitate the degree of inflammation at each evaluation." However, it is not clearly stated whether these observers were aware of the intervention assignment. Also, masking of personnel assessing visual acuity and intraocular pressure was not described. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No losses to follow‐up or adverse events were reported. |
| Selective reporting (reporting bias) | Unclear risk | We did not have access to the original study protocol to compare planned versus reported outcomes. Outcomes for inflammation score were reported as described in the Methods section. Visual acuity outcomes were not reported in Results section as mentioned in the Methods section; instead, a combined rate of functional success (i.e. visual acuity of at least 6/120) and anatomical success (i.e. intraocular pressure of at least 5 mmHg) was reported in the Results section. |
| Other bias | Unclear risk | Demographic details of each group were reported in tabular form, but no statistics were provided on whether these characteristics differed between groups. |
Gan 2005.
| Methods |
Study design: parallel‐group randomized controlled trial Number randomized: 29 participants in total, 13 in steroid group, 16 in placebo group Exclusions after randomization: none reported Losses to follow‐up: 1 participant in steroid group at 12 months Number analyzed: 29 participants in total, 13 in steroid group (1 participant lost to follow‐up at 12 months not included in 12‐month analysis), 16 in placebo group Unit of analysis: participant (1 eye per participant) |
|
| Participants |
Country: the Netherlands Setting: The Rotterdam Eye Hospital (Rotterdam) Study period: April 1999 to June 2000 (terminated prematurely because study drug dexamethasone sodium diphosphate was no longer available) Median age (years): 82 (steroid), 76 (placebo) Gender: 8 males and 5 females (steroid), 4 males and 12 females (placebo) Inclusion criteria: "consecutive patients with a diagnosis of suspected bacterial post‐cataract endophthalmitis" (defined as "(1) severe, sudden visual deterioration and (2) an inflammatory response deemed excessive (relative to the anticipated course after surgery) with cells and hypopyon in the anterior chamber or posterior segment with loss of fundus detail") Exclusion criteria:
Equivalence of baseline characteristics: not reported |
|
| Interventions |
Intervention (n = 13): intravitreal dexamethasone sodium diphosphate 0.4 mg (Decadron 20 mg/mL with 1 mg/mL paraben as preservative, resulting in 0.025 mg paraben in 0.1 mL) with intravitreal 0.2 mg vancomycin in 0.1 mL phosphate‐buffered saline and 0.05 mg gentamicin in 0.1 mL phosphate‐buffered saline Comparator (n = 16): placebo in 0.1 mL phosphate‐buffered saline with intravitreal 0.2 mg vancomycin in 0.1 mL phosphate‐buffered saline and 0.05 mg gentamicin in 0.1 mL phosphate‐buffered saline Additional interventions (all participants): "A limited core vitrectomy with an anterior chamber infusion was performed in patients with light perception only [prior to intravitreal injection] ... . The intravitreal injection of 0.2 mg vancomycin and dexamethasone or placebo was repeated once after 3 or 4 days ... . If the Gram‐staining or culture of the first biopsy material yielded Gram‐negative bacteria, 1 mg ceftazidime was immediately injected intravitreally, followed by continuous intravenous infusion of ceftazidime (6 g per day). After the first biopsy, patients used Predforte eye drops 6 times a day in a tapering schedule over the next 6 weeks and atropine 1% for 4 weeks. Additional procedures were allowed ... if the treating surgeon thought it to be in the patients’ best interest." Length of follow‐up: 12 months |
|
| Outcomes |
Outcomes:
Other findings reported: % positive culture rate overall and per group, most common organisms cultured Adverse events reported: retinal detachment, hypotony, seclusion of pupil, pucker, dropped intraocular lens Intervals at which outcomes were assessed: baseline and 3, 12 months |
|
| Notes |
References to other relevant studies: none Trial registration: none reported Funding source: none reported Declarations of interest: none reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | "The patients were randomized to either intravitreal dexamethasone or placebo as adjunct to standard antibiotic treatment." However, the randomization method was not described. |
| Allocation concealment (selection bias) | Unclear risk | Allocation concealment was not described. |
| Masking of participants and personnel (performance bias) | Unclear risk | Masking of participants and personnel was not described. |
| Masking of outcome assessment (detection bias) | Unclear risk | Masking of outcome assessment was not described. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 1 participant in steroid group did not return for her last follow‐up visit at 12 months, so her visual acuity data at 12 months were not included in the analysis. |
| Selective reporting (reporting bias) | Unclear risk | We did not have access to the original study protocol to compare planned versus reported outcomes. |
| Other bias | Unclear risk | Demographic characteristics of each group were reported in tabular form, but no statistics were provided on whether these characteristics differed between groups. Also, the major limitation of the study was the small sample size; the study stopped enrolling participants prematurely because the manufacturer of dexamethasone withdrew it from the market. |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Baum 1976 | This study is an interventional case series that discussed the clinical outcomes of 8 people with bacterial endophthalmitis who all received the same therapeutic regimen. |
| Breucker 1968 | This is a retrospective study that compared the clinical outcomes of treatment without versus with oral prednisone therapy in people with exogenous endophthalmitis. |
| Doft 1994 | This study is a prospective comparison of immediate vitrectomy versus vitreous tap and treatment with or without intravenous antibiotics. All participants received oral corticosteroids. |
| Rehak 2007 | This is a prospective, non‐randomized study examining the role of adjunctive systemic steroids in the treatment of acute postoperative endophthalmitis; 12 of 34 consecutive patients received systemic prednisolone in addition to a vitrectomy and intravitreous antibiotics. |
Characteristics of ongoing studies [ordered by study ID]
Lindstedt 2014.
| Trial name or title | Intravitreal dexamethasone treatment of bacterial endophthalmitis |
| Methods |
Study design: parallel‐group, double‐masked, randomized controlled trial Expected number to be randomized: 150 participants targeted in total; 81 total participants included in interim safety analysis, but number of participants per group was not reported Unit of analysis: participant (1 eye per participant) |
| Participants |
Country: the Netherlands Setting: University Medical Center Utrecht (Utrecht), Radboud University Nijmegen Medical Centre (Nijmegen), The Rotterdam Eye Hospital (Rotterdam) Age: No age restrictions were specified. No demographic characteristics were reported in interim analysis. Gender: No gender restrictions were specified. No demographic characteristics were reported in interim analysis. Inclusion criteria: "consecutive patients with a diagnosis of suspected bacterial post‐cataract surgery endophthalmitis" as defined by "(1) a decrease in visual acuity; and (2) an inflammatory response deemed excessive (relative to the anticipated course after surgery) with the presence of cells or hypopyon in the anterior chamber or the posterior segment with a loss of fundus detail" Exclusion criteria:
|
| Interventions |
Intervention 1: intravitreal 0.2 mg vancomycin in 0.1 mL phosphate‐buffered saline and 0.05 mg gentamicin in 0.1 mL phosphate‐buffered saline, followed by intravitreal 0.4 mg dexamethasone sodium diphosphate Intervention 2: intravitreal 0.2 mg vancomycin in 0.1 mL phosphate‐buffered saline and 0.05 mg gentamicin in 0.1 mL phosphate‐buffered saline, followed by intravitreal placebo in 0.1 mL sodium hydroxide‐buffered saline Additional interventions (all participants): "The intravitreal injection of 0.2 mg vancomycin and dexamethasone or placebo was repeated once after 3 or 4 days. The moment gram‐staining or culture of the vitreous biopsy revealed gram‐negative bacteria 1 mg ceftazidime was injected intravitreally followed by continuous intravenous infusion of ceftazidime (6 g per day) for two weeks. Patients used prednisolone 1 % eye drops 6 times a day after the biopsy in a tapering schedule over the next 6 weeks and atropine 1 % for 3 weeks. If the treating surgeon thought it to be in the patients’ best interest additional procedures were allowed." Length of follow‐up: 12 months |
| Outcomes |
Outcomes:
Intervals at which outcomes assessed: 3, 6, and 12 months |
| Starting date | February 2004 (as reported by interim safety analysis) |
| Contact information |
Contact author: Jan C van Meurs, MD Address: The Rotterdam Eye Hospital, Schiedamsevest 180, 3011 BH Rotterdam, the Netherlands Email: vanMeurs@oogziekenhuis.nl |
| Notes |
References to relevant studies (in interim study analysis): none Trial registration: Nederlands Trial Register, NTR 189 Funding source: Stichting Wetenschappelijk Onderzoek het Oogziekenhuis Notes: Participants were enrolled between November 2004 and October 2009 for the interim study analysis. The study drug was not available from 15 March 2005 to 28 June 2005 and from 20 July 2007 to 26 December 2008, so participant enrollment was interrupted during these time periods. The results of the safety analysis did not warrant premature discontinuation of the trial. According to the clinical trial register, the trial is finished (last updated on 26 January 2016). |
Differences between protocol and review
We intended to include participants with clinically diagnosed, culture‐positive endophthalmitis following intraocular procedure. However, we realized that due to the emergent nature of endophthalmitis, initial treatment with antibiotics with or without steroids is administered before the culture results are known. Also, none of the included trials reported outcomes separately for culture‐positive cases and culture‐negative cases (except for inflammation scoring in Das 1999, which was not included in the evidence for this review). We therefore amended the Types of participants section to state that participants would be included regardless of their culture result.
Since we did not have sufficient data and the number of trials was small, we did not perform any subgroup analyses. We also did not perform any sensitivity analyses because no included studies fitted any of the following criteria: high risk of bias in one or more domains, only unpublished outcome data, or industry funded.
Contributions of authors
Conceiving the review: CK, AC Designing the review: CK, MC, AC Co‐ordinating the review: CK Undertaking manual searches: CK, MC Screening search results: CK, MC Organizing retrieval of papers: CK Screening retrieved papers against inclusion criteria: CK, MC Appraising risk of bias of papers: CK, MC Abstracting data from papers: CK, MC Writing to authors of papers for additional information: CK Managing data for the review: CK Entering data into Review Manager 5: CK Analysis of data: CK Interpretation of data: CK, MC, AC Writing the review: CK, MC, AC
Sources of support
Internal sources
No sources of support supplied
External sources
Methodological support provided by the Cochrane Eyes and Vision US Project, supported by grant 1 U01 EY020522, National Eye Institute, National Institutes of Health, USA.
-
National Institute for Health Research (NIHR), UK.
- Richard Wormald, Co‐ordinating Editor for Cochrane Eyes and Vision (CEV) acknowledges financial support for his CEV research sessions from the Department of Health through the award made by the National Institute for Health Research to Moorfields Eye Hospital NHS Foundation Trust and UCL Institute of Ophthalmology for a Specialist Biomedical Research Centre for Ophthalmology.
- The NIHR also funds the CEV Editorial Base in London.
The views expressed in this publication are those of the authors and not necessarily those of the NIHR, NHS, or the Department of Health.
Declarations of interest
CK: None known. MC: None known. AC: None known.
New
References
References to studies included in this review
Albrecht 2011 {published data only}
- Albrecht E, Richards JC, Pollock T, Cook C, Myers L. Adjunctive use of intravitreal dexamethasone in presumed bacterial endophthalmitis: a randomised trial. British Journal of Ophthalmology 2011;95(10):1385‐8. [DOI] [PubMed] [Google Scholar]
Das 1999 {published data only}
- Das T, Jalali S, Gothwal VK, Sharma S, Naduvilath TJ. Dexamethasone in endophthalmitis treatment trial. A pilot study. American Academy of Ophthalmology Annual Meeting; 1996 October 30; Chicago (IL). Chicago (IL): American Academy of Ophthalmology, 1996:125.
- Das T, Jalali S, Gothwal VK, Sharma S, Naduvilath TJ. Dexamethasone in endophthalmitis treatment trial. Results of a pilot study. Investigative Ophthalmology & Visual Science 1997;38:ARVO E‐Abstract 2234. [Google Scholar]
- Das T, Jalali S, Gothwal VK, Sharma S, Naduvilath TJ. Intravitreal dexamethasone in exogenous bacterial endophthalmitis. A prospective randomized trial. American Academy of Ophthalmology Annual Meeting; 1995; San Francisco (CA). Chicago (IL): American Academy of Ophthalmology, 1994:131.
- Das T, Jalali S, Gothwal VK, Sharma S, Naduvilath TJ. Intravitreal dexamethasone in exogenous bacterial endophthalmitis: results of a prospective randomised study. British Journal of Ophthalmology 1999;83(9):1050‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gan 2005 {published data only}
- Gan IM, Ugahary LC, Dissel JT, Feron E, Peperkamp E, Veckeneer M, et al. Intravitreal dexamethasone as adjuvant in the treatment of postoperative endophthalmitis: a prospective randomized trial. Graefe's Archive for Clinical and Experimental Ophthalmology 2005;243(12):1200‐5. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Baum 1976 {published data only}
- Baum JL, Rao G. Treatment of postcataract bacterial endophthalmitis with periocular and systemic antibiotics and corticosteroids. Transactions. Section on Ophthalmology. American Academy of Ophthalmology and Otolaryngology 1976;81(5):952. [PubMed] [Google Scholar]
Breucker 1968 {published data only}
- Breucker G. On prednisone treatment in exogenous endophthalmitis. Klinische Monatsblätter für Augenheilkunde 1968;153(5):649‐51. [PubMed] [Google Scholar]
Doft 1994 {published data only}
- Doft BH, Kelsey SF, Wisniewski S, Metz DJ, Lobes L, Rinkoff J, et al. Treatment of endophthalmitis after cataract extraction. Retina 1994;14(4):297‐304. [DOI] [PubMed] [Google Scholar]
Rehak 2007 {published data only}
- Rehak M, Blatz R, Wiedemann P, Meier P. Primary vitrectomy with intraocular antibiotic application in postoperative endophthalmitis. Ophthalmologe 2007;104(11):958‐64. [DOI] [PubMed] [Google Scholar]
References to ongoing studies
Lindstedt 2014 {published and unpublished data}
- Lindstedt EW, Bennebroek CA, Werf DJ, Veckeneer M, Norel AO, Nielsen CC, et al. A prospective multicenter randomized placebo‐controlled trial of dexamethasone as an adjuvant in the treatment of postoperative bacterial endophthalmitis: interim safety analysis of the study drug and analysis of overall treatment results. Graefe's Archive for Clinical and Experimental Ophthalmology 2014;252(10):1631‐7. [DOI] [PubMed] [Google Scholar]
- NTR189. Intravitreal Dexamethason treatment of bacterial endophthalmitis. trialregister.nl/trialreg/admin/rctview.asp?TC=189 (date first received 23 August 2005).
Additional references
Barry 2013
- Barry P, Cordovés L, Gardner S. ESCRS guidelines for prevention and treatment of endophthalmitis following cataract surgery: data, dilemmas and conclusions. www.escrs.org/downloads/Endophthalmitis‐Guidelines.pdf 2013 (accessed 23 February 2016).
Barza 1997
- Barza M, Pavan PR, Doft BH, Wisniewski SR, Wilson LA, Han DP, et al. Evaluation of microbiological diagnostic techniques in postoperative endophthalmitis in the Endophthalmitis Vitrectomy Study. Archives of Ophthalmology 1997;115(9):1142‐50. [DOI] [PubMed] [Google Scholar]
Bui 2014
- Bui DK, Carvounis PE. Evidence for and against intravitreous corticosteroids in addition to intravitreous antibiotics for acute endophthalmitis. International Ophthalmology Clinics 2014;54(2):215‐24. [DOI] [PMC free article] [PubMed] [Google Scholar]
Callegan 2002
- Callegan MC, Engelbert M, Parke DW, Jett BD, Gilmore MS. Bacterial endophthalmitis: epidemiology, therapeutics, and bacterium‐host interactions. Clinical Microbiology Reviews 2002;15(1):111‐24. [DOI] [PMC free article] [PubMed] [Google Scholar]
Callegan 2006
- Callegan MC, Gilmore MS, Parke II DW. Chapter 48: The pathogenesis of infectious endophthlamitis. Duane's Foundations of Clinical Ophthalmology. CD‐ROM. Vol. 2, Lippincott Williams & Wilkins, 2006. [Google Scholar]
Coleman 2015
- Coleman AL. How big data informs us about cataract surgery: the LXXII Edward Jackson Memorial Lecture. American Journal of Ophthalmology 2015;160(6):1091‐103. [DOI] [PubMed] [Google Scholar]
Deeks 2011
- Deeks JJ, Higgins JP, Altman DG, editor(s). Chapter 9: Analysing data and undertaking meta‐analyses. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Dev 2005
- Dev S, Han DP, Mieler WF, Gonzalez‐Vivas R, Pulido JS, Mittra RA, et al. The role of intravitreal dexamethasone as an adjunct in the management of postoperative bacterial endophthalmitis. EVRS Educational Electronic Journal 2005;1(5):10‐7. [Google Scholar]
Du 2014
- Du DT, Wagoner A, Barone SB, Zinderman CE, Kelman JA, Macurdy TE, et al. Incidence of endophthalmitis after corneal transplant or cataract surgery in a Medicare population. Ophthalmology 2014;121(1):290‐8. [DOI] [PubMed] [Google Scholar]
Durand 2013
- Durand ML. Endophthalmitis. Clinical Microbiology and Infection 2013;19(3):227‐34. [DOI] [PMC free article] [PubMed] [Google Scholar]
Eifrig 2003
- Eifrig CW, Scott IU, Flynn HW Jr, Miller D. Endophthalmitis caused by Pseudomonas aeruginosa. Ophthalmology 2003;110(9):1714‐7. [DOI] [PubMed] [Google Scholar]
ESCRS Study 2007
- Endophthalmitis Study Group, European Society of Cataract & Refractive Surgeons. Prophylaxis of postoperative endophthalmitis following cataract surgery: results of the ESCRS multicenter study and identification of risk factors. Journal of Cataract & Refractive Surgery 2007;33(6):978‐88. [DOI] [PubMed] [Google Scholar]
EVS Group 1995
- Endophthalmitis Vitrectomy Study Group. Results of the Endophthalmitis Vitrectomy Study. A randomized trial of immediate vitrectomy and of intravenous antibiotics for the treatment of postoperative bacterial endophthalmitis. Archives of Ophthalmology 1995;113(12):1479‐96. [PubMed] [Google Scholar]
GRADEpro 2014 [Computer program]
- GRADE Working Group, McMaster University. GRADEpro GDT. Version accessed 13 January 2017. Hamilton (ON): GRADE Working Group, McMaster University, 2014.
Hall 2008
- Hall EF, Scott GR, Musch DC, Zacks DN. Adjunctive intravitreal dexamethasone in the treatment of acute endophthalmitis following cataract surgery. Clinical Ophthalmology 2008;2(1):139‐45. [DOI] [PMC free article] [PubMed] [Google Scholar]
Han 1996
- Han DP, Wisniewski SR, Wilson LA, Barza M, Vine AK, Doft BH, et al. Spectrum and susceptibilities of microbiologic isolates in the Endophthalmitis Vitrectomy Study. American Journal of Ophthalmology 1996;122(1):1‐17. [DOI] [PubMed] [Google Scholar]
Hatch 2009
- Hatch WV, Cernat G, Wong D, Devenyi R, Bell CM. Risk factors for acute endophthalmitis after cataract surgery: a population‐based study. Ophthalmology 2009;116(3):425‐30. [DOI] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JP, Altman DG, Sterne JAC, editor(s). Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Ianchulev 2016
- Ianchulev T, Litoff D, Ellinger D, Stiverson K, Packer M. Office‐based cataract surgery: population health outcomes study of more than 21,000 cases in the United States. Ophthalmology 2016;123(4):723‐8. [DOI] [PubMed] [Google Scholar]
Jackson 2003
- Jackson TL, Eykyn SJ, Graham EM, Stanford MR. Endogenous bacterial endophthalmitis: a 17‐year prospective series and review of 267 reported cases. Survey of Ophthalmology 2003;48(4):403‐23. [DOI] [PubMed] [Google Scholar]
Jackson 2014
- Jackson TL, Paraskevopoulos T, Georgalas I. Systematic review of 342 cases of endogenous bacterial endophthalmitis. Survey of Ophthalmology 2014;59(6):627‐35. [DOI] [PubMed] [Google Scholar]
Keay 2012
- Keay L, Gower EW, Cassard SD, Tielsch JM, Schein OD. Postcataract surgery endophthalmitis in the United States: analysis of the complete 2003 to 2004 Medicare database of cataract surgeries. Ophthalmology 2012;119(5):914‐22. [DOI] [PMC free article] [PubMed] [Google Scholar]
Keynan 2012
- Keynan Y, Finkelman Y, Lagace‐Wiens P. The microbiology of endophthalmitis: global trends and a local perspective. European Journal of Clinical Microbiology 2012;31(11):2879‐86. [DOI] [PubMed] [Google Scholar]
Koul 1989
- Koul S, Philipson BT, Philipson A. Visual outcome of endophthalmitis in Sweden. Acta Ophthalmologica 1989;67(5):504‐9. [DOI] [PubMed] [Google Scholar]
Kunimoto 1999
- Kunimoto DY, Das T, Sharma S, Jalali S, Majji AB, Gopinathan U, et al. Microbiologic spectrum and susceptibility of isolates: part I. Postoperative endophthalmitis. Endophthalmitis Research Group. American Journal of Ophthalmology 1999;128(2):240‐2. [DOI] [PubMed] [Google Scholar]
Lalwani 2008
- Lalwani GA, Flynn HW Jr, Scott IU, Quinn CM, Berrocal AM, Davis JL, et al. Acute‐onset endophthalmitis after clear corneal cataract surgery (1996‐2005). Clinical features, causative organisms, and visual acuity outcomes. Ophthalmology 2008;115(3):473‐6. [DOI] [PubMed] [Google Scholar]
Lemley 2007
- Lemley CA, Han DP. Endophthalmitis: a review of current evaluation and management. Retina 2007;27(6):662‐80. [DOI] [PubMed] [Google Scholar]
Lyall 2012
- Lyall DM, Tey A, Foot B, Roxburgh ST, Virdi M, Robertson C, et al. Post‐intravitreal anti‐VEGF endophthalmitis in the United Kingdom: incidence, features, risk factors, and outcomes. Eye 2012;26(12):1517‐26. [DOI] [PMC free article] [PubMed] [Google Scholar]
Majji 1999
- Majji AB, Jalali S, Das T, Gopinathan U. Role of intravitreal dexamethasone in exogenous fungal endophthalmitis. Eye 1999;13(Pt 5):660‐5. [DOI] [PubMed] [Google Scholar]
Mamalis 2002
- Mamalis N, Kearsley L, Brinton E. Postoperative endophthalmitis. Current Opinion in Ophthalmology 2002;13(1):14‐8. [DOI] [PubMed] [Google Scholar]
Mao 1993
- Mao LK, Flynn HW Jr, Miller D, Pflugfelder SC. Endophthalmitis caused by Staphylococcus aureus. American Journal of Ophthalmology 1993;116(5):584‐9. [DOI] [PubMed] [Google Scholar]
Miller 2004
- Miller JJ, Scott IU, Flynn HW Jr, Smiddy WE, Corey RP, Miller D. Endophthalmitis caused by Streptococcus pneumoniae. American Journal of Ophthalmology 2004;138(2):231‐6. [DOI] [PubMed] [Google Scholar]
Packer 2011
- Packer M, Chang DF, Dewey SH, Little BC, Mamalis N, Oetting TA, et al. Prevention, diagnosis, and management of acute postoperative bacterial endophthalmitis. Journal of Cataract & Refractive Surgery 2011;37(9):1699‐714. [DOI] [PubMed] [Google Scholar]
Petropoulos 2006
- Petropoulos IK, Vantzou CV, Lamari FN, Karamanos NK, Anastassiou ED, Pharmakakis NM. Expression of TNF‐alpha, IL‐1beta, and IFN‐gamma in Staphylococcus epidermidis slime‐positive experimental endophthalmitis is closely related to clinical inflammatory scores. Graefe's Archive for Clinical and Experimental Ophthalmology 2006;244(10):1322‐8. [DOI] [PubMed] [Google Scholar]
Pijl 2010
- Pijl BJ, Theelen T, Tilanus MA, Rentenaar R, Crama N. Acute endophthalmitis after cataract surgery: 250 consecutive cases treated at a tertiary referral center in the Netherlands. American Journal of Ophthalmology 2010;149(3):482‐7. [DOI] [PubMed] [Google Scholar]
Pollack 2004
- Pollack JS. Preference and trends survey [Online]. Retina Times. Chicago, IL: American Society of Retina Specialists, 2004; Vol. 22 (Issue 31).
Review Manager 5 2014 [Computer program]
- Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Sadaka 2012
- Sadaka A, Durand ML, Gilmore MS. Bacterial endophthalmitis in the age of outpatient intravitreal therapies and cataract surgeries: host‐microbe interactions in intraocular infection. Progress in Retinal and Eye Research 2012;31(4):316‐31. [DOI] [PMC free article] [PubMed] [Google Scholar]
Shah 2000
- Shah GK, Stein JD, Sharma S, Sivalingam A, Benson WE, Regillo CD. Visual outcomes following the use of intravitreal steroids in the treatment of postoperative endophthalmitis. Ophthalmology 2000;107(3):486‐9. [DOI] [PubMed] [Google Scholar]
Simunovic 2012
- Simunovic MP, Rush RB, Hunyor AP, Chang AA. Endophthalmitis following intravitreal injection versus endophthalmitis following cataract surgery: clinical features, causative organisms and post‐treatment outcomes. British Journal of Ophthalmology 2012;96(6):862‐6. [DOI] [PubMed] [Google Scholar]
Stein 2008
- Stein JD, Ruiz D Jr, Belsky D, Lee PP, Sloan FA. Longitudinal rates of postoperative adverse outcomes after glaucoma surgery among Medicare beneficiaries 1994 to 2005. Ophthalmology 2008;115(7):1109‐16. [DOI] [PMC free article] [PubMed] [Google Scholar]
West 2005
- West ES, Behrens A, McDonnell PJ, Tielsch JM, Schein OD. The incidence of endophthalmitis after cataract surgery among the U.S. Medicare population increased between 1994 and 2001. Ophthalmology 2005;112(8):1388‐94. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Kim 2016
- Kim CH, Chen MF, Coleman AL. Adjunctive steroid therapy versus antibiotics alone for acute endophthalmitis after intraocular procedure. Cochrane Database of Systematic Reviews 2016, Issue 3. [DOI: 10.1002/14651858.CD012131] [DOI] [PMC free article] [PubMed] [Google Scholar]


