Abstract
Background
Routine immunisation with pneumococcal conjugate vaccines (PCV7/10/13) has reduced invasive pneumococcal disease (IPD) due to vaccine serotypes significantly. However, an increase in disease due to non-vaccine types, or serotype replacement, has been observed. Serotypes’ individual contributions to IPD play a critical role in determining the overall effects of PCVs. This study examines the distribution of pneumococcal serotypes in children to identify leading serotypes associated with IPD post-PCV introduction.
Methods
A systematic search was performed to identify studies and surveillance reports (published between 2000 and December 2015) of pneumococcal serotypes causing childhood IPD post-PCV introduction. Serotype data were differentiated based on the PCV administered during the study period: PCV7 or higher valent PCVs (PCV10 or PCV13). Meta-analysis was conducted to estimate the proportional contributions of the most frequent serotypes in childhood IPD in each period.
Results
We identified 68 studies reporting serotype data among IPD cases in children. We analysed data from 38 studies (14 countries) where PCV7 was administered and 20 (24 countries) where PCV10 or PCV13 have been introduced. Studies reported early and late periods of PCV7 administration (range: 2001∓13). In these settings, serotype 19A was the most predominant cause of childhood IPD, accounting for 21.8% (95%CI 18.6∓25.6) of cases. In countries that have introduced higher valent PCVs, study periods were largely representative of the transition and early years of PCV10 or PCV13. In these studies, the overall serotype-specific contribution of 19A was lower (14.2% 95%CI 11.1∓18.3). Overall, non-PCV13 serotypes contributed to 42.2% (95%CI 36.1∓49.5%) of childhood IPD cases. However, regional differences were noted (57.8% in North America, 71.9% in Europe, 45.9% in Western Pacific, 28.5% in Latin America, 42.7% in one African country, and 9.2% in one Eastern Mediterranean country). Predominant non-PCV13 serotypes overall were 22F, 12F, 33F, 24F, 15C, 15B, 23B, 10A, and 38 (descending order), but their rank order varied by region.
Conclusion
Childhood IPD is associated with a wide number of serotypes. In the early years after introduction of higher valent PCVs, non-PCV13 types caused a considerable proportion of childhood IPD. Serotype data, particularly from resource-limited countries with high burden of IPD, are needed to assess the importance of serotypes in different settings. The geographic diversity of pneumococcal serotypes highlights the importance of continued surveillance to guide vaccine design and recommendations.
Introduction
Streptococcus pneumoniae is the major cause of serious invasive diseases such as bacterial pneumonia, septicaemia, and meningitis in young children worldwide. An estimated 14.5 million cases of invasive pneumococcal disease (IPD) occurred globally in 2000 before introduction of pneumococcal conjugate vaccines (PCVs). Widespread availability of PCVs has reduced the burden of IPD substantially, from over 800,000 annual deaths before PCV introduction to 541,000 deaths in 2008 [1, 2]. Although two formulations of PCVs are available to protect against disease, S. pneumoniae still poses a significant burden on individuals and healthcare systems.
PCVs have been largely effective in reducing IPD associated with serotypes included in currently available formulations. Yet, concerns exist about their long-term effects since these contain a limited number of serotypes and the potential role of non-vaccine serotypes. The first PCV, licensed in 2000, offered protection against 7 serotypes (4, 6B, 9V, 14, 18C, 19F, 23F). By 2015, PCV7 had been replaced, and PCV10 (PCV7 + 1, 3, 7F) or PCV13 (PCV10 + 19A, 6A, 3) have been introduced in over 130 countries [3, 4]. Before the introduction of PCVs, vaccine-targeted serotypes were associated with a 82∓88% of IPD in North America, 72∓88% in Europe, 68∓79% in Oceania, 58∓82% in Latin America, 49∓77% in Africa, and 52∓74% in Asia [5]. After the introduction and scale up of PCV7 in routine immunisation programmes, IPD caused by these 7 serotypes decreased significantly in children and other age groups [6, 7]. Nasopharyngeal carriage of S. pneumoniae, which can lead to invasive disease [8], was also dramatically impacted by PCVs. Studies indicated that PCVs reduced the prevalence of targeted serotypes among carriers [9] to the extent that these strains have nearly disappeared in some settings in both the vaccinated and unvaccinated persons [10].
In light of global efforts for universal immunisation with PCV, it is important to examine which serotypes are associated with IPD in the post-PCV era to gain insights into the evolving epidemiology of S. pneumoniae. Serotype replacement, an increase in incidence of disease due to serotypes [8] not included in PCV7, was noticed among young children using data from 21 large surveillance systems [11]. In these industrialised settings, by the 7th year of PCV7, the rate ratio of vaccine-related IPD (RR 0.03 95%CI 0.01∓0.10) decreased substantially as compared to the ratio of non-PCV7 types (RR 2.81 95%CI 2.12∓3.71). Thus, there is a clear need for close monitoring of serotypes causing IPD to inform public health actions [8]. We aimed to systematically assess and describe the global distribution of serotypes causing IPD in young children after the introduction of PCVs to better understand the contribution of individual serotypes, including non-vaccine types and inform recommendations for vaccine use.
Methods
Search methods and inclusion criteria
We conducted a systematic review of the literature according to the PRISMA guidelines. We searched electronic medical databases for studies and surveillance reports (published between 1 January 2000 and 31 December 2015) reporting serotype data from children with IPD after the introduction of a PCV in the study area. Search strategies with database-specific MeSH and free search terms were developed with the assistance of a medical librarian for Medline, Embase, and Global Health (via Ovid); EMRO, SEARO, and WPRO regional databases (Global Health Library), LILACS, and Web of Science. Strategies were designed to retrieve records that included the following terms: Streptococcus pneumoniae and, IPD or syndromes, and vaccines, and serotypes (search strategies are available in S1 Table).
We included peer-reviewed studies and annual surveillance reports from Department of Health websites from countries from which the electronic medical databases retrieved national surveillance publications. IPD was defined as the identification of S. pneumoniae from a normally sterile site (e.g. blood, cerebrospinal, pleural effusions, or joint fluid) in children. No language restrictions or publication type were imposed at this stage. We also scanned the reference list of included studies to identify any additional studies. Eligibility criteria were as described in Box 1, which were modified from previous reviews assessing the serotype distribution in childhood IPD [5, 11].
Box 1. Eligibility criteria for this review
Inclusion
Observational studies or annual national surveillance reports from selected Departments of Health from settings with PCV uptake of at least 25% during the study period described
Studies reporting at least 20 serotyped isolates overall and with at least 50% of reported IPD cases were serotyped
Study reports data on different serogroups/types for invasive disease (defined as isolates from normally sterile sites) in children
Study population was representative of the general population, not a selected group with specific co-morbidities
Surveillance conducted for at least 12 continuous months
Exclusion
Case reports, narrative reviews, quarterly or province-level surveillance reports, if annual and/or national were available
Serotype data from studies with high risk of bias: e.g. studies focused on serotypes from severe cases, most frequent types, or antimicrobial resistance
Data for years after PCV introduction are not extractable independently or the study does not provide a description of PCV use in the area
Data only reported for serotypes included in PCVs (PCV7, PCV10, PCV13)
Serotype data included isolates obtained from non-sterile sites (e.g. nasopharynx) or not extractable specifically for otherwise healthy children (i.e. study population includes all immunocompromised population or adults)
Data overlap with other studies included in the analysis (Studies with the longest study period or larger sample size were preferred)
Serotypes in Pneumococcal conjugate vaccines
PCV7: 4, 6B, 9V, 14, 18C, 19F, 23F
PCV10: PCV7 + 1, 5, 7F
PCV13: PCV10 + 3, 6A, 19A
Non-PCV13: those not included in PCV13
Data collection and analysis
Two reviewers (EB, LG) independently reviewed identified publications and extracted relevant data into a template (Microsoft Excel), which was piloted before use. Datathief III software (http://datathief.org/) was used to extract data from images. Study periods were classified according to the PCV administered during the study period as either PCV7 or higher valent (PCV10 or PCV13, hereafter PCV10/13). From each study included in analysis, data for young children (<5 years) was preferred, if other age groups were available to minimise bias. Non-vaccine types were those currently not included in PCV13 (non-PCV13).
Meta-analysis
Serotype-specific percentages were calculated for each study (% serotype x = total number of cases serotype x over total IPD isolates with serotype data x 100). Serogroup data were not redistributed into serotypes. Using metan in Stata 13 (Statacorp, College Station, TX), pooled estimates and 95% confidence intervals (95%CI) were calculated for serotype-specific proportions using the transformed log of the proportion and random effects model (DerSimonian-Laird method). A continuity correction was used to include data from studies with zero counts in independent meta-analyses. If there were multiple studies from the same setting, the most representative of childhood IPD (i.e. larger sample size, or reporting data for any IPD rather than a single syndrome) was included in meta-analysis. Most data for settings where PCV10 or PCV13 have been introduced were from countries that had transitioned from PCV7, except Brazil, Chile, and Colombia. The first year of PCV10 or PCV13 introduction for these 3 Latin American countries was excluded from analyses to allow time for scaling up of vaccine uptake. For other settings, data from the transition year from PCV7 to higher valent PCVs was included in meta-analysis, as it was not possible to exclude this year from analysis for all studies. We used data exclusively for years with either PCV7 or the current higher valent PCV, except when disaggregation was not possible. We report overall and regional pooled estimates with 95%CIs for vaccine types and individual non-PCV13 types causing at least 1% of IPD in the dataset of countries that have introduced PCV10 or PCV13.
Results
Literature review
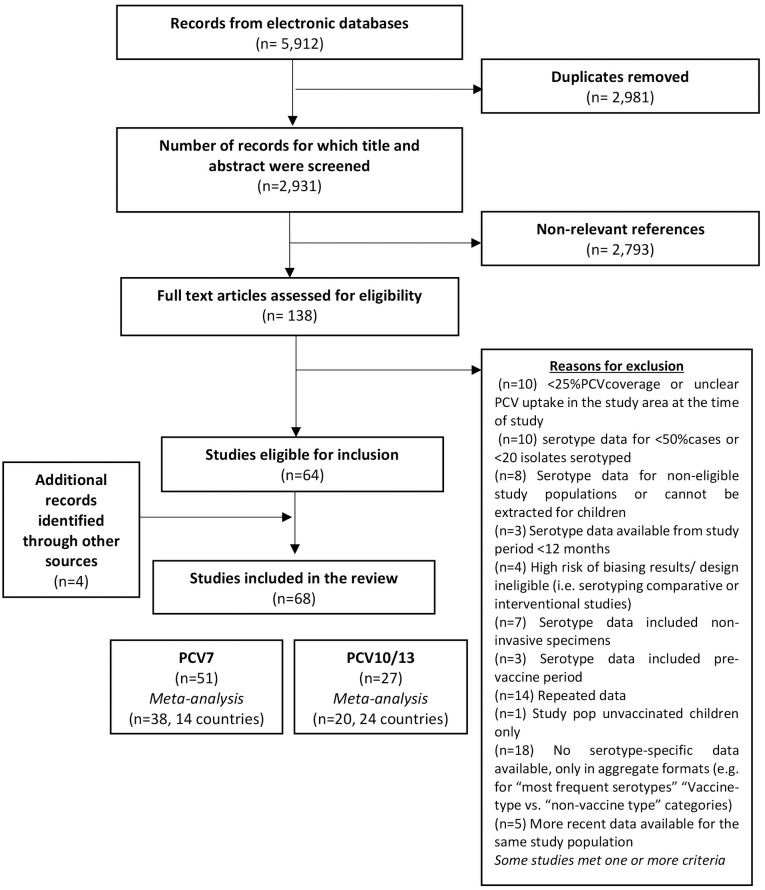
We identified 5,912 records through databases search and identified 138 articles for full text examination (Fig 1). Of these, 64 [6, 7, 12–73] articles met our pre-defined eligibility criteria and we identified an additional 4 surveillance reports [74–77] from online searches.
Fig 1. PRISMA flowchart—Literature review process.
Characteristics of included publications
A total of 27 countries were represented in the 68 publications included in the review. Surveillance data were retrieved from online reports for Latin America (SIREVA II reports 2009–12, considered as 1 publication), Scotland (UK), Australia, New Zealand, and Singapore. Studies identified overlapping geographic, time period or for individual clinical syndromes were excluded from meta-analyses (details in S2 Table).
Main characteristics of studies included in meta-analysis are available in Table 1. Eight publications reported serotype data for years after both time periods after PCV7 and PCV10/13 implementation. These were extracted separately for analysis. After classification of the studies, we analysed 67 data points assessing childhood IPD, primarily from North America (n studies = 17), Europe (n = 28), and Latin America (n = 11), but also from Western Pacific (n = 8), Eastern Mediterranean (n = 2) and Africa (n = 1).
Table 1. Main characteristics of studies included in the review and meta-analyses.
| Last author (publication year) | Country | PCV | Introduction year | Study years (PCV uptake) | Isolates | PCV13 types (%) | Non-PCV13 (%) |
|---|---|---|---|---|---|---|---|
| Rendi-Wagner (2009) | Austria | PCV7 | 2001* | 2005–06 (25%) | 36 | 97.2 | 2.8 |
| Lepoutre (2015) | France | PCV7 | 2003*, 2006† | 2008–09 (83–95%) | 483 | 71.4 | 28.6 |
| Varon (2015) | France | PCV7 | 2006† | 2008–09 (>86%) | 388 | 67.5 | 32.5 |
| van der Linden (2015) | Germany | PCV7 | 2006† | 2007–10 (80–85%) | 542 | 68.1 | 31.9 |
| Knol (2015) | Netherlands | PCV7 | 2006† | 2008–11 (94–95%) | 128 | 52.3 | 47.7 |
| Steens (2013) | Norway | PCV7 | 2006† | 2006–10 (86–92%) | 165 | 69.1 | 30.9 |
| Aguiar (2008) | Portugal | PCV7 | 2001* | 2003–05 (33%) | 90 | 85.6 | 14.4 |
| Aristegui (2007) | Spain | PCV7 | 2001* | 2002–03 (28–45%) | 77 | 88.3 | 11.7 |
| Barricarte (2007) | Spain | PCV7 | 2001* | 2001–05 (45%) | 85 | 87.1 | 12.9 |
| Calbo (2006) | Spain | PCV7 | 2001* | 2002–04 (34%) | 64 | 78.1 | 21.9 |
| Guevara (2014) | Spain | PCV7 | 2001* | 2004–09 (25–61%) | 106 | 78.3 | 21.7 |
| Munoz-Almagro (2011) | Spain | PCV7 | 2001* | 2009§ | 130 | 80.0 | 20.0 |
| Perez-Trallero (2009) | Spain | PCV7 | 2001* | 2002–07 (~50%) | 45 | 86.7 | 13.3 |
| Picazo (2011) | Spain | PCV7 | 2001* | 2007–09 (~50%) | 330 | 79.1 | 20.9 |
| Rodriguez (2011) | Spain | PCV7 | 2006† | 2007–09 (94.5%) | 366 | 74.9 | 25.1 |
| Salleras (2009) | Spain | PCV7 | 2001* | 2005–07 (35%) | 240 | 77.9 | 22.1 |
| Vila-Corcoles (2013) | Spain | PCV7 | 2001* | 2002–09 (13–47%) | 65 | 84.6 | 15.4 |
| Ceyhan (2011) | Turkey | PCV7 | 2008† | 2008–10§ | 146 | 77.4 | 22.6 |
| Miller (2011) | UK | PCV7 | 2006† | 2008–10§ | 528 | 69.7 | 30.3 |
| Moore (2014) | UK | PCV7 | 2006† | 2006–10 (92–87%) | 65 | 49.2 | 50.8 |
| Parra (2013) | Colombia | PCV7 | 2009 | 2010–11 (69.9%) | 84 | 70.2 | 29.8 |
| Bettinger (2010) | Canada | PCV7 | 2005† | 2006–07 (80–90%) | 212 | 69.8 | 30.2 |
| De Wals (2012) | Canada | PCV7 | 2004 | 2007 (>90%) | 113 | 63.7 | 36.3 |
| Kellner (2009) | Canada | PCV7 | 2002 | 2003-07(87–91%) | 67 | 76.1 | 23.9 |
| Black (2007) | USA | PCV7 | 2000† | 2001–05 (86%) | 84 | 34.5 | 65.5 |
| Bruce (2015) | USA | PCV7 | 2001† | 2005–08§ | 126 | 61.9 | 38.1 |
| Byington (2005) | USA | PCV7 | 2000† | 2001–03§ | 105 | 74.3 | 25.7 |
| Croney (2013) | USA | PCV7 | 2000† | 2002–10§ | 157 | 60.5 | 39.5 |
| Hsu (2010) | USA | PCV7 | 2000† | 2001-02/06-07§ | 130 | 66.2 | 33.8 |
| Kaplan (2013) | USA | PCV7 | 2000† | 2007–09§ | 609 | 67.7 | 32.3 |
| Pilishvili (2010) | USA | PCV7 | 2000† | 2006–07§ | 519 | 67.9 | 32.1 |
| Schutze (2004) | USA | PCV7 | 2000† | 2001–03§ | 75 | 77.3 | 22.7 |
| Sharma (2013) | USA | PCV7 | 2000† | 2008–09§ | 47 | 59.6 | 40.4 |
| Weatherholtz (2010) | USA | PCV7 | 2000† | 2001–06§ | 115 | 76.5 | 23.5 |
| Williams (2011) | Australia | PCV7 | 2001*, 2005† | 2006–07§ | 201 | 67.2 | 32.8 |
| Chiba (2014) | Japan | PCV7 | 2010/11* | 2011–12 (50–90%) | 302 | 55.0 | 45.0 |
| Ishiwada (2014) | Japan | PCV7 | 2010*, 2011† | 2012–13§ | 33 | 51.5 | 48.5 |
| Suga (2015) | Japan | PCV7 | 2010* | 2011–13 (89%) | 308 | 68.8 | 31.2 |
| von Gottberg (2013) | South Africa | PCV13 | 2011† | 2011–12§ | 839 | 57.3 | 42.7 |
| Al-Sheikh (2014) | Saudi Arabia | PCV13 | 2010† | 2009–12§ | 78 | 85.9 | 14.1 |
| Shibl (2012) | Saudi Arabia | PCV13 | 2010§ | 108 | 94.4 | 5.6 | |
| Varon (2015) | France | PCV13 | 2010† | 2012–13 (>92%) | 181 | 17.7 | 82.3 |
| van der Linden (2015) | Germany | PCV13 | 2009 Dic† | 2010–14 (80–85%) | 567 | 35.4 | 64.6 |
| Knol (2015) | Netherlands | PCV10 | 2011† | 2011–14 (94–95%) | 57 | 21.1 | 78.9 |
| Steens (2013) | Norway | PCV13 | 2011† | 2011–12 (92%) | 47 | 55.3 | 44.7 |
| Guevara (2014) | Spain | PCV13 | 2010 | 2010–13 (78%) | 25 | 52.0 | 48.0 |
| Moore (2014) | UK | PCV13 | 2010† | 2010–13§ | 48 | 37.5 | 62.5 |
| Scotland Surveillance | UK | PCV13 | 2010† | 2010–15§ | 206 | 19.9 | 80.1 |
| Waight (2015) | UK | PCV13 | 2010† | 2013–14§ | 247 | 14.2 | 85.8 |
| SIREVA (Brazil) | Brazil | PCV10 | 2010‡ | 2011–12 (81.5%) | 416 | 68.8 | 31.3 |
| SIREVA (Chile) | Chile | PCV10 | 2011‡ | 2012 (54.0%) | 168 | 72.6 | 27.4 |
| SIREVA (Colombia) | Colombia | PCV10 | 2010‡ | 2011–12 (69.8%) | 208 | 74.0 | 26.0 |
| SIREVA (Costa Rica) | Costa Rica | PCV13 | 2011† | 2011–12 (78.0%) | 38 | 76.3 | 23.7 |
| SIREVA (Ecuador) | Ecuador | PCV10 | 2010† | 2011–12 (71.0%) | 62 | 80.6 | 19.4 |
| SIREVA (El Salvador) | El Salvador | PCV13 | 2011† | 2011–12 (98.1%) | 31 | 74.2 | 25.8 |
| SIREVA (Mexico) | Mexico | PCV13 | 2011† | 2012 (97.8%) | 105 | 66.7 | 33.3 |
| SIREVA (Panama) | Panama | PCV13 | 2011† | 2011–12 (61.8%) | 68 | 88.2 | 11.8 |
| SIREVA (Peru) | Peru | PCV10 | 2011† | 2011–12 (81.9%) | 23 | 69.6 | 30.4 |
| SIREVA (Uruguay) | Uruguay | PCV13 | 2010† | 2010–12 (92.0%) | 96 | 50.0 | 50.0 |
| Demczuk (2013) | Canada | PCV13 | 2010† | 2010–12 (74–90%) | 886 | 55.5 | 44.5 |
| Bruce (2015) | USA | PCV13 | 2010† | 2010–13 (86–96%) | 52 | 25.0 | 75.0 |
| Kaplan (2013) | USA | PCV13 | 2010† | 2010–11§ | 283 | 56.5 | 43.5 |
| Moore (2015) | USA | PCV13 | 2010 | 2012–13 (63–76%) | 177 | 19.2 | 80.8 |
| Australia Surveillance | Australia | PCV13 | 2011† | 2012§ | 184 | 43.5 | 56.5 |
| Nakano (2015) | Japan | PCV13 | 2013† | 2014§ | 126 | 28.6 | 71.4 |
| N Zealand Surveillance | New Zealand | PCV13 | 2014 | 2014–15 (93%) | 78 | 51.3 | 48.7 |
| Singapore Surveillance | Singapore | PCV13 | 2010* 2011† | 2012–14§ | 65 | 86.2 | 13.8 |
Notes—Definitions for PCV uptake varied by study. Uptake is presented as reported in each study.
*PCV was first recommended, approved or licenced,
†Included in national immunisation programmes/Universal administration recommended
‡ First year of PCV implementation in the country. Settings where national or universal PCV administration is recommended, where eligible for inclusion.
§Data not reported.
We analysed serotype data from 7,366 IPD isolates (range: 33∓609) from 38 studies in 14 countries for study periods when PCV7 was administered (Table 1). Study periods ranged from 2001, in the USA [19, 21, 38, 61, 72] until 2013 in Japan [24, 40, 66]. We excluded studies reporting <25% PCV coverage, but studies included serotype data from different uptake levels. For instance, from settings where PCV7 had been recently licensed, introduced into national immunisation programmes, or it was largely available through the private system, for instance, in Spain [16, 17, 22, 34, 52, 56, 57, 59, 60, 69], Portugal [12], and Austria [58]. Other studies were from settings where vaccine uptake was high or PCV was recommended universally in the study setting, such as in Canada [18, 26, 43], USA, UK, Colombia (Bogota) [55], France [13].
We analysed 20 publications from 24 countries where PCV10 or PCV13 have been introduced. The Americas region was the most represented (n = 12 countries). PCV10 was the primary vaccine in 6 countries (Brazil, Chile, Colombia, Ecuador, Peru, and the Netherlands), while PCV13 had been introduced in the remaining 18. Most study periods included the year of transition from PCV7 or the year after introduction of the current higher valent. PCV. A total of 5,469 IPD isolates with serotype data were included in meta-analysis. The number of isolates in individual studies ranged from 23 to 886. The countries with the largest number of isolates were Canada and South Africa (n = 886 and 839, respectively). Six publications reported data for fewer than 50 isolates and an additional 8 reported less than 100 cases.
Serotype distribution among paediatric IPD cases following PCV introduction
PCV-targeted serotypes
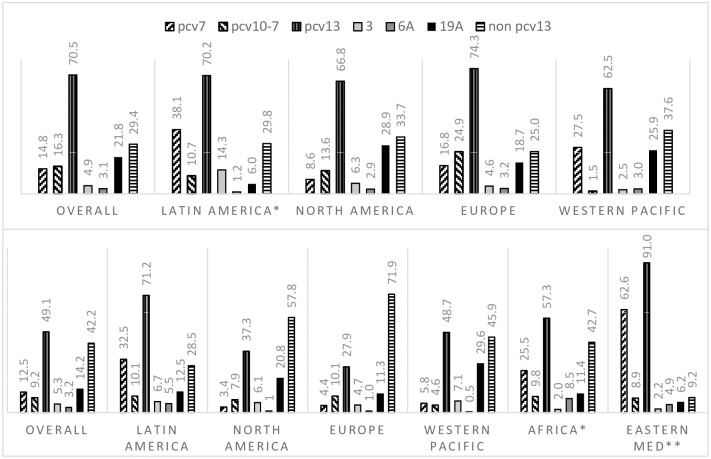
In studies where PCV7 was administered, serotypes included in the heptavalent conjugate vaccine accounted for an overall 14.8% (95%CI 11.4∓19.1) of childhood IPD cases. In studies with study periods after higher valent PCVs were introduced, the proportional contribution of these seven serotypes to IPD in children was 12.5% (95%CI 8.8∓17.7). In the latter, all countries, except the Netherlands, identified PCV7 types. PCV7 types were most frequently isolated from childhood IPD cases in Latin America, Africa, and Eastern Mediterranean regions (32.5%, 25.5%, 62.6% of IPD cases, respectively). Conversely, PCV7 types accounted for lower proportions of IPD in North America, Europe, and the Western Pacific, between 3.4∓5.8%, after introduction of PCV10 or PCV13. Fig 2 and Tables 2 and 3 show the overall meta-estimate and regional stratifications results per period.
Fig 2. Estimates of serotype-specific contributions to IPD in children (%, 95%CI) reported are based on random effects model from meta-analysis of 3 or more studies, unless indicated [one study (*) or two (**)].
Table 2. Serotype-specific contributions (%) to paediatric IPD cases by region in studies with PCV7 implementation.
| OVERALL (N = 38) |
LATIN AMERICA (N = 1) |
NORTH AMERICA (N = 13) |
EUROPE (N = 20) |
WESTERN PACIFIC (N = 4) |
|||
|---|---|---|---|---|---|---|---|
| PCV7 | 14.8 (11.4–19.1) | * | 38.1 (26.9–53.9) | 8.6 (4.9–15.2) | 16.8 (11.9–23.9) | 27.5 (19.8–38) | |
| PCV10-7 | 16.3 (13.5–19.8) | * | 10.7 (5.6–20.6) | 13.6 (10.8–17) | 24.9 (20.7–30) | 1.5 (0.8–2.9) | |
| PCV13 | 70.5 (67.6–73.5) | * | 70.2 (54.4–90.7) | 66.8 (62.3–71.6) | 74.3 (70.6–78.2) | 62.5 (54.9–71.3) | |
| 19A | 21.8 (18.6–25.6) | * | 6 (2.5–14.3) | 28.9 (23–36.4) | 18.7 (15.4–22.7) | 25.9 (17.7–37.9) | |
| 3 | 4.9 (4.2–5.8) | * | 14.3 (8.1–25.2) | 6.3 (5.1–7.8) | 4.6 (4–5.4) | 2.5 (1.4–4.6) | |
| 6A | 3.1 (2.4–4.1) | * | 1.2 (0.2–8.5) | 2.9 (1.5–5.6) | 3.2 (2.2–4.5) | 3 (2–4.5) | |
| NON PCV13 | 29.4 (26.8–32.4) | * | 29.8 (20.1–44) | 33.7 (29.4–38.7) | 24.9 (21.7–28.8) | 37.6 (30.1–46.9) | |
| 22F | 3.5 (2.9–4.3) | * | 1.2 (0.2–8.5) | 4.9 (4–6) | 2.2 (1.5–3.3) | 4 (2.8–5.6) | |
| 12F | 2.3 (1.8–2.9) | − | 3.1 (1.6–6) | 1.9 (1.5–2.4) | * | 0.3 (0–2.3) | |
| 33F | 3.4 (2.6–4.6) | − | 4.8 (3.7–6.3) | 3 (1.7–5.1) | 2.1 (1.3–3.4) | ||
| 24F | 4 (3.4–4.8) | * | 1.2 (0.2–8.5) | − | 3.3 (2.5–4.5) | ** | 5 (3.1–8) |
| 15C | 2.8 (2.3–3.4) | * | 2.4 (0.6–9.5) | 2.6 (1.7–3.8) | 2.2 (1.7–2.9) | 4.7 (3.3–6.7) | |
| 15B | 2.4 (2–3) | * | 3.6 (1.2–11.1) | 2.4 (1.6–3.5) | 2.3 (1.8–3) | ** | 2.8 (1.8–4.6) |
| 23B | 1.7 (1.4–2.1) | * | 1.2 (0.2–8.5) | 2 (1.4–2.8) | 1.5 (1.1–2) | − | |
| 10A | 2.6 (2–3.3) | − | 2.5 (1.7–3.5) | 2.5 (1.6–3.8) | 2.5 (1.2–5.4) | ||
| 38 | 2.3 (1.8–2.8) | − | 2.7 (1.8–4.2) | 1.9 (1.4–2.7) | 1.8 (1–3.2) | ||
| 15A | 1.8 (1.2–2.7) | * | 2.4 (0.6–9.5) | 1.5 (1–2.3) | 1.1 (0.8–1.6) | 7.1 (5–10.2) | |
| 35B | 1.2 (0.9–1.6) | * | 2.4 (0.6–9.5) | 1.5 (0.9–2.6) | 0.7 (0.4–1.1) | 1.6 (0.8–3.1) | |
| 6C | 2.8 (2.2–3.4) | − | 3.4 (2.4–4.8) | 1.1 (0.7–1.7) | ** | 4.2 (2.7–6.8) | |
| 8 | 1.4 (1.1–1.9) | * | 2.4 (0.6–9.5) | 0.9 (0.5–1.8) | 1.6 (1.1–2.2) | − | |
| 11A | 1.3 (1–1.8) | * | 2.4 (0.6–9.5) | 1.1 (0.6–2) | 1.2 (0.8–1.7) | ** | 1.8 (0.7–4.5) |
| 23A | 1.6 (1.2–2.1) | − | 1.8 (1.2–2.7) | 0.7 (0.4–1.2) | ** | 1.4 (0.2–9.1) | |
| 9N | 0.9 (0.6–1.2) | − | 0.7 (0.4–1.3) | 1 (0.6–1.7) | − | ||
Estimates of serotype-specific contributions to IPD in children (% 95%CI) reported are based on random effects model from meta-analysis of 3 or more studies, unless indicated [one study (*) or two (**)].–indicates no data reported. Meta-analyses were conducted individually, thus the sum of each of the serotypes in a category may exceed 100%
Table 3. Serotype-specific contributions (%) to paediatric IPD cases in settings that have introduced PCV10 or PCV13.
| OVERALL (N = 20) |
LATIN AMERICA (N = 1†) |
NORTH AMERICA (N = 4) |
EUROPE (N = 8) |
WESTERN PACIFIC (N = 4) |
AFRICA (N = 1) |
EASTERN MEDITERRANEAN (N = 2) |
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PCV7 | 12.5 (8.8–17.7) | 32.5 (25.5–41.3) | 3.4 (2.5–4.5) | 4.4 (3.3–5.7) | 5.8 (1.6–21.1) | * | 25.5 (22.3–29.2) | ** | 62.6 (46.3–84.7) | |||
| PCV10-7 | 9.2 (6.9–12.2) | 10.1 (5–20.4) | 7.9 (4.5–13.9) | 10.1 (6.6–15.4) | 4.6 (1.5–14) | * | 9.8 (7.9–12.2) | ** | 8.9 (1–81.8) | |||
| PCV13 | 49.1 (42.3–56.9) | 71.2 (65.9–77) | 37.3 (25.4–54.7) | 27.9 (19.9–39.1) | 48.7 (31.6–74.8) | * | 57.3 (52.4–62.6) | ** | 91 (78.2–100) | |||
| 19A | 14.2 (11.1–18.3) | 12.5 (7.7–20.2) | 20.8 (13.3–32.6) | 11.3 (7.6–17) | 29.6 (20.3–43.1) | * | 11.4 (9.3–13.9) | ** | 6.2 (3.4–11.2) | |||
| 3 | 5.3 (4.2–6.7) | 6.7 (4.7–9.7) | 6.1 (3.9–9.3) | 4.7 (3.6–6.1) | 7.1 (3.4–14.9) | * | 2 (1.3–3.2) | * | 2.2 (0.8–5.8) | |||
| 6A | 3.2 (2.1–4.9) | 5.5 (4.2–7.1) | ** | 1 (0.6–1.8) | 1 (0.5–2) | * | 0.5 (0.1–3.8) | * | 8.5 (6.8–10.8) | ** | 4.9 (2.2–11.3) | |
| NON-PCV13 | 42.2 (36.1–49.5) | 28.5 (23.4–34.7) | 57.8 (41.6–80.4) | 71.9 (63.1–82) | 45.9 (30.9–68.2) | * | 42.7 (38.5–47.4) | * | 9.2 (3.7–22.9) | |||
| 22F | 5.3 (4.2–6.7) | 2.3 (1.4–3.7) | 6.9 (4.5–10.4) | 5.2 (3.6–7.5) | 7.4 (4.6–12) | − | − | |||||
| 12F | 4.3 (3.5–5.3) | 4 (2.8–5.7) | ** | 3.3 (1.3–8.3) | 5.6 (4.4–7) | − | * | 4 (2.9–5.6) | * | 1.3 (0.2–9.1) | ||
| 33F | 4.5 (3.4–5.9) | ** | 2 (0.8–4.8) | 4.9 (2.2–10.8) | 4.9 (3.3–7.4) | 4.3 (2.5–7.5) | − | − | ||||
| 24F | 4.2 (2.6–6.8) | 2.4 (1.6–3.8) | − | 6.7 (3.8–11.9) | * | 3.9 (0.1–12.4) | − | − | ||||
| 15C | 4 (3.1–5) | 1.5 (0.8–2.7) | 3.9 (2.4–6.5) | 5.9 (4.4–7.8) | * | 4.3 (2.2–8.7) | − | − | ||||
| 15B | 3.7 (3.1–4.4) | 2.5 (1.5–4.3) | 4.2 (2.3–7.8) | 3.7 (2.6–5.2) | ** | 3.3 (1.7–6.7) | * | 4.3 (3.1–5.9) | − | |||
| 23B | 3.4 (2.6–4.3) | 2.4 (1.4–4.1) | 3 (2–4.4) | 4.1 (2.6–6.5) | ** | 3.9 (2.1–7.3) | − | * | 1.3 (0.2–9.1) | |||
| 10A | 3.4 (2.2–5.4) | 1.3 (0.8–2.3) | ** | 2.6 (1.7–3.8) | 6.7 (3.7–12.4) | ** | 3 (1.6–5.8) | − | − | |||
| 38 | 3.4 (2.4–4.8) | 1.8 (0.6–5.5) | ** | 5 (1.7–14.3) | 3.2 (1.8–5.6) | ** | 0.7 (0.2–2.6) | − | − | |||
| 15A | 2.9 (1.9–4.4) | 1.4 (0.8–2.5) | 3.3 (1.5–7.4) | 3.9 (1.9–7.9) | ** | 3 (0.2–38.9) | − | − | ||||
| 35B | 2.6 (1.8–3.8) | 1.2 (0.2–6.4) | 4.5 (1.7–11.4) | 1.4 (0.9–2.3) | ** | 3.9 (2.2–6.9) | − | − | ||||
| 6C | 2.4 (1.8–3) | 2.4 (1.6–3.7) | 2.3 (1.6–3.3) | 1.6 (0.7–3.6) | 3 (1.2–7.1) | − | − | |||||
| 8 | 2.2 (1.3–3.8) | 1.2 (0.6–2.3) | ** | 1.5 (0.9–2.5) | 2.9 (1.3–6.2) | * | 3.8 (1.2–11.9) | * | 8.3 (6.6–10.5) | * | 1.3 (0.2–9.1) | |
| 11A | 2 (1.6–2.6) | 2.2 (1.5–3.3) | ** | 2.5 (1.6–3.7) | 1.7 (1.1–2.6) | ** | 0.7 (0.2–2.6) | − | − | |||
| 23A | 2 (1.6–2.6) | 2 (1.2–3.6) | 2.2 (1.5–3.2) | 1.6 (1–2.5) | 2.4 (1.1–5) | − | * | 2.6 (0.6–10.2) | ||||
| 9N | 1.3 (1–1.8) | 1.3 (0.6–2.8) | ** | 1.3 (0.7–2.4) | 1.3 (0.7–2.4) | ** | 1.5 (0.5–4.7) | * | 1.8 (1.1–3) | − | ||
Estimates of serotype-specific contributions to IPD in children (% 95%CI) reported are based on random effects model from meta-analysis of 3 or more studies, unless indicated [one study (*) or two (**)].–indicates no data reported.
†10 countries from the SIREVA surveillance network. Meta-analyses were conducted individually, thus the sum of each of the serotypes in a category may exceed 100%
PCV10-specific serotypes
Serotypes 1, 5, and 7F were identified in all studies with periods of PCV7 implementation, accounting for an overall 16.3% (95%CI 13.5∓91.8) of childhood IPD cases. These serotypes comprised 9.2% (95%CI 6.9∓12.2) in settings where PCV10 or PCV13 have been introduced; which were reported in most countries (except France and Singapore). In this most recent period, the contributions to paediatric IPD cases due to PCV10-specific serotypes were similar in all regions (approximately 8∓9%), except in the Western Pacific (4.6%).
PCV13-specific serotypes
In both periods, the most frequent PCV13 type was 19A, followed by 3, and then 6A. Serotype 3 accounted for 5∓6% of childhood IPD cases after the introduction of either PCV, while 6A was associated with approximately 3% of all cases. In studies from countries where PCV10 or PCV13 have been introduced, serotype 19A accounted for 14.2% (95% CI 11.1∓18.3%) of childhood IPD cases across all regions (and at least 10% in each of the regions).
Non-PCV13
There was wide-variation in the number of non-PCV13 strains reported. In addition to the 16 non-PCV13 types included in meta-analysis, at least 1 case was reported for over 60 different serotypes currently absent in any of the PCV formulations (a list is provided in the S3 Table).
The overall contribution of non-PCV13 types to IPD in children was 29.4% (95%CI 26.8∓32.4) in periods of PCV7 administration and 42.2% (95%CI 36.1∓49.5) in studies where higher valent PCVs have been introduced. In each period, differences between the individual proportional contribution of non-PCV13 serotypes to childhood IPD were small and their overall ranking varied within regions (Tables 2 and 3). Based on data from countries where PCV10 or PCV13 have been introduced, 22F was the most common serotype (5% of overall childhood IPD cases analysed), followed by 12F, 15C, 24F, 33F (4% each). Combined, the leading non-PCV13 serotypes 22F, 12F, 33F accounted for 4∓16% of IPD cases in children across regions, except in the Eastern Mediterranean region. Notably, serotype 24F appears to be prevalent in Europe and Western Pacific regions, but not North America.
Contributions of serotypes to childhood IPD between periods
We compared the contribution of various serotypes to childhood IPD in the two sets of studies (PCV7 implementation and PCV10 or PCV13 introduction). We observed decreases in the percentage point differences in PCV7 types (-2.3%), PCV10-only strains (-7.1%) and 19A (-7.6%). Changes observed between periods for overall meta-estimates of non-PCV13 types were small. Comparisons between the regions with the longest history of PCV use (North America, Europe, and Western Pacific) show variations in the percentage point reductions in PCV13 serotypes among childhood IPD cases:-29.5%, -46.4%, and -13.8% in North America, Europe, and Western Pacific regions, respectively. Notably, studies from these regions reported a range of early and late periods of PCV7 use in individual study settings, while data from countries that have introduced higher valent PCVs were largely representative of the transition and early years of the vaccines. Non-PCV13 serotype 22F accounted individually for at least 5% of childhood IPD in North America, an increase of approximately 2 percentage points, compared to the post-PCV7 period. Similar changes in this serotype was also seen in Europe and Western Pacific (+3%). Other non-PCV13 serotypes with an increase in their proportional contribution to paediatric IPD after introduction of PCV10/13 differ between regions. For instance, serotype 38 (+2.7%) accounted for more childhood IPD cases in North America while 12F and 15C (+3.7%) and 10A (+4.2%) were more prominent in Europe.
Discussion
This is the first study to comprehensively report S. pneumoniae serotype-specific contributions to IPD in young children subsequent to the introduction of PCVs. Our estimates, based on a systematic analysis of data available to-date indicate that, in countries that have introduced higher-valent PCVs, approximately 42% (95%CI 36∓49) of childhood IPD cases were caused by non-PCV13 serotypes. Our results provide insights into the relative importance of serotypes in childhood IPD after widespread implementation of PCVs and highlight regional differences.
It is well documented that routine immunisation with PCVs has led to significant decreases in both colonisation and IPD by vaccine-targeted types in children and that it has indirectly impacted non-targeted populations [78]. It was estimated that PCV7-targeted serotypes accounted for 49∓82% of childhood IPD before PCV introduction [5]. We found that, after PCV7 implementation, their proportional contribution was approximately 11∓19%. This is consistent with other regional estimates, as prior to the introduction of PCV13 in Europe PCV7-type IPD was approximately 19% among children based on data from 26 countries [79]. Subsequent to the introduction of higher valent PCVs, the proportional contribution of serotypes targeted by PCVs continued to decrease, with differences across regions.
Concerns exist about optimum prevention of PCV13-types 3 and 19A−IPD in young children. We estimated that serotype 3−specific contribution to childhood IPD was approximately 5% in the two PCV periods analysed. These estimates are noteworthy, as serotype 3 has been associated with cases of vaccine failure [80]. Our meta-estimates also show the extent of 19A’s predominant contribution to childhood IPD in different world regions after the introduction of PCVs. Subsequent to the introduction of PCV7, 19A was consistently identified as the most frequent serotype associated with childhood IPD cases in the Americas, Europe, and Western Pacific regions (causing approximately 20% of cases). We also found an important contribution to childhood IPD cases of serotype 19A (14.2% 95%CI 11.1∓18.3) in the early years of higher valent PCVs. More recent data suggest different experiences regarding changes in the contribution of 19A at the country level. In the USA, significant reductions of IPD after 5 years of use of PCV13 have been driven by decreases in 19A [81]. However, in other settings in Europe, this serotype continues to pose challenges as a disease-causing strain despite widespread use of PCVs [82, 83]. Differences in vaccine schedules and catch-up campaigns between these settings, as well as time required to observe a decrease in disease due to vaccine-targeted strains are key factors that will influence the epidemiology of S. pneumoniae strains. The estimated contributions of serotypes to childhood IPD in this study emphasise the need for a better understanding of factors associated with disease due to vaccine types, especially as PCV10 and PCV13 continue to be administered worldwide.
Widespread use of PCVs has resulted in dramatic reductions of IPD associated with vaccine serotypes among young children in high-income countries in North America, Europe, and the Western Pacific [11, 51, 79]. Knowledge of the serotypes that are associated with IPD among the target population is important for the development of new vaccines that extend protection against non-PCV13, which could also affect other age groups. We found that subsequent to the introduction of higher valent PCVs in North America and Europe, approximately half of childhood IPD cases were due to serotypes for which there is no protection via immunisation. Serotypes likely to provide further reductions in disease are 22F, 33F, 15B, 38, and 35B (25% combined) in North America. In Europe, the leading disease-causing non-PCV13 strains were 12F, 10A, 24F, 22F, and 15C, accounting for 30% of IPD. Serotypes 12F, 22F, 24F, and 33F have been identified to have high invasive disease potentials [68, 84, 85]. Studies have also shown that non-PCV13 15B/C/A, 23B, 24F, 35B are not only important causes of IPD but also are common nasopharyngeal colonisers and have high prevalence of antibiotic resistance [86–90]. Considering the important contribution of non-PCV13 serotypes to childhood IPD in these settings, next generation PCVs with greater coverage will be needed to reduce the remaining burden of pneumococcal disease.
There is paucity of data on burden of pneumococcal disease in Latin American countries. Nevertheless, 1,500,000 cases and 28,000 deaths of IPD are estimated to occur annually in children less than 5 years of age in Latin America [91]. We analysed data from 10 countries in Latin America, all from SIREVA surveillance systems, of which 5 currently use PCV10. Compared with other regions, the proportions of IPD caused by vaccine types were highest in this region. The contribution of PCV7 serotypes for IPD was 10-fold higher in Latin America than in the North America. Given the predominant role of vaccine types, the expansion of immunisation programmes with PCVs and surveillance of epidemiological changes is critical for effectively reducing the burden of pneumococcal disease in Latin American countries.
Before PCVs became available, the highest proportion of deaths (60%) associated with childhood IPD were estimated to occur in Africa and Asia [1]. A limited number of countries in the South East Asian and Mediterranean region have introduced PCVs and are evaluating their impact [92]. A recent systematic review of serotypes associated with IPD found that 1, 14, and 19F were common in South Asian countries. Further, serotypes varied across countries and there was low prevalence of serotype 19A [93]. We only included serotype data for two countries from two of these regions—South Africa and Saudi Arabia. In South Africa, serotype 8 was the most frequent serotype in childhood IPD post-PCV. This is in contrast to its position during the pre-PCV era in Africa, when its contribution was approximately 1% of IPD, ranking 8th [5]. Thus, our overall pooled estimates for the so-called African and Eastern Mediterranean regions in this study should be interpreted with caution. Our review of the literature shows that evidence of the effects of PCV in countries in Asia, Africa, and Eastern Mediterranean is urgently needed to further understand the role of serotypes following PCV10 and PCV13 introduction in settings with high burden of S. pneumoniae disease.
Serotype-specific meta-analysis, as performed in this study, provides evidence on potential serotypes of interest in the post-PCV era. However, changes in the proportional contribution of different strains to childhood IPD need to be interpreted carefully and in the context of changes in IPD incidence in a particular setting. For example, if the percentage of isolates for a serotype doubled, but the IPD decreased by 50%, it would indicate that there was no change in the absolute number of cases for particular serotypes. Without incidence data, we are unable to indicate if there has been a change in the absolute number of IPD cases in any given serotype in the post-PCV era or to account for varying factors, such as temporal changes in the study population and vaccine utilisation [11]. Yet, evidence in this review and meta-estimates adds to the information base about serotypes’ individual importance in IPD and emphasise the need for closer attention to regional differences in the post-PCV era.
Limitations
This study is not exempt from limitations of systematic reviews and meta-analysis. For some studies, case counts by serotype were not exact as we extracted data from images or calculated them from proportions. In other instances, serotype data were only available in a grouped format (e.g. “other”) or not all IPD cases were serotyped/reported. Missing information is likely to affect serotypes considered “rare” or “infrequent”, which will vary in each setting. These issues can potentially introduce biases to our results since not all serotypes could be assessed individually. Our meta-estimates could also be influenced by the differences across studies and are not exempt from risk of under or over-estimation. Although we sought to include large studies, datasets were heterogeneous, reported different sample sizes, observed diverse populations with varying rates of vaccine coverage, immunisation schedules, and methods for case detection of IPD (either by clinical and testing practices). Additionally, analyses were conducted individually per serotype, thus the sum of serotypes into categories may exceed 100%. Limitations withstanding, the large number of countries represented and isolates analysed is a strength of our review. We aimed to address issues of heterogeneity by analysing the most comparable data and case definitions. We also stratified analysis by PCV formulation and used data for children of the same age (<5 years) whenever possible. We would recommend future studies assessing serotype distribution after PCV introduction to report a clear definition and description of vaccine coverage in order contextualise results and better understand vaccine impact.
Conclusion
In the post-PCV era, childhood IPD is associated with a wide number of serotypes. After PCV7 and in the early years after introduction of higher valent PCVs, 19A was the most commonly identified serotype in different world regions. Non-PCV13 serotypes caused a considerable proportion of childhood IPD, which emphasises the need for new vaccines with additional serotypes to reduce the remaining burden of childhood pneumococcal disease. Data on serotypes causing IPD from the regions with the highest burden were not available to draw robust regional conclusions. The geographic diversity of serotypes and changing epidemiology of S. pneumoniae underscores the importance of continued surveillance of pneumococcal serotypes to guide vaccine recommendations.
Supporting information
(DOC)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Acknowledgments
We thank Marshall Dozier, medical librarian, and Claire Parker from the University of Edinburgh for their support in the development of search strategies and management of this study, respectively.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
This study was financially supported by Sanofi Pasteur. MK is an employee of Sanofi Pasteur and had a critical role in study design, data collection and analysis and preparation of the manuscript.
References
- 1.O'Brien KL, Wolfson LJ, Watt JP, Henkle E, Deloria-Knoll M, McCall N, et al. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: global estimates. Lancet. 2009;374(9693):893–902. 10.1016/S0140-6736(09)61204-6 [DOI] [PubMed] [Google Scholar]
- 2.World Health Organization. Estimated Hib and pneumococcal deaths for children under 5 years of age, 2008 2008 [cited 2016 28 March]. http://www.who.int/immunization/monitoring_surveillance/burden/estimates/Pneumo_hib/en/.
- 3.World Health Organization. WHO vaccine-preventable diseases: monitoring system. 2015 global summary 2015. http://apps.who.int/immunization_monitoring/globalsummary/schedules.
- 4.International Vaccine Access Center (IVAC), Johns Hopkins Bloomberg School of Public Health. Vaccine Information Management System (VIMS) Global Vaccine Introduction Report 2015. www.jhsph.edu/ivac/vims.html.
- 5.Johnson HL, Deloria-Knoll M, Levine OS, Stoszek SK, Hance LF, Reithinger R, et al. Systematic evaluation of serotypes causing invasive pneumococcal disease among children under five: The Pneumococcal Global Serotype Project. PLos Medicine. 2010;7(10). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pilishvili T, Lexau C, Farley MM, Hadler J, Harrison LH, Bennett NM, et al. Sustained reductions in invasive pneumococcal disease in the era of conjugate vaccine. Journal of Infectious Diseases. 2010;201(1):32–41. 10.1086/648593 [DOI] [PubMed] [Google Scholar]
- 7.Miller E, Andrews NJ, Waight PA, Slack MP, George RC. Herd immunity and serotype replacement 4 years after seven-valent pneumococcal conjugate vaccination in England and Wales: an observational cohort study. Lancet Infectious Diseases. 2011;11(10):760–68. 10.1016/S1473-3099(11)70090-1 [DOI] [PubMed] [Google Scholar]
- 8.Weinberger DM, Malley R, Lipsitch M. Serotype replacement in disease after pneumococcal vaccination. Lancet. 2011;378(9807):1962–73. 10.1016/S0140-6736(10)62225-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fleming-Dutra KE, Conklin L, Loo JD, Knoll MD, Park DE, Kirk J, et al. Systematic review of the effect of pneumococcal conjugate vaccine dosing schedules on vaccine-type nasopharyngeal carriage. The Pediatric infectious disease journal. 2014;33 Suppl 2:S152–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Loo JD, Conklin L, Fleming-Dutra KE, Knoll MD, Park DE, Kirk J, et al. Systematic review of the indirect effect of pneumococcal conjugate vaccine dosing schedules on pneumococcal disease and colonization. The Pediatric infectious disease journal. 2014;33 Suppl 2:S161–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Feikin DR, Kagucia EW, Loo JD, Link-Gelles R, Puhan MA, Cherian T, et al. Serotype-specific changes in invasive pneumococcal disease after pneumococcal conjugate vaccine introduction: a pooled analysis of multiple surveillance sites. PLoS Medicine. 2013;10(9):e1001517 10.1371/journal.pmed.1001517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Aguiar SI, Serrano I, Pinto FR, Melo-Cristino J, Ramirez M. Changes in Streptococcus pneumoniae serotypes causing invasive disease with non-universal vaccination coverage of the seven-valent conjugate vaccine. Clinical Microbiology and Infection. 2008;14(9):835–43. 10.1111/j.1469-0691.2008.02031.x [DOI] [PubMed] [Google Scholar]
- 13.Alexandre C, Dubos F, Courouble C, Pruvost I, Varon E, Martinot A. Rebound in the incidence of pneumococcal meningitis in northern France: Effect of serotype replacement. Acta Paediatrica, International Journal of Paediatrics. 2010;99(11):1686–90. [DOI] [PubMed] [Google Scholar]
- 14.Al-Sheikh YA, G L K, Mohammed Ali MM, John J, Khaled Homoud Mohammed D, Chikkabidare Shashidhar P. Distribution of serotypes and antibiotic susceptibility patterns among invasive pneumococcal diseases in Saudi Arabia.[Erratum appears in Ann Lab Med. 2014 Sep;34(5):411 Note: A Al-Sherikh, Yazeed [corrected to Al-Sheikh, Yazeed A]]. Annals of Laboratory Medicine. 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Angoulvant F, Levy C, Grimprel E, Varon E, Lorrot M, Biscardi S, et al. Early impact of 13-valent pneumococcal conjugate vaccine on community-acquired pneumonia in children. Clinical Infectious Diseases. 2014;58(7):918–24. 10.1093/cid/ciu006 [DOI] [PubMed] [Google Scholar]
- 16.Aristegui J, Bernaola E, Pocheville I, Garcia C, Arranz L, Duran G, et al. Reduction in pediatric invasive pneumococcal disease in the Basque Country and Navarre, Spain, after introduction of the heptavalent pneumococcal conjugate vaccine. European Journal of Clinical Microbiology and Infectious Diseases. 2007;26(5):303–10. 10.1007/s10096-007-0294-4 [DOI] [PubMed] [Google Scholar]
- 17.Barricarte A, Castilla J, Gil-Setas A, Torroba L, Navarro-Alonso JA, Irisarri F, et al. Effectiveness of the 7-valent pneumococcal conjugate vaccine: a population-based case-control study. Clinical Infectious Diseases. 2007;44:1436–41. 10.1086/516779 [DOI] [PubMed] [Google Scholar]
- 18.Bettinger JA, Scheifele DW, Kellner JD, Halperin SA, Vaudry W, Law B, et al. The effect of routine vaccination on invasive pneumococcal infections in Canadian children, Immunization Monitoring Program, Active 2000–2007. Vaccine. 2010;28(9):2130–6. 10.1016/j.vaccine.2009.12.026 [DOI] [PubMed] [Google Scholar]
- 19.Black S, France EK, Isaacman D, Bracken L, Lewis E, Hansen J, et al. Surveillance for invasive pneumococcal disease during 2000–2005 in a population of children who received 7-valent pneumococcal conjugate vaccine. Pediatric Infectious Disease Journal. 2007;26(9):771–7. 10.1097/INF.0b013e318124a494 [DOI] [PubMed] [Google Scholar]
- 20.Bruce MG, Singleton R, Bulkow L, Rudolph K, Zulz T, Gounder P, et al. Impact of the 13-valent pneumococcal conjugate vaccine (pcv13) on invasive pneumococcal disease and carriage in Alaska. Vaccine. 2015;33(38):4813–9. 10.1016/j.vaccine.2015.07.080 [DOI] [PubMed] [Google Scholar]
- 21.Byington CL, Samore MH, Stoddard GJ, Barlow S, Daly J, Korgenski K, et al. Temporal trends of invasive disease due to Streptococcus pneumoniae among children in the Intermountain West: Emergence of nonvaccine serogroups. Clinical Infectious Diseases. 2005;41(1):21–9. 10.1086/430604 [DOI] [PubMed] [Google Scholar]
- 22.Calbo E, Diaz A, Canadell E, Fabrega J, Uriz S, Xercavins M, et al. Invasive pneumococcal disease among children in a health district of Barcelona: Early impact of pneumococcal conjugate vaccine. Clinical Microbiology and Infection. 2006;12(9):867–72. 10.1111/j.1469-0691.2006.1502_1.x [DOI] [PubMed] [Google Scholar]
- 23.Ceyhan M, Gurler N, Yaman A, Ozturk C, Oksuz L, Ozkan S, et al. Serotypes of Streptococcus pneumoniae isolates from children with invasive pneumococcal disease in Turkey: Baseline evaluation of the introduction of the pneumococcal conjugate vaccine nationwide. Clinical and Vaccine Immunology. 2011;18(6):1028–30. 10.1128/CVI.00526-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chiba N, Morozumi M, Shouji M, Wajima T, Iwata S, Ubukata K. Changes in capsule and drug resistance of pneumococci after introduction of PCV7, Japan, 2010–2013. Emerging Infectious Diseases. 2014;20(7):1132–9. 10.3201/eid2007.131485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Croney CM, Coats MT, Nahm MH, Briles DE, Craina MJ. PspA family distribution, unlike capsular serotype, remains unaltered following introduction of the heptavalent pneumococcal conjugate vaccine. Clinical and Vaccine Immunology. 2012;19(6):891–6. 10.1128/CVI.05671-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.De Wals P, Lefebvre B, Defay F, Deceuninck G, Boulianne N. Invasive pneumococcal diseases in birth cohorts vaccinated with PCV-7 and/or PHiD-CV in the province of Quebec, Canada. Vaccine. 2012;30(45):6416–20. 10.1016/j.vaccine.2012.08.017 [DOI] [PubMed] [Google Scholar]
- 27.del Amo E, Brotons P, Monsonis M, Trivino M, Inigo M, Selva L, et al. High invasiveness of pneumococcal serotypes included in the new generation of conjugate vaccines. Clinical Microbiology & Infection. 2014;20(684–89). [DOI] [PubMed] [Google Scholar]
- 28.del Amo E, Selva L, de Sevilla MF, Ciruela P, Brotons P, Trivino M, et al. Estimation of the invasive disease potential of Streptococcus pneumoniae in children by the use of direct capsular typing in clinical specimens. European Journal of Clinical Microbiology and Infectious Diseases. 2015;34(4):705–11. 10.1007/s10096-014-2280-y [DOI] [PubMed] [Google Scholar]
- 29.Demczuk WH, Martin I, Griffith A, Lefebvre B, McGeer A, Lovgren M, et al. Serotype distribution of invasive Streptococcus pneumoniae in Canada after the introduction of the 13-valent pneumococcal conjugate vaccine, 2010–2012. Canadian Journal of Microbiology. 2013;59:778–88. 10.1139/cjm-2013-0614 [DOI] [PubMed] [Google Scholar]
- 30.Desai AP, Sharma D, Crispell EK, Baughman W, Thomas S, Tunali A, et al. Decline in pneumococcal nasopharyngeal carriage of vaccine serotypes after the introduction of the 13-valent pneumococcal conjugate vaccine in children in Atlanta, Georgia. Pediatric Infectious Disease Journal. 2015;34(11):1168–74. 10.1097/INF.0000000000000849 [DOI] [PubMed] [Google Scholar]
- 31.Doit C, Mariani-Kurkdjian P, Mahjoub-Messai F, Bidet P, Bonacorsi S, Carol A, et al. Epidemiology of pediatric community-acquired bloodstream infections in a children hospital in Paris, France, 2001 to 2008. Diagnostic Microbiology and Infectious Disease. 2010;66(3):332–5. 10.1016/j.diagmicrobio.2009.10.012 [DOI] [PubMed] [Google Scholar]
- 32.Foster D, Walker AS, Paul J, Griffiths D, Knox K, Peto TE, et al. Reduction in invasive pneumococcal disease following implementation of the conjugate vaccine in the Oxfordshire region, England. Journal of Medical Microbiology. 2011;60(1):91–7. [DOI] [PubMed] [Google Scholar]
- 33.Guevara M, Ezpeleta C, Gil-Setas A, Torroba L, Beristain X, Aguinaga A, et al. Reduced incidence of invasive pneumococcal disease after introduction of the 13-valent conjugate vaccine in Navarre, Spain, 2001–2013. Vaccine. 2014;32(22):2553–62. 10.1016/j.vaccine.2014.03.054 [DOI] [PubMed] [Google Scholar]
- 34.Herrero M, Alcalde M, Gomez B, Hernandez JL, Sota M, Benito J, et al. Invasive bacterial infections in a paediatric emergency department in the era of the heptavalent pneumococcal conjugate vaccine. European Journal of Emergency Medicine. 2012;19(2):89–94. 10.1097/MEJ.0b013e3283484bbc [DOI] [PubMed] [Google Scholar]
- 35.Herz AM, Greenhow TL, Alcantara J, Hansen J, Baxter RP, Black SB, et al. Changing epidemiology of outpatient bacteremia in 3- to 36-month-old children after the introduction of the heptavalent-conjugated pneumococcal vaccine. Pediatric Infectious Disease Journal. 2006;25(4):293–300. 10.1097/01.inf.0000207485.39112.bf [DOI] [PubMed] [Google Scholar]
- 36.Hicks LA, Harrison LH, Flannery B, Hadler JL, Schaffner W, Craig AS, et al. Incidence of pneumococcal disease due to non- pneumococcal conjugate vaccine (PCV7) serotypes in the United States during the era of widespread PCV7 vaccination, 1998–2004. Journal of Infectious Diseases. 2007;196(9):1346–54. 10.1086/521626 [DOI] [PubMed] [Google Scholar]
- 37.Hsu K, Pelton S, Karumuri S, Heisey-Grove D, Klein J. Population-based surveillance for childhood invasive pneumococcal disease in the era of conjugate vaccine. Pediatric Infectious Disease Journal. 2005;24(1):17–23. [DOI] [PubMed] [Google Scholar]
- 38.Hsu KK, Shea KM, Stevenson AE, Pelton SI. Changing serotypes causing childhood invasive pneumococcal disease: Massachusetts, 2001–2007. Pediatric Infectious Disease Journal. 2010;29(4):289–93. [DOI] [PubMed] [Google Scholar]
- 39.Imohl M, Moller J, Perniciaro S, van der Linden M, Aktas O. Pneumococcal meningitis and vaccine effects in the era of conjugate vaccination: Results of 20 years of nationwide surveillance in Germany. BMC Infectious Diseases. 2015;15(1). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ishiwada N, Hishiki H, Nagasawa K, Naito S, Sato Y, Chang B, et al. The incidence of pediatric invasive Haemophilus influenzae and pneumococcal disease in Chiba prefecture, Japan before and after the introduction of conjugate vaccines. Vaccine. 2014;32(42):5425–31. 10.1016/j.vaccine.2014.07.100 [DOI] [PubMed] [Google Scholar]
- 41.Kaplan SL, Barson WJ, Lin PL, Romero JR, Bradley JS, Tan TQ, et al. Early trends for invasive pneumococcal infections in children after the introduction of the 13-valent pneumococcal conjugate vaccine. Pediatric Infectious Disease Journal. 2013;32(3):203–7. 10.1097/INF.0b013e318275614b [DOI] [PubMed] [Google Scholar]
- 42.Kaplan SL, Barson WJ, Lin PL, Stovall SH, Bradley JS, Tan TQ, et al. Serotype 19A is the most common serotype causing invasive pneumococcal infections in children. Pediatrics. 2010;125(3):429–36. 10.1542/peds.2008-1702 [DOI] [PubMed] [Google Scholar]
- 43.Kellner JD, Vanderkooi OG, MacDonald J, Church DL, Tyrrell GJ, Scheifele DW. Changing epidemiology of invasive pneumococcal disease in Canada, 1998–2007: Update from the calgary-area Streptococcus pneumoniae research (Casper) study. Clinical Infectious Diseases. 2009;49(2):205–12. 10.1086/599827 [DOI] [PubMed] [Google Scholar]
- 44.Knol MJ, Wagenvoort GHJ, Sanders EAM, Elberse K, Vlaminckx BJ, de Melker HE, et al. Invasive pneumococcal disease 3 years after introduction of 10-valent pneumococcal conjugate vaccine, the Netherlands. Emerging Infectious Diseases. 2015;21(11):2040–4. 10.3201/eid2111.140780 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lepoutre A, Varon E, Georges S, Dorleans F, Janoir C, Gutmann L, et al. Impact of the pneumococcal conjugate vaccines on invasive pneumococcal disease in France, 2001–2012. Vaccine. 2015;33(2):359–66. 10.1016/j.vaccine.2014.11.011 [DOI] [PubMed] [Google Scholar]
- 46.Lepoutre A, Varon E, Georges S, Gutmann L, Levy-Bruhl D. Impact of infant pneumococcal vaccination on invasive pneumococcal diseases in France, 2001–2006. Euro surveillance. 2008;13(35). [DOI] [PubMed] [Google Scholar]
- 47.Levy C, Varon E, Bingen E, Lecuyer A, Boucherat M, Cohen R. Pneumococcal meningitis in French children before and after the introduction of pneumococcal conjugate vaccine. Pediatric Infectious Disease Journal. 2011;30(2):168–70. [DOI] [PubMed] [Google Scholar]
- 48.Levy C, Varon E, Picard C, Bechet S, Martinot A, Bonacorsi S, et al. Trends of pneumococcal meningitis in children after introduction of the 13-valent pneumococcal conjugate vaccine in France. Pediatric Infectious Disease Journal. 2014;33(12):1216–21. 10.1097/INF.0000000000000451 [DOI] [PubMed] [Google Scholar]
- 49.Mistry RD, Wedin T, Balamuth F, McGowan KL, Ellison AM, Nelson KA, et al. Emergency department epidemiology of pneumococcal bacteremia in children since the institution of widespread PCV7 vaccination. Journal of Emergency Medicine. 2013;45(6):813–20. 10.1016/j.jemermed.2013.04.029 [DOI] [PubMed] [Google Scholar]
- 50.Moore CE, Paul J, Foster D, Mahar SA, Griffiths D, Knox K, et al. Reduction of invasive pneumococcal disease 3 years after the introduction of the 13-valent conjugate vaccine in the Oxfordshire region of England. Journal of Infectious Diseases. 2014;210(7):1001–11. 10.1093/infdis/jiu213 [DOI] [PubMed] [Google Scholar]
- 51.Moore MR, Link-Gelles R, Schaffner W, Lynfield R, Lexau C, Bennett NM, et al. Effect of use of 13-valent pneumococcal conjugate vaccine in children on invasive pneumococcal disease in children and adults in the USA: analysis of multisite, population-based surveillance. Lancet Infectious Diseases. 2015;15(3):301–9. 10.1016/S1473-3099(14)71081-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Munoz-Almagro C, Ciruela P, Esteva C, Marco F, Navarro M, Bartolome R, et al. Serotypes and clones causing invasive pneumococcal disease before the use of new conjugate vaccines in Catalonia, Spain. Journal of Infection. 2011;63(2):151–62. 10.1016/j.jinf.2011.06.002 [DOI] [PubMed] [Google Scholar]
- 53.Nakano S, Fujisawa T, Ito Y, Chang B, Suga S, Noguchi T, et al. Serotypes, antimicrobial susceptibility, and molecular epidemiology of invasive and non-invasive Streptococcus pneumoniae isolates in paediatric patients after the introduction of 13-valent conjugate vaccine in a nationwide surveillance study conducted in Japan in 2012–2014. Vaccine. 2016;34(1):67–76. 10.1016/j.vaccine.2015.11.015 [DOI] [PubMed] [Google Scholar]
- 54.Olarte L, Barson WJ, Barson RM, Lin PL, Romero JR, Tan TQ, et al. Impact of the 13-valent pneumococcal conjugate vaccine on pneumococcal meningitis in us children. Clinical Infectious Diseases. 2015;61(5):767–75. 10.1093/cid/civ368 [DOI] [PubMed] [Google Scholar]
- 55.Parra EL, De La Hoz F, Diaz PL, Sanabria O, Realpe ME, Moreno J. Changes in Streptococcus pneumoniae serotype distribution in invasive disease and nasopharyngeal carriage after the heptavalent pneumococcal conjugate vaccine introduction in Bogota, Colombia. Vaccine. 2013;31(37):4033–8. 10.1016/j.vaccine.2013.04.074 [DOI] [PubMed] [Google Scholar]
- 56.Perez-Trallero E, Marimon JM, Ercibengoa M, Vicente D, Perez-Yarza EG. Invasive Streptococcus pneumoniae infections in children and older adults in the north of Spain before and after the introduction of the heptavalent pneumococcal conjugate vaccine. European Journal of Clinical Microbiology and Infectious Diseases. 2009;28(7):731–8. 10.1007/s10096-008-0693-1 [DOI] [PubMed] [Google Scholar]
- 57.Picazo J, Ruiz-Contreras J, Casado-Flores J, Giangaspro E, Del Castillo F, Hernandez-Sampelayo T, et al. Relationship between serotypes, age, and clinical presentation of invasive pneumococcal disease in Madrid, Spain, after introduction of the 7-valent pneumococcal conjugate vaccine into the vaccination calendar. Clinical and Vaccine Immunology. 2011;18(1):89–94. 10.1128/CVI.00317-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Rendi-Wagner P, Paulke-Korinek M, Kundi M, Burgmann H, Georgopoulos A, Vecsei A, et al. National paediatric immunization program of high risk groups: No effect on the incidence of invasive pneumococcal diseases. Vaccine. 2009;27(30):3963–8. 10.1016/j.vaccine.2009.04.044 [DOI] [PubMed] [Google Scholar]
- 59.Rodriguez MA, Gonzalez AV, Gavin MA, Martinez FM, Marin NG, Blazquez BR, et al. Invasive pneumococcal disease: association between serotype, clinical presentation and lethality. Vaccine. 2011;29(34):5740–46. 10.1016/j.vaccine.2011.05.099 [DOI] [PubMed] [Google Scholar]
- 60.Salleras L, Dominguez A, Ciruela P, Izquierdo C, Navas E, Torner N, et al. Changes in serotypes causing invasive pneumococcal disease (2005–2007 vs. 1997–1999) in children under 2 years of age in a population with intermediate coverage of the 7-valent pneumococcal conjugated vaccine. Clinical Microbiology & Infection. 2009;15(11):997–1001. [DOI] [PubMed] [Google Scholar]
- 61.Schutze GE, Tucker NC, Mason EO Jr. Impact of the conjugate pneumococcal vaccine in Arkansas. Pediatric Infectious Disease Journal. 2004;23(12):1125–9. [PubMed] [Google Scholar]
- 62.Sharma D, Baughman W, Holst A, Thomas S, Jackson D, Da Gloria Carvalho M, et al. Pneumococcal carriage and invasive disease in children before introduction of the 13-valent conjugate vaccine: Comparison with the era before 7-valent conjugate vaccine. Pediatric Infectious Disease Journal. 2013;32(2):e45–e53. 10.1097/INF.0b013e3182788fdd [DOI] [PubMed] [Google Scholar]
- 63.Shibl AM, Memish ZA, Al-Kattan KM. Antibiotic resistance and serotype distribution of invasive pneumococcal diseases before and after introduction of pneumococcal conjugate vaccine in the Kingdom of Saudi Arabia (KSA). Vaccine. 2012;30(SUPPL. 6):G32–G6. [DOI] [PubMed] [Google Scholar]
- 64.SIREVA II. Informe Regional de SIREVA II, 2009–12: Datos por país y por grupos de edad sobre las características de los aislamientos de Streptococcus pneumoniae, Haemophilus influenzae y neisseria meningitidis, en procesos invasores SIREVA II Regional Report, 2009–12: Data by country and by age group on the characteristics of isolates of Streptococcus pneumoniae, Haemophilus influenzae and Neisseria meningitidis in invasive processes 2012. http://new.paho.org/hq/index.php?option=com_docman&task=doc_view&gid=21402&Itemid=.
- 65.Steens A, Bergsaker MAR, Aaberge IS, Ronning K, Vestrheim DF. Prompt effect of replacing the 7-valent pneumococcal conjugate vaccine with the 13-valent vaccine on the epidemiology of invasive pneumococcal disease in Norway. Vaccine. 2013;31(52):6232–8. 10.1016/j.vaccine.2013.10.032 [DOI] [PubMed] [Google Scholar]
- 66.Suga S, Chang B, Asada K, Akeda H, Nishi J, Okada K, et al. Nationwide population-based surveillance of invasive pneumococcal disease in Japanese children: Effects of the seven-valent pneumococcal conjugate vaccine. Vaccine. 2015;33(45):6054–60. 10.1016/j.vaccine.2015.07.069 [DOI] [PubMed] [Google Scholar]
- 67.Van Der Linden M, Falkenhorst G, Perniciaro S, Imohl M. Effects of infant pneumococcal conjugate vaccination on serotype distribution in invasive pneumococcal disease among children and adults in Germany. PLoS ONE. 2015;10(7). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Varon E, Cohen R, Bechet S, Doit C, Levy C. Invasive disease potential of pneumococci before and after the 13-valent pneumococcal conjugate vaccine implementation in children. Vaccine. 2015;33(46):6178–85. 10.1016/j.vaccine.2015.10.015 [DOI] [PubMed] [Google Scholar]
- 69.Vila-Corcoles A, Ochoa-Gondar O, Guzman-Avalos A, Gomez-Bertomeu F, Figuerola-Massana E, Raga-Luria X, et al. Incidence of pneumococcal infections among children under 15 years in southern Catalonia throughout the heptavalent conjugate vaccine era, 2002–2009. Infection. 2013;41(2):439–46. 10.1007/s15010-012-0345-3 [DOI] [PubMed] [Google Scholar]
- 70.Von Gottberg A, De Gouveia L, Tempia S, Quan V, Meiring S, Von Mollendorf C, et al. Effects of vaccination on invasive pneumococcal disease in South Africa. New England Journal of Medicine. 2014;371(20):1889–99. 10.1056/NEJMoa1401914 [DOI] [PubMed] [Google Scholar]
- 71.Waight PA, Andrews NJ, Ladhani SN, Sheppard CL, Slack MP, Miller E. Effect of the 13-valent pneumococcal conjugate vaccine on invasive pneumococcal disease in England and Wales 4 years after its introduction: an observational cohort study. Lancet Infectious Diseases. 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Weatherholtz R, Millar EV, Moulton LH, Reid R, Rudolph K, Santosham M, et al. Invasive pneumococcal disease a decade after pneumococcal conjugate vaccine use in an American Indian population at high risk for disease. CID. 2010. [DOI] [PubMed] [Google Scholar]
- 73.Williams SR, Mernagh PJ, Lee MHT, Tan JT. Changing epidemiology of invasive pneumococcal disease in Australian children after introduction of a 7-valent pneumococcal conjugate vaccine. Medical Journal of Australia. 2011;194(3):116–20. [DOI] [PubMed] [Google Scholar]
- 74.Australian Goverment Department of Health. Invasive Pneumococcal Disease Surveillance Australia: National Notifiable Diseases Surveillance; 2012 [cited 2016 January 15]. http://www.health.gov.au/internet/main/publishing.nsf/Content/cda-surveil-nndss-ipd-reports.htm.
- 75.Health Protection Scotland. Pneumococcal Disease 2015 [cited 2016 January 15]. http://www.hps.scot.nhs.uk/resp/pneumococcaldisease.aspx.
- 76.New Zealand Ministry of Health. Invasive Pneumococcal Disease Reports 2015 [cited 2016 January 15]. https://surv.esr.cri.nz/surveillance/IPD.php.
- 77.Ministry of Health Singapore. Epidemiological News Bulletin Laboratory data on surveillance of invasive pneumococcal diseases in Singapore 2012–14. https://www.moh.gov.sg/content/dam/moh_web/Statistics/Epidemiological_News_Bulletin/2013/ENB02Q_13.pdf https://www.moh.gov.sg/content/dam/moh_web/Publications/Reports/2014/Communicable%20Diseases%20Surveillance%20in%20Singapore%202013/Full%20version.pdf https://www.moh.gov.sg/content/dam/moh_web/Publications/Reports/2015/Air%20Droplet-Borne%20Diseases.pdf.
- 78.Hausdorff WP, Hanage WP. Interim results of an ecological experiment—Conjugate vaccination against the pneumococcus and serotype replacement. Human Vaccines Immunotherapies. 2016;12(2):358–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Navarro Torne A, Dias JG, Quinten C, Hruba F, Busana MC, Lopalco PL, et al. European enhanced surveillance of invasive pneumococcal disease in 2010: Data from 26 European countries in the post-heptavalent conjugate vaccine era. Vaccine. 2014;32(29):3644–50. 10.1016/j.vaccine.2014.04.066 [DOI] [PubMed] [Google Scholar]
- 80.Oligbu G, Collins S, Andrews N, Sheppard C, Fry N, Slack PEM, et al., editors. Pneumococcal Conjugate Vaccine Failure in children younger than 5 years old in England and Wales, 2006–14. 10th International Symposium on Pneumococci & Pneumococcal Diseases; 2016; Glasgow, UK.
- 81.Pilishvili T, Gierke R, Farley M, Schaffner W, Thomas A, Reingold A, et al., editors. Changes in invasive pneumococcal disease (IPD) following 5 years of 13-valent pneumococcal conjugate vaccine in the U.S. 10th International Symposium on Pneumococci & Pneumococcal Diseases; 2016; Glasgow, UK.
- 82.Fry N, Kapatai G, Sheppard C, Litt D, Collins S, Ladhani S, et al., editors. The fall and rise of serotype 19A in invasive pneumococcal disease: application of whole genome sequencing to investigate the recent rise in England and Wales. 10th International Symposium on Pneumococci & Pneumococcal Diseases; 2016; Glasgow, UK.
- 83.Corcoran M, Vickers I, Fitzgerald M, Mereckiene J, Murchan S, Cotter S, et al., editors. The persistence of serotype 19A—Despite the introduction of PCV13 vaccine. 10th International Symposium on Pneumococci & Pneumococcal Diseases; 2016; Glasgow, UK.
- 84.Brueggemann AB, Peto TE, Crook DW, Butler JC, Kristinsson KG, Spratt BG. Temporal and geographic stability of the serogroup-specific invasive disease potential of Streptococcus pneumoniae in children. 2004. [DOI] [PubMed] [Google Scholar]
- 85.Yildirim I, Hanage WP, Lipsitch M, Shea KM, Stevenson A, Finkelstein J, et al. Serotype specific invasive capacity and persistent reduction in invasive pneumococcal disease. Vaccine. 2010;29(2):283–8. 10.1016/j.vaccine.2010.10.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Lee GM, Kleinman K, Pelton SI, Hanage W, Huang SS, Lakoma M, et al. Impact of 13-valent pneumococcal conjugate vaccination on Streptococcus pneumoniae carriage in young children in Massachusetts. Journal of the Pediatric Infectious Diseases Society. 2014;3(1):23–32. 10.1093/jpids/pit057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Metcalf BJ, Gertz RE Jr., Gladstone RA, Walker H, Sherwood LK, Jackson D, et al. Strain features and distributions in pneumococci from children with invasive disease before and after 13-valent conjugate vaccine implementation in the USA. Clinical microbiology and infection: the official publication of the European Society of Clinical Microbiology and Infectious Diseases. 2016;22(1):60 e9–e29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kim L, McGee L, Tomczyk S, Beall B. Biological and Epidemiological Features of Antibiotic-Resistant Streptococcus pneumoniae in Pre- and Post-Conjugate Vaccine Eras: a United States Perspective. Clin Microbiol Rev. 2016;29(3):525–52. 10.1128/CMR.00058-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Camilli R, Daprai L, Cavrini F, Lombardo D, D'Ambrosio F, Del Grosso M, et al. Pneumococcal carriage in young children one year after introduction of the 13-valent conjugate vaccine in Italy. PLoS ONE. 2013;8(10):e76309 10.1371/journal.pone.0076309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Van Hoek AJ, Sheppard CL, Andrews NJ, Waight PA, Slack MPE, Harrison TG, et al. Pneumococcal carriage in children and adults two years after introduction of the thirteen valent pneumococcal conjugate vaccine in England. Vaccine. 2014;32(34):4349–55. 10.1016/j.vaccine.2014.03.017 [DOI] [PubMed] [Google Scholar]
- 91.Valenzuela MT, O'Loughlin R, De La Hoz F, Gomez E, Constenla D, Sinha A, et al. The burden of pneumococcal disease among Latin American and Caribbean children: Review of the evidence. Revista Panamericana de Salud Publica/Pan American Journal of Public Health. 2009;25(3):270–9. [DOI] [PubMed] [Google Scholar]
- 92.(IVAC) IVAC. State of PCV Use and Impact Evaluations: The Johns Hopkins Bloomberg School of Public Health; 2016. http://www.jhsph.edu/research/centers-and-institutes/ivac/resources/PCVImpactGapAnalysis_MAR2016_FINAL_public.pdf.
- 93.Jaiswal N, Singh M, Das RR, Jindal I, Agarwal A, Thumburu KK, et al. Distribution of serotypes, vaccine coverage, and antimicrobial susceptibility pattern of Streptococcus pneumoniae in children living in SAARC countries: a systematic review. PloS ONE. 2014;9(9):e108617 10.1371/journal.pone.0108617 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOC)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.