Abstract
Left ventricular (LV) remodeling after tetralogy of Fallot (TOF) repair may influence LV stiffness. We hypothesized that LV stiffness is altered after TOF repair and related to myocardial calibrated integrated backscatter (cIB) and LV diastolic myocardial deformation. Seventy-seven TOF patients and 80 controls were studied. LV stiffness was assessed by diastolic wall strain (DWS) as defined by (LVPWsystole-LVPWdiastole)/LVPWsystole, where LVPW is LV posterior wall thickness, and stiffness index as defined by (E/e/LV end-diastolic dimension), where E and e are respectively early diastolic transmitral inflow and mitral annular velocities. Septal and LVPW cIB and LV diastolic strain rates were determined. Patients had significantly lower DWS (p < 0.001), higher stiffness index (p < 0.001), and greater cIB (p < 0.001). LV DWS correlated negatively with LV stiffness index (r = −0.31, p < 0.001), septal cIB (r = −0.21, p = 0.01), E/e ratio (r = −0.30, p < 0.001) and RV end-diastolic area (r = −0.31, p < 0.001), and positively with LV early (r = 0.33, p < 0.001) and late (r = 0.20, p = 0.01) diastolic strain rates and RV fractional area change (FAC) (r = 0.24, p = 0.003). Multivariate analysis revealed E/e (β = −0.26, p = 0.008), RV end-diastolic area (β = −0.20, p = 0.02), and RV FAC (β = 0.18, p = 0.01) as significant correlates of DWS. Left ventricular stiffening occurs after TOF repair and is related to impaired LV diastolic myocardial deformation, myocardial cIB, and RV volume overload.
Introduction
Right ventricular (RV) dilation and dysfunction secondary to chronic severe pulmonary regurgitation are well documented in patients late after repair of tetralogy of Fallot (TOF)1–3. On the other hand, increasing evidence suggests that left ventricular (LV) dysfunction may also have prognostic significance in these patients4. Left ventricular systolic dysfunction has been attributed to several factors including preoperative hypoxaemia5, LV fibrosis6, 7, LV dyssynchrony8 and adverse right-left ventricular interaction9. On the other hand, the phenotype and understanding of pathogenetic mechanism of LV diastolic dysfunction in repaired TOF is less clear.
Diastolic ventricular dysfunction may be related to relaxation abnormality and/or stiffening of the myocardium. Previous studies in repaired TOF patients have primarily assessed indirectly LV early diastolic relaxation and estimated late diastolic filling using Doppler imaging10, 11. Importantly, however, recent data suggest that potential alteration of myocardial substrates occurs in repaired TOF, which may predispose to stiffening of LV myocardium. In these patients, increased levels of circulating biomarkers of collagen synthesis have been reported12, 13. Cardiac magnetic resonance (CMR) imaging with late gadolinium enhancement6 and T1 mapping7 techniques has further revealed evidence of LV fibrosis. The potential consequence on LV stiffness of LV remodeling related to myocardial fibrosis and geometric eccentricity secondary to RV volume overload has, however, not been explored in patients after TOF repair.
Recently, there has been increasing use of echocardiography for non-invasive assessment of myocardial stiffness. The parameter of diastolic wall strain (DWS)14, which quantifies the thinning of myocardium during diastole, has been found to be useful in assessing diastolic myocardial stiffness and prognostication in heart failure patients with preserved ejection fraction15. Another potentially useful LV stiffness index that relates Doppler-estimated LV filling pressure to LV end-diastolic dimension has been used to interrogate myocardial stiffness in athletes16. In this study we aimed to utilize these indices to test the hypothesis that LV stiffness is altered in adolescents and young adults with repaired TOF and to determine its relationship to myocardial calibrated integrated backscatter (cIB) as a marker of fibrosis17 and LV diastolic myocardial deformation.
Methods
Subjects
This was a retrospective study of our echocardiographic database of congenital heart disease that included 77 consecutively studied TOF patients post total surgical repair. The following patient data were retrieved from case notes: cardiac and associated lesions, type of operation performed, date and age of operation, and the need for additional procedures. A total of 80 age-matched healthy controls were retrieved from the healthy control database for the purpose of comparisons. These included healthy staff volunteers, their friends, and subjects with chest pain or palpitation for which no organic causes had been identified. The body weight and height of all subjects were measured at the time of echocardiographic acquisitions and the body surface area was calculated accordingly. The total number of around 80 subjects in each group gave a statistical power of 80% with 5% level of significance with two-sided testing to detect a minimum DWS difference of 0.03 with a variation of 0.08 based on the previously reported standard deviation15. This study was approved by the Institutional Review Board of the University of Hong Kong/Hospital Authority Hong Kong West Cluster, Hong Kong, and all of the methods as described were performed in accordance with the approved guidelines and regulations. Informed consent had been obtained from all of the participants at the time of acquisition of echocardiographic images.
Conventional echocardiographic assessment
Echocardiographic acquisitions were made using Vivid 7 ultrasound machine (General Electric, Vingmed, Horten, Norway). Offline analyses of the recordings were performed using EchoPAC software (General Electric, Vingmed, Horten, Norway). Measurements of all echocardiographic parameters were made in three cardiac cycles and the average was taken for statistical analyses.
From the four-chamber view, RV end-diastolic and end-systolic areas were measured and RV fractional area change was calculated accordingly. Transmitral pulsed-wave Doppler examination was performed to obtain peak early diastolic inflow velocity (E), late diastolic inflow velocity (A), E/A ratio, and E deceleration time. Tissue Doppler echocardiography was performed with sample volume positioned at the basal LV free wall-mitral annular junction to obtain the peak early diastolic myocardial tissue velocity (e), late diastolic myocardial tissue velocity (a), e/a ratio, and E/e ratio. Severity of pulmonary regurgitation was graded semi-quantitatively as mild, moderate, or severe by color flow mapping18.
Assessment of LV stiffness
Based on M-mode assessment of the parasternal short-axis view, the LV posterior wall (LVPW) thickness at systole and diastole was determined. Diastolic wall strain (DWS) was calculated as (LVPW at systole - LVPW at diastole)/LVPW at systole14. This parameter reflected thinning of the LV posterior wall during diastole. Based on the linear elastic theory, this change in wall thickness reflected resistance to deformation in diastole and hence myocardial stiffness14. Our group had previously reported on high reproducibility of DWS measurements19.
Stiffness index was calculated as (E/e)/LVEDd, where LVEDd is LV end-diastolic dimension derived from M-mode measurement16. This parameter was used to provide an estimate of the pressure-to-volume relationship, with LV filling measure being estimated by E/e ratio and end-diastolic LV volume being estimated by LVEDd.
Measurement of cIB
Integrated backscatter of the ventricular septum and posterior LV wall was measured from the parasternal short-axis view at the papillary muscle level at end-diastole as described previously20. The sample volume was tracked manually to maintain the same region throughout the cardiac cycle. Calibrated integrated backscatter was calculated as the difference between integrated backscatter at the two regions and that at the pericardium. The average cIB at the two sites was taken for statistical analysis. Our group has previously reported on high reproducibility of cIB measurement20.
Quantification of LV diastolic myocardial deformation
Left ventricular diastolic myocardial deformation in the longitudinal dimension was interrogated using speckle tracking echocardiography as reported previously21. By tracking the entire LV contour, the global LV longitudinal early and late diastolic strain rates were determined from the apical four chamber view using EchoPAC software (GE Medical Systems).
Statistical analysis
Data was expressed as mean ± standard deviation. Differences in demographic and echocardiographic parameters between repaired TOF patients and controls were compared using the Student’s t-test and Fisher’s exact test where appropriate. Relationships between LV stiffness indices and cIB and indices of LV diastolic deformation were explored using Pearson correlation analysis. Multivariate analysis using multiple linear regression was performed to determine significant correlates of DWS and stiffness index respectively with adjustments of variables having a p value < 0.05 by univariate analysis. A p value < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS version 17.0 (SPSS, Inc., Chicago, IL, USA).
Results
Subjects
Of the 77 patients, 72 had TOF with pulmonary stenosis, while 5 had pulmonary atresia. The patients (43 males) were aged 18.5 ± 8.1 years and studied at 14.6 ± 7.3 years after total surgical repair. Twenty patients had systemic-to-pulmonary arterial shunt palliation before total surgical repair. Of the 72 TOF patients with pulmonary stenosis, 58 required transannular patch repair of RV outflow. Pulmonary valve replacement was performed subsequently in four of the 77 patients. Six of the patients had syndromal associations including DiGeorge syndrome in 3, Down syndrome in 1, trisomy 20p in 1, and VATER association in 1. The 80 controls (41 males) were aged 16.5 ± 7.0 years (p = 0.11). Compared with controls, patients had similar body weight (46.2 ± 16.3 kg vs 49.9 ± 16.3 kg, p = 0.18), body height (152.0 ± 17.7 cm vs 155.0 ± 22.0 cm, p = 0.35), and body surface area (1.38 ± 0.33 m2 vs 1.46 ± 0.34 m2, p = 0.14).
Conventional and speckle tracking echocardiographic findings
Table 1 summarized the echocardiographic findings in patients and controls. The RV end-diastolic area indexed by body surface area was significantly greater in patients than controls (p < 0.001), although there was no significant difference in RV fractional area change between the two groups (p = 0.14). Of 77 repaired TOF patients, 11 (14.3%), 14 (18.2%) and 52 (67.5%) had respectively mild, moderate and severe degree of pulmonary regurgitation.
Table 1.
Echocardiographic indices in patients and controls.
| Patients (n = 77) | Controls (n = 80) | p | |
|---|---|---|---|
| Mitral inflow Doppler indices | |||
| E (cm/s) | 97.6 ± 21.7 | 97.8 ± 22.5 | 0.94 |
| A (cm/s) | 44.7 ± 14.8 | 50.8 ± 14.3 | 0.01* |
| E/A ratio | 2.37 ± 0.85 | 2.02 ± 0.51 | 0.002* |
| E deceleration time (ms) | 155 ± 52 | 147 ± 34 | 0.28 |
| Mitral annular tissue Doppler | |||
| e (cm/s) | 15.0 ± 3.71 | 17.5 ± 2.5 | <0.001* |
| a (cm/s) | 6.0 ± 2.0 | 6.6 ± 1.5 | 0.02* |
| e/a ratio | 2.70 ± 1.03 | 2.75 ± 0.72 | 0.71 |
| E/e ratio | 6.95 ± 2.41 | 5.66 ± 1.30 | <0.001* |
| LV longitudinal diastolic deformation | |||
| SRe (/s) | 1.52 ± 0.45 | 2.05 ± 0.52 | <0.001* |
| SRa (/s) | 0.59 ± 0.19 | 0.72 ± 0.19 | <0.001* |
| 2D echocardiographic measurements | |||
| RV EDA (cm2/m2) | 17.6 ± 4.7 | 9.7 ± 2.1 | <0.001* |
| RV FAC (%) | 54.4 ± 9.1 | 56.4 ± 7.0 | 0.14 |
| LVEDd (mm) | 39.8 ± 7.8 | 43.2 ± 5.4 | 0.002* |
A, transmitral late diastolic inflow velocity; a, late diastolic annular myocardial velocity; E, transmitral early diastolic inflow velocity; e, early diastolic annular myocardial velocity; FAC, fractional area change; LV, left ventricular; LVEDd, left ventricular end-diastolic dimension; RV, right ventricular; SRa, left ventricular late diastolic strain rate; RV EDA, RV end diastolic area; SRe, left ventricular early diastolic strain rate. *Statistically significant.
Worse LV diastolic mechanics in repaired TOF patients was evidenced by the significantly lower e velocity (p < 0.001) and global longitudinal early (p < 0.001) and late (p < 0.001) diastolic strain rates, and greater E/e ratio (p < 0.001) in patients as compared with those of controls.
LV stiffness and cIB
Compared with controls, TOF patients had significantly lower DWS (0.38 ± 0.10 vs 0.47 ± 0.08, p < 0.001) and higher LV stiffness index (1.82 ± 0.71 vs 1.34 ± 0.38, p < 0.001) (Fig. 1A,B). For the entire cohort, DWS was found to correlate negatively with LV stiffness index (r = −0.31, p < 0.001).
Figure 1.
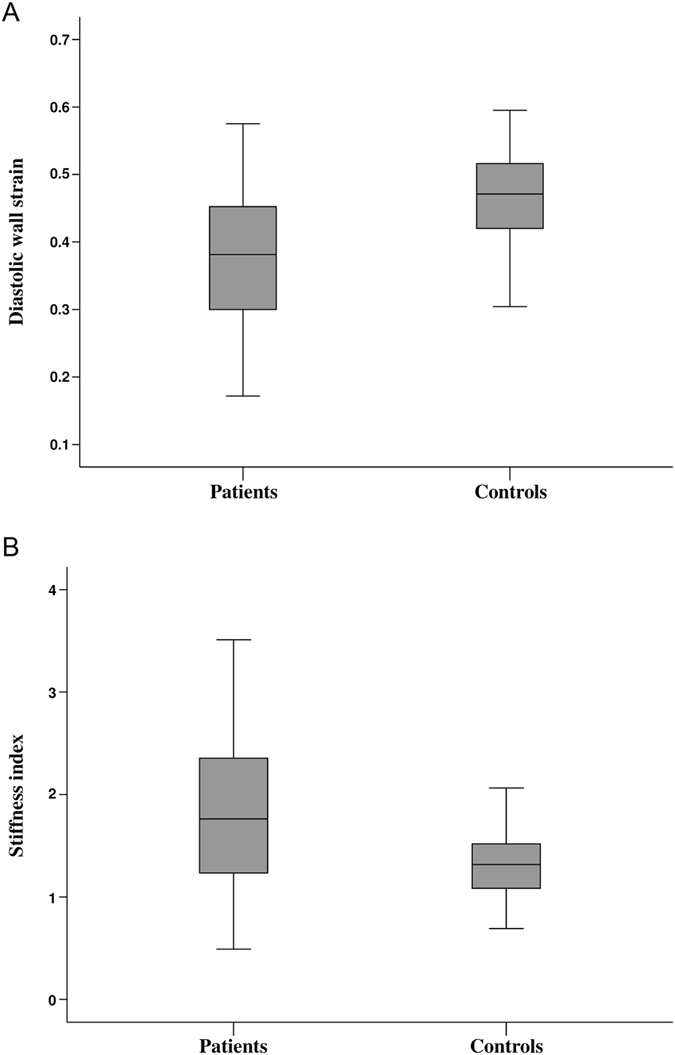
Boxplots showing (A) diastolic wall strain and (B) stiffness index of the left ventricle in patients and controls.
For cIB, patients had significantly greater septal cIB (−19.4 ± 6.6 dB vs −24.9 ± 5.3 dB, p < 0.001), LV posterior wall cIB (−19.4 ± 4.8 dB vs −21.3 ± 4.6 dB, p = 0.01) and average cIB (−19.4 ± 4.7 dB vs −23.1 ± 4.2 dB, p < 0.001).
The septal but not posterior wall cIB was found to correlate negatively with LV DWS (r = −0.21, p = 0.01). Additionally, cIB at septum was found to correlate with mitral annular e velocity (r = −0.17, p = 0.03), LV early diastolic longitudinal strain rate (r = −0.29, p < 0.001), RV volume load (r = 0.24, p = 0.002), and RV fractional area change (r = −0.21, p = 0.007). These were, however, no significant correlation between septal or posterior cIB and LV stiffness index.
Correlates of LV stiffness
Table 2 summarized the correlation analyses of LV DWS and stiffness index. Left ventricular DWS was found to correlate negatively with E/e ratio (p < 0.001) and RV end-diastolic area (p < 0.001), and positively with e velocity (p < 0.001), LV global early (p < 0.001) and late (p = 0.01) diastolic longitudinal strain rates, and RV fractional area change (p = 0.003) (Fig. 2A–D). Multivariate analysis identified E/e ratio (p = 0.008), RV end-diastolic dimension (p = 0.02), and RV fractional area change (p = 0.01) as significant independent correlates of DWS.
Table 2.
Correlation between left ventricular stiffness and indices of left and right ventricular function.
| Diastolic wall strain | Stiffness index | |||||||
|---|---|---|---|---|---|---|---|---|
| Univariate correlation | Multiple regression | Univariate correlation | Multiple regression | |||||
| r | p | beta | p | r | p | beta | p | |
| Mitral inflow Doppler indices | ||||||||
| E | −0.04 | 0.66 | 0.55 | <0.001* | 0.02 | 0.57 | ||
| A | 0.07 | 0.36 | 0.13 | 0.10 | ||||
| E/A ratio | −0.08 | 0.32 | 0.34 | <0.001* | −0.01 | 0.57 | ||
| E deceleration time | −0.05 | 0.56 | 0.09 | 0.28 | ||||
| Mitral annular tissue Doppler | ||||||||
| e | 0.31 | <0.001* | −0.03 | 0.78 | −0.60 | <0.001* | 0.03 | 0.52 |
| a | 0.16 | 0.045* | 0.05 | 0.54 | −0.30 | <0.001* | −0.04 | 0.26 |
| e/a | 0.10 | 0.22 | −0.19 | 0.02* | −0.05 | 0.15 | ||
| E/e ratio | −0.30 | <0.001* | −0.26 | 0.008* | 0.90 | <0.001* | 0.80 | <0.001* |
| LV longitudinal diastolic deformation | ||||||||
| SRe | 0.33 | <0.001* | 0.16 | 0.11 | 0.02 | 0.83 | ||
| SRa | 0.20 | 0.01* | 0.01 | 0.90 | −0.13 | 0.11 | ||
| 2D echocardiographic measurements | ||||||||
| Indexed RV EDA | −0.31 | <0.001* | −0.20 | 0.02* | 0.32 | <0.001* | 0.43 | 0.004* |
| RV FAC | 0.24 | 0.003* | 0.18 | 0.01* | 0.01 | 0.88 | ||
| LVEDd | 0.11 | 0.16 | −0.64 | <0.001* | −0.40 | <0.001* | ||
Abbreviation as in Table 1. *Statistically significant.
Figure 2.
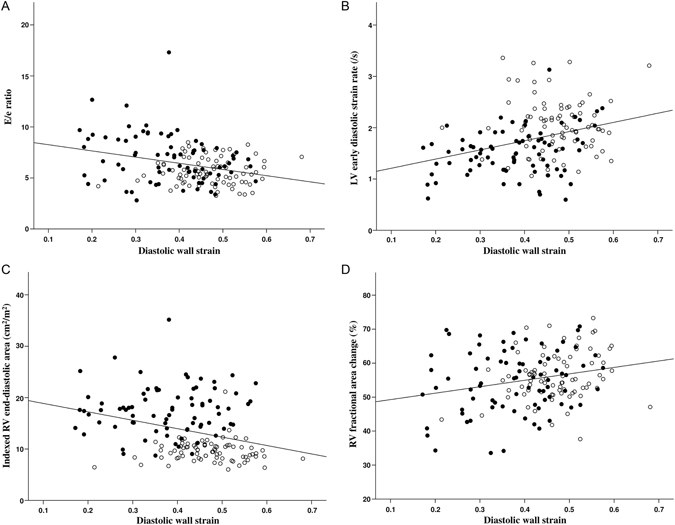
Scatterplots showing the relationships between left ventricular diastolic wall strain and (A) the ratio of transmitral early diastolic flow velocity (E) to mitral annular early diastolic velocity (e), (B) left ventricular (LV) early diastolic strain rate, (C) indexed right ventricular (RV) end-diastolic area, and (D) RV fractional area change. Solid circles represent patients, empty circles represent controls.
On the other hand, the stiffness index expectedly was found to correlate with LV end-diastolic dimension (p < 0.001), transmitral E velocity (p < 0.001), mitral annular e velocity (p < 0.001), and E/e ratio (p < 0.001) based on which the parameter was derived. Additionally, stiffness index was found to correlate positively with RV end-diastolic area (r = 0.32, p < 0.001). Multivariate analysis identified RV end-diastolic area (p = 0.004), LV end-diastolic dimension (p < 0.001) and E/e ratio (p < 0.001) as significant independent correlates.
In patients, there were no significant correlations between severity of pulmonary regurgitation and DWS and stiffness index (both p > 0.05).
Discussion
The present study demonstrates diastolic myocardial stiffening as reflected by reduced LV diastolic wall strain and increased stiffness index in adolescents and young adults with repaired TOF. Increased myocardial stiffness was associated with worse LV diastolic myocardial deformation as characterized by slower global LV early and late diastolic longitudinal strain rates. Furthermore, our data provided evidence that myocardial fibrosis and RV volume overload might be important factors that contributed to LV stiffening late after repair of TOF. To our knowledge, this is the first study to explore LV stiffening and its possible associations with myocardial fibrosis, RV volume load, and LV diastolic myocardial deformation.
There has been increasing interest on ventricular diastolic dysfunction in patients with repaired TOF. Previous studies have focused on interrogation of LV relaxation abnormalities10, 11. Our findings of reduced mitral annular e velocity and global LV longitudinal diastolic strain rate are compatible with impaired LV relaxation and agree with those reported previously11, 12. Increased LV end-diastolic pressure as documented by cardiac catheterization in repaired TOF patients, on the other hand, provides evidence of reduced LV compliance22, although data on stiffening of ventricular myocardium in these patients are lacking.
Direct assessment of LV stiffness is challenging and requires invasive cardiac catheterization for the derivation of the diastolic pressure-volume curve and calculation of chamber stiffness constant23. Recently, the echocardiographic parameter of DWS, which reflects the change of wall thickness and resistance to deformation during diastole, has been validated in animal studies and shown to correlate inversely with the invasively determined myocardial stiffness constant14. In clinical studies, DWS has been shown to predict outcomes of patient with heart failure with preserved15 and reduced24 ejection fraction and in patients with non-ST elevation myocardial infarction25. With regard to the LV stiffness index, which provides an estimate of the relationship between LV filling pressure and end-diastolic volume, this has been reported to be useful in the demonstration of reduced myocardial stiffness in elite athletes16. Our findings of reduced DWS and concomitant increased LV stiffness index in patients with repaired TOF provide therefore evidence of LV myocardial stiffening. Importantly, the magnitude of LV myocardial stiffening was found to relate inversely to indices of LV diastolic deformation. Hence, LV diastolic dysfunction in patients with repaired TOF is attributable in part to increased stiffness of the ventricular myocardium.
While the exact mechanisms of LV myocardial stiffening in our patients remain to be unveiled, the present study suggests that two factors may potentially be important. Firstly, increased cIB of the ventricular septum and LV posterior in our patients suggests that alteration of myocardial substrate with increased deposition of fibrous tissue might be a culprit. Calibrated integrated backscatter has been used to assess myocardial fibrosis in obese subjects26, in patients with metabolic syndrome27, and in those with heart failure28. Our finding of increased cIB in repaired TOF patients are compatible with previous reports of increased levels of circulating biomarkers of collagen synthesis12, 13 and CMR late gadolinium enhancement6 and T1 mapping7 studies demonstrating evidence of LV fibrosis in these patients. Fibrosis increases the viscoelastic burden of the myocardium and impairs relaxation, diastolic suction, and passive stiffness29. Indeed, significant associations between myocardial fibrosis and worsening of LV stiffness have been shown in heart transplant recipients30 and in patients with heart failure with preserved ejection fraction31.
Intriguingly, we found significant negative correlation between DWS and cIB of the septum but not posterior wall. Furthermore, the septal cIB difference between patients and controls was more significant than that of the LV posterior cIB difference. It is interesting to note that preferential fibrotic alteration of the septum has also been shown in patients with metabolic syndrome and LV dysfunction27 and in patients with heart failure with preserved ejection fraction28. In our patients, previous cardiopulmonary bypass and surgical closure of ventricular septal defect might have accounted for our findings although this remains speculative. Notwithstanding, preferential involvement of the septum has been shown to influence adversely the degree of LV diastolic dysfunction32.
The second potentially important factor that may contribute to LV stiffness in our patients is geometric alteration of the ventricles secondary to RV volume overload. Multivariate analysis has identified RV end-diastolic area and fractional area change as independent determinants of both DWS and stiffness index. Our hypothesis is further supported by the study of Schwartz et al. who found a significant relationship between RV end-diastolic volume and LV end-diastolic pressure22. Kim et al. have recently further reported using CMR LGE that non-ischaemic septal fibrosis is independently associated with RV chamber dilation and hypothesized the role of increased RV wall stress33. Additionally, reduced LV preload secondary to altered septal geometry may reduce ventricular compliance as reported similarly in patients with large atrial septal defects34.
While the prognostic implication of LV systolic dysfunction in repair TOF has been documented4, the clinical implication of LV diastolic dysfunction in repaired TOF is less clear. Nonetheless, several considerations warrant comments. Left ventricular dysfunction may impact adversely on pulmonary arterial compliance35, which in turn increases RV afterload and potentially worsens pulmonary regurgitation36. This is of particular relevance in the context of repaired TOF. Utilization of a semi-quantitative method rather than cardiac magnetic resonance to quantify pulmonary regurgitation in this study, however, might have precluded the identification of an association between LV myocardial stiffness and pulmonary regurgitation. Increased LV end-diastolic pressure of 12 mmHg or more has been associated with larger right ventricles22 and shown to be strong predictor of appropriate implantable cardioverter-defibrillator shocks in patients with repaired TOF37. The inclusion of LV diastolic dysfunction when considering the timing of pulmonary valve replacement and assessment of it reversibility after valve replacement should be topics for further research.
Several limitations to the study require comments. Firstly, this cross-sectional study cannot provide data on the relationship between progressive RV dilation and LV stiffening. Further longitudinal studies are required to address the serial changes in LV stiffness post TOF repair and its potential improvement after pulmonary valve replacement. Secondly, the measurement of DWS is regional, based on changes of the LV posterior wall thickness in the cardiac cycle. Heterogeneity of biomechanical properties in different myocardial regions may exist and further work to develop a more global echocardiographic index of myocardial stiffness is indicated. We have therefore also included the LV stiffness index, albeit based the estimates of LV filling pressure and size, to provide a more global assessment. It would have been ideal to be able to assess RV stiffness using similar echocardiographic measurements. However, the use of DWS to quantify RV stiffness has not been reported and yet to be validated. Finally, the use of the new CMR T1 mapping technique, which was not available at the time of study of our patients, may shed more light on the relationship between diffuse myocardial fibrosis and LV diastolic mechanics.
In conclusion, LV stiffening occurs late after repair TOF repair and is related to impaired LV diastolic myocardial deformation, myocardial calibrated integrated backscatter, and RV volume overload.
Author Contributions
C.K.M.Y. designed the study, performed echocardiographic evaluation, analyzed the data, and drafted the manuscript; W.H.S.W. assisted in statistical analysis; V.W.Y.L. assisted in the echocardiographic evaluation and data analysis; Y.F.C. was involved in the conception and design of the study, data interpretation, and finalizing the manuscript. All of the authors critically reviewed the final version of the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Gatzoulis MA, et al. Right and left ventricular systolic function late after repair of tetralogy of Fallot. Am J Cardiol. 2000;86:1352–1357. doi: 10.1016/S0002-9149(00)01241-8. [DOI] [PubMed] [Google Scholar]
- 2.D’Andrea A, et al. Right ventricular myocardial dysfunction in adult patients late after repair of tetralogy of fallot. Int J Cardiol. 2004;94:213–220. doi: 10.1016/j.ijcard.2003.04.033. [DOI] [PubMed] [Google Scholar]
- 3.Nair KK, et al. Asymptomatic right ventricular dysfunction in surgically repaired adult tetralogy of fallot patients. Ann Pediatr Cardiol. 2013;6:24–28. doi: 10.4103/0974-2069.107229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ghai A, et al. Left ventricular dysfunction is a risk factor for sudden cardiac death in adults late after repair of tetralogy of Fallot. J Am Coll Cardiol. 2002;40:1675–1680. doi: 10.1016/S0735-1097(02)02344-6. [DOI] [PubMed] [Google Scholar]
- 5.Hausdorf G, Hinrichs C, Nienaber CA, Schark C, Keck EW. Left ventricular contractile state after surgical correction of tetralogy of Fallot: risk factors for late left ventricular dysfunction. Pediatr Cardiol. 1990;11:61–68. doi: 10.1007/BF02239563. [DOI] [PubMed] [Google Scholar]
- 6.Babu-Narayan SV, et al. Ventricular fibrosis suggested by cardiovascular magnetic resonance in adults with repaired tetralogy of fallot and its relationship to adverse markers of clinical outcome. Circulation. 2006;113:405–413. doi: 10.1161/CIRCULATIONAHA.105.548727. [DOI] [PubMed] [Google Scholar]
- 7.Kozak MF, et al. Diffuse myocardial fibrosis following tetralogy of Fallot repair: a T1 mapping cardiac magnetic resonance study. Pediatr Radiol. 2014;44:403–409. doi: 10.1007/s00247-013-2840-9. [DOI] [PubMed] [Google Scholar]
- 8.Liang XC, Cheung EWY, Wong SJ, Cheung YF. Impact of right ventricular volume overload on three-dimensional global left ventricular mechanical dyssynchrony after surgical repair of tetralogy of Fallot. Am J Cardiol. 2008;102:1731–1736. doi: 10.1016/j.amjcard.2008.07.062. [DOI] [PubMed] [Google Scholar]
- 9.Davlouros PA, et al. Right ventricular function in adults with repaired tetralogy of Fallot assessed with cardiovascular magnetic resonance imaging: detrimental role of right ventricular outflow aneurysms or akinesia and adverse right-to-left ventricular interaction. J Am Coll Cardiol. 2002;40:2044–2052. doi: 10.1016/S0735-1097(02)02566-4. [DOI] [PubMed] [Google Scholar]
- 10.Aboulhosn JA, et al. Left and right ventricular diastolic function in adults with surgically repaired tetralogy of Fallot: a multi-institutional study. Can J Cardiol. 2013;29:866–872. doi: 10.1016/j.cjca.2012.11.003. [DOI] [PubMed] [Google Scholar]
- 11.Friedberg MK, et al. Impaired right and left ventricular diastolic myocardial mechanics and filling in asymptomatic children and adolescents after repair of tetralogy of Fallot. Eur Heart J Cardiovasc Imaging. 2012;13:905–913. doi: 10.1093/ehjci/jes067. [DOI] [PubMed] [Google Scholar]
- 12.Chen CA, et al. Circulating biomarkers of collagen type I metabolism mark the right ventricular fibrosis and adverse markers of clinical outcome in adults with repaired tetralogy of Fallot. Int J Cardiol. 2013;167:2963–2968. doi: 10.1016/j.ijcard.2012.08.059. [DOI] [PubMed] [Google Scholar]
- 13.Lai CT, Chan KW, Wong SJ, Chow PC, Cheung YF. Circulating levels of biomarkers of collagen synthesis and ventricular function and dyssynchrony in adolescents and young adults after repair of tetralogy of Fallot. Am Heart J. 2011;162:467–473. doi: 10.1016/j.ahj.2011.05.027. [DOI] [PubMed] [Google Scholar]
- 14.Takeda Y, et al. Noninvasive assessment of wall distensibility with the evaluation of diastolic epicardial movement. J Card Fail. 2009;15:68–77. doi: 10.1016/j.cardfail.2008.09.004. [DOI] [PubMed] [Google Scholar]
- 15.Ohtani T, et al. Diastolic stiffness as assessed by diastolic wall strain is associated with adverse remodelling and poor outcomes in heart failure with preserved ejection fraction. Eur Heart J. 2012;33:1742–1749. doi: 10.1093/eurheartj/ehs135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.King GJ, et al. Alterations in myocardial stiffness in elite athletes assessed by a new Doppler index. Heart. 2008;94:1323–1325. doi: 10.1136/hrt.2008.142083. [DOI] [PubMed] [Google Scholar]
- 17.Jellis C, Martin J, Narula J, Marwick TH. Assessment of nonischemic myocardial fibrosis. J Am Coll Cardiol. 2010;56:89–97. doi: 10.1016/j.jacc.2010.02.047. [DOI] [PubMed] [Google Scholar]
- 18.Zoghbi WA, et al. Recommendations for evaluation of the severity of native valvular regurgitation with two-dimensional and Doppler echocardiography. J Am Soc Echocardiogr. 2003;16:777–802. doi: 10.1016/S0894-7317(03)00335-3. [DOI] [PubMed] [Google Scholar]
- 19.Li, V. W. et al. Myocardial stiffness as assessed by diastolic wall strain in adult survivors of childhood leukaemias with preserved left ventricular ejection fraction. Eur Heart J Cardiovasc Imaging. pii, jew098 (2016). [DOI] [PubMed]
- 20.Ho PK, Lai CT, Wong SJ, Cheung YF. Three-dimensional mechanical dyssynchrony and myocardial deformation of the left ventricle in patients with tricuspid atresia after Fontan procedure. J Am Soc Echocardiogr. 2012;25:393–400. doi: 10.1016/j.echo.2012.01.003. [DOI] [PubMed] [Google Scholar]
- 21.Cheung EWY, Liang XC, Lam WW, Cheung YF. Impact of right ventricular dilation on left ventricular myocardial deformation in patients after surgical repair of tetralogy of fallot. Am J Cardiol. 2009;104:1264–1270. doi: 10.1016/j.amjcard.2009.06.043. [DOI] [PubMed] [Google Scholar]
- 22.Schwartz MC, et al. Relation of left ventricular end diastolic pressure to right ventricular end diastolic volume after operative treatment of tetralogy of Fallot. Am J Cardiol. 2012;109:417–422. doi: 10.1016/j.amjcard.2011.09.028. [DOI] [PubMed] [Google Scholar]
- 23.Zile MR, Baicu CF, Gaasch WH. Diastolic heart failure – Abnormalities in active relaxation and passive stiffness of the left ventricle. N Engl J Med. 2004;350:1953–1959. doi: 10.1056/NEJMoa032566. [DOI] [PubMed] [Google Scholar]
- 24.Soyama, Y. et al. Prognostic value of diastolic wall strain in patients with chronic heart failure with reduced ejection fraction. Heart and vessels. 1–8 (2016). [DOI] [PubMed]
- 25.Obasare E, et al. Diastolic wall strain predicts mortality in patients with non-ST elevation myocardial infarction. J Am Coll Cardiol. 2016;67:486. doi: 10.1016/S0735-1097(16)30487-9. [DOI] [Google Scholar]
- 26.Kosmala W, Przewlocka-Kosmala M, Szczepanik-Osadnik H, Mysiak A, Marwick TH. Fibrosis and cardiac function in obesity: a randomised controlled trial of aldosterone blockade. Heart. 2013;99:320–326. doi: 10.1136/heartjnl-2012-303329. [DOI] [PubMed] [Google Scholar]
- 27.Kosmala W, Przewlocka-Kosmala M, Wojnalowicz A, Mysiak A, Marwick TH. Integrated backscatter as a fibrosis marker in the metabolic syndrome: association with biochemical evidence of fibrosis and left ventricular dysfunction. Eur Heart J Cardiovasc Imaging. 2012;13:459–467. doi: 10.1093/ejechocard/jer291. [DOI] [PubMed] [Google Scholar]
- 28.Carluccio E, et al. Fibrosis assessment by integrated backscatter and its relationship with longitudinal deformation and diastolic function in heart failure with preserved ejection fraction. Int J Cardiovasc Imaging. 2016;32:1071–1080. doi: 10.1007/s10554-016-0881-5. [DOI] [PubMed] [Google Scholar]
- 29.Burlew BS, Weber KT. Cardiac fibrosis as a cause of diastolic dysfunction. Herz. 2002;27:92–98. doi: 10.1007/s00059-002-2354-y. [DOI] [PubMed] [Google Scholar]
- 30.Ellims AH, et al. Diffuse myocardial fibrosis evaluated by post-contrast T1 mapping correlates with left ventricular stiffness. J Am Coll Cardiol. 2014;63:1112–1118. doi: 10.1016/j.jacc.2013.10.084. [DOI] [PubMed] [Google Scholar]
- 31.Rommel KP, et al. Left Ventricular STIFFness vs. Fibrosis Quantification by T1 MAPping in Heart Failure With Preserved Ejection Fraction-STIFFMAP-HFpEF. Circulation. 2015;132:18283. [Google Scholar]
- 32.Moreo, A. et al. Myocardial fibrosis severity and location: influence on diastolic dysfunction. Eur Heart J. 34, 10.1093/eurheartj/eht308.1003 (2013).
- 33.Kim J, et al. Association of right ventricular pressure and volume overload with non-ischemic septal fibrosis on cardiac magnetic resonance. PLoS One. 2016;11:e0147349. doi: 10.1371/journal.pone.0147349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Booth DC, Wisenbaugh T, Smith M, DeMaria AN. Left ventricular distensibility and passive elastic stiffness in atrial septal defect. J Am Coll Cardiol. 1988;12:1231–1236. doi: 10.1016/0735-1097(88)92605-8. [DOI] [PubMed] [Google Scholar]
- 35.Dupont M, et al. Prognostic role of pulmonary arterial capacitance in advanced heart failure. Circ Heart Fail. 2012;5:778–785. doi: 10.1161/CIRCHEARTFAILURE.112.968511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kilner PJ, et al. Pulmonary regurgitation: the effects of varying pulmonary artery compliance, and of increased resistance proximal or distal to the compliance. Int J Cardiol. 2009;133:157–166. doi: 10.1016/j.ijcard.2008.06.078. [DOI] [PubMed] [Google Scholar]
- 37.Khairy P, et al. Implantable cardioverter-defibrillators in tetralogy of Fallot. Circulation. 2008;117:363–370. doi: 10.1161/CIRCULATIONAHA.107.726372. [DOI] [PubMed] [Google Scholar]


