Supplemental Digital Content is available in the text.
Keywords: antibiotic-associated colitis, epithelial apoptosis, fecal microbiota transplantation, high-volume diarrhea, microbiota depletion
Abstract
Objective:
Antibiotic therapy is a major risk factor for the development of diarrhea and colitis with varying severity. Often the origin of antibiotic-associated gastrointestinal deterioration remains elusive and no specific infectious agents could be discerned.
Patients:
We represent three cases of intractable high-volume diarrhea associated with combined antibiotic and steroid therapy in critically ill patients not fitting into established disease entities. Cases presented with severe apoptotic enterocolitis resembling acute intestinal graft-versus-host-disease. Microbiologic workup precluded known enteropathogens, but microbiota analysis revealed a severely depleted gut microbiota with concomitant opportunistic pathogen overgrowth.
Interventions:
Fecal microbiota transplantation, performed in one patient, was associated with correction of dysbiosis, rapid clinical improvement, and healing of enterocolitis.
Conclusions:
Our series represents a severe form of antibiotic-associated colitis in critically ill patients signified by microbiota depletion, and reestablishment of a physiologic gastrointestinal microbiota might be beneficial for this condition.
Diarrhea and colitis are frequent complications of antibiotic treatment, particularly in the critically ill (1, 2). Besides direct drug-induced toxicity of antibiotics, depletion of the endogenous microbiota and subsequent pathogen overgrowth are major disease causes, exemplified by colitis due to Clostridium difficile and Klebsiella oxytoca (3, 4). Importantly, in many cases of antibiotic-associated colitis (AAC), no causal pathogens can be identified (5). In addition, immunosuppression sets individuals at risk for diarrhea and gastrointestinal inflammation (6, 7). A balanced interaction between the intestinal microbiota and mucosal immune system is required for homeostasis of the gastrointestinal tract (8). Thus, combined antibiotic and immunosuppressant therapies may disturb microbiota-gut homeostasis even more than individual treatments. In this context, we report on a series of severe apoptotic enterocolitis of critically ill patients following combined treatment with broad-spectrum antibiotics and steroids. Cases showed a severe depletion of the endogenous gastrointestinal microbiota, and the condition might be improved by restoration of a physiologic microbiota, for example, by means of fecal microbiota transplantation (FMT).
CASE DESCRIPTIONS
Case A, a 28-year-old woman was hospitalized for fever of unknown origin and subsequently admitted to the medical ICU for systemic inflammatory response syndrome (SIRS). Prior to ICU admission, 100 mg/d prednisolone was initiated because of suspected Still’s disease. SIRS persisted and several empiric courses of antibiotics were given. No infectious etiology for SIRS was detectable. Diarrhea started on ICU day 15, the patient still being under steroid therapy, and aggravated to peak stool volumes of up to 4.4 L/d. Diarrhea was accompanied by severe vomiting resulting in inability for oral nutritional intake for 14 days. Cessation of antibiotics and steroid tapering was accompanied by a decrease of diarrhea. The patient clinically improved and was transferred to the normal ward to receive fluid and electrolyte replacement therapy for 2 further weeks. Diarrhea subsided after 45 days.
Case B, a 46-year-old man with rheumatoid arthritis was hospitalized for methotrexate-induced pneumonitis. He was admitted to the medical ICU due to acute respiratory distress syndrome (ARDS) and SIRS to receive mechanical ventilation and antibiotic therapy. No infectious etiology for ARDS or SIRS was detectable. Glucocorticoid therapy initiated 8 months ahead was stopped at hospital admission but recommenced to 50 mg/d prednisolone upon ICU admission. On ICU day 25, massive vomiting and watery diarrhea developed, which reached peak volumes of up to 6 L/d. Despite discontinuation of steroids and changes in the antibiotic therapy regimen, severe diarrhea persisted and the patient died from multiple organ failure at ICU day 66.
Case C, a 16-year-old girl was admitted to the neurosurgical ICU after multiple trauma including severe head injury subsequent to a car accident. Dexamethasone (80 mg/d) and antibiotic therapy were initiated. On ICU day 11, diarrhea started and culminated in stool volumes of 7.2 L/d on ICU day 39 accompanied by high gastric residue with inability of adequate enteral nutrition. Conservative approaches including probiotic supplementation administered over 42 days did neither lead to reduced diarrheal volumes nor to endoscopic or histologic improvement. Seventy-two days after onset of diarrhea, FMT was performed according to a recently described protocol on compassionate use basis (9). Donor feces was provided by the patient’s mother, and a total of 400 mL of fecal solution was instilled into the ileum and colon by ileocolonoscopy. Two days after FMT, diarrhea improved with significantly decreased stool volumes (1 L/d), leading finally to full clinical and histologic recovery (last follow-up 97 wk after FMT).
Detailed descriptions of the individual disease courses and therapies applied are shown in Fig. S1 (Supplemental Digital Content 1, http://links.lww.com/CCM/C408) and Tables S1 and S2 (Supplemental Digital Content 2, http://links.lww.com/CCM/C409). Microbiologic workup included repeated testing for C. difficile (polymerase chain reaction for C. difficile toxin A and B, enzyme-linked immunosorbent assay C. difficile toxin A), K. oxytoca, campylobacter, salmonella, yersinia, shigella, Enterohemorrhagic Escherichia coli in stools of all cases and testing for viruses in biopsies and blood (Tables S3 and S4, Supplemental Digital Content 2, http://links.lww.com/CCM/C409). All these tests did not reveal any enteropathogen in any case. Additional microbiologic investigations done are shown in Table S5 (Supplemental Digital Content 2, http://links.lww.com/CCM/C409).
METHODS
Details for methods used are given in the supplemental material (Supplemental Digital Content 1, http://links.lww.com/CCM/C408). Mucosal biopsies and stool samples for microbiota analyses (cases B and C) were frozen at –20°C until further processing. 16S rRNA gene pyrosequencing data have been deposited in the European Nucleotide Archive under the accession number ERP013256. Investigations were approved by the review board of the Medical University of Graz (EK-23-212-ex-10/11). Informed consent for FMT (compassionate use) was obtained from the patient’s guardian.
RESULTS
Endoscopic and Histopathologic Findings in Antibiotic-Associated Apoptotic (AAA) Enterocolitis
All patients underwent repeated gastro-duodenoscopies and colonoscopies. At presentation, a severe inflammation was evident in the small and large intestines (Fig. 1) (Fig. S2, Supplemental Digital Content 1, http://links.lww.com/CCM/C408). Macroscopically, segmental loss of folds and large erosions were seen in the duodenum and terminal ileum of all patients, whereas colonic manifestations varied from mosaic-like patterns to large “geographic” ulcerations and areas with a completely denuded mucosa. The histopathologic hallmark feature was epithelial mass apoptosis reminiscent of severe acute graft-versus-host-disease (GvHD) leading to (sub)total crypt losses and moderate-to-severe segmental villus blunting in the small intestines, colonic features ranged from individual crypt losses to large areas with completely lost epithelium (Table S4, Supplemental Digital Content 2, http://links.lww.com/CCM/C409). Goblet and Paneth cells were detectable in all cases, no signs suggestive of viral infection were evident, and repeated testing of biopsies for viruses was negative (Table S4, Supplemental Digital Content 2, http://links.lww.com/CCM/C409). The lamina propria showed a moderately dense mixed infiltrate, CD8+ T cells were significantly increased in the colon, and CD4+ T cells concomitantly reduced (CD4-to-CD8, 1:19; normal, 3:1). Regulatory T cells were not changed compared with healthy controls (Fig. S3, Supplemental Digital Content 1, http://links.lww.com/CCM/C408).
Figure 1.

Endoscopy and histology in antibiotic-associated apoptotic (AAA) enterocolitis. (A) Hemorrhagic inflammation with loss of folds in the duodenum (top). Complete loss of vascularization, “geographic” ulcerations with abundant fibrin and denudation in the colon (bottom). (B) Segmental loss of crypts and villi and abundant apoptoses (arrows) in the duodenal mucosa (inset: activated caspase-3 positivity). (C) Large areas completely devoid of crypts and surface epithelium in the colon (inset: crypt epithelial apoptosis). Activated caspase-3 positive crypt epithelial cells and cell debris (bottom). Images: A (top) and B, case A onset of diarrhea; A (bottom), case C 45 d after diarrhea onset; C (top), case C 15 d after diarrhea onset; C (bottom) and inset C, case C 17 d after onset of diarrhea.
Microbiota Profile in AAA Enterocolitis
The clinical findings in the index patient (case A) prompted us to perform microbiota analyses in cases B and C subsequently. Fecal and mucosal specimens collected during acute disease showed a severely depleted microbiota characterized by significantly reduced richness, diversity, and microbial load, as well as an overabundance of Proteobacteria (99.9% ± 0.1% in case B, 98.6% ± 1.9% in case C), and minimal fractions of Firmicutes (0.02% ± 0.03%) and Bacteroidetes (0.01% ± 0.02%) (Fig. 2, A–C) (Fig. S4, Supplemental Digital Content 1, http://links.lww.com/CCM/C408). Interestingly, microbiota underwent extreme compositional shifts over time. In case B, concentrations of a single Pseudomonas taxon shifted from 98.8% on day 2 to 2% ± 0.5% on day 24, with the latter time point being dominated by an unclassified Enterobacteriaceae (97.5% ± 0.9%). In case C, samples were dominated by different strains from an administered probiotic (Lactobacillus species, 60.4% ± 18.7% on day 33; Enterococcus species, 52.6% ± 9.5% on day 46), as well as typical skin commensals (Staphylococcus epidermidis 34.5% ± 10.4% and Propionibacterium acnes 1.8% ± 3.2%). After cessation of probiotics Haemophilus parainfluenzae, a Proteobacterium typically colonizing the oropharynx, accounted for 74.2% ± 21.2% (Fig. 2A). Taken together, these dramatic and apparently random fluctuations in gut microbiota composition are suggestive of a lost intrinsic colonization resistance of the gut microbial ecosystem (10).
Figure 2.
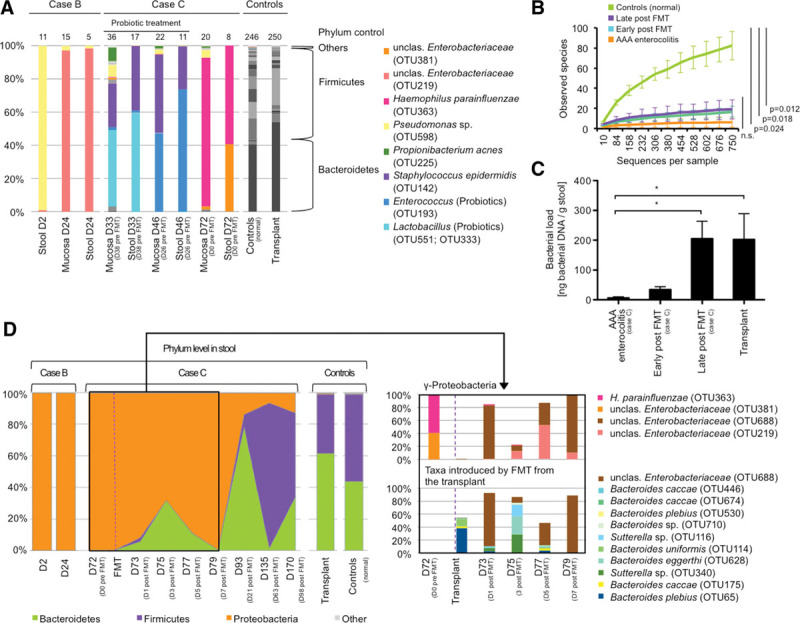
Microbiota depletion and effects of fecal microbiota transplantation (FMT) in antibiotic-associated apoptotic (AAA) enterocolitis. (A) Fluctuating taxa during acute disease in cases B and C. A normal colonic microbiota is shown as control (gray bars). The numbers above bars indicate number of prevalent taxa. (B) Microbial richness is impaired in AAA enterocolitis. Neither probiotic treatment nor FMT is able to restore reduced richness (Two-sample t test, 999 Monte-Carlo permutations, Bonferroni-correction, samples were rarefied to 750 sequences per sample; sample designations are given in Table S4). (C) Microbial load is significantly reduced and increases after FMT (one-way analysis of variance [ANOVA], Bonferroni posttest; * p < 0,05). (D) Phylum level microbiota dynamics in case C before and after FMT compared with case B and controls (left). Operational taxonomic unit (OTU) level analysis indicates removal of Haemophilus parainfluenzae after FMT and recolonization with Gammaproteobacteria followed by Bacteroidetes originating from the transplant (right).
Attenuation of Severe Dysbiosis by FMT Was Associated With Resolution of Symptoms and Mucosal Healing in a Patient With AAA Enterocolitis
Case C clinically deteriorated, severe diarrhea persisted (2–7 L/d), and endoscopy showed persistent severe inflammation predominantly in the left colon. Given the severe microbiota depletion, FMT was performed on compassionate use basis 72 days after diarrhea onset. The transplant represented a physiologic fecal microbiota (61.7% Bacteroidetes and 37.3% Firmicutes; Fig. 2D). After FMT stool volumes continuously declined and sigmoidoscopy showed improvement as early as 7 days after FMT, histologically confirmed by regenerating crypts and a reestablished epithelial lining (Fig. 3A). By day 1 after FMT, H. parainfluenzae was considerably reduced in the feces (1.5%). Initially, the patient’s fecal microbiota was dominated by Enterobacteriaceae, originating from the transplant, followed by reestablishment of a physiologic colonic microbiota dominated by Bacteroidetes and Firmicutes (Fig. 2D). Bacterial load in stools significantly increased over time (Fig. 2C) although levels of microbial richness and diversity remained reduced compared with controls (Fig. 2B) (Fig. S4, Supplemental Digital Content 1, http://links.lww.com/CCM/C408). During the acute phase of disease, short-chain fatty acids (SCFAs) in stools were reduced. After FMT, acetate and subsequently propionate levels increased (Fig. S5, Supplemental Digital Content 1, http://links.lww.com/CCM/C408) and butyrate levels remained low, consistent with reduced amounts of butyrate-producing genes in the fecal metagenome (Fig. S6, Supplemental Digital Content 1, http://links.lww.com/CCM/C408). The CD4-to-CD8 ratio reversed to normal levels in colonic biopsies after FMT; no significant changes in regulatory T-cell counts were detected (Fig. 3B). Ninety-seven days after FMT, the gastrointestinal mucosa showed a normal gross appearance (Fig. S7, Supplemental Digital Content 1, http://links.lww.com/CCM/C408).
Figure 3.
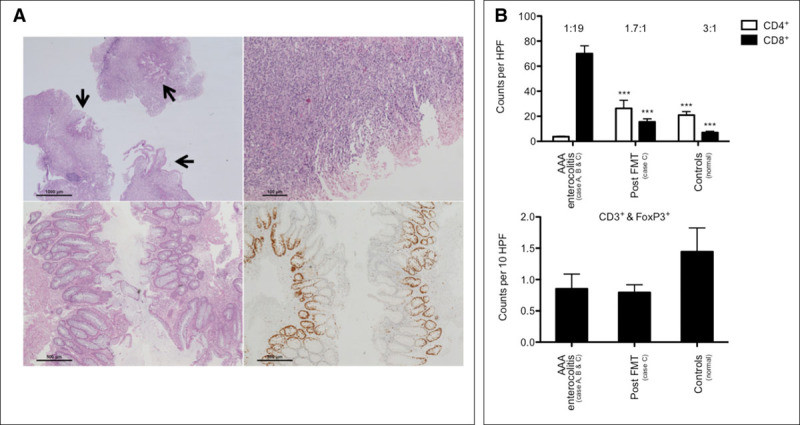
Histopathology and mucosal immunophenotype pre- and post fecal microbiota transplantation (FMT). (A) Colon histopathology 12 d ahead (top left) and at the day of FMT (top right). Only focal residual epithelium is present (arrows). Seven days after FMT, the epithelial lining is reestablished (bottom left). Ki-67 immunohistochemistry identifies proliferating crypt epithelia (bottom right). (B) Significantly increased CD8+ T cells and significantly reduced CD4+ T cells during acute disease in the colon, this immunophenotype is reversible after FMT (top); CD4-to-CD8 ratio is given above bars (one-way analysis of variance [ANOVA], Bonferroni-corrected, *** p < 0.0001). CD3+FoxP3+ double-positive Tregs are not significantly altered (bottom; one-way ANOVA, p = 0.2801). AAA = antibiotic-associated apoptotic, HPF = high power fields.
DISCUSSION
We herein describe severe antibiotic-associated enterocolitis in patients treated at the ICU because of a severe underlying disease. All three patients exhibited several common features: 1) all patients were concomitantly treated with broad-spectrum antibiotics and steroids when intractable watery high-volume diarrhea and gastrointestinal failure started with sudden onset; 2) histopathology revealed severe apoptotic enteropathy, reminiscent of acute GvHD in the absence of transplantations; and 3) despite extensive microbiologic workup, no established enteropathogens could be found. The disease course was death in one patient, spontaneous recovery after discharge from the ICU after 2 months in the second patient, and improvement to FMT in the third patient.
Several recent reports indicate that critical illness is often accompanied by gastrointestinal microbiota depletion and severe insult or trauma alone, already favor this condition (11, 12). In our cases, it is likely that the prolonged antibiotic therapy lead to microbiota depletion but also additional factors like other medications given (e.g., high doses of proton pump inhibitors) might have aggravated the situation (13–15). Importantly, such patients may fail to reestablish a normal microbiota, as they lack direct microbial input from the diet as well as microbial growth substrates (e.g., fiber) due to parenteral nutrition or sterile tube feeding (Table S2, Supplemental Digital Content 2, http://links.lww.com/CCM/C409). A lack of intestinal colonization resistance is especially highlighted by the observation that noncommensal probiotic bacteria could transiently colonize but without altering disease course (16). Depleted ecological niches may predispose for opportunistic pathogen colonization (17). The preferential dominance of facultative anaerobic Proteobacteria might be due to high prevalence of antibiotic resistance in this taxonomic group and due to their ability to proliferate during inflammation and oxidative stress, which has been described also for Haemophilus, the opportunist identified in case C (18, 19). Intriguingly, a recent study of antibiotic-induced microbiota depletion in mice showed that the persisting taxon, a Pseudomonas similarly as in case B, was able to inhibit mitochondrial gene expression and biogenesis leading to apoptotic epithelial cell death phenotypes similar to our findings (20). In addition, the gastrointestinal pathogenicity of Pseudomonas is highlighted also by its ability to cause gut-derived septicemia (21). Furthermore, reduced concentrations of SCFAs, major metabolites of microbial fermentation required for mucosal homeostasis, have been shown to drive mass apoptosis of the gastrointestinal epithelium (22, 23). In case C, SCFAs in stools were highly reduced consistent with reduced amounts of butyrate-producing genes in feces. Thus, direct effects of persisting opportunists like Haemophilus or Pseudomonas, the lack of SCFAs after microbiota depletion in addition to toxicity of antibiotics might altogether promote epithelial apoptosis and enterocolitis.
Of note, the term “AAA” enterocolitis employed in this report is used in a descriptive manner highlighting the histologic hallmark features but is not specifying a distinct entity. Several other conditions like infections (e.g., cytomegalovirus), immunodeficiencies, autoimmune enteropathy, cord colitis, or thymoma could be accompanied by gastrointestinal epithelial apoptosis; however, the severe phenotype reported here is mainly seen in intestinal GvHD (24, 25). Interestingly, cytotoxic CD8+ T cells were significantly increased in the colon, CD4+ T cells concomitantly reduced during acute disease, whereas regulatory T cells were not changed in cases. A similar immune phenotype is reported for GvHD, which is also signified by severe dysbiosis suggesting parallels in the pathogeneses of GvHD and the enterocolitis observed in our cases (26–28). Of note, the CD4-to-CD8 ratio reversed to normal levels in colonic biopsies in case C after FMT.
AAC constitutes one of the most frequent side effects of antimicrobial therapy with variable clinical presentation (1, 5). Often the cause of AAC remains elusive; thus, new clues about disease pathogenesis are needed. Our series and recent reports point towards a role of microbiota depletion as a possible contributing factor, wherein overgrowth of opportunistic pathogens and also the lack of microbial metabolites, like SCFAs, might support disease development. Attention to new, alternative disease models for AAC are therefore warranted. Our case series, however, is not able to infer causality between reported findings and the pathogenesis of enterocolitis, since other so far not identified factors might have also contributed. For instance, our standard microbiology and 16S rRNA gene-based microbiota investigations might have missed certain pathogens, such as virus or fungi not captured with the applied methods.
Although FMT seemed to have a beneficial effect on the course of patient C, also other factors could have contributed to improvement such as withdrawal of antibiotics, discontinuation of steroids, and continued supportive care as occurred with patient A. FMT is an established treatment for therapy-refractory recurrent C. difficile infection (CDI) (29–31) and is also increasingly used for severe and life-threatening CDI (32–35). Despite application of billions of microorganisms into an inflamed gastrointestinal tract, which poses the danger of initiating gut-derived septicemia, short-term infectious complications seem to be relatively rare (36, 37). Even in immunocompromised patients, only few side effects were reported (38). Dangers are related mostly to the application form of FMT (36, 37), like aspiration of fecal suspension after application into the upper gastrointestinal tract, leading to severe pneumonia and death (39, 40). In contrast, application into the lower gastrointestinal tract seems to have a better safety profile (36, 41). Nevertheless, translocation of microorganisms leading to septicemia poses a severe complication that needs to be considered. FMT might also lead to overstimulation of the immune system, which has been recognized as inflammatory bowel disease flares after FMT (42). Although most reported side effects are self-limited, long-term safety data of FMT are missing (34) and physicians should consider this especially if they plan to use FMT as a treatment option in non-CDI indications on a compassionate use basis.
In conclusion, our report outlines severe enterocolitis in critically ill patients associated with antibiotic and steroid treatment. It is characterized by severe apoptosis of the gastrointestinal epithelium that might be the consequence of a deteriorated microbiota-gastrointestinal tract homeostasis. Reestablishment of a physiologic gastrointestinal microbiota, which might not spontaneously happen in the ICU but could be achieved by FMT, possibly relieves this condition (29, 43, 44).
ACKNOWLEDGMENTS
We thank S. Schauer, E. Steinbauer, M. Gogg-Kammerer, S. Eidenhammer, I. Klymiuk, M. Trötzmüller, and H. Köfeler for technical support and W. F. Fricke for critically reviewing the article.
Supplementary Material
Footnotes
*See also p. 1106.
Mr. Wurm and Dr. Spindelboeck contributed equally. Mr. Wurm, Dr. Spindelboeck, Dr. Högenauer, and Dr. Gorkiewicz contributed in conceptualization and methodology. Mr. Wurm, Dr. Spindelboeck, Dr. Krause, Dr. Plank, Dr. Fuchs, Mr. Bashir, Dr. Petritsch, Dr. Langner, Dr. Högenauer, and Dr. Gorkiewicz contributed in investigations and formal analysis. Drs. Högenauer and Gorkiewicz contributed in writing—original draft; Mr. Wurm, Dr. Spindelboeck, Dr. Petritsch, Dr. Halwachs, Dr. Krause, Dr. Plank, Mr. Bashir, Dr. Högenauer, and Dr. Gorkiewicz contributed in writing—review and editing; Dr. Gorkiewicz contributed in funding acquisition; Drs. Halwachs, Högenauer, and Gorkiewicz contributed in resources; Drs. Högenauer and Gorkiewicz contributed in supervision.
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal’s website (http://journals.lww.com/ccmjournal).
Supported, in part, by the Austrian Science Fund (FWF W1241-B18) and BioTechMed Graz.
Mr. Wurm, Dr. Bashir, Dr. Halwachs, and Dr. Gorkiewicz received support for article research from the Austrian Science Fund (FWF) and BioTechMed Graz. The remaining authors have disclosed that they do not have any potential conflicts of interest.
REFERENCES
- 1.Högenauer C, Hammer HF, Krejs GJ, et al. Mechanisms and management of antibiotic-associated diarrhea. Clin Infect Dis 1998; 27:702–710. [DOI] [PubMed] [Google Scholar]
- 2.Reintam Blaser A, Deane AM, Fruhwald S. Diarrhoea in the critically ill. Curr Opin Crit Care 2015; 21:142–153. [DOI] [PubMed] [Google Scholar]
- 3.Leffler DA, Lamont JT. Clostridium difficile infection. N Engl J Med 2015; 372:1539–1548. [DOI] [PubMed] [Google Scholar]
- 4.Högenauer C, Langner C, Beubler E, et al. Klebsiella oxytoca as a causative organism of antibiotic-associated hemorrhagic colitis. N Engl J Med 2006; 355:2418–2426. [DOI] [PubMed] [Google Scholar]
- 5.Gorkiewicz G. Nosocomial and antibiotic-associated diarrhoea caused by organisms other than Clostridium difficile. Int J Antimicrob Agents 2009; 33(Suppl 1):S37–S41. [DOI] [PubMed] [Google Scholar]
- 6.Krones E, Högenauer C. Diarrhea in the immunocompromised patient. Gastroenterol Clin North Am 2012; 41:677–701. [DOI] [PubMed] [Google Scholar]
- 7.Lai KK, Lamps LW. Enterocolitis in immunocompromised patients. Semin Diagn Pathol 2014; 31:176–191. [DOI] [PubMed] [Google Scholar]
- 8.Maynard CL, Elson CO, Hatton RD, et al. Reciprocal interactions of the intestinal microbiota and immune system. Nature 2012; 489:231–241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kump PK, Gröchenig HP, Lackner S, et al. Alteration of intestinal dysbiosis by fecal microbiota transplantation does not induce remission in patients with chronic active ulcerative colitis. Inflamm Bowel Dis 2013; 19:2155–2165. [DOI] [PubMed] [Google Scholar]
- 10.Lawley TD, Walker AW. Intestinal colonization resistance. Immunology 2013; 138:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hayakawa M, Asahara T, Henzan N, et al. Dramatic changes of the gut flora immediately after severe and sudden insults. Dig Dis Sci 2011; 56:2361–2365. [DOI] [PubMed] [Google Scholar]
- 12.Shimizu K, Ogura H, Goto M, et al. Altered gut flora and environment in patients with severe SIRS. J Trauma 2006; 60:126–133. [DOI] [PubMed] [Google Scholar]
- 13.Jackson MA, Goodrich JK, Maxan ME, et al. Proton pump inhibitors alter the composition of the gut microbiota. Gut 2016; 65:749–756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Seto CT, Jeraldo P, Orenstein R, et al. Prolonged use of a proton pump inhibitor reduces microbial diversity: Implications for Clostridium difficile susceptibility. Microbiome 2014; 2:42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Freedberg DE, Toussaint NC, Chen SP, et al. Proton pump inhibitors alter specific taxa in the human gastrointestinal microbiome: A crossover trial. Gastroenterology 2015; 149:883–885.e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lee SM, Donaldson GP, Mikulski Z, et al. Bacterial colonization factors control specificity and stability of the gut microbiota. Nature 2013; 501:426–429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zaborin A, Smith D, Garfield K, et al. Membership and behavior of ultra-low-diversity pathogen communities present in the gut of humans during prolonged critical illness. MBio 2014; 5:e01361–e01314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Winter SE, Winter MG, Xavier MN, et al. Host-derived nitrate boosts growth of E. coli in the inflamed gut. Science 2013; 339:708–711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Harrison A, Bakaletz LO, Munson RS., Jr Haemophilus influenzae and oxidative stress. Front Cell Infect Microbiol 2012; 2:40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Morgun A, Dzutsev A, Dong X, et al. Uncovering effects of antibiotics on the host and microbiota using transkingdom gene networks. Gut 2015; 64:1732–1743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chuang CH, Wang YH, Chang HJ, et al. Shanghai fever: A distinct Pseudomonas aeruginosa enteric disease. Gut 2014; 63:736–743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Luciano L, Hass R, Busche R, et al. Withdrawal of butyrate from the colonic mucosa triggers “mass apoptosis” primarily in the G0/G1 phase of the cell cycle. Cell Tissue Res 1996; 286:81–92. [DOI] [PubMed] [Google Scholar]
- 23.Hass R, Busche R, Luciano L, et al. Lack of butyrate is associated with induction of Bax and subsequent apoptosis in the proximal colon of guinea pig. Gastroenterology 1997; 112:875–881. [DOI] [PubMed] [Google Scholar]
- 24.Masia R, Peyton S, Lauwers GY, et al. Gastrointestinal biopsy findings of autoimmune enteropathy: A review of 25 cases. Am J Surg Pathol 2014; 38:1319–1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Soldini D, Gaspert A, Montani M, et al. Apoptotic enteropathy caused by antimetabolites and TNF-α antagonists. J Clin Pathol 2014; 67:582–586. [DOI] [PubMed] [Google Scholar]
- 26.Rieger K, Loddenkemper C, Maul J, et al. Mucosal FOXP3+ regulatory T cells are numerically deficient in acute and chronic GvHD. Blood 2006; 107:1717–1723. [DOI] [PubMed] [Google Scholar]
- 27.Taur Y, Jenq RR, Perales MA, et al. The effects of intestinal tract bacterial diversity on mortality following allogeneic hematopoietic stem cell transplantation. Blood 2014; 124:1174–1182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jenq RR, Ubeda C, Taur Y, et al. Regulation of intestinal inflammation by microbiota following allogeneic bone marrow transplantation. J Exp Med 2012; 209:903–911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.van Nood E, Vrieze A, Nieuwdorp M, et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med 2013; 368:407–415. [DOI] [PubMed] [Google Scholar]
- 30.Debast SB, Bauer MP, Kuijper EJ; European Society of Clinical Microbiology and Infectious Diseases: Update of the treatment guidance document for Clostridium difficile infection. Clin Microbiol Infect 2014; 20(Suppl 2):1–26. [DOI] [PubMed] [Google Scholar]
- 31.Kelly CR, Khoruts A, Staley C, et al. Effect of fecal microbiota transplantation on recurrence in multiply recurrent Clostridium difficile infection: A randomized trial. Ann Intern Med 2016; 165:609–616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Aroniadis OC, Brandt LJ, Greenberg A, et al. Long-term follow-up study of fecal microbiota transplantation for severe and/or complicated Clostridium difficile infection: A multicenter experience. J Clin Gastroenterol 2016; 50:398–402. [DOI] [PubMed] [Google Scholar]
- 33.Fischer M, Sipe BW, Rogers NA, et al. Faecal microbiota transplantation plus selected use of vancomycin for severe-complicated Clostridium difficile infection: Description of a protocol with high success rate. Aliment Pharmacol Ther 2015; 42:470–476. [DOI] [PubMed] [Google Scholar]
- 34.Agrawal M, Aroniadis OC, Brandt LJ, et al. The Long-term efficacy and safety of fecal microbiota transplant for recurrent, severe, and complicated Clostridium difficile infection in 146 elderly individuals. J Clin Gastroenterol 2016; 50:403–407. [DOI] [PubMed] [Google Scholar]
- 35.Lagier JC, Delord M, Million M, et al. Dramatic reduction in Clostridium difficile ribotype 027-associated mortality with early fecal transplantation by the nasogastric route: A preliminary report. Eur J Clin Microbiol Infect Dis 2015; 34:1597–1601. [DOI] [PubMed] [Google Scholar]
- 36.Baxter M, Colville A. Adverse events in faecal microbiota transplant: A review of the literature. J Hosp Infect 2016; 92:117–127. [DOI] [PubMed] [Google Scholar]
- 37.Wang S, Xu M, Wang W, et al. Systematic review: Adverse events of fecal microbiota transplantation. PLoS One 2016; 11:e0161174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kelly CR, Ihunnah C, Fischer M, et al. Fecal microbiota transplant for treatment of Clostridium difficile infection in immunocompromised patients. Am J Gastroenterol 2014; 109:1065–1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Baxter M, Ahmad T, Colville A, et al. Fatal aspiration pneumonia as a complication of fecal microbiota transplant. Clin Infect Dis 2015; 61:136–137. [DOI] [PubMed] [Google Scholar]
- 40.Vermeire S, Joossens M, Verbeke K, et al. Donor species richness determines faecal microbiota transplantation success in inflammatory bowel disease. J Crohns Colitis 2016; 10:387–394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kump P, Högenauer C. Any future for fecal microbiota transplantation as treatment strategy for inflammatory bowel diseases? Dig Dis 2016; 34(Suppl 1):74–81. [DOI] [PubMed] [Google Scholar]
- 42.Khoruts A, Rank KM, Newman KM, et al. Inflammatory bowel disease affects the outcome of fecal microbiota transplantation for recurrent Clostridium difficile infection. Clin Gastroenterol Hepatol 2016; 14:1433–1438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Buffie CG, Bucci V, Stein RR, et al. Precision microbiome reconstitution restores bile acid mediated resistance to Clostridium difficile. Nature 2015; 517:205–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Li Q, Wang C, Tang C, et al. Successful treatment of severe sepsis and diarrhea after vagotomy utilizing fecal microbiota transplantation: A case report. Crit Care 2015; 19:37. [DOI] [PMC free article] [PubMed] [Google Scholar]


