Abstract
Papillary Thyroid Cancer (PTC) is an endocrine malignancy in which BRAFV600E oncogenic mutation induces the most aggressive phenotype. In this way, considering that lncRNAs are arising as key players in oncogenesis, it is of high interest the identification of BRAFV600E-associated long noncoding RNAs, which can provide possible candidates for secondary mechanisms of BRAF-induced malignancy in PTC. In this study, we identified differentially expressed lncRNAs correlated with BRAFV600E in PTC and, also, extended the cohort of paired normal and PTC samples to more accurately identify differentially expressed lncRNAs between these conditions. Indirectly validated targets of the differentially expressed lncRNAs in PTC compared to matched normal samples demonstrated an involvement in surface receptors responsible for signal transduction and cell adhesion, as well as, regulation of cell death, proliferation and apoptosis. Targets of BRAFV600E-correlated lncRNAs are mainly involved in calcium signaling pathway, ECM-receptor interaction and MAPK pathway. In summary, our study provides candidate lncRNAs that can be either used for future studies related to diagnosis/prognosis or as targets for PTC management.
Introduction
Thyroid cancer is the endocrine malignancy1 that, although stable until the 1990s, has progressively and greatly increased thereafter2, 3. The vast majority of the thyroid cancers originate from the follicular cell epithelium1, which includes papillary thyroid carcinoma (PTC) that accounts for approximately 80% of all thyroid malignancies4.
Thyroid oncogenesis is still under investigation, however a high frequency (70%) of activating mutations in components of the mitogen-activated protein kinase (MAPK) pathway was reported, such as BRAFV600E 5, 6 and HRAS/NRAS/KRAS point mutations7, 8. Also, fusions involving the RET9 and NTRK1 tyrosine kinases10 were described to promote thyroid cancers. More recently, the set of known PTC driver alterations was extended to include EIF1AX, PPM1D, and CHEK27.
Additionally of being the most frequent mutation in many types of cancers including PTC7, 11, BRAFV600E confers poorer prognosis compared to other oncogenes. There is a growing number of evidence demonstrating that BRAFV600E correlates with metastasis, cancer recurrence12 and higher mortality in PTC13. BRAFV600E-expressing cells have a diversity of malignant characteristics, including increased DNA synthesis, dedifferentiation, and chromosomal instability14. Also, BRAFV600E stimulates more actively MEK-dependent invasion than the expression of RET/PTC oncoprotein through the expression of matrix metalloproteinases (e.g. MMP-3, MMP-9 and MMP-13), which, in part, can explain the more aggressive BRAFV600E-induced phenotype15.
Similarly to melanoma16, BRAF mutation occurs at early stages of PTC development11, 17. Besides all BRAF oncogenic activities, its single exacerbated stimulation of the MAPK pathway is not sufficient to sustain malignant transformation, resulting in induced senescence18 that confers a barrier to tumor progression19. To bypass BRAF-induced senescence, cells may suffer a second event that allows malignant transformation, as possibly the epigenetic silencing of tumor suppressors DAPK, TIMP320, SLC5A820, 21 and hMLH122 and other BRAF-induced mechanisms that remain to be discovered11. In thyroid cancers, Thyroid-stimulating Hormone (TSH) is more involved in overcoming senescence; while BRAF overexpression suppresses thyroid hormone biosynthesis and leads to elevated TSH levels in vivo 14; it was shown that TSH signaling inhibits BRAFV600E-induced senescence through DUSP623.
Recently, BRAFV600E-associated mRNA signature was determined in a mouse model and human samples24, which identified new genes not previously reported as related to BRAF mutation in thyroid cancer (e.g. MMD, ITPR3, AACS, LAD1, PVRL3, ALDH3B1, and RASA1) that will provide further support for future research on BRAF-induced PTC24. However, this analysis did not evaluate the expression of long noncoding RNAs (lncRNAs), which are progressively shown to be of fundamental importance in other types of cancer25, 26. Such analysis is necessary for the identification of BRAFV600E-correlated long noncoding RNAs.
LncRNAs are RNAs longer than 200 nucleotides that have no coding potential27 and are involved in several processes, such as gene expression regulation through chromatin modulation28, 29, epigenetic control30, association with translational apparatus31, improving other mRNA stability32, serving as a scaffold for protein33, acting as decoys for miRNAs34, altering protein turnover35, among others.
To date, as per the authors’ knowledge, only a few published studies identified differentially expressed (DE) lncRNAs between normal (N) and tumoral thyroid (T) in a limited set of paired samples36, 37. Although these findings laid the foundation for further investigation of lncRNAs related to PTC36, they need to be confirmed in a more numerous group of patients.
In this study we confirmed previously reported lncRNAs and determined new DE lncRNAs in PTC in a larger set of samples and also identified BRAFV600E-correlated lncRNAs, providing possible candidates that can constitute secondary mechanisms of BRAF- induced malignancy in PTC.
Results
Comparative analyses identified lncRNAs deregulated in PTC and correlated with BRAFV600E mutation
Comparative analysis between 59 pairs of matched normal and papillary thyroid cancer samples identified 455 differentially expressed lncRNAs (log2 fold change > 1 or < −1; adj. p-value < 1 × 10−7; Fig. 1A), being 71 lncRNAs upregulated and 384 lncRNAs downregulated in PTC (Supplemental Table S1). The same samples presented a total of 2016 mRNAs (log2 fold change > 1 or <−1; adj. p-value < 0.05; Supplemental Table S2) and 186 microRNAs (log2 fold change > 1 or < −1; adj. p-value < 0.05; Supplemental Table S3) differentially expressed.
Figure 1.
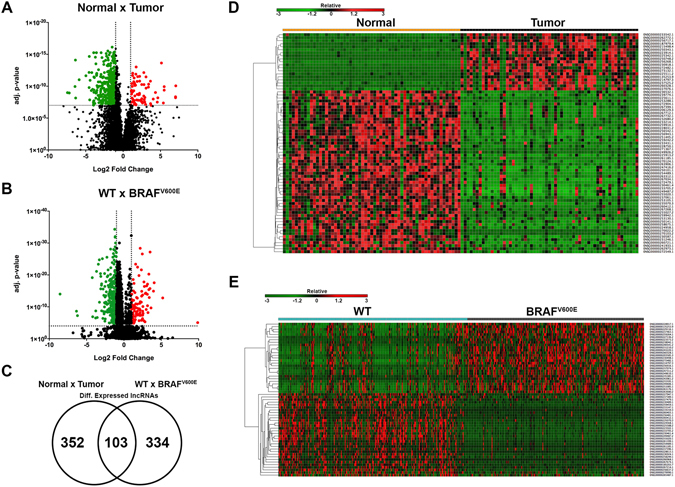
Differentially expressed lncRNAs between N × T and WT × BRAFV600E were identified. (A) Volcano plot of DE lncRNAs between N × T (log2 fold change > 1 or < −1; adj. p-value < 1 × 10−7). (B) Volcano plot of DE lncRNAs between WT × BRAFV600E (log2 fold change > 1 or < −1; adj. p-value < 1 × 10−4). (C) Venn Diagram of common DE lncRNAs between N × T and WT × BRAFV600E. (D) Heatmap* of DE lncRNAs between N × T (log2 fold change > 3 or < −3; adj. p-value < 1 × 10−7). (E) Heatmap* of DE lncRNAs between WT × BRAFV600E (log2 fold change > 2.5 or < −2.5; adj. p-value < 1 × 10−4). *For hierarchical clustering, one minus Spearman rank correlation was performed.
Differential expression analyses were also performed to identify BRAFV600E-correlated lncRNAs. The comparison between BRAF wild type (WT) patients (n = 242) and BRAFV600E patients (n = 226), determined 437 differentially expressed lncRNAs (log2 fold change > 1 or < −1; adj. p-value 1X10−4; Fig. 1B), being 117 upregulated and 320 downregulated (Supplemental Table S4). The same comparison found a total of 924 mRNAs (log2 fold change > 1 or < −1; adj. p-value < 0.05; Supplemental Table S5) and 94 microRNAs (log2 fold change > 1 or < −1; adj. p-value < 0.05; Supplemental Table S6) differentially expressed. A total of 103 lncRNAs was differentially expressed in both analyses [(Normal × Tumor and WT × BRAFV600E), (Fig. 1C, Table 1 and Supplemental Table S7)].
Table 1.
Top 5 upregulated and 5 downregulated common DE lncRNAs between Normal × Tumor and WT × BRAFV600E.
| Ensembl | log2 FC | Adj. p-value | log2 FC | Adj. p-value |
|---|---|---|---|---|
| N × T | N × T | WT × BRAFV600E | WT × BRAFV600E | |
| ENSG00000214797.3 | 7.01 | 6.39E-09 | 5.19 | 4.92E-12 |
| ENSG00000273132.1 | 5.49 | 4.17E-10 | 1.69 | 3.70E-12 |
| ENSG00000230918.1 | 5.49 | 1.17E-10 | 1.36 | 3.89E-05 |
| ENSG00000260328.1 | 5.39 | 4.27E-08 | 4.04 | 2.26E-16 |
| ENSG00000256268.1 | 5.12 | 2.27E-14 | 1.00 | 4.52E-13 |
| ENSG00000261185.1 | −6.42 | 1.13E-10 | −2.81 | 3.55E-06 |
| ENSG00000254489.1 | −6.37 | 7.98E-15 | −3.27 | 8.33E-11 |
| ENSG00000260412.1 | −6.33 | 7.11E-09 | −3.77 | 2.59E-08 |
| ENSG00000235070.3 | −4.97 | 2.28E-12 | −3.01 | 1.10E-15 |
| ENSG00000247416.2 | −4.55 | 6.89E-13 | −2.40 | 1.09E-16 |
Experimental validation using qRT-PCR was performed to demonstrate the reliability of the bioinformatics analyses applied. From the top 25 positively DE lncRNAs and from the top 20 negatively DE lncRNAs, it were selected for validation those lncRNAs with low adjusted p-values to minimize expression variability, especially in the comparison BRAFWT × BRAFV600E tumor, among others characteristics (for detailed information see Methods). From a total of 5 DE lncRNAs selected for validation from the TCGA analysis (Fig. 2 and Supplemental Fig. S1, upper part), 4 DE lncRNAs were validated using thyroid cell lines (Fig. 2 and Supplemental Fig. S1, lower part), which strengths the reliability of this bioinformatics analysis, although the experimentally tested set of lncRNAs constitutes a relatively small sampling. Considering all comparisons, we obtained a very expressive validation efficiency [from a total of 8 different comparisons (Normal × Tumor and BRAFWT × BRAFV600E), 6 were validated]. Downregulation of ENSG00000235070.3 and ENSG00000255020.1 in PTC was confirmed in the tumor cell lines TPC1 (BRAFWT) and BCPAP (BRAFV600E) compared to the normal immortalized cell line NTHY (Fig. 2). Also, downregulation of their expression was in accordance to the bioinformatics analysis, since lower expression for both of them was observed in BCPAP (BRAFV600E) compared to TPC1 (BRAF wild type) (Fig. 2). Noteworthy, is that due to the very low abundance of ENSG00000255020.1 in the BCPAP cell line, qRT-PCR resulted in two unspecific melting peaks, which did not influenced the results. Upregulation of ENSG00000273132.1 in PTC was confirmed, however its overexpression in BRAFV600E tumors was not observed in the cell line BCPAP compared to TPC1 (Fig. 2), maybe due to the small log2 fold change value (1.69) of this comparison. Overexpression of ENSG00000230498.1 in BRAFV600E PTC compared to BRAF wild type tumors was also confirmed (Supplemental Fig. S1); nevertheless ENSG00000247311.2 was undetectable in both TPC1 and BCPAP cells (Supplemental Fig. S1).
Figure 2.
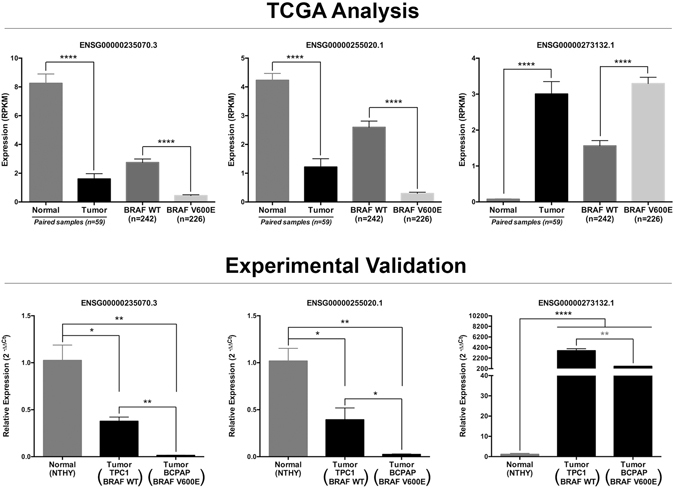
Experimental validation of DE lncRNAs. Upper part of panel displays the expression levels of the indicated lncRNAs in the TCGA analyses. The nonparametric Mann–Whitney test was applied due to the non-Gaussian expression distribution and p-value was assigned. Lower part of panel displays the experimental validation of these lncRNAs measured by qRT-PCR and calculated with 2−ΔΔCt method using RPL19 (Ribosomal Protein L19) as endogenous control. Experiments with three biological replicates were performed using two technical replicates for each sample. These results are representative of at least two independent experiments. Values are plotted as expression mean ± Standard Error of Mean (SEM). Unpaired two-tailed t-Test assigned the p-value.
Clustering lncRNAs identifies two groups with similar expression patterns
For downstream analyses, we increased the stringency of differentially expressed lncRNAs between Normal × Tumor (log2 fold change > 3 or < −3; adj. p-value <1 × 10−7, n = 73; Fig. 1D) and between WT × BRAFV600E (log2 fold change >2.5 or < −2.5; adj. p-value < 1 × 10−4, n = 59; Fig. 1E) to analyze the lncRNAs that were most DE. Hierarchical clustering was used to organize patients or lncRNAs into groups according to the expression levels of DE lncRNAs.
Results demonstrated that this set of lncRNAs is capable of clustering, majorly, normal and cancer patients in two distinct groups (Supplemental Fig. S2A). Clustering lncRNAs by Spearman correlation among all DE lncRNAs also identified two groups of lncRNAs highly positively correlated or negatively correlated (Supplemental Fig. S3A).
Hierarchical clustering was also performed with a more stringent set of DE lncRNAs between WT and BRAFV600E, which allowed the clustering of two groups enriched with WT and BRAFV600E patients, respectively (Supplemental Fig. S2B). Clustering lncRNAs by Spearman correlation among all DE lncRNAs also identified two groups highly positively correlated or negatively correlated lncRNAs (Supplemental Fig. S3B).
Indirectly validated lncRNAs’ targets are involved in several oncogenic processes
As almost the totality of the identified DE lncRNAs in both conditions (Normal × Tumor and WT × BRAFV600E) is uncharacterized, we used prediction methods to identify a possible interaction between lncRNAs and mRNAs/microRNAs. Predicted mRNAs and microRNAs (targets of DE lncRNAs) were compared to differentially expressed mRNAs and microRNAs (log2 fold change >1 or <−1; adj. p-value < 0.05) calculated from the same TCGA patients. Predicted mRNAs/microRNAs that were also identified as DE were considered as indirectly validated targets.
A total of 1109 DE mRNAs (Table 2 and Supplemental Table S8) and 26 DE microRNAs (Supplemental Table S9) were found to be predicted targets of the DE lncRNAs between Normal and Tumor samples and were considered as indirectly validated targets. Gene ontology and KEGG pathways enrichment of these validated mRNAs demonstrated that most of the genes are involved in surface receptors responsible for signal transduction and cell adhesion, as well as, regulation of cell death, proliferation and apoptosis (Fig. 3A). Enriched pathways (Fig. 3B) were composed of cytokine-cytokine receptor interaction, pathways in cancer (Fig. 3C), focal adhesion, MAPK pathway and calcium signaling pathway. Validated microRNAs were also used to determine enriched pathways based on their predicted targets calculated elsewhere (Fig. 3D). Genes involved in cancer and MAPK pathways were the most enriched pathways. Interestingly, some genes predicted to be targets of the validated microRNAs were also DE expressed in our analysis (Supplemental Table S2), such as upregulation of the MAPK constituents, CACNG4, CACNA1E, DUSP4, TGFBR1, FGF1, FGF2 and MAP3K1. Enriched pathways were extended to genes involved in cancer, focal adhesion and calcium signaling (Fig. 3D).
Table 2.
Top 5 upregulated and 5 downregulated DE lncRNAs between paired Normal × Tumor with examples of indirectly validated targets.
| Ensembl | log2 FC | Adj. p-value | Upregulated indirectly validated targets | Downregulated indirectly validated targets |
|---|---|---|---|---|
| N × T | N × T | |||
| ENSG00000223914.1 | 7.04 | 8.69E-11 | VGLL1, GDF6, FAM19A2, HRH1 | RPS6KA5, RNF150, ANK2, FOSB |
| ENSG00000250748.2 | 7.01 | 4.56E-09 | FUT3, GPR115, CAPN8, COL7A1 | SVEP1, LMOD1, DPT, KCNA1 |
| ENSG00000214797.3 | 7.01 | 6.39E-09 | TMEM130, HES2, KCP, DTX4 | CDHR3, PAK3, RASSF6, NWD1 |
| ENSG00000273132.1 | 5.49 | 4.17E-10 | KLK6, ELFN2, C19orf59, SHISA6 | SRF, CPXM1, LAYN, FAM163A |
| ENSG00000230918.1 | 5.49 | 1.17E-10 | GRM4, DPP4, LRP4, SHISA6 | EGR1, TFCP2L1, FOXJ1, ABCA9 |
| ENSG00000253288.1 | −7.43 | 1.03E-09 | SLC6A20, KLK10, FUT3, HRH1 | HAP1, SH2D6, FOXP2, ADH1B |
| ENSG00000272479.1 | −7.19 | 1.16E-09 | DMBX1, TMPRSS6, TMPRSS4, PPP1R1B | CUX2, PAX1, CLCNKB, FOSB |
| ENSG00000261185.1 | −6.42 | 1.13E-10 | B3GNT3, ELFN2, LRP4, SHISA6 | NR4A1, C1QTNF7, RNF150, FAM180B |
| ENSG00000254489.1 | −6.37 | 7.98E-15 | SYTL5, HPCAL4, KCNQ3, CPNE4 | RBM24, PGA3, GFRA1, RNF150 |
| ENSG00000260412.1 | −6.33 | 7.11E-09 | CLDN16, PDE4C, LRG1, SHROOM4 | SLC26A4, CDHR3, PAK3, NWD1 |
Figure 3.
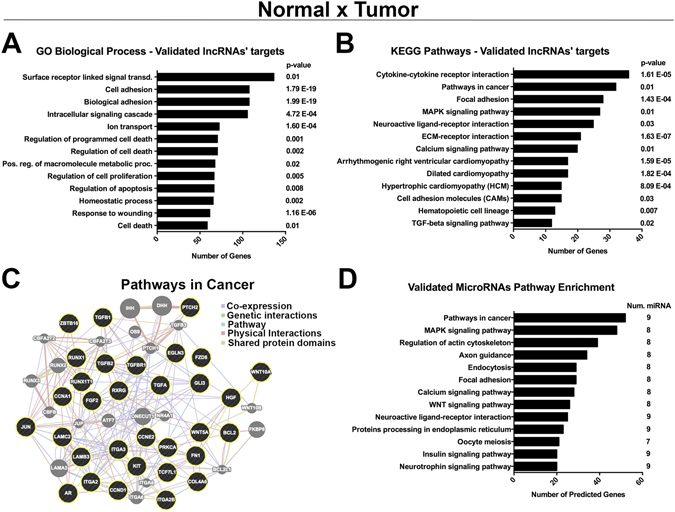
Indirectly validated targets of the DE lncRNAs between N × T are involved in cancer-related processes. (A) GO biological processes and (B) KEGG enriched pathways of the indirectly validated mRNA targets of the DE lncRNAs between N × T. (C) Proteins’ network of genes linked to the pathways in cancer, where black circles are validated targets and grey circles are connective proteins. (D) KEGG Pathways enrichment of the indirectly validated microRNAs targets of the DE lncRNAs between N × T.
Between WT and BRAFV600E, 471 DE mRNAs (Table 3 and Supplemental Table S10) and 11 DE microRNAs (Supplemental Table S11) were indirectly validated. Gene ontology of these mRNAs demonstrated that most of the genes are also related to surface receptors involved in signal transduction and cell adhesion, but, additionally, with response to hormone stimulus and transmembrane transport (Fig. 4A). Enriched pathways (Fig. 4B) were constituted of calcium signaling pathway (Fig. 4C), cardiomyopathies and ECM-receptor interaction. KEGG enrichment pathway analysis of the validated DE microRNAs demonstrated participation of the MAPK and WNT pathways, as well as regulation of actin cytoskeleton and focal adhesion (Fig. 4D). Several pro-oncogenic genes were found to be upregulated in our analysis and were described as predicted targets of the validated DE microRNAs, as the example of MET and TGFBR1 genes (Supplemental Table S5).
Table 3.
Top 5 upregulated and 5 downregulated DE lncRNAs between WT × BRAFV600E with examples of indirectly validated targets.
| Ensembl | log2 FC | Adj. p-value | Upregulated indirectly validated targets | Downregulated indirectly validated targets |
|---|---|---|---|---|
| WT × BRAF V600E | WT × BRAF V600E | |||
| ENSG00000255595.1 | 9.87 | 5.68E-05 | TCAP, ITGA2, LY6G6C, BEND6 | SLC5A5, ASTN1, PART1, IRX6 |
| ENSG00000214797.3 | 5.19 | 4.92E-12 | HES2, DTX4, KCP, LDLR | RNF157, HAP1, NWD1, SLC14A2 |
| ENSG00000260328.1 | 4.04 | 2.26E-16 | TMPRSS4 | PRND, TMPRSS3, PREX2, CNTNAP2 |
| ENSG00000230498.1 | 3.95 | 5.74E-14 | C1orf106, ADAMTS14, DMBX1, DUSP13 | FCGBP, GCGR, SOX3, PPP2R2C |
| ENSG00000256916.1 | 3.62 | 3.51E-09 | ELFN2, SPTBN2, MUC16, ZNF469 | SSPO, ASXL3, CTNND2, CNTNAP2 |
| ENSG00000267674.1 | −8.55 | 3.89E-13 | VSIG1, LDLR | ADM2, ST3GAL6, NWD1, SLC5A8 |
| ENSG00000237396.1 | −6.35 | 3.05E-08 | SIGLEC6, SDK1, C1QL2, MUC21 | TFCP2L1, SCUBE1, SBSN, PAX1 |
| ENSG00000227947.1 | −6.22 | 5.72E-07 | ELFN2, EPHA10, SLC30A3, SYT1 | SFTPC, GATA5, SFRP1, MATN1 |
| ENSG00000224568.1 | −4.23 | 8.95E-18 | SLC6A14, C1orf106 | HIF3A, SYT13, SLC29A4, ARSF |
| ENSG00000267214.1 | −4.18 | 1.17E-06 | ELFN2, COL7A1, B4GALNT3 | FAM124A, SULT1A2, SLC29A4, CNTNAP2 |
Figure 4.
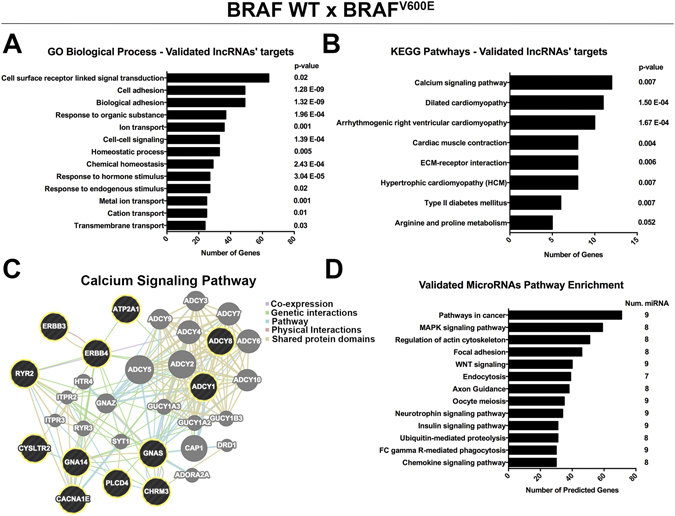
Indirectly validated targets of the DE lncRNAs between WT × BRAFV600E are involved in oncogenic pathways. (A) GO biological processes and (B) KEGG enriched pathways of the indirectly validated mRNA targets of the DE lncRNAs between WT × BRAFV600E. (C) Proteins’ network of genes linked to calcium signaling pathway, where black circles are validated targets and grey circles are connective proteins. (D) KEGG pathways enrichment of the indirectly validated microRNAs targets of the DE lncRNAs between WT × BRAFV600E.
Discussion
Long noncoding RNAs are arising as key participants in cancer establishment and progression by several oncogenic mechanisms30, 32. On the other hand, it is of urge interest the determination of how these lncRNAs are activated and how they can be associated with specific events or genotypes, such as point mutations. BRAFV600E is the driver oncogenic mechanism with the greatest incidence in PTC7 and, therefore, any event correlated with this mutation will be necessary to understand BRAFV600E-induced aggressiveness.
This is the first study to identify DE lncRNAs correlated with BRAFV600E in PTC and, besides that, we extended the cohort of paired normal and PTC samples to more accurately determine DE lncRNAs between these conditions.
We have identified 455 DE lncRNAs between paired normal and PTC samples. A total of 76 (log2 fold change >1 or < −1; adj. p-value < 1 × 10−7) lncRNA were previously reported as DE in thyroid cancer compared to adjacent normal thyroid36 (Fig. 5A and Supplemental Table S12). This validation set, together with the lncRNAs confirmed by experimental approaches (Fig. 2 and Supplemental Fig. S1), confers consistency to our analysis. Additionally, a diversity of DE lncRNAs identified in our analysis were reported in individual studies as altered in PTC samples, such as ENSG00000259104.2 38, 39, ENSG00000236130 40, ENSG00000226363 37, ENSG00000271086 39, 41, ENSG00000223914 37 and ENSG00000187979 37.
Figure 5.
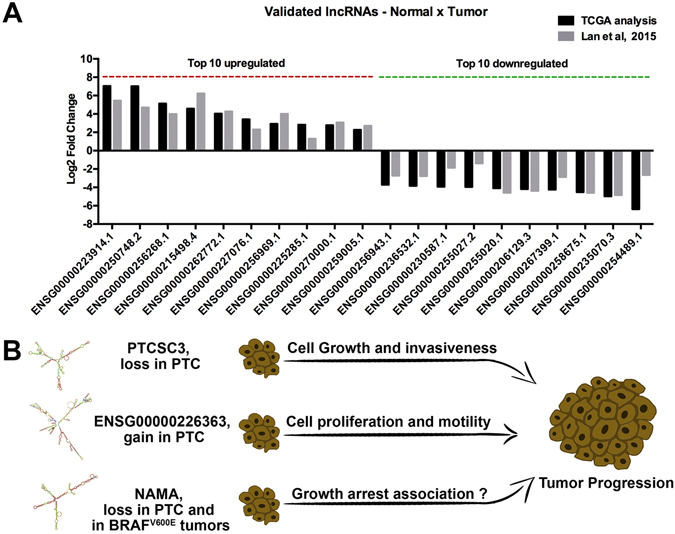
Experimentally validated lncRNAs are important to tumor malignancy. (A) Examples of DE lncRNAs between Normal and PTC, which were confirmed in a validation set36. (B) Differentially expressed lncRNAs in PTC that alter tumor malignancy.
ENSG00000259104.2 (PTCSC3), which is downregulated in the tumor samples (log2 fold change −1.40; adj. p-value 1.11E-12) was previously reported as having thyroid-specific expression and decreased expression in PTC38, 39. Interestingly, the risk allele [T] associated with SNP rs944289, located at PTCSC3’s promoter, affects the binding site of C/EBPα and C/EBPβ (PTCSC3 activators), reducing its expression. Restoration of PTCSC3 expression in PTC cells inhibited cell growth and affected the expression of genes involved in DNA replication/repair, cellular movement and cell death38. Also PTCSC3 ectopic expression reduces cell proliferation and increases cell cycle arrest and apoptosis39, while reducing cell motility and invasiveness through S100A4 downregulation42 (Fig. 5B).
ENSG00000236130 (PTCSC2), was also reported as having decreased expression in PTC40, which was confirmed in our analysis (N × T log2 fold change −1.03; adj. p-value 3.12E-09). The risk allele [A] of rs965513 was significantly associated with low expression of unspliced PTCSC2 in unaffected thyroid tissue, however this correlation was not extended to PTC samples40.
ENST00000426615 (ENSG00000226363) is another lncRNA that we identified as upregulated in PTC (NxT log2 fold change 3.87; adj. p-value 2.36E-05), which was experimentally demonstrated to be overexpressed in this cancer, inducing cell proliferation and motility37 (Fig. 5B).
Our analysis also confirmed the differential expression (N × T: log2 fold change −2.42; adj. p-value 3.96E-11) of the previously reported lncRNA ENSG00000271086 (NAMA), which is downregulated in PTC compared to normal tissues39, 41 and in BRAFV600E tumors compared to wild type tumors39 (Fig. 5B). NAMA is induced by inhibition of the MAPK pathway, growth arrest and DNA damage41 and our analysis also demonstrated that NAMA is downregulated in BRAFV600E patients (WT × BRAFV600E log2 fold change −1.66; adj. p-value 2.02E-15). All these independently validated lncRNAs demonstrate the reliability of our study (Fig. 5).
Similarity matrix based on Spearman correlation identified clusters of DE lncRNAs between Normal × Tumor (Supplemental Fig. S3A) and WT × BRAFV600E (Supplemental Fig. S3B) with similar expression patterns, which can provide evidence for further studies to determine common upstream regulators.
Indirectly validated targets of the DE lncRNAs between Normal × Tumor are involved in a diversity of biological processes (Fig. 3A). For instance, it was noticed an overrepresentation of adhesion molecules, such as downregulation of CDH16, which was already reported as a potential marker for PTC43. Along with CDH16, many other cadherins were identified as validated targets of DE lncRNAs, such as CDH2, CDH3, CDH4, CDH6, CDH11 and CDH24 (Supplemental Table S8). Another highly enriched biological process was the regulation of programmed cell death (Fig. 3A), represented by the upregulation of the antiapoptotic SOX444 and TP6345 in PTC samples. Enriched pathways (Fig. 3B) as cytokine-cytokine receptor interaction, focal adhesion and MAPK pathways were already reported in the first study of DE lncRNA with paired Normal × PTC samples36, providing further support for future research. It was observed an enrichment of MAPK-related genes, represented in our results by upregulation, in the tumor samples, of TGB1, TGFB2 and TGFBR1 that were shown to activate the MAPK pathway46. Interestingly, pathway enrichment analysis of indirectly validated microRNAs (Fig. 3D) demonstrated a convergent tendency to genes involved in cancer, MAPK pathway and focal adhesion, which were also observed with the validated mRNAs.
Indirectly validated targets of the DE lncRNAs between WT × BRAFV600E tumors were demonstrated to be involved with cell surface receptors responsible for signal transduction and with cell adhesion (Fig. 4A). Pathway enrichment analysis, identified genes involved in calcium signaling and ECM-receptor interaction, which were already reported as an early transcriptome change in BRAFV600E-associated mouse model24. Interestingly, we also observed several genes correlated with cardiomyopathies that are mostly related to calcium regulation in cardiac muscle cells (Fig. 4B). Calcium (Fig. 4C) and MAPK cascade (represented by BRAFV600E group) are tightly involved, where calcium modulates the protein interaction properties of ERKs, affecting the subcellular localization and influencing the distribution of their targets47. Calcium can also stimulates MEK through Ras activation48. Therefore, these results can support a future investigation to answer if BRAFV600E-stimulated MAPK activation can be reinforced by calcium modulation induced by the DE lncRNAs. Additionally, MAPK stimulation may be supported by the indirectly validated microRNAs, since pathway analysis demonstrated an enrichment of microRNAs’ targets in cancer and MAPK pathways (Fig. 4D). Interestingly, predicted targets of the DE microRNAs were also differentially expressed in our analysis, such as the upregulation of TGFBR1 and downregulation of PRKACB (Supplemental Table S5).
Concluding, our extended cohort of paired Normal and PTC patients identified new DE lncRNAs and confirmed many other lncRNAs already reported. Additionally, to our knowledge, this is the first study to identify BRAFV600E-correlated lncRNAs in PTC, which will provide support for future studies aiming to identify BRAFV600E-linked events in attempt to optimize therapeutic treatment and diagnosis/prognosis of this aggressive PTC genotype.
Methods
Data analysis
Thyroid Carcinoma (THCA) clinical information, mRNA and microRNA data expression data were downloaded from The Cancer Genome Atlas (TCGA) online platform (https://tcga-data.nci.nih.gov/tcga/), as January 2016. Mutations data were retrieved through cBioPortal49. LncRNA RPKM expression levels corresponding to TCGA patients were downloaded through TANRIC50, which obtained the genomic coordinates of 13,870 human lncRNAs from the GENCODE Resource (version 19)51 and further filtered out those lncRNA exons that overlapped with any known coding genes based on the gene annotations of GENCODE and RefGene, resulting in 12,727 lncRNAs50.
BRAFV600E patients were selected to form the BRAFV600E group (n = 226), which excluded any other type of BRAF mutations. Wild Type group (n = 242) was formed by patients without any somatic mutation in BRAF gene, but patients with mutations in HRAS, NRAS, KRAS, EIF1AX, PPM1D, RET and NTRK1 were considered. It were selected only the patients with papillary thyroid cancer diagnosis.
Differential Expression Analysis
For differential expression analysis of mRNA and microRNA was used edgeR package52 through TCGAbiolinks53. To identify differentially expressed lncRNAs between groups, it was used the paired/unpaired Student t test to assess the statistical difference of mean expression values between the two groups50. LncRNA with median value equal zero were excluded and fold change was calculated using median expression values. In all differential analysis, p-values were adjusted for False Discovery Rate (FDR) < 0.05 as multiple hypothesis test correction method.
RNA extraction, Reverse-transcription and qPCR
Total RNA was phenol-chloroform extracted from cell lines Nthy-ori3-1 (NTHY–immortalized human thyroid follicular epithelial cell), TPC1 (papillary thyroid carcinoma- RET/PTC1 rearrangement) and BCPAP (papillary thyroid carcinoma–BRAFV600E) using TRIzol reagent (Invitrogen) according to the manufacter’s instructions. Four µg of total RNA was reverse transcribed using M-MLV Reverse Transcriptase (Invitrogen) in the presence of 100 ng of random hexamers primers according to the manufacter’s protocol. qPCR reaction was performed using 100 ng of cDNA, 1X Power SYBR Green PCR Master Mix (Applied Biosystems) and specific primers. Amplification and detection were performed using ViiA7 TM Real-Time PCR System (Applied Biosystems). Relative gene expression was calculated using the QGENE program and calculated with 2−ΔΔCt method using RPL19 (Ribosomal Protein L19) as endogenous control.
Primers used (5′-3′): RPL19 (Fw-TCTCATGGAACACATCCACAA; Rv-TGGTCAGCCAGGAGCTTCTT), ENSG00000273132.1 (Fw-CTAGCTGCCAGCAGTGACAA; Rv-GCGAGAGCACAGATGACCAC), ENSG00000230498.1 (Fw-CCCTGGGTGATGAAGATGAG; Rv-TGGGATCCCTTTTTTGTCCG), ENSG00000235070.3 (Fw-TGACTCCAAGTTCACGCAGC; Rv-GTGGATGAGTTGTGTGCTGG), ENSG00000255020.1 (Fw-AGTGACGTGGGGAAGAAACG; Rv-CGACATATTTCAAGGGCGCC) and ENSG00000247311.2 (Fw-GCTGTGAGTGACTCTTCAGC and ACAGACACACCCAGGAACAA).
To select the above DE lncRNAs for validation, it was taken in consideration one of the major characteristics of lncRNA, that is, high heterogeneous expression across the same tumor and even the same cell line. Long noncoding RNA’s expression is tightly regulated by a wild range of cellular responses and, due to the markedly lower transcriptional levels of lncRNAs, the expression variability inside the same group of patients is expected. Taking this aspects in consideration, from the top 25 positively DE lncRNAs and from the top 20 negatively DE lncRNAs, it were selected for validation those lncRNAs with low adjusted p-values to avoid this variability, especially in the comparison BRAFWT × BRAFV600E tumor, which is the focus of this research. As another desirable characteristic, most lncRNAs selected, presented at least in one group (normal thyroid, PTC, BRAFWT or BRAFV600E) a median expression greater than 1 RPKM (reads per kilo base per million mapped reads). Added to that, it was given preference for those lncRNAs without isoforms (seen that many lncRNAs have annotation errors) and that present at least 2 exons, which are more stable and would allow the PCR primers to be located in different exons.
Prediction of lncRNAs targets
To identify the possible target genes of the selected (Fig. 1D and E) differentially expressed lncRNAs via cis- or trans-regulatory effects, two previously described approaches were used36, 54. The genes transcribed within a 10 kb window upstream or downstream of lncRNAs were considered as cis-target genes36, 54. The second method was used to identify trans-targets and is based on mRNA and microRNA sequence complementarity with the query lncRNA. For mRNA interactions we used a pre-computed database that catalogs the predicted lncRNA–RNA interactions55, where the accessible regions of the query lncRNA and possible targets (mRNA/lncRNA) are extracted, the binding energies of pairs of sequences (target and query) around the seed matches are evaluated and the minimum interaction energy of the joint secondary structures is calculated55. The 500 predicted targets (mostly constituting repeated targets with different interaction sites) with the lowest minimum free energies under −20 kcal/mol were taken in consideration for downstream analysis. For lncRNA and microRNA interaction prediction it was used rna22, a method for identifying microRNA-binding sites and their corresponding heteroduplexes56. It were selected those microRNAs with a folding energy lower than −20 kcal/mol.
Indirect Validation
As interaction prediction methods are susceptible to error and to minimize this, we compared the predicted targets of the differentially expressed lncRNAs with the differentially expressed mRNAs and microRNAs36 calculated with the TCGA patients, because we consider that the targets of DE lncRNAs would possibly be DE in TCGA analysis. With this approach, we intended to enrich our analysis for targets with a greater propensity to be occurring biologically.
Gene ontology, pathway enrichment and protein-protein interaction network
Indirect validated targets of the DE lncRNAs were loaded into the Database for Annotation, Visualization and Integrated Discovery (DAVID)57, which returned the gene ontology of the query genes and identified enriched Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways58. miRSystem59 was used to calculate enriched pathways based on the predicted targets of the query microRNAs, which in this case, were the DE microRNAs in both conditions (Normal × Tumor and WT × BRAFV600E). For protein-protein interaction network it was used Genemania60.
Electronic supplementary material
Acknowledgements
This work was supported by grants to EME from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP- 2014/18189-5) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq Research Fellowship- 309187/2015-0 and 506780/2013-9). FAPESP provided fellowships to LG (2014/07726-0), C.S.F. (2014/50521-0) and M.C.R.M. (2015/13396-5). Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) provided fellowship to L.G., J.R.P. and J.F.S. (CAPES-PROCAD-2961/2014). Fundação de Amparo à Pesquisa do Estado da Bahia (FAPESB) provided fellowship to P.P.A. (6693/2015). CNPq provided fellowship to D.R.P. and A.C.C. and provided financial support to E.T.K. (458505/2014-5 and 308527/2013-5). L.G., J.R.P., W.A.S.J. and E.M.E. are members of Center For Cell-Therapy, CEPID/FAPESP (grant 2013/08135-2). Some of the results published here are in whole or part based upon data generated by the TCGA Research Network: http://cancergenome.nih.gov/.
Author Contributions
L.G., J.R.P., C.S.F., M.C.R.M., D.R.P., P.P.A., T.P.S., J.F.S., A.C.C., I.E.G.P. and M.M.C. conducted the analyses and experiments. L.G., J.R.P., D.R.P., P.P.A., T.P.S., J.F.S., A.C.C., I.E.G.P., M.M.C., E.T.K., W.A.S.J. and E.M.E. contributed to the writing of the manuscript and jointly developed the structure and arguments for the paper. All authors reviewed and approved the final manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Lucas Goedert and Jessica Rodrigues Plaça contributed equally to this work.
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-01957-0
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Segev DL, Umbricht C, Zeiger MA. Molecular pathogenesis of thyroid cancer. Surgical oncology. 2003;12:69–90. doi: 10.1016/S0960-7404(03)00037-9. [DOI] [PubMed] [Google Scholar]
- 2.Vigneri R, Malandrino P, Vigneri P. The changing epidemiology of thyroid cancer: why is incidence increasing? Current opinion in oncology. 2015;27:1–7. doi: 10.1097/CCO.0000000000000148. [DOI] [PubMed] [Google Scholar]
- 3.Guay B, Johnson-Obaseki S, McDonald JT, Connell C, Corsten M. Incidence of differentiated thyroid cancer by socioeconomic status and urban residence: Canada 1991–2006. Thyroid: official journal of the American Thyroid Association. 2014;24:552–555. doi: 10.1089/thy.2013.0308. [DOI] [PubMed] [Google Scholar]
- 4.Fagin JA, Mitsiades N. Molecular pathology of thyroid cancer: diagnostic and clinical implications. Best practice & research. Clinical endocrinology & metabolism. 2008;22:955–969. doi: 10.1016/j.beem.2008.09.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cohen Y, et al. BRAF mutation in papillary thyroid carcinoma. Journal of the National Cancer Institute. 2003;95:625–627. doi: 10.1093/jnci/95.8.625. [DOI] [PubMed] [Google Scholar]
- 6.Kimura ET, et al. High prevalence of BRAF mutations in thyroid cancer: genetic evidence for constitutive activation of the RET/PTC-RAS-BRAF signaling pathway in papillary thyroid carcinoma. Cancer research. 2003;63:1454–1457. [PubMed] [Google Scholar]
- 7.Integrated genomic characterization of papillary thyroid carcinoma Integrated genomic characterization of papillary thyroid carcinoma. Cell. 2014;159:676–690. doi: 10.1016/j.cell.2014.09.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lemoine NR, et al. Activated ras oncogenes in human thyroid cancers. Cancer research. 1988;48:4459–4463. [PubMed] [Google Scholar]
- 9.Grieco M, et al. PTC is a novel rearranged form of the ret proto-oncogene and is frequently detected in vivo in human thyroid papillary carcinomas. Cell. 1990;60:557–563. doi: 10.1016/0092-8674(90)90659-3. [DOI] [PubMed] [Google Scholar]
- 10.Pierotti MA, et al. Rearrangements of TRK proto-oncogene in papillary thyroid carcinomas. Journal of endocrinological investigation. 1995;18:130–133. doi: 10.1007/BF03349721. [DOI] [PubMed] [Google Scholar]
- 11.Knauf JA, Fagin JA. Role of MAPK pathway oncoproteins in thyroid cancer pathogenesis and as drug targets. Current opinion in cell biology. 2009;21:296–303. doi: 10.1016/j.ceb.2009.01.013. [DOI] [PubMed] [Google Scholar]
- 12.Kebebew, E. et al. The prevalence and prognostic value of BRAF mutation in thyroid cancer. Annals of surgery246, 466–470, discussion 470–461, doi:10.1097/SLA.0b013e318148563d (2007). [DOI] [PMC free article] [PubMed]
- 13.Xing M, et al. Association between BRAF V600E mutation and mortality in patients with papillary thyroid cancer. Jama. 2013;309:1493–1501. doi: 10.1001/jama.2013.3190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Knauf JA, et al. Targeted expression of BRAFV600E in thyroid cells of transgenic mice results in papillary thyroid cancers that undergo dedifferentiation. Cancer research. 2005;65:4238–4245. doi: 10.1158/0008-5472.CAN-05-0047. [DOI] [PubMed] [Google Scholar]
- 15.Mesa C, Jr., et al. Conditional activation of RET/PTC3 and BRAFV600E in thyroid cells is associated with gene expression profiles that predict a preferential role of BRAF in extracellular matrix remodeling. Cancer research. 2006;66:6521–6529. doi: 10.1158/0008-5472.CAN-06-0739. [DOI] [PubMed] [Google Scholar]
- 16.Pollock PM, et al. High frequency of BRAF mutations in nevi. Nature genetics. 2003;33:19–20. doi: 10.1038/ng1054. [DOI] [PubMed] [Google Scholar]
- 17.Adeniran AJ, et al. Correlation between genetic alterations and microscopic features, clinical manifestations, and prognostic characteristics of thyroid papillary carcinomas. The American journal of surgical pathology. 2006;30:216–222. doi: 10.1097/01.pas.0000176432.73455.1b. [DOI] [PubMed] [Google Scholar]
- 18.Michaloglou C, et al. BRAFE600-associated senescence-like cell cycle arrest of human naevi. Nature. 2005;436:720–724. doi: 10.1038/nature03890. [DOI] [PubMed] [Google Scholar]
- 19.Bartkova J, et al. Oncogene-induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature. 2006;444:633–637. doi: 10.1038/nature05268. [DOI] [PubMed] [Google Scholar]
- 20.Hu S, et al. Association of aberrant methylation of tumor suppressor genes with tumor aggressiveness and BRAF mutation in papillary thyroid cancer. International journal of cancer. Journal international du cancer. 2006;119:2322–2329. doi: 10.1002/ijc.22110. [DOI] [PubMed] [Google Scholar]
- 21.Porra V, et al. Silencing of the tumor suppressor gene SLC5A8 is associated with BRAF mutations in classical papillary thyroid carcinomas. The Journal of clinical endocrinology and metabolism. 2005;90:3028–3035. doi: 10.1210/jc.2004-1394. [DOI] [PubMed] [Google Scholar]
- 22.Guan H, et al. Hypermethylation of the DNA mismatch repair gene hMLH1 and its association with lymph node metastasis and T1799A BRAF mutation in patients with papillary thyroid cancer. Cancer. 2008;113:247–255. doi: 10.1002/cncr.23548. [DOI] [PubMed] [Google Scholar]
- 23.Kim YH, et al. TSH signaling overcomes B-RafV600E-induced senescence in papillary thyroid carcinogenesis through regulation of DUSP6. Neoplasia (New York, N.Y.) 2014;16:1107–1120. doi: 10.1016/j.neo.2014.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rusinek D, et al. BRAFV600E-Associated Gene Expression Profile: Early Changes in the Transcriptome, Based on a Transgenic Mouse Model of Papillary Thyroid Carcinoma. PloS one. 2015;10:e0143688. doi: 10.1371/journal.pone.0143688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Flockhart RJ, et al. BRAFV600E remodels the melanocyte transcriptome and induces BANCR to regulate melanoma cell migration. Genome research. 2012;22:1006–1014. doi: 10.1101/gr.140061.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Goedert, L. et al. RMEL3, a novel BRAFV600E-associated long noncoding RNA, is required for MAPK and PI3K signaling in melanoma. Oncotarget, 8, doi:10.18632/oncotarget.9164 (2016). [DOI] [PMC free article] [PubMed]
- 27.Kapusta A, Feschotte C. Volatile evolution of long noncoding RNA repertoires: mechanisms and biological implications. Trends in genetics: TIG. 2014;30:439–452. doi: 10.1016/j.tig.2014.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Penny GD, Kay GF, Sheardown SA, Rastan S, Brockdorff N. Requirement for Xist in X chromosome inactivation. Nature. 1996;379:131–137. doi: 10.1038/379131a0. [DOI] [PubMed] [Google Scholar]
- 29.Saxena A, Carninci P. Long non-coding RNA modifies chromatin: epigenetic silencing by long non-coding RNAs. BioEssays: news and reviews in molecular, cellular and developmental biology. 2011;33:830–839. doi: 10.1002/bies.201100084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Huang MD, et al. Long non-coding RNA ANRIL is upregulated in hepatocellular carcinoma and regulates cell apoptosis by epigenetic silencing of KLF2. Journal of hematology & oncology. 2015;8:50. doi: 10.1186/s13045-015-0146-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yoon JH, et al. LincRNA-p21 suppresses target mRNA translation. Molecular cell. 2012;47:648–655. doi: 10.1016/j.molcel.2012.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yuan SX, et al. Antisense long non-coding RNA PCNA-AS1 promotes tumor growth by regulating proliferating cell nuclear antigen in hepatocellular carcinoma. Cancer letters. 2014;349:87–94. doi: 10.1016/j.canlet.2014.03.029. [DOI] [PubMed] [Google Scholar]
- 33.Yoon JH, et al. Scaffold function of long non-coding RNA HOTAIR in protein ubiquitination. Nature communications. 2013;4:2939. doi: 10.1038/ncomms3939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Karreth FA, et al. The BRAF pseudogene functions as a competitive endogenous RNA and induces lymphoma in vivo. Cell. 2015;161:319–332. doi: 10.1016/j.cell.2015.02.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Greco S, Gorospe M, Martelli F. Noncoding RNA in age-related cardiovascular diseases. Journal of molecular and cellular cardiology. 2015;83:142–155. doi: 10.1016/j.yjmcc.2015.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lan X, et al. Genome-wide analysis of long noncoding RNA expression profile in papillary thyroid carcinoma. Gene. 2015;569:109–117. doi: 10.1016/j.gene.2015.05.046. [DOI] [PubMed] [Google Scholar]
- 37.Xu B, et al. The Long Non-Coding RNA ENST00000537266 and ENST00000426615 Influence Papillary Thyroid Cancer Cell Proliferation and Motility. Cellular physiology and biochemistry: international journal of experimental cellular physiology, biochemistry, and pharmacology. 2016;38:368–378. doi: 10.1159/000438637. [DOI] [PubMed] [Google Scholar]
- 38.Jendrzejewski J, et al. The polymorphism rs944289 predisposes to papillary thyroid carcinoma through a large intergenic noncoding RNA gene of tumor suppressor type. Proceedings of the National Academy of Sciences of the United States of America. 2012;109:8646–8651. doi: 10.1073/pnas.1205654109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zheng, H. et al. BRAF-activated Long Non-coding RNA Modulates Papillary Thyroid Carcinoma Cell Proliferation through Regulating Thyroid Stimulating Hormone Receptor. Cancer research and treatment: official journal of Korean Cancer Association. doi:10.4143/crt.2015.118 (2015). [DOI] [PMC free article] [PubMed]
- 40.He H, et al. Genetic predisposition to papillary thyroid carcinoma: involvement of FOXE1, TSHR, and a novel lincRNA gene, PTCSC2. The Journal of clinical endocrinology and metabolism. 2015;100:E164–172. doi: 10.1210/jc.2014-2147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yoon H, et al. Identification of a novel noncoding RNA gene, NAMA, that is downregulated in papillary thyroid carcinoma with BRAF mutation and associated with growth arrest. International journal of cancer. Journal international du cancer. 2007;121:767–775. doi: 10.1002/ijc.22701. [DOI] [PubMed] [Google Scholar]
- 42.Jendrzejewski J, et al. PTCSC3 Is Involved in Papillary Thyroid Carcinoma Development by Modulating S100A4 Gene Expression. The Journal of clinical endocrinology and metabolism. 2015;100:1370–1377. doi: 10.1210/jc.2015-2247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Li, W. B. et al. Identification of Genes Associated with Papillary Thyroid Carcinoma (PTC) for Diagnosis by Integrated Analysis. Hormone and metabolic research = Hormon- und Stoffwechselforschung = Hormones et metabolisme, doi:10.1055/s-0035-1569289 (2016). [DOI] [PubMed]
- 44.Pramoonjago P, Baras AS, Moskaluk CA. Knockdown of Sox4 expression by RNAi induces apoptosis in ACC3 cells. Oncogene. 2006;25:5626–5639. doi: 10.1038/sj.onc.1209566. [DOI] [PubMed] [Google Scholar]
- 45.Matin RN, et al. p63 is an alternative p53 repressor in melanoma that confers chemoresistance and a poor prognosis. The Journal of experimental medicine. 2013;210:581–603. doi: 10.1084/jem.20121439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Mulder KM. Role of Ras and Mapks in TGFbeta signaling. Cytokine & growth factor reviews. 2000;11:23–35. doi: 10.1016/S1359-6101(99)00026-X. [DOI] [PubMed] [Google Scholar]
- 47.Chuderland D, Seger R. Calcium regulates ERK signaling by modulating its protein-protein interactions. Communicative & integrative biology. 2008;1:4–5. doi: 10.4161/cib.1.1.6107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rosen LB, Ginty DD, Weber MJ, Greenberg ME. Membrane depolarization and calcium influx stimulate MEK and MAP kinase via activation of Ras. Neuron. 1994;12:1207–1221. doi: 10.1016/0896-6273(94)90438-3. [DOI] [PubMed] [Google Scholar]
- 49.Cerami E, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer discovery. 2012;2:401–404. doi: 10.1158/2159-8290.CD-12-0095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Li J, et al. TANRIC: An Interactive Open Platform to Explore the Function of lncRNAs in Cancer. Cancer research. 2015;75:3728–3737. doi: 10.1158/0008-5472.CAN-15-0273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Djebali S, et al. Landscape of transcription in human cells. Nature. 2012;489:101–108. doi: 10.1038/nature11233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Robinson MD, McCarthy DJ, Smyth GK. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics (Oxford, England) 2010;26:139–140. doi: 10.1093/bioinformatics/btp616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Colaprico, A. et al. TCGAbiolinks: an R/Bioconductor package for integrative analysis of TCGA data. Nucleic Acids Research. https://www.bioconductor.org/packages/release/bioc/html/TCGAbiolinks.html (2015). [DOI] [PMC free article] [PubMed]
- 54.Han L, et al. LncRNA pro fi le of glioblastoma reveals the potential role of lncRNAs in contributing to glioblastoma pathogenesis. International journal of oncology. 2012;40:2004–2012. doi: 10.3892/ijo.2012.1413. [DOI] [PubMed] [Google Scholar]
- 55.Terai G, Iwakiri J, Kameda T, Hamada M, Asai K. Comprehensive prediction of lncRNA-RNA interactions in human transcriptome. BMC genomics. 2016;17(Suppl 1):12. doi: 10.1186/s12864-015-2307-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Miranda KC, et al. A pattern-based method for the identification of MicroRNA binding sites and their corresponding heteroduplexes. Cell. 2006;126:1203–1217. doi: 10.1016/j.cell.2006.07.031. [DOI] [PubMed] [Google Scholar]
- 57.Huang da W, Sherman BT, Lempicki RA. Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic acids research. 2009;37:1–13. doi: 10.1093/nar/gkn923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kanehisa M, Goto S. KEGG: kyoto encyclopedia of genes and genomes. Nucleic acids research. 2000;28:27–30. doi: 10.1093/nar/28.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Lu TP, et al. miRSystem: an integrated system for characterizing enriched functions and pathways of microRNA targets. PloS one. 2012;7:e42390. doi: 10.1371/journal.pone.0042390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Warde-Farley D, et al. The GeneMANIA prediction server: biological network integration for gene prioritization and predicting gene function. Nucleic acids research. 2010;38:W214–220. doi: 10.1093/nar/gkq537. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


