Abstract
Introduction: For chronic prosthetic joint infections (PJI), complete removal of the infected prosthesis is necessary in order to cure the infection. Unfortunately, a subgroup of patients is not able to undergo a revision surgery due to high surgical risk. Alternatively, these patients can be treated with antibiotic suppressive therapy (AST) to suppress the infection. Aim: To evaluate the efficacy and tolerability of AST. Methods: We retrospectively collected data (period 2009-2015) from patients with a PJI (of hip, knee or shoulder) who were treated with AST at the University Medical Center Groningen, the Netherlands. AST was defined as antibiotic treatment for PJI that was started after the usual 3 months of antibiotic treatment. The time of follow-up was defined from the time point AST was started. Treatment was considered as failed, when the patient still experienced joint pain, when surgical intervention (debridement, removal, arthrodesis or amputation) was needed to control the infection and/or when death occurred due to the infection. Results: We included 21 patients with a median age of 67 years (range 21 - 88) and with a median follow-up of 21 months (range 3 - 81). Coagulase negative staphylococci (CNS) (n=6), S. aureus (n=6) and polymicrobial flora (n=4) were the most frequently found causative pathogens. Most patients with CNS and S. aureus were treated with minocycline (67%) and clindamycin (83%) as AST, respectively. Overall, treatment was successful in 67% of patients. Failure was due to persistent joint pain (n=1), surgical intervention because of an uncontrolled infection (n=3), and death due the infection (n=3). We observed a treatment success of 90% in patients with a 'standard' prosthesis (n=11), compared to only 50% in patients with a tumor-prosthesis (n=10). Also, treatment was successful in 83% of patients with a CNS as causative microorganism for the infection, compared to 50% in patients with a S. aureus. Patients who failed on AST had a higher ESR in comparison to patients with a successful treatment (mean 73 ± 25SD versus 32 ± 19SD mm/hour (p = 0.007), respectively. 43% of patients experienced side effects and led to a switch of antibiotic treatment or a dose adjustment in almost all of these patients. Conclusions: Removal of the implant remains first choice in patients with chronic PJI. However, AST is a reasonable alternative treatment option in a subgroup of patients with a PJI who are no candidate for revision surgery, in particular in patients with a 'standard' prosthesis and/or CNS as the causative micro-organism.
Keywords: prosthetic joint infection, antibiotic suppressive therapy, side effects
Introduction
For patients with a chronic prosthetic joint infection (PJI), removal of the infected prosthesis via a one- or two-stage revision surgery is required to cure the infection. The chance to cure the infection depends on multiple factors, including the affected joint, the microorganism(s) that are involved in the infection and several host-factors.1-4 In general, if the infection is considered as difficult to treat, a two-stage surgical revision is indicated. Overall, the current success rates of revision surgery are quite high, on average, around 90% for both one-stage and two-stage procedures.1-4
Unfortunately, this means that in roughly 10% of patients, re-infection occurs and re-revision surgery is needed. Multiple revision surgeries ultimately diminish the chance to cure the infection due to poor bone stock and soft tissue injury.5 As a result, complete removal of the prosthesis resulting in a Girdlestone situation, arthrodesis or lower-limb amputation is, in most cases, inevitable in order to eliminate the infection. Antibiotic suppressive therapy (AST) can be subscribed for these patients as alternative treatment to suppress the infection without removal of the prosthesis as long as possible, and to avoid these disabling surgeries. This conservative antibiotic treatment approach can also be applied in patients with a poor prognosis due to cancer, and in elderly patients or patients with severe comorbidity that are not vital enough to undergo revision surgery.
Apart from its main benefit, i.e. avoidance of complex and potentially harmful surgery, important disadvantages of AST may include treatment failure and unmanageable antibiotic side effects. Because of the ageing population and the subsequent rise of joint arthroplasties performed, it is expected that chronic PJI's and the number of 'inoperable patients' increase.6 Therefore, in the following decades, AST will probably play a more prominent role in the treatment of chronic PJI's. Patients, as well as treating orthopedic surgeons, infectiologists and medical microbiologists, should know of the advantages and disadvantages of AST. However, scientific data are scarce.
To determine the efficacy and tolerability of AST in patients who are treated with AST, we retrospectively evaluated our cohort of patients with a PJI who were treated according to this regime, in order to inform and guide physicians in making a balanced treatment decision for their patients.
Material and Methods
Data collection
We retrospectively collected data from the period of January 2009 until January 2015 from patients with a chronic PJI that were treated with AST and followed at the University Medical Center Groningen. PJI was diagnosed using the diagnostic criteria described by the Infectious Diseases Society of America.7 A chronic PJI was defined as symptoms that existed for more than 3 weeks. PJI was categorized in early (< 3 month after implantation), delayed (3 months - 2 years after implantation) and late infection (> 2 years after implantation). We collected the data by analyzing the reports of our weekly multidisciplinary meetings, where all patients with a PJI are discussed. Therefore, all subsequent patients in our center treated with AST are included in the study. AST was defined as antibiotic treatment that was started after the standard 3 months of 'regular' antibiotic treatment (in most cases 2 weeks of intravenous therapy and 10 weeks of oral therapy). The choice for AST was based upon patients' comorbidity, prognosis and the degree of tissue vitality and bone stock. The choice for AST was made by a multidisciplinary team consisting of orthopedic surgeons, infectiologists and medical microbiologists.
Besides demographic characteristics, we collected several patients' parameters considered to be related to outcome; i.e. the patients' underlying disease that led to joint arthroplasty, the affected joint, the number of revision surgeries that were performed in the affected joint prior to the start of AST, the type of prosthesis that was used (i.e. a standard or tumor prosthesis), the cultured micro-organisms that were accounted as the causative pathogens of the infection, the number of surgical debridements and lavages before suppression therapy was started and the indication reported for AST. We also evaluated the degree of inflammation by collecting the C-reactive protein (CRP), Erythrocyte Sedimentation Rate (ESR) and leucocytes in blood closest around the start of AST.
Outcome; definition of treatment failure
Treatment was considered as failed, when: 1) the patient still reported joint pain during follow-up visits at the outpatient clinic, 2) when surgical intervention was needed to control the infection (i.e. removal of the prosthesis (Girdlestone or arthrodesis), revision surgery and/or amputation/dysarticulation) and/or 3) when death occurred due to the infection.
Antibiotic treatment and side effects
The choice for the type of AST was based on the cultured micro-organism(s) and its susceptibility pattern(s), and the expected (long-term) side effects. The antibiotic treatment was advised by the involved medical microbiologist and/or infectiologist. If micro-organisms were considered as a contaminant and were not covered by the chosen antibiotic treatment, this was reported. Side effects of antibiotic treatment mentioned by patients during outpatient clinic visits were collected and noted whether these side effects led to dose adjustments, switch of therapy and/or discontinuation of treatment.
Follow-up
Our patients visited the outpatient clinic at regular intervals (on average with a 3-6 month interval) to evaluate treatment response. When AST was successful for a longer time-period and patients experienced no side effects, some patients were followed by the general practitioner and were instructed to contact their treating orthopedic surgeon if problems arose. The time of follow-up was defined from the time point AST was started. The end-point of follow-up of our study was defined as: the patients' last visit at our outpatient clinic and/or orthopedic ward during admission, or when treatment failed (as described above under: 'outcome; definition of treatment failure').
Statistical analysis
Since multiple variables may affect treatment outcome, we considered the number of included patients too small in order to perform a multiple regression analysis. Therefore, descriptive statistics were used to summarize the results. For continuous data (i.e. inflammatory parameters), the successful and failed treatment group were compared by using the Mann-Whitney U-test. All significance tests were performed 2-tailed. P-values <0.05 were considered as statistically significant. Statistics was performed using Prism 7 for Mac OS X (1994-2016 GraphPad Software, Inc.).
Results
Patient characteristics
In the period between January 2009 and January 2015, 24 patients in our center were started with oral AST. We excluded 3 patients from the analysis; 1 because of patient non-compliance (refused to take antibiotic therapy), 1 patient died from another cause than infection shortly after AST was started, and 1 patient was transferred to another hospital and was lost to follow-up. In total, 21 patients were included in the study. Table 1 shows the patients' baseline characteristics and treatment outcome. The median age of the included patients was 67 years (range 21 - 88). A large proportion of our patients (45%) was obese (BMI ≥ 30), 54% were known with coronary heart disease, 14% with rheumatoid arthritis and 9% with diabetes mellitus. The majority of implants were revised prostheses (67%). PJI was caused by the following micro-organisms: S. epidermidis (n=6), S. aureus (n=6), polymicrobial flora (n=4), P. acnes (n=2), S. agalactiae (n=1), S. pyogenes (n=1) and B. fragilis (n=1). The median onset of PJI after implantation was 10 months (range 3-81 months). PJI was classified as an early infection in 24%, as a delayed infection in 33%, and as a late infection in 43% of patients. The indication to choose for AST was in 43% of patients because of poor bone stock and/or severe tissue injury. In most cases this was due to a malignancy or several revision surgeries in the past. 48% of patients were on AST because of a poor prognosis and/or severe comorbidity. In two cases, AST was because of patients' refusal of surgery. 14 of the 21 patients (67%) underwent a debridement and/or lavage of the affected joint (mean number of procedures per patient: 1.5 (± 0.6 SD) before AST was started.
Table 1.
Overview of patient characteristics and outcome of antibiotic suppressive therapy (AST).
| Pt | Sex | Age | Indication AST | Indication prosthesis |
Revised prosthesis |
Affected joint |
Type of prosthesis |
Months after implantation |
Surgeries before start AST (n) |
Micro- organism(s) |
Outcome | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | M | 70 | comorbidity | osteoarthritis | yes | hip | standard | 54 (late) | lavage (3) | S. pyogenes | Successful | |
| 2 | M | 69 | comorbidity/prognosis | malignancy | yes | hip | tumor | 1 (early) | no | S. epidermidis | Successful | |
| 3 | M | 40 | poor soft tissue | malignancy | yes | hip | tumor | 1 (early) | lavage (1) | S. aureus | Successful | |
| 4 | M | 71 | prognosis | malignancy | yes | hip | standard | 1 (early) | no | S. epidermidis, E.cloacae | Successful | |
| 5 | F | 76 | comorbidity | osteoarthritis | yes | knee | standard | 30 (late) | no | S. epidermidis | Successful | |
| 6 | F | 55 | comorbidity/prognosis | malignancy | yes | hip | standard | 1 (early) | lavage (1) | S. aureus, E. faecalis | Successful | |
| 7 | F | 71 | patient wish | osteoarthritis | yes | hip | standard | 39 (late) | lavage (1) | Bacteroides fragilis | Successful | |
| 8 | F | 47 | poor soft tissue/bone stock | osteoarthritis | yes | hip | standard | 1 (early) | DAIR (1), lavage (1) | S. epidermidis (rifampin R) | Successful | |
| 9 | M | 80 | comorbidity | fracture | no | shoulder | standard | 1 (early) | lavage (1) | P. acnes | Successful | |
| 10 | M | 52 | pore bonestock/prognosis | malignancy | no | knee | tumor | 6 (delayed) | DAIR (1) | S. aureus | Successful | |
| 11 | M | 35 | pore bonestock | osteomyelitis | yes | hip | tumor | 44 (late) | no | S. epidermidis | Successful | |
| 12 | M | 21 | prognosis | malignancy | no | knee | tumor | 24 (late) | excision sarcoma | P. acnes | Successful | |
| 13 | M | 73 | poor soft tissue/bone stock | fracture | yes | hip | standard | 78 (late) | DAIR (1), lavage (2) | S. aureus | Successful | |
| 14 | M | 88 | comorbidity/prognosis | osteoarthritis | no | knee | standard | 1 (early) | lavage (2) | S. epidermidis | Successful | |
| 15 | F | 54 | poor bonestock | RA | yes | knee | tumor | 27 (late) | no | S. aureus | Failed 1 | |
| 16 | M | 70 | comorbidity/prognosis | RA | yes | knee | standard | 176 (late) | lavage (1) | S. aureus | Failed 2 | |
| 17 | F | 59 | poor soft tissue/bone stock | osteoarthritis | yes | hip | tumor | 6 (delayed) | resposition (3) | S. aureus, S. epidermidis | Failed 1 | |
| 18 | F | 67 | poor soft tissue | fracture | no | shoulder | standard | 7 (delayed) | lavage (2) | S. aureus | Failed 2 | |
| 19 | M | 58 | patient wish | malignancy | no | hip | tumor | 2 (early) | DAIR (2) | E.coli (ESBL+), E. faecalis | Failed 2 | |
| 20 | M | 68 | poor soft tissue | fracture | no | hip | tumor | 43 (late) | DAIR (1), lavage (2) | S. agalactiae | Failed 3 | |
| 21 | F | 65 | comorbidity | RA | yes | hip | tumor | 9 (delayed) | DAIR (1) | S. epidermidis | Failed 1 | |
DAIR: debridement, antibiotics, irrigation and retention.
Definition of failed outcome: 1 surgical intervention needed, 2 death due to persistent prosthetic joint infection, 3 persistent pain. RA: rheumatoid arthritis.
Treatment outcome and follow-up
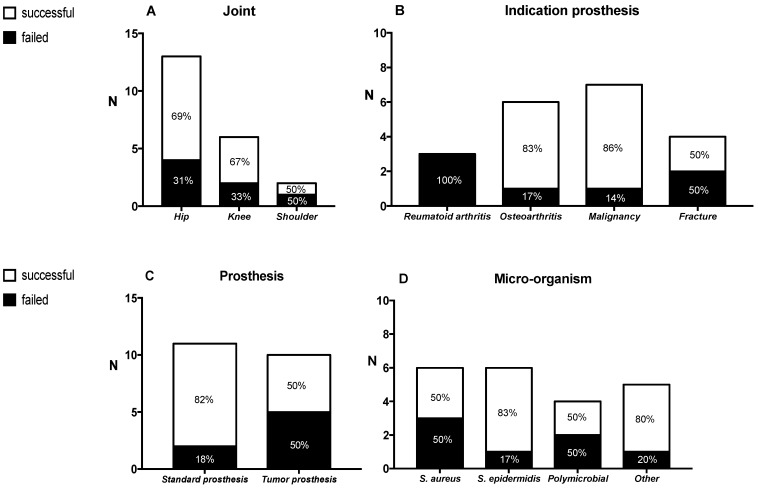
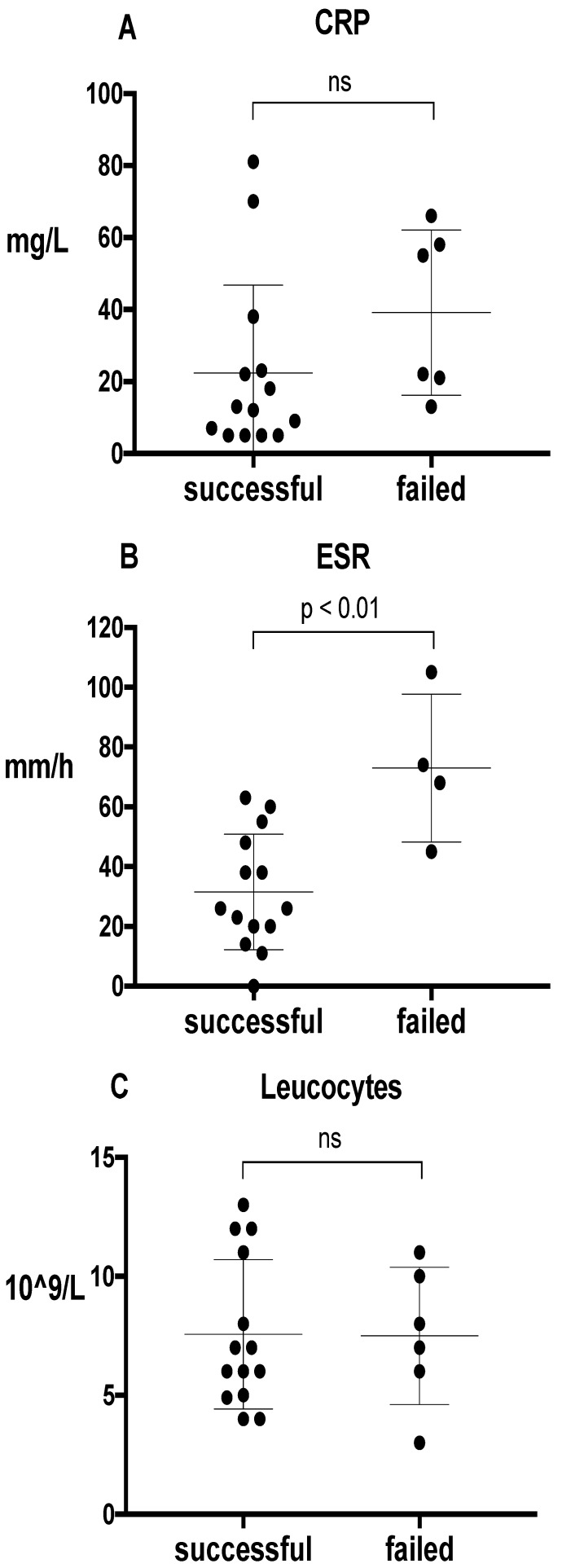
The median follow-up of patients was 21 months (range: 3-81). Overall, AST was successful in 67% of patients (Table 1). Failure of treatment was due to persistent joint pain (n=1), surgical intervention because of an uncontrolled infection (n=3), and death due the infection (n=3). The number of debridement and/or lavage of the affected joint prior to the start of AST was approximately equal between the successful and failed treatment group (9 out of 14 patients (64%) versus 5 out of 7 patients (71%) respectively). Figure 1 outlines the treatment outcome in association with several parameters. In general, we observed a treatment success of 90% in patients with a 'standard' prosthesis (n=11). This success was only 50% in patients with a tumor-prosthesis (n=10). Also, treatment was successful in 83% of patients with a S. epidermidis as causative microorganism for the infection. For S. aureus, treatment success was only 50%. All patients with rheumatoid arthritis (n=3) failed on AST. All of these three patients were treated with methotrexate or prednisone. In our study, no difference in treatment outcome was observed between prosthetic knee and hip infections. Patients who failed on AST did not have a longer follow-up in comparison to patients whose treatment was successful (median follow up 15 months versus 27 months, respectively). Figure 2 shows the treatment outcome in relation to inflammatory parameters. The median time point of laboratory values around the start (day 0) of AST, was +3 days (range -19 to -20 days). Patients who failed on AST had a higher ESR in comparison to patients with a successful treatment (mean 73 ± 25SD versus 32 ± 19SD mm/hour (p = 0.007), respectively).
Figure 1.
Outcome of antibiotic suppressive therapy (n=21) subdivided in several parameters; the affected joint (A), the indication for the prosthesis / underlying condition (B), the type of prosthesis (C) and the causative pathogen / micro-organism for the prosthetic joint infection (PJI) (D). The definition of successful treatment and failed treatment is descripted in the text.
Figure 2.
Outcome of antibiotic suppressive therapy (n=21) in relation to inflammatory parameters before suppression therapy was started); C-reactive protein (CRP) (A), Estimated Sedimentation Rate (ESR) (B), and leucocytes (C). The definition of successful treatment and failed treatment is descripted in the text. Bars represent the median, the 25th and the 75th percentile.
Antibiotic suppressive therapy and reported side effects
The 'regular' antibiotic treatment, the type of AST, the reported side effects and whether these side effects led to a change in antibiotic treatment or dose adjustments, are shown in Table 2. During the first chosen AST regime, nearly half of the patients reported side effects (43%), which led to a change in treatment in almost all of these patients, from which 50% was a dose adjustment and 50% a change in antibiotic treatment. On the second AST regime, only 3 out of 8 patients reported side effects. None of these 3 led to dose adjustments or a change in the antibiotic regime. Reported side effects were equal between the failed and successful treatment group. Overall, clindamycin was best tolerated; side effects were reported in only 2 out of 7 patients. Loss of appetite improved in one patient after the dose was adjusted to 300 mg TID. Minocycline was the least tolerated drug. Side effects were reported in 4 out of 6 patients, and led to a change of treatment or dose adjustment in all of these patients.
Table 2.
Overview of the initial treatment and the antibiotic suppressive therapy (AST) per patient, the reported side effects and subsequent intervention.
| Pt | Intravenous treatment1 |
Oral treatment2 | 1st AST | Side effects AST |
Change of treatment |
2nd AST | Side effects AST | Change of treatment |
|---|---|---|---|---|---|---|---|---|
| 1 | Penicillin 12 mU /day | Amoxicillin 1000 mg TID | Amoxicillin 500 mg TID | No | - | - | - | - |
| 2 | Vancomycin/day3, 4w | Minocycline 100 mg BID | Minocycline 100 mg BID | Nausea, phototoxicity | Yes | Minocycline 100 mg QD | Phototoxicity | No |
| 3 | Penicillin 12 mU/day | Moxifloxacin 400 mg QD Rifampin4 450 mg BID |
Moxifloxacin 400 mg QD | No | - | - | - | - |
| 4 | Vancomycin/day3 Ciprofloxacin 400 mg TID |
Ciprofloxacin 750 mg BID Rifampin4 450 mg BID Linezolid 600 mg BID, 4w Mincocyline 100 mg BID, 6w |
Minocycline 100 mg BID | Nausea, vomiting | Yes | Doxycyclin 100 mg QD | No | - |
| 5 | - | - | Moxifloxacin 400 mg QD | Nausea | No | - | - | - |
| 6 | Amoxicillin 12 g/day Flucloxacillin 12 g/day Gentamicin 3 mg/kg/day |
Amox/clav 500/125 QID Rifampin4 450 mg BID |
Amox/clav 500/125 mg TID | No | - | - | - | - |
| 7 | - - |
Metronidazole 500 mg QID Amox/clav 750/125 TID |
Amox/clav 500/125 mg TID | Nausea | Yes | Minocycline 100 mg QD | Dizziness | No |
| 8 | Vancomycin/day3 | Mincocyline 100 mg BID | Minocycline 100 mg QD | No | - | - | - | - |
| 9 | Ceftriaxone 2 g QD | Clindamycin 600 mg TID Rifampin4 450 mg BID |
Amoxicillin 500 mg TID | Dry mouth | Yes | Amoxicillin 375 mg TID | Malaise | No |
| 10 | Flucloxacillin 12 g/day | Moxifloxacin 400 mg QD Rifampin4 450 mg BID |
Clindamycin 450 mg TID | No | - | - | - | - |
| 11 | - | Linezolid 600 mg BID, 4w Mincocyline 100 mg BID, 8w |
Minocycline 100 mg BID | Diarrhea | Yes | Minocycline 100 mg QD | No | - |
| 12 | Flucloxacillin 12 g/day | Moxifloxacin 400 mg QD Rifampin4 450 mg BID |
Clindamycin 450 mg TID | Nausea | Yes | Moxifloxacin 400 mg QD | No | - |
| 13 | Flucloxacillin 12 g/day | Moxifloxacin 400 mg QD Rifampin4 450 mg BID |
Clindamycin 600 mg TID | No | - | - | - | - |
| 14 | Vancomycin/day3 | Linezolid 600 mg BID, 4w Mincocyline 100 mg BID, 6w |
Minocycline 100 mg QD | No | - | - | - | - |
| 15 | - | Clindamycin 600 mg TID | Clindamycin 600 mg TID | Loss of appetite | Yes | Clindamycin 300 mg TID | No | - |
| 16 | Flucloxacillin 8 g5/day, 1w | Clindamycin 600 mg TID | Clindamycin 600 mg TID | No | - | - | - | - |
| 17 | Daptomycin 8 mg/kg/day6 | Mincocyline 100 mg BID Rifampin4 450 mg BID |
Minocycline 100 mg BID | Skin rash | Yes | Cotrimoxazole 960 mg BID | No | - |
| 18 | Flucloxacillin 8 g/day5 | Moxifloxacin 400 mg QD Rifampin4 450 mg BID |
Clindamycin 600 mg TID | No | - | - | - | - |
| 19 | Meropenem 2 g TID Vancomycin/day3 Gentamicin 3 mg/kg/day |
Ciprofloxacin 750 mg BID Amoxicillin 1000 mg TID |
Cotrimoxazole 960 mg BID Amoxicillin 1000 mg TID |
No No |
- - |
- - |
- - |
- - |
| 20 | Penicillin 12 mU/day, 4w Gentamicin 3 mg/kg/day, 4w |
Amoxicillin 1000 mg TID, 8w | Amoxicillin 500 mg TID | No | - | - | - | - |
| 21 | - | - | Clindamycin 450 mg TID | No | - | - | - | - |
1 Intravenous treatment comprises 2 weeks of treatment, unless otherwise stated. 2 Oral treatment comprises 10 weeks of treatment, unless otherwise stated. 3 The dose of vancomycin was determined by therapeutic drug monitoring, aiming towards a serum concentration level of ±25 mg/L (continuous infusion). 4 In general, rifampin is added during intravenous treatment after susceptibility is known (after ± 5 days). 5 8 grams of flucloxacillin was chosen because of renal insufficiency. 6 Daptomycin was chosen because of allergy to penicillin.
Discussion
With a median follow-up of almost two years, we evaluated the efficacy and tolerability of AST in patients with a chronic PJI. With an overall treatment success of 67% - with the highest success rate observed in patients with a 'standard' prosthesis and a S. epidermidis infection- our data showed that AST is a reasonable alternative treatment option for patients in whom revision surgery is not feasible or attractive because of a variety of reasons. Our treatment success is in agreement with previous studies, which report success rates between 63%-86%.8-11 Reported success rates of >80% in some studies can be explained by the inclusion of acute PJI's, from which it is known that retention of the prosthetic joint can be achieved.7
Based on our data, we cannot draw conclusions about prognostic factors for treatment success or failure. We did observe that treatment succeeded in almost all of our patients with a PJI caused by a S. epidermidis. This finding may not sound surprising since S. epidermidis has low virulence and its natural course of infection is often smouldering and low-grade in nature. However, Barberan et al. did show a 50% relapse rate in patients with a PJI caused by coagulase negative staphylococci (CNS) and who were treated with a conservative treatment regime (i.e. surgical debridement with implant retention and a 3 month course of antibiotic treatment). In addition, Byren et al. showed that when antibiotic treatment is discontinued, even after a long period of suppression therapy (with a minimum of 180 days), relapse of infection occurs in around 30% of patients.12 Therefore, long-term antibiotic treatment in patients with a PJI due CNS does have additional value to suppress the infection, and its favorable outcome is not merely due to its natural course. We also observed a high success rate in patients with a 'standard' prosthesis, in comparison with patients with a 'tumor' prosthesis, in whom 50% of AST failed. In previous studies on AST, the type of prosthesis is not mentioned. However, it is known, that tumor prostheses are more prone to infection in comparison to standard prostheses. This may be partly explained by poor soft tissue and/or bone stock due to radiation therapy, and the larger surface area of a 'tumor' prosthesis.13 In contrast to other studies, in our cohort of patients, we did not observe higher failure rates in patients with multiple prior revision surgeries or in patients with knee arthroplasties.8
To our knowledge, we are the first who evaluated the degree of inflammation in relation to treatment outcome. Although laboratory data were not available for every patient shortly before or after the start of AST, we believe our results do suggest that patients who have an ESR > 65 mm/h when suppression therapy is started, probably have a higher chance of treatment failure. Our three patients with rheumatoid arthritis all failed on AST, two of them had an ESR > 65 mm/h. In previous studies, rheumatoid arthritis and/or the use of immunosuppressive medication has not been evaluated as variable for treatment outcome, and would be interesting to include as potential prognostic factor in future studies.
Our data give a good impression about the challenges patients and treating physicians face regarding potential side effects of AST. 43% our patients experienced side effect during antibiotic therapy. Although the majority of them needed a switch of therapy or an alternation of dose, the antibiotic treatment was ultimately well tolerated. The number of side effects whilst on the 'first' antibiotic regime is in accordance with the study of Tsukayama et al.14 They also report side effects in 38% of patients that led to a change in antibiotic therapy. In our study, no severe side effects occurred. One previous study reported a 22% incidence (4 out of 18 patients) of Clostridum difficile infection.15 This high incidence might be due to the longer follow-up of this study (at least 4 years) or due to the use of a different antibiotic treatment regime in the 4 patients with a Clostridium infection (2 patients were treated with with dicloxacillin).16-17
In conclusion, for patients with a PJI who are no candidate for revision surgery antibiotic suppressive therapy seems a reasonable alternative treatment, with the highest success rates in patients with a standard-prosthesis or with a S. epidermidis infection. Although side effects occur frequently, most patients finally tolerate treatment after a change in therapy has been made. Although numbers are low, antibiotic suppressive therapy failed for all our patients with rheumatoid arthritis.
Abbreviations
- AST
Antibiotic Suppressive therapy
- CNS
Coagulase Negative Staphylococci
- CRP
C-Reactive Protein
- DAIR
Debridement, Antibiotics, Irrigation and Retention
- ESR
Erythrocyte Sedimentation Rate
- PJI
Prosthetic Joint Infection
- SD
Standard Deviation
- RA
Rheumatoid Arthritis
References
- 1.Lange J, Troelsen A, Thomsen RW. et al. Chronic infections in hip arthroplasties: comparing risk of reinfection following one-stage and two-stage revision: a systematic review and meta-analysis. Clin Epidemiol. 2012;4:57–73. doi: 10.2147/CLEP.S29025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Masters JPM, Smith NA, Foguet P. et al. A systematic review of the evidence for single and two-stage revision of infected knee replacement. BMC Musculoskeletal Disorders. 2013;14:222. doi: 10.1186/1471-2474-14-222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kunutsor SK, Whitehouse MR, Blom AW. et al. Re-infection outcomes following one- and two-stage surgical revision of infected hip prosthesis: a systematic review and meta-analysis. PLoS One. 2015;10(9):3–14. doi: 10.1371/journal.pone.0139166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kunutsor SK, Whitehouse MR, Lenguerrand E. et al. Re-infection outcomes following one- and two-stage surgical revision of infected knee prosthesis: a systematic review and meta-analysis. PLoS One. 2016;11(3):1–15. doi: 10.1371/journal.pone.0151537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cochran AR, Ong KL, Lau E. et al. Risk of reinfection after treatment of infected knee arthroplasty. J Arthroplasty. 2016;31(9 Suppl):156–61. doi: 10.1016/j.arth.2016.03.028. [DOI] [PubMed] [Google Scholar]
- 6.Otten R, van Roermund PM, Picavet HS. Trends in the number of knee and hip arthroplasties: considerably more knee and hip prostheses due to osteoarthritis in 2030. Ned Tijdschr Geneeskd. 2010;154:A1534. [PubMed] [Google Scholar]
- 7.Osmon DR, Berbari EF, Berendt AR. et al. Diagnosis and management of prosthetic joint infection: clinical practice guidelines by the infectious diseases society of America. Clin Inf Dis. 2013;56(1):e1–25. doi: 10.1093/cid/cis803. [DOI] [PubMed] [Google Scholar]
- 8.Siqueira MBP, Saleh A, Klika AK. et al. Chronic suppression of periprosthetic joint infections with oral antibiotics increases infection-free survivorship. J Bone Joint Surg Am. 2015;97:1220–32. doi: 10.2106/JBJS.N.00999. [DOI] [PubMed] [Google Scholar]
- 9.Rao N, Crossett LS, Sinha RK. et al. Long-term suppression of infection in total joint arthroplasty. Clin Orthop Rel Res. 2003;414:55–60. doi: 10.1097/01.blo.0000087321.60612.cf. [DOI] [PubMed] [Google Scholar]
- 10.Pavoni GL, Giannella M, Falcone M. et al. Conservative medical therapy of prosthetic joint infections: retrospective analysis of an 8-year experience. Clin Microbiol Infect. 2004;10:831–837. doi: 10.1111/j.1469-0691.2004.00928.x. [DOI] [PubMed] [Google Scholar]
- 11.Goulet JA, Pellicci PM, Brause BD. et al. Prolonged suppression of infection in total hip arthroplasty. J Arthroplasty. 1988;3(2):109–116. doi: 10.1016/s0883-5403(88)80075-5. [DOI] [PubMed] [Google Scholar]
- 12.Byren I, Bejon P, Atkins BL. et al. One hundred and twelve infected arthroplasties treated with 'DAIR' (debridement, antibiotics and implant retention): antibiotic duration and outcome. J Antimicrob Chemother. 2009;63(6):1264–71. doi: 10.1093/jac/dkp107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Grimer RJ, Aydin BK, Wafa H. et al. Very long-term outcomes after endoprosthetic replacement for malignant tumours of bone. Bone Joint J. 2016;98B(6):857–64. doi: 10.1302/0301-620X.98B6.37417. [DOI] [PubMed] [Google Scholar]
- 14.Tsukayama DT, Wicklund B, Gustilo RB. Suppressive antibiotic therapy in chronic prosthetic joint infections. Orthopedics. 1991;14(8):841–4. doi: 10.3928/0147-7447-19910801-07. [DOI] [PubMed] [Google Scholar]
- 15.Segreti J, Nelson JA, Trenholme GM. Prolonged suppressive antibiotic therapy for infected orthopedic prostheses. Clin Infect Dis. 1998;27(4):711–3. doi: 10.1086/514951. [DOI] [PubMed] [Google Scholar]
- 16.Vesteinsdottir I, Gudlaugsdottir S, Einarsdottir R. et al. Risk factors for Clostridium difficile toxin-positive diarrhea: a population-based prospective case-control study. Eur J Clin Microbiol Infect Dis. 2012;31(10):2601–10. doi: 10.1007/s10096-012-1603-0. [DOI] [PubMed] [Google Scholar]
- 17.Søes LM, Holt HM, Böttiger B. et al. Risk factors for Clostridium difficile infection in the community: a case-control study in patients in general practice, Denmark, 2009-2011. Epidemiol Infect. 2014;142(7):1437–48. doi: 10.1017/S0950268813002380. [DOI] [PMC free article] [PubMed] [Google Scholar]