Abstract
High-risk patients with rheumatic heart disease (RHD) who were undergoing valve replacement surgery (VRS) were not identified entirely. This study included 1782 consecutive patients with RHD who were undergoing VRS to explore the relationship between hypoalbuminemia and adverse outcomes and to confirm whether hypoalbuminemia plays a role in risk evaluation. A total of 27.3% of the RHD patients had hypoalbuminemia. In-hospital deaths were significantly higher in the hypoalbuminemic group than in the non-hypoalbuminemic group (6.6% vs 3.1%, P = 0.001). Hypoalbuminemia was an independent predictor of in-hospital death (OR = 1.89, P = 0.014), even after adjusting for the Euro score. The addition of hypoalbuminemia to Euro score enhanced net reclassification improvement (0.346 for in-hospital death, P = 0.004; 0.306 for 1-year death, p = 0.005). A Kaplan-Meier curve analysis revealed that the cumulative rate of 1-year mortality after the operation was higher in patients with a new Euro score ≥6. These findings indicated that hypoalbuminemia was an independent risk factor for in-hospital and 1-year mortality after VRS in patients with RHD, which might have additive prognostic value to Euro score.
Introduction
Rheumatic heart disease (RHD) is the permanent damage to the heart valves caused by an abnormal immune reaction to group A streptococcal infection. RHD is a global threat to human health, especially in developing countries1. Valve replacement surgery (VRS) is an effective therapy for RHD. Epidemiological data2 have indicated that 4.7% (698/14799) of patients died soon after cardiac surgery, among which 0.8% (36/4529) were low-risk patients (European System for Cardiac Operative Risk Evaluation, Euro score ≤2), 3.0% (182/5977) were medium-risk patients (Euro score 3–5) and 11.2% (480/4293) were high-risk patients (Euro score ≥6). The Euro score cannot, however, determine all high-risk patients3. Therefore, it is important to explore an index in which more high-risk patients can be identified.
Albumin is an important protein of human serum, and has multiple physiological functions in the body4. Hypoalbuminemia is common in patients with critical illnesses5. Hypoalbuminemia has been associated with poor prognosis in several conditions, such as acute coronary syndrome, acute aortic dissection, heart failure6–8, and in patients undergoing coronary artery bypass9. However, there are fewer reports that have investigated the relationship between low serum albumin (SA) and adverse outcomes in patients with RHD undergoing VRS. In addition, whether adding hypoalbuminemia to the Euro score can improve the prediction of poor outcomes is unclear. For these reasons, this study was conducted to assess the prognostic value of hypoalbuminemia in patients with RHD undergoing VRS.
Methods
Patient population
This was an observational, single-center study. Between March 2009 and July 2013, patients with RHD undergoing VRS were enrolled from Guangdong General Hospital, Guangzhou, China. All patients underwent coronary angiography to exclude coronary heart disease prior to the operation. Patients without SA level measured on admission was excluded in this analysis. Exclusion criteria also included if patients had any condition relating to hypoalbuminemia: (1) patients with end-stage renal disease (estimated glomerular filtration rate (eGFR) <30 mL/min/1.73 m2) or significant hepatic dysfunction (alanine transaminase >3* the upper limit of normal)10, 11, (2) patients with infections or autoimmune diseases and (3) patients with a history of malignancy. Written informed consent was received from all patients, and the study was passed by the ethics committee of the hospital and in accordance with the relevant guidelines and regulations.
Clinical examination
SA levels and other blood variables were measured under fasting conditions in the morning of day following admission. SA was measured using a Beckman Coulter AU5821 or AU5831 (Beckman Coulter Inc, California). Echocardiography was routinely performed within 24 hours after admission to the hospital. Left ventricular ejection fraction (LVEF) was evaluated using the Simpson’s biplane method. The eGFR was calculated by the Modification of Diet in Renal Disease equation for Chinese patients12. Demographic and clinical characteristics including variables of Euro score were collected by one researcher with the use of an electronic case report form and then confirmed by another researcher. Euro score developed by Nashef SA et al. was calculated in this analysis2.
Definition and end points
Hypoalbuminemia was defined as SA < 35 g/L. Stenosis of greater than 50% in the major coronary arteries was considered as coronary artery disease. The primary endpoint in this analysis was in-hospital mortality. All causes of death in the year after the operation, and in-hospital events (including death, dialysis and stroke) were considered as secondary end points.
Statistical analysis
All statistical analyses were performed using SPSS software version 13.0 (SPSS, Inc., Chicago, Illinois). Patients were classified into two groups according to the SA level on admission: a non-hypoalbuminemic group and a hypoalbuminemic group. Continuous variables were shown as mean ± standard deviation or as medians and interquartile ranges, according to different distribution types. An independent sample T-test or non-parameters test was then performed, accordingly. Categorical variables were presented as absolute counts and percentages, and analyzed using the x2 test. Variables whose p value was less than 0.05 (except for components of the Euro score) in univariate logistic regression analysis were included in the multivariable analysis. In addition, these might affect SA level or prognosis in the clinical practice were also included in multiple analysis to rule out the interference. Receiver operating characteristic (ROC) curve analysis was conducted to determine the best cut-off value of SA and of the Euro score for predicting in-hospital mortality. The areas under the curve (AUCs) were compared using MedCalc statistical software (version 12.7.10, Ostend, Belgium). Continuous net reclassification improvement (NRI) was also used to discover the difference of predictive value. The Kaplan–Meier method was applied to assess the cumulative rate of 1-year mortality after the operation, and survival differences between groups were compared using the log-rank test. A 2-tailed P-value <0.05 was considered statistically significant.
Results
Baseline clinical characteristics
Between March 2009 and July 2013, 1858 patients with rheumatic heart disease undergoing valve replacement surgery and preoperative coronary angiography were screened. A total of two patients killed themselves during hospitalization and 40 patients did not have their SA levels measured upon admission. In addition, patients with eGFR <30 mL/min/1.73 m2 (n = 13) and alanine transaminase >120 U/L (n = 21) were excluded to avoid the confounding influence on albumin levels. Thus, 581 (32.6%) males and 1201 (67.4%) females were selected in this study with a mean age of 58 ± 6 years.
A total of 27.3% of patients also had hypoalbuminemia (SA < 35 g/L). In addition, there were 23.9% of patients with hypoalbuminemia in patients with a Euro score of ≤2, 26.1% in patients with a Euro score of 3–5 and 37.2% in patients with a Euro score of ≥6 (Fig. 1). Included patients were classified into two groups according to SA levels at admission: a non-hypoalbuminemic group (n = 1295) and a hypoalbuminemic group (n = 487). Patients with hypoalbuminemia were significantly older (58.2 ± 5.9 vs 57.4 ± 5.6, p = 0.005) and less likely to be female (62.8% vs 69.1%, p = 0.012). There was a larger proportion of smokers, New York Heart Association (NYHA) Functional Classification >II, pulmonary artery systolic pressure (PASP) > 60 mmHg and history of hypertension in the hypoalbuminemic group. Lower eGFR and LVEF were seen in patients with hypoalbuminemia in whom the Euro score was higher. A total of 72 patients died while in hospital: 40 (3.1%) in the non-hypoalbuminemic group and 32 (6.6%) in the hypoalbuminemic group. There was a statistically significant difference between the two groups (p = 0.001, Table 1).
Figure 1.
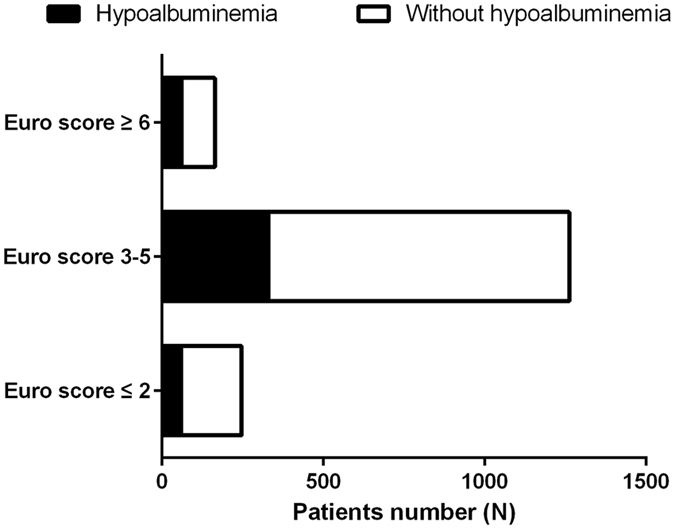
Distribution of patients with hypoalbuminemia with different Euro scores.
Table 1.
Demographic and clinical characteristics in the two different preoperative plasma albumin groups.
| Clinical variables | Non-hypoalbuminemic group (n = 1295) | Hypoalbuminemic group (n = 487) | P |
|---|---|---|---|
| Age (year) | 57.4 ± 5.6 | 58.2 ± 5.9 | 0.005 |
| Females, n (%) | 895 (69.1) | 306 (62.8) | 0.012 |
| Previous stroke, n (%) | 46 (3.6) | 13 (2.7) | 0.353 |
| Hypertension, n (%) | 143 (11.0) | 74 (15.2) | 0.017 |
| Diabetes mellitus, n (%) | 76 (5.9) | 34 (7.0) | 0.384 |
| Previous valve replacement | 26 (2.0) | 9 (1.8) | 0.829 |
| Coronary artery disease, n (%) | 71 (5.5) | 32 (6.6) | 0.380 |
| NYHA > II, n (%) | 527 (40.7) | 261 (53.6) | <0.001 |
| Serum creatinine (µmol/L) | 78.2 ± 20.5 | 85.3 ± 23.4 | <0.001 |
| eGFR (mL/min/1.73 m2) | 90.4 ± 25.0 | 82.6 ± 23.0 | <0.001 |
| ALT (U/L) | 21.0 (17.0, 27.0) | 21.0 (16.0, 30.0) | 0.135 |
| SA (g/L) | 38.9 ± 2.7 | 32.2 ± 2.6 | <0.001 |
| LVEF (%) | 62.3 ± 8.6 | 59.8 ± 10.0 | <0.001 |
| Valve intervention | |||
| Aortic | 526 (40.6) | 199 (40.9) | 0.925 |
| Mitral | 1217 (94.0) | 463 (95.1) | 0.375 |
| Tricuspid | 990 (76.4) | 401 (82.3) | 0.007 |
| CABG | 63 (4.9) | 21 (4.3) | 0.624 |
| Euro score | 3.6 ± 1.3 | 3.9 ± 1.4 | <0.001 |
| In-hospital Death | 40 (3.1) | 32 (6.6) | 0.001 |
| In-hospital events | 55 (4.2) | 41 (8.4) | 0.001 |
NYHA, New York Heart Association; eGFR, estimated glomerular filtration rate; ALT, alanine transaminase; SA, serum albumin; LVEF, left ventricular ejection fraction; CABG, coronary artery bypass grafting.
Correlation analysis revealed that there was a negative relation between SA and C-reactive protein (CRP) (r = −0.212, p < 0.001).
Role of hypoalbuminemia for in-hospital mortality
The univariate analyses for in-hospital mortality are shown in Table 2. Results revealed that age, previous valve replacement, NYHA Functional Classification > II, eGFR < 60 mL/min/1.73 m2, hypoalbuminemia, coronary artery bypass grafting (CABG) and Euro score were related to an increased in-hospital mortality. These variables (except for components of the Euro score) were studied by multiple logistic regression analysis, and it was revealed that hypoalbuminemia was a reliable predictor of in-hospital death (OR = 1.89, 95% CI, 1.14–3.13, P = 0.014), even after adjusting for the Euro score.
Table 2.
Univariate analysis and multiple logistic regression analysis for in-hospital deaths.
| Clinical variables | Univariate analysis | Multiple logistic regression | |||
|---|---|---|---|---|---|
| OR | P | OR | 95% CI | P | |
| Age | 1.10 | <0.001 | |||
| Female | 0.75 | 0.247 | |||
| Hypertension | 1.47 | 0.237 | |||
| Diabetes mellitus | 1.97 | 0.081 | 1.79 | 0.81, 3.93 | 0.148 |
| Previous valve replacement | 4.18 | 0.004 | |||
| Coronary artery disease | 1.51 | 0.347 | |||
| NYHA > II | 2.17 | 0.002 | |||
| eGFR < 60 mL/min/1.73 m2 | 2.07 | 0.023 | |||
| ALT | 1.01 | 0.267 | 1.01 | 1.00, 1.03 | 0.175 |
| Hypoalbuminemia | 2.21 | 0.001 | 1.89 | 1.14, 3.13 | 0.014 |
| LVEF | 0.98 | 0.104 | |||
| Aortic valve intervention | 1.40 | 0.164 | |||
| Mitral valve intervention | 0.55 | 0.142 | |||
| Tricuspid valve intervention | 1.78 | 0.096 | |||
| CABG | 2.69 | 0.012 | 2.50 | 1.11, 5.61 | 0.026 |
| Euro score | 1.62 | <0.001 | 1.61 | 1.37, 1.89 | <0.001 |
The variables in the Euro score were not included in the multiple analysis. NYHA, New York Heart Association; eGFR, estimated glomerular filtration rate; ALT, alanine transaminase; SA, serum albumin; LVEF, left ventricular ejection fraction; CABG, coronary artery bypass grafting.
According to the odds ratio from multivariate analyses, we assigned one point for hypoalbuminemia. The associations between in-hospital mortality and the old and new risk stratification assessment score are displayed in Table 3. The new risk score was able to identify more low-risk patients than the old one. In addition, the ROC curve showed that SA < 36.6 had a sensitivity of 57.3% and a specificity of 63.9% for predicting in-hospital deaths (AUC = 0.623, 95% CI, 0.558–0.688, P < 0.001). For the Euro score, the AUC was 0.690 (95% CI 0.627–0.753, P < 0.001) and the sensitivity and specificity were 58.0% and 73.7%, respectively. There was no significant difference between their AUCs (z = 1.468, p = 0.142). The inclusion of hypoalbuminemia in the Euro score produced a slight improvement (z = 1.569, p = 0.117, Fig. 2). We also calculated the continuous NRI and the result demonstrated that the addition of hypoalbuminemia to Euro score enhanced net reclassification improvement for in-hospital mortality (NRI = 0.346, 95% CI: 0.108–0.584, P = 0.004).
Table 3.
Risk of in-hospital death stratified by different Euro scores.
| Euro score | Euro score + hypoalbuminemia | |||
|---|---|---|---|---|
| N (%) | In-hospital mortality, % | N (%) | In-hospital mortality, % | |
| Low, ≤2 | 247 (13.9) | 2 (0.8) | 188 (10.5) | 0 |
| Middle, 3–5 | 1262 (70.8) | 50 (4.0) | 1228 (68.9) | 40 (3.3) |
| High, ≥6 | 164 (9.2) | 17 (10.4) | 257 (14.4) | 29 (11.3) |
Figure 2.
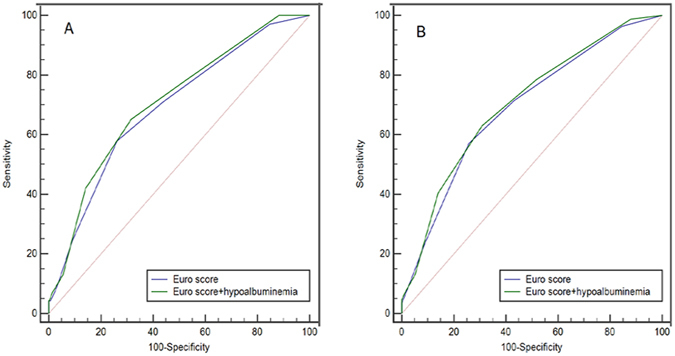
Comparison of areas under the curve for in-hospital (A) and 1-year mortality (B).
Hypoalbuminemia and 1-year mortality
All enrolled patients were followed up for 1 year after the operation. In this follow-up period, 193 (10.8%) patients were lost to follow-up and 87 (4.9%) died. The ROC curves of Euro score and new one showed no differences in their AUCs (z = 1.326, P = 0.185, Fig. 2). However, compared with the Euro score alone, combined use of hypoalbuminemia and Euro score was associated with significant improvements in the ability to predict 1-year death (NRI = 0.306, 95% CI: 0.091–0.521, P = 0.005). Kaplan-Meier curve analysis was shown in Fig. 3, revealing that the cumulative rate of 1-year mortality after the operation was higher in patients with a new Euro score of ≥6 (Log-rank = 47.72, p < 0.001). Multivariate Cox proportional hazard modelling revealed that new Euro score remained a significant predictor for 1-year death (HR = 1.56, 95% CI, 1.38–1.77, P < 0.001), even after adjusting potential risk factors (Table 4).
Figure 3.
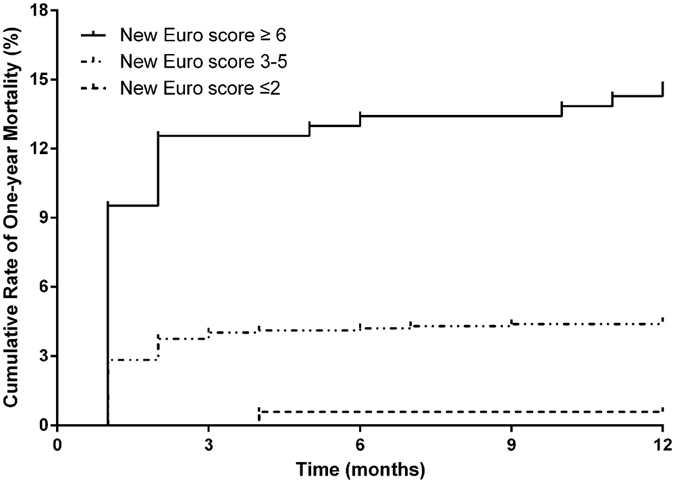
Cumulative rate of 1-year mortality after the operation, in patients with different risk scores.
Table 4.
Multivariate Cox proportional hazard modelling results of 1-year mortality.
| Clinical variables | Hazard ratio (HR) | 95% Confidence interval (CI) | p-value |
|---|---|---|---|
| Diabetes mellitus | 1.55 | 0.78, 3.10 | 0.215 |
| ALT | 1.01 | 0.99, 1.02 | 0.427 |
| CABG | 2.41 | 1.24, 4.68 | 0.009 |
| New Euro score | 1.56 | 1.38, 1.77 | <0.001 |
The variables in the New Euro score were not included in the multiple analysis. ALT, alanine transaminase; CABG, coronary artery bypass grafting.
Discussion
This is the first study to assess the prognostic value in patients with RHD undergoing VRS. The main finding was that hypoalbuminemia was an independent risk factor for in-hospital and 1-year mortality after VRS in patients with RHD. In addition, including hypoalbuminemia in the Euro score produced a slight improvement, which might identify more patients at high risk of poor outcomes than the Euro score alone.
A total of 27.3% patients with RHD suffered from hypoalbuminemia on admission to this study, for three possible reasons. The first reason was the relationship between age and SA; Weaving et al. discovered that, at around age 20, SA reached a peak value and then decreased with age13. The average age in this study was 58 ± 6 years. Secondly, all the RHD patients in this study required an operation, which reflected the fact that patients experienced symptoms such as fatigue, loss of appetite and shortness of breath, which could influence the protein intake. Thirdly, patients with RHD often suffer from cardiac dysfunction14. Previous investigations have indicated that hypoalbuminemia was common in patients with stable chronic15 or acute heart failure16. Hepatocellular function, nutritional status, inflammation and congestion may account for this phenomenon17.
This study demonstrated that preoperative hypoalbuminemia was related to poor outcomes in patients with RHD undergoing VRS. This could be explained by several theories. SA is a good index for indicating nutritional status18, and malnutrition has been reported to be associated with increased mortality in cardiac surgery patients19. Low SA, as a sign of malnutrition, was shown to be an independent predictor of increased mortality after cardiac surgery20. SA has also been associated with inflammation, in which the catabolism will increase, including reduced protein synthesis rates and increased protein degradation rates21. Therefore, hypoalbuminemia could represent inflammatory conditions. In this study, we also found that SA was negatively correlated with CRP. In addition, SA participated in modulating prostaglandin E2 (PGE2)-mediated immunosuppression22. Hypoalbuminemia has been shown to be a risk factor for surgical site infection after a gastrointestinal operation23. Another theory that can be considered is the relationship between low SA and postoperative acute kidney injury (AKI). Lee et al. reported that hypoalbuminemia was an independent predictor for AKI and that AKI was related to poor outcomes after off-pump coronary artery bypass surgery24. In the meta-analysis conducted by Wiedermann, a similar result was shown in that low SA was associated with AKI and mortality following development of AKI25. All these results support our data.
The mortality rate in this analysis was 4.0%, which was in accordance with a previous study26. Euro score is a risk assessment method that can calculate operative mortality in patients after cardiac surgery. However, the Euro score could not identify all patients at risk of death27. In this study, we found that adding hypoalbuminemia to the Euro score might identify more patients at high risk of poor outcomes than the Euro score alone. In clinical practice, patients presenting with higher Euro score were at high risk of poor outcomes and may need more attention and care in cases with hypoalbuminemia and high Euro score.
Limitations
Several limitations should be considered in this analysis. Firstly, this study was a retrospective analysis based on prospectively collected data. Therefore, some admission SA levels were missing or confounding, which may affect the results. Secondly, SA was not dynamically detected. The relationship between prognosis and SA measured at different time points is unknown. Future studies should be performed to confirm whether receiving albumin treatment could improve outcomes.
Conclusions
This study demonstrated that hypoalbuminemia was an independent risk factor for in-hospital and 1-year mortality after VRS in patients with RHD. In addition, inclusion of hypoalbuminemia in the Euro score produced a slight improvement in prognosis, which might identify more patients at high risk of poor outcomes than the Euro score alone.
Author Contributions
Ning Tan and Dan-qing Yu conceived and designed the experiments; Du Feng and Peng-cheng He analyzed the data; Xue-biao Wei and Lei Jiang wrote the paper. Ji-yan Chen and Yuan-hui Liu critically revised the manuscript. Everyone gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Xue-biao Wei and Lei Jiang contributed equally to this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Dan-qing Yu, Email: gdydq100@126.com.
Ning Tan, Email: ningtan888@yeah.net.
References
- 1.Remenyi B, ElGuindy A, Smith SJ, Yacoub M, Holmes DJ. Valvular Aspects of Rheumatic Heart Disease. Lancet. 2016;387:1335–1346. doi: 10.1016/S0140-6736(16)00547-X. [DOI] [PubMed] [Google Scholar]
- 2.Nashef SA, et al. European System for Cardiac Operative Risk Evaluation (EuroSCORE) Eur J Cardiothorac Surg. 1999;16:9–13. doi: 10.1016/S1010-7940(99)00134-7. [DOI] [PubMed] [Google Scholar]
- 3.Geissler HJ, et al. Risk Stratification in Heart Surgery: Comparison of Six Score Systems. Eur J Cardiothorac Surg. 2000;17:400–406. doi: 10.1016/S1010-7940(00)00385-7. [DOI] [PubMed] [Google Scholar]
- 4.Rothschild MA, Oratz M, Schreiber SS. Serum Albumin. Hepatology. 1988;8:385–401. doi: 10.1002/hep.1840080234. [DOI] [PubMed] [Google Scholar]
- 5.Vincent JL, Dubois MJ, Navickis RJ, Wilkes MM. Hypoalbuminemia in Acute Illness: Is there a Rationale for Intervention? A Meta-Analysis of Cohort Studies and Controlled Trials. Ann Surg. 2003;237:319–334. doi: 10.1097/01.SLA.0000055547.93484.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yang LJ, et al. Serum Albumin Levels Might be an Adverse Predictor of Long Term Mortality in Patients with Acute Myocardial Infarction. INT J CARDIOL. 2016;223:647–648. doi: 10.1016/j.ijcard.2016.08.251. [DOI] [PubMed] [Google Scholar]
- 7.Liu M, et al. Albumin Levels Predict Survival in Patients with Heart Failure and Preserved Ejection Fraction. Eur J Heart Fail. 2012;14:39–44. doi: 10.1093/eurjhf/hfr154. [DOI] [PubMed] [Google Scholar]
- 8.Hartopo AB, Gharini PP, Setianto BY. Low Serum Albumin Levels and In-Hospital Adverse Outcomes in Acute Coronary Syndrome. Int Heart J. 2010;51:221–226. doi: 10.1536/ihj.51.221. [DOI] [PubMed] [Google Scholar]
- 9.de la Cruz KI, et al. Hypoalbuminemia and Long-Term Survival After Coronary Artery Bypass: A Propensity Score Analysis. Ann Throac Surg. 2011;91:671–675. doi: 10.1016/j.athoracsur.2010.09.004. [DOI] [PubMed] [Google Scholar]
- 10.Gonzalez-Pacheco H, et al. Prognostic Implications of Serum Albumin Levels in Patients with Acute Coronary Syndromes. AM J Cardiol. 2017;119:951–958. doi: 10.1016/j.amjcard.2016.11.054. [DOI] [PubMed] [Google Scholar]
- 11.Abboud G, Kaplowitz N. Drug-Induced Liver Injury. Drug Saf. 2007;30:277–294. doi: 10.2165/00002018-200730040-00001. [DOI] [PubMed] [Google Scholar]
- 12.Ma YC, et al. Modified Glomerular Filtration Rate Estimating Equation for Chinese Patients with Chronic Kidney Disease. J Am Soc Nephrol. 2006;17:2937–2944. doi: 10.1681/ASN.2006040368. [DOI] [PubMed] [Google Scholar]
- 13.Weaving G, Batstone GF, Jones RG. Age and Sex Variation in Serum Albumin Concentration: An Observational Study. Ann Clin Biochem. 2016;53:106–111. doi: 10.1177/0004563215593561. [DOI] [PubMed] [Google Scholar]
- 14.Marijon E, Mirabel M, Celermajer DS, Jouven X. Rheumatic Heart Disease. Lancet. 2012;379:953–964. doi: 10.1016/S0140-6736(11)61171-9. [DOI] [PubMed] [Google Scholar]
- 15.Horwich TB, Kalantar-Zadeh K, MacLellan RW, Fonarow GC. Albumin Levels Predict Survival in Patients with Systolic Heart Failure. Am Heart J. 2008;155:883–889. doi: 10.1016/j.ahj.2007.11.043. [DOI] [PubMed] [Google Scholar]
- 16.Uthamalingam S, et al. Serum Albumin and Mortality in Acutely Decompensated Heart Failure. Am Heart J. 2010;160:1149–1155. doi: 10.1016/j.ahj.2010.09.004. [DOI] [PubMed] [Google Scholar]
- 17.Bonilla-Palomas JL, et al. Hypoalbuminemia in Acute Heart Failure Patients: Causes and its Impact On Hospital and Long-Term Mortality. J Card Fail. 2014;20:350–358. doi: 10.1016/j.cardfail.2014.01.016. [DOI] [PubMed] [Google Scholar]
- 18.McClave SA, Snider HL, Spain DA. Preoperative Issues in Clinical Nutrition. Chest. 1999;115:64S–70S. doi: 10.1378/chest.115.suppl_2.64S. [DOI] [PubMed] [Google Scholar]
- 19.Evans AS, Hosseinian L, Mohabir T, Kurtis S, Mechanick JI. Nutrition and the Cardiac Surgery Intensive Care Unit Patient–An Update. J Cardiothorac Vasc Anesth. 2015;29:1044–1050. doi: 10.1053/j.jvca.2015.03.021. [DOI] [PubMed] [Google Scholar]
- 20.Engelman DT, et al. Impact of Body Mass Index and Albumin On Morbidity and Mortality After Cardiac Surgery. J Thorac Cardiovasc Surg. 1999;118:866–873. doi: 10.1016/S0022-5223(99)70056-5. [DOI] [PubMed] [Google Scholar]
- 21.Don BR, Kaysen G. Serum Albumin: Relationship to Inflammation and Nutrition. Semin Dial. 2004;17:432–437. doi: 10.1111/j.0894-0959.2004.17603.x. [DOI] [PubMed] [Google Scholar]
- 22.Choe WH, Baik SK. Prostaglandin E2 -Mediated Immunosuppression and the Role of Albumin as its Modulator. Hepatology. 2015;61:1080–1082. doi: 10.1002/hep.27644. [DOI] [PubMed] [Google Scholar]
- 23.Hennessey DB, et al. Preoperative Hypoalbuminemia is an Independent Risk Factor for the Development of Surgical Site Infection Following Gastrointestinal Surgery: A Multi-Institutional Study. Ann Surg. 2010;252:325–329. doi: 10.1097/SLA.0b013e3181e9819a. [DOI] [PubMed] [Google Scholar]
- 24.Lee EH, et al. Preoperative Hypoalbuminemia is a Major Risk Factor for Acute Kidney Injury Following Off-Pump Coronary Artery Bypass Surgery. Intensive Care Med. 2012;38:1478–1486. doi: 10.1007/s00134-012-2599-8. [DOI] [PubMed] [Google Scholar]
- 25.Wiedermann CJ, Wiedermann W, Joannidis M. Hypoalbuminemia and Acute Kidney Injury: A Meta-Analysis of Observational Clinical Studies. Intensive Care Med. 2010;36:1657–1665. doi: 10.1007/s00134-010-1928-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Roques F, et al. Risk Factors and Outcome in European Cardiac Surgery: Analysis of the EuroSCORE Multinational Database of 19030 Patients. Eur J Cardiothorac Surg. 1999;15:816–822. doi: 10.1016/S1010-7940(99)00106-2. [DOI] [PubMed] [Google Scholar]
- 27.Pitkanen O, Niskanen M, Rehnberg S, Hippelainen M, Hynynen M. Intra-Institutional Prediction of Outcome After Cardiac Surgery: Comparison Between a Locally Derived Model and the EuroSCORE. Eur J Cardiothorac Surg. 2000;18:703–710. doi: 10.1016/S1010-7940(00)00579-0. [DOI] [PubMed] [Google Scholar]


