Abstract
Acute lung injury (ALI) is an acute failure of the respiratory system for which effective treatment is urgently necessary. Previous studies found that several peptides potently inhibited the production of cytokines induced by lipopolysaccharide (LPS). In this study, we synthetized a cell-permeable TIR domain-derived decoy peptide (TM6) and examined its substance for the ability to inhibit TLR signaling in the model of ALI induced by LPS. We demonstrated that TM6 (2.5, 5 and 10 nmol/g) alleviated the histological changes in the lung tissues as well as myeloperoxtidase (MPO) activity, lung W/D ratio, the production of TNF-α, IL-1β and IL-6 induced by LPS. Furthermore, the numbers of total cells, neutrophils and macrophages in the BALF were suppressed by TM6. In vitro, TM6 (5, 10 and 20 µM) inhibited the production of TNF-α, IL-1β and IL-6 in LPS-stimulated alveolar macrophages. Moreover, the activation of Nuclear factor-kappaB (NF-κB) and Mitogen activated protein kinases (MAPK) signaling pathways induced by LPS were also inhibited by TM6. Collectively, our results suggested that TM6 was an effective inhibitor of ALI induced by LPS, and this peptide may very well serve as a future treatment for ALI.
Introduction
Acute lung injury (ALI) has been recognized as a life-threatening disease, which is usually caused by pneumonia, sepsis, shock and aspiration1, 2. The major characteristic of ALI, - which is the key factor leading to Acute Respiratory Distress Syndrome (ARDS), is the neutrophil accumulation, increasing vascular permeability and arousing pulmonary edema3, 4. It is a common clinical problem that presents high morbidity and mortality worldwide5. Although there are some medicine to treat ALI patients, the high mortality rate of ALI even reaches a rate of 30–50%6. Therefore, to cure ALI, specific and effective treatments are urgently needed. Lipopolysaccharide (LPS) is widely accepted to establish ALI models. LPS has the ability to induce the releases of numerous inflammatory mediators, including TNF-α, IL-1β, IL-6, NO and superoxide anions7–9. Additionally, LPS could activate Toll like receptor 4 (TLR4), which subsequently induces the activation of NF-κB and MAPK signaling pathways10, 11.
Activation of TLR4 induces the accumulation of TIR domain-containing adapter protein, including MyD88, TRIAP, TRIF, and TRAM. Upon TLR4 activated, TRAM was served as a bridging adapter directly to recruit TRIF and render the production of cytokines. It has been suggested in some study that the recruitment of adapter proteins is through the interaction of TIR-TIR domains. The TIR domain of TLR4 is consists of five-stranded parallel β-sheet and five α-helices12, 13. We synthetized some TRAM TIR domain-derived decoy peptides and indicated that TM6 or TR6 inhibited the development of mastitis induced by LPS in mice14, 15. It has been reported that certain decoy peptides have been demonstrated modest inhibition to several the signaling outcomes, suggesting the possibility of pathway-specific interaction surfaces13. Pfalzraff A found that synthetic antimicrobial and LPS-neutralising peptides inhibited the production of inflammatory cytokines through inhibiting NF-κB and MAPK signaling pathway16. Other studies have confirmed that TM6 was capable to efficiently prevent the LPS-induced TLR4/MyD88 association and significantly decrease circulating levels of both cytokines17. The TM6 (sequence: “N”-RQIKIWFQNRRMKWK and,-KENFLRDTWCNFQFY-“C”) is derived from the third helical region of TIR domains of TRAM. TM6 exhibited anti-inflammatory effects through the blocking of the docking site of its prototype function protein, which lead to the inhibition of the interaction of TRAM with downstream adaptor molecular. The therapeutic potential of TM6 has ever been mentioned in individual papers14, 17. However, the effects of TM6 on LPS-induced ALI remains unclear. In the present study, we investigated the therapeutic effects and mechanisms of TM6 on LPS-induced ALI both in vivo and in vitro.
Results
The effect of TM6 on histopathology changes in lung tissues
7 h after LPS stimulation, histopathology changes were determined by H&E staining. The lung tissues of LPS group showed several obvious inflammatory changes, such as lung edema, pulmonary congestion, alveolar hemorrhage, thickening of the alveolar wall and areas of inflammatory infiltration (Fig. 1B). Treatment with TM6 (2.5, 5 and 10 nmol/g) and DEX (5 mg/kg) markedly alleviated the histopathology changes induced by LPS (Fig. 1C,D,E,F). In addition, the scores was evaluated to determine the thickness of alveolar walls and epithelium, the increased number of infiltration cells in perbronchial, and perivascular cuff area. As it is shown in Fig. 1G, the mean pathological score was significantly increased after LPS administration compared with control group (0.30 ± 0.003 vs 4.86 ± 0.08). However, the pathological scores were reduced by treatment of TM6 (4.15 ± 0.09, 3.72 ± 0.06, 1.81 ± 0.04, 1.84 ± 0.06 vs 4.86 ± 0.08) in a dose-dependent manner.
Figure 1.
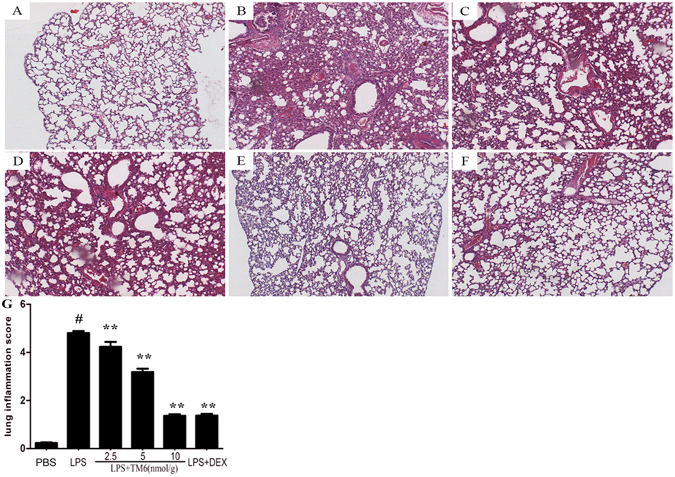
The effect of TM6 on lung tissue histopathological changes. Histopathologic sections of lung tissues (H and E, ×100). (A) Lung tissues from control, (B) LPS, (C) LPS + TM6 (2.5 nmol/g), (D) LPS + TM6 (5 nmol/g), (E) LPS + TM6 (10 nmol/g), and (F) LPS + DEX (5 mg/kg) mice, (G) the pathological scores was evaluate during ALI.
The effect of TM6 on wet/dry lung ratios
Lung W/D ratios were evaluated 7 h after LPS stimulation. The lung W/D ratio was markedly higher after LPS administration compared to the control group. However, TM6 (2.5, 5 and 10 nmol/g) significantly reduced the lung W/D ratio induced by LPS. In addition, DEX (5 mg/kg) also reduced the Lung W/D ratios after LPS stimulation (Fig. 2).
Figure 2.
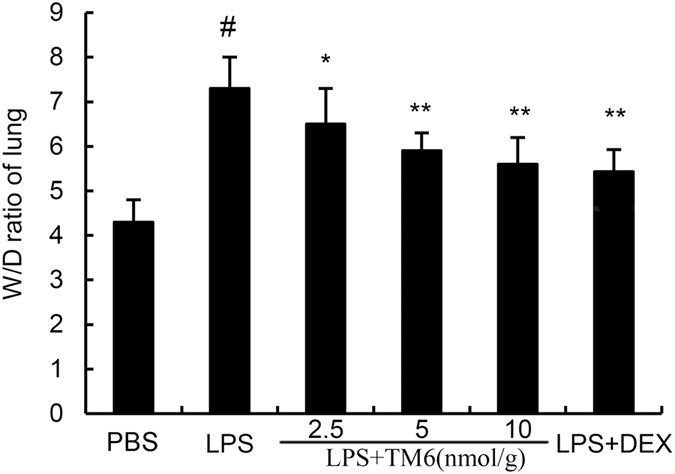
The effect of TM6 on lung wet/dry radio. Mice were pretreated with TM6 (2.5, 5 and 10 nmol/g) and DEX (5 mg/kg) 1 h prior to an i.n. administration of LPS. The lung wet/dry ratio was determined 7 h after LPS challenge. The values presented are the means ± SEM (n = 6). #P < 0.01 is significantly different from the control group; *P < 0.05 and **P < 0.01 are significantly different from the LPS group.
The effect of TM6 on inflammatory cell counts in the Bronchial Alveolar Lavage Fluid (BALF)
Seven hours after LPS stimulation, the numbers of total cells, neutrophils, and macrophages in BALF were measured. The results showed that stimulation with LPS caused a markedly increase in total cells compared with control group (10.93 ± 0.18 vs 2.51 ± 0.06 × 108/ml), and macrophages (4.02 ± 0.12 vs 1.16 ± 0.03 × 108/ml) and neutrophil production (6.17 ± 0.20 vs 1.82 ± 0.08 × 108/ml). However, treatment of TM6 (2.5, 5 and 10 nmol/g) does-dependently suppressed the numbers of total cells (8.82 ± 0.11, 7.92 ± 0.09, 6.21 ± 0.10 × 108/ml), neutrophils (5.52 ± 0.18, 4.49 ± 0.15, 3.82 ± 0.17 × 108/ml) and macrophages (3.61 ± 0.13, 3.38 ± 0.14, 2.33 ± 0.08 × 108/ml) induced by LPS (Fig. 3). DEX also reduced the total cells (6.02 ± 0.10 × 108/ml), neutrophils (3.72 ± 0.13 × 108/ml), and macrophages (2.31 ± 0.13 × 108/ml) production compared with LPS group (Fig. 3).
Figure 3.
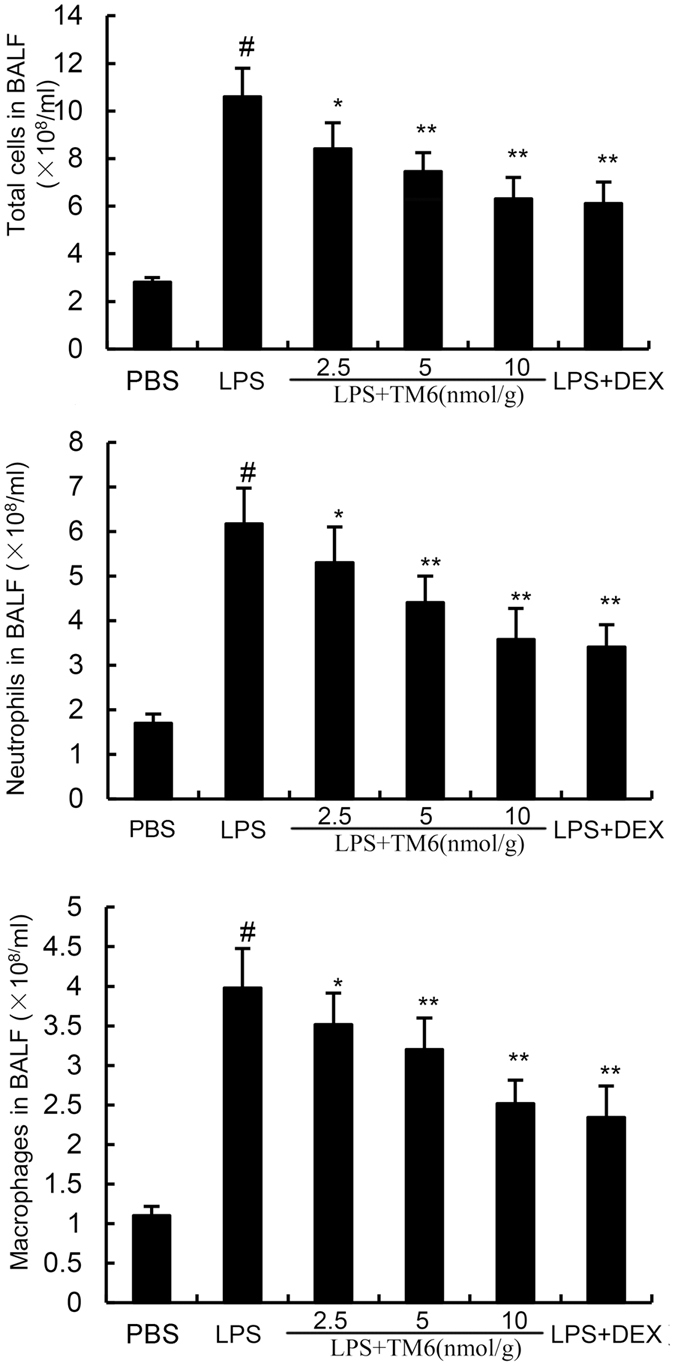
The effects of TM6 on the number of total cells, neutrophils, and macrophages in the BALF. Mice were given an intraperitoneal injection of TM6 (2.5, 5 and 10 nmol/g) and DEX (5 mg/kg) 1 h prior to an i.n. administration of LPS. BALF was collected 7 h after LPS administration to measure the number of total cells (a), neutrophils (b), and macrophages (c). The values presented are the means ± SEM (n = 6). #P < 0.01 is significantly different from the control group; *P < 0.05 and **P < 0.01 are significantly different from the LPS group.
The effect of TM6 on the production of cytokines in BALF
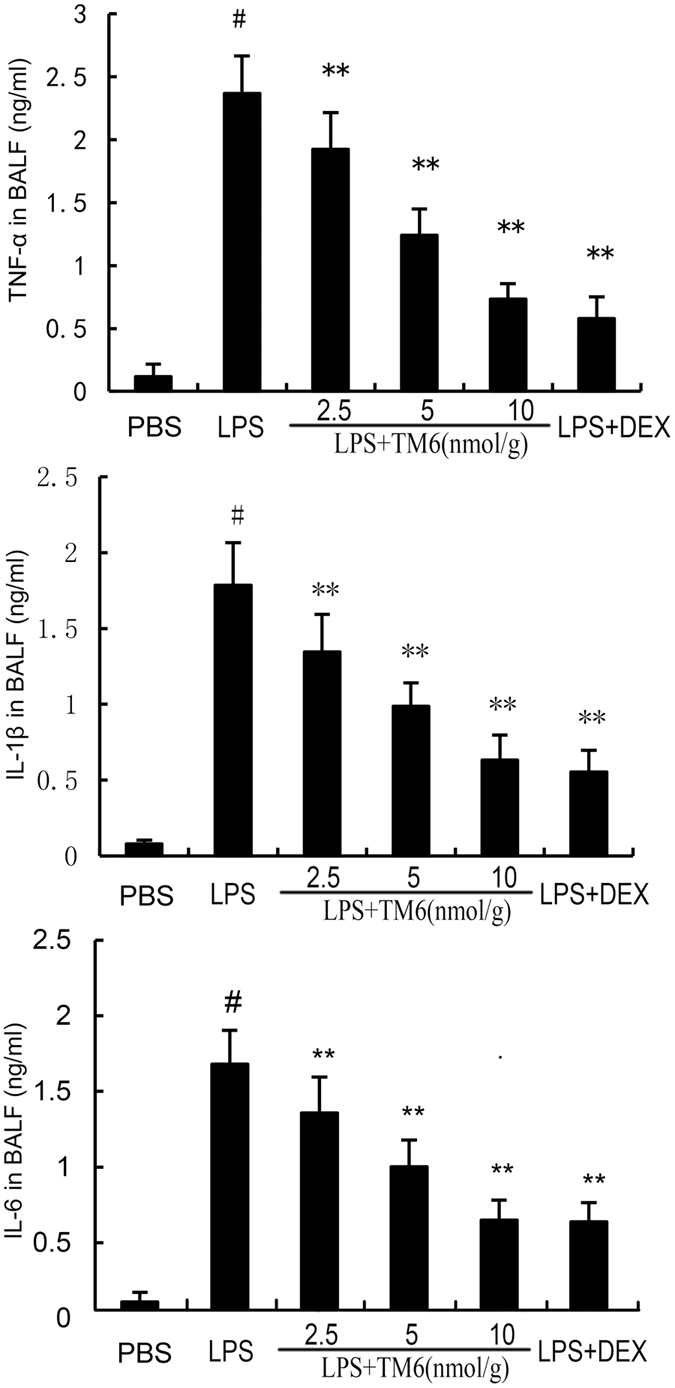
To illustrate the effect of TM6 on inflammatory cytokines production, we tested the levels of TNF-α, IL-1β and IL-6 by ELISA. 7 h after LPS stimulation, an evident elevation of TNF-α (2.48 ± 0.08 vs 0.15 ± 0.02 ng/ml), IL-1β (1.81 ± 0.06 vs 0.11 ± 0.01 ng/ml) and IL-6 (1.62 ± 0.05 vs 0.09 ± 0.007 ng/ml) in BALF of LPS group were observed when compared with control group. The levels of TNF-α (1.87 ± 0.09, 1.12 ± 0.05, 0.70 ± 0.02, 0.51 ± 0.04 vs 2.48 ± 0.08 ng/ml), IL-1β (1.43 ± 0.06, 1.13 ± 0.02, 0.72 ± 0.03, 0.66 ± 0.02 vs 1.81 ± 0.06 ng/ml) and IL-6 (1.42 ± 0.07, 1.01 ± 0.04, 0.62 ± 0.018, 0.62 ± 0.019 vs 1.62 ± 0.05 ng/ml) in TM6 (2.5, 5, and 10 nmol/g) or DEX (5 mg/kg) groups decreased significantly when compared with the LPS group (Fig. 4).
Figure 4.
The effect of TM6 on cytokines production in BALF. Mice were given an intraperitoneal injection of TM6 (2.5, 5 and 10 nmol/g) and DEX (5 mg/kg) 1 h prior to an i.n. administration of LPS. BALF was collected 7 h following LPS challenge to analyze the inflammatory cytokines TNF-α, IL-1β, and IL-6. The values presented are means ± SEM (n = 6). # P < 0.01 is significantly different from the control group; * P < 0.05 and ** P < 0.01 are significantly different from the LPS group.
The effect of TM6 on myeloperoxtidase (MPO) activity
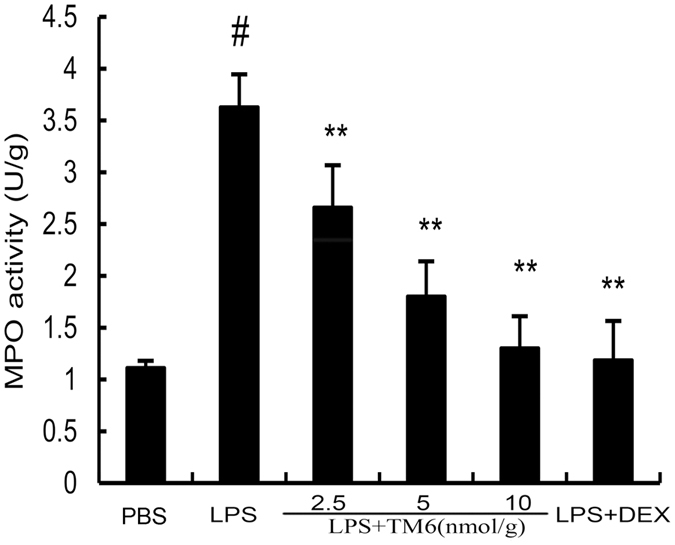
MPO activity served as a marker of the development of LPS induced ALI. MPO activity was evaluated 7 h after LPS stimulation. The results showed that lung MPO activity was up-regulated after LPS administration compared with control group. TM6 (2.5, 5, and 10 nmol/g) and DEX (5 mg/kg) administration prevented elevated MPO activity after LPS administration (Fig. 5).
Figure 5.
The effect of TM6 on MPO activity. MPO activity was determined 7 h after LPS administration. The values presented are the means ± SEM. (n = 6) # P < 0.01 is significantly different from the control group; * P < 0.05 and ** P < 0.01 are significantly different from the LPS group.
The effect of TM6 on cell viability
MTT assay was used to measure the effect of TM6 on the viability of alveolar macrophages. The results showed that TM6 did not affect alveolar macrophages viability at concentrations with 5, 10 and 20 µM (Fig. 6). It suggests that TM6 at the concentrations of 5, 10 and 20 µM did not have any cytotoxic effects on alveolar macrophages.
Figure 6.
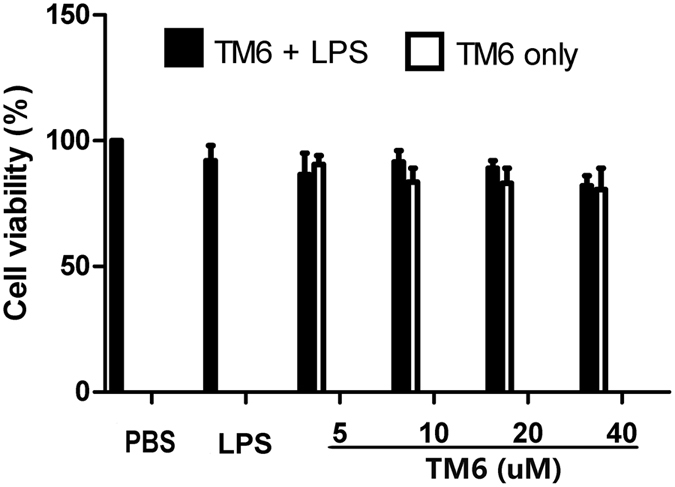
The effect of TM6 on cell viability. Alveolar macrophages extract from mice were treatment with different concentrations of TM6 (0–40 µM) in the absence or presence LPS (1 µg/ml) for 24 hours. The cells viability was tested by MTT assay. The values presented are means ± SEM (n = 6). # P < 0.01 is significantly different from the control group; * P < 0.05 and ** P < 0.01 are significantly different from the LPS group.
The effect of TM6 on inflammatory cytokines production in alveolar macrophages
Alveolar macrophages were treated with TM6 (5, 10 and 20 µM) and DEX (1 mM) 1 hour before LPS treatment. 24 hours later, the inflammatory cytokines TNF-α, IL-1β and IL-6 in the supernatant of alveolar macrophages were tested by ELISA. As shown in Fig. 7, after LPS treatment, the production of TNF-α, IL-1β and IL-6 were significantly increased compared with control group. However, TM6 dose-dependently inhibited the production of TNF-α, IL-1β and IL-6 after stimulation with LPS in alveolar macrophages.
Figure 7.
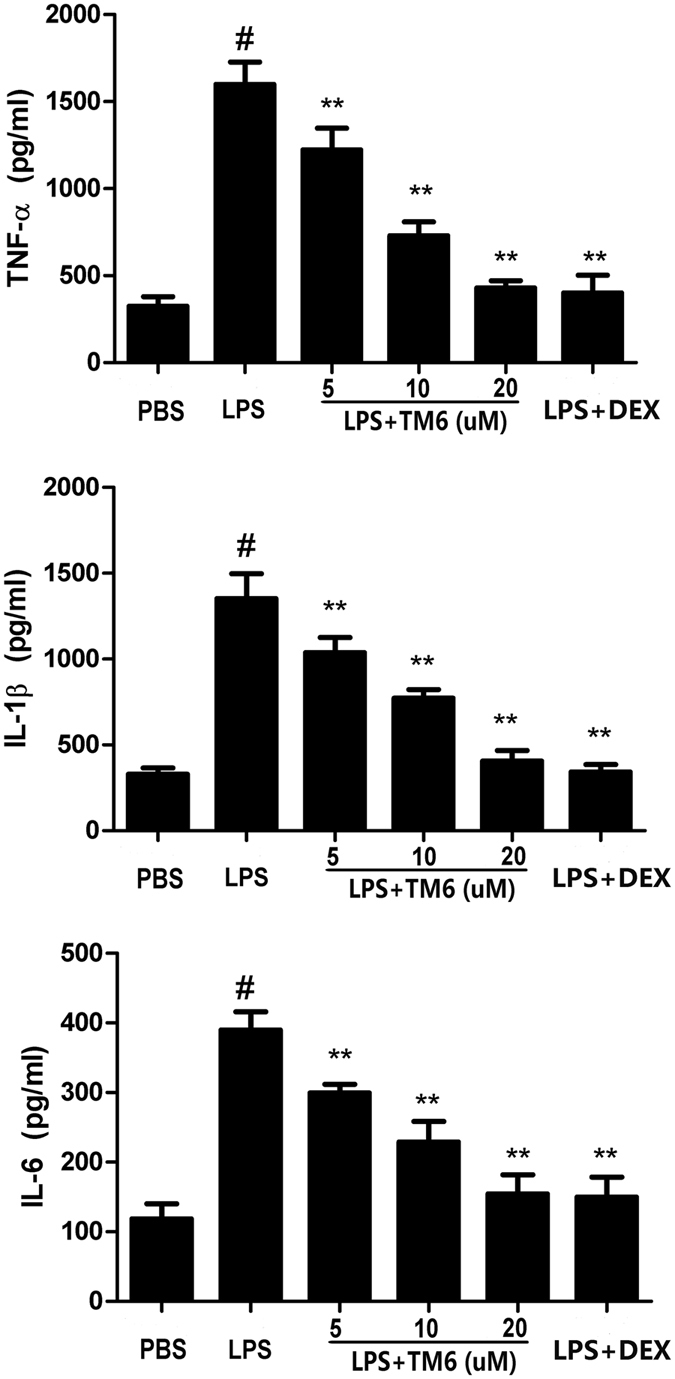
The effect of TM6 on inflammatory cytokines production in alveolar macrophages. Alveolar macrophages were treated with TM6 (5, 10 and 20 µM) and DEX (1 mM) 1 hour prior to stimulation with LPS. 24 hours later, the suspension was collected to test the levels of TNF-α, IL-1β, and IL-6. The values presented are means ± SEM (n = 6). # P < 0.01 is significantly different from the control group; * P < 0.05 and ** P < 0.01 are significantly different from the LPS group.
The effect of TM6 on NF-κB and MAPK signaling pathways expression
To investigate the- protective mechanisms by which TM6 inhibits inflammatory cytokines production in alveolar macrophages, we detected NF-κB and MAPK signaling pathways by western blot analysis. The results showed that expression of p-IκB-α, p-p65, p-p38, p-ERK, and p-JNK increased significantly after LPS stimulation. However, TM6 (5, 10 and 20 µM) dose-dependently inhibited the expression p-IκB-α, p-p65, p-p38, p-ERK, and p-JNK induced by LPS (Figs 8 and 9).
Figure 8.
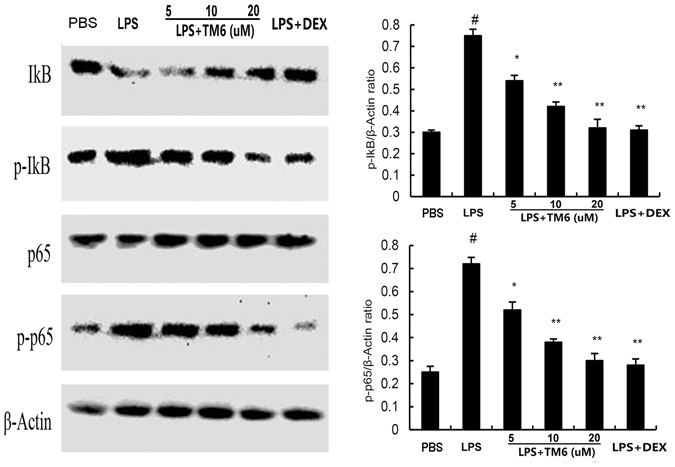
The effect of TM6 on NF-κB pathway. The effects of TM6 on the expression of the NF-κB pathway that was induced by LPS. Alveolar macrophages were pretreated with TM6 (5, 10 and 20 µM) for 1 hour and then treated with LPS for 1 hour. NF-κB protein samples were analyzed by western blot with specific antibodies. β-actin was used as a control. The values presented are the means ± SEM of three independent experiments, # P < 0.01 is significantly different from the control group; * P < 0.05 and ** P < 0.01 are significantly different from the LPS group.
Figure 9.
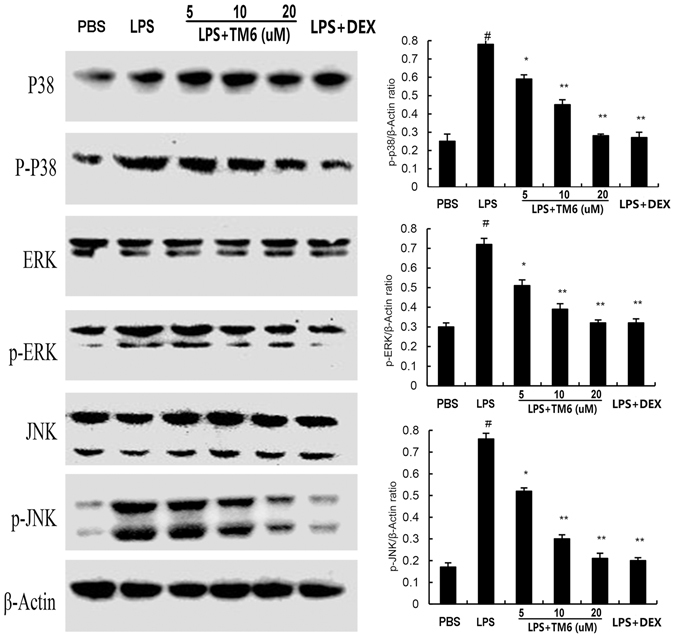
The effect of TM6 on MAPK pathway. Alveolar macrophages were pretreated with TM6 (5, 10 and 20 µM) for 1 hour and then treated with LPS for 1 hour. MAPK protein samples were analyzed by western blot with specific antibodies. β-actin was used as a control. The values presented are the means ± SEM of three independent experiments, # P < 0.01 is significantly different from the control group; * P < 0.05 and ** P < 0.01 are significantly different from the LPS group.
Discussion
Acute lung injury (ALI), a frequent complication of sepsis, can be caused by many factors, such as sepsis, trauma and bacterial pneumonia18. The development of ALI is associated with a large number of inflammatory cells migrating to the lung. Stimulation of these inflammatory cells lead to the releasing of inflammatory mediators. These inflammatory mediators disrupt pulmonary endothelial cells, pulmonary epithelial integrity and finally leads to lung edema19, 20. Corticosteroids, including DEX, triamcinolone acetonide, flunisolide and so on, have been widely used to treat inflammatory diseases like acute lung injury. Previous studies reported that treatment with DEX could improve the pathological process in ALI21. However, clinical application had suggested that these agents had local or systemic side effects. The reports have been suggested that some cell-permeable peptide can inhibited the development of inflammation in vivo and vitro 12, 13. Our previously studies also demonstrated that the peptide derived from TRAM or TIRAP also protected the development of mastitis induced by LPS in mice14, 15. In our study, we synthesized a cell-permeable TIR domain of TRAM-derived decoy peptide (TM6) and tested the protective effect of TM6 on LPS-induced ALI in mice. The results showed that the administration of TM6 markedly decreased lung wet/dry ratios, inflammatory cells production, pro-inflammatory cytokines production, and MPO activity. Moreover, TM6 significantly alleviated LPS-induced lung histopathological changes. In vitro, TM6 also inhibited the production of TNF-α, IL-1β and IL-6 and the expression of NF-κB and MAPK. For further research, we also tested the effect of intranasally administered of TM6 in LPS induced ALI. The results suggested that TM6 also inhibited the production of inflammatory cells and pro-inflammation cytokines in BLAF, as well as MPO activity in lung tissues. But the protective effect is not as effective compared with TM6 administered by intraperitoneal injection (data not shown). These findings suggested that TM6 may have potential value as a future therapeutic treatment to prevent ALI.
It is well known that edema is a typical symptom of ALI. Endothelial injury associated with microvascular leakage, was believed to be the main contributor of lung edema19. To analyze the magnitude of lung edema, the lung W/D ratio was measured. The results showed that administration of TM6 apparently decreased the lung W/D ratio, which suggested it suppressed the accumulation of serous fluid in lung tissues. From analyzing the lung pathology sections with light microscopy, we also found a resolution in pulmonary edema and most of the histopathological changes. MPO, the influx of a marker of neutrophil into the tissue, has been associated with tissue damage in many diseases22. In this study, our results showed that TM6 significantly inhibited LPS-induced MPO activity. These results suggested that TM6 had the ability to protect against LPS-induced ALI.
LPS has been reported to be one of the major factors that induces inflammatory response23, 24. In our model of ALI, LPS significantly increased the production of inflammatory cytokines including TNF-α, IL-1β and IL-6, which has been shown to be involved in the development of ALI25, 26. TNF-α is a primary pro-inflammatory factor, and is produced initially during infection27. IL-1β, another important cytokine, is one of the crucial mediators in ALI. Moreover, IL-1β is involved in the process of LPS-induced endotoxic shock and caused multiple organ failure28. IL-6, another pro-inflammatory cytokine, was considered to be a marker in the LPS-induced inflammatory response27. In this study, we measured the production of the cytokines TNF-α, IL-1β and IL-6, both in vivo and vitro. The results showed that TM6 significantly inhibited LPS-induced inflammatory cytokine production both in vivo and in vitro. These results indicated that TM6 has anti-inflammatory property to inhibit the production of cytokines during LPS-induced ALI.
NF-κB and MAPK, served as the major transcription factors, have been reported to play vital roles in the regulation of inflammatory cytokines production29, 30. Generally, once stimulated, NF-κB activation is initiated by the IκB-α and then NF-κB p65 is translocated to the nucleus. This process may promote the transcription of TNF-α, IL-1β, and IL-631. MAPK family proteins including p38, JNK and ERK, all participate in the production of inflammatory cytokines32. To investigate the anti-inflammatory mechanism of TM6 on LPS-induced ALI, we determined the effects of TM6 on NF-κB and MAPK activation. The results showed that LPS-induced NF-κB and MAPK activation were markedly blocked by administration of TM6.
In conclusion, our study showed that TM6 was able to decrease inflammatory cell infiltration into the lung tissues, lung W/D ratio, MPO activity, inflammatory cytokines production, and lung histopathological changes. Moreover, the protective effect of TM6 may be related to attenuation of inflammatory reactions and inhibition of the activation of NF-κB and MAPK signaling pathways. These findings provide powerful evidence that TM6 may be a potential treatment for preventing ALI induced by LPS.
Materials and Methods
Animals
Adult male Balb/c mice were provided by the Experimental Animal Center of Baiqiuen Medical College of Jilin University (Changchun, China). The procedures were approved by the Jilin University Animal Care and Use Committee. The mice were supplied with food and water in a constant environment with the temperature at approximately 24 ± 1 °C and 40% to 80% relative humidity. All experiments followed the regulations for the Use and Care of Laboratory Animals Manual published by the US National Institutes of Health.
Reagents
TM6 (Purity > 95%) was purchased from and synthesized by SBS Genetech Co. (Beijing, China). LPS (Escherichia coli 055:B5) was obtained from Sigma (St. Louis, MO, USA). Dexamethasone (Purity > 99%) was provided by Changle Pharmaceutical Co. (Xinxiang, Henan, China). ELISA kits were purchased from Biolegend (San Diego, CA, USA). Phospho-IκBα and, phospho-NF-κB p65 antibodies were purchased from Cell Signaling Technology Inc. (Beverly, MA). All other chemicals were reagent grade.
ALI model
Mice were randomly divided into six groups: a control group, an LPS group, TM6 (2.5, 5 and 10 nmol/g) + LPS groups, and a DEX (5 mg/kg) + LPS group. The mice of TM6 (2.5, 5 and 10 nmol/g) + LPS groups were received an intraperitoneal (i.p.) injection of TM6 (2.5, 5 and 10 nmol/g) 1 h before LPS treatment. The mice of DEX + LPS group (5 mg/kg DEX) were received i.p. injections using the same method to avoid confounding procedural differences between groups. An equal volume of PBS, instead of TM6 or DEX, was given to the LPS group and control group. Then, 1 h later, 10 µg of LPS in 50 µl of PBS was introduced intranasally (i.n.) to produce acute lung injury. Control mice received 50 µl of PBS. All mice were humanely sacrificed 7 h after LPS treatment. Then, the bronchoalveolar lavage fluid (BALF) were collected for subsequent analysis.
Histopathological evaluation
Histopathological analysis proceeded via H & E staining and light microscopy. Lung tissues were fully fixed with 10% buffered formalin for approximately 1 week, followed by paraffin-embedding and eosin staining. Histopathological changes of the lungs were examined with a light microscope (Olympus, Japan). The scores of histological changes in the lungs were evaluated as previously described33. Each animal is the average score of the above four criteria, including thickening of alveolar walls and epithelium, the numbers of infiltration cell, increase in peribronchial, and perivascular cuff area. The histological changes were scored 0 to 5.
Lung wet/dry ratio
The lung lobe tissues were extracted and weighted to obtain the wet weight. Then, the tissues were incubated in 80 °C for 48 h to obtain the dry weight. Finally, the lung W/D ratio was calculated.
Inflammatory cell numbers of BALF
BALF was collected 7 h after ALI was induced by LPS. After centrifugation (3000 rpm at 4 °C) for 10 min, the supernatants were collected and used to analyze the production of inflammatory cells.
Pulmonary myeloperoxidase activity analysis
MPO activity reflects accumulation of macrophages and neutrophils in the lung tissues. The MPO activity in the lung tissues was measured by MPO kit according to the manufacturer’s instructions.
Cell culture and treatment
Mice alveolar macrophages were isolated and cultured as previously described34. In brief, the mice were killed and the lungs were lavaged with PBS. The fluids were collected and centrifuged at 1000 g for 10 min at 4 °C. Then, the supernatants were removed and the cells were resuspended with RPMI 1640 media. The cells were plated in well plate and cultured at 37 °C in 5% CO2 incubator for 4 hours. Then the cells were washed once a day for 3 days. The nonadherent cells were removed and attached cells were cultured in RPMI 1640 media with 10% fetal calf serum. The media was changed once every day. In the study, the mice alveolar macrophages were preincubated with or without TM6 (5, 10 and 20 µM) for 1 hour before stimulation with LPS (1 µg/ml).
Cell viability assays
Cell viability was tested by MTT assay. The alveolar macrophages were plated at a density of 4 × 105 cells/ml in 96-well plates at 37 °C. 1 hour later, the cells were treated with TM6 at the dose with 0–40 µM and then cultured for another 1 hour. Next, the cells were treatment with LPS for 18 hour, followed by treatment with MTT (5 mg/ml). The cells were incubated for an additional 4 hours. Then, the supernatants were removed and DMSO (150 µl/well) were added to each well. The OD was read at 570 nm on a microplate reader.
Inflammatory cytokine assays
BALF was collected and centrifuged (3000 rpm, 4 °C) for 20 min. Supernatant samples were used to analyze the production of TNF-α, IL-1β and IL-6 using special ELISA kits according to the instructions provided by the manufacturer.
Western blot analysis
The proteins from the alveolar macrophages were extracted with mammal protein extraction reagent (M-PER) according to the manufacturer’s instructions. Equal amounts of protein (30 µg) were subjected to SDS-PAGE on 10% gels and transferred to PVDF membranes. The membranes were blocked with 3% BSA at room temperature on a rotary shaker for 2 h. Incubation with specific primary antibodies (1:1000) in TBS-T was performed overnight at 4 °C. Following washing, the membranes were incubated with secondary antibody for 1 h. The proteins bands were detected using an enhanced chemiluminescence western blotting detection kit.
Statistical analyses
All values are expressed as the means ± SEM. Differences between groups were analyzed using a one-way ANOVA (Dunnett’s t-test) and a two-tailed Student’s t-test. The results were considered statistically significant at p < 0.05 or p < 0.01.
Acknowledgements
This work was supported by a grant from the National Natural Science Foundation of China (Nos 31472248, 31602122, 30972225, 30771596) and Project 2016114 Supported by Graduate Innovation Fund of Jilin University.
Author Contributions
Xiaoyu Hu contributed to article writing, literature search, results evaluation. Yuan Tian performed histologic analysis and article revision. Shihui Qu and Shumin Li performed the final revision of the article. Yongguo Cao, Wenlong Zhang and Zecai Zhang contributed to literature search and results evaluation. Yunhe Fu performed the final revision of the article and results evaluation. Yunhe Fu and Naisheng Zhang contributed to study design.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Xiaoyu Hu and Yuan Tian contributed equally to this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Naisheng Zhang, Email: zhangns@jlu.edu.cn.
Yunhe Fu, Email: fuyunhesky@sina.com.
References
- 1.Fein AM, Calalang-Colucci MG. Acute lung injury and acute respiratory distress syndrome in sepsis and septic shock. Critical care clinics. 2000;16:289. doi: 10.1016/S0749-0704(05)70111-1. [DOI] [PubMed] [Google Scholar]
- 2.Kodavanti UP, Schladweiler MCJ, Richards JR, Costa DL. Acute lung injury from intratracheal exposure to fugitive residual oil fly ash and its constituent metals in normo- and spontaneously hypertensive rats. Inhalation toxicology. 2001;13:37–54. doi: 10.1080/089583701459056. [DOI] [PubMed] [Google Scholar]
- 3.Epidemiology of transfusion related acute lung injury in Gifit, the French hemovigilance database. A study of the French hemovigilance network. Vox Sang89, 8–8 (2005).
- 4.Brun-Buisson C, et al. Epidemiology and outcome of acute lung injury in European intensive care units - Results from the ALIVE study (vol 30, pg 51, 2003) Intensive care medicine. 2004;30:524–524. doi: 10.1007/s00134-003-2136-x. [DOI] [PubMed] [Google Scholar]
- 5.Niu XF, et al. Protective effects of Isofraxidin against lipopolysaccharide-induced acute lung injury in mice. Int Immunopharmacol. 2015;24:432–439. doi: 10.1016/j.intimp.2014.12.041. [DOI] [PubMed] [Google Scholar]
- 6.Matthay MA, Ware LB, Zimmerman GA. The acute respiratory distress syndrome. J Clin Invest. 2012;122:2731–2740. doi: 10.1172/JCI60331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Park EJ, et al. Dehydrocostuslactone inhibits LPS-induced inflammation by p38MAPK-dependent induction of hemeoxygenase-1 in vitro and improves survival of mice in CLP-induced sepsis in vivo. Int Immunopharmacol. 2014;22:332–340. doi: 10.1016/j.intimp.2014.07.012. [DOI] [PubMed] [Google Scholar]
- 8.Wu GT, et al. The total alkaloids of Aconitum tanguticum protect against lipopolysaccharide-induced acute lung injury in rats. J Ethnopharmacol. 2014;155:1483–1491. doi: 10.1016/j.jep.2014.07.041. [DOI] [PubMed] [Google Scholar]
- 9.Li W, et al. Viola yedoensis Liposoluble Fraction Ameliorates Lipopolysaccharide-Induced Acute Lung Injury in Mice. Am J Chinese Med. 2012;40:1007–1018. doi: 10.1142/S0192415X12500747. [DOI] [PubMed] [Google Scholar]
- 10.Khan S, et al. Molecular mechanism of capillarisin-mediated inhibition of MyD88/TIRAP inflammatory signaling in in vitro and in vivo experimental models. J Ethnopharmacol. 2013;145:626–637. doi: 10.1016/j.jep.2012.12.001. [DOI] [PubMed] [Google Scholar]
- 11.Bhaskaran N, Shukla S, Srivastava JK, Gupta S. Chamomile: An anti-inflammatory agent inhibits inducible nitric oxide synthase expression by blocking RelA/p65 activity. Int J Mol Med. 2010;26:935–940. doi: 10.3892/ijmm_00000545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dunne A, Ejdeback M, Ludidi PL, O’Neill LA, Gay NJ. Structural complementarity of Toll/interleukin-1 receptor domains in Toll-like receptors and the adaptors Mal and MyD88. J Biol Chem. 2003;278:41443–41451. doi: 10.1074/jbc.M301742200. [DOI] [PubMed] [Google Scholar]
- 13.Toshchakov VY, Szmacinski H, Couture LA, Lakowicz JR, Vogel SN. Targeting TLR4 signaling by TLR4 Toll/IL-1 receptor domain-derived decoy peptides: identification of the TLR4 Toll/IL-1 receptor domain dimerization interface. J Immunol. 2011;186:4819–4827. doi: 10.4049/jimmunol.1002424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hu X, et al. TRAM-Derived Decoy Peptides inhibits the inflammatory response in mouse mammary epithelial cells and a mastitis model in mice. European journal of pharmacology. 2015;764:607–612. doi: 10.1016/j.ejphar.2015.06.022. [DOI] [PubMed] [Google Scholar]
- 15.Hu X, et al. The anti-inflammatory effect of TR6 on LPS-induced mastitis in mice. Int Immunopharmacol. 2016;30:150–156. doi: 10.1016/j.intimp.2015.12.003. [DOI] [PubMed] [Google Scholar]
- 16.Pfalzgraff A, et al. Synthetic antimicrobial and LPS-neutralising peptides suppress inflammatory and immune responses in skin cells and promote keratinocyte migration. Scientific reports. 2016;6:31577. doi: 10.1038/srep31577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Piao W, Vogel SN, Toshchakov VY. Inhibition of TLR4 signaling by TRAM-derived decoy peptides in vitro and in vivo. J Immunol. 2013;190:2263–2272. doi: 10.4049/jimmunol.1202703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hammer J. Acute lung injury: pathophysiology, assessment and current therapy. Paediatric respiratory reviews. 2001;2:10–21. doi: 10.1053/prrv.2000.0096. [DOI] [PubMed] [Google Scholar]
- 19.Morita, S., Takimoto, T., Kawahara, K., Nishi, K. & lino, M. Clinical course of acute chemical lung injury caused by 3-chloropentafluoropene. BMJ case reports2013, doi:10.1136/bcr-2013-201089 (2013). [DOI] [PMC free article] [PubMed]
- 20.Martin GS, Bernard GR. Acute respiratory distress syndrome: innovative therapies. Seminars in respiratory and critical care medicine. 2001;22:293–306. doi: 10.1055/s-2001-15786. [DOI] [PubMed] [Google Scholar]
- 21.Huang B, Wang DX, Deng W. Protective effects of dexamethasone on early acute lung injury induced by oleic acid in rats. International journal of clinical and experimental medicine. 2014;7:4698–4709. [PMC free article] [PubMed] [Google Scholar]
- 22.Par A. [Clinical Implications of Inflammation and Immunity in Acute and Chronic Liver Disease: Advances in Diagnosis, Treatment and Clinical Practice. AASLD Liver Meeting, Boston, November 7–11, 2014] Orvosi hetilap. 2015;156:869–872. doi: 10.1556/650.2015.21M. [DOI] [PubMed] [Google Scholar]
- 23.Hu JJ, Zhang DD, Chen JJ, Chen CS, Li YP. [Effect of pretreatment with puerarin on activation of LPS-induced RAW264. 7 cells] Zhongguo Zhong yao za zhi=Zhongguo zhongyao zazhi=China journal of Chinese materia medica. 2012;37:3112–3116. [PubMed] [Google Scholar]
- 24.Wang YR, Yang F, Liu J, Zhou Y. [Effect of sophoridine and TLR4/MD-2 blocking agent on pathway of LPS-induced RAW264. 7 macrophage TLR4-NF-kappaB-TNF-alpha] Zhongguo Zhong yao za zhi=Zhongguo zhongyao zazhi=China journal of Chinese materia medica. 2012;37:3107–3111. [PubMed] [Google Scholar]
- 25.Zhao DM, et al. Heparin Rescues Sepsis-Associated Acute Lung Injury and Lethality Through the Suppression of Inflammatory Responses. Inflammation. 2012;35:1825–1832. doi: 10.1007/s10753-012-9503-0. [DOI] [PubMed] [Google Scholar]
- 26.Fu, P. K., Wu, C. L., Tsai, T. H. & Hsieh, C. L. Anti-Inflammatory and Anticoagulative Effects of Paeonol on LPS-Induced Acute Lung Injury in Rats. Evid-Based Compl Alt, doi:Artn 83751310.1155/2012/837513 (2012). [DOI] [PMC free article] [PubMed]
- 27.Yang LL, Ji XP, Liu Z. Effects of hypercapnia on nuclear factor-kappaB and tumor necrosis factor-alpha in acute lung injury models. Chinese medical journal. 2004;117:1859–1861. [PubMed] [Google Scholar]
- 28.Wang S, et al. Deferoxamine attenuates lipopolysaccharide-induced inflammatory responses and protects against endotoxic shock in mice. Biochem Biophys Res Commun. 2015;465:305–311. doi: 10.1016/j.bbrc.2015.08.032. [DOI] [PubMed] [Google Scholar]
- 29.Qian Z, et al. Mulberry fruit prevents LPS-induced NF-kappaB/pERK/MAPK signals in macrophages and suppresses acute colitis and colorectal tumorigenesis in mice. Scientific reports. 2015;5:17348. doi: 10.1038/srep17348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yeom M, et al. Xanthii fructus inhibits inflammatory responses in LPS-stimulated RAW 264.7 macrophages through suppressing NF-kappaB and JNK/p38 MAPK. J Ethnopharmacol. 2015;176:394–401. doi: 10.1016/j.jep.2015.11.020. [DOI] [PubMed] [Google Scholar]
- 31.Deshpande R, Khalili H, Pergolizzi RG, Michael SD, Chang MD. Estradiol down-regulates LPS-induced cytokine production and NFkB activation in murine macrophages. American journal of reproductive immunology. 1997;38:46–54. doi: 10.1111/j.1600-0897.1997.tb00275.x. [DOI] [PubMed] [Google Scholar]
- 32.Hu HX, et al. GuaLou GuiZhi decoction inhibits LPS-induced microglial cell motility through the MAPK signaling pathway. Int J Mol Med. 2013;32:1281–1286. doi: 10.3892/ijmm.2013.1522. [DOI] [PubMed] [Google Scholar]
- 33.Bao S, et al. Ginsenoside Rg1 improves lipopolysaccharide-induced acute lung injury by inhibiting inflammatory responses and modulating infiltration of M2 macrophages. Int Immunopharmacol. 2015;28:429–434. doi: 10.1016/j.intimp.2015.06.022. [DOI] [PubMed] [Google Scholar]
- 34.Fu YH, et al. Cyanidin-3-O-beta-glucoside ameliorates lipopolysaccharide-induced acute lung injury by reducing TLR4 recruitment into lipid rafts. Biochemical pharmacology. 2014;90:126–134. doi: 10.1016/j.bcp.2014.05.004. [DOI] [PubMed] [Google Scholar]


