Abstract
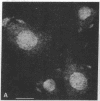
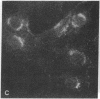
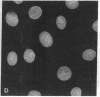
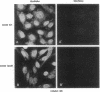
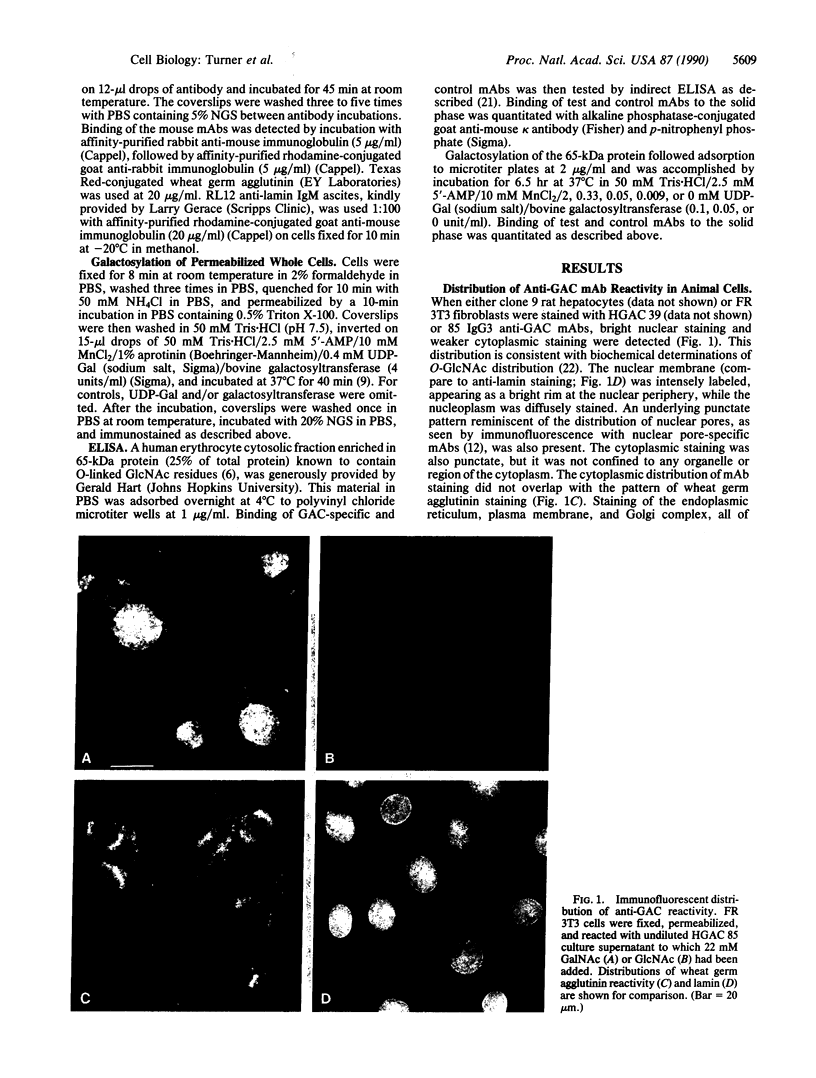
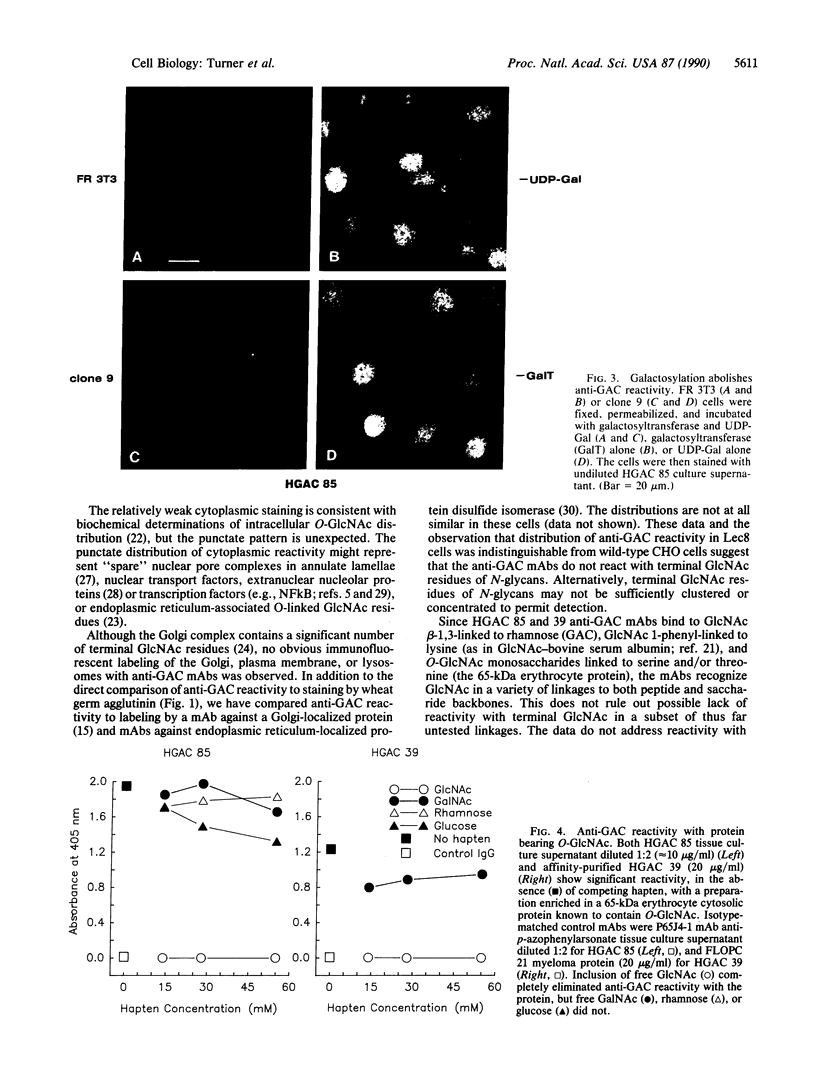
Recent studies have demonstrated the existence of single O-linked N-acetylglucosamine (O-GlcNAc) residues on cytoplasmic and nuclear glycoproteins. Labeled lectin and enzymatic techniques have been used to identify O-GlcNAc-bearing proteins, but no antibodies generally reactive with such O-linked GlcNAc moieties have been described. We have previously characterized monoclonal antibodies (mAbs) specific for the GlcNAc residues of streptococcal group A carbohydrate, which is composed of a polyrhamnose backbone with GlcNAc side chains. We now report that these mAbs recognize O-GlcNAc-bearing proteins. By immunofluorescence, the mAbs reacted strongly with the nuclear periphery and nucleoplasm of mammalian cells and stained the cytoplasm less intensely. The distribution was not consistent with labeling of the endoplasmic reticulum, Golgi complex, or plasma membrane. Furthermore, the staining pattern of a mutant cell line, which retains terminal GlcNAc residues on many N-linked glycans, was indistinguishable from that of wild-type cells. Nuclear and cytoplasmic staining were inhibited by free GlcNAc and were completely abolished by galactosylation of terminal GlcNAc residues. Indirect ELISA demonstrated GlcNAc- and galactosylation-inhibitable binding of the mAbs to a 65-kDa human erythrocyte cytosolic protein known to contain O-GlcNAc. Thus, these mAbs react with O-GlcNAc without apparent influence of peptide determinants, do not show detectable binding to N- or O-glycans, and, therefore, represent a valuable tool for the study of O-GlcNAc moieties. In addition, these mAbs provide the first cytologic analysis of the distribution of O-GlcNAc residues throughout the nucleus and the cytoplasm of mammalian cells.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abeijon C., Hirschberg C. B. Intrinsic membrane glycoproteins with cytosol-oriented sugars in the endoplasmic reticulum. Proc Natl Acad Sci U S A. 1988 Feb;85(4):1010–1014. doi: 10.1073/pnas.85.4.1010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergeron J. J., Rachubinski R. A., Sikstrom R. A., Posner B. I., Paiement J. Galactose transfer to endogenous acceptors within Golgi fractions of rat liver. J Cell Biol. 1982 Jan;92(1):139–146. doi: 10.1083/jcb.92.1.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borer R. A., Lehner C. F., Eppenberger H. M., Nigg E. A. Major nucleolar proteins shuttle between nucleus and cytoplasm. Cell. 1989 Feb 10;56(3):379–390. doi: 10.1016/0092-8674(89)90241-9. [DOI] [PubMed] [Google Scholar]
- Brew K., Vanaman T. C., Hill R. L. The role of alpha-lactalbumin and the A protein in lactose synthetase: a unique mechanism for the control of a biological reaction. Proc Natl Acad Sci U S A. 1968 Feb;59(2):491–497. doi: 10.1073/pnas.59.2.491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briles E. B., Li E., Kornfeld S. Isolation of wheat germ agglutinin-resistant clones of Chinese hamster ovary cells deficient in membrane sialic acid and galactose. J Biol Chem. 1977 Feb 10;252(3):1107–1116. [PubMed] [Google Scholar]
- Caulfield M. J., Cerny J. Specific antigen/antibody complexes induce the in vivo production of a parallel set of nonantigen-binding idiotype-positive antibodies. Eur J Immunol. 1988 Mar;18(3):439–443. doi: 10.1002/eji.1830180318. [DOI] [PubMed] [Google Scholar]
- Chen T. Y., Merisko E. M. Annulate lamellae: comparison of antigenic epitopes of annulate lamellae membranes with the nuclear envelope. J Cell Biol. 1988 Oct;107(4):1299–1306. doi: 10.1083/jcb.107.4.1299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chicheportiche Y., Vassalli P., Tartakoff A. M. Characterization of cytoplasmically oriented Golgi proteins with a monoclonal antibody. J Cell Biol. 1984 Dec;99(6):2200–2210. doi: 10.1083/jcb.99.6.2200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coligan J. E., Kindt T. J., Krause R. M. Structure of the streptococcal groups A, A-variant and C carbohydrates. Immunochemistry. 1978 Nov;15(10-11):755–760. doi: 10.1016/0161-5890(78)90105-0. [DOI] [PubMed] [Google Scholar]
- Deutscher S. L., Hirschberg C. B. Mechanism of galactosylation in the Golgi apparatus. A Chinese hamster ovary cell mutant deficient in translocation of UDP-galactose across Golgi vesicle membranes. J Biol Chem. 1986 Jan 5;261(1):96–100. [PubMed] [Google Scholar]
- Goldstein I., Rebeyrotte P., Parlebas J., Halpern B. Isolation from heart valves of glycopeptides which share immunological properties with Streptococcus haemolyticus group A polysaccharides. Nature. 1968 Aug 24;219(5156):866–868. doi: 10.1038/219866a0. [DOI] [PubMed] [Google Scholar]
- Greenspan N. S., Dacek D. A., Cooper L. J. Fc region-dependence of IgG3 anti-streptococcal group A carbohydrate antibody functional affinity. I. The effect of temperature. J Immunol. 1988 Dec 15;141(12):4276–4282. [PubMed] [Google Scholar]
- Greenspan N. S., Davie J. M. Analysis of idiotope variability as a function of distance from the binding site for anti-streptococcal group A carbohydrate antibodies. J Immunol. 1985 Sep;135(3):1914–1921. [PubMed] [Google Scholar]
- Greenspan N. S., Davie J. M. Serologic and topographic characterization of idiotopes on murine monoclonal anti-streptococcal group A carbohydrate antibodies. J Immunol. 1985 Feb;134(2):1065–1072. [PubMed] [Google Scholar]
- Greenspan N. S., Monafo W. J., Davie J. M. Interaction of IgG3 anti-streptococcal group A carbohydrate (GAC) antibody with streptococcal group A vaccine: enhancing and inhibiting effects of anti-GAC, anti-isotypic, and anti-idiotypic antibodies. J Immunol. 1987 Jan 1;138(1):285–292. [PubMed] [Google Scholar]
- Hart G. W., Haltiwanger R. S., Holt G. D., Kelly W. G. Glycosylation in the nucleus and cytoplasm. Annu Rev Biochem. 1989;58:841–874. doi: 10.1146/annurev.bi.58.070189.004205. [DOI] [PubMed] [Google Scholar]
- Holt G. D., Haltiwanger R. S., Torres C. R., Hart G. W. Erythrocytes contain cytoplasmic glycoproteins. O-linked GlcNAc on Band 4.1. J Biol Chem. 1987 Nov 5;262(31):14847–14850. [PubMed] [Google Scholar]
- Holt G. D., Hart G. W. The subcellular distribution of terminal N-acetylglucosamine moieties. Localization of a novel protein-saccharide linkage, O-linked GlcNAc. J Biol Chem. 1986 Jun 15;261(17):8049–8057. [PubMed] [Google Scholar]
- Holt G. D., Snow C. M., Senior A., Haltiwanger R. S., Gerace L., Hart G. W. Nuclear pore complex glycoproteins contain cytoplasmically disposed O-linked N-acetylglucosamine. J Cell Biol. 1987 May;104(5):1157–1164. doi: 10.1083/jcb.104.5.1157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishibashi M., Maizel J. V., Jr The polypeptides of adenovirus. VI. Early and late glycopolypeptides. Virology. 1974 Apr;58(2):345–361. doi: 10.1016/0042-6822(74)90070-1. [DOI] [PubMed] [Google Scholar]
- Jackson S. P., Tjian R. O-glycosylation of eukaryotic transcription factors: implications for mechanisms of transcriptional regulation. Cell. 1988 Oct 7;55(1):125–133. doi: 10.1016/0092-8674(88)90015-3. [DOI] [PubMed] [Google Scholar]
- Kaetzel C. S., Rao C. K., Lamm M. E. Protein disulphide-isomerase from human placenta and rat liver. Purification and immunological characterization with monoclonal antibodies. Biochem J. 1987 Jan 1;241(1):39–47. doi: 10.1042/bj2410039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly W. G., Hart G. W. Glycosylation of chromosomal proteins: localization of O-linked N-acetylglucosamine in Drosophila chromatin. Cell. 1989 Apr 21;57(2):243–251. doi: 10.1016/0092-8674(89)90962-8. [DOI] [PubMed] [Google Scholar]
- Kingston D., Glynn L. E. Anti-streptococcal antibodies reacting with brain tissue. I. Immunofluourescent studies. Br J Exp Pathol. 1976 Feb;57(1):114–128. [PMC free article] [PubMed] [Google Scholar]
- Krisher K., Cunningham M. W. Myosin: a link between streptococci and heart. Science. 1985 Jan 25;227(4685):413–415. doi: 10.1126/science.2578225. [DOI] [PubMed] [Google Scholar]
- Monsigny M., Roche A. C., Sene C., Maget-Dana R., Delmotte F. Sugar-lectin interactions: how does wheat-germ agglutinin bind sialoglycoconjugates? Eur J Biochem. 1980 Feb;104(1):147–153. doi: 10.1111/j.1432-1033.1980.tb04410.x. [DOI] [PubMed] [Google Scholar]
- Nahm M. H., Clevinger B. L., Davie J. M. Monoclonal antibodies to streptococcal group A carbohydrate. I. A dominant idiotypic determinant is located on Vk. J Immunol. 1982 Oct;129(4):1513–1518. [PubMed] [Google Scholar]
- Schindler M., Hogan M., Miller R., DeGaetano D. A nuclear specific glycoprotein representative of a unique pattern of glycosylation. J Biol Chem. 1987 Jan 25;262(3):1254–1260. [PubMed] [Google Scholar]
- Shirakawa F., Mizel S. B. In vitro activation and nuclear translocation of NF-kappa B catalyzed by cyclic AMP-dependent protein kinase and protein kinase C. Mol Cell Biol. 1989 Jun;9(6):2424–2430. doi: 10.1128/mcb.9.6.2424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snow C. M., Senior A., Gerace L. Monoclonal antibodies identify a group of nuclear pore complex glycoproteins. J Cell Biol. 1987 May;104(5):1143–1156. doi: 10.1083/jcb.104.5.1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres C. R., Hart G. W. Topography and polypeptide distribution of terminal N-acetylglucosamine residues on the surfaces of intact lymphocytes. Evidence for O-linked GlcNAc. J Biol Chem. 1984 Mar 10;259(5):3308–3317. [PubMed] [Google Scholar]
- Wysocki L., Manser T., Gefter M. L. Somatic evolution of variable region structures during an immune response. Proc Natl Acad Sci U S A. 1986 Mar;83(6):1847–1851. doi: 10.1073/pnas.83.6.1847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoneda Y., Imamoto-Sonobe N., Yamaizumi M., Uchida T. Reversible inhibition of protein import into the nucleus by wheat germ agglutinin injected into cultured cells. Exp Cell Res. 1987 Dec;173(2):586–595. doi: 10.1016/0014-4827(87)90297-7. [DOI] [PubMed] [Google Scholar]