Abstract
Semi-dwarf traits have been widely introgressed into cereal crops to improve lodging resistance. In sorghum (Sorghum bicolor L. Moench), four major unlinked dwarfing genes, Dw1-Dw4, have been introduced to reduce plant height, and among them, Dw3 and Dw1 have been cloned. Dw3 encodes a gene involved in auxin transport, whereas, Dw1 was recently isolated and identified as a gene encoding a protein of unknown function. In this study, we show that DW1 is a novel component of brassinosteroid (BR) signaling. Sorghum possessing the mutated allele of Dw1 (dw1), showed similar phenotypes to rice BR-deficient mutants, such as reduced lamina joint bending, attenuated skotomorphogenesis, and insensitivity against feedback regulation of BR-related genes. Furthermore, DW1 interacted with a negative regulator of BR signaling, BRASSINOSTEROID INSENSITIVE 2 (BIN2), and inhibited its nuclear localization, indicating that DW1 positively regulates BR signaling by inhibiting the function of BIN2. In contrast to rice and wheat breeding which used gibberellin (GA) deficiency to reduce plant height, sorghum breeding modified auxin and BR signaling. This difference may result from GA deficiency in rice and wheat does not cause deleterious side effects on plant morphology, whereas in sorghum it leads to abnormal culm bending.
Introduction
In crop breeding, breeders have significantly changed plant stature during the selection of improved grain crops. One famous example is the introduction of a semi-dwarf trait into rice and wheat in the 1960s. Compared to normal plants, semi-dwarf plants have lower center of gravity, which increases lodging resistance and thus enables plant to sustain high grain yield. This phenomenon was later referred as the ‘green revolution’. The mechanism of semi-dwarfism that contributed to the ‘green revolution’ was the introduction of mutated alleles of gibberellin (GA) 20-oxidase (semidwarf 1; sd1) in rice1, 2 and DELLA (Rht) in wheat3, encoding a GA biosynthesis enzyme and a dominant repressor of GA signal transduction, respectively. Furthermore, it has also been reported that the semi-dwarfism of barley, caused by the introgression of semi-dwarf 1 (sdw1/denso) into cultivars grown in Europe, probably depends on a defect in an ortholog of rice SD1 4–6. Such wide usage of GA-related mutations to produce semi-dwarf plants has been possible due to a unique feature of GA. That is, GA deficiency specifically causes a decrease in plant height without deleterious side effects on other morphologies or physiologies, whereas dwarfism caused by other mechanisms often induces undesired phenotypes, such as abnormal leaf structure, abnormal internode elongation, and stunted seeds7–10. Therefore, it is rare that mutations involved in mechanisms other than GA deficiency were used to improve lodging resistance in crop breeding. One rare exception is a semi-dwarf barley mutant containing semi-brachytic 1 (uzu), a weak allele of the brassinosteroid (BR) receptor, which is grown in a limited region of East Asia, including areas of Japan, Korea, and China11. Recently, Dockter and Hanssen12 reported that some barley (Hordeum vulgare) mutants defective in BR synthesizing genes, such as genes encoding Δ5-sterol-Δ24-reductase (HvDIMINUTO), C-23 α-hydroxylase (HvCPD), and BR-6-oxidase (HvBRD) also reduce plant height, but their contributions to breeding are very limited. Except for these barley mutants, there has not been a report, to our knowledge, that BR-related mutations have been used to affect plant height in cereal crops.
However, the situation in sorghum (Sorghum bicolor L. Moench) seems to differ from other cereal crops in terms of lodging-resistant breeding. Sorghum is the fifth most important cereal crop, and its grain is the dietary staple for more than 500 million people13. In sorghum breeding, mutations in four major unlinked dwarfing genes, Dw1-Dw4, have been combined to reduce plant height to increase lodging resistance and improve mechanized harvesting14. Among these dwarfing genes, Dw3 and Dw1 have been cloned, but both are unrelated to GA. Dw3 is orthologous to maize Brachytic2, which encodes a gene involved in auxin transport15. Dw1 was recently isolated through quantitative trait locus (QTL) analysis and encodes an uncharacterized protein, whose homologs are widely found in monocots and dicots including Arabidopsis 16, 17. A histological analysis of a SIL-05 near-isogenic line in which Dw1 is replaced by the mutant allele dw1 [Near isogenic line-dw1 (NIL-dw1)] revealed that the longitudinal parenchymal cell lengths of the internode were almost the same between NIL-dw1 and SIL-05 (Dw1), while the number of cells per internode was significantly reduced in NIL-dw1, demonstrating that dw1 reduces the cell proliferation activity in the internodes17. In accordance, knockdown and T-DNA insertion plants of its orthologous genes in rice and Arabidopsis, respectively, showed similar semi-dwarfism17, indicating that sorghum Dw1 and its orthologs are possibly involved in a novel mechanism to regulate plant size. From physiological perspective, it seems reasonable that sorghum does not use GA deficiency, but instead uses other mechanisms for semi-dwarfism, because in contrast to other cereals, GA deficiency in sorghum induces culm bending, which inevitably causes abnormal plant architecture18.
In this study, we characterized the molecular function of DW1 and revealed that Dw1 encodes a novel component of BR signaling. BRs are plant steroid hormones that regulate multiple aspects of growth and development, including cell elongation, cell division, photomorphogenesis, and organ formation19–21. We suggest that DW1 acts as a positive modulator of BR signaling through inhibiting the function of a negative regulator of BR signaling, BRASSINOSTEROID INSENSITIVE2 (BIN2).
Results
DW1 is involved in BR signaling
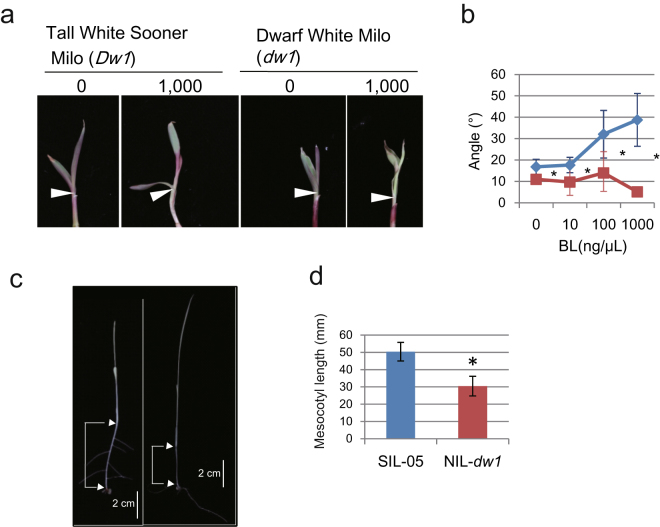
The predicted amino acid sequence of DW1 (Sobic.009G229800) does not contain any characteristic domains or sequence and no role has been proposed for DW1 orthologs in rice or Arabidopsis 17. However, rice in which two Dw1 orthologs (Os01g01390 and Os03g16400) have been down-regulated by RNAi, showed a semi-dwarf phenotype17, erect leaves, and shorter and rounded seeds (Fig. 1), which are typical phenotypes of rice BR-deficient or -insensitive mutants in rice7–10. This prompted us to investigate the BR-related phenotypes of sorghum dw1. First, lamina joint bending upon BR treatment was analyzed. The lamina joint is an organ between the leaf blade and leaf sheath, which acts as a hinge to bend the leaf blade. The degree of lamina inclination in rice is known to be regulated by the synergistic action of BRs and auxin22, 23, although there is no report whether such a phenomenon also occurs in sorghum. The wild type sorghum, ‘Tall White Sooner Milo’, responded to an active form of BR, brassinolide (BL) (Fig. 2a), demonstrating that the lamina inclination test can also be applied to test BR sensitivity in sorghum plants. Thus, we examined the sensitivity of the mutant carrying dw1, ‘Dwarf White Milo’, by using this test. Tall White Sooner Milo carrying Dw1 started to respond to an active form of BL at a concentration of 10 ng/μL (20.8 μM) and continuously increased its bending angle up to a concentration of 1000 ng/μL BL, while Dwarf White Milo (dw1) did not show any clear response even at a concentration of 1000 ng/μL BL (Fig. 2a,b), suggesting that DW1 is involved in BR response. The fact that Dwarf White Milo (dw1) did not respond to exogenously applied BL further suggests that it is not the BR synthesis, but BR signaling that is defective in dw1 sorghum. Next, we examined the effect of DW1 on skotomorphogenesis, which is another phenomenon regulated by BR. We grew sorghum plants in complete darkness and measured the mesocotyl length (Fig. 2c,d). For this experiment, we used a NIL of SIL-05 carrying dw1 (NIL-dw1)17 and compared it to SIL-05 (Dw1). SIL-05 developed an elongated mesocotyl in response to darkness, while NIL-dw1 produced shorter mesocotyls than SIL-05 (Fig. 2c,d). This observation again demonstrates that DW1 is involved in BR response.
Figure 1.
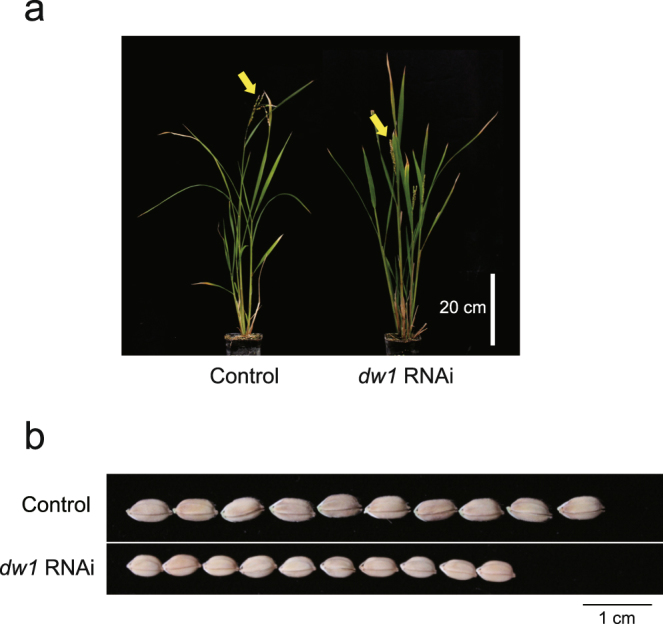
Phenotypes of Os01g01390 and Os03g16400 rice RNAi plant (dw1 RNAi). (a) Plant stature of dw1 RNAi plant. Vector control (Control) and dw1 RNAi plants are presented. The positions of the panicles are shown by yellow arrows. (b) dw1 RNAi plant shows short and rounded seeds. Ten seeds of vector control (Control) and RNAi plants are presented.
Figure 2.
Plants carrying the dw1 allele show defects in BL response. (a) Lamina joint bending test. Arrowheads indicate the position of the lamina joint. The addition of 1,000 ng/μL of BL increased the lamina joint bending of Tall White Sooner Milo (Dw1), but not of Dwarf White Milo (dw1). (b) Dose response of lamina joint bending in Tall White Sooner Milo (blue line) and Dwarf White Milo (red line) to BL (n = 10). Asterisks indicate P < 0.05 using Student’s t-test. (c) SIL-05 (left) and NIL-dw1 (right) plants grown in complete darkness. The size of the mesocotyl is shown by square brackets. (d) Quantitative representation of (c) (n = 11). Asterisks indicate P < 0.05 using Student’s t-test.
Expression of BR-related genes in dw1
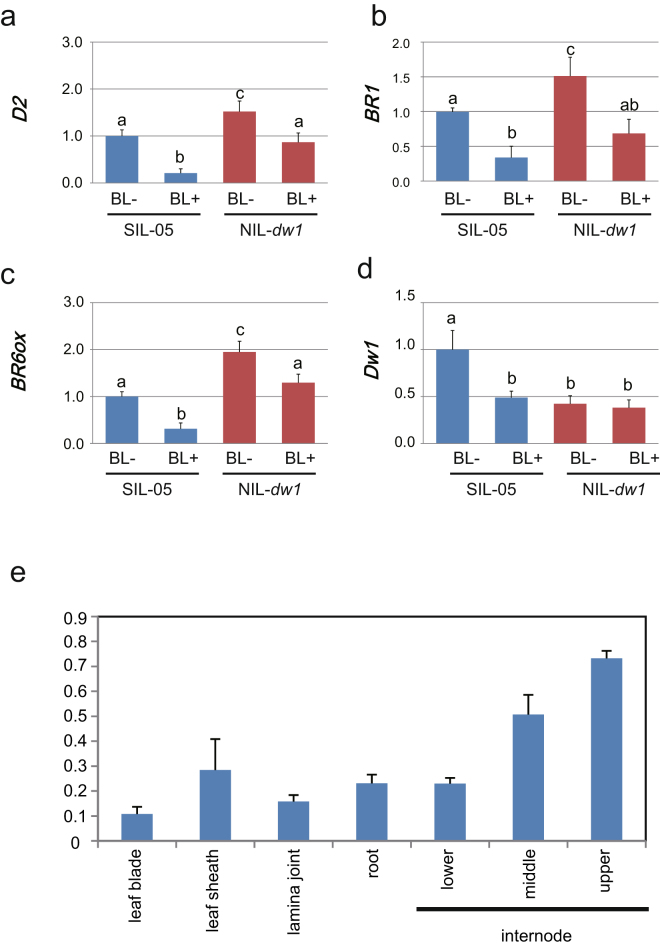
To further confirm that DW1 is involved in BR signaling, we examined the expression of BR-related genes in dw1. Activation of BR signaling in turn inhibits transcription of some BR biosynthesis and signaling genes in rice and Arabidopsis as a feedback mechanism to fine-tune BR response24–26. Sorghum BR genes, which correspond to the ebisu dwarf (SbD2), BR 6-oxidase (SbBR6ox), and BRASSINOSTEROID INSENSITIVE 1 (SbBRI1) genes were identified through phylogenetic analysis (see Supplementary Figs S1–S3), and their expression was analyzed. D2 and BR6ox encode enzymes for the late steps of BR biosynthesis and BRI1 encodes a BR receptor. The corresponding genes of sorghum showed significantly higher expression in NIL-dw1 than in its original strain, SIL-05 (Fig. 3a–c). When BL was applied to these seedlings, their expression was repressed in both plants; however, the suppressive effect of BL was milder in NIL-dw1 than that in SIL-05 (Fig. 3a–c), indicating that BR signaling is attenuated in NIL-dw1. The BL treatment also repressed the expression of Dw1 in SIL-05 but not in NIL-dw1 (Fig. 3d), also confirming that DW1 is related to BR signaling. The reason for the reduced expression of dw1 in NIL-dw1 (BL−) compared to Dw1 in SIL-05 (BL−) is unknown. Because dw1 does not contain a mutation in its promoter region and is assumed to be a null allele caused by a premature stop codon in its coding region, the altered mRNA might have affected dw1 expression.
Figure 3.
Expression of BR-related genes. (a–d) BL feedback regulation of Dw1 and BR-related genes. Expression of SIL-05 (blue bars) and NIL-dw1 (red bars) treated with or without BL are shown. Bars labeled with different letters differ significantly (Tukey–Kramer HSD test, P < 0.05). (e) Expression of Dw1 in various organs of Tall White Milo. Expression levels are shown relative to the expression of a sorghum ubiquitin gene. More than 3 individuals were analyzed.
Dw1 is highly expressed in the young internode
We next analyzed the expression of Dw1. Field grown Tall White Milo (Dw1) 1–2 weeks before the heading stage was used for the analysis. Expression of Dw1 was detected in all of the organs analyzed, including the leaf blade, the sheath, and the root (Fig. 3e). When internodes were divided into lower, middle, and upper regions, the expression increased from the lower to the upper part of the internode. The lower part of the internodes elongate at the early vegetative stage, whereas the uppermost internode extensively elongates just prior to the heading stage. That Dw1 showed the highest expression in the upper internode is consistent with DW1 functioning in internode elongation16, 17.
DW1 inhibits nuclear localization of BIN2
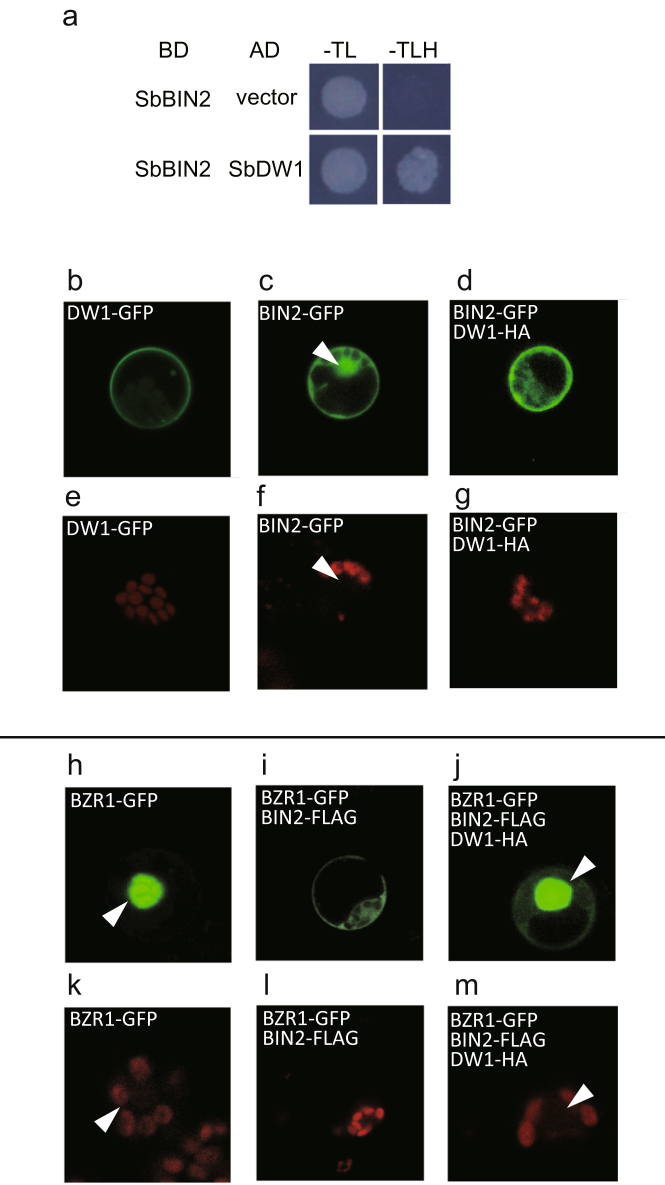
Various factors that play a positive role in BR signaling interact with BIN2. Thus, we performed a yeast two-hybrid assay (Y2H) to test the physical interaction between DW1 and BIN2. For this purpose, we isolated sorghum ortholog of BIN2 (SbBIN2) (see Supplementary Fig. S4). As expected, DW1 and SbBIN2 tightly interacted in yeast cells (Fig. 4a). Similarly, rice DW1 orthologs (LOC_Os01g01390 and LOC_Os03g16400) also interacted with rice BIN2 (OsBIN2), respectively (see Supplementary Fig. S5). To further investigate the effect of the DW1-BIN2 interaction in planta, cellular localization of sorghum DW1, BIN2, and BRASSINAZOLE RESISTANT 1 (BZR1) were analyzed (Fig. 4b–d). The analysis was conducted in rice protoplasts because the transient expression using sorghum protoplasts did not work. When DW1 fused with Green Fluorescent Protein (GFP) was expressed, the GFP signal was localized on the plasma membrane or in the cytosol (Fig. 4b), whereas as previously reported, BIN2 was localized in the cytosol or in the nucleus (Fig. 4c, arrowhead indicates the nucleus). When both BIN2-GFP and DW1-Human influenza hemagglutinin (HA) were co-transformed, the GFP signal of BIN2 disappeared from the nucleus (Fig. 4d), suggesting that DW1 inhibits the nuclear localization of BIN2. Next, because BIN2 is known to phosphorylate BZR1 and direct BZR1 out of the nucleus, the effect of DW1 on BZR1 localization was investigated. BZR1-GFP alone localized to the nucleus and the cytosol (Fig. 4h), whereas co-transformation with BIN2-FLAG led the GFP signal to be restricted to the cytosol (Fig. 4i), which is consistent with previous reports that BIN2 phosphorylates and inhibits the transcriptional activity of BZR1 by directing BZR1 out of the nucleus27, 28. When BZR1-GFP, BIN2-FLAG, and DW1-HA were co-transformed, the GFP signal of BZR1 was localized to the nucleus (Fig. 4j). Similar results were also obtained when DW1, BIN2, and BZR1 of Arabidopsis were expressed in Arabidopsis protoplasts (see Supplementary Fig. S6). These observations suggest that DW1 localizes to the plasma membrane and the cytosol and interacts with BIN2 to suppress the repressive function of BIN2, allowing BZR1 to localize to the nucleus (see Discussion).
Figure 4.
DW1 interacts with BIN2 and inhibits BIN2 from localizing to the nucleus. (a) Interaction of SbDW1 and SbBIN2 in a Y2H assay. The growth of yeast strain AH109 transformants is shown. SbBIN2 was used as the bait, and SbDW1 was used as the prey. −TL, synthetic complete medium lacking Trp and Leu; −TLH, synthetic complete medium lacking Trp, Leu, and His. (b–d, h–j) Transient expression of DW1, BIN2, and BZR1 in rice protoplasts. (b) DW1 localizes on the plasma membrane and in the cytosol. (c) BIN2 localizes to the nucleus and the cytosol. (d) Simultaneous expression of DW1 and BIN2 inhibits nuclear localization of BIN2. (h) BZR1 localizes to the nucleus and the cytosol. (i) Simultaneous expression of BZR1 and BIN2 inhibits nuclear localization of BZR1. (j) DW1, BIN2, and BZR1 co-expression results in BZR1 localizing to the nucleus. Rice protoplasts were transformed with either BIN2-GFP, BZR1-GFP, DW1-GFP, and co-transformed with BIN2-FLAG, DW1-HA, or BIN2-FLAG/DW1-HA. (e–g, k–m) Chloroplast autofluorescence of rice protoplasts are shown as the red channel.
Emergence of DW1 in the plant lineage
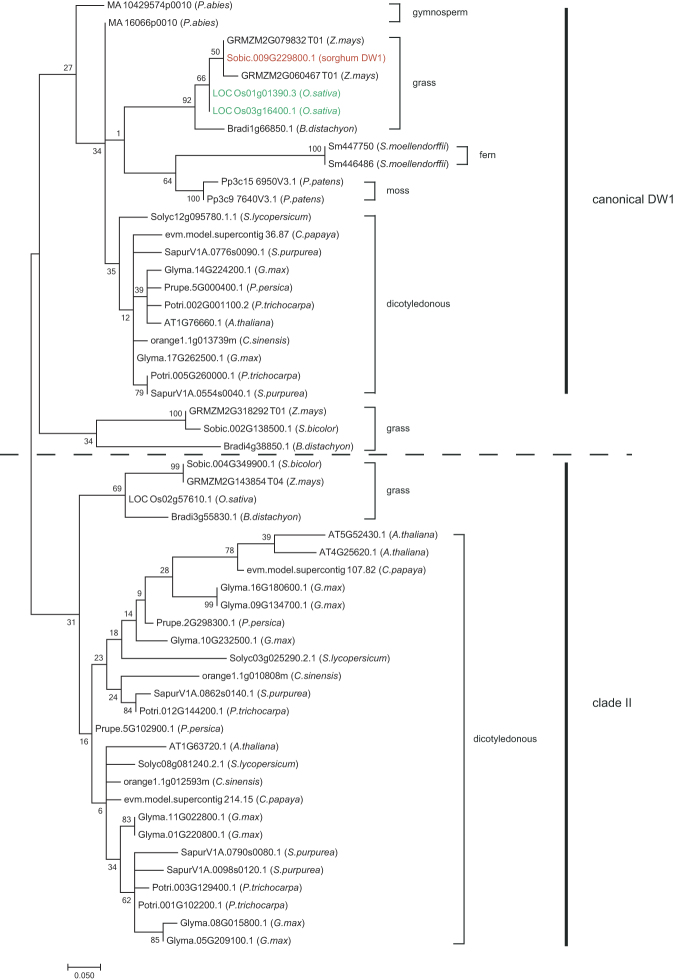
Wang et al.29 proposed that canonical BR signaling originated after the split of gymnosperms and angiosperms. This assumption is based on BRI1 KINASE INHIBITOR 1 (BKI1), a negative regulator of BR signaling, being present only in the angiosperm lineage, while homologs or orthologs of the other 8 BR signaling components [BRI1, BRASSINOSTEROIDINSENSITIVE1-ASSOCIATED RECEPTOR KINASE1 (BAK1), CONSTITUTIVE DIFFERENTIAL GROWTH1 (CDG1), BRASSINOSTEROID INSENSITIVE1 SUPPRESSOR1 (BSU1), BIN2, BZRs, PROTEIN PHOSPHATASE 2A (PP2A), GENERAL REGULATORYFACTORS (GRFs)] are found in all the land plants and also in algae (Fig. 5). The only exception is that BZRs are missing in some of the algae species including red algae. To investigate when DW1 originated in plant evolution, we searched for the existence of DW1 homologs in 15 land plants and 5 chlorophytes, for which the whole genome sequences are available (https://phytozome.jgi.doe.gov/pz/portal.html, http://congenie.org/). Our data set includes the genome sequences of 4 grasses (Sorghum bicolor, Oryza sativa, Brachypodium distachyon, and Zea mays), 8 dicots (Arabidopsis thaliana, Glycine max, Solanum lycopersicum, Prunus persica, Citrus sinensis, Carica papaya, Salix purpurea, and Populus trichocarpa), 1 gymnosperm (Picea abies), 1 lycophyte (Selaginella moellendorffii, a fern), 1 bryophyte (Physcomitrella patens, a moss), and 5 chlorophytes (Chlamydomonas reinhardtii, Coccomyxa subellipsoidea, Micromonas pusilla, Ostreococcus lucimarinus, and Volvox carteri). Similar to those observed for 8 BR signaling components, DW1 homologs were present in all of the land plants including S. moellendorffii and P. patens with varying copy numbers of homologs in each species (see Supplementary Table S1). However, a DW1 homolog could not be found in any of the 5 chlorophyte genomes, indicating that DW1 arose in the land plant lineage after the divergence from chlorophytes. When a phylogenetic tree of DW1 homologs was constructed, two large clades were observed (Fig. 6, see also Supplementary Figs S7 and S8). One clade consisted of SbDW1 and thus we named it as the canonical DW1 clade. Within this clade, there were DW1 homologs of fern (Sm44686 and Sm447750) and moss (Pp3c9 7640v3.1 and Pp3c15 6950V3.1), whereas, in the second clade (clade II), no homologs of fern and moss were present, indicating that clade II DW1 homologs originated from canonical DW1 in seed plants. Within the canonical DW1 clade, each plant species contained either one or several homologs, suggesting that either some species lost or duplicated the Dw1 gene homologs during evolution.
Figure 5.
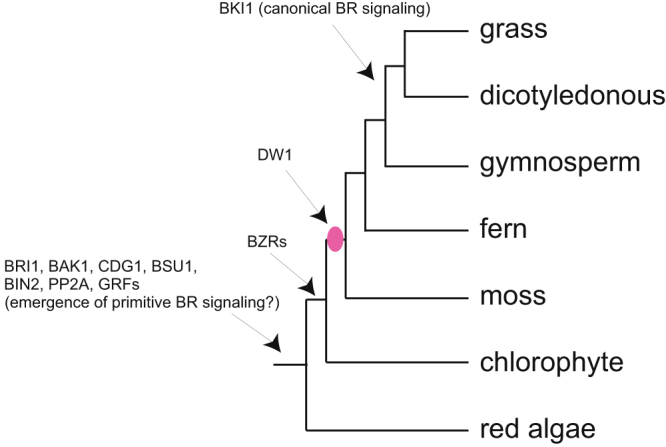
Origin of DW1 in plants. The pink circle indicates the point of possible emergence of DW1. BKI1 arose after the split of gymnosperms and angisperms, whereas the other 8 BR signaling components arose at early stages of plant evolution29.
Figure 6.
Phylogenetic analysis of DW1 homologs. A maximum likelihood tree based on the JTT model38 was obtained. The horizontal branch lengths are proportional to the estimated number of amino acid substitutions per residue. Bootstrap values were obtained by 100 bootstrap replicates. SbDW1 are shown in red, and rice DW1 homologs used to create the RNAi rice are shown in green.
Discussion
In this study, molecular biological experiments demonstrated that DW1 is positively involved in BR signaling. Simultaneous knockdown of two rice canonical Dw1s led to semi-dwarfism with erect leaves and shortened and rounded seeds (Fig. 1). Dwarf White Milo carrying dw1 did not respond to BL treatment in a lamina joint inclination assay (Fig. 2a,b). In addition, compared to SIL-05 (Dw1), NIL-dw1 had a shorter mesocotyl length when grown in the dark (Fig. 2c,d). We also observed that the feedback regulation of BR-related genes in SIL-05 was attenuated in NIL-dw1 (Fig. 3a–d).
DW1 directly interacted with a negative regulator of BR signaling, BIN2 (Fig. 4a), and our transient expression analyses showed that DW1, which localizes to the plasma membrane and the cytosol, functions as a modulator of BR signaling by regulating the intracellular localization of BIN2. That is, expression of DW1 eliminated BIN2 from the nucleus, which in turn allowed nuclear localization of BZR1 (Fig. 4d,j). Up to now, three BIN2 inactivating mechanisms have been proposed. In the first mechanism, BRs enhance BIN2 degradation mediated by the 26S proteasome by an unknown mechanism30. Another mechanism is that direct binding of HISTONE DEACETYLASE 6 to BIN2 deacetylates BIN2, which leads to inhibition of BIN2 kinase activity and represses BIN2 function31. The third inactivating mechanism is similar to that proposed for DW1. OCTOPUS (OPS), a polarly localized plasma membrane-associated protein that functions in phloem differentiation, recruits BIN2 to the plasma membrane32. Localization of BIN2 to the plasma membrane by OPS prevents BIN2 from phosphorylating BZR1 and BES1 in the nucleus. Although OPS is specifically expressed in the phloem, it would be interesting to examine if any interaction occurs between OPS and DW1 or if they each independently function to inhibit BIN2 to the localize to the nucleus.
Our comparative genomic analysis suggests that DW1 arose in the land plant lineage after the divergence from chlorophyta (Fig. 5). This is in clear contrast to the other BR components, which are present even in algae, with the exception of BKI1 which originated after the split of gymnosperms and angiosperms29. It is tempting to speculate that if a primitive BR signaling lacking BKI1 exists in plants other than angiosperms, the emergence of DW1 in land plants should have enabled plants to fine-tune BR signaling through BIN2 inactivation.
To reduce plant height to aid cultivation, sorghum was bred to contain four mutant genes, Dw1-Dw4, whereas in rice and wheat, modifying a single gene related to GA was sufficient. Rice and wheat can repress GA signaling to a certain degree without accompanying deleterious effects on plant morphology, but in sorghum, it leads to abnormal culm bending18. This led to a conundrum in sorghum because reducing plant height by severely modifying other mechanisms often leads to negative side effects in plants. To avoid these side effects in sorghum, it might have been necessary to combine several mutations, each only mildly affecting different hormone signaling. A mutation in one of these four genes, Dw1, may cause only a mild effect on BR signaling because it is not an evolutionarily conserved component of BR signaling. Our phylogenic analysis of DW1 homologs shows that DW1 arose at a later stage in plant evolution than other BR signaling factors and therefore might have been a suitable component to mutate as it functions in fine-tuning BR signaling rather than being a central component of it.
Methods
Rice transformation
Construction and transformation of RNAi transgenic rice to simultaneously knockdown Os01g01390 and Os03g16400 were previously described17.
Lamina joint inclination assay and skotomorphogenesis analysis
The lamina joint inclination assay was performed according to Fujioka et al.33. BL was dissolved in ethanol at final concentrations of 10 ng/μL, 100 ng/μL, and 1000 ng/μL. Germinated seeds were selected for uniformity of coleoptile length, transplanted onto 0.7% agar medium, and grown at 30 °C for 2 days. The seedlings were injected at the top of the lamina with 1 μL of ethanol solution containing 0, 10, 100, or 1,000 ng of BR intermediates with 5 μg indole-3-acetic acid. After incubating for 3 days, the angle between the lamina and its leaf sheath was measured33. There were 10 plants in each treatment. For the skotomorphogenesis analysis, NILs were germinated and grown on 0.65% agar in complete darkness at 30 °C for 2 weeks and then the lengths of mesocotyls were measured.
RNA extraction and semi-quantitative reverse transcription polymerase chain reaction (RT-PCR) analyses
To analyze feedback regulation using semi-quantitative RT-PCR analysis, SIL-05 and NIL-dw1 were grown on Murashige and Skoog basal salt mixture (MS) agar in a growth chamber for 2 weeks under constant light at 30 °C. For BL treatment, BL (Daiichi Fine Chemical) dissolved in 0.01% ethanol was included in the MS agar. Mock-treated plants were grown on MS agar containing 0.01% ethanol. Total RNA was isolated from whole seedlings using an RNeasy Plant Mini Kit (Qiagen). The first-strand complementary DNA (cDNA) was synthesized using an Omniscript RT Kit (Qiagen) and was used in a real-time PCR detection system (Bio-Rad CFX96). Gene expression was normalized to that of sorghum ubiquitin (Sobic.001G311100; SbUbi), and ratios of the expression levels in NIL-dw1 compared to SIL-05 (set as 1.0) were calculated. Three repeats were performed for each gene, and the average values and standard deviations are shown. To analyze the global expression of Dw1, field grown Tall White Milo was collected before heading. Other procedures were conducted identical to those stated above. Primer sequences are listed in Supplementary Table S2.
Yeast Two-Hybrid
To construct DNA binding domain (BD)-SbBIN2 and Activation domain (AD)-SbDW1, SbBIN2 and SbDW1 cDNA were PCR amplified and cloned into the EcoRI/SalI site of pGBKT7 and BamHI/XhoI site of pGADT7, respectively. The yeast two-hybrid assay (Y2H) was performed as described previously34 using the BD Matchmaker Two-Hybrid System 3 (Clontech). Vector cassettes for the DNA-AD were used as negative controls, and Saccharomyces cerevisiae strain AH109 was used as the host. Details of the methods used for the yeast assays can be found in the manufacturer’s instructions (Yeast Protocols Handbook #PT3024-1; http://www.clontech.com/).
Transient expression assay using Arabidopsis mesophyll protoplasts
Three plasmids, Dw1-GFP/pE2113, BIN2-GFP/pE2113, and BZR1-GFP/pE2113 were constructed for the transient assay as follows. A PCR-amplified GFP gene fragment was introduced into the SpeI/StuI site of the pE2113_GW_SAS vector35. Then PCR-amplified protein-coding regions of Dw1 (Sobic.009g229800), BIN2 (At4g18710) and BZR1 (At1g75080) were introduced into the XbaI/SpeI site of the vector in the same reading frame as GFP. To construct the plasmids Dw1-HA/pE2113 and BIN2-FLAG/pE2113, a synthetic DNA encoding the HA tag and a PCR-amplified FLAG fragment were introduced into SpeI/StuI site instead of GFP. All of the genes were driven by the 35S promoter, and all of the constructs were carried out using the In-Fusion HD Cloning Kit (Clontech). The plasmids were extracted using the QIAGEN Plasmid Maxi Kit (QIAGEN) and used for the transient expression assay. The sequences of the primers used for PCR amplification or oligonucleotides used in this study are listed in Supplementary Table S2. Isolation of mesophyll protoplasts and transient gene expression assays were performed according to Zhang et al.36, Wu et al.37 and Ryu et al.27. GFP fluorescence was recorded by a LSM 700 confocal microscope (Zeiss).
Phylogenetic tree analysis
The amino acid sequence of sorghum DW1 was used as a query to screen databases (https://phytozome.jgi.doe.gov/pz/portal.html, http://congenie.org/) by tblastn and candidate genes were selected for phylogenetic analyses. The e-value threshold was set at 5.8E-07. Sequences were aligned with ClustalW (Mega 7) with default parameters, and phylogenetic trees (maximum likelihood, neighbor-joining, and maximum parsimony methods) were created by Mega 7 with default parameters. Bootstrap values were obtained by 100 bootstrap replicates.
Electronic supplementary material
Acknowledgements
This work was partially supported by a grant from the Core Research for Evolutional Science and Technology (CREST) Program “Novel techniques of tailor-made breeding for energy crop improvement using high-throughput genotyping” of Japan Science and Technology Agency (JST); Grants-in-Aid from the NC-CARP project, the Ministry of Education, Science, Sports and Culture of Japan. We thank Y. Tahara for plant cultures in the field. We also thank H. Kitano and M. Matsuoka for helpful suggestions.
Author Contributions
A.F., S.K. and T.S. performed the sorghum field experiments. H.F., S.A.-N., and T.S. performed the lamina joint bending and skotomorphogenesis experiments. K.H., M.K., M.Y., S.A.-N., K.O.-S., and H.S. constructed the plasmids. K.H., M.K., and S.A.-N. performed the transgenic experiments. K.H., S.A.-N., K.O.-S., and H.F. performed the rice experiments. K.H., and M.K. performed the Y2H. S.A.-N., M.Y., and H.S. performed the transient assays. K.H., and T.S. wrote the paper.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-00096-w
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Sasaki A, et al. Green revolution: a mutant gibberellin-synthesis gene in rice. Nature. 2002;416:701–702. doi: 10.1038/416701a. [DOI] [PubMed] [Google Scholar]
- 2.Ashikari M, et al. Loss-of-function of a rice gibberellin biosynthetic gene, GA20 oxidase (GA20ox-2), led to the rice “green revolution”. Breed. Sci. 2002;52:143–150. doi: 10.1270/jsbbs.52.143. [DOI] [Google Scholar]
- 3.Peng J, et al. ‘Green revolution’ genes encode mutant gibberellin response modulators. Nature. 1999;400:256–261. doi: 10.1038/22307. [DOI] [PubMed] [Google Scholar]
- 4.Dahleen LS, van der Wal LJ, Franckowiak JD. Characterization and molecular mapping of genes determining semidwarfism in barley. J. Heredity. 2005;96:654–662. doi: 10.1093/jhered/esi125. [DOI] [PubMed] [Google Scholar]
- 5.Jia Q, et al. GA-20 oxidase as a candidate for the semidwarf gene sdw1/denso in barley. Funct. Integr. Genomics. 2009;9:255–262. doi: 10.1007/s10142-009-0120-4. [DOI] [PubMed] [Google Scholar]
- 6.Jia Q, et al. Expression level of a gibberellin 20-oxidase gene is associated with multiple agronomic and quality traits in barley. Theor. Appl. Genet. 2011;122:1451–1460. doi: 10.1007/s00122-011-1544-5. [DOI] [PubMed] [Google Scholar]
- 7.Hong Z, et al. Loss-of-function of a rice brassinosteroid biosynthetic enzyme, C-6 oxidase, prevents the organized arrangement and polar elongation of cells in the leaves and stem. Plant J. 2002;32:495–508. doi: 10.1046/j.1365-313X.2002.01438.x. [DOI] [PubMed] [Google Scholar]
- 8.Hong Z, et al. A rice brassinosteroid-deficient mutant, ebisu dwarf (d2), is caused by a loss of function of a new member of cytochrome P450. Plant Cell. 2003;12:2900–2910. doi: 10.1105/tpc.014712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hong Z, et al. The rice brassinosteroid-deficient dwarf2 mutant, defective in the rice homolog of Arabidopsis DIMINUTO/DWARF1, is rescued by the endogenously accumulated alternative bioactive brassinosteroid, dolichosterone. Plant Cell. 2005;17:2243–2254. doi: 10.1105/tpc.105.030973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yamamuro C, et al. Loss of function of a rice brassinosteroid insensitive1 homolog prevents internode elongation and bending of the lamina joint. Plant Cell. 2000;12:1591–1605. doi: 10.1105/tpc.12.9.1591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chono M, et al. A semidwarf phenotype of barley uzu results from a nucleotide substitution in the gene encoding a putative BR receptor. Plant Physiol. 2003;133:1209–1219. doi: 10.1104/pp.103.026195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dockter C, Hansson M. Improving barley culm robustness for secured crop yield in a changing climate. J. Exp. Bot. 2015;66:3499–3509. doi: 10.1093/jxb/eru521. [DOI] [PubMed] [Google Scholar]
- 13.Wang, Y. H., Upadhyaya, H. D. & Kole, C. Genetics, Genomics and Breeding of Sorghum. 233 (CRC Press, 2014).
- 14.Quinby JR, Karper RE. Inheritance of height in sorghum. Agron. J. 1954;46:211–216. doi: 10.2134/agronj1954.00021962004600050007x. [DOI] [Google Scholar]
- 15.Multani DS, et al. Loss of an MDR transporter in compact stalks of maize br2 and sorghum dw3 mutants. Science. 2003;302:81–84. doi: 10.1126/science.1086072. [DOI] [PubMed] [Google Scholar]
- 16.Hilley J, Truong S, Olson S, Morishige D, Mullet J. Identification of Dw1, a Regulator of Sorghum Stem Internode Length. PLoS ONE. 2016;11:e0151271. doi: 10.1371/journal.pone.0151271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yamaguchi M, et al. Sorghum Dw1, an agronomically important gene for lodging resistance, encodes a novel protein involved in cell proliferation. Sci Rep. 2016;6:28366. doi: 10.1038/srep28366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ordonio RL, et al. Gibberellin deficiency pleiotropically induces culm bending in sorghum: an insight into sorghum semi-dwarf breeding. Sci. Rep. 2014;4:5287. doi: 10.1038/srep05287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Clouse SD, Sasse JM. Brassinosteroids: essential regulators of plant growth and development. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1998;49:427–451. doi: 10.1146/annurev.arplant.49.1.427. [DOI] [PubMed] [Google Scholar]
- 20.Wang ZY, He JX. Brassinosteroid signal transduction-choices of signals and receptors. Trends Plant Sci. 2004;9:91–96. doi: 10.1016/j.tplants.2003.12.009. [DOI] [PubMed] [Google Scholar]
- 21.Zhu JY, Sae-Seaw J, Wang ZY. Brassinosteroid signalling. Development. 2013;140:1615–1620. doi: 10.1242/dev.060590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wada K, et al. A rice lamina inclination test-a microquantitative bioassay for brassinosteroids. Agric. Biol. Chem. 1984;48:719–726. [Google Scholar]
- 23.Takeno K, Pharis RP. Brassinosteroid-induced bending of the leaf lamina of dwarf rice seedlings: an auxin-mediated phenomenon. Plant Cell Physiol. 1982;23:1275–1281. doi: 10.1093/oxfordjournals.pcp.a076471. [DOI] [Google Scholar]
- 24.He JX, et al. BZR1 is a transcriptional repressor with dual roles in brassinosteroid homeostasis and growth responses. Science. 2005;307:1634–1638. doi: 10.1126/science.1107580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sun Y, et al. Integration of brassinosteroid signal transduction with the transcription network for plant growth regulation in Arabidopsis. Dev. Cell. 2010;19:765–777. doi: 10.1016/j.devcel.2010.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yu X, et al. A brassinosteroid transcriptional network revealed by genome-wide identification of BESI target genes in Arabidopsis thaliana. Plant J. 2011;65:634–646. doi: 10.1111/j.1365-313X.2010.04449.x. [DOI] [PubMed] [Google Scholar]
- 27.Ryu H, et al. Nucleocytoplasmic shuttling of BZR1 mediated by phosphorylation is essential in Arabidopsis brassinosteroid signaling. Plant Cell. 2007;19:2749–2762. doi: 10.1105/tpc.107.053728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Belkhadir Y, Jaillais Y. The molecular circuitry of brassinosteroid signaling. New Phytologist. 2015;206:522–540. doi: 10.1111/nph.13269. [DOI] [PubMed] [Google Scholar]
- 29.Wang H, Mao H. On the origin and evolution of plant brassinosteroid receptor kinases. J Mol Evol. 2014;78:118–129. doi: 10.1007/s00239-013-9609-5. [DOI] [PubMed] [Google Scholar]
- 30.Peng P, Yan Z, Zhu Y, Li J. Regulation of the Arabidopsis GSK3-like kinase BRASSINOSTEROID-INSENSITIVE 2 through proteasome-mediated protein degradation. Mol Plant. 2008;1:338–346. doi: 10.1093/mp/ssn001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hao Y, Wang H, Qiao S, Leng L, Wang X. Histone deacetylase HDA6 enhances brassinosteroid signaling by inhibiting the BIN2 kinase. Proc. Natl. Acad. Sci. U S A. 2016;113:10418–10423. doi: 10.1073/pnas.1521363113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Anne P, et al. OCTOPUS negatively regulates BIN2 to control phloem differentiation in Arabidopsis thaliana. Curr Biol. 2015;25:2584–2590. doi: 10.1016/j.cub.2015.08.033. [DOI] [PubMed] [Google Scholar]
- 33.Fujioka S, Noguchi T, Takatsuto S, Yoshida S. Activity of brassinosteroids in the dwarf rice lamina inclination bioassay. Phytochemistry. 1998;49:1841–1848. doi: 10.1016/S0031-9422(98)00412-9. [DOI] [Google Scholar]
- 34.Hirano K, et al. The suppressive function of the rice DELLA protein SLR1 is dependent on its transcriptional activation activity. Plant J. 2012;71:443–453. doi: 10.1111/j.1365-313X.2012.05000.x. [DOI] [PubMed] [Google Scholar]
- 35.Yoshida H, et al. DELLA protein functions as a transcriptional activator through the DNA binding of the INDETERMINATE DOMAIN family proteins. Proc. Natl. Acad. Sci. USA. 2014;111:7861–7866. doi: 10.1073/pnas.1321669111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang Y, et al. A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant Methods. 2011;7:30. doi: 10.1186/1746-4811-7-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wu FH, et al. Tape-Arabidopsis Sandwich-a simpler Arabidopsis protoplast isolation method. Plant Methods. 2009;5:16. doi: 10.1186/1746-4811-5-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jones DT, Taylor WR, Thornton JM. The rapid generation of mutation data matrices from protein sequences. Comput. Appl. Biosci. 1992;8:275–282. doi: 10.1093/bioinformatics/8.3.275. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.