Abstract
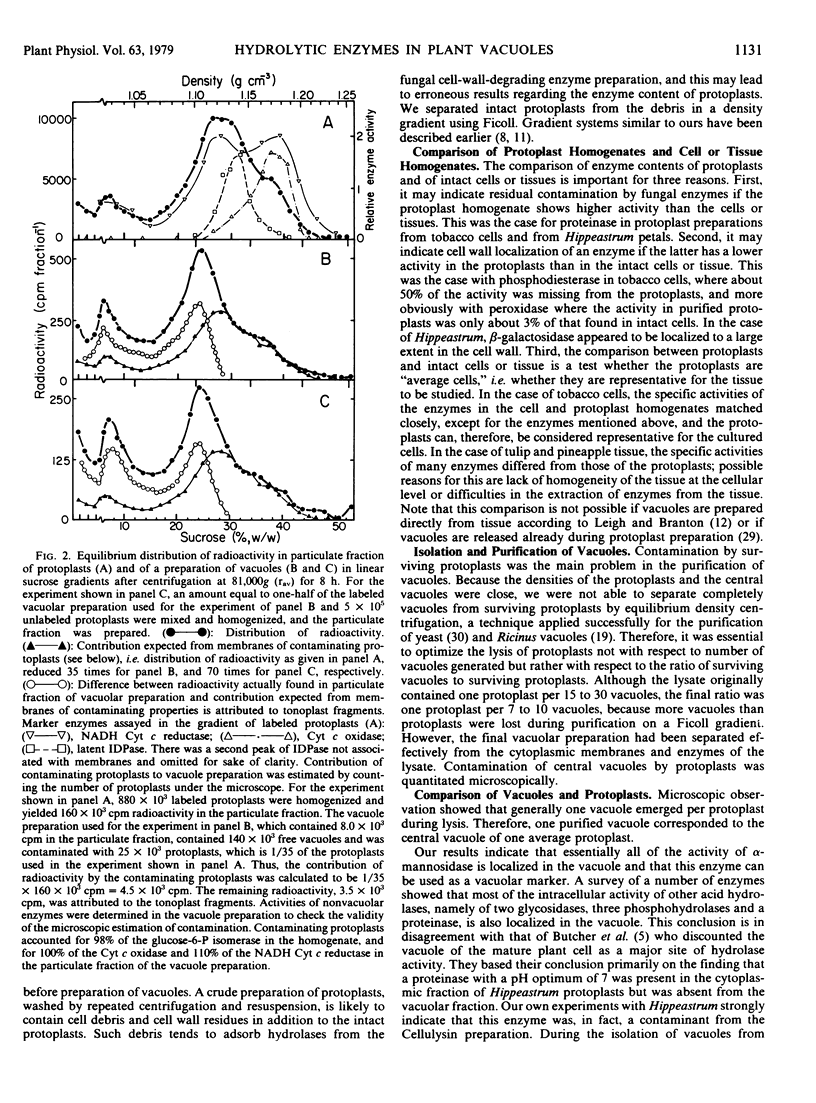
The hydrolase content of vacuoles isolated from protoplasts of suspension-cultured tobacco cells, of tulip petals, and of pineapple leaves, and the sedimentation behavior of tobacco tonoplasts were studied. Three precautions were found to be important for the analysis of vacuolar hydrolases and of the tonoplast. (a) Purification of protoplasts in a Ficoll gradient was necessary to remove cell debris which contained contaminating hydrolases adsorbed from the fungal cell-wall-degrading enzyme preparation. (b) Hydrolase activities in the homogenates of the intact cells or the tissue used and of the purified protoplasts had to be compared to verify the absence of contaminating hydrolases in the protoplast preparation. (c) Vacuoles obtained from the protoplasts by an osmotic shock had to be purified from the lysate in a Ficoll gradient. Since the density of the central vacuole approximates that of the protoplasts, about a 10% contamination of the vacuolar preparation by surviving protoplasts could not be eliminated and had to be taken into account when the distribution of enzymes and of radioactivity was calculated.
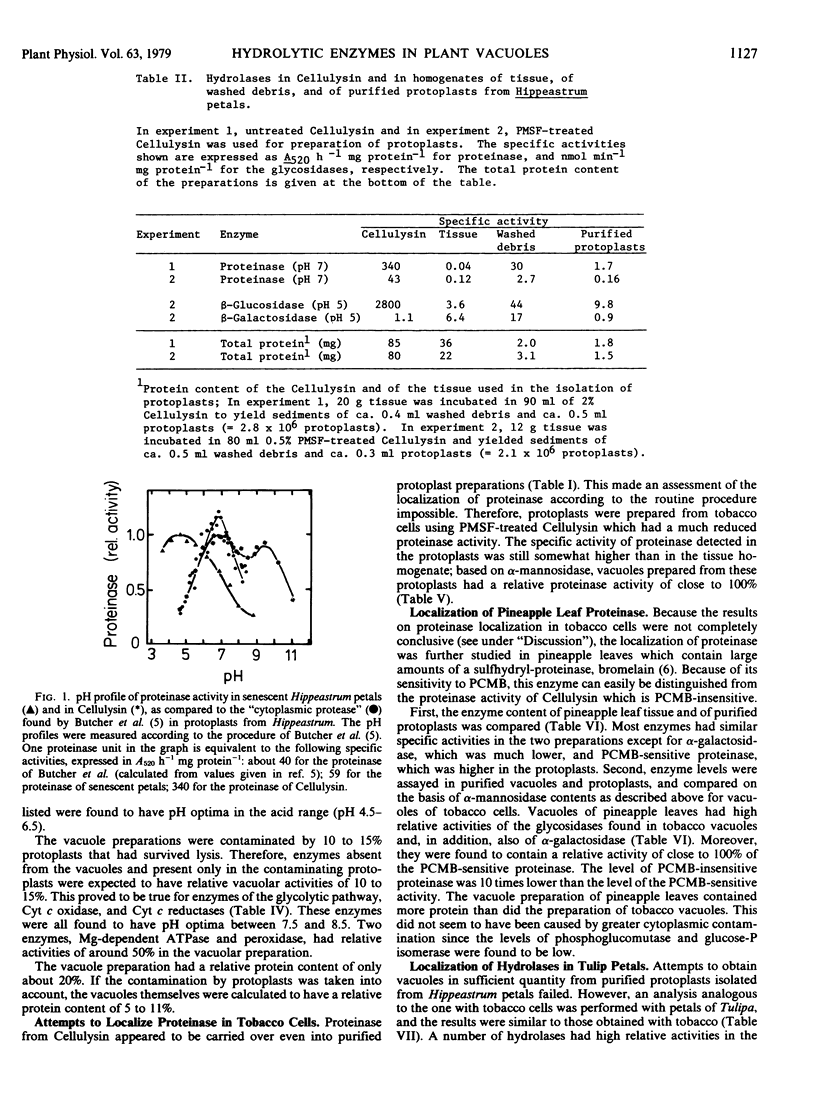
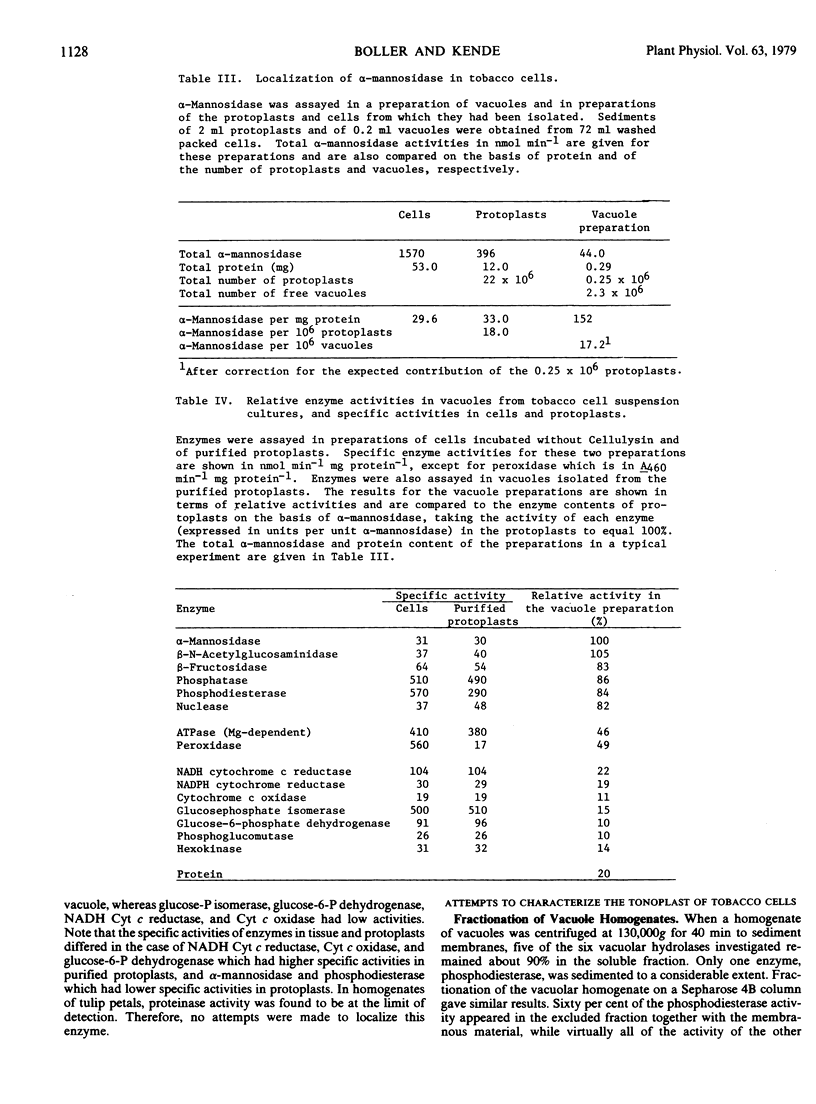
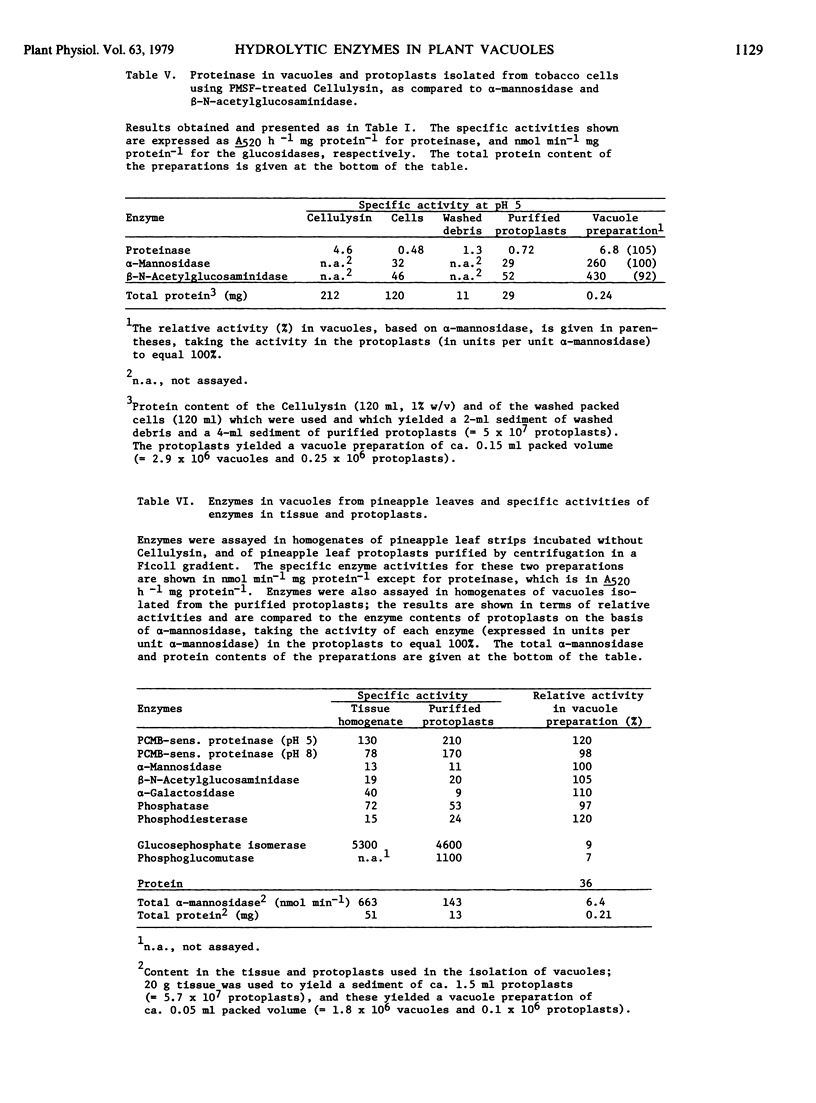
The intracellular activities of the following acid hydrolases were primarily localized in the vacuole of tobacco cells: α-mannosidase, β-N-acetylglucosaminidase, β-fructosidase, nuclease, phosphatase, phosphodiesterase. A similar composition of acid hydrolases was found in vacuoles obtained from protoplasts of tulip petals. Proteinase, a hydrolase with low activity in tobacco cells and tulip petals and therefore difficult to localize unequivocally, was found to be vacuolar in pineapple leaves, a tissue containing high levels of this enzyme. Our data support the hypothesis that the central vacuole of higher plant cells has an enzyme composition analogous to that of the animal lysosome.
None of the vacuolar enzymes investigated was found to be bound to the tonoplast. When vacuoles were isolated from cells labeled with radioactive choline, the vacuolar membrane was found to contain radioactivity. On sucrose gradients, the label incorporated into tonoplasts banded around a density of 1.10 grams per cubic centimeter (24% sucrose, w/w).
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Butcher H. C., Wagner G. J., Siegelman H. W. Localization of Acid hydrolases in protoplasts: examination of the proposed lysosomal function of the mature vacuole. Plant Physiol. 1977 Jun;59(6):1098–1103. doi: 10.1104/pp.59.6.1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dürr M., Boller T., Wiemken A. Polybase induced lysis of yeast spheroplasts. A new gentle method for preparation of vacuoles. Arch Microbiol. 1975 Nov 7;105(3):319–327. doi: 10.1007/BF00447152. [DOI] [PubMed] [Google Scholar]
- Galbraith D. W., Northcote D. H. The isolation of plasma membrane from protoplasts of soybean suspension cultures. J Cell Sci. 1977 Apr;24:295–310. doi: 10.1242/jcs.24.1.295. [DOI] [PubMed] [Google Scholar]
- Hodges T. K., Leonard R. T. Purification of a plasma membrane-bound adenosine triphosphatase from plant roots. Methods Enzymol. 1974;32:392–406. doi: 10.1016/0076-6879(74)32039-3. [DOI] [PubMed] [Google Scholar]
- Leigh R. A., Branton D. Isolation of Vacuoles from Root Storage Tissue of Beta vulgaris L. Plant Physiol. 1976 Nov;58(5):656–662. doi: 10.1104/pp.58.5.656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin W., Wagner G. J., Siegelman H. W., Hind G. Membrane-bound ATPase of intact vacuoles and tonoplasts isolated from mature plant tissue. Biochim Biophys Acta. 1977 Feb 14;465(1):110–117. doi: 10.1016/0005-2736(77)90359-5. [DOI] [PubMed] [Google Scholar]
- Matile P., Wiemken A. The vacuole as the lysosome of the yeast cell. Arch Mikrobiol. 1967 Feb 20;56(2):148–155. doi: 10.1007/BF00408765. [DOI] [PubMed] [Google Scholar]
- McEwen C. R. Tables for estimating sedimentation through linear concentration gradients of sucrose solution. Anal Biochem. 1967 Jul;20(1):114–149. doi: 10.1016/0003-2697(67)90271-0. [DOI] [PubMed] [Google Scholar]
- Nishimura M., Beevers H. Hydrolases in vacuoles from castor bean endosperm. Plant Physiol. 1978 Jul;62(1):44–48. doi: 10.1104/pp.62.1.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohnishi T., Gall R. S., Mayer M. L. An improved assay of inorganic phosphate in the presence of extralabile phosphate compounds: application to the ATPase assay in the presence of phosphocreatine. Anal Biochem. 1975 Nov;69(1):261–267. doi: 10.1016/0003-2697(75)90585-0. [DOI] [PubMed] [Google Scholar]
- Ray P. M., Shininger T. L., Ray M. M. ISOLATION OF beta-GLUCAN SYNTHETASE PARTICLES FROM PLANT CELLS AND IDENTIFICATION WITH GOLGI MEMBRANES. Proc Natl Acad Sci U S A. 1969 Oct;64(2):605–612. doi: 10.1073/pnas.64.2.605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saunders J. A., Conn E. E. Presence of the cyanogenic glucoside dhurrin in isolated vacuoles from sorghum. Plant Physiol. 1978 Feb;61(2):154–157. doi: 10.1104/pp.61.2.154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simcox P. D., Reid E. E., Canvin D. T., Dennis D. T. Enzymes of the Glycolytic and Pentose Phosphate Pathways in Proplastids from the Developing Endosperm of Ricinus communis L. Plant Physiol. 1977 Jun;59(6):1128–1132. doi: 10.1104/pp.59.6.1128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stafford H. A., Bravinder-Bree S. Peroxidase isozymes of first internodes of sorghum: tissue and intracellular localization and multiple peaks of activity isolated by gel filtration chromatography. Plant Physiol. 1972 Jun;49(6):950–956. doi: 10.1104/pp.49.6.950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker-Simmons M., Ryan C. A. Immunological Identification of Proteinase Inhibitors I and II in Isolated Tomato Leaf Vacuoles. Plant Physiol. 1977 Jul;60(1):61–63. doi: 10.1104/pp.60.1.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiemken A. Isolation of vacuoles from yeasts. Methods Cell Biol. 1975;12:99–109. doi: 10.1016/s0091-679x(08)60954-1. [DOI] [PubMed] [Google Scholar]


