ABSTRACT
Human cytomegalovirus (HCMV) is the most common congenital infection worldwide and the leading infectious cause of neurologic deficits and hearing loss in newborns. Development of a maternal HCMV vaccine to prevent vertical virus transmission is a high priority, yet protective maternal immune responses following acute infection are poorly understood. To characterize the maternal humoral immune response to primary CMV infection, we investigated the plasmablast and early antibody repertoire using a nonhuman primate model with two acutely rhesus CMV (RhCMV)-infected animals—a CD4+ T cell-depleted dam that experienced fetal loss shortly after vertical RhCMV transmission and an immunocompetent dam that did not transmit RhCMV to her infant. Compared to the CD4+ T cell-depleted dam that experienced fetal loss, the immunocompetent, nontransmitting dam had a more rapid and robust plasmablast response that produced a high proportion of RhCMV-reactive antibodies, including the first identified monoclonal antibody specific for soluble and membrane-associated RhCMV envelope glycoprotein B (gB). Additionally, we noted that plasmablast RhCMV-specific antibodies had variable gene usage and maturation similar to those observed in a monkey chronically coinfected with simian immunodeficiency virus (SIV) and RhCMV. This study reveals characteristics of the early maternal RhCMV-specific humoral immune responses to primary RhCMV infection in rhesus monkeys and may contribute to a future understanding of what antibody responses should be targeted by a vaccine to eliminate congenital HCMV transmission. Furthermore, the identification of an RhCMV gB-specific monoclonal antibody underscores the possibility of modeling future HCMV vaccine strategies in this nonhuman primate model.
KEYWORDS: congenital cytomegalovirus, nonhuman primates, vaccine
INTRODUCTION
Human cytomegalovirus (HCMV) is a ubiquitous herpesvirus with seroprevalence rates ranging from 40 to 90% of the world's adult population (1). HCMV is the most common cause of congenital infection worldwide, affecting 0.64% of all live-born infants, totaling 40,000 newborns per year in the United States alone (2). Of these 40,000 annual congenital HCMV infections, it is estimated that 5,400 cases result in long-term neurological sequelae, and an additional 400 cases result in fetal death (3). One of the most common neurological manifestations of congenital HCMV infection is microcephaly, a condition that has gained global attention following the recent outbreak of Zika virus in the Americas (4). HCMV is also the leading cause of sensorineural hearing loss in newborns, accounting for up to 25% of all childhood deafness (5–7). In recognition of the major impact of congenital HCMV infection on pediatric health, the development of a protective maternal HCMV vaccine has remained a “top priority” of the National Academy of Medicine for the past 15 years (8–10).
The rate of congenital HCMV transmission is higher among women who acquire primary infection during pregnancy (30 to 35%) than among HCMV-seropositive women (0.2 to 2%) (2, 11–13). Furthermore, in utero HCMV transmission following primary maternal CMV infection more frequently results in permanent and severe neurological deficits, especially when the virus is transmitted during the earliest stages of fetal development (14). Collectively, these findings suggest that preexisting maternal immunity can provide partial protection against congenital transmission and plays a role in the prevention of adverse pregnancy outcomes such as neurological impairment, and, in some cases, fetal loss. However, the maternal immune correlates of protection, which will frame the immunologic targets of an HCMV vaccine, are not yet clearly defined.
Previous human cohort studies have suggested that antibodies likely play an important role in controlling vertical transmission of HCMV and reducing the severity of clinical outcomes following infant infection. Pregnant women with early avidity maturation of HCMV-specific IgG antibodies appear to be at a lower risk of vertical transmission (15). Additionally, in comparison to HCMV-transmitting mothers, nontransmitting mothers exhibit earlier antibody responses to the surface glycoprotein pentameric complex (gH/gL/UL128/UL130/UL131A) (PC), and antibodies with this specificity were found to inhibit HCMV cell-to-cell spread and infection of cells by free virus (16, 17). Furthermore, infants with an increased number of B cells present at birth had a reduced incidence of long-term impairment resulting from congenital HCMV infection, suggesting that antibodies may be critical for effective anti-HCMV immunity during fetal development (18). Despite these findings, attempts to prevent transplacental HCMV transmission by passive infusion of HCMV-specific hyperimmune globulin to pregnant women following primary infection did not result in a significant reduction in rates of congenital infection (19). However, perhaps the elicitation or delivery of more potent functional antibodies may be an optimal strategy to prevent placental HCMV transmission (16).
The role of maternal humoral immunity in preventing severe fetal outcomes is further supported by our recently described rhesus monkey model of congenital CMV transmission (20). In our model, two groups of rhesus CMV (RhCMV)-seronegative pregnant females (immunocompetent and CD4+ T cell-depleted dams) were inoculated intravenously with a swarm of three RhCMV strains (180.92, UCD52, and UCD59) at 8 weeks of gestation. In two of three immunocompetent females, intrauterine RhCMV transmission occurred between 2 and 4 weeks postinfection. Of interest, while all fetuses of the immunocompetent dams were carried to term and there were no signs of congenital CMV pathogenesis, a single RhCMV-seronegative, immunocompetent dam did not transmit RhCMV to her infant despite the absence of preexisting immunity. In contrast, intrauterine transmission occurred within 1 to 3 weeks of infection in four of four CD4+ T cell-depleted pregnant dams, of which three experienced fetal loss at week 3 postinfection and the fourth animal delivered a full-term infant with symptomatic CMV disease. Most notably, CD4+ T cell depletion caused a delay in the appearance of RhCMV-neutralizing antibodies until 3 weeks postinfection, which may have contributed to protection against the severe fetal outcome (20). Given the rapidity of intrauterine transmission, dampened maternal anti-RhCMV antibody function, and fetal loss and/or disease after inoculation of CD4+ T cell-depleted dams, we sought to characterize the acute maternal B cell responses in both a CD4+ T cell-depleted, RhCMV-transmitting dam and an immunocompetent nontransmitting dam in order to define the early humoral immune response to RhCMV infection during pregnancy.
Here, we applied a newly developed rhesus plasmablast phenotyping and sorting methodology (21) to define the kinetics and monoclonal antibody (MAb) repertoire of the plasmablast response to acute RhCMV infection in both an RhCMV-transmitting dam and a nontransmitting dam. We also compared the early antibody responses to that of the memory B cell repertoire of a rhesus monkey that was chronically coinfected with RhCMV and simian immunodeficiency virus (SIV). The recovered monoclonal antibodies were further assessed for RhCMV antigen-specific binding and genetic/maturation characteristics in comparison to those isolated from the memory B cell population of a chronically RhCMV/SIV-coinfected rhesus monkey. Our characterization of the plasmablast response in rhesus monkeys following primary RhCMV infection will guide future studies evaluating immunologic responses to acute viral infection in rhesus monkey models. Furthermore, our dissection of the potentially protective role of the early maternal humoral immune response against congenital RhCMV transmission could inform our understanding of the characteristics of maternal antibodies that prevent congenital HCMV transmission in humans.
RESULTS
Kinetics of the early antibody and plasmablast responses in acutely RhCMV-infected pregnant rhesus monkeys.
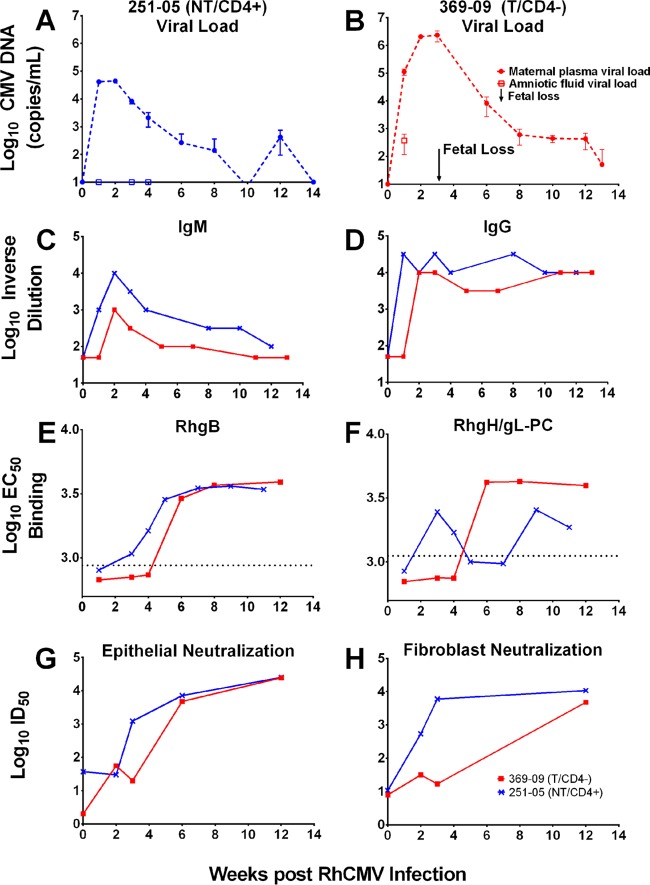
Two RhCMV-seronegative, pregnant rhesus macaques (251-05 and 369-09) at 8 weeks of gestation were intravenously inoculated with a swarm of RhCMV strains. Animal 369-09, which had been treated with a CD4+ T cell-depleting antibody 1 week prior to inoculation, had a high maternal plasma viral load and evidence of intrauterine transmission (virus present in amniotic fluid by PCR) within 1 week of RhCMV infection. Fetal loss occurred in this animal at 3 weeks postinfection. In contrast, animal 251-05 did not receive the CD4+ T cell-depleting antibody prior to RhCMV inoculation, had a peak maternal plasma viral load approximately 2 logs lower than that of the CD4+ T cell-depleted dam, and did not transmit the virus to her fetus (Fig. 1A and B). In the setting of acute primary RhCMV infection, both animals had peak plasma anti-RhCMV binding IgM and IgG responses by 2 weeks postinfection (Fig. 1C and D), but the magnitude of the IgM and IgG responses of animal 369-09 (transmitting, CD4+ T cell depleted [T/CD4−]) was 0.5 to 1 log lower than that of animal 251-05 (nontransmitting, immunocompetent [NT/CD4+]) (Fig. 1C and D). Importantly, RhCMV antigen-specific (RhCMV glycoprotein B [RhgB], RhCMV PC [RhPC]) IgG antibodies were detectable by 2 weeks postinfection in the NT/CD4+ dam but were delayed in the T/CD4− female until 5 weeks postinfection (Fig. 1E and F). The higher titer of RhPC-specific antibodies in the T/CD4− dam from weeks 6 to 12 postinfection was unexpected but could be due to greater antigen exposure in this CD4+ T cell-depleted animal with an increased plasma viral load (Fig. 1F). Furthermore, antibodies capable of neutralizing RhCMV infection of fibroblasts and epithelial cells were produced by the NT/CD4+ dam by 2 and 3 weeks postinfection, respectively, yet were delayed until more than 3 weeks postinfection in the T/CD4− dam (Fig. 1G and H). The viral load and antibody kinetics data shown in Fig. 1 were adapted from a previous publication (20) that established this rhesus macaque model of congenital cytomegalovirus transmission.
FIG 1.
Differences in the early RhCMV-specific antibody responses between an RhCMV-nontransmitting (NT/CD4+) pregnant monkey and an RhCMV-transmitting (T/CD4−) pregnant monkey following acute maternal RhCMV challenge. (A and B) Maternal plasma (●) and amniotic fluid (□) RhCMV DNA loads in an immunocompetent, RhCMV-nontransmitting dam (NT/CD4+, 251-05; in blue throughout) and a CD4+ T cell-depleted RhCMV-transmitting dam (T/CD4−, 369-09; in red throughout). An arrow indicates the time of fetal loss due to spontaneous abortion. (C and D) IgM and IgG responses measured against whole RhCMV virions. (E and F) RhCMV gB and RhPC glycoprotein-specific responses. The dotted lines indicate the limits of detection at week 0 + 2 SD. (G and H) Neutralizing titers assessed against RhCMV variants in fibroblasts and epithelial cells. The viral load and antibody kinetics data shown were adapted from reference 20 with permission of the publisher.
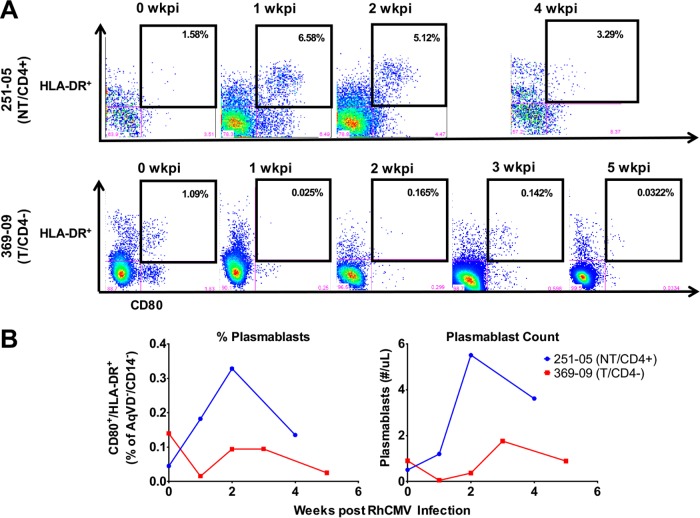
To complement our assessment of the early antibody kinetics in each of the two monkeys, we measured the kinetics of the plasmablast response over the first 4 to 5 weeks of primary RhCMV infection. Plasmablasts, defined as CD14−/CD16−/CD3−/CD20−/CD11c−/CD123−/CD80+/HLADR+ lymphocytes (see Fig. S1 and S2 in the supplemental material) (21), were detectable in peripheral blood as early as 1 week after infection in the NT/CD4+ dam, continued their expansion through week 2 postinfection, and diminished by week 4 postinfection (Fig. 2A). In contrast, no clear peripheral plasmablast population expansion was observed in the T/CD4− dam through week 5. Notably, the population of plasmablasts detected in peripheral blood at week 0 in the T/CD4− dam may be a response to the CD4+ T cell-depleting antibody that was administered intravenously the week before. The absolute number of plasmablasts in the periphery of the T/CD4− dam, calculated using the circulating absolute lymphocyte count as determined by the complete blood count, was lower than that in the NT/CD4+ monkey with absolute peak plasmablast counts of 1.8 × 103/μl and 5.5 × 103/μl (3 weeks postinfection), respectively. Thus, CD4+ T cell depletion delayed the peak plasmablast response following RhCMV infection and may have resulted in incomplete development of the overall plasmablast response.
FIG 2.
Kinetics of plasmablast response in an RhCMV-nontransmitting, immunocompetent (NT/CD4+) dam (251-05) and an RhCMV-transmitting (T/CD4−) dam following acute maternal RhCMV challenge. (A) The plasmablast populations (defined as CD80+/HLADR+ of negatively selected lineage), outlined in black, following primary RhCMV infection in 251-05 (NT/CD4+, upper panel) and in 369-09 (T/CD4−, lower panel) are shown. The percentages of CD80+/HLADR+ populations out of the negatively selected lineage are indicated. wkpi, weeks postinfection. (B) Percent plasmablasts (calculated as CD80+/HLADR+ lymphocytes of total viable lymphocytes [AqVD−/CD14−]) (left) and absolute plasmablast counts (right) are shown for animal 251-05 (NT/CD4+; in blue) and animal 369-09 (T/CD4−; in red) acutely infected with RhCMV.
Isolation and characterization of the acute plasmablast antibody repertoire during primary RhCMV infection.
Plasmablasts were single-cell sorted from the NT/CD4+ dam at week 2 postinfection and the T/CD4− dam at week 3 postinfection using the following flow cytometry phenotype: CD14−/CD16−/CD3−/CD20−/HLADR+/CD11c−/CD123−/CD80+/IgG+. Of 80 single-sorted plasmablasts isolated from both the NT/CD4+ and T/CD4− monkeys, immunoglobulin variable-gene PCR amplification, purification, and sequencing yielded 14 heavy- and light-chain pairs without nonsense mutations (17.5% efficiency) and 9 functional heavy- and light-chain pairs (11.3% efficiency) for the NT/CD4+ and T/CD4− monkeys, respectively.
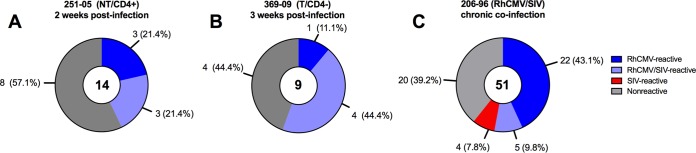
To examine the specificity of the plasmablast antibodies at the peak plasmablast response during acute primary RhCMV infection, we screened the antibodies that were generated from the isolated plasmablasts for reactivity against RhCMV virions and virus lysates. Antibody polyreactivity was assessed by binding to control antigen SIVmac gp120. Of the 14 antibodies generated by plasmablasts at week 2 postinfection in the NT/CD4+ female, 3 (21.4%) were RhCMV reactive, 3 (21.4%) were polyreactive and bound to both RhCMV antigens and SIVmac gp120, and 8 (57.1%) were not reactive to either RhCMV or SIV antigens (Fig. 3A). Among the 9 antibodies generated from the plasmablasts of the T/CD4− female, only 1 (11.1%) was solely RhCMV specific, 4 (44.4%) were polyreactive to RhCMV virions and SIVmac gp120, and 4 (44.4%) were nonreactive (Fig. 3B; Data Set S1).
FIG 3.
RhCMV reactivity of antibodies isolated from plasmablasts of dams acutely infected with RhCMV and from total memory B cells of a monkey chronically coinfected with RhCMV/SIV. Plasmablasts at peak response were sorted from animal 251-05 (NT/CD4+) at 2 weeks postinfection (wkpi) (A) and animal 369-09 (T/CD4−) at 3 wkpi (B) and screened for RhCMV reactivity and specificity. Total memory B cells were sorted from peripheral blood of 206-96, a rhesus monkey chronically coinfected with RhCMV and SIV for 1 year (C). Proportions of isolated monoclonal antibodies with RhCMV or SIV reactivity and antibodies that were nonreactive to RhCMV or SIV are shown.
We also compared the acutely elicited monoclonal antibodies isolated from plasmablasts of the two acutely RhCMV-infected monkeys to those of circulating memory B cells harvested from an adult female rhesus monkey (206-96) chronically coinfected with RhCMV and SIV (Data Set S2). In rhesus monkey breeding colonies, RhCMV seroconversion typically occurs by 1 year of age (22), and therefore it is expected that the memory B cell response to RhCMV in this monkey may have undergone affinity maturation over several years, leading to a higher degree of antigen affinity. In addition, RhCMV/SIV-coinfected monkeys often sustain RhCMV disease due to an inability to contain viral replication (23) yet also experience B cell dysregulation. Remarkably, of the 51 antibodies isolated from total circulating memory B cells of the RhCMV/SIV-coinfected monkey, 27 bound to either RhCMV lysate and/or intact virions (52.9%) (Fig. 3C). Relatively few bound only to whole RhCMV virions (n = 2, 3.9%), which may reflect binding to conformation-dependent surface glycoprotein complexes, whereas 15.7% bound to only viral lysate (n = 8). The majority of the remaining RhCMV-specific antibodies bound to both whole virions and viral lysate (n = 17, 33.3%) (Fig. 3C; Data Set S2). In this monkey, a relatively small percentage of RhCMV-reactive antibodies (n = 5, 9.8%) were polyreactive with SIVmac gp120 (Fig. 3C; Data Set S2). Additionally, only 4 of 52 isolated antibodies were reactive to SIVmac gp120 alone (7.8%), despite coinfection with SIV for more than 1 year. The remaining 20 of the 52 antibodies from this chronically RhCMV/SIV-coinfected monkey were reactive to neither RhCMV nor SIVmac gp120 (Fig. 3C; Data Set S2).
Classification of plasmablast antibody isotype and gene usage in acute primary RhCMV infection versus memory B cell antibodies during chronic RhCMV/SIV coinfection.
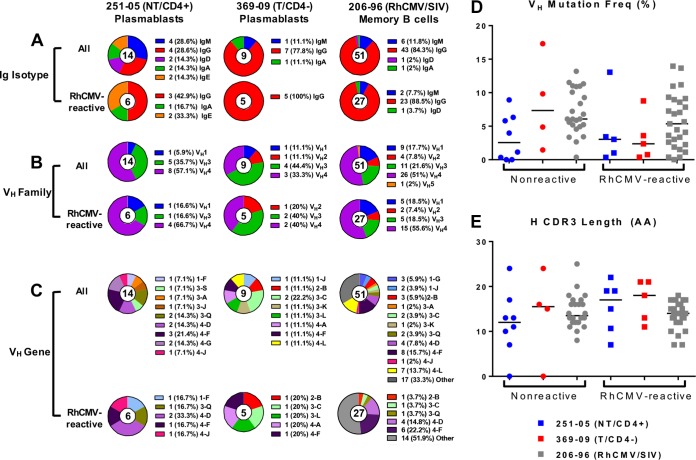
Antibodies isolated from the plasmablast response following acute RhCMV infection of the NT/CD4+ dam were equally distributed between IgM and IgG isotypes (28.6% each), whereas isolated plasmablasts from the T/CD4− female and chronically RhCMV-infected memory B cell population were predominantly IgG isotype antibodies (77.8% and 84.3%) (Fig. 4A). The predominant variable heavy-chain families used by all plasmablasts isolated during acute RhCMV infection were VH3 and VH4, consistent with those used by antibodies isolated from memory B cells of the chronically RhCMV/SIV-coinfected animal (Fig. 4B). When the total antibodies isolated from each of the three animals were compared, the specific VH gene usage had similar distributions (Fig. 4C, upper pie charts). In acute infection, 4-F was the predominant VH gene used by the NT/CD4+ dam (21.4%), whereas the T/CD4− female showed no VH gene preference. When specific VH gene usages of antibodies reactive with RhCMV antigens were compared, VH gene 4-D was predominant in the NT/CD4+ (33.3%) monkey and second-most predominant in the RhCMV/SIV (14.8%)-coinfected monkey, yet the T/CD4− female showed no clear preference for any particular VH gene (Fig. 4D). VH gene 4-F was most predominant in the RhCMV/SIV-coinfected monkey (22.2%) and also appeared in the antibody repertoire in acute RhCMV infection in both the NT/CD4+ dam and the T/CD4− dam.
FIG 4.
Isotype, subclass, and genetic analysis of antibodies in acute and chronic RhCMV infection. Distribution of immunoglobulin isotype (A), VH family (B), and VH gene usage (C) from total monoclonal antibodies (upper pie charts) and RhCMV-reactive antibodies (lower pie charts) isolated from an NT/CD4+ dam (251-05; in blue), an T/CD4− dam (369-09; in red) during acute infection, and a chronically RhCMV/SIV-coinfected monkey (206-96; in gray). VH mutation frequencies (D) and HCDR3 lengths (E) of isolated RhCMV-reactive (●) and nonreactive (■) monoclonal antibodies were similar between each monkey. AA, amino acids.
Mutation frequency and heavy-chain complementarity-determining region 3 (HCDR3) length of plasmablast antibodies in primary maternal RhCMV infection versus those in circulating memory B cell antibodies during chronic RhCMV/SIV coinfection.
To examine the genetic characteristics and degree of affinity maturation within the plasmablast response of the NT/CD4+ and T/CD4− monkeys, we analyzed the heavy-chain somatic hypermutation frequency as well as HCDR3 of antibodies at the peak of the plasmablast response following acute RhCMV infection (Fig. 4D and E). For comparison, we included antibodies isolated from the chronically RhCMV/SIV-coinfected monkey. The median variable heavy-chain (VH) somatic hypermutation rates for nonreactive antibodies from the NT/CD4+, T/CD4−, and chronically RhCMV/SIV-coinfected monkeys were 2.6%, 7.3%, and 6.1%, respectively, values that were not statistically different between any of the animals (P value, nonsignificant [NS]; Kruskal-Wallis test). The median HCDR3 lengths of nonreactive antibodies were also found to be similar among all three monkeys (P value, NS; Kruskal-Wallis test). No differences were observed in the median VH mutation frequencies in RhCMV-reactive antibodies in the three monkeys: 3%, 2.4%, and 5.4% in the NT/CD4+, T/CD4−, and RhCMV/SIV-coinfected monkeys, respectively (P value, NS; Kruskal-Wallis test). Additionally, no significant differences were found in the HCDR3 lengths of RhCMV-reactive antibodies produced by the acutely infected NT/CD4+ and T/CD4− dams or the chronically RhCMV/SIV-infected animal, which had median lengths of 17, 18, and 14 amino acids, respectively (P value, NS; Kruskal-Wallis test).
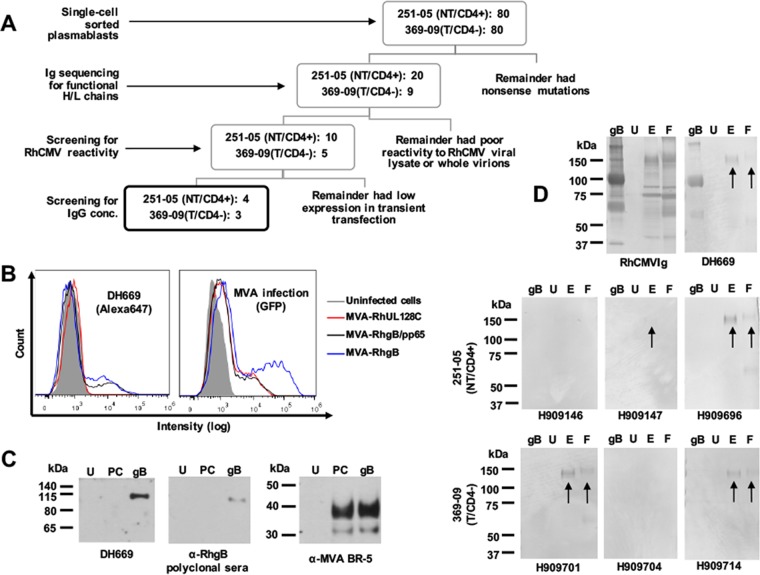
Evaluation of RhCMV-reactive plasmablast MAb specificities.
We investigated the specificity of isolated plasmablast MAbs that bound strongly to whole RhCMV virions. The process of antibody screening is detailed in Fig. 5A. A total of seven MAbs exhibited binding against whole RhCMV virions and were efficiently produced in transient transfection (concentration of at least 0.75 μg/ml)—four from animal 251-05 (NT/CD4+) and three from animal 369-09 (T/CD4−) (Fig. 5A; Data Set S1). These MAbs were purified and screened for RhCMV glycoprotein recognition. Antibody specificity was first assessed by intracellular flow cytometry staining of permeabilized BHK-21 cells infected with modified vaccinia Ankara (MVA) vector expressing the RhCMV gH/gL/UL128/UL130/UL131A pentameric complex (termed MVA-UL128C), RhCMV gB, or a combination of RhCMV gB and tegument protein, RhCMV pp65. Of the antibodies screened, only a single antibody (DH669) isolated from the NT/CD4+ dam exhibited specificity for RhgB (Fig. 5B; Fig. S3), an essential surface glycoprotein required for entry into all cell types. Recognition of RhgB by DH669 was confirmed by Western blotting against both MVA-expressed (Fig. 5C) and soluble (Fig. 5D) RhgB protein. Furthermore, this MAb was not cross-reactive with soluble HCMV gB protein by enzyme-linked immunosorbent assay (ELISA). DH669 is a member of the IgG1 subclass with the variable heavy-chain gene 1∼F*02 (3% mutated, CDR3 length of 7 amino acids) and the kappa variable light-chain gene 4∼3*01. The neutralization activity of DH669 was assessed against strain 180.92 in telomerized rhesus fibroblasts (teloRF) and against strain UCD52 in monkey kidney epithelial (MKE) cells (starting concentration, 100 μg/ml), but no neutralizing activity was detected in either cell line. No tested MAbs demonstrated binding to either MVA-expressed or soluble RhCMV gH/gL/UL128/UL130/UL131A pentameric complex (Fig. S4).
FIG 5.
Characterization of plasmablast MAb glycoprotein binding specificities. (A) Flow chart depicting the sorting/screening of MAbs from plasmablasts of two monkeys. (B) BHK-21 cells infected with MVA recombinants were analyzed by flow cytometry for intracellular staining of RhCMV UL128C (expressing gH/gL/UL128/UL130/UL131A), gB, or gB/pp65. Left panel, staining of MVA-infected cells was performed with DH669 as the primary antibody and Alexa Fluor 647-coupled secondary anti-rhesus IgG Ab. Right panel, GFP expression was analyzed for confirming MVA infection of BHK-21 cells since all the constructs contain a GFP expression cassette. Uninfected cells were used as a control. (C) Recognition of linear RhgB by DH669 (left panel) and anti-RhgB polyclonal serum (center panel). Immunoblots were performed by using a lysate from MVA-RhUL128C (PC)- or MVA-RhgB (gB)-infected BHK-21 cells. Uninfected cells (U) were used as a control. For a loading control, samples were analyzed with anti-MVA BR-5 Ab (right panel). (D) The binding of RhCMV Ig (whole IgG from RhCMV-seropositive monkeys) and DH669 (upper panels), plasmablast MAbs from 251-05 (NT/CD4+) (middle panels), and plasmablast MAbs from 369-09 (T/CD4−) (lower panels) to soluble RhgB and to membrane-associated proteins from uninfected cells (U), epithelial cell-tropic UCD52-infected cells (E), and fibroblast-tropic 180.92-infected cells (F) was assessed. Arrows indicate MAb binding to membrane-associated RhgB. Mass markers (in kilodaltons) are shown next to each panel.
We further investigated binding of these seven MAbs against cytosolic and membrane-associated proteins isolated from RhCMV-infected fibroblasts. We first confirmed that the cytosolic protein preparations isolated from RhCMV-infected cells contained a variety of RhCMV antigens, including RhCMV IE-1 and RhCMV pp65b (Fig. S4). Moreover, we noted that DH669 bound to membrane-associated RhgB from both strain 180.92 (fibroblast tropic) and strain UCD52 (epithelial cell tropic) in addition to the soluble RhgB protein (Fig. 5D; Fig. S4). Intriguingly, four of the remaining six MAbs also exhibited binding to membrane-associated RhgB (Fig. 5D; Fig. S4). Furthermore, these MAbs were reactive against RhgB expressed on the surface of both UCD52-infected and 180.92-infected cells and are therefore not specific to RhgB from a single viral strain (Fig. 5D; Fig. S4). These membrane-associated RhgB-binding MAbs were isolated from plasmablasts of both animal 251-05 (NT/CD4+) and animal 369-09 (T/CD4−), suggesting that they are frequently elicited in both settings.
DISCUSSION
There remains a gap in our understanding of the immune responses to primary HCMV infection during pregnancy and in particular those that prevent viral transmission to the fetus (1). Using a rhesus monkey model of congenital RhCMV transmission, we previously observed that CD4+ T cell-depleted monkeys had delayed development of maternal RhCMV-neutralizing antibodies, which was associated with high rates of congenital transmission and fetal loss (20). Given this observation, this study sought to characterize the early maternal humoral response following primary RhCMV infection by dissecting the antibody repertoire of plasmablasts. Compared to the immunocompetent RhCMV-nontransmitting dam, the CD4+ T cell-depleted dam had slower kinetics and reduced magnitude of a peripheral plasmablast response, as well as distinct antibody specificity and VH gene expression.
To our knowledge, this is the first study to evaluate the plasmablast response to acute viral infection in rhesus monkeys and to characterize the plasmablast antibody repertoire in the setting of primary maternal CMV infection. Plasmablast responses can be triggered by a T cell-independent (24) or T cell-dependent (24, 25) pathway. In the former, plasmablasts are activated to produce polyspecific antibodies with poor affinity maturation (26). In the T cell-dependent pathway, CD4+ follicular helper T cells secrete interleukin 21 (IL-21), which facilitates germinal center B cell selection events and differentiation of activated B cells into plasmablasts (27). By studying the antibody repertoire in acutely infected monkeys with differential CD4+ T cell status, we have gained insight into the importance of each of these pathways in the kinetics and specificity of an early antibody response in acute RhCMV infection. We realize the limitations of our study due to the antibody repertoire of a small number of animals being represented. However, our data suggest that a rapid plasmablast response against RhCMV potentially contributes to protection against congenital transmission and is dependent upon CD4+ T cell help. Therefore, the role of CD4+ T cells in the development of RhCMV-specific antibody responses to primary infection should be investigated further.
In addition to distinct kinetics of the plasmablast response in the dams with disparate immune competencies and fetal outcomes, we also observed possible differences in the plasmablast antibody repertoires. RhCMV-reactive plasmablasts from animal 251-05 (NT/CD4+) showed a preference for the VH4 family, including genes 4-D and 4-F, which resembles the VH gene expression profile from the monkey chronically coinfected with RhCMV/SIV. In contrast, RhCMV-reactive antibodies from animal 369-09 (T/CD4−) showed no clear preference for VH family or gene expression. Comparison of the genetic characteristics of these antibodies to those isolated from memory B cells from two acutely infected animals with a chronic RhCMV/SIV coinfection allowed us to assess the relationship between RhCMV-binding antibodies produced early after infection to the highly evolved antibody repertoire present during chronic RhCMV infection. The similarity of VH gene usage between animal 251-05 (NT/CD4+) and the chronically RhCMV/SIV-coinfected monkey suggests that this immunocompetent dam had the potential to develop a high-affinity RhCMV-binding clonal lineage. There was no discernible difference in either the percentages of plasmablast RhCMV-specific antibodies or mutation frequencies between the NT/CD4+ dam and the T/CD4− dam. However, there was a higher percentage of polyreactive antibodies in the T/CD4− dam (3/14, 21%) than in the NT/CD4+ dam (4/9, 44%), perhaps suggesting decreased affinity maturation in the absence of CD4+ T cell help.
One surprising observation was the remarkable 53% of the memory B cell pool that was RhCMV specific in the chronically RhCMV/SIV-coinfected monkey, compared to just 17.6% of the antibodies that were SIV reactive. This trend correlates well with the highly prevalent HCMV-specific memory T cell responses in HCMV-seropositive individuals, comprising approximately 10% of both the CD4+ and CD8+ memory T cell compartments in blood (28). Additionally, it has been shown that a higher proportion of HCMV-specific memory CD4+ T cells than human immunodeficiency virus type 1 (HIV-1)-specific CD4+ T cells exists in individuals with chronic HCMV/HIV coinfections (29).
Of the seven antibodies tested for RhCMV glycoprotein specificity, we were able to identify that only one (DH669) recognized MVA-expressed or soluble RhgB. Intriguingly, four additional MAbs bound to membrane-associated RhgB, though not to MVA-expressed or soluble RhgB. We hypothesize that this binding variability could indicate protein conformational differences between the MVA-expressed/soluble RhgB protein and the RhgB expressed on the surface of RhCMV-infected cells, raising the possibility that these MAbs may be directed against prefusion or cell-associated RhgB protein epitopes. gB is the primary protein that mediates viral fusion with the host cell and has been a topic of vaccine research for many decades (30), culminating in multiple phase II clinical trials in which a gB subunit vaccine demonstrated moderate (∼50%) efficacy (31, 32). This is the first reported isolation of an anti-RhCMV monoclonal antibody, suggesting the future possibility of modeling antibody-based prophylaxis/treatment of congenital HCMV transmission in rhesus monkeys with RhCMV glycoprotein-specific MAbs. It is noteworthy that DH669 was nonneutralizing. Though the majority of gB-specific MAbs are nonneutralizing in humans (33), gB is recognized as an important target of neutralizing activity (34). Furthermore, it is unknown whether neutralization, and not other antibody functions such as antibody-dependent cellular cytotoxicity (ADCC), are protective against congenital CMV transmission.
Of note, none of the isolated plasmablast MAbs bound to MVA-expressed or soluble RhCMV pentameric complex (gH/gL/UL128/UL130/UL131A) (RhPC). One potential explanation is that our MAb isolation and glycoprotein-specificity analysis were completed at 1 to 2 weeks postinfection. RhgB, similar to HCMV gB, is an immunodominant target and elicits high titers of antibodies early during the course of infection (35). Indeed, in humans, the development of HCMV PC-specific antibodies is delayed several weeks following the appearance of RhCMV gB-reactive antibodies (36). The timing of the development of RhPC-reactive MAbs in this model is important, since the pentameric complex is known to be a target of neutralizing antibodies for both HCMV (37, 38) and RhCMV (39) and therefore is perhaps an important vaccine immunogen. It remains unknown whether the kinetics of the rhesus monkey anti-RhPC antibody response following natural infection are analogous to the human humoral response to HCMV PC, and more extensive mapping of the plasmablast and B cell repertoire following primary RhCMV infection is needed.
Our study of the plasmablast antibody repertoire in rhesus monkeys is limited by the small number of experimental subjects. However, we were able to investigate the early maternal humoral response to acute RhCMV infection, which is important for modeling prevention of primary congenital CMV transmission and disease. Our analysis suggests that the delayed plasmablast response in the CD4+ T cell-depleted dam may have contributed to congenital infection and/or disparate fetal outcomes. This work will enable future studies to identify the characteristics, specificity, and function of antibodies that ought to be elicited by maternal vaccination to bring an end to the leading infectious cause of brain damage and sensorineural hearing loss in infants.
MATERIALS AND METHODS
Characterization and isolation of plasmablasts from acutely RhCMV-infected rhesus dams.
The plasmablast populations in peripheral blood mononuclear cells (PBMCs) from RhCMV-transmitting and -nontransmitting dams during primary RhCMV infection were phenotypically characterized and isolated using a panel of fluorescently conjugated monoclonal antibodies and flow cytometry as previously described (21). Plasmablasts were defined by the following flow cytometry phenotype: CD14−/CD16−/CD3−/CD20−/HLADR+/CD11c−/CD123−/CD80+ (see Fig. S1 in the supplemental material). Gating controls were determined using fluorescence minus one (FMO) controls for the full panel (CD3, CD20, CD14, CD16, CD11c, CD123, HLADR, CD80, surface IgG, AqVD) (Table S1). Plasmablasts were isolated using the flow cytometry phenotype CD14−/CD16−/CD3−/CD20−/HLADR+/CD11c−/CD123−/CD80+/surface IgG+; surface IgG expression was added to the sorting criteria to potentially enhance the yield of RhCMV-specific antibody-producing plasmablasts. Although well defined in humans, plasmablasts are not as well defined phenotypically in rhesus monkeys. Thus, plasmablast responses were first characterized in a rhesus monkey who received a DTaP booster vaccination (Fig. S2). This technique was then applied to PBMCs isolated from the immunocompetent RhCMV-nontransmitting dam (251-05) in addition to a CD4+ T cell-depleted, RhCMV-transmitting monkey (369-09) for comparison. Complete blood counts were performed on peripheral blood at each time point in order to calculate the absolute numbers of plasmablasts.
Isolation of memory B cells from a chronically RhCMV/SIV-coinfected monkey.
Memory B cells, defined as CD20+/CD3−/surface IgD− lymphocytes, were identified and sorted from PBMCs collected from an adult rhesus monkey naturally infected with RhCMV and intravenously inoculated with SIV 1 year prior to evaluation. Memory B cells were sorted as single cells into 96-well plates containing an RNA-stabilizing mixture (40). Of note, the use of an SIV-infected monkey was a matter of convenience and was not intended to model HCMV/HIV coinfection.
PCR amplification, sequencing, and data analysis of immunoglobulin heavy (VH)- and light (VL)-chain variable genes isolated from plasmablasts and memory B cells.
The immunoglobulin (Ig) VHDHJH and VLJL genes of the sorted plasma cell and memory B cells were amplified by reverse transcription (RT) and nested PCR using the method and primer sets reported previously (40). Nested VH and VL gene PCR methods were completed as described previously (41). Briefly, PCR products of Ig VH and VL genes were purified using a PCR purification kit and sequenced in forward and reverse directions using an ABI 3700 instrument and a BigDye sequencing kit. The isotypes of Ig heavy chain and light chain were determined by comparing the constant-region sequences of the isolated Ig VH and VL PCR products with the constant- and variable-region sequences of defined rhesus Ig genes. Functional VH and VL gene pairing and inferred unmutated ancestors were determined using Cloanalyst, as well as gene segment usage, somatic mutations, and complementarity-determining region 3 (CDR3) length.
Expression of VH and VL as full-length IgG1 recombinant monoclonal antibodies.
Using the methods described previously (41), the isolated Ig VH and VL gene pairs were assembled by PCR into the linear full-length Ig heavy- and light-chain gene expression cassettes for production of recombinant monoclonal antibodies by transfection in the human embryonic kidney cell line 293T (40). Three days following transfection with purified PCR products of the paired Ig heavy- and light-chain gene expression cassettes, cell culture supernatants containing recombinant monoclonal antibodies were harvested, quantified for expressed IgG levels, and screened for antibody reactivity (41).
Evaluation of plasma and monoclonal antibody reactivity to RhCMV.
To test transiently expressed recombinant monoclonal antibodies and plasma for RhCMV-specific reactivity, transfection supernatants were assessed for binding to whole free virus and viral lysates prepared from fibroblast-adapted strain 180.92 (grown in primary rhesus fibroblasts and passaged in telomerized rhesus fibroblasts [teloRF]) and epithelial cell-tropic strains UCD59/UCD52 (grown/passaged in MKE cells) (42, 43) by ELISA as previously described (20). The positivity cutoff was defined as an optical density (OD) of >0.1, which was twice that of antibody-negative control wells. Antibodies were also screened for polyreactivity against an SIV envelope (Env) antigen, SIVmac gp120 (44). Antibodies with reactivity against SIVmac gp120, similarly defined by an OD of >0.1 and twice that of irrelevant transfected antibody-negative control wells, were considered to demonstrate SIV Env reactivity. Rhesus anti-influenza virus hemagglutinin (HA) IgG monoclonal antibody (CH65) was included as a negative control.
Assessment of plasmablast MAb specificity by RhCMV glycoprotein-expressing modified vaccinia Ankara (MVA) vectors.
Flow cytometry was used to investigate rhesus monkey antibody binding specificity as described previously (45), with the following modifications. Briefly, BHK-21 cells were infected with MVA-RhUL128C (expressing all five pentameric complex proteins, gH/gL/UL128/UL130/UL131A), MVA-RhgB, or MVA-RhgB/pp65 at a multiplicity of infection (MOI) of 5. At 4 h postinfection, the cells were fixed and permeabilized using a Cytofix/Cytoperm solution (BD Biosciences). Rhesus monoclonal antibodies were diluted in Perm/Wash buffer (BD Biosciences) to obtain a concentration of 500 ng/ml and added to the cells for 1 h at 4°C. After washing with Perm/Wash buffer, Alexa Fluor 647 goat anti-rhesus IgG (H+L) (SouthernBiotech) was added at a dilution of 1:2,000 for 1 h at 4°C. After an additional wash, the cells were resuspended in phosphate-buffered saline (PBS)–0.1% bovine serum albumin (BSA) and acquired using a Gallios flow cytometer (Beckman Coulter). Analysis was performed using FlowJo software (Tree Star). Uninfected BHK-21 cells were used as a control. Green fluorescent protein (GFP) expression from infected cells was used to confirm MVA infection, since all the constructs contained a GFP expression cassette (39, 46). Immunoblotting was performed to confirm antibody binding using lysates from BHK-21 cells infected with MVA-RhUL128C or MVA-RhgB at an MOI of 5 for 24 h. Infected and uninfected cells (2.5 × 105) were resuspended in 250 μl reducing Laemmli buffer (dithiothreitol [DTT], 0.1 M). Proteins were boiled, electrophoretically separated by SDS-PAGE, and blotted onto a polyvinylidene difluoride (PVDF) membrane. Purified rhesus monoclonal antibody was incubated with the membrane at a concentration of 10 ng/ml. Polyclonal anti-RhgB mouse antiserum (47) was used at a dilution of 1:3,000, and hybridoma supernatant of anti-BR-5 was used at a dilution of 1:1,500. Proteins were visualized with secondary antibodies; anti-rhesus (KPL) or anti-mouse IgG (Thermo Scientific) Ab coupled to horseradish peroxidase (HRP), followed by chemiluminescence detection using a Pierce ECL Western blot substrate (Thermo Fisher Scientific).
Assessment of plasmablast MAb binding to soluble and membrane-associated RhCMV glycoproteins.
First, to isolate membrane-associated RhCMV proteins, confluent teloRF were infected with RhCMV (either UCD52 or 180.92) at an MOI of 1. At 72 hours postinfection (hpi), cells were harvested by manual cell scraping and washed in PBS, and then the cytosolic and membrane protein fractions were isolated using the Mem-PER Plus membrane protein extraction kit (Thermo Fisher Scientific). Next, soluble RhCMV glycoprotein complexes (gB and gH/gL/UL128/UL131/UL131A) and prepared RhCMV membrane-associated proteins from virally infected cells were electrophoretically separated by SDS-PAGE (2 μg/well) on a NuPAGE Novex 4-to-12% Bis-Tris protein gel (Thermo Fisher Scientific) under nonreducing conditions and then blotted onto a PVDF membrane (Thermo Fisher Scientific). The membrane was blocked with casein blocking buffer, and then purified monoclonal antibodies were incubated with the membrane at dilutions of 1:100 for rhesus MAbs, 1:500 for IE-1 and pp65b MAbs (kindly provided by Daniel Cawley, Oregon Health Sciences University), and 1:10,000 for RhCMV Ig (whole IgG purified from RhCMV-seropositive monkeys). Proteins were detected using alkaline phosphatase-conjugated secondary antibodies (anti-monkey antibody for rhesus MAbs and anti-mouse antibody for IE-1 and pp65b; Thermo Fisher Scientific) and then visualized with stabilized alkaline phosphatase substrate (Promega).
Neutralization assays.
The neutralization methods were completed as described previously (20). Briefly, teloRF and MKE cells were seeded into 96-well plates and incubated for 2 days at 37°C and 5% CO2 to achieve 100% confluence. After 2 days, serial dilutions (1:10 to 1:30,000) of heat-inactivated rhesus plasma were incubated with RhCMV 180.92 or RhCMV UCD52 in a 50-μl volume for 45 min at 37°C. The virus-plasma dilutions were then added in duplicate to wells containing teloRF or MKE cells and incubated at 37°C for 2 h. After washing, cells were incubated at 37°C for an additional 3 h. Infected cells were then fixed for 20 min at −20°C with 1:1 methanol-acetone, rehydrated in PBS with calcium (three times for 5 min each), and processed for immunofluorescence with 0.6 mg/ml mouse anti-RhCMV IE-1 monoclonal antibody (Daniel Cawley, Oregon Health Sciences University), followed by a 1:500 dilution of goat anti-mouse IgG–Alexa Fluor 488 antibody (Millipore). Nuclei were stained with DAPI (4′,6-diamidino-2-phenylindole) for 5 min (Pierce) and imaged at ×10 magnification. ImageJ software was used to automatically count cells from a single field of view.
Statistical analyses.
Comparisons of the VH mutation frequencies and VH CDR3 lengths for animal 251-05 (NT/CD4+), animal 369-09 (T/CD4−), and the chronically RhCMV/SIV-infected monkey were performed using the nonparametric Kruskal-Wallis test. A P value of <0.05 was considered significant and was not adjusted for pairwise comparisons. All comparisons were performed using GraphPad Prism version 6.04 and were two-tailed.
Supplementary Material
ACKNOWLEDGMENTS
This work was supported by the following grants from the National Institutes of Health: (i) a DP2 grant to S.R.P. for “Maternal immune protection against congenital CMV infection” (DP2HD075699), (ii) a grant to the Duke University Center for AIDS Research (CFAR, 5P30AI064518), (iii) a fellowship grant to C.S.N. (F30HD089577A), and (iv) R01 grants to D.J.D. and P.A.B. (R01AI103960/R01AI063356). D.J.D. was partially supported by grants CA077544/CA181045, and the City of Hope Cancer Center is supported by grant CA033572.
We thank Xinzhen Yang and Pfizer, Inc., for the generous gift of research materials. Furthermore, we gratefully acknowledge Minyue Amy Wang and Kevin Wiehe at the Duke Human Vaccine Institute for their assistance with antibody sequence analysis, Tom Kepler for his work in developing the Cloanalyst software, and Chi Wei Cliburn Chan for his assistance with the statistical analysis.
The authors have no commercial affiliations or financial conflicts of interest to disclose. The funders had no role in study design, data collection and interpretation, decision to publish, or the preparation of the manuscript. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Footnotes
Supplemental material for this article may be found at https://doi.org/10.1128/CVI.00510-16.
REFERENCES
- 1.Cannon MJ, Schmid DS, Hyde TB. 2010. Review of cytomegalovirus seroprevalence and demographic characteristics associated with infection. Rev Med Virol 20:202–213. doi: 10.1002/rmv.655. [DOI] [PubMed] [Google Scholar]
- 2.Kenneson A, Cannon MJ. 2007. Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev Med Virol 17:253–276. doi: 10.1002/rmv.535. [DOI] [PubMed] [Google Scholar]
- 3.Manicklal S, Emery VC, Lazzarotto T, Boppana SB, Gupta RK. 2013. The “silent” global burden of congenital cytomegalovirus. Clin Microbiol Rev 26:86–102. doi: 10.1128/CMR.00062-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mlakar J, Korva M, Tul N, Popović M, Poljšak-Prijatelj M, Mraz J, Kolenc M, Resman Rus K, Vesnaver Vipotnik T, Fabjan Vodušek V, Vizjak A, Pižem J, Petrovec M, Avšič Županc T. 2016. Zika virus associated with microcephaly. N Engl J Med 374:951–958. doi: 10.1056/NEJMoa1600651. [DOI] [PubMed] [Google Scholar]
- 5.Cheeran MC, Lokensgard JR, Schleiss MR. 2009. Neuropathogenesis of congenital cytomegalovirus infection: disease mechanisms and prospects for intervention. Clin Microbiol Rev 22:99–126. doi: 10.1128/CMR.00023-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Grosse SD, Ross DS, Dollard SC. 2008. Congenital cytomegalovirus (CMV) infection as a cause of permanent bilateral hearing loss: a quantitative assessment. J Clin Virol 41:57–62. doi: 10.1016/j.jcv.2007.09.004. [DOI] [PubMed] [Google Scholar]
- 7.Korver AMH, de Vries JJC, Konings S, de Jong JW, Dekker FW, Vossen ACTM, Frijns JHM, Oudesluys-Murphy AM, DECIBEL Collaborative Study Group . 2009. DECIBEL study: congenital cytomegalovirus infection in young children with permanent bilateral hearing impairment in the Netherlands. J Clin Virol 46:S27–S31. doi: 10.1016/j.jcv.2009.09.007. [DOI] [PubMed] [Google Scholar]
- 8.Schleiss MR. 2010. Cytomegalovirus vaccines and methods of production (WO20009049138): the emerging recognition of the importance of virus neutralization at the epithelial/endothelial interface. Expert Opin Ther Pat 20:597–602. doi: 10.1517/13543770903584882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hassan J, Connell J. 2007. Translational mini-review series on infectious disease: congenital cytomegalovirus infection: 50 years on. Clin Exp Immunol 149:205–210. doi: 10.1111/j.1365-2249.2007.03454.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Krause PR, Bialek SR, Boppana SB, Griffiths PD, Laughlin CA, Ljungman P, Mocarski ES, Pass RF, Read JS, Schleiss MR, Plotkin SA. 2013. Priorities for CMV vaccine development. Vaccine 32:4–10. doi: 10.1016/j.vaccine.2013.09.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Boppana SB, Rivera LB, Fowler KB, Mach M, Britt WJ. 2001. Intrauterine transmission of cytomegalovirus to infants of women With preconceptional immunity. N Engl J Med 344:1366–1371. doi: 10.1056/NEJM200105033441804. [DOI] [PubMed] [Google Scholar]
- 12.Wang C, Zhang X, Bialek S, Cannon MJ. 2011. Attribution of congenital cytomegalovirus infection to primary versus non-primary maternal infection. Clin Infect Dis 52:e11–e13. doi: 10.1093/cid/ciq085. [DOI] [PubMed] [Google Scholar]
- 13.Mussi-Pinhata MM, Yamamoto AY, Moura Brito RM, de Lima Isaac M, de Carvalho e Oliveira PF, Boppana S, Britt WJ. 2009. Birth prevalence and natural history of congenital cytomegalovirus infection in a highly seroimmune population. Clin Infect Dis 49:522–528. doi: 10.1086/600882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pass RF, Fowler KB, Boppana SB, Britt WJ, Stagno S. 2006. Congenital cytomegalovirus infection following first trimester maternal infection: symptoms at birth and outcome. J Clin Virol 35:216–220. doi: 10.1016/j.jcv.2005.09.015. [DOI] [PubMed] [Google Scholar]
- 15.Furione M, Rognoni V, Sarasini A, Zavattoni M, Lilleri D, Gerna G, Revello MG. 2013. Slow increase in IgG avidity correlates with prevention of human cytomegalovirus transmission to the fetus. J Med Virol 85:1960–1967. doi: 10.1002/jmv.23691. [DOI] [PubMed] [Google Scholar]
- 16.Lilleri D, Gerna G, Bruno F, Draghi P, Gabanti E, Fornara C, Meloni F. 2013. Systemic and local human cytomegalovirus-specific T-cell response in lung transplant recipients. New Microbiol 36:267–277. [PubMed] [Google Scholar]
- 17.Boppana SB, Britt WJ. 1995. Antiviral antibody responses and intrauterine transmission after primary maternal cytomegalovirus infection. J Infect Dis 171:1115–1121. doi: 10.1093/infdis/171.5.1115. [DOI] [PubMed] [Google Scholar]
- 18.Rovito R, Korndewal MJ, van Zelm MC, Ziagkos D, Wessels E, van der Burg M, Kroes AC, Langerak AW, Vossen AC. 2017. T and B cell markers in dried blood spots of neonates with congenital cytomegalovirus infection: B cell numbers at birth are associated with long-term outcomes. J Immunol 198:102–109. doi: 10.4049/jimmunol.1601182. [DOI] [PubMed] [Google Scholar]
- 19.Revello MG, Lazzarotto T, Guerra B, Spinillo A, Ferrazzi E, Kustermann A, Guaschino S, Vergani P, Todros T, Frusca T, Arossa A, Furione M, Rognoni V, Rizzo N, Gabrielli L, Klersy C, Gerna G, CHIP Study Group. 2014. A randomized trial of hyperimmune globulin to prevent congenital cytomegalovirus. N Engl J Med 370:1316–1326. doi: 10.1056/NEJMoa1310214. [DOI] [PubMed] [Google Scholar]
- 20.Bialas KM, Tanaka T, Tran D, Varner V, Cisneros De La Rosa E, Chiuppesi F, Wussow F, Kattenhorn L, Macri S, Kunz EL, Estroff JA, Kirchherr J, Yue Y, Fan Q, Lauck M, O'Connor DH, Hall AH, Xavier A, Diamond DJ, Barry PA, Kaur A, Permar SR. 2015. Maternal CD4+ T cells protect against severe congenital cytomegalovirus disease in a novel nonhuman primate model of placental cytomegalovirus transmission. Proc Natl Acad Sci U S A 112:13645–13650. doi: 10.1073/pnas.1511526112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Silveira ELV, Kasturi SP, Kovalenkov Y, Rasheed AU, Yeiser P, Jinnah ZS, Legere TH, Pulendran B, Villinger F, Wrammert J. 2015. Vaccine-induced plasmablast responses in rhesus macaques: phenotypic characterization and a source for generating antigen-specific monoclonal antibodies. J Immunol Methods 416:69–83. doi: 10.1016/j.jim.2014.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Barry PA, Lockridge KM, Salamat S, Tinling SP, Yue Y, Zhou SS, Gospe SM, Britt WJ, Tarantal AF. 2006. Nonhuman primate models of intrauterine cytomegalovirus infection. ILAR J 47:49–64. doi: 10.1093/ilar.47.1.49. [DOI] [PubMed] [Google Scholar]
- 23.Powers CJ, Fruh K. 2008. Signal peptide-dependent inhibition of MHC class I heavy chain translation by rhesus cytomegalovirus. PLoS Pathog 4:e1000150. doi: 10.1371/journal.ppat.1000150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fink K. 2012. Origin and function of circulating plasmablasts during acute viral infections. Front Immunol 3:78. doi: 10.3389/fimmu.2012.00078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bortnick A, Chernova I, Quinn WJ, Mugnier M, Cancro MP, Allman D. 2012. Long-lived bone marrow plasma cells are induced early in response to T cell-independent or T cell-dependent antigens. J Immunol 188:5389–5396. doi: 10.4049/jimmunol.1102808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dörner T, Radbruch A. 2007. Antibodies and B cell memory in viral immunity. Immunity 27:384–392. doi: 10.1016/j.immuni.2007.09.002. [DOI] [PubMed] [Google Scholar]
- 27.Zotos D, Coquet JM, Zhang Y, Light A, D'Costa K, Kallies A, Corcoran LM, Godfrey DI, Toellner K-M, Smyth MJ, Nutt SL, Tarlinton DM. 2010. IL-21 regulates germinal center B cell differentiation and proliferation through a B cell-intrinsic mechanism. J Exp Med 207:365–378. doi: 10.1084/jem.20091777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sylwester AW, Mitchell BL, Edgar JB, Taormina C, Pelte C, Ruchti F, Sleath PR, Grabstein KH, Hosken NA, Kern F, Nelson JA, Picker LJ. 2005. Broadly targeted human cytomegalovirus-specific CD4+ and CD8+ T cells dominate the memory compartments of exposed subjects. J Exp Med 202:673–685. doi: 10.1084/jem.20050882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Harari A. 2002. Analysis of HIV-1- and CMV-specific memory CD4 T-cell responses during primary and chronic infection. Blood 100:1381–1387. doi: 10.1182/blood-2001-11-0080. [DOI] [PubMed] [Google Scholar]
- 30.McVoy MA. 2013. Cytomegalovirus vaccines. Clin Infect Dis 57:S196–S199. doi: 10.1093/cid/cit587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pass RF, Zhang C, Evans A, Simpson T, Andrews W, Huang ML, Corey L, Hill J, Davis E, Flanigan C, Cloud G. 2009. Vaccine prevention of maternal cytomegalovirus infection. N Engl J Med 360:1191–1199. doi: 10.1056/NEJMoa0804749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bernstein DI, Munoz FM, Callahan ST, Rupp R, Wootton SH, Edwards KM, Turley CB, Stanberry LR, Patel SM, McNeal MM, Pichon S, Amegashie C, Bellamy AR. 2016. Safety and efficacy of a cytomegalovirus glycoprotein B (gB) vaccine in adolescent girls: a randomized clinical trial. Vaccine 34:313–319. doi: 10.1016/j.vaccine.2015.11.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Potzsch S, Spindler N, Wiegers AK, Fisch T, Rucker P, Sticht H, Grieb N, Baroti T, Weisel F, Stamminger T, Martin-Parras L, Mach M, Winkler TH. 2011. B cell repertoire analysis identifies new antigenic domains on glycoprotein B of human cytomegalovirus which are target of neutralizing antibodies. PLoS Pathog 7:e1002172. doi: 10.1371/journal.ppat.1002172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zydek M, Petitt M, Fang-Hoover J, Adler B, Kauvar LM, Pereira L, Tabata T. 2014. HCMV infection of human trophoblast progenitor cells of the placenta is neutralized by a human monoclonal antibody to glycoprotein B and not by antibodies to the pentamer complex. Viruses 6:1346–1364. doi: 10.3390/v6031346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yue Y, Zhou SS, Barry PA. 2003. Antibody responses to rhesus cytomegalovirus glycoprotein B in naturally infected rhesus macaques. J Gen Virol 84:3371–3379. doi: 10.1099/vir.0.19508-0. [DOI] [PubMed] [Google Scholar]
- 36.Lilleri D, Kabanova A, Revello MG, Percivalle E, Sarasini A, Genini E, Sallusto F, Lanzavecchia A, Corti D, Gerna G. 2013. Fetal human cytomegalovirus transmission correlates with delayed maternal antibodies to gH/gL/pUL128-130-131 complex during primary infection. PLoS One 8:e59863. doi: 10.1371/journal.pone.0059863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Loughney JW, Rustandi RR, Wang D, Troutman MC, Dick LW Jr, Li G, Liu Z, Li F, Freed DC, Price CE, Hoang VM, Culp TD, DePhillips PA, Fu TM, Ha S. 2015. Soluble human cytomegalovirus gH/gL/pUL128-131 pentameric complex, but not gH/gL, inhibits viral entry to epithelial cells and presents dominant native neutralizing epitopes. J Biol Chem 290:15985–15995. doi: 10.1074/jbc.M115.652230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Freed DC, Tang Q, Tang A, Li F, He X, Huang Z, Meng W, Xia L, Finnefrock AC, Durr E, Espeseth AS, Casimiro DR, Zhang N, Shiver JW, Wang D, An Z, Fu TM. 2013. Pentameric complex of viral glycoprotein H is the primary target for potent neutralization by a human cytomegalovirus vaccine. Proc Natl Acad Sci U S A 110:E4997–5005. doi: 10.1073/pnas.1316517110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wussow F, Yue Y, Martinez J, Deere JD, Longmate J, Herrmann A, Barry PA, Diamond DJ. 2013. A vaccine based on the rhesus cytomegalovirus UL128 complex induces broadly neutralizing antibodies in rhesus macaques. J Virol 87:1322–1332. doi: 10.1128/JVI.01669-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Liao H- X, Levesque MC, Nagel A, Dixon A, Zhang R, Walter E, Parks R, Whitesides J, Marshall DJ, Hwang K-K, Yang Y, Chen X, Gao F, Munshaw S, Kepler TB, Denny T, Moody MA, Haynes BF. 2009. High-throughput isolation of immunoglobulin genes from single human B cells and expression as monoclonal antibodies. J Virol Methods 158:171–179. doi: 10.1016/j.jviromet.2009.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Liao H- X, Chen X, Munshaw S, Zhang R, Marshall DJ, Vandergrift N, Whitesides JF, Lu X, Yu J-S, Hwang K-K, Gao F, Markowitz M, Heath SL, Bar KJ, Goepfert PA, Montefiori DC, Shaw GC, Alam SM, Margolis DM, Denny TN, Boyd SD, Marshal E, Egholm M, Simen BB, Hanczaruk B, Fire AZ, Voss G, Kelsoe G, Tomaras GD, Moody MA, Kepler TB, Haynes BF. 2011. Initial antibodies binding to HIV-1 gp41 in acutely infected subjects are polyreactive and highly mutated. J Exp Med 208:2237–2249. doi: 10.1084/jem.20110363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rivailler P, Kaur A, Johnson RP, Wang F. 2006. Genomic sequence of rhesus cytomegalovirus 180.92: insights into the coding potential of rhesus cytomegalovirus. J Virol 80:4179–4182. doi: 10.1128/JVI.80.8.4179-4182.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Oxford KL, Strelow L, Yue Y, Chang WLW, Schmidt KA, Diamond DJ, Barry PA. 2011. Open reading frames carried on UL/b′ are implicated in shedding and horizontal transmission of rhesus cytomegalovirus in rhesus monkeys. J Virol 85:5105–5114. doi: 10.1128/JVI.02631-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Amos JD, Himes JE, Armand L, Gurley TC, Martinez DR, Colvin L, Beck K, Overman RG, Liao H-X, Moody MA, Permar SR. 2015. Rapid development of gp120-focused neutralizing B cell responses during acute simian immunodeficiency virus infection of African green monkeys. J Virol 89:9485–9498. doi: 10.1128/JVI.01564-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chiuppesi F, Wussow F, Johnson E, Bian C, Zhuo M, Rajakumar A, Barry PA, Britt WJ, Chakraborty R, Diamond DJ. 2015. Vaccine-derived neutralizing antibodies to the human cytomegalovirus gH/gL pentamer potently block primary cytotrophoblast infection. J Virol 89:11884–11898. doi: 10.1128/JVI.01701-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wussow F, Chiuppesi F, Martinez J, Campo J, Johnson E, Flechsig C, Newell M, Tran E, Ortiz J, La Rosa C, Herrmann A, Longmate J, Chakraborty R, Barry PA, Diamond DJ. 2014. Human cytomegalovirus vaccine based on the envelope gH/gL pentamer complex. PLoS Pathog 10:e1004524. doi: 10.1371/journal.ppat.1004524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yue Y, Kaur A, Eberhardt MK, Kassis N, Zhou SS, Tarantal AF, Barry PA. 2007. Immunogenicity and protective efficacy of DNA vaccines expressing rhesus cytomegalovirus glycoprotein B, phosphoprotein 65-2, and viral interleukin-10 in rhesus macaques. J Virol 81:1095–1109. doi: 10.1128/JVI.01708-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.