Abstract
Type 2 diabetes mellitus is a complex metabolic disease that has reached epidemic proportions in the United States and around the world. This disease is characterized by loss of insulin secretion and eventually destruction of insulin-producing pancreatic beta cells. Controlling type 2 diabetes is often difficult as pharmacological management routinely requires complex therapy with multiple medications, and loses its effectiveness over time. The objective of this study was to explore the effectiveness of a novel, non-pharmacological approach that utilizes the application of ultrasound energy to augment insulin release from rat INS 832/13 beta cells. The cells were exposed to unfocused ultrasound for 5 min at peak intensity of 1 W/cm2 and frequencies of 400 kHz, 600 kHz, 800 kHz and 1 MHz. Insulin release was measured with enzyme-linked immunosorbent assay (ELISA) and cell viability was assessed via trypan blue dye exclusion test. A marked release (approximately 150 ng/106 cells, p < 0.05) of insulin was observed when beta cells were exposed to ultrasound at 400 kHz and 600 kHz as compared to their initial control values, however this release was accompanied with a substantial loss in cell viability. Ultrasound application at frequencies of 800 kHz resulted in 24 ng/106 cells of released insulin (p < 0.05) as compared to its unstimulated base level, while retaining cell viability. Insulin release from beta cells caused by application of 800 kHz ultrasound was comparable to that reported by secretagogue glucose, thus operating within physiological secretory capacity of these cells. Ultrasound has a potential to find an application as a novel and alternative method to current approaches aimed at correcting secretory deficiencies in patients with type 2 diabetes.
Keywords: Ultrasound Therapy, Cell Stimulation, Insulin, Pancreatic Beta Cells, Type 2 Diabetes
Introduction
Type 2 diabetes mellitus is a complex and heterogeneous metabolic disease that has reached epidemic proportions (CDC 2013; Wild et al. 2004; Zimmet et al. 2001). As of 2016, it is estimated that 29 million Americans have type 2 diabetes while 86 million are living with prediabetes. Main characteristics of type 2 diabetes are the insufficient ability of pancreatic beta cells to secrete insulin, and decreased insulin sensitivity of peripheral tissues (Ferrannini and Mari 2004; Festa et al. 2008; Kahn 2001). People with prediabetes can start showing signs of impaired fasting glucose (IFG) and impaired glucose tolerance (IGT) years before the onset of the disease. Similarly, people with family history of diabetes can start showing early signs of insulin resistance long before they develop overt diabetes. If not identified and addressed in time, subjects with prediabetes and/or family history of diabetes can also develop serious cardiovascular diseases such as atherosclerosis: a leading cause of morbidity and mortality for people with diabetes. Insulin, a peptide hormone, is the main glucose regulator in human body. Insulin is synthesized and stored in secretory vesicles within the pancreatic beta cells, and is released in a calcium-dependent manner in response to changes in blood sugar levels. An accepted model of stimulus-secretion coupling of beta cells attributes glucose-induced insulin secretion to a sequence of events involving closure of ATP-sensitive potassium channels, membrane depolarisation, influx of calcium and a rise in cytosolic free calcium concentration, and calcium-triggered exocytosis of insulin (Henquin 2009; Sakurada et al. 1993). Over time, in patients with type 2 diabetes large population of beta cells undergoes apoptosis or becomes “glucose-blind”. Although remaining beta cells in diabetic patients still produce and store insulin, glucose does not mobilize intracellular calcium and subsequently does not release insulin from these dysfunctional beta cells (Ferrannini and Mari 2004; Israili 2011). To counteract this, some pharmaceutical approaches in the treatment of type 2 diabetes utilize sulfonylureas class of drugs which can change the permeability of beta cell membranes (by targeting ATP sensitive potassium channels) to allow calcium influx and triggering of insulin release (Neumiller and Setter 2009). However, this class of drugs is also shown to promote failure of beta cells (Raskin 2010).
Currently available interventions in the treatment of type 2 diabetes usually fail over time, and new modes of therapy are needed that will target directly the underlying causes of abnormal glucose metabolism, such as beta cell dysfunction (Spellman 2007). In this study, we explored a novel, non-thermal and non-invasive approach that utilizes the application of ultrasound energy to augment insulin release from beta cells as alternative to traditional (pharmacological) approaches. Therapeutic ultrasound has been used for non-invasive and selective targeting of various internal organs including human pancreas in treatment of malignancies (Leslie and Kennedy 2007; Zhao et al. 2010), and with appropriate reduction in ultrasound intensities a similar method may be adopted for stimulation of insulin release. Few studies exist to date that address application of low-intensity therapeutic ultrasound for functional modification of cells and tissues, though most of this work has been done in the area of wound healing. The mechanical effects of ultrasound have been shown to cause modification of cell membrane permeability leading to different rates of transports of ions and molecules across the membrane (Dinno et al. 1989; Hassan et al. 2010; Hsu and Huang 2004; Robinson et al. 1996; Tsukamoto et al. 2011). For example, studies have indicated that ultrasound application can lead to reversible modulation of neural tissues by activating voltage-gated sodium channels, as well as voltage-gated calcium channels (Tyler et al. 2008). These effects were followed by SNARE (Soluble NSF Attachment Protein Receptor)-mediated synaptic vesicle exocytosis indicating that ultrasound may be capable of stimulating exocytosis in other cell types such as pancreatic beta cells (Wheeler et al. 1996). In an earlier study, ultrasound was applied to bovine adrenal chromaffin cells leading to transient influx of calcium which triggered exocytosis of catecholamines, a process known to be similar to the mechanism leading to insulin exocytosis from pancreatic beta cells (Robinson et al. 1996). Further, it has been reported that ultrasound can be used to increase release of a protein hormone adiponectin (by approximately 70%) from adipose cells (Fujii et al. 2006). However, the exact mechanisms leading to this and other ultrasound-stimulated release of hormones are still unknown.
Our study is to the best of our knowledge the first study to explore the potential effects of ultrasound (or any other energy-based modality) on insulin release. In this study, we assessed the effectiveness and safety of ultrasound stimulation in evoking secretory responses in pancreatic beta cells. Further, we performed preliminary studies of thermal and mechanical ultrasound effects involved in stimulation of insulin release. We identified ultrasound parameters that showed to be safe and effective at enhancing insulin secretions from beta cells, offering a potential novel method in correcting insulin secretory deficiency in type 2 diabetes.
Methods and Materials
Rat INS 832/13 cells, an insulin secreting insulinoma beta cell-derived cell line with doubling time of 100 h (INS-1-derived cell line), were routinely cultured in RPMI-1640 tissue culture medium (11.1 mM glucose, pH 7.4) supplemented with 2 mM L-glutamine, 10 mM HEPES, 1 Mm sodium-pyruvate, 0.05 mM 2-mercaptoethanol and 10% fetal calf serum in a 37°C incubator) (VWR International, Radnor, PA, USA ) with 5% CO2 and 95% air. The cells were a gift from Christopher Newgard's laboratory at Duke University (Hohmeier et al. 2000). The cell lines were maintained in 14 ml of culture medium in 75 cm2 sterile vented tissue culture flasks (Greiner GmbH, Pleidelsheim, Germany). Prior to treatment, trypsinated cells were collected and centrifuged for 10 min at 1000 rpm. The supernatant was removed and the cells were re-suspended in 1 ml of modified Krebs bicarbonate solution (sodium chloride 138 mM, potassium chloride 5.4 mM, calcium chloride 2.6 mM, sodium bicarbonate 5 mM, magnesium chloride 1 mM, 10 mM HEPES, pH 7.4) supplemented with 0.1% bovine serum albumin. The cell suspension was then loaded into a 3-D printed exposure chamber made in-house out of polylactic acid (PLA) and with 0.18 mm thick acoustically transparent windows made out of Mylar® (1.5 cm × 1 cm) (Karshafian et al. 2009).
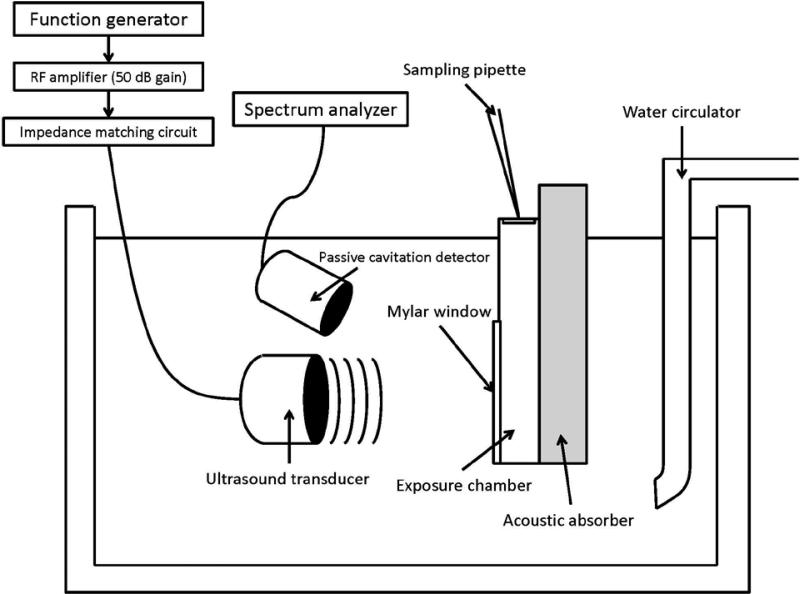
The exposure chamber was placed in a water bath maintained at 37°C (Thermo Haake DC10-P21, Fisher Scientific, Waltham, MA, USA) as shown in Figure 1. Circular planar ultrasound transducers with an active diameter of 1.5 cm and center frequencies of 400 kHz, 600 kHz, 800 kHz and 1 MHz (Sonic Concepts, Inc. Bothell, WA, USA) were directed towards the exposure chamber at a distance of 1.5 cm, 2.25 cm, 3 cm and 3.75 cm, which corresponded to their respective near-field to far-field distances (dff) (Christensen 1988). Ultrasound waveforms were generated using an Agilent 33220A function generator (Agilent Technologies, Santa Clara, CA, USA) and were further amplified (50 dB gain) using a 150A100B RF amplifier (Amplifier Research, Souderton, PA, USA). Ultrasound intensity at different input settings was measured using a reflective radiation force balance (Ultrasound power meter; Ohmic Instruments, Easton, MD, USA). A 3-D micropositioning system with 0.025 mm resolution was used to control the distance between the ultrasound transducer and the exposure chamber. An ultrasound absorber (Precision Acoustics LTD, Dorchester, United Kingdom) was placed in the back of the exposure chamber in order to minimize the production of standing waves. Cell samples with density roughly around 2-5×106 cells/ml were suspended in 1 ml of glucose-free Krebs bicarbonate solution (KBS), placed inside the exposure chamber and treated for 5 min with ultrasound at intensity ISATA of 1 W/cm2 in a continuous mode using the previously mentioned center frequencies. Aliquots of 100 μL were acquired prior to the start of the treatment (t = 0 min), immediately after treatment (t = 5 min) and 30 minutes after treatment (t = 35 min) for analysis. The cells in the chamber were re-suspended with a micropipette immediately before collecting each alliquot. To serve as positive controls, insulin release was measured from cells suspended in glucose-supplemented (12 mM) modified Krebs Ringer bicarbonate buffer (pH 7.4). Sham treatment consisted in placing the cell suspension in the exposure chamber and following the same sample acquisition procedure performed on ultrasound-treated cells but without exposing them to ultrasound.
Figure 1.
In vitro experimental setup for beta cell stimulation experiments. Ultrasound transducer and cell exposure chamber were placed inside the temperature-controlled water bath.
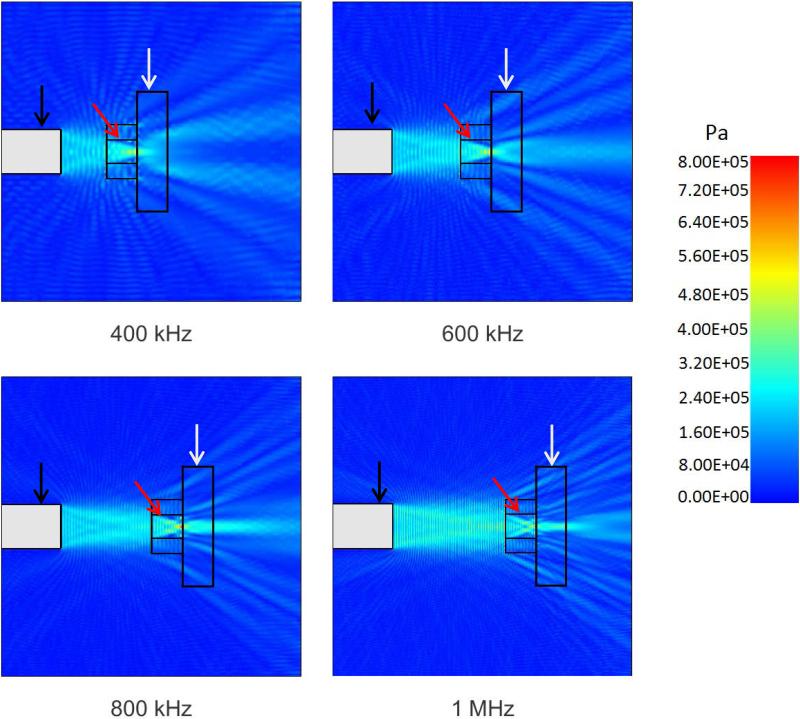
The experimental setup shown in Figure 1 was modeled using PZFlex modeling software (Weidlinger Associates, Mountain View, CA, USA). The purpose of these simulations was to establish a range of pressures to which the cells were exposed to as result of any potential formation of standing waves within the exposure chamber. Further, simulations provided pressure maps at very high spatial resolution, therefore better characterizing the acoustic field affecting the cells. Simulation parameters in the PZFlex model were established as previously reported (Hensel et al. 2011). Material properties, parameters and dimensions were obtained from our measurements, manufacturers’ data and published data. The grid size was set to one fifteenth of the exposure wavelength to ensure proper spatial resolution as recommended by the PZFlex software manufacturer (Nabili et al. 2015). The acoustic absorber was assumed to absorb 99% of the incident energy per the manufacturer's specifications. Pressure maps of our experimental setup were generated for the different ultrasound frequencies used experimentally (Figure 2). Simulations showed that cells in the chamber were exposed to peak pressures of 227 ± 80.23 kPa, 218 ± 90.25 kPa, 228 ± 96.15 kPa and 220 ± 83.38 kPa when exposed to ultrasound beams with frequencies of 400 kHz, 600 kHz, 800 kHz and 1 MHz, respectively. Peak rarefactional pressures were calculated to be −221 ± 82.61 kPa, −229 ± 97.83 kPa, −221 ± 90.85 kPa and −220 ± 85.83 kPa for frequencies of 400 kHz, 600 kHz, 800 kHz and 1 MHz, respectively. The results suggested that some standing waves were generated due to reflective patterns formed inside the cell exposure chamber. However, most regions in the chamber were exposed to pressures only slightly higher than the ultrasound beam's peak pressure of 0.18 MPa (corresponding to ISATA of 1 W/cm2) potentially due to the natural focusing of the acoustic field at dff distance. Simulated pressure calculations were compared to point measurements obtained experimentally with an acoustic hydrophone (HGL-0085, Onda Corporation, Sunnyvale, CA) resulting in differences no higher than 20%.
Figure 2.
Simulated pressures in the water bath experimental setup during ultrasound application at different frequencies (view from top). Simulations were done using PZFlex modeling software. Ultrasound transducer is shown with the black arrow, cell exposure chamber is shown with the red arrow, and acoustic absorber with the white arrow.
For cell viability studies, the number of viable beta cells before and after the treatment was determined using a trypan blue dye exclusion test (Tennant 1964). Ten μL (2-5×106 cells/ml) of each cell sample was acquired and mixed with 10 μL of 0.5% trypan blue solution (Bio-Rad Laboratories, Inc. Hercules, CA, USA). Ten μL of the mix were acquired and placed on a dual chamber cell counting slide (Bio-Rad Laboratories, Inc. Hercules, CA, USA). The cell counting slide was then loaded in a TC20 automatic cell counter (Bio-Rad Laboratories, Inc. Hercules, CA, USA) to determine the proportion of the cells which excluded the dye. Results were presented as the percent ratio of viable cells to the total number of cells in the sample. Percent cell loss during treatment was further estimated by taking the difference between the cell count before and immediately after treatment and dividing the difference by the former.
To determine extracellular insulin concentration after the treatment, the samples acquired from ultrasound-treated, glucose-supplemented (positive control) and sham groups were centrifuged for 10 min at 1000 rpm and the supernatants were collected for insulin quantification. Insulin concentration in collected supernatants was determined using enzyme-linked immunosorbent assay (ELISA) Insulin Kit (Millipore Corporation, Billerica, MA, USA; Inter-assay (%): 9.1–11.4; Intra-assay (%): 4.6–7.0) using a SpectraMax M5 Spectrometer (Molecular Devices, Sunnyvale, CA, USA). The amount of insulin in a specific sample, measured in ng/ml, is linearly proportional to the fluorescence detected by the spectrometer in the range of 0.2 ng/ml to 10 ng/ml. Collected supernatants were diluted until their measured insulin concentrations fell within the linear range of the ELISA assay (0.2 ng/ml to 10 ng/ml). The final insulin concentration was determined by multiplying the measured concentration by its respective dilution factor. Measured insulin concentrations in each sample were adjusted to their respective initial cell count as determined using a TC20 automatic cell counter (Bio-Rad Laboratories, Inc. Hercules, CA, USA). Insulin release at t = 5 min and t = 35 min was quantified by taking the difference between their measured extracellular insulin concentrations and their respective control value at t = 0 min. We expect to measure positive differences for enhanced insulin release, differences close to zero for no effect on insulin release and negative differences for decreased extracellular insulin content. Samples acting as positive controls were suspended in KBS supplemented with 12 mM glucose, a concentration shown to naturally induce insulin secretion in INS 832/13 cell lines (Hohmeier et al. 2000). Results were presented as the average insulin concentration ± SEM in units of ng/106 cells (n = 6).
In a separate and limited set of experiments, changes in intracellular insulin content in cells treated with ultrasound and sham groups were determined. The amount of insulin in INS 832/13 beta cells is estimated to be 1440 ± 348 ng/106 cells (mean ± SEM) (Hohmeier et al. 2000). These experiments were performed to ensure that any released insulin detected in the extracellular space of the beta cells came from healthy, non-lysed cells and not from cells whose membrane integrity may have been irreversibly compromised as result of ultrasound exposure. Briefly, cell samples acquired from ultrasound-treated and sham groups were washed twice in modified Krebs Ringer bicarbonate buffer (pH 7.4), re-suspended in RIPA 1X buffer for lysing and kept on ice for 90 min. Samples were then centrifuged for 10 min at 14000 rpm, and supernatants were collected and stored at −70°C for subsequent insulin quantification with ELISA. Similar to measurements of released insulin, samples were diluted until their measured insulin concentrations fell within the linear range of the ELISA assay (0.2 ng/ml to 10 ng/ml). Further, intracellular insulin contents measured in samples acquired immediately after the treatment (t = 5 min) and 30 min after the treatment (t = 35 min) were adjusted to their respective cell count as determined using a TC20 automatic cell counter (Bio-Rad Laboratories, Inc. Hercules, CA, USA). Changes in intracellular insulin content at t = 5 min and t = 35 min were quantified by taking the difference between their measured intracellular insulin concentrations and their respective baseline value at t = 0 min. We expect to measure negative differences in intracellular insulin for enhanced insulin release, values close to zero for no effect on insulin release and positive differences for increased insulin content. Results were presented as the average insulin concentration measured in all the samples belonging to both sham and 800 kHz group (n=6) along with their respective standard error of the mean (SEM).
Graph Pad Prism 5 Program (GraphPad Software, Inc., La Jolla, CA, USA) was used for plotting and statistical analysis of collected data. For multiple comparisons between the control and treated groups data was analyzed by one-way ANOVA test. Significance was established at p < 0.05. For the case of intracellular insulin content comparison between sham group and cells exposed to 800 kHz ultrasound, a Student's t-test assuming unequal variances was used (as this data only contained two groups) and significance established at p < 0.05.
The cavitation activity inside the chamber was characterized using passive cavitation detection (PCD) for all ultrasound frequencies used in this study. A single-element PCD transducer (bandwidth of 2.8 MHz to 4.2 MHz; ISO304HP, CTS Valpey Corporation, Hopkinton, MA, USA) was aimed at the exposure chamber thus intersecting with the ultrasound beam's path inside the chamber (see Figure 1). The signals obtained by the PCD transducer were sent to a spectrum analyzer (MDO3024, Tektronix, Arlington, VA), and the digitized data was acquired for further analysis in MATLAB. The presence of stable cavitation during ultrasound exposure was determined by identifying the subharmonic and ultraharmonics of all the frequencies used in our study, while the presence of inertial cavitation was determined by identifying the presence of broadband noise across the frequency spectrum of the acquired signal (Leighton 1994). Broadband noise was quantified in two ways. In Method 1, an eighth-order polynomial was fitted to the frequency spectrum in order to omit the signal's harmonic peaks. The fitted signal was then integrated across the detector's bandwidth in a similar manner as described in previous studies (Li et al. 2014; Rabkin et al. 2005). In Method 2, a frequency window was selectively picked that was common to all four frequencies used in this study. This window was chosen on the basis of not containing any of the four frequencies’ harmonics while being located within the detector's bandwidth (Chen et al. 2003). This window was chosen to be from 3.05 MHz to 3.15 MHz and the region of the spectrum was integrated as defined by this window for quantification of inertial cavitation.
In a separate set of experiments, temperature changes due to ultrasound exposure were monitored by inserting a thin-wire thermocouple (range: −200°C to 650°C; resolution: 0.1°C; accuracy: 0.1%) in the cell exposure chamber during the ultrasound treatment. The signal from the thermocouple was recorded using a dual input thermometer (Wavetek Waterman TMD90). Readings from the thermometer were recorded at t = 0 min (right before the treatment) and then every 30 s for 5 min of ultrasound treatment at different frequencies and for additional 5 min after the ultrasound treatment (n=3).
Results
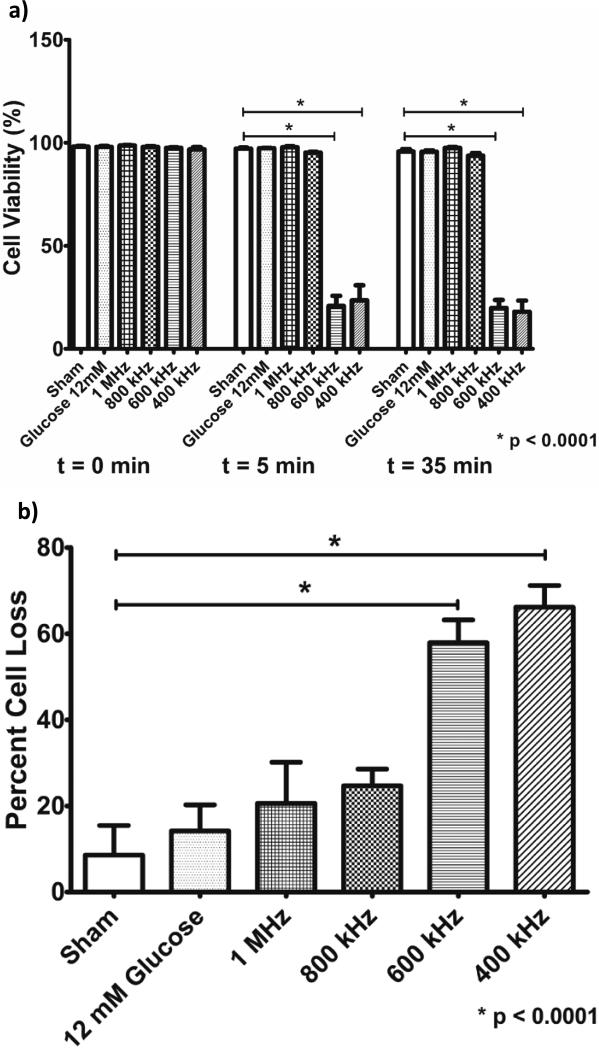
Cell viability studies were performed to assess the safety of the chosen ultrasound parameters (Figure 3a). Cell viability was significantly reduced by almost 80% when beta cells were treated with ultrasound at frequencies of 400 kHz and 600 kHz (from 96.8% to 23.5% and from 97.5% to 20.8% in cell viability respectively, p < 0.0001). In contrast, little to no harmful effects were seen in cell samples treated with ultrasound at 800 kHz (from 97.8% to 95.8% cell viability, p > 0.05) and 1 MHz (from 98.7% to 97.8% in cell viability, p > 0.05) as compared to untreated samples (from 98.1% to 97.1% cell viability in sham group, p > 0.05). As expected, insulin secretagogue glucose had no significant effect on cell viability throughout the experiment (from 98% to 97.3% cell viability). When considering percent cell loss after treatment, we see that all groups experienced some cell loss (Figure 3b). In agreement with cell viability data, there appears to be a notable difference between cells exposed to lower ultrasound frequencies of 400 kHz and 600 kHz (66% and 55% cell loss, respectively) and cell exposed to higher ultrasound frequencies of 800 kHz and 1 MHz (24% and 21% cell loss, respectively). However, statistical significance was only achieved in the 600 kHz and 400 kHz groups as compared to the sham group (p < 0.0001). The percent cell loss in the sham group and the positive control group were 9% and 14%, respectively.
Figure 3.
a) Cell viability (mean ± SEM) measured immediately before the treatment (t=0 min), immediately after the treatment (t=5 min) and 30 min after the treatment (t=35 min) for pancreatic beta cells exposed to ultrasound at different frequencies (at 1 W/cm2 for 5 min), sham-treated cells, and positive control (12 Mm glucose – a natural secretagogue) (n=6 per group). b) Percent cell loss (mean ± SEM) measured between the cell count before the treatment (t=0 min) and immediately after the treatment (t=5 min) for pancreatic beta cells exposed to ultrasound at different frequencies (at 1 W/cm2 for 5 min), sham-treated cells, and positive control (12 Mm glucose) (n=6 per group). A significant reduction in cell viability and percent cell loss was observed after ultrasound application at frequencies of 400 kHz and 600 kHz.
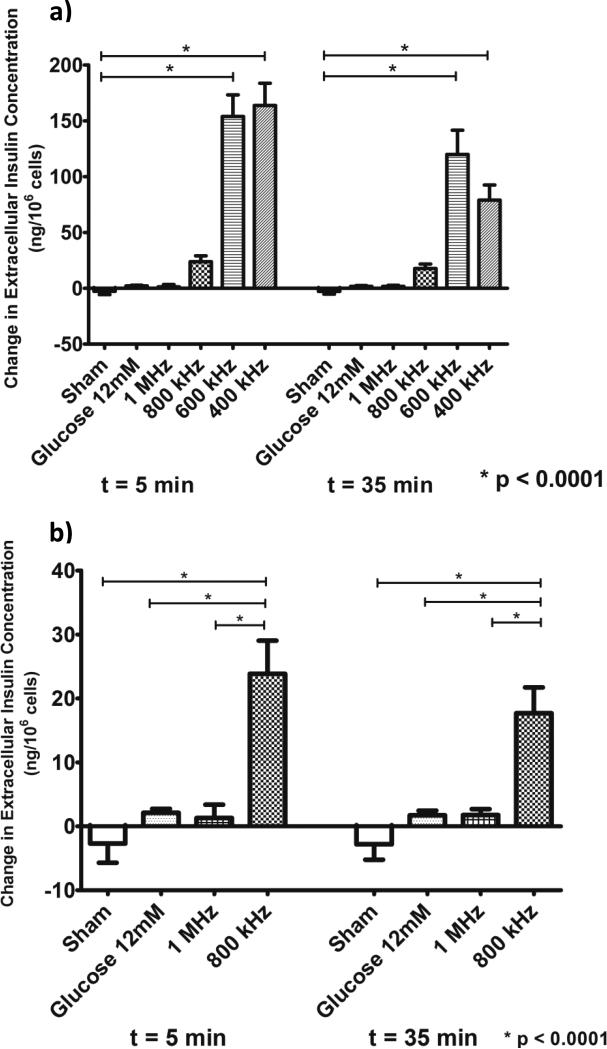
Changes in extracellular insulin concentration (as compared to their baseline at t = 0 min, immediately before treatment) in response to treatment are shown in Figure 4. As expected, extracellular insulin levels in the sham-treated group at t = 5 min (immediately after the treatment) and t = 35 min (30 min after the treatment) did not show any significant changes from their base value at t = 0 min (immediately before treatment) as the suspension medium did not contain any glucose or any other insulin secretagogue (Figure 4). In fact, we measured a small, non-significant decrease in the extracellular insulin content of the sham group averaging approximately 3 ng/106 cells as compared to its base value at t = 0 min (as shown by the negative change). Cell exposure to 800 kHz ultrasound resulted in release of 24 ng of insulin per 106 cells (p < 0.0001, Figure 4) with no significant effect on cell viability (see Figure 3). Furthermore, no further increase in measured levels of insulin was observed 30 min after the end of exposure, indicating that insulin release stopped immediately after the end of treatment at t = 5 min. In comparison, cells suspended in 12 mM secretagogue glucose (serving as positive control) showed a modest stimulatory effect on insulin release averaging 1.8 ng/106 cells respective to its value at t = 0 min. Beta cells exposed to 1 MHz ultrasound also showed a small and non-significant increase in released insulin (approximately 2 ng/106 cells) as compared to control conditions, thus revealing a potential frequency threshold for insulin release by ultrasound in these cells (Figure 4). However, it is possible that clinically-meaningful insulin secretion may still be achieved when using 1 MHz ultrasound at different parameters (e.g. slightly higher peak intensities). Further, significant amounts of insulin (approximately 150 ng/106 cells, p < 0.0001) were released from beta cells exposed to 400 kHz and 600 kHz ultrasound (Figure 4) but at the cost of significantly reduced cell viability (see Figure 3).
Figure 4.
(a) Quantification of insulin released (mean ± SEM) from pancreatic beta cells exposed to ultrasound at different frequencies as compared to sham treatment and positive control (12 mM glucose) immediately after the 5 min long ultrasound treatment (t=5 min) and 30 min after the treatment (t=35 min) (n=6 per group). (b) Insulin release results at ultrasound frequencies for which no loss in cell viability was observed (see Figure 3). Ultrasound applied at 800 kHz (1 W/cm2 for 5 min) was able to release insulin within clinically-relevant levels of insulin (p<0.0001). No further insulin release was observed at 30 min after the ultrasound treatment indicating that no irreparable damage was produced in the cell membranes.
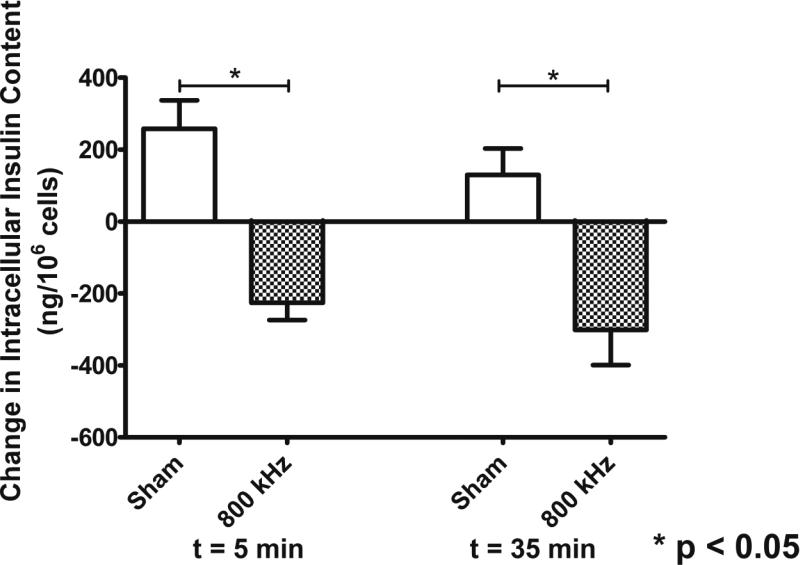
Measurements of changes in intracellular insulin content (as compared to their baseline at t = 0 min, immediately before treatment, Figure 5) were in agreement with results obtained from experiments that tested changes in extracellular insulin (see Figure 4) in showing a general reduction of intracellular insulin content in cells exposed to 800 kHz ultrasound. Our results showed a reduction of approximately 250 ng/106 cells both immediately after the treatment (t = 5 min) (p < 0.05) and 30 min after the treatment (t = 35 min) (p < 0.05) in the intracellular insulin content of the beta cells in ultrasound-treated samples (tested frequency of 800 kHz), thus indicating increased insulin release from the beta cells in response to ultrasound exposure. In the case of the sham group, we actually measured higher levels of intracellular insulin (see Figure 5) both at t = 5 min (approximately 200 ng/106 cells) and t = 35 min (approximately 130 ng/106 cells).
Figure 5.
Quantification of changes intracellular insulin content (mean ± SEM) in pancreatic beta cells exposed to 800 kHz ultrasound (1 W/cm2 for 5 min), and sham-treated cells (n=7 per group). A significant reduction (p < 0.05) in the intracellular insulin content was observed immediately after the completion of the ultrasound treatment (t=5 min) and 30 min after the treatment (t=35 min), thus indicating insulin release from the beta cells due to ultrasound exposure.
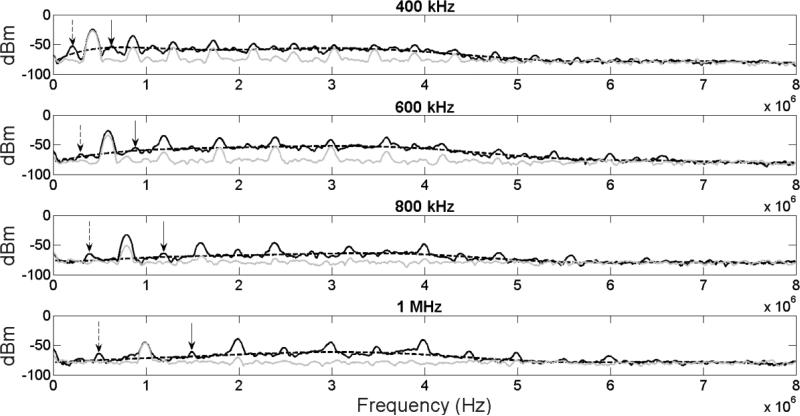
Spectra obtained using PCD measurements during ultrasound application for stimulation of beta cells are shown in Figure 6. Stable cavitation, as measured by the presence of the frequencies’ subharmonics and ultraharmonics (dashed and solid black arrows respectively), was shown to be present inside the cell exposure chamber for all four ultrasound frequencies used. Inertial cavitation was also observed at all four tested frequencies as shown by the presence of broadband noise and estimated by the eighth-order polynomial fitting of the four frequency spectra (dashed curve, Figure 6). The fitting of all four spectra was calculated to have R2 of over 0.95. To highlight the presence of broadband noise, spectra was also generated for all frequencies at an intensity of 0.1 W/cm2 (gray solid line), which is expected to generate little to no inertial cavitation. The results of inertial cavitation measurements at the ultrasound parameters used in our beta cell stimulation studies (1 W/cm2, 400-1000 kHz) are shown in Table 1. It can be seen that there is a distinct increase in measured inertial cavitation in the spectra corresponding to 400 kHz and 600 kHz ultrasound when compared to the spectra at higher frequencies, suggesting that this increase in inertial cavitation may be involved in the significant reduction in the cell viability observed in Figure 3.
Figure 6.
Spectra obtained using PCD during ultrasound application for stimulation of pancreatic beta cells (solid black curve, intensity of 1 W/cm2, frequencies of 400-1000 kHz). Subharmonics (dashed black arrows), ultraharmonics (solid black arrows) and broadband noise were observed in all cases at the parameters used in the beta cells stimulation experiments. Inertial cavitation was estimated by the eighth-order polynomial fitting of the four frequency spectra (dashed curve). Spectra obtained at an intensity of 0.1 W/cm2 is represented by a solid gray curve (for comparison reasons).
Table 1.
Quantification of inertial cavitation measured during ultrasound application for stimulation of pancreatic beta cells.
| Frequency (kHz) | Method 1 (VRMS) | Method 2 (VRMS) |
|---|---|---|
| 400 | 1.38×10−4 | 1.80×10−6 |
| 600 | 1.33×10−4 | 2.76×10−6 |
| 800 | 9.42×10−5 | 5.75×10−7 |
| 1000 | 7.51×10−5 | 9.20×10−7 |
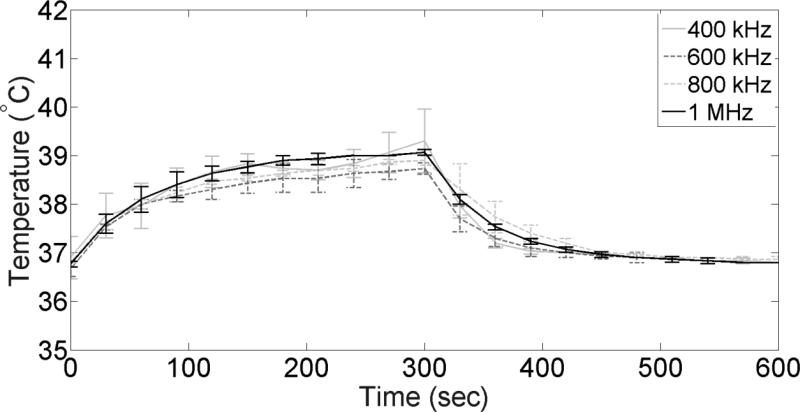
Figure 7 shows measurements of temperature elevations in the exposure chamber during ultrasound treatment at all four frequencies used in our studies. It can be seen that a 5 min ultrasound exposure caused temperature increase no higher than 3°C at all four tested frequencies. Furthermore, the trend appears to be the same in all cases thus potentially indicating that the observed differences in cell viability and insulin release among different ultrasound frequencies are due to mechanical rather than thermal effects.
Figure 7.
Temperature measurements inside the cell exposure chamber during ultrasound treatments at different frequencies (n=3).
Discussion
The results of our experiments suggested that ultrasound exposure could stimulate insulin release from pancreatic beta cells in a safe and controlled manner. Application of 800 kHz ultrasound resulted in significant (24 ng/106 cells) increase in insulin release from the beta cells, whereas the cells exposed to 1 MHz ultrasound showed a lesser (2 ng/106 cells on average) increase in insulin release (see Figure 4), both of which could be useful in fighting hyperglycemia in type 2 diabetes. It is possible that 1 MHz ultrasound can also safely stimulate significant insulin release from beta cells at a slightly higher intensity. Both 800 kHz and 1 MHz frequencies exhibited similarly high levels of cell viability (96% and 98%, respectively) with no statistical significance compared to sham group and positive controls (see Figure 3a). Percent cell loss appears to be higher, on average, in ultrasound-treated cells at 1 MHz and 800 kHz (21% and 24% cell loss, respectively) as compared to the sham group, though no statistical significance was achieved in either case (see Figure 3b). Increased cell loss in ultrasound treated samples at 1 MHz and 800 kHz could be the result of cell lysing (unlikely as we would expect to measure significantly higher levels of released insulin in the 1 MHz group in a such scenario), or cells lost during transfer as all groups exhibited some level of cell loss. Our data also showed that insulin release stopped immediately after ultrasound treatment as no further increase in extracellular insulin content was detected at t = 35 min, thus highlighting the fact that ultrasound-induced insulin release could be controlled. It is important to note that 800 kHz ultrasound stimulation of INS 832/13 beta cells produced insulin release that is comparable to the response reportedly evoked by natural secretagogue glucose under non-hyperglycemic conditions (Hohmeier et al. 2000). This is important because too much insulin release can also be harmful to patients as it can lead to hypoglycemia. Positive controls with 12 mM glucose showed a very modest increase in released insulin over the treatment period. This lack of glucose responsiveness was largely due to particular experimental conditions in that prior to glucose treatment, these cells were routinely cultured and incubated in 11.1 mM glucose-supplemented media for an extended period of time (72 h), to mimic hyperglycemic conditions found in diabetic patients. As expected, subsequent exposure of these cells to hyperglycemic conditions (12 mM glucose supplemented KBS) had a low stimulatory effect on insulin release as demonstrated in Figure 4. However, in contrast to high glucose stimulus, ultrasound showed its effectiveness to stimulate significant insulin release from cells that experienced prolonged hyperglycemic conditions while in culture. The beta cells exposed to lower frequencies of 400 kHz and 600 kHz experienced significant decrease in cell viability and percent cell loss (of approximately 80% and 60%, respectively) which resulted in substantial amounts of insulin released into the extracellular space (average of 150 ng/106 cells). Release of large amounts of insulin from cells exposed to 400 kHz and 600 kHz ultrasound might be the result of a stimulatory effect at these frequencies or insulin that may have leaked out of cells suffering membrane damage. The amount of insulin released following exposure to 400 kHz and 600 kHz ultrasound accounted for approximately 10% of the total insulin content reported and measured in INS 832/13 insulinoma cell line (1427 ± 258 ng/106 cells (mean ± SEM)), which leads us to believe that the effects of ultrasound exposure may affect mostly vesicles fused to or near the cell's plasma membrane. It is estimated that approximately 7% of all vesicles in beta cells are docked to the plasma membrane (Olofsson et al. 2002).
Measurements of changes in intracellular insulin content supported the observation of enhanced insulin release from cells exposed to 800 kHz ultrasound as compared to those belonging to the sham group. In these experiments, intracellular insulin content was reduced in cells treated with 800 kHz ultrasound (as represented by the negative change in intracellular insulin) while it appears to have increased in the sham group (as represented by the positive change in intracellular insulin). We believe that the increased insulin content in the sham group may be due to potential factors such as extracellular insulin uptake through endocytosis or continuous insulin synthesis (Eliasson et al. 1996; Kaelin et al. 1978; Orci et al. 1973; Rorsman and Renström 2003). Though the general trends of insulin release measurements into the extracellular space and changes in intracellular insulin concentration are in good agreement, their measured magnitudes appear to be notably different. We believe that this is because measurements of intracellular insulin content required substantial dilution with KBS (500 to 1000 times) due to high insulin content in INS 832/13 beta cells. Such dilution could have substantially amplified inter- and intra-assay variations as the measured insulin concentrations were later corrected for their dilution factors. The insulin content in these cells was measured to be 1427 ± 258 ng/106 cells (mean ± SEM), which is consistent with values of 1440 ± 348 ng/106 cells reported in published literature (Hohmeier et al. 2000).
Our thermal measurements showed that 5 min ultrasound exposure raised the temperature of the cell medium by no higher than 3°C for all frequencies considered, which is unlikely to have any damaging effect on the cells. Our study of cavitation activity showed that stable cavitation and inertial cavitation were present in the cell chamber when ultrasound was applied at all frequencies under study. However, lower frequencies of 400 kHz and 600 kHz exhibited distinctively higher levels of inertial cavitation compared to frequencies of 800 kHz and 1 MHz, suggesting that higher levels of inertial cavitation may play an increasingly detrimental role regarding cell viability. Further studies are required to determine the exact mechanisms involved in ultrasound-enhanced insulin release from pancreatic beta cells. In addition to cavitation, other mechanisms documented in literature could play an important role in this process (Suarez Castellanos et al. 2016).
Insulin is naturally secreted from pancreatic beta cells in a calcium-dependent manner, where calcium influx is the last triggering step before exocytosis of insulin containing vesicles in glucose-stimulated insulin secretion. Furthermore, ultrasound induced-bioeffects have been shown to produce intracellular calcium transients in various cell types, which have triggered Ca2+-dependent exocytosis of secretory vesicles. For example, Tyler et al. (2008) showed in an ex vivo study that low-intensity, low-frequency ultrasound was capable of activating Ca2+ transients followed by SNARE-mediated synaptic vesicle exocytosis. Another study showed that ultrasound exposure of chromaffin cells was capable of releasing catecholamines via Ca2+-mediated exocytosis (Robinson et al. 1996). It is therefore possible that the observed increase in insulin secretion from pancreatic beta cells when treated with ultrasound is the result of ultrasound-induced calcium transients and subsequent triggering of insulin vesicle exocytosis. Calcium currents have also been shown to be essential to a cell's resealing process after exposure to acoustic cavitation and other mechanical stresses generated by 24 kHz ultrasound exposures (Schlicher et al. 2006).
Ultrasound-induced changes in the cell membrane permeability have been strongly correlated with cavitation activity, which leads to formation of reversible pits in the cell membranes thus allowing delivery of genes, drugs, and macromolecules into the cells or release of the cell components (Guzman et al. 2002; Zderic et al. 2004). Specifically, a study showed that cavitation generated by ultrasound enhanced cellular incorporation of macromolecules of up to 28 nm in radius through repairable micron-scale holes in the membrane of DU 145 prostate cancer cells which were shown to reseal after 1 minute (using native cell healing response involving endogenous vesicle-based membrane resealing)(Schlicher et al. 2006). Tsukamoto et al. (2011) demonstrated that cytoplasmic calcium in fibroblasts cultured in vitro was increased in response to stable cavitation generated by exposure to 1 MHz pulsed ultrasound. In a similar study, Mortimer et al. (1988) showed that ultrasound treatment increased calcium uptake in 3T3 fibroblasts by almost 20% after a 5 minute exposure. Similar ultrasound effects could potentially lead to the formation of reversible pits in the membrane of pancreatic beta cells, creating a calcium influx and the subsequent release of insulin. Our study on cavitation activity showed the presence of both stable and inertial cavitation at ultrasound frequencies that showed increased insulin release from pancreatic beta cells. It is therefore possible that either one or both types of cavitation activity are involved in ultrasound enhanced-insulin release from pancreatic beta cells though further studies are required to confirm this hypothesis.
Other possible mechanisms responsible for enhanced membrane permeability and subsequent insulin release include mechanical stimulation of mechano-sensitive proteins in the plasma membrane of the beta cell. Studies have shown that ionic mechanisms other than the inhibition of KATP channels may be involved in membrane depolarization caused by higher glucose concentrations. One of these mechanisms is believed to be beta cell swelling induced by high concentration glucose and dependent on glucose metabolism (Helen et al. 2007; Semino et al. 1990; Takii et al. 2006). Beta cell swelling is believed to be caused by increased intracellular lactate (Best 1999), Na+ and Cl− (Best et al. 1997) concentrations due to increased beta cell metabolic activity, leading to intracellular hyperosmolarity and ultimately, insulin secretion. Increased insulin secretion caused by osmotic cell swelling has been attributed to stimulation of stretch-activated cation channels (SAC) sensitive to mechanical stretching of the plasma membrane (Best et al. 2010) and volume-regulated anion channels (VRAC) sensitive to cell volume changes due to hypotonicity-induced cell swelling (Takii et al. 2006). Therefore, it is possible that SAC and VRAC channels are activated by physical and subcellular perturbations of the beta cell structure in response to ultrasound exposure. Activation of these channels could in turn be responsible for membrane depolarization, activation of voltage-gated Ca2+ channels and subsequent insulin secretion.
Many studies have been aimed at identifying ultrasound-mediated bioeffects that can mechanically activate membrane proteins and modulate intracellular pathways, a process often referred to as mechanotransduction. In particular, low-intensity ultrasound was shown to cause morphological changes to neuronal cells, a process that the authors believe could have implications in neuronal cell growth and other downstream cellular processes mediated by the cytoskeleton of the cell (Hu et al. 2013; Hu et al. 2014). These cells were shown to recover their original pre-exposure size within 30 minutes after the end of exposure. Transient changes in cell morphology and cytoskeletal disruptions caused by ultrasound exposure could stimulate machano-sensitive membrane proteins (VRAC or SAC), depolarizing the membrane to levels necessary to open Ca2+ channels and consequently stimulate insulin secretion. Another effect of ultrasound that could play a role in modulating membrane channels is a process known as “intramembrane cavitation”. Krasovitski et al. (2011) suggested that the cell membrane is capable of transforming oscillating acoustic pressure waves into intracellular deformations. Such cyclic deformations could stimulate cycles of stretch and release in the cell membrane and the cytoskeleton, which could in turn stimulate mechano-sensitive proteins, increase membrane permeability and depolarize the cell's membrane. Other ultrasound bioeffects that could play important roles in beta cell stimulation include cell responses to mechanical stresses caused by acoustic radiation force (Morris and Juranka 2007).
Finally, it is also possible that enhanced insulin release resulting from ultrasound exposure is the result of insulin granules leaking out of the cell through transient membrane pores created by ultrasound cavitation. It is estimated that around 700 out of the total 10000 insulin containing granules are docked to the plasma membrane, 200 of which are primed and readily releasable (Olofsson et al. 2002). Granules that are docked and primed at the beta cell's membrane are said to belong to the RRP, while the rest are considered to belong to the reserve pool (RP) (Olofsson et al. 2002). Therefore, it is possible that transient poration of the plasma membrane caused by acoustic cavitation may also be permeating the cell membrane to either insulin leaking directly from the RRP, or insulin granules being released into the extracellular space and subsequently being ruptured by ultrasound bioeffects.
Cell viability as assessed by trypan blue dye exclusion test provides a measure of cell viability by assessing plasma membrane integrity. This measurement is of great relevance to our study since we hypothesize that transient disruption of the plasma membrane may play a role in ultrasound-enhanced insulin secretion. However, previous studies have shown that certain ultrasound exposures can result in other bioeffects that can be harmful to the cells. In particular, it has been observed that cells can become apoptotic or necrotic through Ca2+-dependent pathways and/or mitochondria-caspase pathways when exposed to ultrasound exposures inducing inertial cavitation (Honda et al. 2004; Kumon et al. 2009). Careful optimization of frequencies and duration of stimulation may be effectively used to stimulate insulin-release by non-invasive methods while retaining cell viability as our results indicate.
Our future studies will focus on further studying the effects of ultrasound on cell viability and elucidating the mechanisms involved in ultrasound-enhanced insulin release. Longevity studies are required in order to ensure that cells maintain their ability to proliferate as well as their secretory capabilities long after ultrasound treatment. These studies will include assessment of apoptosis (early and delayed) and necrosis following ultrasound exposure for up to 48 h after treatment. Studies highlighting the role of Ca2+ in ultrasound-stimulated insulin secretion will also be explored. Eventually, we will aim to test the proposed technology in an animal model of type 2 diabetes. An eventual medical device based on this technology could potentially be applied non-invasively from the patient's abdomen similar to the ultrasound application in the treatment of pancreatic cancer (Kotopoulis et al. 2013) or alternatively via implanted ultrasound transducer. Ultrasound would either be applied following a similar regime as pharmacological management (a few times a day, usually after meals) or used in a closed-loop system with wireless feedback from a constant glucose monitor. Our proposed approach may offer the capability of adjusting various ultrasound parameters (frequency, intensity, pulse duration, duty cycle) to deliver an optimal dose of insulin to the patient. Therefore, we hope that this therapeutic strategy will provide the patient with a more personalized approach for the treatment and management of their condition. If our work is shown successful, it could potentially lead to a whole new area of therapeutic ultrasound research. For example, low-intensity therapeutic ultrasound could be tested for enhancement of secretion of other hormones such as thyroid hormones.
Conclusion
The objective of this study was to demonstrate that low-intensity ultrasound can be used to safely stimulate insulin release from pancreatic beta cells in an in vitro environment. Our findings show that ultrasound, when applied at a frequency of 800 kHz and intensity of 1 W/cm2, can induce insulin secretion from pancreatic beta cells similar to secretagogue glucose while preserving cell viability. The mechanisms by which ultrasound can lead to enhanced insulin secretion remain unknown and will be studied in our future experiments. Experiments aimed at fully understanding the ultrasound-induced bioeffects involved in this process and their role in modulating Ca2+ dynamics are needed and will follow.
Acknowledgements
The authors would like to thank Dr. Sanghamitra Singh and Diti Chatterjee Bhowmick for productive discussions and advice. The authors would also like to thank Tania Singh and Bogdan Balteanu for their help during this project. This work was supported by NIH grant 1R03EB019065-01.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Bibliography
- Best L. Cell-attached recordings of the volume-sensitive anion channel in rat pancreatic beta-cells. Biochim Biophys Acta. 1999;1419:248–256. doi: 10.1016/s0005-2736(99)00071-1. [DOI] [PubMed] [Google Scholar]
- Best L, Brown PD, Sener A, Malaisse WJ. Electrical activity in pancreatic islet cells: The VRAC hypothesis. Islets. 2010:2. doi: 10.4161/isl.2.2.11171. [DOI] [PubMed] [Google Scholar]
- Best L, Brown PD, Tomlinson S. Anion fluxes, volume regulation and electrical activity in the mammalian pancreatic beta-cell. Exp Physiol. 1997;82:957–966. doi: 10.1113/expphysiol.1997.sp004081. [DOI] [PubMed] [Google Scholar]
- CDC . Number (in Millions) of Civilian, Noninstitutionalized Persons with Diagnosed Diabetes, United States, 1980–2011. 2013. [Google Scholar]
- Chen W-S, Brayman AA, Matula TJ, Crum LA, Miller MW. The pulse length-dependence of inertial cavitation dose and hemolysis. Ultrasound Med Biol. 2003;29:739–748. doi: 10.1016/s0301-5629(03)00029-2. [DOI] [PubMed] [Google Scholar]
- Christensen D. Ultrasonic Bioinstrumentation. John Wiley & Sons, Inc.; United States: 1988. [Google Scholar]
- Dinno MA, Dyson M, Young SR, Mortimer AJ, Hart J, Crum LA. The significance of membrane changes in the safe and effective use of therapeutic and diagnostic ultrasound. Phys Med Biol. 1989;34:1543–1552. doi: 10.1088/0031-9155/34/11/003. [DOI] [PubMed] [Google Scholar]
- Eliasson L, Proks P, Ammala C, Ashcroft FM, Bokvist K, Renstrom E, Rorsman P, Smith PA, Ammälä C, Ashcroft FM, Bokvist K, Renström E, Rorsman P, Smith PA. Endocytosis of secretory granules in mouse pancreatic beta-cells evoked by transient elevation of cytosolic calcium. J Physiol. 1996;493:755–767. doi: 10.1113/jphysiol.1996.sp021420. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8799897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrannini E, Mari A. Beta cell function and its relation to insulin action in humans: a critical appraisal. Diabetologia. 2004;47:943–956. doi: 10.1007/s00125-004-1381-z. [DOI] [PubMed] [Google Scholar]
- Festa A, Williams K, Hanley AJG, Haffner SM. β-cell dysfunction in subjects with impaired glucose tolerance and early type 2 diabetes comparison of surrogate markers with first-phase insulin secretion from an intravenous glucose tolerance test. Diabetes Am Diabetes Assoc. 2008;57:1638–1644. doi: 10.2337/db07-0954. [DOI] [PubMed] [Google Scholar]
- Fujii Y, Taniguchi N, Satoh M, Irie T, Itoh K. Effect of repeated US stimulation on adiponectin secretion by adipocytes of obese human subjects. AIP Proc 5th Int Symp Ther Ultrasound. 2006:64–69. [Google Scholar]
- Guzmán HR, Nguyen DX, McNamara AJ, Prausnitz MR. Equilibrium loading of cells with macromolecules by ultrasound: Effects of molecular size and acoustic energy. J Pharm Sci. 2002;91:1693–1701. doi: 10.1002/jps.10156. [DOI] [PubMed] [Google Scholar]
- Hassan MA, Campbell P, Kondo T. The role of Ca 2+ in ultrasound-elicited bioeffects: progress, perspectives and prospects. Drug Discov Today Elsevier. 2010;15:892–906. doi: 10.1016/j.drudis.2010.08.005. [DOI] [PubMed] [Google Scholar]
- Helen ME, Sheader EA, Brown PD, Best L. Glucose-induced swelling in rat pancreatic B-cells. Mol Cell Endocrinol. 2007;264:61–67. doi: 10.1016/j.mce.2006.10.005. [DOI] [PubMed] [Google Scholar]
- Henquin JC. Regulation of insulin secretion: A matter of phase control and amplitude modulation. Diabetologia. 2009;52:739–751. doi: 10.1007/s00125-009-1314-y. [DOI] [PubMed] [Google Scholar]
- Hensel K, Mienkina MP, Schmitz G. Analysis of ultrasound fields in cell culture wells for in vitro ultrasound therapy experiments. Ultrasound Med Biol. 2011;37:2105–2115. doi: 10.1016/j.ultrasmedbio.2011.09.007. [DOI] [PubMed] [Google Scholar]
- Hohmeier HE, Mulder H, Chen G, Henkel-rieger R, Prentki M, Newgard CB. Isolation of INS-1– Derived Cell Lines With Robust ATP-Sensitive K + Channel-Dependent and -Independent Glucose-Stimulated Insulin Secretion. 2000:49. doi: 10.2337/diabetes.49.3.424. [DOI] [PubMed] [Google Scholar]
- Honda H, Kondo T, Zhao Q-L, Feril LB, Kitagawa H. Role of intracellular calcium ions and reactive oxygen species in apoptosis induced by ultrasound. Ultrasound Med Biol. 2004;30:683–692. doi: 10.1016/j.ultrasmedbio.2004.02.008. [DOI] [PubMed] [Google Scholar]
- Hong Chen TL, Khokhlova T, Wang Y-N, Kreider W, He X, Hwang JH. Passive cavitation detection during pulsed HIFU exposures of ex vivo tissues and in vivo mouse pancreatic tumors. Ultrasound Med Biol. 2014;40:1523–1534. doi: 10.1016/j.ultrasmedbio.2014.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu S, Huang T. Bioeffect of ultrasound on endothelial cells in vitro. Biomol Eng Elsevier. 2004;21:99–104. doi: 10.1016/j.bioeng.2004.06.001. [DOI] [PubMed] [Google Scholar]
- Hu Y, Wan JMF, Yu ACH. Cytomechanical Perturbations during Low-Intensity Ultrasound Pulsing. Ultrasound Med Biol. 2014;40:1587–1598. doi: 10.1016/j.ultrasmedbio.2014.01.003. [DOI] [PubMed] [Google Scholar]
- Hu Y, Zhong W, Wan JMF, Yu ACH. Ultrasound can Modulate Neuronal Development: Impact on Neurite Growth and Cell Body Morphology. Ultrasound Med Biol. 2013;39:915–925. doi: 10.1016/j.ultrasmedbio.2012.12.003. [DOI] [PubMed] [Google Scholar]
- Israili ZH. Advances in the treatment of type 2 diabetes mellitus. Am J Ther. 2011;18:117–152. doi: 10.1097/MJT.0b013e3181afbf51. [DOI] [PubMed] [Google Scholar]
- Kaelin D, Renold AE, Sharp GWG. Glucose stimulated proinsulin biosynthesis. Diabetologia. 1978;14:329–335. doi: 10.1007/BF01223025. Available from: http://dx.doi.org/10.1007/BF01223025. [DOI] [PubMed] [Google Scholar]
- Kahn SE. Beta cell failure: causes and consequences. Int J Clin Pract Suppl. 2001:13–18. [PubMed] [Google Scholar]
- Karshafian R, Bevan PD, Williams R, Samac S, Burns PN. Sonoporation by ultrasound-activated microbubble contrast agents: Effect of acoustic exposure parameters on cell membrane permeability and cell viability. Ultrasound Med Biol. 2009;35:847–860. doi: 10.1016/j.ultrasmedbio.2008.10.013. [DOI] [PubMed] [Google Scholar]
- Krasovitski B, Frenkel V, Shoham S, Kimmel E. Intramembrane cavitation as a unifying mechanism for ultrasound-induced bioeffects. Proc Natl Acad Sci U S A. 2011;108:3258–3263. doi: 10.1073/pnas.1015771108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumon RE, Aehle M, Sabens D, Parikh P, Han YW, Kourennyi D, Deng CX. Spatiotemporal Effects of Sonoporation Measured by Real-Time Calcium Imaging. Ultrasound Med Biol. 2009;35:494–506. doi: 10.1016/j.ultrasmedbio.2008.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leighton TG. The Acoustic Bubble. United States: Academic Press; 1994. [Google Scholar]
- Leslie T a Kennedy JE. High intensity focused ultrasound in the treatment of abdominal and gynaecological diseases. Int J Hyperthermia. 2007;23:173–182. doi: 10.1080/02656730601150514. [DOI] [PubMed] [Google Scholar]
- Morris CE, Juranka PF. Nav Channel Mechanosensitivity : Activation and Inactivation Accelerate Reversibly with Stretch. Biophys J Elsevier. 2007;93:822–833. doi: 10.1529/biophysj.106.101246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mortimer a. J, Dyson M. The effect of therapeutic ultrasound on calcium uptake in fibroblasts. Ultrasound Med Biol. 1988;14:499–506. doi: 10.1016/0301-5629(88)90111-1. [DOI] [PubMed] [Google Scholar]
- Nabili M, Geist C, Zderic V. Thermal safety of ultrasound-enhanced ocular drug delivery: A modeling study. Med Phys American Association of Physicists in Medicine. 2015;42:5604–5615. doi: 10.1118/1.4929553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neumiller JJ, Setter SM. Pharmacologic management of the older patient with type 2 diabetes mellitus. Am J Geriatr Pharmacother Excerpta Medica Inc. 2009;7:324–342. doi: 10.1016/j.amjopharm.2009.12.002. [DOI] [PubMed] [Google Scholar]
- Olofsson CS, Göpel SO, Barg S, Galvanovskis J, Ma X. Fast insulin secretion reflects exocytosis of docked granules in mouse pancreatic B-cells. 2002:43–51. doi: 10.1007/s00424-002-0781-5. [DOI] [PubMed] [Google Scholar]
- Orci L, Malaisse-Lagae F, Ravazzola M, Amherdt M, Renold AE. Exocytosis-Endocytosis Coupling in the Pancreatic Beta Cell. Science (80-) 1973;181:561–562. doi: 10.1126/science.181.4099.561. [DOI] [PubMed] [Google Scholar]
- Rabkin BA, Zderic V, Vaezy S. Hyperecho in ultrasound images of HIFU therapy: Involvment of cavitation. Ultrasound Med Biol. 2005;31:947–956. doi: 10.1016/j.ultrasmedbio.2005.03.015. [DOI] [PubMed] [Google Scholar]
- Raskin P. Why insulin sensitizers but not secretagogues should be retained when initiating insulin in type 2 diabetes. Diabetes Metab Res Rev. 2010;26:446–447. doi: 10.1002/dmrr.783. [DOI] [PubMed] [Google Scholar]
- Robinson IM, Kinnick RR, Greenleaf JF, Fernandez JM. Stimulation of secretion from bovine adrenal chromaffin cells by microsecond bursts of therapeutic levels of ultrasound. J Physiol. 1996;492(Pt 1):257–263. doi: 10.1113/jphysiol.1996.sp021306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rorsman P, Renström E. Insulin granule dynamics in pancreatic beta cells. Diabetologia. 2003;46:1029–1045. doi: 10.1007/s00125-003-1153-1. [DOI] [PubMed] [Google Scholar]
- Sakurada M, Kanatsuka A, Saitoh T, Makino H, Yamamura K, Miyazaki J-I, Kikuchi M, Yoshida S. Relation between Glucose-Stimulated Superfusion Pancreatic. Endocrinology. 1993:132. doi: 10.1210/endo.132.6.8504766. [DOI] [PubMed] [Google Scholar]
- Schlicher RK, Radhakrishna H, Tolentino TP, Apkarian RP, Zarnitsyn V, Prausnitz MR. Mechanism of intracellular delivery by acoustic cavitation. Ultrasound Med Biol. 2006;32:915–924. doi: 10.1016/j.ultrasmedbio.2006.02.1416. [DOI] [PubMed] [Google Scholar]
- Semino MC, Gagliardino AM, Bianchi C, Rebolledo OR, Gagliardino JJ. Early changes in the rat pancreatic B cell size induced by glucose. Cells Tissues Organs Karger Publishers. 1990;138:293–296. doi: 10.1159/000146958. [DOI] [PubMed] [Google Scholar]
- Spellman CW. Islet cell dysfunction in progression to diabetes mellitus. J Am Osteopath Assoc. 2007;107(Suppl):S1–S5. [PubMed] [Google Scholar]
- Suarez Castellanos IM, Balteanu B, Singh T, Zderic V. Therapeutic Modulation of Calcium Dyanamis Using Ultrasound and Other Energy-Based Techniques. IEEE Rev Biomed Eng. 2016;9:177–191. doi: 10.1109/RBME.2016.2555760. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22435804. [DOI] [PubMed] [Google Scholar]
- Takii M, Ishikawa T, Tsuda H, Kanatani K, Sunouchi T, Kaneko Y, Nakayama K, Na K. Involvement of stretch-activated cation channels in hypotonically induced insulin secretion in rat pancreatic B-cells. Am J Physiol - Cell Physiol. 2006;291:1405–1411. doi: 10.1152/ajpcell.00519.2005. [DOI] [PubMed] [Google Scholar]
- Tennant JR. Evaluation of the Trypan Blue Technique for Determination of Cell Viability. Transplantation. 1964:685–694. doi: 10.1097/00007890-196411000-00001. [DOI] [PubMed] [Google Scholar]
- Tsukamoto A, Higashiyama S, Yoshida K, Watanabe Y, Furukawa KS, Ushida T. Stable cavitation induces increased cytoplasmic calcium in L929 fibroblasts exposed to 1-MHz pulsed ultrasound. Ultrasonics Elsevier. 2011;51:982–990. doi: 10.1016/j.ultras.2011.05.014. [DOI] [PubMed] [Google Scholar]
- Tyler WJ, Tufail Y, Finsterwald M, Tauchmann ML, Olson EJ, Majestic C. Remote excitation of neuronal circuits using low-intensity, low-frequency ultrasound. PLoS One. 2008:3. doi: 10.1371/journal.pone.0003511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wheeler B, Sheu L, Beaudoin R, Bennett MK, Gaisanot Y, G DBG, Sherbrooke U De. Characterization of SNARE protein expression in beta cell lines and pancreatic islets. Endocrinology. 2015;137:1340–8. doi: 10.1210/endo.137.4.8625909. [DOI] [PubMed] [Google Scholar]
- Wild S, Roglic G, Green A, Sicree R, King H. Estimates for the year 2000 and projections for 2030. World Health. 2004;27:1047–1053. doi: 10.2337/diacare.27.5.1047. [DOI] [PubMed] [Google Scholar]
- Zderic V, Clark JI, Martin RW, Vaezy S. Transcorneal drug delivery using 880 KHz ultrasound. Cornea. 2004a;23:804–811. doi: 10.1097/01.ico.0000134189.33549.cc. [DOI] [PubMed] [Google Scholar]
- Zderic V, Clark JI, Vaezy S. Drug Delivery Into the Eye. J Ultrasound Med. 2004b;23:1349–1359. doi: 10.7863/jum.2004.23.10.1349. [DOI] [PubMed] [Google Scholar]
- Zhao H, Yang G, Wang D, Yu X, Zhang Y, Zhu J, Ji Y, Zhong B, Zhao W, Yang Z, Aziz F. Concurrent gemcitabine and high-intensity focused ultrasound therapy in patients with locally advanced pancreatic cancer. Anticancer Drugs. 2010;21:447–452. doi: 10.1097/CAD.0b013e32833641a7. [DOI] [PubMed] [Google Scholar]
- Zimmet P, Alberti KGMM, Shaw J. Diabetes Epidemic. Nature. 2001;414:782–787. doi: 10.1038/414782a. [DOI] [PubMed] [Google Scholar]