Abstract
Huntington’s disease is caused by the expansion of a polyglutamine repeat (>37 glutamines) in the disease protein huntingtin, which results in preferential neuronal loss in distinct brain regions. Mutant huntingtin causes late-onset neurological symptoms in patients in middle life, though the expression of mutant huntingtin is ubiquitous from early life. Thus, it is important to understand why mutant huntingtin selectively causes neuronal loss in an age-dependent manner. Transgenic animal models have been essential tools for uncovering the pathogenesis and therapeutic targets of neurodegenerative diseases. Genetic mouse models have been investigated extensively and have revealed the common pathological hallmark of age-dependent formation of aggregates or inclusions consisting of misfolded proteins. However, most genetic mouse models lack striking neurodegeneration that has been found in patient brains. Since there are considerable species differences between small and large animals, large animal models of Huntington’s disease may allow one to identify the pathological features that are more similar to those in patients and also help uncover more effective therapeutic targets. This chapter will focus on the important findings from large animal models of Huntington’s disease and discusses the use of large animal models to investigate the pathogenesis of Huntington’s disease and develop new therapeutic strategies.
Keywords: Aging, Huntington’s disease, Neurodegeneration, Species differences, Polyglutamine, Transgenic animals
1 Introduction
Alzheimer’s disease (AD), Parkinson’s disease (PD), Huntington’s disease (HD), and several other neurodegenerative diseases share a common pathological feature, which is selective neurodegeneration that occurs in an age-dependent manner. All these diseases are caused by misfolded proteins that form aggregates in the brains of affected patients. For example, in the brains of patients with AD, extracellular aggregates (senile plaques), which are formed by beta amyloid (Aβ) proteins, are the pathological hallmark of AD, whereas Lewy bodies, which contain alpha-synuclein, are a characteristic brain pathology of patients with PD. In HD, nuclear inclusions and neuropil aggregates are formed by N-terminal mutant huntingtin fragments that carry more than 37 polyglutamine repeats (Gusella et al. 1993; Vonsattel and DiFiglia 1998). Although whether these aggregates are toxic or protective remains to be investigated, it is clear that these aggregates are formed by small misfolded peptides and result from the accumulation of misfolded proteins. Consistently, the age-dependent formation of these aggregates in patient brains is correlated with the progression of neurological symptoms in AD, PD, and HD.
All these neurodegenerative diseases affect distinct types of neuronal cells despite the widespread expression of the disease proteins. For example, AD mainly affects the cortical neurons, whereas PD selectively affects dopaminergic neurons in the brain region known as the substantia nigra. HD, on the other hand, preferentially affects the medium spiny neurons in the striatum (Vonsattel et al. 1998). The selective neuropathology in each disease indicates that protein context is important for the specific neuronal toxicity in these diseases. Because all these disease proteins have different functions and interact with different partners, protein misfolding is likely to alter their functions and association with other proteins, resulting in a gain of toxicity in specific types of neurons.
HD shares common pathological changes with other neurodegenerative diseases but is caused by an autosomal dominant genetic mutation, making HD an ideal model to study how protein misfolding leads to selective neurodegeneration. The mechanistic insight obtained from studying HD would be very helpful for understanding other neurodegenerative diseases, such as AD and PD, which are also caused by protein misfolding. Thus, this chapter will focus on HD and its animal models.
2 HD and Polyglutamine Expansion
HD is characterized by motor dysfunction, cognitive decline, and psychological dysfunction. HD displays selective neurodegeneration that occurs preferentially in the brain striatum (Gusella et al. 1993; Vonsattel and DiFiglia 1998). The majority of patients with HD show symptoms in midlife and often die 10–15 years after the onset of symptoms. The genetic cause of HD is the expansion of a CAG repeat (>36 CAGs) in exon1 of the HD gene. Thus, the CAG repeat expansion results in an expanded polyglutamine (polyQ) tract in the N-terminal region of htt, a large-sized protein (3,144 amino acids) that is ubiquitously expressed in various types of cells and interacts with a number of proteins (Harjes and Wanker 2003; Li and Li 2004). As a result, mutant htt with an expanded polyQ tract forms insoluble aggregates or inclusions in the brains of patients with HD in an age-dependent manner.
The selective neurodegeneration in HD occurs early in the medium spiny neurons in the striatum. Other brain regions, such as the deep layers of the cortex, the hypothalamus, and the hippocampus, also undergo neurodegeneration in the later stages of HD (Vonsattel et al. 1998). While the primary function of htt has yet to be determined, it is known to be essential for early development and probably plays a role in cellular trafficking as a scaffold protein (Harjes and Wanker 2003; Li and Li 2004). It is evident that only N-terminal mutant htt is able to form aggregates and is more toxic than full-length mutant htt (Gutekunst et al. 1999; Zhou et al. 2003), which has led to extensive studies to identify the proteolytic cleavage sites mediated by various proteases, including calpains, aspartyl proteases, and caspases, which can degrade mutant htt to generate N-terminal htt fragments (Qin and Gu 2004). In addition to having the abilities to misfold and aggregate, N-terminal mutant htt fragments can also accumulate in the nucleus, whereas the majority of full-length mutant htt remains in the cytoplasm (DiFiglia et al. 1997; Gutekunst et al. 1999). The nuclear localization of N-terminal mutant htt can lead to abnormal binding of mutant htt to various transcription factors, subsequently affecting transcriptional expression (Harjes and Wanker 2003; Li and Li 2004).
3 HD Mouse Models
Identification of the genetic mutation for HD has led to the establishment of various transgenic HD mouse models. These models include transgenic mice (R6/2, N171-82Q) expressing N-terminal mutant htt (Davies et al. 1997; Schilling et al. 1999), full-length mutant htt transgenic mice (YAC and BAC) (Slow et al. 2003; Gray et al. 2008), and HD repeat knock-in (KI) mice (Wheeler et al. 2000; Lin et al. 2001; Menalled et al. 2002). R6/2 and N171-82Q mice display abundant htt aggregates in their brains at 3–4 months, as well as severe neurological symptoms and earlier death at 3–6 months (Davies et al. 1997; Schilling et al. 1999). Yeast artificial chromosome transgenic mice (YAC128), HD KI mice, and BACHD transgenic mice, which express full-length mutant human htt with an expanded polyQ repeat (114–150Q), display obvious htt aggregates only at older ages (7–10 months), show milder neurological symptoms than R/2 and N171-82Q mice, and can survive as wild-type mice (Lin et al. 2001; Slow et al. 2003; Gray et al. 2008; Wang et al. 2008). A careful analysis of HD150Q KI mice at 22 months, however, demonstrated that these mice develop the well-characterized htt aggregates, which could be seen in R6/2 mice at the age of 12 weeks (Woodman et al. 2007). Thus, characterization of various HD mouse models provides clear evidence that small N-terminal htt fragments with expanded polyQ tracts become misfolded and form aggregates. Consistently, polyQ-containing N-terminal htt fragments also form aggregates in HD cellular models (Li and Li 1998) and in HD patient brains (DiFiglia et al. 1997; Gutekunst et al. 1999). Despite the milder phenotypes of HD mice that express full-length mutant htt, such as YAC128 and HD knock-in mice, these mice show preferential accumulation of mutant htt in the striatum, consistent with the preferential loss of the medium spiny neurons in the striatum of HD patients. Thus, the accumulation of mutant htt in neuronal cells is clearly a prerequisite for neuronal dysfunction and degeneration.
Since mutant htt is ubiquitously expressed in various types of cells and localized in different subcellular regions including the nucleus and synapses, it would be important to investigate how mutant htt in different cell types and subcellular localization contribute to the disease progression. Transgenic mouse models allow for the selective expression mutant htt in neuronal and glial cells in the brain. Expression of N-terminal mutant htt in astrocytes is sufficient to cause age-dependent neurological symptoms (Bradford et al. 2009). Recently, transgenic mice that selectively express N-terminal mutant htt in presynaptic terminals were established and also show severe neurological phenotypes, providing convincing evidence for the critical role of synaptic mutant htt in the disease (Xu et al. 2013).
Although HD mouse models have been used widely to uncover the pathogenesis of HD and to develop treatments, most of these mouse models show no apoptosis or overt neurodegeneration in their brains. Similarly, in other polyQ mouse models, the lack of striking neurodegeneration is also a noteworthy phenomenon. Further, transgenic mice for AD and PD do not show typical neurodegeneration, either (Lee et al. 2012; LaFerla and Green 2012), although neurodegeneration is the major pathological event in AD and PD patients (Mattson 2000; Yuan and Yankner 2000). All these facts point out that, although mouse models are used widely to investigate the pathogenesis of HD and other neurological diseases, they have their limitations and do not replicate the full range of neurological phenotypes seen in human diseases. Such limitations reflect the importance of species differences in the development of neurodegeneration.
4 The Large Animal Models of HD
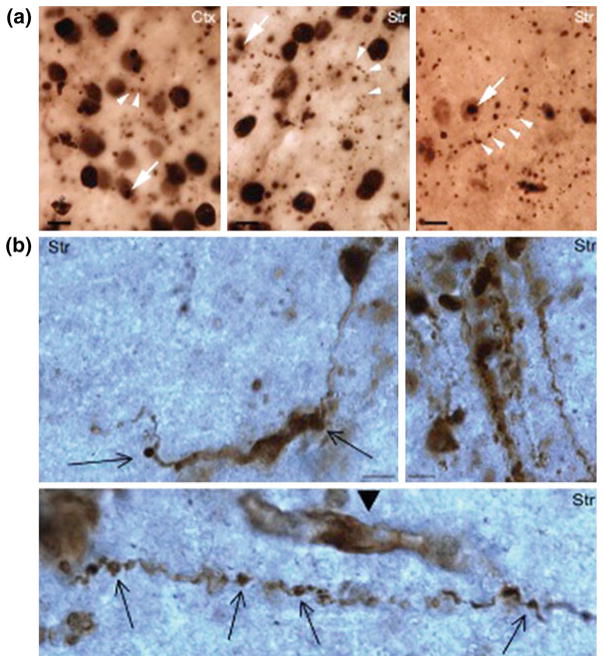
Although biological differences between humans and mice may account for the failure of some mouse models to replicate pathology in humans, whether larger transgenic animal models can mimic important neurodegenerative features caused by misfolded proteins remains to be rigorously tested. The creation of a transgenic monkey in 2001 (Chan et al. 2001) demonstrated that the monkey genome could be genetically modified and has led to the generation of transgenic nonhuman primate models expressing disease genes or exogenous foreign genes (Yang et al. 2008; Sasaki et al. 2009; Niu et al. 2010). Of which, transgenic HD rhesus monkeys express exon1 mutant htt with 84Q under the control of the human ubiquitin promoter (Yang et al. 2008). These HD monkeys were generated by injecting lentiviruses into fertilized oocytes to express mutant htt. Unlike transgenic mice, which can survive after birth when expressing the same exon1 mutant htt with an even longer polyQ repeat (150Q) (Davies et al. 1997; Cheng et al. 2013), HD transgenic monkeys with 84Q could die postnatally and this early death is associated with the levels of mutant htt (Yang et al. 2008). Despite their early death, some transgenic monkeys developed key clinical HD features including dystonia, chorea, and seizure (Yang et al. 2008), which have not been replicated by mouse models and other small animal models. Like the brains of HD mouse models and patients, the HD monkey brains also show abundant htt aggregates in the neuronal nuclei and neuronal processes (Fig. 1a). More importantly, the transgenic HD monkeys display the degeneration of axons and neuronal processes in the absence of obvious cell body degeneration, suggesting that neuronal degeneration in HD may initiate from neuronal processes (Fig. 1b). Such findings provide us with valuable information to understand the pathogenesis of HD.
Fig. 1.
Axonal degeneration in transgenic HD monkey brains. (a) Htt (EM48) immunostaining showing the presence of htt aggregates (arrows) in the neuronal nuclei in the cortex and striatum and in the neuronal processes (multiple arrowheads). (b) Htt immunostaining also revealed axonal degeneration (arrows) in HD monkey brain. Arrowhead indicates a glial cell
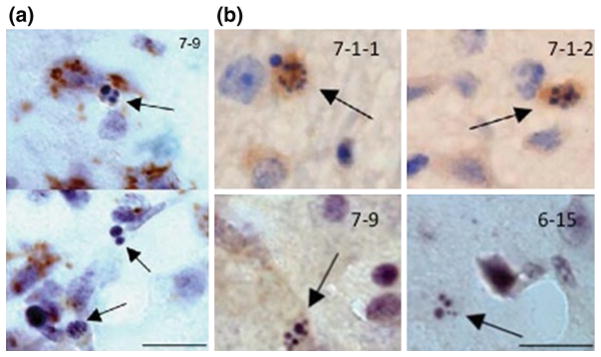
Our collaboration with Dr. Liangxue Lai at the Guangzhou Institutes of Biomedicine and Health (GIBH), Chinese Academy of Sciences, also led to the generation of transgenic HD pigs that express N-terminal mutant htt consisting of the first 208 amino acids with 105Q (N208-105Q) (Yang et al. 2010). The transgenes were expressed under the control of the cytomegalovirus enhancer and chicken beta-actin (CAG) promoter to allow the ubiquitous expression of transgenes in all tissues. Primary porcine fetal fibroblast cells expressing this mutant htt fragment were used to generate transgenic HD pigs via nuclear transfer. Six early pregnancies were established, and four of them went to term, with five live births. Like transgenic monkey models of HD, most of these transgenic HD piglets die postnatally, and some transgenic HD pigs show a severe chorea phenotype before death. We also generated transgenic mice expressing the same mutant htt and found that transgenic HD mice could live up to 9 months. Thus, the postnatal death of transgenic HD piglets also suggests that mutant htt is more toxic to larger animals. More importantly, in all transgenic pig brains examined, there were apoptotic cells (Fig. 2, Yang et al. 2010), which have not been reported in any HD mouse models.
Fig. 2.
Apoptotic cells in the brains of transgenic HD pigs. (a) Anti-polyQ (1C2) immunocytochemistry revealed the presence of mutant htt in the neurons of the brain striatal (upper) and cortical (lower) sections of HD transgenic pig (7-9). (b) Htt (EM48) immunocytochemistry also revealed apoptotic neurons (arrows) in transgenic HD pigs (7-1-1, 7-1-2, 7-9, and 6-15). Scale bars: 10 μm
However, transgenic HD sheep expressing full-length mutant htt with a 73Q tract live normally and show only a decrease in the expression of the medium spiny neuron marker DARPP-32 (Jacobsen et al. 2010). The differences between full-length htt and N-terminal htt transgenic pigs provide further evidence for the toxicity of N-terminal mutant htt. Thus, as with HD mouse models, the expression of N-terminal mutant htt can cause robust neurological phenotypes and pathological changes in large animals. These studies also suggest that protein context and the length of htt fragments may determine the nature of the neuropathology. For example, exon1 (1–67 amino acids) mutant htt in monkey brains causes axonal degeneration, whereas N-terminal 208 amino acids of mutant htt in pig brains can mediate apoptosis; however, in transgenic pigs (Uchida et al. 2001; Baxa et al. 2013) expressing a larger mutant htt fragment and in transgenic HD sheep (Jacobsen et al. 2010) expressing full-length (3,144 amino acids) mutant htt, there was no apoptosis, early animal death, or neurological phenotype reported. It is possible that neurodegeneration in large animals only occurs when sufficient degraded N-terminal fragments have accumulated in old animals. Thus, expressing N-terminal mutant htt fragments can facilitate disease progression, resulting in the early postnatal death of transgenic HD pigs and monkeys.
5 Insights from HD Models
It is clear that species differences play a critical role in the neurological phenotype differences in small and large animal models. There are considerable differences in development, life span, physiology, genetics, and anatomy between small and large animals (Table 1). An interesting issue is what the mechanisms behind these differences are. Certainly, there are a number of possible explanations. The short life span of mice is often believed to be responsible for the failure of HD mouse models to develop overt neurodegeneration. It is also possible that the misfolded form of N-terminal mutant htt is more toxic to the neuronal cells of pigs and monkeys than to rodent neurons. Considering that gestation in monkeys and pigs is much longer than in mice, this longer time period may allow overexpression of the toxic form of mutant proteins, such as N-terminal mutant htt, to cause more severe neurotoxicity in the pig and monkey brains. Also, because the brain circuitry in pigs and monkeys is more complex than in mice, this complexity may render neurons in large animals more vulnerable to misfolded mutant htt. Finally, the cellular ability to cope with misfolded proteins during development and adulthood may be different between species. The rapid maturation of rodent neurons during early brain development may reduce their sensitivity to misfolded proteins, which can also explain why mouse models can survive to adulthood even when they express the same mutant htt N-terminal fragment.
Table 1.
Major differences in some species
| Species | Sex maturity | Generation (day) | Life span (year) | Body weight (kg) |
|---|---|---|---|---|
| Human | 15–18 years | 266 | 75 | 50 |
| Monkey (Rhesus) | 3–5 years | 165 | 25 | 6 |
| Pig | 9–11 months | 114 | 7 | 80 |
| Mouse | 7 weeks | 19–21 | 2 | 0.03 |
Considering that HD and other neurodegenerative disease are age-dependent disorders, it is important to understand the differences in the aging process among different animal species when these animals are used to model neurodegenerative diseases. The life spans of these species differ drastically, indicating that aging processes in different species are not identical. Also, the early development of these mammalian species requires significantly different periods of time. For example, the gestation period for mice is 21 days, whereas pigs and monkeys require 4–5 months to reach full-term development. In addition to these significant differences, the anatomy, physiology, function, and circuitry of pig and monkey brains are much more complex than mouse brains (Table 1). These differences clearly indicate that monkeys and pigs are much closer to humans than mice and also explain why larger animal models would be better to mimic the pathological features seen in human patients. Indeed, a genetic pig model of cystic fibrosis replicates abnormalities seen in cystic fibrosis patients that do not occur in mouse models (Rogers et al. 2008). Of the large animals, monkeys are the best to model neurological diseases of humans, especially for cognitive behavioral analysis. Pigs, on the other hand, have a long life span (12–15 years), are easily bred, and reach puberty at 5–6 months, so they also offer advantages for biomedical research over other large animals, such as primates, for ethical and economic reasons (Lind et al. 2007).
6 Future Studies with Large Animal Models
Nevertheless, generation of genetic models using large animals is much more challenging than establishing genetic mouse models. So far, there are no ES cells from pigs, monkeys, or other large animals that can be used for generating gene-targeted animals. Induced pluripotent stem (iPS) cells are similar to embryonic cells and can be potentially used for altering endogenous genes and gene targeting in various species of animals. However, use of iPS cells for gene targeting is still under development. Most work done on transgenic monkeys involved the use of lentiviral vector infection of fertilized oocytes and embryo transplantation (Yang et al. 2008; Sasaki et al. 2009; Niu et al. 2010), which requires a considerable number of donor and surrogate monkeys. Successful generation of transgenic pigs can also be achieved via nuclear transfer, a cloning strategy that has a low rate (<1–2 %) for transferred pig embryos to develop to term (Lai and Prather 2003). In addition, the costs in maintaining and breeding large animals as well as the ethical concerns and strict regulation of the use of large animals also make it difficult to use them for biomedical research.
Recent advances in gene targeting have opened new avenues for gene targeting in large animals. The new technology, called TALEN and cas9, enable gene targeting via transcription activator-like effector nucleases (TALENs) and the Cas9 endonuclease from the type II bacterial CRISPR/Cas system, respectively. Gene targeting via TALENs or Cas9 can occur in embryos without involving ES cells (Shen et al. 2013; Wang et al. 2013). The new technology involves the use of DNA binding peptides that can bind specific target DNA sequences to allow cleavage DNAs by nuclease, thus creating the loss of function of a specific gene. This approach can be applied to one-cell fertilized embryos to cause null mutations of specific genes in the body. It has been reported that replacement of an endogenous gene via Cas9-mediated DNA cleavage can also be achieved (Wang et al. 2013).
Although the rodent models of neurodegenerative diseases will remain as a major modeling system for investigating a variety of diseases because of the difficulty and expense of generating and characterizing large animals, large animal models will make a more rigorous system for validating the relevance of critical findings from small animal models. The large animal models will help us to address the following important issues. It would be interesting to know how mutant htt possesses neuronal toxicity in adult transgenic animals. Such studies would require new transgenic animal models that can survive to adulthood, which can be accomplished by using different transgenic vectors that express mutant htt at a lower level or in an inducible manner. To verify that N-terminal mutant htt, rather than its overexpression, is indeed toxic, it would also be important to use a knock-in approach to express N-terminal mutant htt at the endogenous level. Transgenic models using higher mammalian species or large animals to model important neurodegenerative diseases would give us deeper insight into the pathogenesis of neurodegenerative diseases. In addition, given the frequent failures when it comes to clinical trials of drugs that have been found to work in small animal models, transgenic large animals could yield a more reliable system for verifying therapeutic efficacy before moving to clinical trials.
Acknowledgments
This work was supported by NIH grants NS036232, AG019206, NS041669 for X.J.L., and AG031153 for S.H.L.
Contributor Information
Xiao-Jiang Li, Department of Human Genetics, Emory University School of Medicine, 615 Michael St 347, Atlanta, GA 30322, USA. Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, No.1 West Beichen Road, Chaoyang District 100101 Beijing, China.
Shihua Li, Department of Human Genetics, Emory University School of Medicine, 615 Michael St 347, Atlanta, GA 30322, USA.
References
- Baxa M, Hruska-Plochan M, Juhas S, Vodicka P, Pavlok A, Juhasova J, Miyanohara A, Nejime T, Klima J, Macakova M, Marsala S, Weiss A, Kubickova S, Musilova P, Vrtel R, Sontag EM, Thompson LM, Schier J, Hansikova H, Howland DS, Cattaneo E, DiFiglia M, Marsala M, Motlik J. A transgenic minipig model of huntington’s disease. J Huntington Dis. 2013;2:47–68. doi: 10.3233/JHD-130001. [DOI] [PubMed] [Google Scholar]
- Bradford J, Shin JY, Roberts M, Wang CE, Li XJ, Li S. Expression of mutant huntingtin in mouse brain astrocytes causes age-dependent neurological symptoms. Proc Natl Acad Sci U S A. 2009;106:22480–22485. doi: 10.1073/pnas.0911503106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan AW, Chong KY, Martinovich C, Simerly C, Schatten G. Transgenic monkeys produced by retroviral gene transfer into mature oocytes. Science. 2001;291:309–312. doi: 10.1126/science.291.5502.309. [DOI] [PubMed] [Google Scholar]
- Cheng PH, Li CL, Chang YF, Tsai SJ, Lai YY, Chan AW, Chen CM, Yang SH. miR-196a Ameliorates phenotypes of Huntington disease in cell, transgenic mouse, and induced pluripotent stem cell models. Am J Hum Genet. 2013 doi: 10.1016/j.ajhg.2013.05.025. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies SW, Turmaine M, Cozens BA, DiFiglia M, Sharp AH, Ross CA, Scherzinger E, Wanker EE, Mangiarini L, Bates GP. Formation of neuronal intranuclear inclusions underlies the neurological dysfunction in mice transgenic for the HD mutation. Cell. 1997;90:537–548. doi: 10.1016/s0092-8674(00)80513-9. [DOI] [PubMed] [Google Scholar]
- Dawson TM, Ko HS, Dawson VL. Genetic animal models of Parkinson’s disease. Neuron. 2010;66:646–661. doi: 10.1016/j.neuron.2010.04.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiFiglia M, Sapp E, Chase KO, Davies SW, Bates GP, Vonsattel JP, Aronin N. Aggregation of huntingtin in neuronal intranuclear inclusions and dystrophic neurites in brain. Science. 1997;277:1990–1993. doi: 10.1126/science.277.5334.1990. [DOI] [PubMed] [Google Scholar]
- Gray M, Shirasaki DI, Cepeda C, Andre VM, Wilburn B, Lu XH, Tao J, Yamazaki I, Li SH, Sun YE, et al. Full-length human mutant huntingtin with a stable polyglutamine repeat can elicit progressive and selective neuropathogenesis in BACHD mice. J Neurosci. 2008;28:6182–6195. doi: 10.1523/JNEUROSCI.0857-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gusella JF, MacDonald ME, Ambrose CM, Duyao MP. Molecular genetics of Huntington’s disease. Arch Neurol. 1993;50:1157–1163. doi: 10.1001/archneur.1993.00540110037003. [DOI] [PubMed] [Google Scholar]
- Gutekunst CA, Li SH, Yi H, Mulroy JS, Kuemmerle S, Jones R, Rye D, Ferrante RJ, Hersch SM, Li XJ. Nuclear and neuropil aggregates in Huntington’s disease: relationship to neuropathology. J Neurosci. 1999;19:2522–2534. doi: 10.1523/JNEUROSCI.19-07-02522.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harjes P, Wanker EE. The hunt for huntingtin function: interaction partners tell many different stories. Trends Biochem Sci. 2003;28:425–433. doi: 10.1016/S0968-0004(03)00168-3. [DOI] [PubMed] [Google Scholar]
- Jacobsen JC, Bawden CS, Rudiger SR, McLaughlan CJ, Reid SJ, Waldvogel HJ, MacDonald ME, Gusella JF, Walker SK, Kelly JM, et al. An ovine transgenic Huntington’s disease model. Hum Mol Genet. 2010;19:1873–1882. doi: 10.1093/hmg/ddq063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LaFerla FM, Green KN. Animal models of Alzheimer disease. Cold Spring Harb Perspect Med. 2012:2. doi: 10.1101/cshperspect.a006320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai L, Prather RS. Production of cloned pigs by using somatic cells as donors. Cloning Stem Cells. 2003;5:233–241. doi: 10.1089/153623003772032754. [DOI] [PubMed] [Google Scholar]
- Lee Y, Dawson VL, Dawson TM. Animal models of Parkinson’s disease: vertebrate genetics. Cold Spring Harb Perspect Med. 2012:2. doi: 10.1101/cshperspect.a009324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li SH, Li XJ. Aggregation of N-terminal huntingtin is dependent on the length of its glutamine repeats. Hum Mol Genet. 1998;7:777–782. doi: 10.1093/hmg/7.5.777. [DOI] [PubMed] [Google Scholar]
- Li SH, Li XJ. Huntingtin-protein interactions and the pathogenesis of Huntington’s disease. Trends Genet. 2004;20:146–154. doi: 10.1016/j.tig.2004.01.008. [DOI] [PubMed] [Google Scholar]
- Lin CH, Tallaksen-Greene S, Chien WM, Cearley JA, Jackson WS, Crouse AB, Ren S, Li XJ, Albin RL, Detloff PJ. Neurological abnormalities in a knock-in mouse model of Huntington’s disease. Hum Mol Genet. 2001;10:137–144. doi: 10.1093/hmg/10.2.137. [DOI] [PubMed] [Google Scholar]
- Lind NM, Moustgaard A, Jelsing J, Vajta G, Cumming P, Hansen AK. The use of pigs in neuroscience: modeling brain disorders. Neurosci Biobehav Rev. 2007;31:728–751. doi: 10.1016/j.neubiorev.2007.02.003. [DOI] [PubMed] [Google Scholar]
- Matsuyama N, Hadano S, Onoe K, Osuga H, Showguchi-Miyata J, Gondo Y, Ikeda JE. Identification and characterization of the miniature pig Huntington’s disease gene homolog: evidence for conservation and polymorphism in the CAG triplet repeat. Genomics. 2000;69:72–85. doi: 10.1006/geno.2000.6317. [DOI] [PubMed] [Google Scholar]
- Mattson MP. Apoptosis in neurodegenerative disorders. Nat Rev Mol Cell Biol. 2000;1:120–129. doi: 10.1038/35040009. [DOI] [PubMed] [Google Scholar]
- Menalled LB, Sison JD, Wu Y, Olivieri M, Li XJ, Li H, Zeitlin S, Chesselet MF. Early motor dysfunction and striosomal distribution of huntingtin microaggregates in Huntington’s disease knock-in mice. J Neurosci. 2002;22:8266–8276. doi: 10.1523/JNEUROSCI.22-18-08266.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niu Y, Yu Y, Bernat A, Yang S, He X, Guo X, Chen D, Chen Y, Ji S, Si W, et al. Transgenic rhesus monkeys produced by gene transfer into early-cleavage-stage embryos using a simian immunodeficiency virus-based vector. Proc Natl Acad Sci U S A. 2010;107:17663–17667. doi: 10.1073/pnas.1006563107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orr HT, Zoghbi HY. Trinucleotide repeat disorders. Ann Rev Neurosci. 2007;30:575–621. doi: 10.1146/annurev.neuro.29.051605.113042. [DOI] [PubMed] [Google Scholar]
- Perutz MF, Johnson T, Suzuki M, Finch JT. Glutamine repeats as polar zippers: their possible role in inherited neurodegenerative diseases. Proc Natl Acad Sci U S A. 1994;91:5355–5358. doi: 10.1073/pnas.91.12.5355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qin ZH, Gu ZL. Huntingtin processing in pathogenesis of Huntington disease. Acta Pharmacol Sin. 2004;25:1243–1249. [PubMed] [Google Scholar]
- Rogers CS, Stoltz DA, Meyerholz DK, Ostedgaard LS, Rokhlina T, Taft PJ, Rogan MP, Pezzulo AA, Karp PH, Itani OA, et al. Disruption of the CFTR gene produces a model of cystic fibrosis in newborn pigs. Science. 2008;321:1837–1841. doi: 10.1126/science.1163600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasaki E, Suemizu H, Shimada A, Hanazawa K, Oiwa R, Kamioka M, Tomioka I, Sotomaru Y, Hirakawa R, Eto T, et al. Generation of transgenic non-human primates with germline transmission. Nature. 2009;459:523–527. doi: 10.1038/nature08090. [DOI] [PubMed] [Google Scholar]
- Schilling G, Becher MW, Sharp AH, Jinnah HA, Duan K, Kotzuk JA, Slunt HH, Ratovitski T, Cooper JK, Jenkins NA, et al. Intranuclear inclusions and neuritic aggregates in transgenic mice expressing a mutant N-terminal fragment of huntingtin. Hum Mol Genet. 1999;8:397–407. doi: 10.1093/hmg/8.3.397. [DOI] [PubMed] [Google Scholar]
- Shen B, Zhang J, Wu H, Wang J, Ma K, Li Z, Zhang X, Zhang P, Huang X. Generation of gene-modified mice via Cas9/RNA-mediated gene targeting. Cell Res. 2013;23:720–723. doi: 10.1038/cr.2013.46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slow EJ, van Raamsdonk J, Rogers D, Coleman SH, Graham RK, Deng Y, Oh R, Bissada N, Hossain SM, Yang YZ, et al. Selective striatal neuronal loss in a YAC128 mouse model of Huntington disease. Hum Mol Genet. 2003;12:1555–1567. doi: 10.1093/hmg/ddg169. [DOI] [PubMed] [Google Scholar]
- Uchida M, Shimatsu Y, Onoe K, Matsuyama N, Niki R, Ikeda JE, Imai H. Production of transgenic miniature pigs by pronuclear microinjection. Transgenic Res. 2001;10:577–582. doi: 10.1023/a:1013059917280. [DOI] [PubMed] [Google Scholar]
- Vonsattel JP, DiFiglia M. Huntington disease. J Neuropathol Exp Neurol. 1998;57:369–384. doi: 10.1097/00005072-199805000-00001. [DOI] [PubMed] [Google Scholar]
- Wang CE, Tydlacka S, Orr AL, Yang SH, Graham RK, Hayden MR, Li S, Chan AW, Li XJ. Accumulation of N-terminal mutant huntingtin in mouse and monkey models implicated as a pathogenic mechanism in Huntington’s disease. Hum Mol Genet. 2008;17:2738–2751. doi: 10.1093/hmg/ddn175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H, Yang H, Shivalila CS, Dawlaty MM, Cheng AW, Zhang F, Jaenisch R. One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell. 2013;153:910–918. doi: 10.1016/j.cell.2013.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wheeler VC, White JK, Gutekunst CA, Vrbanac V, Weaver M, Li XJ, Li SH, Yi H, Vonsattel JP, Gusella JF, et al. Long glutamine tracts cause nuclear localization of a novel form of huntingtin in medium spiny striatal neurons in HdhQ92 and HdhQ111 knock-in mice. Hum Mol Genet. 2000;9:503–513. doi: 10.1093/hmg/9.4.503. [DOI] [PubMed] [Google Scholar]
- Woodman B, Butler R, Landles C, Lupton MK, Tse J, Hockly E, Moffitt H, Sathasivam K, Bates GP. The Hdh(Q150/Q150) knock-in mouse model of HD and the R6/2 exon 1 model develop comparable and widespread molecular phenotypes. Brain Res Bull. 2007;72:83–97. doi: 10.1016/j.brainresbull.2006.11.004. [DOI] [PubMed] [Google Scholar]
- Xu QQ, Huang SS, Song M, Wang CE, Yan S, Liu XD, Gaetig MA, Yu SP, Li H, Li S, Li X-J. Synaptic mutant Huntingtin inhibits synapsin-1 phosphorylation and causes neurological symptoms. J Cell Biol. 2013 doi: 10.1083/jcb.201303146. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang D, Wang CE, Zhao B, Li W, Ouyang Z, Liu Z, Yang H, Fan P, O’Neill A, Gu W, et al. Expression of Huntington’s disease protein results in apoptotic neurons in the brains of cloned transgenic pigs. Hum Mol Genet. 2010;19:3983–3994. doi: 10.1093/hmg/ddq313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang SH, Cheng PH, Banta H, Piotrowska-Nitsche K, Yang JJ, Cheng EC, Snyder B, Larkin K, Liu J, Orkin J, et al. Towards a transgenic model of Huntington’s disease in a nonhuman primate. Nature. 2008;453:921–924. doi: 10.1038/nature06975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan J, Yankner BA. Apoptosis in the nervous system. Nature. 2000;407:802–809. doi: 10.1038/35037739. [DOI] [PubMed] [Google Scholar]
- Zhou H, Cao F, Wang Z, Yu ZX, Nguyen HP, Evans J, Li SH, Li XJ. Huntingtin forms toxic NH2-terminal fragment complexes that are promoted by the age-dependent decrease in proteasome activity. J Cell Biol. 2003;163:109–118. doi: 10.1083/jcb.200306038. [DOI] [PMC free article] [PubMed] [Google Scholar]