Abstract
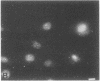
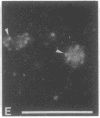
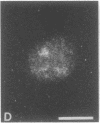
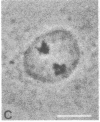
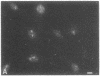
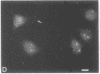
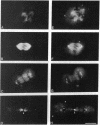
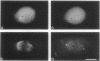
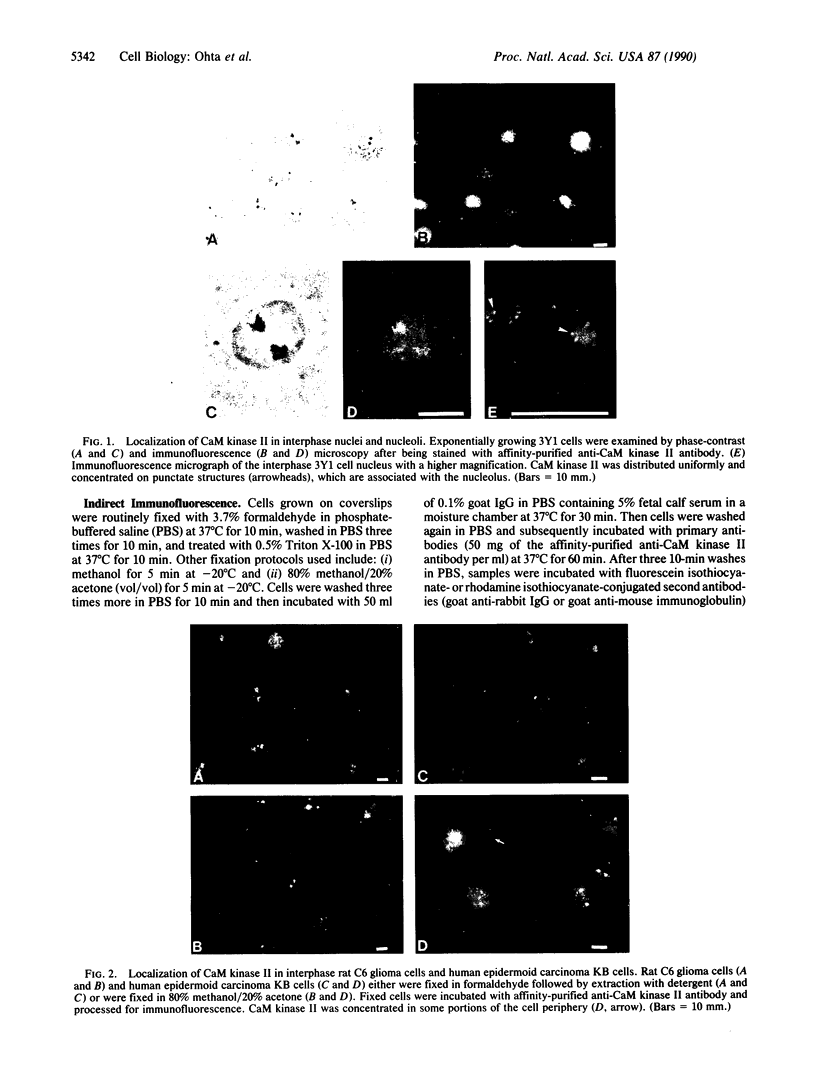
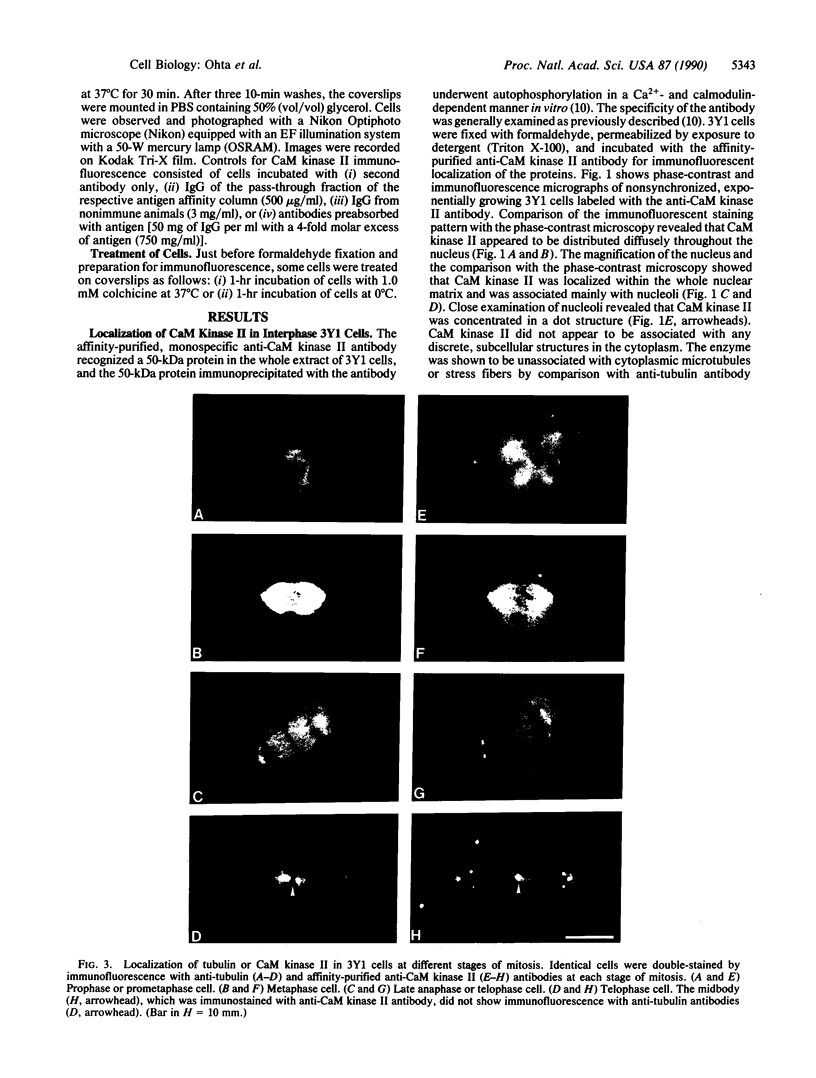
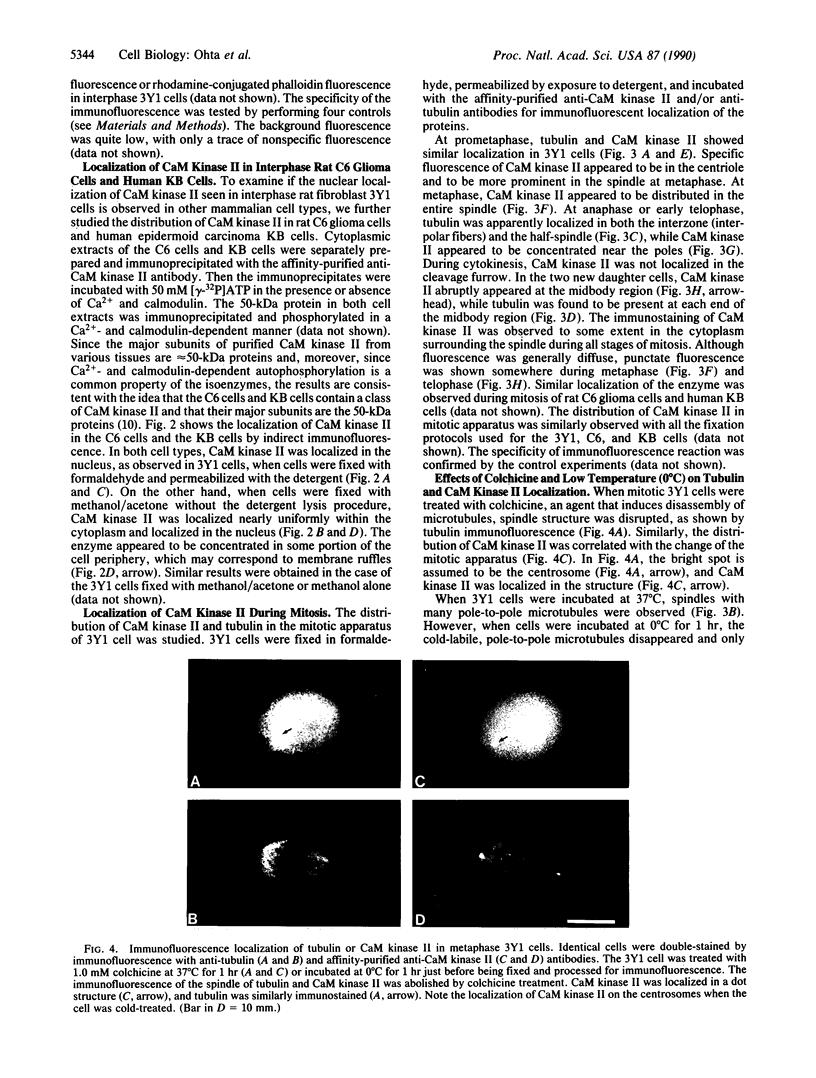
Indirect immunofluorescence was used to determine the distribution of Ca2+/calmodulin-dependent protein kinase II (CaM kinase II) in rat embryo fibroblast 3Y1 cells, rat C6 glioma cells, and human epidermoid carcinoma KB cells. During interphase at growing phase, CaM kinase II was localized diffusely in the cytoplasm and in the nucleus. In the nucleus, the enzyme was localized within the whole nuclear matrix in which the enzyme was specially concentrated in nucleoli. During mitosis, CaM kinase II was found to be a dynamic component of the mitotic apparatus, particularly present at microtubule-organizing centers. In metaphase and anaphase, CaM kinase II was observed at centrosomes and between the spindle poles. During telophase, CaM kinase II was condensed as a bright fluorescent dot at the midzone of the intercellular bridge between two daughter cells, while tubulin was found at each side of the midbody. Colchicine, a microtubule inhibitor, disorganized the tubulin- and CaM kinase II specific fluorescent structure of mitotic 3Y1 cells. In cold-treated cells, CaM kinase II was localized predominantly at centrosomes. The localization of CaM kinase II in the cell nucleus and the mitotic apparatus suggests that the enzyme may play a role in the cell cycle progression of mammalian cells.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brinkley B. R. Microtubule organizing centers. Annu Rev Cell Biol. 1985;1:145–172. doi: 10.1146/annurev.cb.01.110185.001045. [DOI] [PubMed] [Google Scholar]
- Chackalaparampil I., Shalloway D. Altered phosphorylation and activation of pp60c-src during fibroblast mitosis. Cell. 1988 Mar 25;52(6):801–810. doi: 10.1016/0092-8674(88)90422-9. [DOI] [PubMed] [Google Scholar]
- Connolly J. A., Kalnins V. I., Cleveland D. W., Kirschner M. W. Intracellular localization of the high molecular weight microtubule accessory protein by indirect immunofluorescence. J Cell Biol. 1978 Mar;76(3):781–786. doi: 10.1083/jcb.76.3.781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis F. M., Tsao T. Y., Fowler S. K., Rao P. N. Monoclonal antibodies to mitotic cells. Proc Natl Acad Sci U S A. 1983 May;80(10):2926–2930. doi: 10.1073/pnas.80.10.2926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinsmore J. H., Sloboda R. D. Calcium and calmodulin-dependent phosphorylation of a 62 kd protein induces microtubule depolymerization in sea urchin mitotic apparatuses. Cell. 1988 Jun 3;53(5):769–780. doi: 10.1016/0092-8674(88)90094-3. [DOI] [PubMed] [Google Scholar]
- Draetta G., Beach D. Activation of cdc2 protein kinase during mitosis in human cells: cell cycle-dependent phosphorylation and subunit rearrangement. Cell. 1988 Jul 1;54(1):17–26. doi: 10.1016/0092-8674(88)90175-4. [DOI] [PubMed] [Google Scholar]
- Dunphy W. G., Newport J. W. Unraveling of mitotic control mechanisms. Cell. 1988 Dec 23;55(6):925–928. doi: 10.1016/0092-8674(88)90234-6. [DOI] [PubMed] [Google Scholar]
- Edelman A. M., Blumenthal D. K., Krebs E. G. Protein serine/threonine kinases. Annu Rev Biochem. 1987;56:567–613. doi: 10.1146/annurev.bi.56.070187.003031. [DOI] [PubMed] [Google Scholar]
- Guerriero V., Jr, Rowley D. R., Means A. R. Production and characterization of an antibody to myosin light chain kinase and intracellular localization of the enzyme. Cell. 1981 Dec;27(3 Pt 2):449–458. doi: 10.1016/0092-8674(81)90386-x. [DOI] [PubMed] [Google Scholar]
- Hunter T. A thousand and one protein kinases. Cell. 1987 Sep 11;50(6):823–829. doi: 10.1016/0092-8674(87)90509-5. [DOI] [PubMed] [Google Scholar]
- Izant J. G., Weatherbee J. A., McIntosh J. R. A microtubule-associated protein in the mitotic spindle and the interphase nucleus. Nature. 1982 Jan 21;295(5846):248–250. doi: 10.1038/295248a0. [DOI] [PubMed] [Google Scholar]
- Job D., Rauch C. T., Fischer E. H., Margolis R. L. Regulation of microtubule cold stability by calmodulin-dependent and -independent phosphorylation. Proc Natl Acad Sci U S A. 1983 Jul;80(13):3894–3898. doi: 10.1073/pnas.80.13.3894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai Y., Nairn A. C., Greengard P. Autophosphorylation reversibly regulates the Ca2+/calmodulin-dependence of Ca2+/calmodulin-dependent protein kinase II. Proc Natl Acad Sci U S A. 1986 Jun;83(12):4253–4257. doi: 10.1073/pnas.83.12.4253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller S. G., Kennedy M. B. Regulation of brain type II Ca2+/calmodulin-dependent protein kinase by autophosphorylation: a Ca2+-triggered molecular switch. Cell. 1986 Mar 28;44(6):861–870. doi: 10.1016/0092-8674(86)90008-5. [DOI] [PubMed] [Google Scholar]
- Ohta Y., Ohba T., Fukunaga K., Miyamoto E. Serum and growth factors rapidly elicit phosphorylation of the Ca2+/calmodulin-dependent protein kinase II in intact quiescent rat 3Y1 cells. J Biol Chem. 1988 Aug 15;263(23):11540–11547. [PubMed] [Google Scholar]
- Ratan R. R., Maxfield F. R., Shelanski M. L. Long-lasting and rapid calcium changes during mitosis. J Cell Biol. 1988 Sep;107(3):993–999. doi: 10.1083/jcb.107.3.993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riabowol K., Draetta G., Brizuela L., Vandre D., Beach D. The cdc2 kinase is a nuclear protein that is essential for mitosis in mammalian cells. Cell. 1989 May 5;57(3):393–401. doi: 10.1016/0092-8674(89)90914-8. [DOI] [PubMed] [Google Scholar]
- Sahyoun N., LeVine H., 3rd, Bronson D., Cuatrecasas P. Ca2+-calmodulin-dependent protein kinase in neuronal nuclei. J Biol Chem. 1984 Aug 10;259(15):9341–9344. [PubMed] [Google Scholar]
- Sahyoun N., LeVine H., 3rd, Bronson D., Siegel-Greenstein F., Cuatrecasas P. Cytoskeletal calmodulin-dependent protein kinase. Characterization, solubilization, and purification from rat brain. J Biol Chem. 1985 Jan 25;260(2):1230–1237. [PubMed] [Google Scholar]
- Schworer C. M., Colbran R. J., Soderling T. R. Reversible generation of a Ca2+-independent form of Ca2+(calmodulin)-dependent protein kinase II by an autophosphorylation mechanism. J Biol Chem. 1986 Jul 5;261(19):8581–8584. [PubMed] [Google Scholar]
- Vandre D. D., Davis F. M., Rao P. N., Borisy G. G. Phosphoproteins are components of mitotic microtubule organizing centers. Proc Natl Acad Sci U S A. 1984 Jul;81(14):4439–4443. doi: 10.1073/pnas.81.14.4439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welsh M. J., Dedman J. R., Brinkley B. R., Means A. R. Tubulin and calmodulin. Effects of microtubule and microfilament inhibitors on localization in the mitotic apparatus. J Cell Biol. 1979 Jun;81(3):624–634. doi: 10.1083/jcb.81.3.624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westwood J. T., Church R. B., Wagenaar E. B. Changes in protein phosphorylation during the cell cycle of Chinese hamster ovary cells. J Biol Chem. 1985 Aug 25;260(18):10308–10313. [PubMed] [Google Scholar]
- Willingham M. C., Wehland J., Klee C. B., Richert N. D., Rutherford A. V., Pastan I. H. Ultrastructural immunocytochemical localization of calmodulin in cultured cells. J Histochem Cytochem. 1983 Apr;31(4):445–461. doi: 10.1177/31.4.6338107. [DOI] [PubMed] [Google Scholar]
- Wordeman L., Cande W. Z. Reactivation of spindle elongation in vitro is correlated with the phosphorylation of a 205 kd spindle-associated protein. Cell. 1987 Aug 14;50(4):535–543. doi: 10.1016/0092-8674(87)90026-2. [DOI] [PubMed] [Google Scholar]
- Yamamoto H., Fukunaga K., Tanaka E., Miyamoto E. Ca2+- and calmodulin-dependent phosphorylation of microtubule-associated protein 2 and tau factor, and inhibition of microtubule assembly. J Neurochem. 1983 Oct;41(4):1119–1125. doi: 10.1111/j.1471-4159.1983.tb09060.x. [DOI] [PubMed] [Google Scholar]
- Yamamoto K. K., Gonzalez G. A., Biggs W. H., 3rd, Montminy M. R. Phosphorylation-induced binding and transcriptional efficacy of nuclear factor CREB. Nature. 1988 Aug 11;334(6182):494–498. doi: 10.1038/334494a0. [DOI] [PubMed] [Google Scholar]