Abstract
Introduction
Optimizing outcome in biliary atresia (BA) requires timely diagnosis. Cholestasis is a presenting feature of BA, as well as other diagnoses (Non-BA). Identification of clinical features of neonatal cholestasis that would expedite decisions to pursue subsequent invasive testing to correctly diagnose or exclude BA would enhance outcomes. The analytical goal was to develop a predictive model for BA using data available at initial presentation.
Methods
Infants at presentation with neonatal cholestasis (direct/conjugated bilirubin >2 mg/dl [34.2 μM]) were enrolled prior to surgical exploration in a prospective observational multi-centered study (PROBE–NCT00061828). Clinical features (physical findings, laboratory results, gallbladder sonography) at enrollment were analyzed. Initially, 19 features were selected as candidate predictors. Two approaches were used to build models for diagnosis prediction: a hierarchical classification and regression decision tree (CART) and a logistic regression model using a stepwise selection strategy.
Results
In PROBE April 2004-February 2014, 401 infants met criteria for BA and 259 for Non-BA. Univariate analysis identified 13 features that were significantly different between BA and Non-BA. Using a CART predictive model of BA versus Non-BA (significant factors: gamma-glutamyl transpeptidase, acholic stools, weight), the receiver operating characteristic area under the curve (ROC AUC) was 0.83. Twelve percent of BA infants were misclassified as Non-BA; 17% of Non-BA infants were misclassified as BA. Stepwise logistic regression identified seven factors in a predictive model (ROC AUC 0.89). Using this model, a predicted probability of >0.8 (n = 357) yielded an 81% true positive rate for BA; <0.2 (n = 120) yielded an 11% false negative rate.
Conclusion
Despite the relatively good accuracy of our optimized prediction models, the high precision required for differentiating BA from Non-BA was not achieved. Accurate identification of BA in infants with neonatal cholestasis requires further evaluation, and BA should not be excluded based only on presenting clinical features.
Introduction
Neonatal cholestasis is a relatively common clinical issue that presents a complex diagnostic challenge for clinicians [1]. Cholestasis may not be readily identified at its onset and, as such, may present late in the course of the underlying disease process. An expansive differential diagnosis underlies the condition, which challenges one to prioritize diagnostic evaluations in order to sort through a complex set of etiologies in a relatively short time [2]. Shotgun approaches to diagnosis are typically not feasible in infants, while identification of life-threatening and treatable causes of cholestasis is a high priority. Newborn screening has the potential to identify some of the relevant disease processes.
One of the most important and relatively common specific causes of neonatal cholestasis is biliary atresia (BA). Timely diagnosis of BA is ultimately made by cholangiography at the time of exploratory laparotomy and histologic assessment of the surgically-removed bile duct remnant. Such timely diagnosis has the potential to improve clinical outcomes, as earlier hepatic portoenterostomy is associated with longer survival without liver transplantation [3]. Deciding which infants should undergo surgical exploration is critical. Ideally, one would like to minimize the number of infants who undergo unnecessary surgery, while not missing or delaying the diagnosis of BA. There is no universal consensus on the sequential steps to be taken in the diagnostic evaluation of neonatal cholestasis from the time of presentation leading up to exploratory surgery.
The Childhood Liver Disease Research Network (ChiLDReN), a National Institutes of Health-funded consortium, has conducted a prospective longitudinal study of 875 infants presenting with neonatal cholestasis at 15 clinical sites in the United States and Canada over an 11-year period. Data collected included details of the presenting clinical features, demographics, physical findings, laboratory values, and gallbladder sonography results that are typically available in routine clinical practice. Using these data, the objective of this study was to determine the predictive value for BA of typical testing performed in the evaluation of cholestatic infants prior to the decision for invasive testing (e.g., liver biopsy, cholangiography, exploratory laparotomy). A secondary goal was to develop a diagnostic algorithm to help guide the clinician’s decision-making for invasive testing.
Materials and methods
Study population
Between April 2004 and February 2014, infants presenting with neonatal cholestasis were enrolled in a prospective observational study of infants with cholestasis (PROBE: https://clinicaltrials.gov/ct2/show/NCT00061828, conducted by ChiLDReN). Written informed consent was obtained from the study participants’ parents or guardians, and the protocol was carried out under institutional review board (IRB) approval. Given the age of the participants, assent was not feasible. The IRB at each participating institution has approved PROBE (S1 Table). Inclusion criteria were: 1) age ≤180 days at presentation to a ChiLDReN center; and 2) serum direct or conjugated bilirubin >20% of total bilirubin (TB) and ≥2mg/dl. The PROBE protocol permitted the use of laboratory studies drawn prior to enrollment (“presentation”) to be used for inclusion criteria. Exclusion criteria were: 1) acute liver failure; 2) previous hepatobiliary surgery; 3) bacterial or fungal sepsis; 4) hypoxia, shock, or ischemic hepatopathy; 5) malignancy; 6) primary hemolytic disease; 7) drug or total parenteral nutrition-associated cholestasis; 8) extracorporeal membrane oxygenation (ECMO)-associated cholestasis; or 9) birth weight <1500g in an infant who did not have BA. Presenting clinical features (including stool color), demographics, physical findings, laboratory data, and gallbladder sonography findings were collected prospectively and recorded prior to the ultimate assignment of a clinical diagnosis. Evaluations of neonatal cholestasis were not prescribed and were according to local practice and conducted at local facilities.
Not all participants enrolled in PROBE were included in this analysis of predictors of BA. Participants were included only if they had laboratory studies indicating direct/conjugated hyperbilirubinemia that were performed at the time of “presentation” to the ChiLDReN clinical site. Inclusion in the BA cohort (Group 1) for this analysis required either the performance of a biliary drainage procedure for BA or exploratory surgery with the finding of an atretic extrahepatic bile duct by either inspection or attempted cholangiography. BA could not be definitively “confirmed” in infants who presented “late” in the clinical course and in whom clinicians determined that laparotomy or laparoscopy would not benefit the child or alter management. Inclusion in the Non-BA cohort (Group 2) required the identification of a specific alternative etiology for their cholestasis or cholangiography that excluded BA. For an infant with the clinical diagnosis of idiopathic neonatal hepatitis (INH) or idiopathic cholestasis (IC) to be included in this analysis, resolution of cholestasis was required as defined by a subsequent TB <1.0 mg/dL at >120 days of age (without hepatic portoenterostomy). INH was defined as neonatal cholestasis in which histologic evidence of giant cell hepatitis was present on liver biopsy and for whom no other etiology was confirmed. IC was defined as neonatal cholestasis that resolved in an infant who did not undergo liver biopsy or did not have giant cell hepatitis on a liver biopsy, and for whom no other etiology was confirmed. The outcome variable for this study is a confirmed study definition meeting diagnosis of BA or Non-BA (i.e., Group 1 vs. Group 2).
Candidate predictors
Twenty-two variables collected at the time of the first evaluation at the ChiLDReN center were considered as candidate predictors, including age at disease onset and first evaluation, sex, race, ethnicity, anthropometrics (weight z-score, height z-score, head circumference z-score), palpable liver (including number of centimeters below the costal margin at the midclavicular line), palpable spleen, acholic stools, Alagille “syndromic” facial features, serum TB (defined as conjugated + unconjugated when total not measured), conjugated/direct bilirubin, alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma-glutamyl transpeptidase (GGTP), albumin, platelet count, cholesterol, and gallbladder sonography (presence or absence of the gallbladder, “small” gallbladder equated with presence). Age at first evaluation was defined as the earliest date among dates of study informed consent, diagnosis, or surgery; age at disease onset was defined as the earliest age at which there was caregiver reported icterus of eyes or skin, darkening of urine, or white/pale stools in the initial history case report form.
Statistical analysis
Descriptive statistics for the characteristics listed above were provided for BA and Non-BA subjects included in the model development and those not included (Group 3 = BA not included and Group 4 = Non-BA not included). Differences between Groups 1 and 2 were assessed using two sample t-tests for the continuous parameters. Variables with skewed distributions were analyzed after first applying a log transformation, with the accompanying descriptive statistics reported on the original scale. Categorical variables were assessed using a Chi-Square test or Fisher’s exact test, where cell size(s) were ≤5 participants.
Model development
Two types of model were used to find the best prediction models: a hierarchical classification and regression tree (CART) and a logistic regression model [4]. All 22 factors mentioned above were considered by both approaches, regardless of whether or not they obtained statistical significance in the univariate setting. CART analysis recursively partitions observations to define the optimum cutoff point for continuous predictors and identifies homogeneous groups having the largest difference in the outcome variable (minimum misclassification error rate). Each partition is a binary split based on a single independent variable. This process results in a classification rule with the optimum cut point for continuous variables and is represented as a tree. Once the full tree was grown, a pruning algorithm was run to avoid over-fitting. In the pruning process, the chi-square statistic for 2x2 contingency tables was calculated for each split. Using a pre-selected alpha level (p = 0.10), nodes whose chi-square values–as well as the chi-square values of subsequent splits–did not exceed the predetermined threshold were pruned.
A logistic regression prediction model was constructed using a forward stepwise hierarchical approach, with higher than standard p value, α = 0.10 [5–7]. To avoid losing study sample due to missing data, a sequential regression imputation method was used to impute missing values [8]. Only one randomly selected imputed data set was used for model development [9]. To define appropriate transformation of continuous variables, we used penalized-spline functions to explore the potential nonlinear effect of potential continuous predictors [10]. Potential interaction effects identified through CART analysis were considered in the model development process. The final model consists of only variables maintaining a 0.10 level of significance.
Model evaluation
The ability of the multivariate model to correctly classify patients into the dichotomous disease classification (BA vs. Non-BA) was determined by assessing the area under the receiver operating characteristic (ROC) curve (AUC), where larger values on the 0–1 scale indicate greater concordance between the predicted and observed disease groups. Reapplying the model to our data, we further evaluated the disease misclassification rates at what are considered more definitive predicted probability thresholds.
The CART analysis was performed using R (version 3.2.2) software. Data imputation and all other analyses were conducted using SAS (version 9.3)[4].
Results
During the study period, 875 infants with neonatal cholestasis were enrolled in PROBE. Strict criteria for BA and Non-BA inclusion were used in this analysis to increase the confidence for the predictive value of variables tested. Thus, 401 infants (Group 1) met criteria for the study definition of BA; 102 participants were classified clinically as BA by the study site, but after review of laboratory and operative data at presentation, these patients did not meet the strict study definition of BA and were excluded from analysis (Group 3: 58 excluded for lack of laboratory data at presentation and 44 for lack of operative demonstration of BA). Groups 1 and 3 were generally similar, except for a skewing of data to a “late” presentation in Group 3, which likely accounted for the decision to not proceed with hepatic portoenterostomy, thereby excluding those infants from Group 1 (S2 Table).
There were 259 of 372 infants enrolled in PROBE who did not have a clinical diagnosis of BA and met study criteria for Non-BA (Group 2). There were 113 infants (Group 4) with a clinical diagnosis of Non-BA excluded from analysis for potentially more than one reason, including: 1) inability to definitively exclude BA because, despite having a clinical diagnosis of indeterminate/IC, INH, choledochal cyst, or “other”, either TB was still elevated (>1 mg/dL) beyond 120 days of age and/or there was no cholangiographic evidence of bile duct patency; 2) laboratory data were not available at presentation; and 3) laboratory data at presentation did not meet PROBE entry criteria. Groups 2 and 4 were similar (S3 Table). The clinical phenotype in Group 4 may have been milder, with less apparent hepatomegaly and lower biochemical markers of liver disease (TB, direct bilirubin, conjugated bilirubin, ALT, and AST).
Diagnoses in the 259 Non-BA infants who met study criteria (Group 2) included IC (n = 72), INH (n = 61), alpha-1 antitrypsin deficiency (n = 31), Alagille syndrome (n = 28), panhypopituitarism (n = 12), cytomegalovirus infection (n = 10), bile duct paucity (n = 10), progressive familial intrahepatic cholestasis (n = 8), cystic fibrosis (n = 6), mitochondrial disease (n = 6), bile acid synthesis defect (n = 5), and other (n = 8; 1 each for hemophagocytic lymphohistiocytosis, hereditary spherocytosis, neonatal ascites, Caroli’s disease, perinatal sclerosing cholangitis, porphyria, hyperinsulinism, and duplicate gall bladder). The demographics, salient clinical features, and laboratory values of the BA and Non-BA groups obtained at presentation at the ChiLDReN sites are displayed in Table 1.
Table 1. Comparison of clinical information at presentation between infants with and without BA.
| Variable | Group 1 (BA Included) % or Mean (SD) N = 401 | Group (Non-BA Included) % or Mean (SD) N = 259 | p-value |
|---|---|---|---|
| Race | 0.130 | ||
| White | 244 (63%) | 156 (61.2%) | |
| Black | 61 (15.8%) | 51 (20%) | |
| Asian | 36 (9.3%) | 13 (5.1%) | |
| Other | 46 (11.9%) | 35 (13.7%) | |
| Sex | <0.001 | ||
| Male | 191 (47.6%) | 164 (63.3%) | |
| Female | 210 (52.4%) | 95 (36.7%) | |
| Ethnicity | 0.146 | ||
| Hispanic | 92 (23%) | 60 (23.3%) | |
| Non-Hispanic | 308 (77%) | 197 (76.7%) | |
| Age at First Evaluation (Days) | N = 401 | N = 259 | 0.176 |
| 63.5 (30.9) | 60 (33.3) | ||
| Age at Disease Onset (Days) | N = 401 | N = 259 | <0.001 |
| 12.8 (18.5) | 18.7 (22.1) | ||
| Weight (kg) | N = 398 | N = 257 | <0.001 |
| 4.5 (0.9) | 4.1 (1.1) | ||
| Length (cm) | N = 381 | N = 252 | <0.001 |
| 55.5 (4) | 54.3 (4.3) | ||
| Head Circumference (cm) | N = 336 | N = 215 | 0.015 |
| 37.6 (2.2) | 37.1 (2.6) | ||
| Weight Z-Score | N = 398 | N = 257 | <0.001 |
| -1 (1) | -1.5 (1.2) | ||
| Length Z-Score | N = 381 | N = 252 | <0.001 |
| -0.8 (1.5) | -1.4 (1.5) | ||
| Head Circumference Z-Score | N = 336 | N = 215 | 0.026 |
| -1.1 (1.6) | -1.4 (1.2) | ||
| Acholic Stools | <0.001 | ||
| Absent | 69 (17.6%) | 165 (66%) | |
| Present | 322 (82.4%) | 85 (34%) | |
| Acholic Stools (3 Levels) | <0.001 | ||
| Normal | 69 (17.6%) | 165 (66%) | |
| White or Gray | 184 (47.1%) | 30 (12%) | |
| Pale | 138 (35.3%) | 55 (22%) | |
| Facial Features | <0.001 | ||
| Normal | 380 (95.2%) | 207 (81.2%) | |
| Abnormal | 19 (4.8%) | 48 (18.8%) | |
| Liver Edge Palpable | 0.004 | ||
| Not Palpable | 26 (7.3%) | 34 (14.5%) | |
| Palpable | 332 (92.7%) | 201 (85.5%) | |
| Liver Edge Below Costal Margin (cm) | N = 334 | N = 202 | <0.001 |
| 3.3 (1.6) | 2.5 (1.4) | ||
| Spleen Palpable | 0.018 | ||
| Not Palpable | 188 (50%) | 149 (59.6%) | |
| Palpable | 188 (50%) | 101 (40.4%) | |
| Direct Baseline Bilirubin (mg/dL) | N = 239 | N = 162 | 0.335 |
| 5.7 (2.2) | 5.8 (3.2) | ||
| Conjugated Baseline Bilirubin (mg/dL) | N = 215 | N = 121 | 0.871 |
| 4.3 (1.6) | 4.6 (2.6) | ||
| Total Baseline Bilirubin (mg/dL) | N = 401 | N = 259 | 0.979 |
| 8.3 (3.1) | 8.6 (4.3) | ||
| AST (u/L) | N = 397 | N = 254 | 0.304 |
| 232.1 (206.4) | 284.2 (347.7) | ||
| ALT (u/L) | N = 400 | N = 255 | 0.230 |
| 154.7 (124.3) | 190.7 (232.5) | ||
| Albumin (g/dL) | N = 391 | N = 246 | 0.006 |
| 3.6 (0.5) | 3.5 (0.6) | ||
| GGTP (u/L) | N = 379 | N = 238 | <0.001 |
| 711.9 (537.5) | 299 (380.5) | ||
| Platelets (103/ mm3) | N = 380 | N = 243 | 0.022 |
| 445.2 (180.2) | 419.7 (197.3) | ||
| Alkaline Phosphatase (IU/L) | N = 395 | N = 254 | 0.230 |
| 568.6 (320.7) | 572.1 (252.1) | ||
| Total Cholesterol (mg/dL) | N = 33 | N = 54 | 0.925 |
| 184.2 (61.3) | 190.6 (82.3) | ||
| Gallbladder | <0.001 | ||
| Absent | 125 (39.9%) | 13 (6.5%) | |
| Present | 5 (1.6%) | 1 (1.4%) | |
| Present (Small) | 142 (45.4%) | 81 (40.7%) | |
| Normal | 41 (13.1%) | 105 (52.8%) | |
| Gallbladder (Absent vs. Present) | <0.001 | ||
| Absent | 125 (39.9%) | 13 (6.5%) | |
| Present | 188 (60.1%) | 186 (93.5%) |
Univariate analysis identified 13 variables (Table 1), which were significantly different (in bold) between BA and Non-BA (Group 1 vs. Group 2), including age at disease onset, stool color, sex, facial features, weight z-score, length z-score, head circumference z-score, centimeters of liver palpable below the costal margin, palpable spleen, GGTP, albumin, platelet count, and gallbladder sonography. Infants with BA were more likely to have acholic stools, to be female, to be younger at disease onset, have greater z-score growth parameters, have normal facial features, more significant hepatosplenomegaly, a higher GGTP, albumin, and platelet count, and a sonographically absent gallbladder.
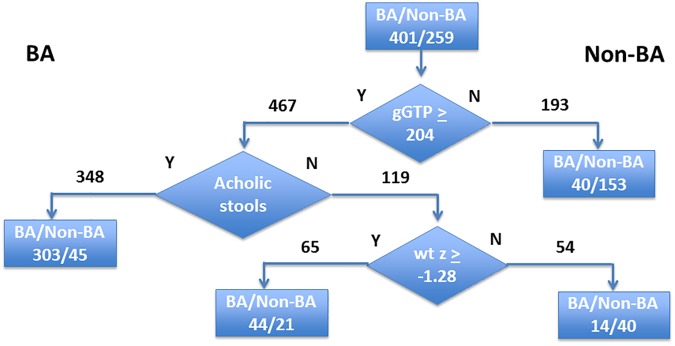
We used a hierarchical CART analysis to create an algorithm that could distinguish BA from Non-BA. In this approach, the population was segregated into either BA or Non-BA in a stepwise manner based on the single most predictive variable, using a threshold value derived empirically from the observed data. After this initial segregation, each newly-created sub-population was again evaluated using the most predictive variable that was redefined for this new subset of the population. In this manner, the predictive power of each variable was maximized at each step. The process of segregation and reanalysis was continued until there was no further improvement in the overall predictive power for the population. The results of this analysis are shown in Fig 1.
Fig 1. Hierarchical CART analysis of the prediction of BA.
A pruned model is shown that uses GGTP level (cut-off 203.5 IU/L), acholic stools, and wt z-score (cut-off -1.28) to segregate BA from Non-BA as indicated.
If the initial discriminator was a GGTP of 204 IU/L, those with lower levels were unlikely to have BA (40 [21%] out of 193 infants). In those with GGTP ≥204 IU/L and acholic stools, BA was likely (303 out of 467 infants). Further discrimination was achieved by incorporating weight z-score. Overall, the predictive capacity for this model was somewhat worse than the logistic regression modeling, with an AUC for the ROC of 0.831. When the three-variable CART analysis was utilized, 12% of infants categorized as Non-BA (n = 247) were misclassified and had BA. Conversely, 17.5% of infants categorized as BA (n = 415) were misclassified and did not have BA.
The best logistic regression model selected included nine predictors: sex, acholic stools, normal facial features, ALT, GGTP, age at disease onset, weight z-score, palpable liver below the costal margin, and a sonographically absent gallbladder, which were associated with a diagnosis of BA (Table 2).
Table 2. Multivariate logistic regression analysis of factors predicting BA.
| Variable | β | Odds Ratio | 95% Confidence Interval | p-value |
|---|---|---|---|---|
| Intercept | -0.367 | |||
| Age at Onset (Days) | -0.011 | 0.989 | (0.98, 1) | 0.0514 |
| Weight Z-Score | 0.305 | 1.357 | (1.11, 1.67) | 0.003 |
| Liver Below Costal Margin (cm) | 0.320 | 1.377 | (1.19, 1.60) | <0.0001 |
| ALT (IU/L) | -0.002 | 0.998 | (0.997, 1) | 0.0265 |
| GGTP(IU/L) | 0.002 | 1.002 | (1.001, 1.002) | <0.0001 |
| Sex: Male vs. Female | -0.312 | 0.540 | (0.35, 0.83) | 0.0049 |
| Acholic Stools: | <0.0001 | |||
| Pale vs. Normal | 0.252 | 4.775 | (2.9, 7.88) | |
| White/Gray vs. Normal | 1.061 | 10.725 | (6.17, 18.66) | |
| Facial Features: Abnormal vs. Normal | -0.755 | 0.221 | (0.10, 0.48) | 0.0001 |
| Gallbladder: Present vs. Absent | -0.820 | 0.194 | (0.11, 0.35) | <0.0001 |
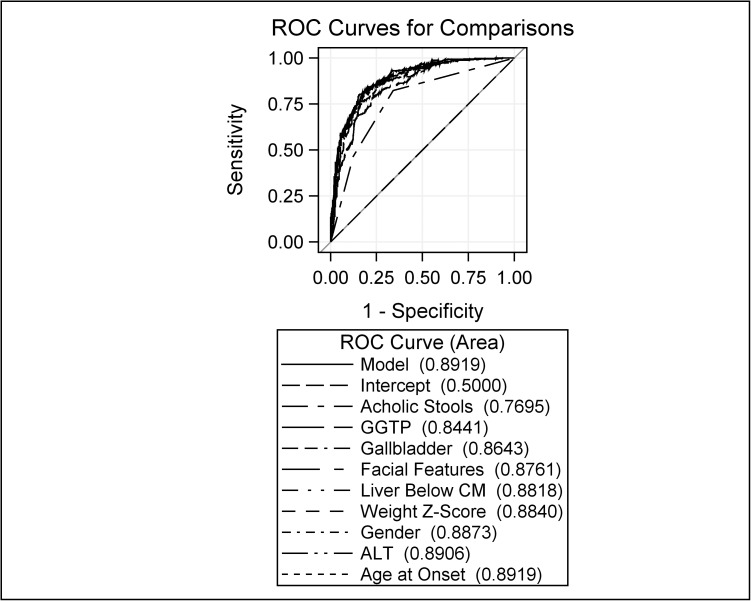
Model discriminating ability was assessed by the ROC curve. Larger values on the 0–1 scale indicated a better predictive model. The final model yielded an AUC for the ROC analysis of 0.892 (Fig 2).
Fig 2. Receiver operator curve analysis of a multivariate model to predict the diagnosis of BA.
The blue solid line is for the final nine-level model. The rest of the curves indicate AUC for a series of models obtained in the stepwise selection procedure. In stepwise order: intercept only, acholic stools, GGTP, gallbladder absence, absence of abnormal facial features, centimeters of liver palpable below the costal margin, weight z-score, sex, ALT, and age of disease onset.
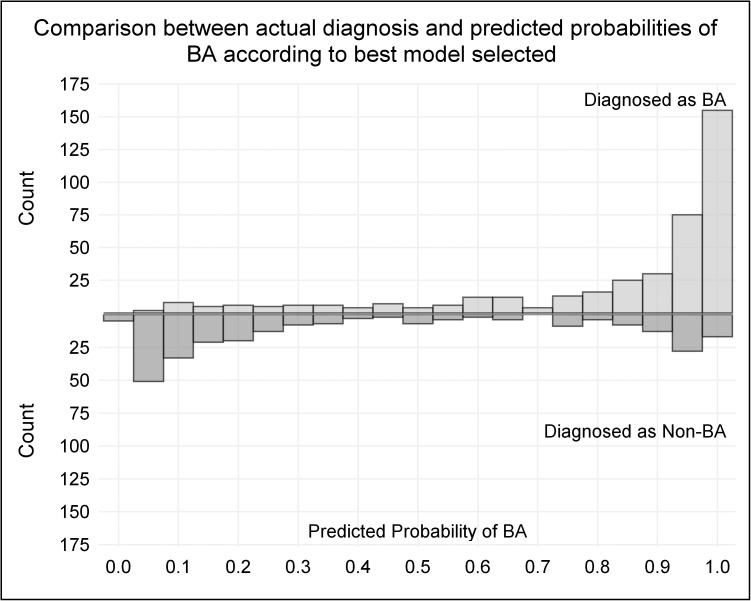
If all 22 candidate predictor features were incorporated into the model, the AUC of the ROC increased marginally to 0.898. Based upon the final model [logit(p) = -0.367–0.011*Age at Onset (Days) + 0.305*Weight Z-Score + 0.320*Liver Below Costal Margin—0.002*ALT(IU/L) + 0.002*GGTP (IU/L)—0.312*Male + 0.252*Pale Stools + 1.061*White/Gray Stools—0.755*Abnormal Facial Features—0.820*Present Gallbladder], a predicted probability of BA was calculated, with 1 indicating the highest chance (100%) of being BA, and 0 being the lowest (0%). The distribution of predicted probabilities for BA and actual study diagnoses of BA and Non-BA are displayed in Fig 3.
Fig 3. Logistic regression model of predicted probability of BA.
Based upon a nine-feature model, a predicted probability of BA was calculated for each participant, with increased probability of BA as the score increased from 0 to 1. The number of participants with the probability scores is shown on the figure, with those with BA above the horizontal line and those with Non-BA below the line.
Three-hundred fifty-seven infants had a predicted probability >0.8, of whom 290 had BA (81.2%). Of the 67 remaining Non-BA infants (19%) with a predicted probability of >0.8, 12 had alpha-1 antitrypsin deficiency, and 10 had Alagille syndrome (Table 3). One-hundred thirty-six infants had a predicted probability of <0.2, of whom 120 had Non-BA (88.2%). Sixteen infants (12%) with scores <0.2 had BA and were evaluated at mean of 63 days of age; most had normally pigmented stools and gallbladder that was present. One-hundred sixty-seven infants had intermediate predicted probability scores between 0.2 and 0.8.
Table 3. Demographics, clinical, and laboratory profile of infants with BA predicted probability >0.8 or <0.2 (BA vs. Non-BA).
| Predicted Probability | Parameter | Group 1 (BA) | Group 2 (Non-BA) | Overall | |||
|---|---|---|---|---|---|---|---|
| N | Mean (SD) or % | N | Mean (SD) or % | N | Mean (SD) or % | ||
| ≥0.80 | Age at First Evaluation (Days) | 290 | 64.6 (31.2) | 67 | 60.6 (36.6) | 357 | 63.9 (32.3) |
| Age at Disease Onset (Days) | 11.8 (17.2) | 15.7 (22.1) | 12.5 (18.3) | ||||
| Weight Z-Scores | -0.8 (1.0) | -1.3 (1.0) | -0.9 (1.0) | ||||
| Liver Edge Below Costal Margin (cm) | 3.4 (1.7) | 2.6 (1.5) | 3.2 (1.7) | ||||
| ALT | 155 (111) | 191 (169) | 162 (124) | ||||
| GGTP | 810 (566) | 478 (543) | 748 (576) | ||||
| Sex | |||||||
| Male | 126 | 43.5% | 39 | 58.2% | 165 | 46.2% | |
| Female | 164 | 56.6% | 28 | 41.8% | 192 | 53.8% | |
| Acholic Stools | |||||||
| Normal | 9 | 3.1% | 4 | 6.0% | 13 | 3.6% | |
| White or Gray | 137 | 47.2% | 7 | 10.5% | 144 | 40.3% | |
| Pale | 144 | 49.7% | 56 | 83.6% | 200 | 56.0% | |
| Facial Features | |||||||
| Normal | 276 | 95.2% | 54 | 80.6% | 330 | 92.4% | |
| Dysmorphic | 14 | 4.8% | 13 | 19.4% | 27 | 7.6% | |
| Gallbladder | |||||||
| Absent | 142 | 49.0% | 11 | 16.4% | 153 | 42.9% | |
| Present | 148 | 51.0% | 56 | 83.6% | 204 | 57.1% | |
| Diagnosis | |||||||
| BA | 285 | 98.3% | 0 | 0 | 285 | 79.8% | |
| Alpha1-Antitrypsin Deficiency | 0 | 0 | 12 | 17.9% | 12 | 3.4% | |
| Hereditary Fructose Intolerance | 0 | 0 | 1 | 1.5% | 1 | 0.3% | |
| Cystic Fibrosis | 0 | 0 | 1 | 1.5% | 1 | 0.3% | |
| PFIC 1, 2, or 3 | 0 | 0 | 1 | 1.5% | 1 | 0.3% | |
| Alagille Syndrome | 1 | 0.3% | 10 | 14.9% | 11 | 3.1% | |
| Operable-Extrahepatic BA and Choledochal Cyst | 2 | 0.7% | 0 | 0 | 2 | 0.6% | |
| INH | 0 | 0 | 15 | 22.4% | 15 | 4.2% | |
| Cholestasis, Indeterminate | 0 | 0 | 18 | 26.9% | 18 | 5.0% | |
| Other | 1 | 0.3% | 8 | 11.9% | 9 | 2.5% | |
| Choledochal Cyst | 1 | 0.3% | 1 | 1.5% | 2 | 1.5% | |
| ≤0.20 | Age at First Evaluation (Days) | 16 | 62.8 (30.8) | 120 | 56.7 (30.3) | 136 | 57.4 (30.3) |
| Age at Disease Onset (Days) | 29.0 (31.4) | 22.7 (23.2) | 23.4 (24.3) | ||||
| Weight Z-Scores | -1.8 (1.1) | -1.8 (1.1) | -1.8 (1.1) | ||||
| Liver Edge Below Costal Margin (cm) | 1.9 (1.5) | 1.9 (1.1) | 1.9 (1.1) | ||||
| ALT | 199 (202) | 194 (285) | 194 (276) | ||||
| GGTP | 289 (174) | 182 (183) | 194 (185) | ||||
| Sex | |||||||
| Male | 6 | 37.5% | 84 | 70.0% | 90 | 66.2% | |
| Female | 10 | 62.5% | 36 | 30.0% | 46 | 33.8% | |
| Acholic Stools | |||||||
| Normal | 15 | 93.8% | 119 | 99.2% | 134 | 98.5% | |
| White or Gray | 1 | 6.3% | 1 | 0.8% | 2 | 1.5% | |
| Pale | 0 | 0 | 0 | 0 | 0 | 0 | |
| Facial Features | |||||||
| Normal | 15 | 93.8% | 95 | 79.2% | 110 | 80.9% | |
| Dysmorphic | 1 | 6.3% | 25 | 20.8% | 26 | 19.1% | |
| Gallbladder | |||||||
| Absent | 1 | 6.3% | 2 | 1.7% | 3 | 2.2% | |
| Present | 15 | 93.8% | 118 | 98.3% | 133 | 97.8% | |
| Diagnosis | |||||||
| BA | 12 | 75.0% | 0 | 0 | 12 | 8.8% | |
| Alpha1-Antitrypsin Deficiency | 0 | 0 | 9 | 7.5% | 9 | 6.6% | |
| Storage Diseases | 1 | 6.3% | 0 | 0 | 1 | 0.7% | |
| Cystic Fibrosis | 0 | 0 | 1 | 0.8% | 1 | 0.7% | |
| PFIC 1, 2, or 3 | 0 | 0 | 2 | 1.7% | 2 | 1.5% | |
| Alagille Syndrome | 1 | 6.3% | 5 | 4.2% | 6 | 4.4% | |
| Bile Acid Synthetic Disorder | 0 | 0 | 1 | 0.8% | 1 | 0.7% | |
| INH | 0 | 0 | 32 | 26.7% | 32 | 23.5% | |
| Cholestasis, Indeterminate | 0 | 0 | 36 | 30.0% | 36 | 26.5% | |
| Other | 0 | 0 | 28 | 23.3% | 28 | 20.6% | |
| Hepatitis Due to CMV | 0 | 0 | 6 | 5.0% | 6 | 4.4% | |
| Choledochal Cyst | 2 | 12.5% | 0 | 0 | 2 | 1.5% | |
Discussion
The quest for finding clinical and laboratory features that distinguish BA from other causes of neonatal cholestasis has been ongoing for over 50 years [11–20]. Early investigations of over 800 infants in five separate reports from Boston, Toronto, London, Houston, and Bicêtre demonstrated a difficulty in clinically distinguishing BA from intrahepatic cholestasis in a significant number of infants [11–15]. Infants with BA more frequently had acholic stools, had less failure to thrive, and had more pronounced elevation in biochemical markers of bile duct and canalicular injury, although these features were not uniformly discriminative. More recent reports have added radiologic and histologic features to the investigative paradigm [17–19]. Most of these studies have been single or two-center studies and retrospective in nature.
The current analysis is based on data obtained in a large, truly multi-centered prospective study, which was particularly rigorous with regard to the study definition of BA and Non-BA and with the application of advanced statistical modeling methods. The purpose of the current study was to attempt to develop a diagnostic algorithm that could distinguish between BA and Non-BA using non-invasive parameters that were typically obtained during initial clinical evaluation of cholestatic infants. An effective algorithm might serve as a guide to physicians as to whether invasive procedures, such as liver biopsy and exploratory laparotomy, are warranted. The three variables in the CART analysis (serum GGTP, acholic stools, and weight z-score) that were statistically derived to achieve the best prediction of BA are simple, mostly objective, and readily available early in the course of the evaluation of cholestasis. Accurate classification of the stool pigmentation is the only somewhat subjective parameter in this algorithm [21]; however, recent simple smartphone technology may overcome this [22]. The predicted probability model that was developed achieved accurate diagnosis of BA in 290 out of 357 cases (81%) when the predictive probability was >0.8. Accuracy in these cases might be enhanced if alpha-1 antitrypsin levels and phenotype were readily available, and if features of Alagille syndrome were carefully assessed. One could argue that, for the infants with a predictive probability of >0.8 who had negative diagnostic testing for alpha-1 antitrypsin deficiency and Alagille syndrome, the next logical step would be exploratory laparotomy, and one might defer liver biopsy. An accurate diagnosis of Non-BA was predicted in 120 of 136 cases (88%) when the predicted probability was <0.2. Conversely, an unsettling number of these infants had BA, whose diagnosis would be delayed or missed if one relied solely on these presenting clinical features to “exclude” BA. In addition, a significant number of infants had intermediate predicted probability scores between 0.2 and 0.8 and could not be classified as either BA or Non-BA.
It is clear from the current detailed analysis that clinicians should be very cautious about either diagnosing or excluding BA on the basis of presenting clinical features in infants with cholestasis. Family history is typically noninformative, but in selected circumstances can direct investigations toward specific inherited disorders like Alagille syndrome or familial intrahepatic cholestasis. Additional diagnostic investigations are typically warranted, and noninvasive approaches are often the first to be considered [23]. In the current study, only the presence of gallbladder was considered on ultrasonography. More detailed evaluation for the triangular cord sign, gallbladder wall characteristics, and hepatic subcapsular blood flow were not conducted, although may have increased the accuracy of the predictive model [18, 24–26]. Hepatobiliary scintigraphy may be especially useful in excluding BA when intestinal excretion of radiotracer is demonstrated, although nonexcretion is less helpful since it is observed in BA and Non-BA [27]. Thus, in 60 of 67 cases where a predictive value of >0.8 erroneously suggested BA, stools were pale or normal; in such infants, hepatobiliary scintigraphy may have been useful.
The current analysis did not attempt to determine the added value of liver histology in the predictive algorithm, as the focus was to determine the predictive value of tests performed prior to subjecting infants to invasive testing. Liver histology can be quite informative in the evaluation of neonatal cholestasis, although false negative rates are disturbing given the consequences of late or missed diagnosis of BA [28, 29]. In addition, the exposure of infants unnecessarily to anesthesia (for liver biopsy, cholangiography, or laparotomy) has become a relevant issue in light of recent reports of potential long-term neurodevelopmental sequelae of general anesthesia in young children [30]. Clinicians should consider this issue when deciding about diagnostic testing that may require general anesthesia, including liver biopsy and endoscopic, percutaneous, or intraoperative cholangiography.
Conclusions
In conclusion, early accurate diagnosis of BA remains challenging. Clinicians are obliged to categorically exclude BA in the setting of neonatal cholestasis, since failure to make this diagnosis has potentially profound adverse consequences. This rigorous prospective analysis of presenting features in neonatal cholestasis was unable to generate a diagnostic algorithm that yielded sufficient ability to discriminate between BA and Non-BA in all patients. Early referral to a specialist, with consideration for possible liver biopsy or intraoperative cholangiography, needs to be entertained as soon as cholestasis is identified. Caution should be exercised in excluding BA based only on clinical non-invasive features. The identification of an alternative definitive diagnosis makes BA unlikely, although the Kasai hepatoportoenterostomy has been performed mistakenly in some infants with alternative diagnoses, including cystic fibrosis, alpha-1 antitrypsin deficiency, and Alagille syndrome [31–35]. Although not necessary for all infants with neonatal cholestasis, surgical exploration with operative cholangiography and/or pathologic examination of a bile duct remnant remains the only definitive means of making the diagnosis of BA.
Supporting information
(DOCX)
(DOCX)
(DOCX)
Acknowledgments
Membership of the Childhood Liver Disease Research Network
The following individuals (who did not contribute as coauthors of this manuscript) are instrumental in the planning and conduct of the Childhood Liver Disease Research Network (ChiLDReN).
Paula M. Hertel; Pediatric Gastroenterology, Hepatology and Nutrition; Baylor College of Medicine; Houston, Texas, United States
Estella M. Alonso; Division of Pediatric Gastroenterology, Hepatology and Nutrition; Ann & Robert H. Lurie Children’s Hospital; Chicago, Illinois, United States
Emily M. Fredericks; Division of Child Behavioral Health; University of Michigan and CS Mott Children’s Hospital; Ann Arbor, Michigan, United States
Barbara H. Haber; Infectious Disease, Clinical Research; Merck; North Wales, Pennsylvania, United States
Kasper S. Wang; Division of Pediatric Surgery; Children's Hospital Los Angeles; Los Angeles, California, United States
Lisa G. Sorensen; Ann & Robert H. Lurie Children’s Hospital of Chicago; Northwestern University Feinberg School of Medicine; Chicago, Illinois, United States
Vicky Lee Ng; Division of Pediatric Gastroenterology, Hepatology and Nutrition; The Hospital for Sick Children; University of Toronto; Toronto, Ontario, Canada
Lee Bass; Pediatrics Division of Gastroenterology, Hepatology, and Nutrition; Ann and Robert H Lurie Children's Hospital of Chicago; Chicago, Illinois, United States
Henry Lin; Children’s Hospital of Philadelphia; Philadelphia, Pennsylvania, United States
Nathan P. Goodrich; Arbor Research Collaborative for Health; Ann Arbor, Michigan, United States
Kieran Hawthorne; Arbor Research Collaborative for Health; Ann Arbor, Michigan, United States
James E. Heubi; Division of Pediatric Gastroenterology, Hepatology and Nutrition; Cincinnati Children’s Hospital Medical Center; Cincinnati, Ohio, United States
Rachel Sheridan; Cincinnati Children’s Hospital Medical Center; Cincinnati, Ohio, United States
Lin Fei; Cincinnati Children’s Hospital Medical Center; Cincinnati, Ohio, United States
Jeffrey Teckman; Saint Louis University; Cardinal Glennon Children's Medical Center; St. Louis, Missouri, United States
Catherine A. Spino; Department of Biostatistics; University of Michigan; Ann Arbor, Michigan, United States
Collaborators
Heather Van Doren, MFA, senior medical editor with Arbor Research Collaborative for Health, provided editorial assistance on this manuscript.
Data Availability
This study is funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). The mechanism for public access to data is via the NIDDK Data Repository (https://www.niddkrepository.org/home/). Use of data via this mechanism is subject to standard NIDDK policies and procedures regarding public access databases and supporting documentation. The NIDDK Data Repository is overseen and managed by Information Management Services (IMS), Inc. IMS anticipates the data supporting the submitted manuscript to be publicly available on or before March 10, 2017. Secure Data Exchange for the NIDDK Data Repository is at https://dataexchange.imsweb.com/general/niddkrepository.
Funding Statement
Funding provided by the following National Institute of Diabetes and Digestive and Kidney Diseases (https://www.niddk.nih.gov/) grants: U01DK103149 (BLS); U01DK062456 (JCM); U01DK062470 and UL1TR000454 (SJK); U01DK062453 and UL1TR001082 (RJS); U01DK062436 and UL1TR000150 (PFW); U01DK062503, UL1TR000424 (KS); U01DK062497 and UL1TR000077 (JAB); U01DK062445 (NK RA); U01DK062500 and UL1TR000004 (PR); U01DK062452 and UL1TR000448 (YPT); U01DK084536 and UL1TR001108 (JPM); U01DK084575, UL1TR000423 and UL1RR025014 (KFM); U01DK062466 and UL1TR000005 (RHS); U01DK103135 (BMK); U01DK062481 (KML); U01DK103140 (SLG). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. National Institute of Diabetes and Digestive and Kidney Diseases, U01DK103149, Dr. Benjamin L. Shneider National Institute of Diabetes and Digestive and Kidney Diseases, U01DK062445, Dr. Nanda Kerkar Dr. Ronen Arnon National Institute of Diabetes and Digestive and Kidney Diseases, U01DK062456, Dr. John C. Magee National Institute of Diabetes and Digestive and Kidney Diseases, U01DK062470, Dr. Saul J. Karpen National Institute of Diabetes and Digestive and Kidney Diseases, UL1TR000454, Dr. Saul J. Karpen National Institute of Diabetes and Digestive and Kidney Diseases, U01DK103135, Dr. Binita M. Kamath National Institute of Diabetes and Digestive and Kidney Diseases, U01DK084536, Dr. Jean P. Molleston National Institute of Diabetes and Digestive and Kidney Diseases, UL1TR001108, Dr. Jean P. Molleston National Institute of Diabetes and Digestive and Kidney Diseases, U01DK062497, Dr. Jorge A. Bezerra National Institute of Diabetes and Digestive and Kidney Diseases, UL1TR000077, Dr. Jorge A. Bezerra National Institute of Diabetes and Digestive and Kidney Diseases, U01DK084575, Dr. Karen F. Murray National Institute of Diabetes and Digestive and Kidney Diseases, UL1TR000423, Dr. Karen F. Murray National Institute of Diabetes and Digestive and Kidney Diseases, UL1RR025014, Dr. Karen F. Murray National Institute of Diabetes and Digestive and Kidney Diseases, U01DK062481, Dr. Kathleen M. Loomes National Institute of Diabetes and Digestive and Kidney Diseases, U01DK062436, Dr. Peter F. Whitington National Institute of Diabetes and Digestive and Kidney Diseases, UL1TR000150, Dr. Peter F. Whitington National Institute of Diabetes and Digestive and Kidney Diseases, U01DK062500, Dr. Philip Rosenthal National Institute of Diabetes and Digestive and Kidney Diseases, UL1TR000004, Dr. Philip Rosenthal National Institute of Diabetes and Digestive and Kidney Diseases, U01DK062466, Dr. Robert H. Squires National Institute of Diabetes and Digestive and Kidney Diseases, UL1TR000005, Dr. Robert H. Squires National Institute of Diabetes and Digestive and Kidney Diseases, U01DK103140, Dr. Stephen L. Guthery National Institute of Diabetes and Digestive and Kidney Diseases, U01DK062503, Dr. Kathleen B. Schwarz National Institute of Diabetes and Digestive and Kidney Diseases, UL1TR000424, Dr. Kathleen B. Schwarz National Institute of Diabetes and Digestive and Kidney Diseases, U01DK062452, Dr. Yumirle P. Turmelle National Institute of Diabetes and Digestive and Kidney Diseases, UL1TR000448, Dr. Yumirle P. Turmelle National Institute of Diabetes and Digestive and Kidney Diseases, U01DK062453, Dr. Ronald J. Sokol National Institute of Diabetes and Digestive and Kidney Diseases, UL1TR001082, Dr. Ronald J. Sokol
References
- 1.Gottesman LE, Del Vecchio MT, Aronoff SC. Etiologies of conjugated hyperbilirubinemia in infancy: a systematic review of 1692 subjects. BMC Pediatr. 2015;15(1):192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gotze T, Blessing H, Grillhosl C, Gerner P, Hoerning A. Neonatal Cholestasis—Differential Diagnoses, Current Diagnostic Procedures, and Treatment. Front Pediatr. 2015;3:43 doi: 10.3389/fped.2015.00043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jimenez-Rivera C, Jolin-Dahel KS, Fortinsky KJ, Gozdyra P, Benchimol EI. International incidence and outcomes of biliary atresia. J Pediatr Gastroenterol Nutr. 2013;56(4):344–54. doi: 10.1097/MPG.0b013e318282a913 [DOI] [PubMed] [Google Scholar]
- 4.Breiman L, Friedman JH, Olshen RA, Stone CI. Classification and regression trees. Belmont, CA: Wadsworth; 1984.
- 5.Ambler G, Brady AR, Royston P. Simplifying a prognostic model: a simulation study based on clinical data. Stat Med. 2002;21(24):3803–22. doi: 10.1002/sim.1422 [DOI] [PubMed] [Google Scholar]
- 6.Lee KI, Koval JJ. Determinants of the best significance level in forward stepwise logistic regression. Comm Stat Sim Comp. 1997;26:559–75. [Google Scholar]
- 7.Steyerberg EW, Eijkemans MJ, Harrell FE Jr., Habbema JD. Prognostic modelling with logistic regression analysis: a comparison of selection and estimation methods in small data sets. Stat Med. 2000;19(8):1059–79. [DOI] [PubMed] [Google Scholar]
- 8.Raghunathan T, Lepkowski J, van Hoewyk J, Solenberger P. A multivariate technique for multiply imputing missing values using a sequence of regression models. Survery methodology. 2001;27(1):85–96. [Google Scholar]
- 9.Steyerberg EW. Clinical Prediction Models: A Practical Approach to Development, Validation, and Updating. New York: Springer; 2009. [Google Scholar]
- 10.Eilers PHC, Marx BD. Flexible smoothing with B-splines and penalties (with discussion). Statistical Science. 1996;11:89–121. [Google Scholar]
- 11.Thaler MM, Gellis SS. Studies in neonatal hepatitis and biliary atresia. IV. Diagnosis. Am J Dis Child. 1968;116(3):280–4. [DOI] [PubMed] [Google Scholar]
- 12.Mowat AP, Psacharopoulos HT, Williams R. Extrahepatic biliary atresia versus neonatal hepatitis. Review of 137 prospectively investigated infants. Arch Dis Child. 1976;51(10):763–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Manolaki AG, Larcher VF, Mowat AP, Barrett JJ, Portmann B, Howard ER. The prelaparotomy diagnosis of extrahepatic biliary atresia. Arch Dis Child. 1983;58(8):591–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ferry GD, Selby ML, Udall J, Finegold M, Nichols B. Guide to early diagnosis of biliary obstruction in infancy. Review of 143 cases. Clin Pediatr (Phila). 1985;24(6):305–11. [DOI] [PubMed] [Google Scholar]
- 15.Maggiore G, Bernard O, Hadchouel M, Lemonnier A, Alagille D. Diagnostic value of serum gamma-glutamyl transpeptidase activity in liver diseases in children. J Pediatr Gastroenterol Nutr. 1991;12(1):21–6. [DOI] [PubMed] [Google Scholar]
- 16.Tang KS, Huang LT, Huang YH, Lai CY, Wu CH, Wang SM, et al. Gamma-glutamyl transferase in the diagnosis of biliary atresia. Acta Paediatr Taiwan. 2007;48(4):196–200. [PubMed] [Google Scholar]
- 17.Poddar U, Thapa BR, Das A, Bhattacharya A, Rao KL, Singh K. Neonatal cholestasis: differentiation of biliary atresia from neonatal hepatitis in a developing country. Acta Paediatr. 2009;98(8):1260–4. doi: 10.1111/j.1651-2227.2009.01338.x [DOI] [PubMed] [Google Scholar]
- 18.El-Guindi MA, Sira MM, Sira AM, Salem TA, El-Abd OL, Konsowa HA, et al. Design and validation of a diagnostic score for biliary atresia. J Hepatol. 2014;61(1):116–23. doi: 10.1016/j.jhep.2014.03.016 [DOI] [PubMed] [Google Scholar]
- 19.Jancelewicz T, Barmherzig R, Chung CT, Ling SC, Kamath BM, Ng VL, et al. A screening algorithm for the efficient exclusion of biliary atresia in infants with cholestatic jaundice. J Pediatr Surg. 2015;50(3):363–70. doi: 10.1016/j.jpedsurg.2014.08.014 [DOI] [PubMed] [Google Scholar]
- 20.Chen X, Dong R, Shen Z, Yan W, Zheng S. Value of Gamma-Glutamyl Transpeptidase for Diagnosis of Biliary Atresia by Correlation with Age. J Pediatr Gastroenterol Nutr. 2016. [DOI] [PubMed] [Google Scholar]
- 21.Bakshi B, Sutcliffe A, Akindolie M, Vadamalayan B, John S, Arkley C, et al. How reliably can paediatric professionals identify pale stool from cholestatic newborns? Arch Dis Child Fetal Neonatal Ed. 2012;97(5):F385–7. doi: 10.1136/fetalneonatal-2010-209700 [DOI] [PubMed] [Google Scholar]
- 22.Franciscovich A, Vaidya D, Doyle J, Bolinger J, Capdevila M, Rice M, et al. PoopMD, a Mobile Health Application, Accurately Identifies Infant Acholic Stools. PLoS One. 2015;10(7):e0132270 doi: 10.1371/journal.pone.0132270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.He JP, Hao Y, Wang XL, Yang XJ, Shao JF, Feng JX. Comparison of different noninvasive diagnostic methods for biliary atresia: a meta-analysis. World J Pediatr. 2016;12(1):35–43. doi: 10.1007/s12519-015-0071-x [DOI] [PubMed] [Google Scholar]
- 24.Choi SO, Park WH, Lee HJ, Woo SK. 'Triangular cord': a sonographic finding applicable in the diagnosis of biliary atresia. J Pediatr Surg. 1996;31(3):363–6. [DOI] [PubMed] [Google Scholar]
- 25.Farrant P, Meire HB, Mieli-Vergani G. Improved diagnosis of extraheptic biliary atresia by high frequency ultrasound of the gall bladder. Br J Radiol. 2001;74(886):952–4. doi: 10.1259/bjr.74.886.740952 [DOI] [PubMed] [Google Scholar]
- 26.Zhou L, Shan Q, Tian W, Wang Z, Liang J, Xie X. Ultrasound for the Diagnosis of Biliary Atresia: A Meta-Analysis. AJR Am J Roentgenol. 2016:W1–W10. [DOI] [PubMed] [Google Scholar]
- 27.Gilmour SM, Hershkop M, Reifen R, Gilday D, Roberts EA. Outcome of hepatobiliary scanning in neonatal hepatitis syndrome. J Nucl Med. 1997;38(8):1279–82. [PubMed] [Google Scholar]
- 28.Russo P, Magee JC, Boitnott J, Bove KE, Raghunathan T, Finegold M, et al. Design and validation of the biliary atresia research consortium histologic assessment system for cholestasis in infancy. Clin Gastroenterol Hepatol. 2011;9(4):357–62 e2. doi: 10.1016/j.cgh.2011.01.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lee JY, Sullivan K, El Demellawy D, Nasr A. The value of preoperative liver biopsy in the diagnosis of extrahepatic biliary atresia: A systematic review and meta-analysis. J Pediatr Surg. 2016. [DOI] [PubMed] [Google Scholar]
- 30.Backeljauw B, Holland SK, Altaye M, Loepke AW. Cognition and Brain Structure Following Early Childhood Surgery With Anesthesia. Pediatrics. 2015;136(1):e1–e12. doi: 10.1542/peds.2014-3526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Greenholz SK, Krishnadasan B, Marr C, Cannon R. Biliary obstruction in infants with cystic fibrosis requiring Kasai portoenterostomy. J Pediatr Surg. 1997;32(2):175–9; discussion 9–80. [DOI] [PubMed] [Google Scholar]
- 32.Tolaymat N, Figueroa-Colon R, Mitros FA. Alpha 1-antitrypsin deficiency (Pi SZ) and biliary atresia. J Pediatr Gastroenterol Nutr. 1989;9(2):256–60. [DOI] [PubMed] [Google Scholar]
- 33.Nord KS, Saad S, Joshi VV, McLoughlin LC. Concurrence of alpha 1-antitrypsin deficiency and biliary atresia. J Pediatr. 1987;111(3):416–8. [DOI] [PubMed] [Google Scholar]
- 34.Lee HP, Kang B, Choi SY, Lee S, Lee SK, Choe YH. Outcome of Alagille Syndrome Patients Who Had Previously Received Kasai Operation during Infancy: A Single Center Study. Pediatr Gastroenterol Hepatol Nutr. 2015;18(3):175–9. doi: 10.5223/pghn.2015.18.3.175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Markowitz J, Daum F, Kahn EI, Schneider KM, So HB, Altman RP, et al. Arteriohepatic dysplasia. I. Pitfalls in diagnosis and management. Hepatology. 1983;3(1):74–6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX)
(DOCX)
(DOCX)
Data Availability Statement
This study is funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). The mechanism for public access to data is via the NIDDK Data Repository (https://www.niddkrepository.org/home/). Use of data via this mechanism is subject to standard NIDDK policies and procedures regarding public access databases and supporting documentation. The NIDDK Data Repository is overseen and managed by Information Management Services (IMS), Inc. IMS anticipates the data supporting the submitted manuscript to be publicly available on or before March 10, 2017. Secure Data Exchange for the NIDDK Data Repository is at https://dataexchange.imsweb.com/general/niddkrepository.