Abstract
Postoperative pancreatic fistula (POPF) is a common complication following distal pancreatectomy (DP). However, the risk factors of this complication in patients after DP still remain controversial. The aim of our study is to estimate the association between potential risk factors and POPF. Relevant articles published up to June 21, 2016 were identified via PubMed, EMBASE, Web of Science, and The Cochrane Library. Studies that examined the risk factors of POPF following DP were enrolled. 20 articles (2070 patients) were finally included in this study. The pooled data suggested that patients with soft pancreas, higher Body Mass Index (BMI), blood transfusion, elevated intraoperative blood loss, and longer operative time had a decreased risk for POPF. However, age, gender, malignant pathology, types of stump closure, octreotide therapy, history of diabetes and chronic pancreatitis, splenectomy, multiorgan resection, main duct ligation, preoperative serum albumin levels, PGA felt wrapping, and extended lymphadenectomy could not be regarded as risk factors for POPF. Our analytic data demonstrated that pancreas texture, BMI, blood transfusion, intraoperative blood loss, and operative time were clinical predictor for POPF. This study may assist surgeons to screen patients with high risk of POPF and select appropriate treatment measures.
Introduction
Distal pancreatectomy is commonly applied for the resection of pancreatic diseases located in the body and/or tail of the pancreas, such as pancreatic ductal adenocarcinoma, cystic neoplasm, neuroendocrine neoplasm, and chronic pancreatitis1–3. Although surgical techniques and perioperative managements for DP were significantly improved in past decades, the complications following DP were still great challenges for surgeons specialized in pancreatic surgery, especially postoperative pancreatic fistula4.
According to the definition provided by the International Study Group of Pancreatic Fistula (ISGPF)5, POPF manifests as a drain (obtained from operatively or postoperatively placed drain) output with amylase content greater than three times the upper limit of the normal level of serum amylase on or after the third postoperative day. Furthermore, ISGPF also divided POPF into three grades, grade A, B, and C6. Grade A pancreatic fistula is an asymptomatic fistula; while grade B and C pancreatic fistula are symptomatic fistula which needs therapeutic intervention (such as antibiotics and/or percutaneous drainage for grade B; resuscitation and/or exploratory laparotomy for grade C). Once postoperative patients suffer from grade B or C pancreatic fistula, a series of other severe complications might develop, including intra-abdominal infection, bleeding, and even shock. Therefore, it is urgent to minutely understand the knowledge of POPF, especially the risk factors which were responsible for the occurrence such complication.
As reported in some studies, a large number of factors could heighten the risk of POPF, such as older age, increased BMI, diabetes history, soft pancreas preoperative blood transfusion, elevated intraoperative blood loss, and longer operative time7–32. However, the results of these studies were controversial, and the sample size of them was relatively limited. Therefore, we performed this systematic review to further assess the predictive roles of these risk factors for POPF in patients with DP.
Results
Study selection
The identification and selection processes for this meta-analysis are illustrated in Fig. 1. A total of 136 potential studies were yielded after our initial search. And then, 110 studies were excluded by scanning titles and abstracts, 26 articles were left for the further full text assessment. According to the inclusion criteria mentioned in materials and methods, 20 articles were finally included to use for this meta analysis. Other articles were excluded due to the reasons as follows: (a) non-focusing on the association between risk factors and POPF after DP; (b) lacking full text; (c) non-providing exact numbers of each group.
Figure 1.
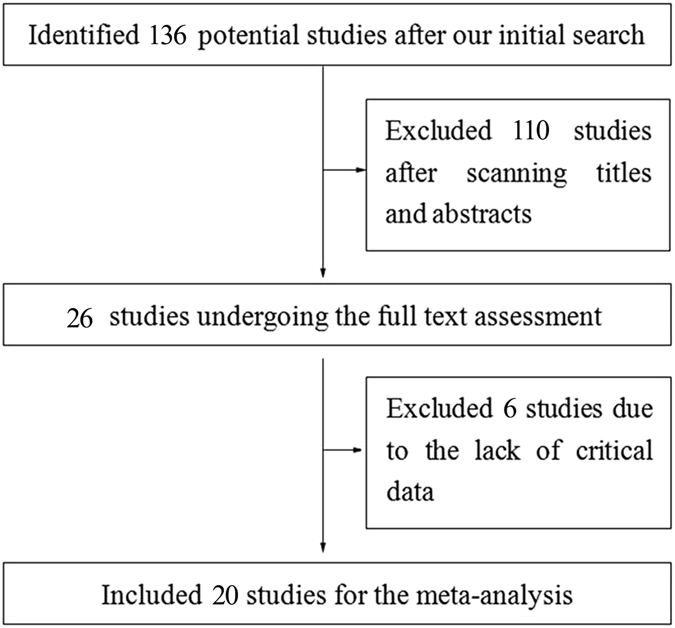
Flow diagram illustrating selection of the articles included in the meta-analysis.
Characteristics of enrolled articles
The main information of these included articles was shown in Table 1. A total of 20 studies with 2070 patients (range from 33–352 per study) were evaluated in this meta analysis7–25, 32. All studies were published from 2006 to 2015. Among them, 12 studies investigated the patients from Asian countries, including Singapore (2), Korea (1), India (1), and Japan (8); 8 studies investigated the patients from European countries, North American countries, and North African countries, including Italy (1), France (1), Germany (2), Poland (1), Sweden (1), USA (1), and Tunis (1). 16 of these articles were retrospective studies, 3 of them were prospective studies, and 1 was randomised, controlled trial (RCT). Furthermore, the results of study quality assessment were also listed in Table 1.
Table 1.
Characteristics of selected studies.
| Author | Country | Year | Study design | Definition of POPF | Sample size | Quality assessment |
|---|---|---|---|---|---|---|
| Marco Pericoli Ridolfini et al. | Italy | 2007 | Retrospective | ISGPF | 64 | 7 |
| Brian K. P. Goh et al. | Singapore | 2008 | Retrospective | ISGPF | 230 | 9 |
| Virginie Pannegeon et al. | France | 2006 | Retrospective | NA | 175 | 7 |
| Ryuji Yoshioka et al. | Japan | 2010 | Retrospective | ISGPF | 100 | 7 |
| Marius Distler et al. | Germany | 2014 | Retrospective | ISGPF | 124 | 8 |
| Rachel M Gomes et al. | India | 2012 | Retrospective | ISGPF | 33 | 6 |
| Koji Soga et al. | Japan | 2011 | Retrospective | ISGPF | 63 | 7 |
| Y. Kawabata et al. | Japan | 2013 | Retrospective | ISGPF | 40 | 7 |
| Hidetoshi Eguchi et al. | Japan | 2011 | Retrospective | ISGPF | 48 | 7 |
| Motokazu Sugimoto et al. | Japan | 2013 | Retrospective | ISGPF | 106 | 8 |
| Chiow Adrian Kah Heng et al. | Singapore | 2009 | Retrospective | ISGPF | 75 | 7 |
| Amin Makni et al. | Tunis | 2012 | Retrospective | ISGPF | 35 | 6 |
| Marek Sierzega et al. | Poland | 2007 | Prospective | ISGPF | 132 | 7 |
| Manabu Kawai et al. | Japan | 2008 | Prospective | NA | 75 | 6 |
| Norihiro Sato et al. | Japan | 2014 | Retrospective | ISGPF | 44 | 6 |
| Preeti D. Subhedar et al. | USA | 2011 | Prospective | ISGPF | 149 | 8 |
| Arturo S. Mendoza III et al. | Korea | 2015 | Retrospective | ISGPF | 143 | 8 |
| Keiichi Okano et al. | Japan | 2011 | Retrospective | ISGPF | 31 | 6 |
| Markus K Diener et al. | Germany | 2011 | RCT | ISGPF | 352 | 9 |
| Farshad Frozanpor et al. | Sweden | 2010 | Retrospective | ISGPF | 51 | 7 |
The association between risk factors and POPF after DP
Non-operation related risk factors
8 non-operation related risk factors were analyzed in this study, including age, gender, BMI, malignant pathology, octreotide therapy, history of diabetes, history of chronic pancreatitis, and preoperative serum albumin levels. All pooled data about these factors were shown in Table 2.
Table 2.
Pooled data about non-operation related risk factors.
| Number of articles | OR | 95% CI | P | I2(%) | P H | |
|---|---|---|---|---|---|---|
| Age | 10 | 1.35 | 0.72–2.52 | 0.350 | 53 | 0.030 |
| Gender | 16 | 1.08 | 0.87–1.35 | 0.480 | 0 | 0.900 |
| BMI | 6 | 2.19 | 1.35–3.56 | 0.001 | 6 | 0.380 |
| Malignant pathology | 15 | 0.89 | 0.70–1.12 | 0.310 | 28 | 0.150 |
| Octreotide therapy | 5 | 1.07 | 0.58–1.99 | 0.820 | 51 | 0.080 |
| History of diabetes | 9 | 0.82 | 0.59–1.13 | 0.210 | 33 | 0.150 |
| History of chronic pancreatitis | 4 | 1.08 | 0.45–2.26 | 0.860 | 64 | 0.004 |
| Preoperative serum albumin levels | 3 | 0.51 | 0.20–1.31 | 0.160 | 0 | 0.980 |
Age: 9 of 10 studies suggested that the risk of POPF was not significantly associated with age, while 1 of 10 studies found that increased risk of POPF was observed in older patients. Our pooled data shown that older patients were not prone to suffer from POFP (Table 2, Fig. 2A). However, subgroup analysis suggested that age is a positive factor for POFP according to pooled data from articles published after 2010 and articles with more than or equal to 100 patients (Supplementary Tables 1 and 2).
Figure 2.
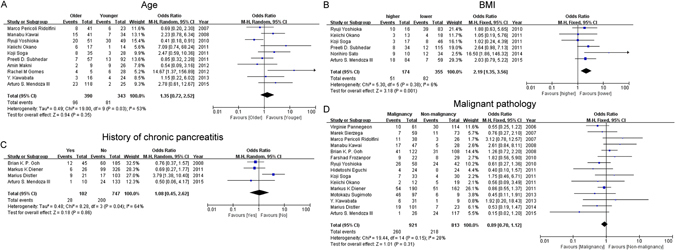
Forest plot of the association between POPF and non-operation related risk factors. (A) The association between POPF and age. (B) The association between POPF and BMI. (C) The association between POPF and history of chronic pancreatitis.
BMI: BMI was mentioned in 6 articles. Although only 1 of 6 articles reported that increased BMI could enhance the incidence of POPF, the combined result supported this finding (Table 2, Fig. 2B), as well as the subgroup results based on the articles with published after 2010, and enrolled more than or equal to 100 patients (Supplementary Tables 1 and 2).
History of chronic pancreatitis: 1 of 4 article suggested that patients with chronic pancreatitis had higher risk to suffer from POFP. However, the combined result (Table 2, Fig. 2C) and all subgroup results all showed that there was no significant difference between patients with or without chronic pancreatitis (Supplementary Tables 1 and 2).
Malignant pathology: all selected articles and the combined data were reported that patients with malignant tumors do not have an increased risk of POPF (Table 2, Fig. 2D). However, in subgroup analysis, the result showed that malignant tumors were associated with the decreased risk of POPF in studies with larger sample size (≥100) (Supplementary Tables 1 and 2).
Other factors: Gender, octreotide therapy, history of diabetes, and preoperative serum albumin levels were respectively reported in 16, 5, 9, 3 enrolled studies. The pooled results illustrated that these non-operation related risk factors mentioned above do not increase or decrease risk of POPF, were in agreement with all articles enrolled in this study (Table 2, Supplementary Fig. 1). Furthermore, the tendency of all subgroup analytic results was in accordance with that of the primary data and combined data (Supplementary Tables 1 and 2).
Operation related risk factors
10 operation related risk factors were assessed in our study, such as pancreas texture, types of stump closure, blood transfusion, intraoperative blood loss, operative time, splenectomy, multiorgan resection, main duct ligation, PGA felt wrapping, and extended lymphadenectomy. All combined data about factors were shown in Table 3.
Table 3.
Combined data about operation related risk factors.
| Number of articles | OR | 95% CI | P | I2(%) | P H | |
|---|---|---|---|---|---|---|
| Pancreas texture | 14 | 1.80 | 1.08–3.02 | 0.030 | 51 | 0.010 |
| Types of stump closure | 8 | 0.75 | 0.42–1.33 | 0.330 | 53 | 0.040 |
| Blood transfusion | 10 | 1.55 | 1.11–2.16 | 0.009 | 0 | 0.640 |
| Intraoperative blood loss | 8 | 2.25 | 1.54–3.29 | <0.0001 | 28 | 0.210 |
| Operative time | 7 | 1.67 | 1.08–2.58 | 0.020 | 31 | 0.190 |
| Splenectomy | 12 | 0.91 | 0.52–1.59 | 0.730 | 52 | 0.020 |
| Multiorgan resection | 8 | 0.77 | 0.54–1.10 | 0.150 | 28 | 0.210 |
| Main duct ligation | 7 | 0.50 | 0.24–1.08 | 0.080 | 61 | 0.020 |
| PGA felt wrapping | 3 | 0.75 | 0.18–3.14 | 0.690 | 61 | 0.080 |
| Extended lymphadenectomy | 4 | 0.82 | 0.56–1.20 | 0.300 | 0 | 0.820 |
Pancreas texture: A total of 14 articles assessed the association between pancreas texture and POPF. 3 of them considered that soft pancreas might increase the risk of POPF, and the pooled OR and 95% CI supported this view (Table 3, Fig. 3A). However, according to the subgroup analysis, we found that similar tendency was just observed in results obtained from studies with larger sample size, and published after 2010 (Supplementary Tables 3 and 4).
Figure 3.
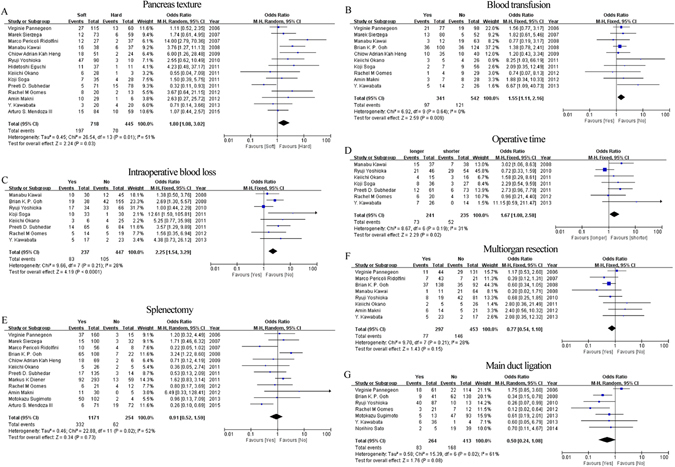
Forest plot of the association between POPF and operation related risk factors. (A) The association between POPF and pancreas texture. (B) The association between POPF and blood transfusion. (C) The association between POPF and intraoperative blood loss. (D) The association between POPF and operative time. (E) The association between POPF and splenectomy. (F) The association between POPF and main duct ligation.
Blood transfusion: Although only 2 of 10 articles reported that patients with blood transfusion had an increased opportunity to occur POPF, our meta analysis still confirmed this tendency (Table 3, Fig. 3B). Further assessment based on the studies published after 2010 also suggested that blood transfusion could enhance the occurrence of POPF, and other further assessment did not have positive results (Supplementary Tables 3 and 4).
Intraoperative blood loss: More intraoperative blood loss might increase the risk of POPF according to the data provided by 3 of 8 articles and the combined data (Table 3, Fig. 3C). All subgroup analysis also showed the coincident results (Supplementary Tabled 3 and 4).
Operative time: Our analytic data showed that longer operative time was another promotive risk factors for POPF (Table 3, Fig. 3D). And this result was in accordance with the results provided by Manabu Kawai et al., the subgroup results based on articles published after 2010, and articles possessed less than 100 patients (Supplementary Tables 3 and 4).
Splenectomy: Brian K. P. Goh et al. suggested that DP combined with splenectomy might increase the development of POPF, but Arturo S. Mendoza III revealed the opposite results. However, our assessed results did not agree with them, there was no difference between DP with and without splenectomy (Table 3, Fig. 3E). Moreover, the further analysis based on sample size and the year of articles published all showed that additional splenectomy do not affect the development of POPF (Supplementary Tables 3 and 4).
Multiorgan resection: Multiorgan resection was mentioned in 8 articles. Although all articles and pooled data reported that this factor do not influence the incidence of POPF (Table 3, Fig. 3F), the data based on the articles published in or before 2010 suggested that patients with multiorgan resection had a decreased risk of POPF (Supplementary Tables 3 and 4).
Main duct ligation: Although 3 of 7 studies agreed that main duct ligation could reduce the incidence rate of POPF, the combined results did not show this tendency (Table 3, Fig. 3G). Moreover, subgroup assessment based on articles published after 2010, and consisted of small sample size also showed this tendency (Supplementary Tables 3 and 4).
Other factors: Not only articles enrolled in this study but also our pooled results suggested that other operation related factors did not show any correlation with the development of POPF, including types of stump closure (8 studies), PGA felt wrapping (3 studies), and extended lymphadenectomy (4 studies) (Table 3, Supplementary Fig. 2). In further assessment, subgroup results based on articles published year and sample size suggested that patients with these factors do not had a decreased or increased risk of POPF (Supplementary Tables 2 and 3).
Discussion
Pancreatic fistula is still a ongoing clinical problem following pancreatic resection, with various technical innovation failing to reduce the occurrence of such complication33. Delays in the treatment of POPF will lead to serious problems, such as abscess, pseudoaneurysm, hemorrhage, and sepsis34–37. Therefore, in-depth studies focused on the epidemic factors of POPF are urgently needed to provide basis for the prevention and management of POPF. In this study, we reviewed several reported operation and non-operation related risk factors of POPF to further confirm their roles.
To the best of our knowledge, this is the first meta analysis to assess the association between several risk factors and POPF comprehensively. According to the combined ORs and 95% CIs, we confirmed that patients with soft pancreas, higher BMI, blood transfusion, a large number of intraoperative blood loss, and extended operative time were prone to suffer from POPF. Therefore, DP patients with above-named factors should get more attention during the perioperation. Moreover, in subgroup analysis, the predictive role of intraoperative blood loss was consistent in all subgroups, suggesting that effective bleeding control was particularly necessary in DP. However, the positive roles of other risk factors were only observed in some special subgroups. Various reasons might be responsible for this phenomenon. The number of studies in some subgroups were relatively small (such as age, BMI and operative time in articles published before and equal to 2010 group; blood transfusion and operative time in sample size more than 100 group). And well conducted studies with large sample size were lacking (such as BMI in sample size less than and equal to 100 group). Furthermore, we also found that many other risk factors were not associated with the incidence of POPF. The role of some factors might be inconclusive due to the lacking of enrolled studies, such as history of chronic pancreatitis, extended lymphadenectomy, PGA felt wrapping, and serum albumin levels. From the above mentioned, therefore, more studies about some potential articles were greatly needed, especially well designed studies with a large number of patients.
There was significant heterogeneity among the articles included in meta-analysis about age, octreotide therapy, pancreas texture, history of chronic pancreatitis, types of stump closure, splenectomy, main duct ligation, and PGA felt wrapping. To evaluate the reasons of heterogeneity, we performed the subgroup analysis on nationality of patients, year of articles published, and sample size. For age and pancreas texture, the nationality of patients was identified as main factor resulting in heterogeneity; for octreotide therapy, the main factor was sample size; and for types of stump closure and main duct ligation, published year was considered as the main factor. Moreover, the reason of the heterogeneity about history of chronic pancreatitis and PGA felt wrapping was not determined because of the lack of enough articles. To avoid the heterogeneity, further articles were needed which was validated against the main factors. Of course, many other factors also could result in the heterogeneity, such as surgical technology and operation method.
There were also some other limitations in this meta analysis. Firstly, some articles reported the association between risk factors and POPF with ORs and 95% CI were not selected in this study. Secondly, the cut off value of several factors in different articles was not coincidental, such age, BMI, blood loss, and operative time. Thirdly, many other factors might be responsible for the development of POPF. However, they were not assessed in our study due to the lack of sufficient articles or effective data. Finally, the design of eligible articles was highly limited. Almost all of them (16 of 20) were retrospective studies, and more prospective studies and randomized controlled trials were need to improve the reliability of reported data.
In conclusion, this meta-analysis indicates that soft pancreas texture, higher BMI, blood transfusion, massive intraoperative blood loss, and prolonged operative time are markedly associated with the increased incidence of POPF. These findings will provide important theoretical basis for surgeons to overcome the POPF. However, due to the limitations mentioned above, additional well-designed studies with larger sample size are required to confirm the predictive roles of those factors.
Materials and Methods
Literature Search
Potential studies were selected by screening PubMed, EMBASE, Web of Science, and The Cochrane Library. The search criteria were ((risk [Title/Abstract]) and distal pancreatectomy [Title/Abstract]) and fistula [Title/Abstract], and ((risk [Title/Abstract]) and left pancreatectomy [Title/Abstract]) and fistula [Title/Abstract]. The last date of retrieval was updated to June 21, 2016. Moreover, the references cited in these articles were also been examined to identify additional relevant studies.
Inclusion and Exclusion Criteria
Studies that
included patients underwent distal pancreatectomy or left pancreatectomy;
investigated postoperative pancreatic fistula;
assessed the association between risk factors and pancreatic fistula in postoperative patients; and
were published as a full paper in English were finally enrolled in this meta-analysis.
The following criteria were applied to exclude studies:
non-human research;
review articles or letters;
articles with repeated data; and
lacking critical data required for further analysis.
Study Quality Assessment
The quality of the eligible studies was assessed by two independent reviewers according to the Newcastle–Ottawa Quality Assessment Scale for cohort studies (NOS) recommended in the Cochrane Handbook version 5.1.038. The eight elements in the NOS are assessed under three aspects: selection (four elements, one star each), comparability (one element, up to two stars) and outcome (three elements, one star each). The high-quality choices for each element are marked with a star, and then the number of stars is counted to evaluate the quality of each study. Studies are considered high quality if they are awarded six stars or more39.
Data Extraction
All data applied in this study were carefully extracted and estimated by two independent researchers, and any controversial data were re-estimated by both two researchers at the same time. Data extracted from each article were shown as follows:
article data: author, year of publication, country, study types, numbers of total DP patients, and sample size;
the definition of POPF and the number of postoperative patients suffered from POPF;
potential risk factors mentioned in more than or equal to three articles, and the number of patients in each group. Age, gender, BMI, malignant pathology, pancreas texture, types of stump closure, octreotide therapy, history of diabetes, history of chronic pancreatitis, blood transfusion, intraoperative blood loss, operative time, splenectomy, multiorgan resection, main duct ligation, preoperative serum albumin levels, PGA felt wrapping, and extended lymphadenectomy were finally selected, and we will analyze the association between the development of POPF and risk factors mentioned above.
Statistical Analysis
Review Manager software (version 5.3; Cochrane Collaboration, Oxford, United Kingdom) was applied to perform this meta analysis and provide relative graphics. The numbers of different groups obtained from each article were used to estimate the ORs and 95% CIs, and further to calculate the combined ORs and 95% CIs. Subgroup analysis was performed based on the year of articles published (>2010 vs. ≤2010), and the sample size (≥100 vs. <100). Cochran’s Q test and the Higgins I-squared statistic were applied to conduct the heterogeneity analysis40. Heterogeneity would not be considered significant if the P-value for Cochran’s Q test was greater than or equal to 0.1. Random effects model was selected if the P-value for Cochran’s Q test was less than 0.1, otherwise fixed effects model was selected41. Two-tailed P-value was considered statistically significant at less than 0.05.
Electronic supplementary material
Acknowledgements
We thank Prof. Markus K Diener for the providing of the primary data of his article. This work was supported by grants from the National Natural Science Foundation of China (81170336, 81272239).
Author Contributions
Dr. Yi Miao designed the study. Dr. Yun-Peng Peng performed the study and wrote the paper. Dr. Xiao-Le Zhu, Ling-Di Yin, and Yi Zhu assessed the study enrolled in this study and collected the data. Dr. Ji-Shu Wei and Jun-Li Wu analyzed the data.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Yun-Peng Peng and Xiao-Le Zhu contributed equally to this work.
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-00311-8
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Parikh PY, Lillemoe KD. Surgical management of pancreatic cancer–distal pancreatectomy. Semin Oncol. 2015;42:110–22. doi: 10.1053/j.seminoncol.2014.12.010. [DOI] [PubMed] [Google Scholar]
- 2.Probst, P. et al. Stapler versus scalpel resection followed by hand-sewn closure of the pancreatic remnant for distal pancreatectomy. Cochrane Database Syst Rev. CD008688 (2015). [DOI] [PMC free article] [PubMed]
- 3.Fernandez-Cruz L, et al. Laparoscopic Distal Pancreatectomy for Pancreatic Tumors: Does Size Matter? Dig Surg. 2016;33:290–8. doi: 10.1159/000445008. [DOI] [PubMed] [Google Scholar]
- 4.Miyasaka, Y., Mori, Y., Nakata, K., Ohtsuka, T. & Nakamura, M. Attempts to prevent postoperative pancreatic fistula after distal pancreatectomy. Surg Today (2016). [DOI] [PubMed]
- 5.Fisher WE, et al. Effect of BioGlue on the incidence of pancreatic fistula following pancreas resection. J Gastrointest Surg. 2008;12:882–90. doi: 10.1007/s11605-008-0479-x. [DOI] [PubMed] [Google Scholar]
- 6.Sudo T, et al. Distal pancreatectomy with duct-to-mucosa pancreaticogastrostomy: a novel technique for preventing postoperative pancreatic fistula. Am J Surg. 2011;202:77–81. doi: 10.1016/j.amjsurg.2010.04.023. [DOI] [PubMed] [Google Scholar]
- 7.Ridolfini MP, et al. Risk factors associated with pancreatic fistula after distal pancreatectomy, which technique of pancreatic stump closure is more beneficial? World J Gastroenterol. 2007;13:5096–100. doi: 10.3748/wjg.v13.i38.5096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Goh BK, et al. Critical appraisal of 232 consecutive distal pancreatectomies with emphasis on risk factors, outcome, and management of the postoperative pancreatic fistula: a 21-year experience at a single institution. Arch Surg. 2008;143:956–65. doi: 10.1001/archsurg.143.10.956. [DOI] [PubMed] [Google Scholar]
- 9.Pannegeon V, et al. Pancreatic fistula after distal pancreatectomy: predictive risk factors and value of conservative treatment. Arch Surg. 2006;141:1071–6. doi: 10.1001/archsurg.141.11.1071. [DOI] [PubMed] [Google Scholar]
- 10.Yoshioka R, et al. Risk factors for clinical pancreatic fistula after distal pancreatectomy: analysis of consecutive 100 patients. World J Surg. 2010;34:121–5. doi: 10.1007/s00268-009-0300-3. [DOI] [PubMed] [Google Scholar]
- 11.Distler M, et al. Chronic pancreatitis of the pancreatic remnant is an independent risk factor for pancreatic fistula after distal pancreatectomy. BMC Surg. 2014;14:54. doi: 10.1186/1471-2482-14-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gomes RM, Doctor N. Three level risk assessment for pancreatic fistula formation after distal pancreatectomy with a strategy for prevention. Trop Gastroenterol. 2012;33:207–13. doi: 10.7869/tg.2012.50. [DOI] [PubMed] [Google Scholar]
- 13.Soga K, et al. Risk factors for postoperative pancreatic fistula in distal pancreatectomy. Hepatogastroenterology. 2011;58:1372–6. doi: 10.5754/hge09255. [DOI] [PubMed] [Google Scholar]
- 14.Kawabata Y, Nishi T, Tanaka T, Yano S, Tajima Y. Distal pancreatectomy utilizing a flexible stapler closure eliminates the risk of pancreas-related factors for postoperative pancreatic fistula. Eur Surg Res. 2013;50:71–9. doi: 10.1159/000349977. [DOI] [PubMed] [Google Scholar]
- 15.Eguchi H, et al. A thick pancreas is a risk factor for pancreatic fistula after a distal pancreatectomy: selection of the closure technique according to the thickness. Dig Surg. 2011;28:50–6. doi: 10.1159/000322406. [DOI] [PubMed] [Google Scholar]
- 16.Sugimoto M, et al. Risk factor analysis and prevention of postoperative pancreatic fistula after distal pancreatectomy with stapler use. J Hepatobiliary Pancreat Sci. 2013;20:538–44. doi: 10.1007/s00534-013-0596-0. [DOI] [PubMed] [Google Scholar]
- 17.Kah Heng CA, Salleh I, San TS, Ying F, Su-Ming T. Pancreatic fistula after distal pancreatectomy: incidence, risk factors and management. ANZ J Surg. 2010;80:619–23. doi: 10.1111/j.1445-2197.2010.05337.x. [DOI] [PubMed] [Google Scholar]
- 18.Makni A, et al. Risk factors associated with pancreatic fistula after distal pancreatectomy. Tunis Med. 2012;90:148–53. [PubMed] [Google Scholar]
- 19.Sierzega M, Niekowal B, Kulig J, Popiela T. Nutritional status affects the rate of pancreatic fistula after distal pancreatectomy: a multivariate analysis of 132 patients. J Am Coll Surg. 2007;205:52–9. doi: 10.1016/j.jamcollsurg.2007.02.077. [DOI] [PubMed] [Google Scholar]
- 20.Kawai M, Tani M, Yamaue H. Transection using bipolar scissors reduces pancreatic fistula after distal pancreatectomy. J Hepatobiliary Pancreat Surg. 2008;15:366–72. doi: 10.1007/s00534-008-1330-1. [DOI] [PubMed] [Google Scholar]
- 21.Sato N, et al. Rapid postoperative reduction in prognostic nutrition index is associated with the development of pancreatic fistula following distal pancreatectomy. Pancreatology. 2014;14:216–20. doi: 10.1016/j.pan.2014.02.006. [DOI] [PubMed] [Google Scholar]
- 22.Subhedar PD, et al. Risk factors for pancreatic fistula after stapled gland transection. Am Surg. 2011;77:965–70. [PubMed] [Google Scholar]
- 23.Mendoza AS, 3rd, et al. Predictive factors associated with postoperative pancreatic fistula after laparoscopic distal pancreatectomy: a 10-year single-institution experience. Surg Endosc. 2016;30:649–56. doi: 10.1007/s00464-015-4255-1. [DOI] [PubMed] [Google Scholar]
- 24.Okano K, et al. Pancreatic thickness as a predictive factor for postoperative pancreatic fistula after distal pancreatectomy using an endopath stapler. Surg Today. 2013;43:141–7. doi: 10.1007/s00595-012-0235-4. [DOI] [PubMed] [Google Scholar]
- 25.Frozanpor F, et al. Impact of pancreatic gland volume on fistula formation after pancreatic tail resection. JOP. 2010;11:439–43. [PubMed] [Google Scholar]
- 26.Kawai M, et al. Stump closure of a thick pancreas using stapler closure increases pancreatic fistula after distal pancreatectomy. Am J Surg. 2013;206:352–9. doi: 10.1016/j.amjsurg.2012.11.023. [DOI] [PubMed] [Google Scholar]
- 27.Belyaev O, et al. Histomorphological features of the pancreatic remnant as independent risk factors for postoperative pancreatic fistula: a matched-pairs analysis. Pancreatology. 2011;11:516–24. doi: 10.1159/000332587. [DOI] [PubMed] [Google Scholar]
- 28.Ochiai T, et al. Application of polyethylene glycolic acid felt with fibrin sealant to prevent postoperative pancreatic fistula in pancreatic surgery. J Gastrointest Surg. 2010;14:884–90. doi: 10.1007/s11605-009-1149-3. [DOI] [PubMed] [Google Scholar]
- 29.Schnelldorfer T, Mauldin PD, Lewin DN, Adams DB. Distal pancreatectomy for chronic pancreatitis: risk factors for postoperative pancreatic fistula. J Gastrointest Surg. 2007;11:991–7. doi: 10.1007/s11605-007-0187-y. [DOI] [PubMed] [Google Scholar]
- 30.Weber SM, et al. Laparoscopic left pancreatectomy: complication risk score correlates with morbidity and risk for pancreatic fistula. Ann Surg Oncol. 2009;16:2825–33. doi: 10.1245/s10434-009-0597-z. [DOI] [PubMed] [Google Scholar]
- 31.Nakamura M, et al. Prediction of Pancreatic Fistula by Preoperatively Assessable Factors; Retrospective Review of Unified Operations by Single Surgeon. Hepatogastroenterology. 2014;61:834–7. [PubMed] [Google Scholar]
- 32.Diener MK, et al. Efficacy of stapler versus hand-sewn closure after distal pancreatectomy (DISPACT): a randomised, controlled multicentre trial. Lancet. 2011;377:1514–22. doi: 10.1016/S0140-6736(11)60237-7. [DOI] [PubMed] [Google Scholar]
- 33.Kuroshima N, et al. Triple-drug therapy to prevent pancreatic fistula after pancreatectomy in a rat model. Pancreatology. 2016;16:917–21. doi: 10.1016/j.pan.2016.06.011. [DOI] [PubMed] [Google Scholar]
- 34.Zhang H, et al. Systematic review and meta-analysis comparing three techniques for pancreatic remnant closure following distal pancreatectomy. Br J Surg. 2015;102:4–15. doi: 10.1002/bjs.9653. [DOI] [PubMed] [Google Scholar]
- 35.Sikora SS, et al. External pancreatic fistula as a sequel to management of acute severe necrotizing pancreatitis. Dig Surg. 2005;22:446–51. doi: 10.1159/000091448. [DOI] [PubMed] [Google Scholar]
- 36.Fujii T, et al. Influence of Food Intake on the Healing Process of Postoperative Pancreatic Fistula After Pancreatoduodenectomy: A Multi-institutional Randomized Controlled Trial. Ann Surg Oncol. 2015;22:3905–12. doi: 10.1245/s10434-015-4496-1. [DOI] [PubMed] [Google Scholar]
- 37.Kwon HJ, Ha HT, Choi YY, Kim SG. The effects of the end-to-side inverted mattress pancreaticojejunostomy on postoperative pancreatic fistula: a single surgeon’s experience. Ann Surg Treat Res. 2015;89:61–7. doi: 10.4174/astr.2015.89.2.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Maxwell L, et al. Method guidelines for Cochrane Musculoskeletal Group systematic reviews. J Rheumatol. 2006;33:2304–11. [PubMed] [Google Scholar]
- 39.Wang J, et al. Statin use and risk of lung cancer: a meta-analysis of observational studies and randomized controlled trials. PLoS One. 2013;8:e77950. doi: 10.1371/journal.pone.0077950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang W, et al. Laparoscopic versus open total gastrectomy for gastric cancer: an updated meta-analysis. PLoS One. 2014;9:e88753. doi: 10.1371/journal.pone.0088753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mantel N, Haenszel W. Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst. 1959;22:719–48. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


