Abstract
Cytochrome P450 lanosterol 14α-demethylase (CYP51) is a key enzyme in sterols and steroids biosynthesis that can induce meiotic resumption in mouse oocytes. The present study investigated the expression mechanism and function of CYP51 during FSH-induced mouse cumulus oocyte complexes (COCs) meiotic resumption. FSH increased cAMP-dependent protein kinase (PKA) RIIβ level and induced cAMP response element-binding protein (CREB) phosphorylation and CYP51 expression in cumulus cells before oocyte meiotic resumption. Moreover, CYP51 and epidermal growth factor (EGF)-like factor [amphiregulin (AR)] expression were blocked by 2-naphthol-AS-Ephosphate (KG-501) (a drug interrupting the formation of CREB functional complex). KG-501 and RS21607 (a specific inhibitor of CYP51 activity) inhibited oocyte meiotic resumption, which can be partially rescued by progesterone. These two inhibitors also inhibited FSH-induced MAPK phosphorylation. EGF could rescue the suppression by KG-501 but not RS21607. Furthermore, type II PKA analog pairs, N6-monobutyryl-cAMP plus 8-bromo-cAMP, increased PKA RIIβ level and mimicked the action of FSH, including CREB phosphorylation, AR and CYP51 expression, MAPK activation, and oocyte maturation. All these data suggest that CYP51 plays a critical role in FSH-induced meiotic resumption of mouse oocytes. CYP51 and AR gene expression in cumulus cells are triggered by FSH via a type II PKA/CREB-dependent signal pathway. Our study also implicates that CYP51 activity in cumulus cells participates in EGF receptor signaling-regulated oocyte meiotic resumption.
FULLY GROWN MAMMALIAN oocytes are arrested at the germinal vesicle (GV) stage of the first meiotic division by meiosis-arresting factors such as cAMP (1, 2) and hypoxanthine (HX) (3, 4). Reinitiation of meiosis occurs as a consequence of the preovulatory surge of gonadotropins, particularly LH (5). Oocytes of GV stage undergo gonadotropin-independent spontaneous meiotic maturation when they are removed from follicles (6). FSH also induces oocyte meiotic resumption in vitro when spontaneous maturation is prevented by HX or cAMP-elevating agents (7, 8). The FSH-induced model of cumulus oocyte complexes (COCs) is generally used to study mechanism of gonadotropins-induced oocyte maturation, because LH receptors are absent or express at very low levels in cumulus cells (9, 10) and LH has no effect on mouse COC maturation (11, 12, 13). It has been shown by many groups that the change of cAMP level and activation of MAPK are essential for gonadotropin-induced oocyte maturation (14, 15). However, the signal transduction pathway between cAMP and MAPK is not well understood.
It has been well established that a notable surge of cAMP in follicle and COC may trigger germinal vesicle breakdown (GVBD) after stimulation with LH or FSH (16, 17). A linear cAMP signaling cascade generally requires activation of cAMP-dependent protein kinase (PKA), which modulates the cAMP response by phosphorylating certain substrates. In the inactive state, PKA is a tetramer consisting of two identical catalytic (C) subunits, which includes at least three subtypes (Cα, Cβ, Cγ), and two regulatory (R) subunits (RI, RII) that each exist as α- and β-isoforms (18, 19). Binding of cAMP to the two sites on each R subunit leads to dissociation of RC dimers, releasing activated C subunits (20). There are two types of PKA, I and II, differing from their R subunits. Unlike type I PKA (PKAI) which is more sensitive to slight increases of cAMP, type II PKA (PKAII) is preferentially responsive to higher and discrete cAMP levels (20, 21). Both type I and II PKA are detected within rodent oocytes and cumulus cells (22, 23). By using site-selective cAMP analogs, recent studies show that activation of PKA I suppresses meiotic resumption of COCs and denuded oocytes (DOs); however, the activation of PKAII induces GVBD of COCs but inhibits spontaneous maturation of DOs (23, 24). These works imply that PKAII in cumulus cells may be the major subtype responsible for inducing oocyte meiotic resumption.
Because the oocyte is lacking gonadotropin receptors, it has been hypothesized that gonadotropin exerts its effect via a positive meiosis factor synthesized by cumulus cells indirectly, rather than a direct action on the oocyte (7). It has been proved that, in lower vertebrates such as Xenopus, progesterone stimulated by gonadotropins serves as such a positive meiosis factor (25, 26), whereas in mammals, the positive factors and their product chain are still not clear. Results by using sterol biosynthesis inhibitors support that follicular fluid meiosis-activating sterols (FF-MAS) is a potentially positive factor of the FSH-induced signal transduction pathway (13, 27). In addition, mouse DOs arrested by 3-isobutyl-1-methylxanthine are triggered to mature by testosterone or estradiol (28). Furthermore, some studies indicate that FSH and LH induce meiotic resumption through a secretion of progesterone by cumulus cells in mouse and pig (29, 30, 31). Thus, gonadotropin-induced steroidogenesis may also play an important role in mammal oocyte maturation. More recently, several studies have demonstrated either of the two gonadotropins uses epidermal growth factor (EGF) receptor (EGFR) signaling as a potential central pathway in murine oocyte maturation. EGF-like factors, especially amphiregulin (AR), participate in gonadotropin-induced mouse oocytes meiotic resumption through a transcriptional regulation (32, 33, 34). Notably, steroid production, such as progesterone, regulated by epidermal EGFR signaling, is necessary for LH-induced oocyte maturation (31). It is interesting to explore the action that gonadotropin exerts on the induction of EGFR signaling and steroidogenesis in cumulus cells.
Involving FF-MAS and steroids biosynthesis, cytochrome P450 lanosterol 14α-demethylase (CYP51) is a rate-limiting enzyme for catalyzing the first step after cyclization in sterol biosynthesis (35). The results that FSH-induced meiotic resumption could be inhibited by specific inhibitors of CYP51 in mouse follicle-enclosed oocytes (FEOs) and porcine COCs (27, 36) reveal that CYP51 plays an important role in FSH-induced oocyte meiotic process. As an evolutionarily conserved gene of the cytochrome P450 superfamily (37), CYP51 gene contains sterol-regulatory element and cAMP response element (CRE), which implies two regulatory mechanisms: general feedback regulation by sterols levels and characteristic cAMP response element-binding protein (CREB)/CRE modulator transcription (35, 38, 39). Similarly, the CRE is also conserved in AR promoter (40). A role of CREB in the regulation of AR mRNA expression is further supported in epithelial cells (41) and tumor cells (42).
To better understand the roles of CREB signaling and steroidogenesis on FSH action, the present experiments were designed to determine 1) whether PKAII and CREB phosphorylation in cumulus cells are involved in FSH-induced oocyte maturation; 2) the effects of PKAII activation and CREB phosphorylation on CYP51 and AR expression; 3) the possible role of CYP51 on MAPK phosphorylation and meiotic resumption.
RESULTS
FSH Increased PKA RIIβ Level during Oocyte Maturation
In the preliminary experiment shown in Fig. 1A, FSH significantly induced GVBD (12 h, 57.78%; 24 h, 78.02%) compared with oocytes cultured in the control treatment (12 h, 15.27%; 24 h, 23.74%, P < 0.01, respectively), which coincided with the results of previous reports (13, 14).
Fig. 1.

The Effect of FSH on PKA RIIβ and C Subunit Levels within Cumulus Cells during Oocyte Meiotic Resumption
COCs were cultured with or without 50 IU/liter FSH in HX medium for 24 h. The rate of GVBD was scored at 4-h intervals during the culture. Data were represented as mean percentage of GVBD ± sem of three independent experiments. *, P < 0.01, compared with each corresponding time point in the control group (A). Cumulus cells from 50 COCs treated with or without FSH were collected at various time points for immunoblotting. The densitometric data of RIIβ and C (Cat) subunits were normalized according to GAPDH to evaluate the relative abundance of the targets. Columns with different letters were significantly different (P < 0.05) (B). The experiments were performed three times with similar results.
Western blotting analysis showed that, under the stimulation of FSH, PKA RIIβ in cumulus cells significantly increased at 2 h and decreased in the following incubation period, whereas only slight RIIβ was detected in the control group during 0–12 h of culture. In addition, there was a lower C subunit level in the FSH group compared with the control group at each corresponding time point (Fig. 1B).
Site-Selective cAMP Analogs of PKAII Increased PKA RIIβ Level and Stimulated Oocyte Meiotic Resumption
As shown in Fig. 2A, using 250 μm N6-monobutyryl-cAMP (N6-cAMP) as a priming compound, 8-bromo-cAMP (8-Br-cAMP) promoted oocyte GVBD in a dose-dependent manner (0 μm: 24.51%; 50 μm: 32.92%; 100 μm: 51.89%; 250 μm: 74.80%; 500 μm: 72.07%; control: 27.71%) and cumulus expansion (data not shown) of COCs, but had no effect on maturation of DOs (data not shown). Using the same drug treatment, PKA RIIβ level within cumulus cells was increased in a dose-dependent fashion, whereas PKA C subunit had no obvious change (Fig. 2B). Treatment of COCs with N6-cAMP and 8-Br-cAMP, each at a concentration of 250 μm, was the most effective for stimulating PKA RIIβ level and oocytes maturation. Therefore, paired combination of these two cAMP analogs, each at 250 μm, was used for activating PKAII in experiments 1–6 (see Materials and Methods).
Fig. 2.
The Effect of Pulsing COCs with PKAII Analog Pairs on PKA RIIβ and C Subunit Levels and Meiotic Maturation
COCs were incubated in HX medium supplemented with 250 μm N6-cAMP (N6) and increasing concentration of 8-Br-cAMP (8-Br) (0–500 μm) for 3 h, and then transferred into a fresh, analog pairs-free HX medium for an additional 20 h. The GVBD rate was recorded after culture. COCs were cultured in HX medium as a control. Columns with different letters were significantly different (P < 0.05) (A). Cumulus cells were collected at 3 h for Western blot analysis of RIIβ and C subunits (Cat) (B). The experiment was performed at least three times with similar results.
Phosphorylation of CREB in Cumulus Cells Involved in FSH- and PKAII Analog Pairs-Induced Oocyte Meiotic Resumption
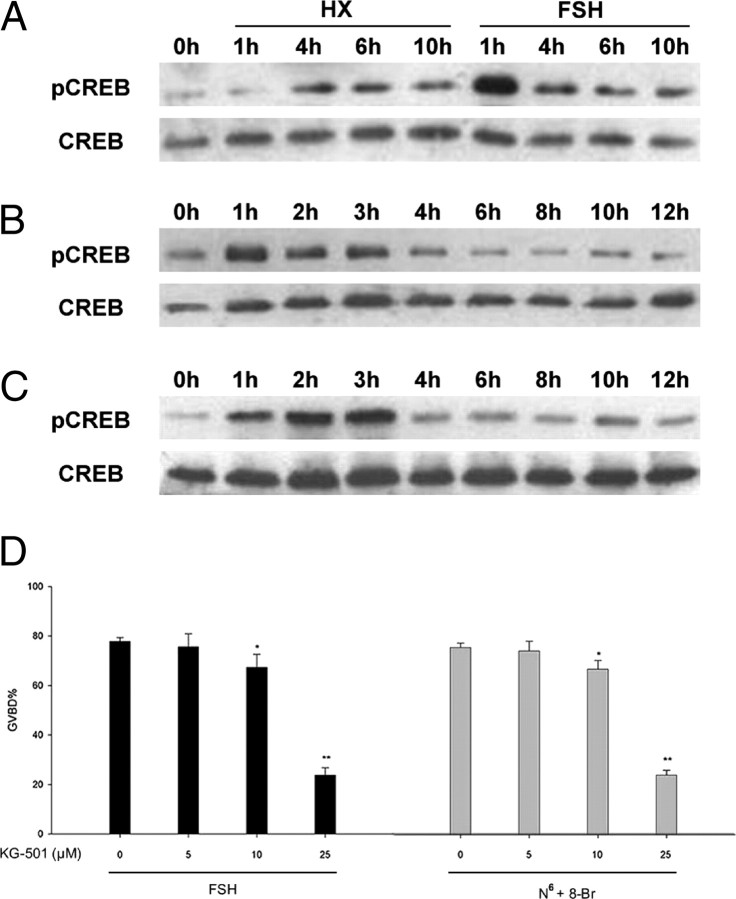
During exposure to FSH, there was a significant increase of phosphorylated CREB in cumulus cells at 1–3 h (Fig. 3B), whereas weak phosphorylated forms with no change was observed in control group (Fig. 3A). Likewise, transient high level of phosphorylation of the CREB in response to N6-cAMP plus 8-Br-cAMP was observed at 2–3 h, followed by immediate dephosphorylation (Fig. 3C).
Fig. 3.
Kinetics of CREB Phosphorylation within Cumulus Cells of COCs in Response to FSH or N6-cAMP Plus 8-Br-cAMP and the Effect of KG-501 on Oocyte Maturation
Cumulus cells from 50 COCs were collected at various time point for immunoblotting with anti-phos CREB and anti-CREB antibodies.COCs were cultured in HX medium with or without 50 IU/liter FSH (A). COCs were cultured in HX medium containing 50 IU/liter FSH (B) or pulsing with 250 μm N6-cAMP and 8-Br-cAMP for 3 h (C). Different concentrations of KG-501 (0–25 μm) were added when mouse COCs were cultured in HX medium containing 50 IU/liter FSH or pulsing with 250 μm N6-cAMP (N6) plus 8-Br-cAMP (8-Br). The percentage of GVBD was recorded at 24 h of culture. Columns with different letters were significantly different (P < 0.05) (D). The experiment was performed three times with similar results. pCREB, Phosphorylated CREB.
2-Naphthol-AS-Ephosphate (KG-501) (a drug interrupting the formation of CREB functional complex) inhibited FSH- and N6-cAMP plus 8-Br-cAMP-induced oocyte meiotic resumption in a dose-dependent manner (0 μm, 78.96% and 76.25%; 5 μm, 74.35% and 72.54%; 10 μm, 68.48% and 65.89%; 25 μm, 24.78% and 25.71%) (Fig. 3D). KG-501 (50 μm) significantly inhibited oocyte maturation induced by FSH but increased the percentage of oocyte degeneration (data not shown). Thus, treatment of COCs with 25 μm KG-501 was the most effective dose and was used in experiments 1–6.
The Role and Kinetics of CYP51 in FSH-Induced Oocyte Meiotic Resumption
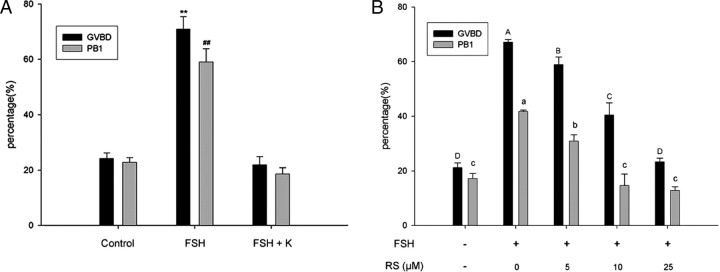
The effect of CYP51 on FSH-induced oocyte maturation was studied by the inhibitors. Under the stimulation of 50 IU/liter FSH, the percentages of the oocyte undergoing GVBD and first polar body (PB1) extrusion increased to 71.00% and 59.18%, respectively. Ketoconazole (10 μm) drastically inhibited GVBD and PB1 extrusion induced by FSH to 21.84% and 18.52%, separately (Fig. 4A). Likewise, RS21607, a more potent and highly specific inhibitor of CYP51 (44), showed a dose-dependent block of GVBD (0 μm, 67.13%; 5 μm, 58.87%; 10 μm, 40.45%; 25 μm, 23.24%) and PB1 extrusion (0 μm, 44.70%; 5 μm, 33.86%; 10 μm, 18.23%; 25 μm, 16.35%). (Fig. 4B). The rates of degeneration of oocytes in all concentration of RS21607 were less than 10% in our experiment (data not shown).
Fig. 4.
Effects of Ketoconazole or RS21607 on FSH-Induced Mouse COCs Meiotic Resumption
COCs were cultured in HX medium containing 50 IU/liter FSH with 10 μm ketoconazole (K) (panel A) or 0, 5, 10, and 25 μm RS21607 (RS) (panel B) for 22–24 h, after which GVBD (%) and PB1 (%) were recorded. Columns with different letters were significantly different (P < 0.05). Each independent experiment was repeated at least three times.
RT-PCR data showed that FSH significantly increased CYP51 mRNA expression after 2 h treatment, arrived at peak at 4 h, which was about 3-fold of the corresponding control group, and decreased after this period (Fig. 5A). A low level of CYP51 gene expression was observed in the control group throughout the process of culture. Concordant with mRNA data, cumulus cell lysates prepared from FSH treatment showed an increase in the cellular CYP51 protein level. As shown in Fig. 5B, FSH increased CYP51 protein level after 4 h of incubation, arrived at maximal level at 6 h, and kept a relative high level to the end of culture. Similar results were obtained when COCs were pulsed with 250 μm N6-cAMP and 8-Br-cAMP: CYP51 gene level increased after 2 h and then rose to maximal level at 4 h, which is about 3-fold of corresponding control group (Fig. 5C). A notable enhancement of CYP51 protein expression occurred at 6–8 h (Fig. 5D).
Fig. 5.
Time-Dependent Changes of CYP51 mRNA and Protein Level within Cumulus Cells of COCs in Response to FSH or N6-cAMP (N6) + 8-Br-cAMP (8-Br)
COCs were treated with 50 IU/liter FSH or pulsed with 250 μm N6-cAMP and 8-Br-cAMP in HX medium. COCs were cultured in HX medium as a control. Sample of cumulus cells at various time points were collected for RT-PCR and immunoblotting. CYP51 mRNA and protein levels induced by FSH (panels A and B) or N6-cAMP plus 8-Br-cAMP (panels C and D) during 0–12 h were analyzed. The densitometric data of CYP51 were normalized according to internal standards (β-actin and GAPDH) to evaluate the relative abundance of the targets. *, P < 0.05, compared with each corresponding group in the control. These results are representative of three experiments.
Phosphorylation of CREB in Cumulus Cells Involved in FSH- and PKAII Analog Pairs-Induced AR and CYP51 Expression
KG-501 attenuated CYP51 mRNA and protein expression within cumulus cells at corresponding high level expression time points in FSH and N6-cAMP plus 8-Br-cAMP groups (Fig. 6, A–D). As shown in Fig. 6, E and F, AR mRNA expression increased at 4 h after FSH or N6-cAMP plus 8-Br-cAMP stimulation, which coincided with Shimada’s report (45). However, 25 μm KG-501 significantly inhibited AR mRNA expression. These results indicate that AR and CYP51 are up-regulated via a CREB/CRE-dependent mechanism during meiotic resumption triggered by FSH and PKAII analog pairs.
Fig. 6.
The Effect of KG-501 on CYP51 and AR Expression Induced by FSH or N6-cAMP + 8-Br-cAMP
COCs were treated with 25 μm KG-501 in HX medium containing 50 IU/liter FSH or pulsing with 250 μm N6-cAMP (N6) plus 8-Br-cAMP (8-Br). Samples of cumulus cells were collected at 3 and 4 h for RT-PCR, and at 6 h and 8 h for Western blot analysis of CYP51 expression, when 25 μm KG-501 was added to FSH group (panels A and B, respectively) or N6-cAMP plus 8-Br-cAMP group (panels C and D, respectively). Cumulus cells at 4 h with above treatment were collected for analyzing AR mRNA expression under the stimulation of FSH (panel E) or N6-cAMP plus 8-Br-cAMP (panel F). Cumulus cells of COCs at 0 h were used for control. The experiment was performed three times with similar results. KG, KG-501.
EGF Could Rescue MAPK Phosphorylation and Oocyte Meiotic Resumption Suppressed by KG-501 But Not by RS21607
As shown in Fig. 7A, 10 ng/ml EGF could reverse the inhibitory effect of 25 μm KG-501 on GVBD induced by FSH (from 24.20% to 86.16%) or N6-cAMP plus 8-Br-cAMP (from 25.13% to 77.20%) but did not have any statistical significant effect on oocyte meiotic resumption blocked by RS21607. Whereas only the low level phosphorylation of MAPK was detected and sustained during procedure of cultivation in control group, FSH or N6-cAMP plus 8-Br-cAMP activated MAPK sequentially in cumulus cells, and reached the maximal phosphorylation level at 8 h (Fig. 7, B and C), which was significantly inhibited by KG-501 and RS21607 (Fig. 7D, lanes 4 and 5; Fig. 7E, lanes 4 and 5). In addition, EGF could reverse the inhibitory effect of KG-501 on MAPK phosphorylation induced by FSH and N6-cAMP plus 8-Br-cAMP (Fig. 7D, lanes 7 and 8) but had no effect on FSH + RS21607 and N6-cAMP plus 8-Br-cAMP + RS21607 groups (Fig. 7E, lanes 7 and 8).
Fig. 7.
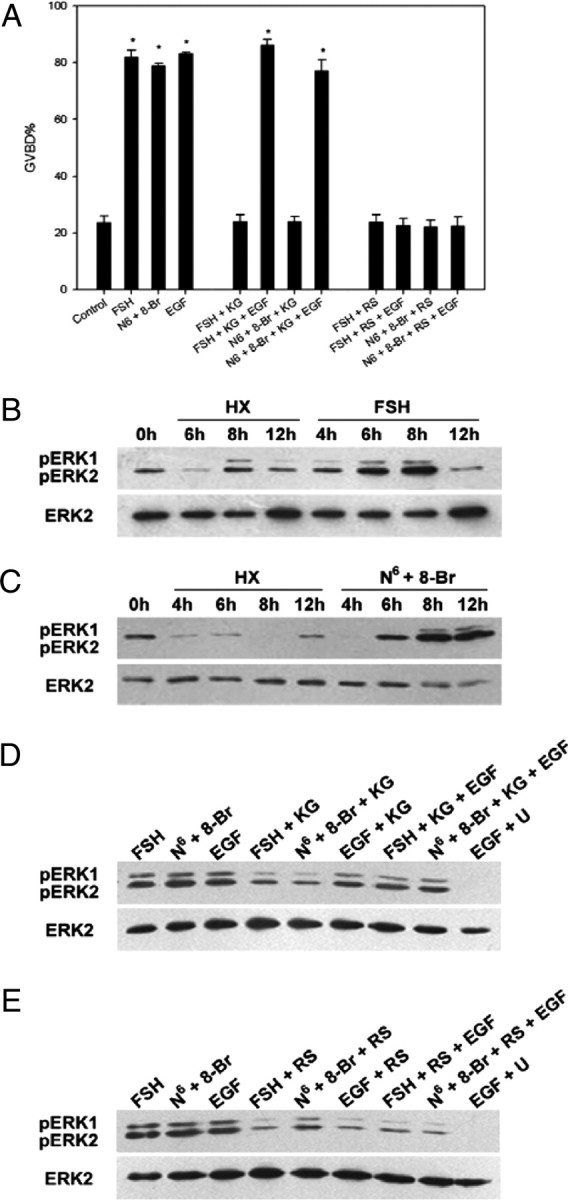
The Effects of PKAII Analog Pairs, EGF, RS21607, or KG-501 on Mouse Oocyte Meiotic Resumption and MAPK Phosphorylation within Cumulus Cells of COCs
COCs were treated with 50 IU/liter FSH, 10 ng/ml EGF, or pulsed with 250 μm N6-cAMP (N6) plus 8-Br-cAMP (8-Br) in HX medium containing 25 μm KG-501(KG) or 25 μm RS21607 (RS). The percentage of GVBD was recorded at 24 h of culture. COCs were cultured in HX medium as a control. Columns with different letters were significantly different (P < 0.01) (panel A). Cumulus cells of COCs were collected at 0, 4, 6, 8, and 12 h of drug treatment for Western blot analysis of MAPK phosphorylation with anti-p-ERK1/2 and anti-ERK2 antibodies: B, HX vs. FSH; C, HX vs. N6-cAMP plus 8-Br-cAMP. Cumulus cell samples were collected at 8 h after different treatment in HX medium for Western blot analysis of MAPK phosphorylation: D, 10 ng/ml EGF was supplemented to assess the potential rescue on MAPK phosphorylation suppressed by KG-501; E, 10 ng/ml EGF was supplemented to assess the potential rescue on MAPK activation inhibited by RS21607. The experiment was performed three times with similar results. pERK, Phosphorylated ERK; U, U0126.
Progesterone Rescued KG-501 and RS21607-Inhibited Oocyte Meiotic Resumption
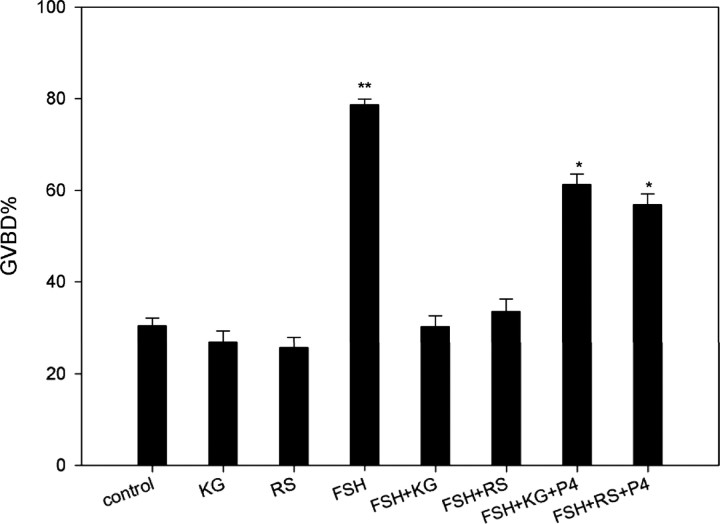
Both 25 μm KG-501 and 25 μm RS21607 had no significant effect on mouse COCs in control group, whereas each drastically inhibited the positive effect of FSH on GVBD (from 78.70% to 33.48% and 30.24%), respectively. The inhibitory effects of KG-501 and RS21607 on meiotic resumption were partially overridden by the addition of 100 ng/ml progesterone (GVBD, 60.25% and 56.88%) (P < 0.05) (Fig. 8).
Fig. 8.
The Effects of Progesterone on KG-01 (KG) or RS21607 (RS)-Inhibited Oocyte Meiotic Resumption
COCs were cultured in HX medium containing 25 μm KG-501 or 25 μm RS21607 with or without 50 IU/liter FSH. Progesterone (P4) (100 ng/ml) was used to rescue inhibition. The percentage of GVBD was recorded at 24 h of culture. COCs were cultured in HX medium as a control. Columns with different letters were significantly different (P < 0.05).
DISCUSSION
Gonadotropin-induced production of sterols and steroid hormones plays an important role in mammalian oocyte maturation (13, 29, 31, 36), and CYP51 is a key enzyme involved in sterol biosynthesis and steroidogenesis. By using in vitro meiosis-induced model of mouse cumulus oocyte complexes (COCs), our studies demonstrate that FSH increased PKA RIIβ level and induced phosphorylation of CREB and CYP51 expression within cumulus cells during oocyte maturation. Moreover, interrupting the formation of CREB functional complex by KG-501 attenuated CYP51 expression and oocyte maturation. RS21607, a specific inhibitor of CYP51, blocked meiosis induced by FSH, which was overridden by progesterone. Furthermore, PKAII analog pairs triggered the similar events, including CREB phosphorylation, AR and CYP51 expression, MAPK activation, and oocyte maturation as FSH did. All these results suggested that FSH induces oocyte meiotic resumption via a PKAII/CREB/CYP51 pathway within cumulus cells.
It was reported that an acute cAMP in response to human chorionic gonadotropin can activate type II PKA in rabbit ovarian follicles (46). In our study, an increase of dissociated PKA RIIβ level were detected in cumulus cells under the stimulation of FSH, which coincided with the timing of a transient cAMP level elevation in response to FSH (16). Generally, cAMP activates PKA by causing the C subunit to physically dissociate from the R subunit. Thus, the increased RIIβ level suggested the activation of type II PKA in FSH-induced oocyte meiotic resumption. A brief treatment with N6-cAMP plus 8-Br-cAMP that increased RIIβ subunit level not only triggered oocyte maturation, which is consistent with the observation of Downs and Hunzicker-Dunn (23), but also performed a sequence of CREB phosphorylation, AR, CYP51 up-expression, and MAPK activation as FSH did in the meiosis process. Taken together, data in this point suggest that FSH induces oocyte maturation by mediating PKAII within cumulus cells of COCs. Although the previous work found that PKAII cAMP analogs inhibit spontaneous maturation of DOs (24), but N6-cAMP plus 8-Br-cAMP did not give an inhibition of GVBD of COCs in the present experiment. It is possibly due to pulsing COCs with PKAII analog pairs, which transiently activates PKAII in cumulus cells and leads to oocyte maturation, whereas constant activation of PKAII maintains arrest of DOs.
As mentioned, dissociation of RC dimers of PKA leads to C subunit activation. In the present experiment, an attenuated PKA C subunit level is detected which seems not to coincide with the increase of PKA RIIβ level in response to FSH. The discordance is likely ascribed to type I and II PKA sharing the same C subunits (18, 19); therefore, C subunit level cannot exactly show the activity either of two types of PKA. Analyzing the present data, it is possible that a majority of PKA C subunit is dissociated from PKAI holoenzyme in the HX group, with respect to lower PKA RIIβ level; in contrast, a significant increase of RIIβ level with a decrease of C subunit level is detected in the FSH group, which implies an increase of PKAII activity concurrent with the activity of PKAI decrease in response to FSH. Namely, PKAI activity is likely down-regulated whereas PKAII activity is up-regulated within the cumulus cells, which mediate meiotic resumption in response to FSH. To confirm this hypothesis, further work on the regulation of PKA RI subunit during gonadotropin-induced oocyte maturation is needed. In any case, PKA RIIβ level within cumulus cells increased in response to FSH, which was evidence for the activation of PKAII in FSH-induced oocyte meiotic resumption.
Mammalian CYP51 encodes lanosterol 14α-demethylase, which is a key enzyme involved in FF-MAS and steroids (e.g. testosterone, progesterone, and estradiol) biosynthesis. In the present experimental model, a dose-dependent inhibitory effect of RS21607 on FSH-induced GVBD and the similar inhibitory effect of ketoconazole were observed. In fact, previous reports about the effect of CYP51 protein inhibitors (e.g. RS21745, RS21607, ketoconazole) on meiotic maturation are inconsistent. Significantly suppressible effects of these inhibitors on FSH-induced GVBD of COCs or FEOs have been reported by the recent research in pig and mouse (27, 36, 47), whereas these inhibitors did not prevent the LH-induced oocyte maturation of FEOs, and did not affect spontaneous maturation of COCs in rat (48, 49). The inconsistent results may be explained by the following aspects. First, ketoconazole is not a specific inhibitor of CYP51 but an inhibitor blocking the activity of nearly all cytochrome P450 enzymes. Furthermore, to rule out the possibility of undefined culture conditions, media, and the variety of animal species (50), the varying results among different research groups may be ascribed to different gonadotropins. It is possible that CYP51/sterol or steroid pathway is necessary for FSH-induced oocyte meiosis but not for LH-induced oocyte maturation. LH and FSH may either directly overlap or utilize redundant pathways in oocyte maturation. However, recent studies support that progesterone is essential for LH-induced oocyte maturation (29, 30, 31). Thus, it is necessary to define in future work whether sterol or steroid pathway is involved in LH-induced oocyte meiotic resumption. Presently, we provided the evidence that CYP51 expression of cumulus cells was significantly up-regulated in response to FSH, which was pre- or perimeiotic resumption; RS21607, a specific CYP51 inhibitor, significantly inhibited FSH-induced oocyte maturation. These results suggest that CYP51 takes part in FSH-induced mouse meiotic resumption. Notably, steroid production, especially progesterone, regulated by EGFR signaling, is necessary for LH-induced oocyte maturation (31). In our study, EGF failed to rescue oocyte meiotic resumption suppressed by RS21607, which implies a regulation of CYP51 activity involved in EGFR signaling. Also, compatible with the earlier study that progesterone could rescue the suppression of GVBD by ketoconazole (30), progesterone here partially overrode RS21607-inhibited oocyte meiotic resumption. Thereby, CYP51 in cumulus cells perhaps exerts its role by facilitating steroidogenesis to influence oocyte maturation in response to gonadotropin.
As a well-known transcription factor employed by cAMP, CREB is generally activated by phosphorylation at serine residue 133 and is believed to induce gene expression by binding to a conserved CRE sequence in the promoter (51). In our study, a transient phosphorylation of CREB could be observed under the stimulation of FSH and PKAII analog pairs, and interrupting the formation of CREB functional complex by KG-501 could inhibit oocyte maturation in a dose-dependent manner; these suggested a CREB-regulated gene transcription involved in FSH/PKAII signal cascades. Meanwhile, numerous genes were regulated by CREB in response to hormone; we demonstrate here that CYP51 and AR are two of the target genes in the process of FSH-induced oocyte maturation, with the results that KG-501 effectively down-regulated the up-expression of CYP51 and AR mRNA induced by FSH. Moreover, EGF successfully reversed the inhibitory effect of KG-501 on GVBD induced by FSH, which supports the finding that gene transcription is required in FSH-induced but not EGF-induced oocyte maturation, as Downs et al. (52) and Farin et al. (53) reported.
Accumulating evidence suggests that the MAPK cascade in cumulus cells is necessary for gonadotropin-induced meiotic resumption (13, 14, 54). However, the regulation of MAPK in cumulus cells is not so clear. In the present study, activation of MAPK occurring downstream of PKAII/CREB/CYP51 within cumulus cells is implicated by the following observations. First, FSH and PKAII analog pairs stimulated phosphorylation of MAPK in cumulus cells. Second, KG-501 or RS21607 blocked both phosphorylation of MAPK and oocyte maturation induced by FSH or PKAII analog pairs. Moreover, EGF appears to be necessary for gonadotropin-induced meiotic resumption, and p42/44MAPK in cumulus cells has been proved to be its downstream signal response (14, 45, 55, 56). EGFR signaling modulates MAPK phosphorylation, and oocyte maturation possibly requires a CYP51 activity, because EGF could rescue the activation of MAPK and oocyte meiotic resumption suppressed by CREB-transcriptional inhibitor but not by the specific inhibitor of CYP51. It has been reported by Norman et al. (57) that steroid hormone interacts with plasma membrane steroid receptors and then activates MAPK cascade to initiate biological responses. Recent studies found that MAPK activity is required for growth differentiation factor 9 to promote cumulus expansion (58). Another finding showed that MAPK mediates LH-induced FEO maturation by interrupting cell-to-cell communication through phosphorylation of connexin43 (54). It will be interesting to investigate the relationship between these events downstream of MAPK and the steroids signal pathway.
Although it is still controversial whether CYP51 mediates LH-induced oocyte maturation, similar events occur between LH- and FSH-induced oocyte meiotic resumption, with respect to Gs-cAMP signaling and MAPK activation. In addition, several studies have demonstrated that both of the two gonadotropins use EGFR signaling as a potential central pathway in murine oocyte maturation (32, 33, 34). These findings indicate that the mechanism of FSH- and LH-induced oocyte maturation may share some similarities. In the present study, a FSH-induced meiosis model in vitro was employed, which is a useful experimental model for examining the potential signal pathways in gonadotropin-induced oocyte maturation and is commonly adopted to identify the role of cumulus cells and the oocyte in the control of the resumption of meiosis (13, 14, 34, 59, 60). Our results indicate a possible signal pathway in FSH-induced oocyte maturation that FSH uses PKAII as a downstream mediator, which leads to phosphorylation of CREB, AR, CYP51 expression, and MAPK activation within cumulus cells. A schematic diagram of these pathways and their interaction is shown in Fig. 9. Our work is expected to provide some basic information for clarifying the physiological mechanism of gonadotropin-induced oocyte maturation and improving in vitro oocyte maturation systems for animal and human clinical applications.
Fig. 9.
A Possible Signaling Pathway that Mediates FSH-Induced Meiotic Resumption of Mouse Oocytes
After binding with its membrane-integrated receptor in cumulus cells, FSH uses PKAII as a downstream mediator after a transient increase of cAMP. PKAII leads to AR and CYP51 expression with the mediation of CREB. CYP51 probably participates in the regulation of MAPK activation by mediating downstream steroidogenesis during oocyte maturation. In addition, CYP51 activity may be involved in EGF-induced MAPK phosphorylation and oocyte meiotic resumption. cat, Catalytic unit; CBP, CREB-binding protein; FSHR, FSH receptor.
MATERIALS AND METHODS
Experimental Animals
Immature 21- to 23-d-old Kunming White female mice (outbreed strain) were used for all experiments. All animal treatment procedures were approved by the Animal Care Committee of China Agricultural University. Mice were housed under controlled temperature (24 ± 1 C) and lighting (12 h light/12 h darkness) with food and water ad libitum. Follicle development was primed by ip injection of each mouse with 5 IU equine chorionic gonadotropin and mice were killed by cervical dislocation 44–48 h later.
Experimental Design
Experiment 1: the Effect of FSH on PKA RIIβ and C subunits Levels.
To investigate whether PKAII signal cascades are activated by FSH, we detected the R and C subunit levels of PKAII within cumulus cells during FSH-induced oocyte meiotic maturation. Mouse COCs were exposed to HX medium containing 50 IU/liter FSH. Samples of cumulus cells were collected at various time points for Western blot analysis of PKA RIIβ and C subunits.
Experiment 2: the Effect of PKAII Analog Pairs on PKA RIIβ and C Subunit Levels and Oocyte Meiotic Maturation.
COCs were pulsed with 250 μm N6-cAMP and increasing concentration of 8-Br-cAMP (0–500 μm) in HX medium. GVBD of oocytes was investigated at 24 h of culture, and cumulus cells were collected at 3 h of incubation for Western blot analysis of PKA RIIβ and C subunits.
Experiment 3: the Role of CREB Phosphorylation in Cumulus Cells during FSH- and PKAII Analog Pairs-Induced Oocyte Maturation.
Mouse COCs were cultured in HX medium containing 50 IU/liter FSH. Cumulus cells were collected at various time points for Western blot analysis of CREB phosphorylation. Further, KG-501 (2-naphthol-AS-Ephosphate), which blocks cAMP induction of CREB-dependent target gene transcription through interference in the binding between the kinase-inducible domain (KID) domain of CREB and KIX domain of CREB-binding protein (43), was used to identify whether a CRE/CREB-dependent signal mechanism mediates oocyte meiotic progression. Mouse COCs were cultured in HX medium containing 50 IU/liter FSH plus increasing concentration of KG-501. GVBD in oocytes were investigated at 24 h of culture. Likewise, PKAII analog pairs (N6-cAMP plus 8-Br-cAMP) were used to mimic the action of FSH in the experiments as mentioned above.
Experiment 4: the Role and Kinetics of CYP51 in FSH-Induced Oocyte Meiotic Resumption.
COCs were cultured for 24 h in HX medium containing 50 IU/liter FSH with 10 μm ketoconazole (a steroidogenetic enzymes inhibitor which also inhibits CYP51) or different concentration of RS21607 (a specific inhibitor of CYP51). Finally the nuclear status of oocytes was determined. In addition, COCs were treated with or without 50 IU/liter FSH in HX medium. Samples of cumulus cells of COCs were collected at various time points for RT-PCR and Western blot analysis of CYP51 expression. Likewise, PKAII analog pairs (N6-cAMP plus 8-Br-cAMP) were used to mimic the action of FSH in the experiments as mentioned above.
Experiment 5: the Effect of KG-501 on FSH- and PKAII Analog Pairs-Induced EGF-Like Factor AR and CYP51 Expression.
COCs were cultured in HX medium, containing 50 IU/liter FSH, N6-cAMP plus 8-Br-cAMP, and/or 25 μm KG-501. Samples of cumulus cells were collected for RT-PCR and Western blot analysis of AR and CYP51 expression.
Experiment 6: the Effects of RS21607 or KG-501 on MAPK Phosphorylation in Cumulus Cells.
COCs were cultured in HX medium, containing 50 IU/liter FSH, 250 μm N6-cAMP plus 8-Br-cAMP, 25 μm KG-501, 25 μm RS21607, and/or 10 ng/ml EGF. GVBD of oocytes was observed at 24 h of culture, and cumulus cells were collected at 0, 4, 6, 8, and 12 h for analyzing the kinetics of MAPK phosphorylation. EGF (10 ng/ml) plus 10 μm U0126 (a specific inhibitor of MAPK phosphorylation) was used for a negative control. In addition, 100 ng/ml progesterone was used to rescued the inhibition of KG-501 or RS21607 to examine the effect of steroid downstream of CYP51 on oocyte maturation.
Chemicals
All reagents and chemicals used in this study were obtained from Sigma-Aldrich Corp (St. Louis, MO), unless otherwise indicated. FSH and EGF were prepared as stock solutions in distilled PBS containing 0.1% BSA, and the final concentrations for culture were 0.05 IU/ml and 10 ng/ml, respectively. RS21607 (a gift of Dr. D. C. Swinney, Roche Bioscience, Indianapolis, IN) and progesterone was dissolved in ethanol to 10 mm and 100 μg/ml, respectively. Ketoconazole was dissolved in 0.5 n HCl, neutralized with 0.5 n NaOH, and diluted with saline to 10 mm. U0126, KG-501 (2-naphthol-AS-Ephosphate), N6-monobutyryl-cAMP (N6-cAMP), and 8-Br-cAMP were prepared in dimethylsulfoxide to 50 mm, and the final concentration of dimethylsulfoxide was less than 0.1%, which had no significant effect on oocyte maturation (data not shown). This stock solution was further diluted for culture with the medium. The test concentrations of these reagents were according to the previous reports (14, 23, 27, 47). Rabbit polyclonal antihuman CYP51 antibody was a gift of Dr. M. R. Waterman (Vanderbilt University, Memphis, TN). Monoclonal mouse anti-MAPK-activated antibody (phosphorylated ERK1 and 2), monoclonal mouse anti-phos CREB (Ser-133) antibody, and polyclonal rabbit anti-CREB antibody were purchased from Cell Signaling Technology (Beverly, MA). Polyclonal rabbit anti-ERK2 antibody, polyclonal goat anti-PKA RIIβ antibody, polyclonal rabbit anti-PKA Cα antibody, monoclonal mouse glyceraldehyde-3-phosphate dehydrogenase (GAPDH) antibody, and relative horseradish peroxidase-conjugated second antibodies were purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA).
Oocyte Isolation and Culture
Cumulus-enclosed and GV intact oocytes were isolated under a stereomicroscope by puncturing the follicles (300–400 μm) with a 27-gauge needle in culture medium. COCs of equal size with several layers of cumulus cells were collected. The culture medium used for this study was M199 (GIBCO-Invitrogen, Carlsbad, CA) medium containing 4 mm HX; 0.23 mm sodium pyruvate; 2 mm glutamine; 3 mg/ml lyophilized crystallized BSA; 75 mg/ml potassium penicillin G; and 50 mg/ml streptomycin sulfate. This medium is designated as HX medium. In each experiment, 50–60 COCs were cultured in a 100-μl drop covered with paraffin oil in a 35-mm culture dish. Oocytes were cultured at 37 C, in an atmosphere of 5% CO2 and 100% humidity. After culture, oocytes were denuded mechanically and assessed for maturation scoring for GV (meiotic arrest), GVBD (meiotic resumption), and the PB1 (the first meiotic maturation). The percent of GVBD (including PB1) per total number of oocytes (% GVBD) and the percent of PB1 per total number of oocytes (% PB1) were calculated. Oocytes that had degenerated were not included.
Site Selectivity of the cAMP Analogs Used in This Study
The site-selective cAMP analogs, 8-Br-cAMP and N6-cAMP, were used to selectively activate PKAII, because the former preferentially binds to site B on RII, especially in mouse COCs (23, 61), and the latter displays a higher affinity for site A on either PKAI or PKAII than for site B (62), the combination of which (termed PKAII analog pairs) preferentially activates PKAII. Considering that PKAII is preferentially activated by a higher cAMP level (63, 64), also for mimicking the remarkable surge of cAMP triggered by FSH (16), oocytes were exposed to PKAII analog pairs (N6-cAMP plus 8-Br-cAMP) for 3 h, washed, and then transferred into a fresh, analog pairs-free HX medium for additional culture if needed. The same operation when using this analog pair to activate PKAII in the experiments.
RNA Isolation and Semiquantitative RT-PCR
Total RNA was extracted from 200 COCs using the TRIzol Reagent (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions and quantified by measuring the absorbance at 260 nm. Reverse transcription was performed according to the protocol described by RT-PCR system (Promega Corp., Madison, WI) from total RNA at 42 C for 1 h. The reverse transcriptase products were then amplified for 25 (β-actin) or 24 (CYP51) cycles, including denaturation at 95 C for 30 sec, annealing at 54 C for 30 sec, and extension at 72 C for 45 sec, finally terminated with an extension of 10 min at 72 C. The PCR products were electrophoresed on 2% agarose gel in Tris-acetate-EDTA (TAE) buffer for analysis. The density of PCR products was measured by AlphaEaseFC Software (Alpha Innotech Corp., San Leandro, CA).
Oligonucleotide primers used for amplification of CYP51, AR, and β-actin were designed according to the mouse sequence from GenBank (CYP51, 9910171; AR, 31981895; β-actin, 6671508) (Table 1). β-Actin was used as a control for reaction efficiency and variations in concentrations of mRNA in the original reverse transcriptase reaction.
Table 1.
Primers Used for Determinations of Mouse CYP51, AR, and β-Actin mRNA by RT-PCR
| mRNA | Primer Sequence | Predicted Product Size (bp) | Amplification Cycle |
|---|---|---|---|
| CYP51 | Sense: 5′-GGGAGAAAGCGGAGAAAGA-3′ | 405 | 24 |
| Antisense: 3′-GTAGGAGGTGGTGGTCACG-5′ | |||
| AR | Sense: 5′-TTCATGGCGAATGCAATGCA-3′ | 402 | 25 |
| Antisense: 3′-ATCCGAAAGCTCCACTTCCT-5′ | |||
| β-Actin | Sense: 5′-TCCAGCCTTCCTTCTTGGGTAT-3′ | 557 | 25 |
| Antisense: 3′-TTTGACCTTGCCACTTCCGC-5′ |
Electrophoresis and Western Blot Analysis
Western blot analysis was carried out with standard techniques as described in previous reports (14, 65). Proteins from cumulus cells of 50 COCs per sample were extracted with double-strength electrophoresis sample buffer after culture, supplemented with 1 mm phenylmethylsulfonylfluoride and 1 mm sodium orthovanadate for 20 min on ice, and stored at −70 C. Before electrophoresis, the lysates were heated to 100 C for 5 min and cooled down on ice immediately and then centrifuged at 12,000 × g for 5 min. The proteins were separated by SDS-PAGE with a 4% stacking gel and a 10% separating gel for 50 min at 160 V, and electrically transferred to a nitrocellulose membrane (Amersham Pharmacia Biotech, Braunschweig, Germany). The membrane was saturated with 5% nonfat dry milk and then incubated with corresponding antibody. Proteins were detected using SuperSignal West Pico (enhanced chemiluminescence) detection system (Pierce Chemical Co., Rockford, IL).
For reprobing total MAPK and CREB, the blots were stripped of the bound antibodies by washing in a stripping buffer (100 mm β-mercaptoethanol; 20% sodium dodecyl sulfate; 62.5 mm Tris, pH 6.7) and then reprobed with anti-ERK2 antibody or anti-CREB antibody. The density of blot was measured by AlphaEaseFC Software if needed.
Statistical Analysis
All experiments were replicated at least three times and the values were given as mean ± sem. Statistical analyses of all data were done by t test or ANOVA followed by Duncan’s multiple range test (SigmaStat; Systat Software, Inc., Richmond, CA). All percentage data were subjected to arc-sine transformation before statistical analysis. P < 0.05 was considered to indicate a significant difference.
Acknowledgments
We thank Dr. D. C. Swinney for kindly providing RS21607 and Dr. M. R. Waterman for generously supplying CYP51 antibody.
Footnotes
This work was supported by grants of Chinese 973 Project (Nos. 2006CB504003 and 2007CB947401) and Chinese Natural Science Foundation (Nos. 30470868, 30571358, and 30671508).
Disclosure Statement: The authors have nothing to declare.
First Published Online May 8, 2008
G.N. and H.O. contributed equally to this work and should be considered as co-first authors.
Abbreviations: AR, Amphiregulin; 8-Br-cAMP, 8-bromo-cAMP; C subunit, catalytic subunit; COCs, cumulus oocyte complexes; CRE, cAMP response element; CREB, cAMP response element-binding protein; CYP51, cytochrome P450 lanosterol 14α-demethylase; DOs, denuded oocytes; EGF, epidermal growth factor; EGFR, EGF receptor; FEOs, follicle-enclosed oocytes; FF-MAS, follicular fluid meiosis-activating sterols; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; GV, germinal vesicle; GVBD, germinal vesicle breakdown; HX, hypoxanthine; KG-501, 2-naphthol-AS-Ephosphate; N6-cAMP, N6-monobutyryl-cAMP; PB1, first polar body; PKA, cAMP-dependent protein kinase; PKAI, type I PKA; PKAII, type II PKA; R subunit, regulatory subunit.
References
- 1.Cho WK, Stern S, Biggers JD 1974. Inhibitory effect of dibutyryl cAMP on mouse oocyte maturation in vitro. J Exp Zool 187:383–386 [DOI] [PubMed] [Google Scholar]
- 2.Dekel N, Beers WH 1978. Rat oocyte maturation in vitro: relief of cyclic AMP inhibition by gonadotropins. Proc Natl Acad Sci USA 75:4369–4373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Eppig JJ, Downs SM 1987. The effect of hypoxanthine on mouse oocyte growth and development in vitro: maintenance of meiotic arrest and gonadotropin-induced oocyte maturation. Dev Biol 119:313–321 [DOI] [PubMed] [Google Scholar]
- 4.Downs SM 1993. Purine control of mouse oocyte maturation: evidence that nonmetabolized hypoxanthine maintains meiotic arrest. Mol Reprod Dev 35:82–94 [DOI] [PubMed] [Google Scholar]
- 5.Lindner HR, Tsafriri A, Lieberman ME, Zor U, Koch Y, Bauminger S, Barnea A 1974. Gonadotropin action on cultured Graafian follicles: induction of maturation division of the mammalian oocyte and differentiation of the luteal cell. Recent Prog Horm Res 30:79–138 [DOI] [PubMed] [Google Scholar]
- 6.Edwards RG 1965. Maturation in vitro of mouse, sheep, cow, pig, rhesus monkey and human ovarian oocytes. Nature 208:349–351 [DOI] [PubMed] [Google Scholar]
- 7.Downs SM, Daniel SA, Eppig JJ 1988. Induction of maturation in cumulus cell-enclosed mouse oocytes by follicle-stimulating hormone and epidermal growth factor: evidence for a positive stimulus of somatic cell origin. J Exp Zool 245:86–96 [DOI] [PubMed] [Google Scholar]
- 8.Eppig JJ 1991. Maintenance of meiotic arrest and the induction of oocyte maturation in mouse oocyte-granulosa cell complexes developed in vitro from preantral follicles. Biol Reprod 45:824–830 [DOI] [PubMed] [Google Scholar]
- 9.Eppig JJ, Wigglesworth K, Pendola F, Hirao Y 1997. Murine oocytes suppress expression of luteinizing hormone receptor messenger ribonucleic acid by granulosa cells. Biol Reprod 56:976–984 [DOI] [PubMed] [Google Scholar]
- 10.Diaz FJ, Wigglesworth K, Eppig JJ 2007. Oocytes determine cumulus cell lineage in mouse ovarian follicles. J Cell Sci 120:1330–1340 [DOI] [PubMed] [Google Scholar]
- 11.Fu M, Chen X, Yan J, Lei L, Jin S, Yang J, Song X, Zhang M, Xia G 2007. Luteinizing hormone receptors expression in cumulus cells closely related to mouse oocyte meiotic maturation. Front Biosci 12:1804–1813 [DOI] [PubMed] [Google Scholar]
- 12.Zhang M, Xia G, Zhou B, Wang C 2007. Gonadotropin-controlled mammal oocyte meiotic resumption. Front Biosci 12:282–296 [DOI] [PubMed] [Google Scholar]
- 13.Byskov AG, Yding Andersen C, Hossaini A, Guoliang X 1997. Cumulus cells of oocyte-cumulus complexes secrete a meiosis-activating substance when stimulated with FSH. Mol Reprod Dev 46:296–305 [DOI] [PubMed] [Google Scholar]
- 14.Su YQ, Wigglesworth K, Pendola FL, O'Brien MJ, Eppig JJ 2002. Mitogen-activated protein kinase activity in cumulus cells is essential for gonadotropin-induced oocyte meiotic resumption and cumulus expansion in the mouse. Endocrinology 143:2221–2232 [DOI] [PubMed] [Google Scholar]
- 15.Dekel N 1988. Regulation of oocyte maturation. The role of cAMP. Ann NY Acad Sci 541:211–216 [DOI] [PubMed] [Google Scholar]
- 16.Salustri A, Petrungaro S, De Felici M, Conti M, Siracusa G 1985. Effect of follicle-stimulating hormone on cyclic adenosine monophosphate level and on meiotic maturation in mouse cumulus cell-enclosed oocytes cultured in vitro. Biol Reprod 33:797–802 [DOI] [PubMed] [Google Scholar]
- 17.Hubbard CJ 1985. The effects of forskolin and LH on cAMP changes and maturation in the follicle-enclosed oocytes of hamsters. Acta Endocrinol (Copenh) 110:413–420 [DOI] [PubMed] [Google Scholar]
- 18.Brandon EP, Idzerda RL, McKnight GS 1997. PKA isoforms, neural pathways, and behaviour: making the connection. Curr Opin Neurobiol 7:397–403 [DOI] [PubMed] [Google Scholar]
- 19.Skalhegg BS, Tasken K 2000. Specificity in the cAMP/PKA signaling pathway. Differential expression, regulation, and subcellular localization of subunits of PKA. Front Biosci 5:D678–D693 [DOI] [PubMed]
- 20.Vigil D, Blumenthal DK, Brown S, Taylor SS, Trewhella J 2004. Differential effects of substrate on type I and type II PKA holoenzyme dissociation. Biochemistry 43:5629–5636 [DOI] [PubMed] [Google Scholar]
- 21.Viste K, Kopperud RK, Christensen AE, Doskeland SO 2005. Substrate enhances the sensitivity of type I protein kinase A to cAMP. J Biol Chem 280:13279–13284 [DOI] [PubMed] [Google Scholar]
- 22.Brown RL, Ord T, Moss SB, Williams CJ 2002. A-kinase anchor proteins as potential regulators of protein kinase A function in oocytes. Biol Reprod 67:981–987 [DOI] [PubMed] [Google Scholar]
- 23.Downs SM, Hunzicker-Dunn M 1995. Differential regulation of oocyte maturation and cumulus expansion in the mouse oocyte-cumulus cell complex by site-selective analogs of cyclic adenosine monophosphate. Dev Biol 172:72–85 [DOI] [PubMed] [Google Scholar]
- 24.Newhall KJ, Criniti AR, Cheah CS, Smith KC, Kafer KE, Burkart AD, McKnight GS 2006. Dynamic anchoring of PKA is essential during oocyte maturation. Curr Biol 16:321–327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Reynhout JK, Taddei C, Smith LD, LaMarca MJ 1975. Response of large oocytes of Xenopus laevis to progesterone in vitro in relation to oocyte size and time after previous HCG-induced ovulation. Dev Biol 44:375–379 [DOI] [PubMed] [Google Scholar]
- 26.Schmitt A, Nebreda AR 2002. Signalling pathways in oocyte meiotic maturation. J Cell Sci 115:2457–2459 [DOI] [PubMed] [Google Scholar]
- 27.Xie H, Xia G, Byskov AG, Andersen CY, Bo S, Tao Y 2004. Roles of gonadotropins and meiosis-activating sterols in meiotic resumption of cultured follicle-enclosed mouse oocytes. Mol Cell Endocrinol 218:155–163 [DOI] [PubMed] [Google Scholar]
- 28.Gill A, Jamnongjit M, Hammes SR 2004. Androgens promote maturation and signaling in mouse oocytes independent of transcription: a release of inhibition model for mammalian oocyte meiosis. Mol Endocrinol 18:97–104 [DOI] [PubMed] [Google Scholar]
- 29.Yamashita Y, Nishibori M, Terada T, Isobe N, Shimada M 2005. Gonadotropin-induced δ14-reductase and δ7-reductase gene expression in cumulus cells during meiotic resumption of porcine oocytes. Endocrinology 146:186–194 [DOI] [PubMed] [Google Scholar]
- 30.Yamashita Y, Shimada M, Okazaki T, Maeda T, Terada T 2003. Production of progesterone from de novo-synthesized cholesterol in cumulus cells and its physiological role during meiotic resumption of porcine oocytes. Biol Reprod 68:1193–1198 [DOI] [PubMed] [Google Scholar]
- 31.Jamnongjit M, Gill A, Hammes SR 2005. Epidermal growth factor receptor signaling is required for normal ovarian steroidogenesis and oocyte maturation. Proc Natl Acad Sci USA 102:16257–16262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Park JY, Su YQ, Ariga M, Law E, Jin SL, Conti M 2004. EGF-like growth factors as mediators of LH action in the ovulatory follicle. Science 303:682–684 [DOI] [PubMed] [Google Scholar]
- 33.Conti M, Hsieh M, Park JY, Su YQ 2006. Role of the epidermal growth factor network in ovarian follicles. Mol Endocrinol 20:715–723 [DOI] [PubMed] [Google Scholar]
- 34.Downs SM, Chen J 2007. EGF-like peptides mediate FSH-induced maturation of cumulus cell-enclosed mouse oocytes. Mol Reprod Dev 75:105–114 [DOI] [PubMed] [Google Scholar]
- 35.Rozman D 2000. Lanosterol 14α-demethylase (CYP51)—a cholesterol biosynthetic enzyme involved in production of meiosis activating sterols in oocytes and testis—a minireview. Pflugers Arch 439:R56–R57 [PubMed]
- 36.Jin S, Zhang M, Lei L, Wang C, Fu M, Ning G, Xia G 2006. Meiosis activating sterol (MAS) regulate FSH-induced meiotic resumption of cumulus cell-enclosed porcine oocytes via PKC pathway. Mol Cell Endocrinol 249:64–70 [DOI] [PubMed] [Google Scholar]
- 37.Debeljak N, Fink M, Rozman D 2003. Many facets of mammalian lanosterol 14α-demethylase from the evolutionarily conserved cytochrome P450 family CYP51. Arch Biochem Biophys 409:159–171 [DOI] [PubMed] [Google Scholar]
- 38.Halder SK, Fink M, Waterman MR, Rozman D 2002. A cAMP-responsive element binding site is essential for sterol regulation of the human lanosterol 14α-demethylase gene (CYP51). Mol Endocrinol 16:1853–1863 [DOI] [PubMed] [Google Scholar]
- 39.Rozman D, Fink M, Fimia GM, Sassone-Corsi P, Waterman MR 1999. Cyclic adenosine 3′,5′-monophosphate(cAMP)/cAMP-responsive element modulator (CREM)-dependent regulation of cholesterogenic lanosterol 14α-demethylase (CYP51) in spermatids. Mol Endocrinol 13:1951–1962 [DOI] [PubMed] [Google Scholar]
- 40.Plowman GD, Green JM, McDonald VL, Neubauer MG, Disteche CM, Todaro GJ, Shoyab M 1990. The amphiregulin gene encodes a novel epidermal growth factor-related protein with tumor-inhibitory activity. Mol Cell Biol 10:1969–1981 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.O'Reilly SM, Leonard MO, Kieran N, Comerford KM, Cummins E, Pouliot M, Lee SB, Taylor CT 2006. Hypoxia induces epithelial amphiregulin gene expression in a CREB-dependent manner. Am J Physiol Cell Physiol 290:C592–C600 [DOI] [PubMed]
- 42.Shao J, Lee SB, Guo H, Evers BM, Sheng H 2003. Prostaglandin E2 stimulates the growth of colon cancer cells via induction of amphiregulin. Cancer Res 63:5218–5223 [PubMed] [Google Scholar]
- 43.Best JL, Amezcua CA, Mayr B, Flechner L, Murawsky CM, Emerson B, Zor T, Gardner KH, Montminy M 2004. Identification of small-molecule antagonists that inhibit an activator: coactivator interaction. Proc Natl Acad Sci USA 101:17622–17627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Burton PM, Swinney DC, Heller R, Dunlap B, Chiou M, Malonzo E, Haller J, Walker KA, Salari A, Murakami S 1995. Azalanstat (RS-21607), a lanosterol 14 α-demethylase inhibitor with cholesterol-lowering activity. Biochem Pharmacol 50:529–544 [DOI] [PubMed] [Google Scholar]
- 45.Shimada M, Hernandez-Gonzalez I, Gonzalez-Robayna I, Richards JS 2006. Paracrine and autocrine regulation of epidermal growth factor-like factors in cumulus oocyte complexes and granulosa cells: key roles for prostaglandin synthase 2 and progesterone receptor. Mol Endocrinol 20:1352–1365 [DOI] [PubMed] [Google Scholar]
- 46.Hunzicker-Dunn M 1981. Selective activation of rabbit ovarian protein kinase isozymes in rabbit ovarian follicles and corpora lutea. J Biol Chem 256:12185–12193 [PubMed] [Google Scholar]
- 47.Lu Z, Xia G, Byskov AG, Andersen CY 2000. Effects of amphotericin B and ketoconazole on mouse oocyte maturation: implications on the role of meiosis-activating sterol. Mol Cell Endocrinol 164:191–196 [DOI] [PubMed] [Google Scholar]
- 48.Vaknin KM, Lazar S, Popliker M, Tsafriri A 2001. Role of meiosis-activating sterols in rat oocyte maturation: effects of specific inhibitors and changes in the expression of lanosterol 14α-demethylase during the preovulatory period. Biol Reprod 64:299–309 [DOI] [PubMed] [Google Scholar]
- 49.Tsafriri A, Popliker M, Nahum R, Beyth Y 1998. Effects of ketoconazole on ovulatory changes in the rat: implications on the role of a meiosis-activating sterol. Mol Hum Reprod 4:483–489 [DOI] [PubMed] [Google Scholar]
- 50.Downs SM, Ruan B, Schroepfer Jr GJ 2001. Meiosis-activating sterol and the maturation of isolated mouse oocytes. Biol Reprod 64:80–89 [DOI] [PubMed] [Google Scholar]
- 51.Johannessen M, Delghandi MP, Moens U 2004. What turns CREB on? Cell Signal 16:1211–1227 [DOI] [PubMed] [Google Scholar]
- 52.Downs SM, Humpherson PG, Martin KL, Leese HJ 1996. Glucose utilization during gonadotropin-induced meiotic maturation in cumulus cell-enclosed mouse oocytes. Mol Reprod Dev 44:121–131 [DOI] [PubMed] [Google Scholar]
- 53.Farin CE, Rodriguez KF, Alexander JE, Hockney JE, Herrick JR, Kennedy-Stoskopf S 2007. The role of transcription in EGF- and FSH-mediated oocyte maturation in vitro. Anim Reprod Sci 98:97–112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sela-Abramovich S, Chorev E, Galiani D, Dekel N 2005. Mitogen-activated protein kinase mediates luteinizing hormone-induced breakdown of communication and oocyte maturation in rat ovarian follicles. Endocrinology 146:1236–1244 [DOI] [PubMed] [Google Scholar]
- 55.Gall L, Boulesteix C, Ruffini S, Germain G 2005. EGF-induced EGF-receptor and MAP kinase phosphorylation in goat cumulus cells during in vitro maturation. Mol Reprod Dev 71:489–494 [DOI] [PubMed] [Google Scholar]
- 56.Li M, Liang CG, Xiong B, Xu BZ, Lin SL, Hou Y, Chen DY, Schatten H, Sun QY 2008. PI3-kinase and mitogen-activated protein kinase in cumulus cells mediate EGF-induced meiotic resumption of porcine oocyte. Domest Anim Endocrinol 34:360–371 [DOI] [PubMed] [Google Scholar]
- 57.Norman AW, Mizwicki MT, Norman DP 2004. Steroid-hormone rapid actions, membrane receptors and a conformational ensemble model. Nat Rev Drug Discov 3:27–41 [DOI] [PubMed] [Google Scholar]
- 58.Su YQ, Wu X, O'Brien MJ, Pendola FL, Denegre JN, Matzuk MM, Eppig JJ 2004. Synergistic roles of BMP15 and GDF9 in the development and function of the oocyte-cumulus cell complex in mice: genetic evidence for an oocyte-granulosa cell regulatory loop. Dev Biol 276:64–73 [DOI] [PubMed] [Google Scholar]
- 59.Coticchio G, Rossi G, Borini A, Grondahl C, Macchiarelli G, Flamigni C, Fleming S, Cecconi S 2004. Mouse oocyte meiotic resumption and polar body extrusion in vitro are differentially influenced by FSH, epidermal growth factor and meiosis-activating sterol. Hum Reprod 19:2913–2918 [DOI] [PubMed] [Google Scholar]
- 60.Denys A, Avazeri N, Lefevre B 2007. The PKC pathway and in particular its β1 isoform is clearly involved in meiotic arrest maintenance but poorly in FSH-induced meiosis resumption of the mouse cumulus cell enclosed oocyte. Mol Reprod Dev 74:1575–1580 [DOI] [PubMed] [Google Scholar]
- 61.Ogreid D, Ekanger R, Suva RH, Miller JP, Doskeland SO 1989. Comparison of the two classes of binding sites (A and B) of type I and type II cyclic-AMP-dependent protein kinases by using cyclic nucleotide analogs. Eur J Biochem 181:19–31 [DOI] [PubMed] [Google Scholar]
- 62.Qi Z, Hao CM, Salter K, Redha R, Breyer MD 1999. Type II cAMP-dependent protein kinase regulates electrogenic ion transport in rabbit collecting duct. Am J Physiol 276:F622–F628 [DOI] [PubMed]
- 63.Feliciello A, Gallo A, Mele E, Porcellini A, Troncone G, Garbi C, Gottesman ME, Avvedimento EV 2000. The localization and activity of cAMP-dependent protein kinase affect cell cycle progression in thyroid cells. J Biol Chem 275:303–311 [DOI] [PubMed] [Google Scholar]
- 64.Porcellini A, Messina S, De Gregorio G, Feliciello A, Carlucci A, Barone M, Picascia A, De Blasi A, Avvedimento EV 2003. The expression of the thyroid-stimulating hormone (TSH) receptor and the cAMP-dependent protein kinase RII β regulatory subunit confers TSH-cAMP-dependent growth to mouse fibroblasts. J Biol Chem 278:40621–40630 [DOI] [PubMed] [Google Scholar]
- 65.Fan HY, Huo LJ, Chen DY, Schatten H, Sun QY 2004. Protein kinase C and mitogen-activated protein kinase cascade in mouse cumulus cells: cross talk and effect on meiotic resumption of oocyte. Biol Reprod 70:1178–1187 [DOI] [PubMed] [Google Scholar]