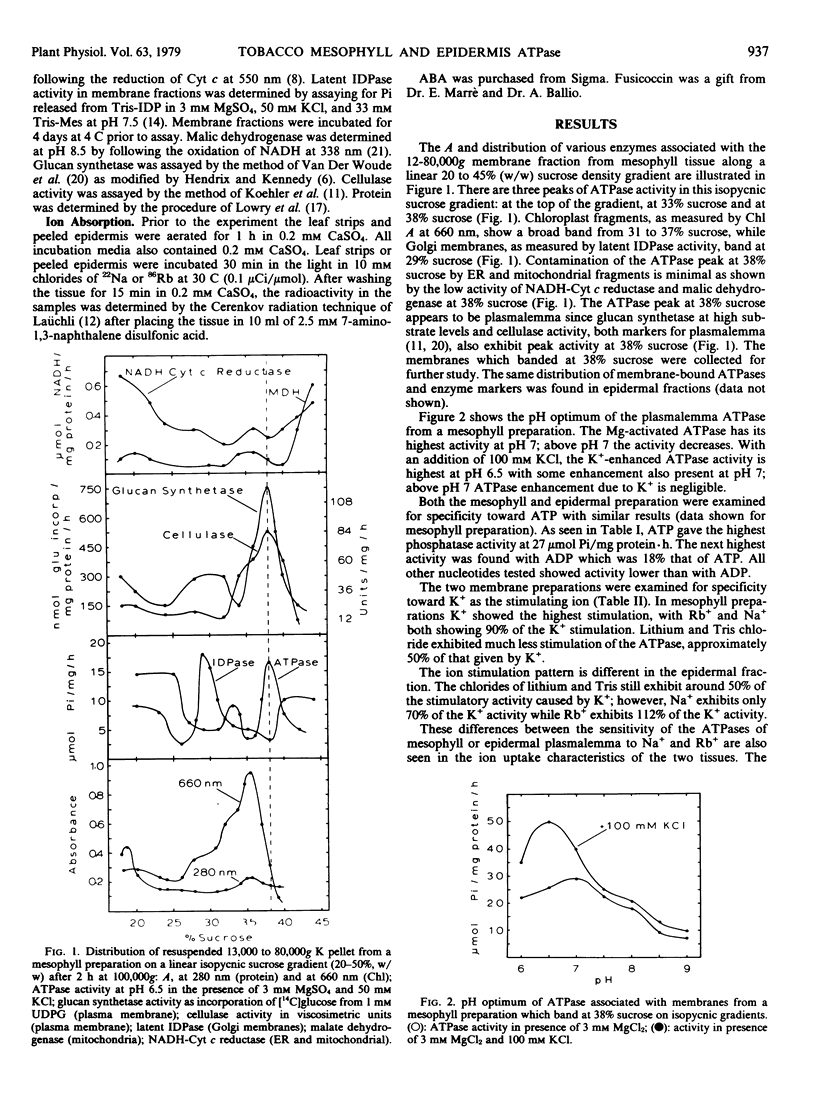
Abstract
An ATPase preparation, presumed to be associated with plasma membrane due to the coincidence on isopycnic gradients of cellulase and β-glucan synthetase at high substrate, has been isolated from the epidermal and mesophyll of tobacco leaf. The ATPase from both tissues was found to prefer ATP over other nucleotides. The pH optimum was 7.0 in the presence of 3 millimolar MgCl2 and pH 6.5 in the presence of 3 millimolar MgCl2 and 100 millimolar KCl. Monovalent ion stimulation patterns of the ATPases from these tissues were found to differ and ion accumulation patterns in these tissues reflect this difference: mesophyll accumulated roughly equal amounts of Na+ and Rb+ and its plasma membrane ATPase is also equally stimulated by these ions; on the other hand, epidermal ATPase preparations showed a stronger stimulation by Rb+ than Na+ and this tissue was found to accumulate Rb+ in preference to Na+. Abscisic acid and fusicoccin affected both ATPase activity and ion uptake, the former inhibiting and the latter stimulating these parameters. These data support the hypothesis that the epidermal plasmalemma ATPase is involved in stomatal opening.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Fisher J. D., Hansen D., Hodges T. K. Correlation between ion fluxes and ion-stimulated adenosine triphosphatase activity of plant roots. Plant Physiol. 1970 Dec;46(6):812–814. doi: 10.1104/pp.46.6.812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hendrix D. L., Kennedy R. M. Adenosine triphosphatase from soybean callus and root cells. Plant Physiol. 1977 Feb;59(2):264–267. doi: 10.1104/pp.59.2.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodges T. K., Leonard R. T. Purification of a plasma membrane-bound adenosine triphosphatase from plant roots. Methods Enzymol. 1974;32:392–406. doi: 10.1016/0076-6879(74)32039-3. [DOI] [PubMed] [Google Scholar]
- Humble G. D., Hsiao T. C. Specific requirement of potassium for light-activated opening of stomata in epidermal strips. Plant Physiol. 1969 Feb;44(2):230–234. doi: 10.1104/pp.44.2.230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacoby B. Light sensitivity of na, rb, and k absorption by different tissues of bean leaves. Plant Physiol. 1975 Jun;55(6):978–981. doi: 10.1104/pp.55.6.978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koehler D. E., Leonard R. T., Vanderwoude W. J., Linkins A. E., Lewis L. N. Association of latent cellulase activity with plasma membranes from kidney bean abscission zones. Plant Physiol. 1976 Sep;58(3):324–330. doi: 10.1104/pp.58.3.324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Leonard R. T., Hansen D., Hodges T. K. Membrane-bound Adenosine Triphosphatase Activities of Oat Roots. Plant Physiol. 1973 Apr;51(4):749–754. doi: 10.1104/pp.51.4.749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leonard R. T., Hotchkiss C. W. Cation-stimulated Adenosine Triphosphatase Activity and Cation Transport in Corn Roots. Plant Physiol. 1976 Sep;58(3):331–335. doi: 10.1104/pp.58.3.331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leonard R. T., Hotchkiss C. W. Plasma Membrane-associated Adenosine Triphosphatase Activity of Isolated Cortex and Stele from Corn Roots. Plant Physiol. 1978 Feb;61(2):175–179. doi: 10.1104/pp.61.2.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Läuchli A. Radioassay for beta-emitters in biological materials using Cerenkov radiation. Int J Appl Radiat Isot. 1969 Apr;20(4):265–270. doi: 10.1016/0020-708x(69)90054-4. [DOI] [PubMed] [Google Scholar]
- Van Der Woude W. J., Lembi C. A., Morré D. J. beta-Glucan Synthetases of Plasma Membrane and Golgi Apparatus from Onion Stem. Plant Physiol. 1974 Sep;54(3):333–340. doi: 10.1104/pp.54.3.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zschoche W. C., Ting I. P. Malate Dehydrogenases of Pisum sativum: Tissue Distribution and Properties of the Particulate Forms. Plant Physiol. 1973 Jun;51(6):1076–1081. doi: 10.1104/pp.51.6.1076. [DOI] [PMC free article] [PubMed] [Google Scholar]



