Abstract
Genome editing has emerged as a technology with a potential to revolutionize plant breeding. In this study, we report on generating, in less than ten months, Tomelo, a non-transgenic tomato variety resistant to the powdery mildew fungal pathogen using the CRISPR/Cas9 technology. We used whole-genome sequencing to show that Tomelo does not carry any foreign DNA sequences but only carries a deletion that is indistinguishable from naturally occurring mutations. We also present evidence for CRISPR/Cas9 being a highly precise tool, as we did not detect off-target mutations in Tomelo. Using our pipeline, mutations can be readily introduced into elite or locally adapted tomato varieties in less than a year with relatively minimal effort and investment.
Introduction
As the world population grows, there is an increasing demand for food. This demand needs to be addressed in a sustainable manner e.g. by creating new crop varieties with valuable traits, such as higher yield, enhanced disease resistance, improved salt and drought tolerance. Traditional plant breeding has been used to generate new crop varieties for decades, but new technologies, such as genome editing, have the potential to generate improved varieties faster and at a lower cost, by precise introduction of favorable alleles into many different, locally adapted elite varieties. Genome editing is achieved using sequence-specific nucleases (SSNs) and results in chromosomal changes, such as nucleotide deletions, insertions or substitutions at specified genetic loci. SSNs include zinc finger nucleases (ZFNs), TAL effector nucleases (TALENs) and, most recently, clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 9 (Cas9) system1. Because certain genetic changes (e.g. nucleotide deletions) introduced using SSNs are indistinguishable from natural mutations, the resulting crop varieties are different from transgenics, aka genetically modified organisms (GMO). In the US, in several cases, genome edited crops have not been regulated, and a decision on regulation of these technologies is forthcoming in Europe2–4.
In this study we report on generating a tomato variety resistant to the powdery mildew fungal pathogen Oidium neolycopersici using the CRISPR/Cas9 technology, which is based on the Cas9 DNA nuclease guided to a specific DNA target by a single guide RNA (sgRNA). Wild-type alleles of MILDEW RESISTANT LOCUS O (Mlo), which encodes a membrane-associated protein with seven transmembrane domains and is conserved throughout monocots and dicots, confer susceptibility to fungi causing the powdery mildew disease5. Therefore, homozygous loss-of-function mutations (mlo) result in resistance to powdery mildew. In tomato, there are sixteen Mlo genes, SlMlo1 to SlMlo16 with SlMlo1 being the major contributor to powdery mildew susceptibility6. Natural non-transgenic loss-of-function slmlo1 mutants are available in tomato, but introgression of such mutations into an elite variety is a lengthy and laborious process. We aimed at determining the speed and efficiency at which we could generate a transgene-free genetically edited slmlo1 tomato variety using the CRISPR/Cas9 system. We targeted the SlMlo1 locus using the double sgRNA strategy as previously described7. The two selected targets within SlMlo1 were positioned on opposite DNA strands and spaced 42 bp apart (Fig. 1a). Ten primary transformants (T0) carrying the T-DNA expressing Cas9 and sgRNAs were analysed for altered electrophoretic mobility of PCR amplicons as an indication of modifications in SlMlo1. Eight out of ten tested T0 transformants showed a mobility shift indicating the presence of mutations (Fig. 1b). We selected three transformants (2, 8 and 10) that showed a band shift consistent with the expected deletion of 48 bp when Cas9 cuts 3 bp away from the PAM motif within the target sequence8 (Fig. 1c). The PCR amplicons were cloned and individual clones sequenced (Fig. 1d). All three transformants that showed the expected band shift proved to carry homozygous (transformants 2 and 8) or biallelic (transformant 10) mutations at the SlMlo1 locus.
Figure 1.
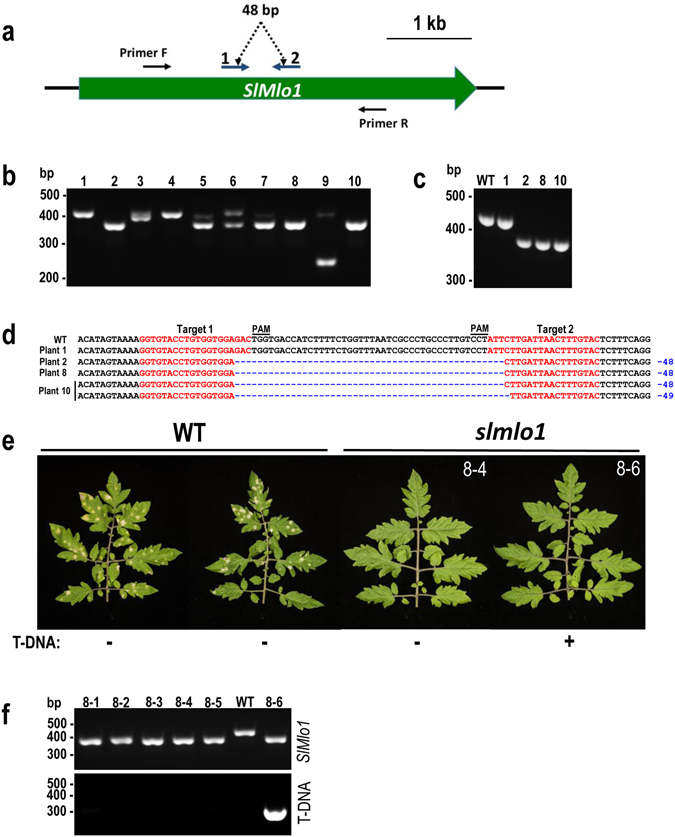
Generating non-transgenic slmlo1 tomato lines resistant to powdery mildew. (a) The SlMlo1 locus was targeted by two sgRNAs; (b) T0 tomato transformants were tested for the presence of deletions using the PCR band shift assay; (c) Selected T0 transformants genotyped using the PCR band shift assay alongside wild type (WT); (d) SlMlo1 sequencing reads from selected T0 transformants; (e) Leaves of tomato plants inoculated with Oidium neolycopersici (5 weeks post inoculation); (f) PCR genotyping of the T1 generation for the presence T-DNA and the slmlo1 mutation. The agarose gels presented in panels (b and c) were cropped.
To generate non-transgenic slmlo1 tomato lines, we segregated away the T-DNA by selfing T0 transformant 8 and cultivating next generation (T1) plants. In the progeny, we identified five slmlo1 T-DNA-free individuals (Fig. 1f). Disease resistance assays with the powdery mildew fungus Oidium neolycopersici revealed that all the slmlo1 mutant plants were fully resistant to the pathogen, whereas all tested wild-type plants were susceptible (Fig. 1e). The slmlo1 mutant plants were not only morphologically similar to the wild type (Supplementary Fig. 1), but they also produced harvested fruit weight similar to the wild type (Supplementary Fig. 2).
To verify that the slmlo1 mutants are indeed free from any T-DNA or transformation vector sequences, and to confirm the presence of a homozygous deletion in SlMlo1, we Illumina sequenced genomic DNA from the five T1 T-DNA free slmlo1 segregants (8-1, 8-2, 8-3, 8-4 and 8-5), one slmlo1 T1 segregant carrying the T-DNA (8-6), as well as the wild type to at least 28x genome coverage (Supplementary Table 1). We first mapped the reads to the complete vector used for T-DNA transformation, allowing for 4% mismatches. In the slmlo1 8-6 plant, in which we had confirmed the presence of T-DNA sequences by PCR, 1,757 reads matching the T-DNA or transformation vector backbone were detected. In contrast, no reads matching the T-DNA or vector were detected in mutants slmlo1 8-2 and 8-4, while only 1 or 2 reads were detected in lines 8-1, 8-3, and 8-5, as well as the wild type (Fig. 2a,b). These reads are most likely the result of low levels of carry-over contamination between runs due to the extreme sensitivity of the DNA sequencing method. We conclude that mutant lines slmlo1 8-2 and 8-4 are unambiguously free of transgenic sequences. The mutants were confirmed to carry the slmlo1 48 bp deletion in a homozygous configuration and to be clearly different from the parental line in this locus based on the whole genome shotgun sequence analysis (Fig. 2c).
Figure 2.
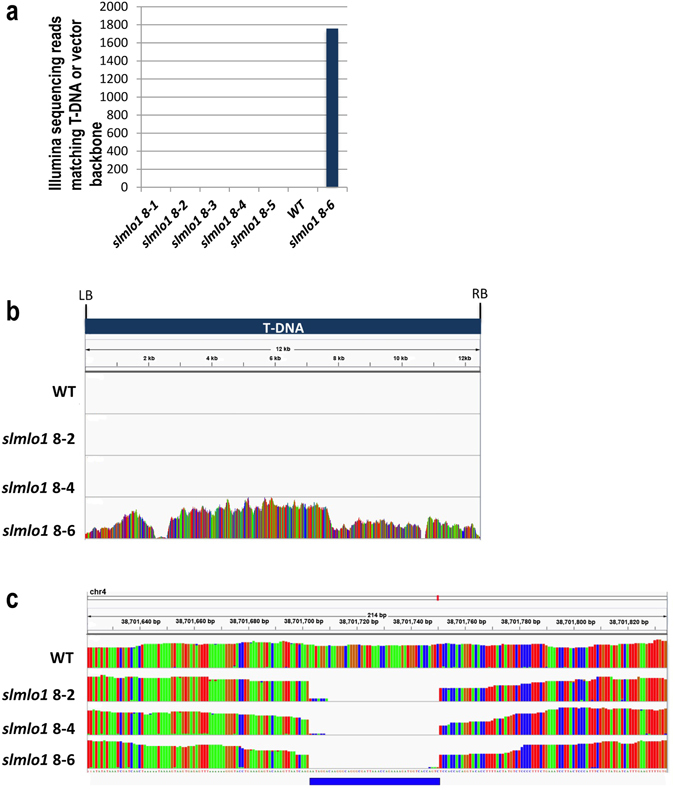
Illumina sequencing data. (a) Quantification of Illumina sequencing reads matching the T-DNA or vector backbone in wild type and slmlo1 T1 progeny lines; (b) Coverage of the T-DNA by Illumina reads; (c) Coverage of the SlMlo1 locus by Illumina reads.
We also evaluated the extent to which the genomes of slmlo1 mutants suffered from off-target mutations caused by the CRISPR/Cas9 system. We generated a list of 145 potential off-targets for both CRISPR/Cas targets with up to 4 mismatches using the Cas-OFFinder online tool (http://www.rgenome.net/cas-offinder/) (Supplementary Table 2). We performed the off-target analysis by mapping the Illumina reads from wild-type and slmlo1 8-2 genomes to the tomato reference genome. Out of 145 putative off-targets, 140 were covered by Illumina reads in both the wild type and slmlo1 8-2 mutant (Supplementary Table 2). The results of the DNA base calling analyses are presented in Supplementary Tables 3 and 4. Side-by-side comparison of DNA bases called in the wild type and slmlo1 8-2 revealed that 12 out of 39 differences (31%) were confined to the targeted SlMlo1 locus on chromosome 4 (Supplementary Tables 5 and 6). The remaining 27 out of 39 differences (69%) that we observed in genomic regions outside of the targeted SlMlo1 locus did not indicate off-target effects (Supplementary Tables 5 and 6). For example, a region of several Mb on chromosome 9 was poorly covered by reads in both the wild type and slmlo1 8-2, with several potential off-targets lacking reads from both the wild type and slmlo1 8-2, but one off-target, at position 38,542,554, was covered by just one read in wild type and no reads in slmlo1 8-2 (Supplementary Table 2 and Supplementary Fig. 3). We note that this sequence carries four mismatches relative to target 2, with two of the mismatches close to the PAM motif, which are expected to dramatically decrease the ability of CRISPR/Cas9 to recognise it8. Therefore, it is likely to be by chance that the off-target was covered in wild type but not in the slmlo1 8-2 line. We also detected heterozygous SNPs at two positions in the slmlo1 8-2 mutant (chr8, positions 21,452,901 and 21,452,908), where the wild type appeared to be homozygous (Supplementary Tables 5 and 6). In addition, we detected heterozygous SNPs at two positions in wild type (chr1, position 43,886,749; chr2, position 2,894,674), where the slmlo1 8-2 mutant appears to be homozygous (Supplementary Tables 5 and 6). Visual examination of the sequence reads at these sites revealed that homozygous SNP calls at all four positions were an artifact resulting from the 0.25 threshold for calling heterozygous positions with both wild type and slmlo1 8-2 showing heterozygous calls when the threshold was lowered to 0.16 (Supplementary Table 7, and Supplementary Figs 4 and 5). Since our inbred wild-type strain is more likely to be homozygous, these heterozygous calls likely resulted from mis-mapping of sequences related to the target locus that are present in the wild type GCR758 accession9, a derivative of the Moneymaker cultivar, and its descendants, but not in the Heinz 1706 strain from which the reference genome sequence was assembled. In conclusion, we did not find credible evidence for mutations at potential off-target positions within the genome of the slmlo1 8-2 line. Our results suggest that CRISPR/Cas9 can be a highly precise genome editing tool when applied in tomato consistent with other recent reports in plants10, 11.
We named the powdery mildew resistant slmlo1 tomato variety ‘Tomelo’. We have unambiguously demonstrated by whole-genome sequencing that Tomelo is non-transgenic, i.e. it does not carry any foreign DNA sequences. Importantly, the 48 bp deletion at SlMlo1 in Tomelo is indistinguishable from naturally occurring mutations. We generated Tomelo in only 9.5 months from the DNA transformation step to recovery of second generation transgene-free segregants (Supplementary Fig. 6). Therefore, using our pipeline, the slmlo1 mutation could be readily introduced into elite or locally adapted varieties in less than a year with relatively little effort or investment. Although the status of genetically edited crop varieties like Tomelo is still debated by regulatory authorities throughout the world, some countries, such as the US, consider transgene-free genetically edited crops as non-GMO2, 3. We hope that plant varieties, such as Tomelo, are adopted world-wide with similar regulatory burden as traditionally bred varieties to meet the food demands of the growing world’s population and promote competitiveness of the agrobiotech sector by reducing chemical input.
Methods
Plasmid construction
The plasmid pAGM4723::Cas9_sgRNA1_sgRNA2 (Supplementary sequence file 1) was assembled as previously described7. Primers Mlo1_target1_f and sgRNAr were used to amplify sgRNA1, and primers Mlo1_target1_f and sgRNAr were used to amplify sgRNA2 (Supplementary Table 8).
Plant transformation
GCR7589, a derivative of tomato cultivar Moneymaker, was transformed with the pAGM4723::Cas9_sgRNA1_sgRNA2 construct as previously described12. T0 transgenic plants were selected in the medium with kanamycin and then transferred into soil.
Plant DNA extraction
The DNA was extracted from the plant tissue using 500 ul of CTAB buffer (2% CTAB, 1.42 M NaCl, 20 mM EDTA and 100 mM Tris pH 8) for 30 min at 60 °C. Then 300 ul of chlorophorm was added. The mix was vortexed and then spun for 20 min at 4,500 rpm. The genomic DNA was precipitated from the supernatant with 0.7 volumes of isopropanol at −20 °C overnight. The mix was centrifuged for 15 min at 12,000 rpm and the pellet was then washed with 1 ml of ice cold 70% ethanol. The pellet was dried and resuspended in water.
Detection of Cas9-induced deletions and T-DNA in plant genomic DNA using PCR genotyping
The SlMlo1 locus (Solyc04g049090) was PCR amplified using Phusion DNA polymerase (New England Biolabs) across targets 1 and 2 using SlMloseq3f and SlMloseq1r primers. The PCR products were run on a 3% agarose gel, extracted from the gel using the Qiagen gel extraction kit and cloned into pCR-Blunt II-TOPO vector. Eight individual clones were sequenced using M13F and M13R primers. The T-DNA was detected by PCR amplification using primers Cas9_2f and Cas9_2r.
Whole genome Illumina sequencing
To avoid amplification artifacts introduced during library preparation for Illumina sequencing all libraries were prepared following the TruSeq DNA PCR-free library preparation guide (Cat. No: FC-121-9006DOC) and using TruSeq DNA PCR-Free LT kit Set A (Cat. No: FC-121-3001). A starting amount of 1 μg DNA was sheared to an average insert size 350 bp by AFA (S2, Covaris) followed by end repair, A-tailing and adapter ligation. We obtained sufficient paired-end DNA for validation and sequencing. The quality of the libraries was checked on a Bioanalyzer (Agilent Technologies) using a High Sensitivity DNA chip and 1 µl of diluted DNA library. Quantification was done by Qubit (Invitrogen) and qPCR.
DNA Sequencing was performed on HiSEQ3000 sequencing system (Illumina), equipped with on-instrument HiSeq control software (HCS) version 3.3.20 and Real time analysis (RTA) version2.5.2. Cluster generation was performed on a cBot (recipe: HiSeq_3000_4000_HB_Exclusion_Amp_v1.0, Illumina) using a patterned flow cell and reagents from PE Cluster Kit (Cat. No: PE-410-1001, Illumina) according to the manufacturer’s instructions. The libraries were sequenced in 2 lanes paired end with 151 bp read length and 8 bp index read using 300cycle HiSEQ3000 SBS kits (Cat No: FC-410-1003). The library was diluted with Resuspension Buffer (Illumina) to 2 nM before clustering. The final DNA concentration on the flowcell was 200 pM which resulted in 2 × 329 M reads (past filter) in the first lane and 2 × 319 M reads in the second lane.
Off-target mutation analysis
Each tomato line was analyzed separately with the SHORE pipeline13, using BWA aln (v0.6.2)14 with option “-n 0.05” to map the reads to the Solanum lycopersicum (version SL2.40.26; ftp://ftp.ensemblgenomes.org/pub/release-26/plants/fasta/solanum_lycopersicum/dna/) reference genome. As a result, .bam files for slmlo1 8-2 and WT have been generated.
We produced a list of 145 potential off-targets for both CRISPR/Cas targets with up to 4 mismatches using the Cas-OFFinder online tool (http://www.rgenome.net/cas-offinder/) (Supplementary Table 2). For each predicted off-target, we calculated nucleotide frequencies based on the coverage of all 23 nucleotide positions (Supplementary Tables 3 and 4). A SNP was called if the rate of a differential nucleotide, as compared to the reference genome, equaled 0.25 or more (Supplementary Table 5).
Pathotest with Oidium neolycopersici
The tomato lines were infected with airborne spores of Oidium neolycopersici propagated on wild type GCR758 tomato plants in an isolated growth room. The experiment was done twice with similar results.
Analysis of the fruit harvest from slmlo1 vs wild type tomato plants
The fruits were collected on nine different days and plants that produced fruits on more than one day were included into the analysis. The statistics was done using the algorithm described15.
Electronic supplementary material
Acknowledgements
We thank Levente Kiss for the Oidium neolycopersici isolate, and Matthew Smoker for tomato transformation and technical advice. This work was supported by the Gatsby Charitable Foundation, the European Research Council (ERC), and the Biotechnology and Biological Sciences Research Council (BBSRC).
Author Contributions
V.N. generated slmlo1 tomato mutant lines and performed phenotypic characterisation. C.W. and J.W. analysed the Illumina data. C.L. generated raw Illumina data. V.N., D.W. and S.K. contributed to the design of the study and wrote the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-00578-x
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Detlef Weigel, Email: detlef.weigel@tuebingen.mpg.de.
Sophien Kamoun, Email: sophien.kamoun@tsl.ac.uk.
References
- 1.Schiml S, Puchta H. Revolutionizing plant biology: multiple ways of genome engineering by CRISPR/Cas. Plant methods. 2016;12:8. doi: 10.1186/s13007-016-0103-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Waltz E. CRISPR-edited crops free to enter market, skip regulation. Nat Biotechnol. 2016;34:582. doi: 10.1038/nbt0616-582. [DOI] [PubMed] [Google Scholar]
- 3.Waltz E. Gene-edited CRISPR mushroom escapes US regulation. Nature. 2016;532:293. doi: 10.1038/nature.2016.19754. [DOI] [PubMed] [Google Scholar]
- 4.Huang S, Weigel D, Beachy RN, Li J. A proposed regulatory framework for genome-edited crops. Nature genetics. 2016;48:109–111. doi: 10.1038/ng.3484. [DOI] [PubMed] [Google Scholar]
- 5.Acevedo-Garcia J, Kusch S, Panstruga R. Magical mystery tour: MLO proteins in plant immunity and beyond. The New phytologist. 2014;204:273–281. doi: 10.1111/nph.12889. [DOI] [PubMed] [Google Scholar]
- 6.Zheng Z, et al. Genome-Wide Study of the Tomato SlMLO Gene Family and Its Functional Characterization in Response to the Powdery Mildew Fungus Oidium neolycopersici. Frontiers in plant science. 2016;7:380. doi: 10.3389/fpls.2016.00380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brooks C, Nekrasov V, Lippman ZB, Van Eck J. Efficient Gene Editing in Tomato in the First Generation Using the Clustered Regularly Interspaced Short Palindromic Repeats/CRISPR-Associated9 System. Plant physiology. 2014;166:1292–1297. doi: 10.1104/pp.114.247577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jinek M, et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012;337:816–821. doi: 10.1126/science.1225829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Balint-Kurti PJ, Jones DA, Jones JD. Integration of the classical and RFLP linkage maps of the short arm of tomato chromosome 1. Theor Appl Genet. 1995;90:17–26. doi: 10.1007/BF00220991. [DOI] [PubMed] [Google Scholar]
- 10.Peterson BA, et al. Genome-Wide Assessment of Efficiency and Specificity in CRISPR/Cas9 Mediated Multiple Site Targeting in Arabidopsis. PLoS One. 2016;11:e0162169. doi: 10.1371/journal.pone.0162169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pan C, et al. CRISPR/Cas9-mediated efficient and heritable targeted mutagenesis in tomato plants in the first and later generations. Scientific reports. 2016;6:24765. doi: 10.1038/srep24765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fillatti JJ, Kiser J, Rose R, Comai L. Efficient Transfer of a Glyphosate Tolerance Gene into Tomato Using a Binary Agrobacterium-Tumefaciens Vector. Bio-Technol. 1987;5:726–730. doi: 10.1038/nbt0787-726. [DOI] [Google Scholar]
- 13.Ossowski S, et al. Sequencing of natural strains of Arabidopsis thaliana with short reads. Genome Res. 2008;18:2024–2033. doi: 10.1101/gr.080200.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009;25:1754–1760. doi: 10.1093/bioinformatics/btp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Petre B, et al. Heterologous Expression Screens in Nicotiana benthamiana Identify a Candidate Effector of the Wheat Yellow Rust Pathogen that Associates with Processing Bodies. PLoS One. 2016;11:e0149035. doi: 10.1371/journal.pone.0149035. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


