Abstract
Mucus forms a major ecological niche for microbiota in various locations throughout the human body such as the gastrointestinal tract, respiratory tract and oral cavity. The primary structural components of mucus are mucin glycoproteins, which crosslink to form a complex polymer network that surrounds microbes. Although the mucin matrix could create constraints that impact inhabiting microbes, little is understood about how this key environmental factor affects interspecies interactions. In this study, we develop an experimental model using gel-forming human salivary mucins to understand the influence of mucin on the viability of two competing species of oral bacteria. We use this dual-species model to show that mucins promote the coexistence of the two competing bacteria and that mucins shift cells from the mixed-species biofilm into the planktonic form. Taken together, these findings indicate that the mucus environment could influence bacterial viability by promoting a less competitive mode of growth.
Mucus lines wet epithelia throughout the human body and is a major ecological niche for microbiota in the respiratory tract, gastrointestinal tract and oral cavity among other locations (Tabak, 1995; Worlitzsch et al., 2002; Derrien et al., 2010). The mucus layer is a three-dimensional hydrogel primarily composed of densely glycosylated polymers called mucins (Tabak et al., 1982; Bansil and Turner, 2006). In the gel, mucin chains crosslink to form a network that surrounds microbes and, consequently, could create geometric and diffusive constraints for biotic and abiotic environmental factors. Little is known, however, about how these constraints influence microbial interactions, such as cell-cell communication and competition, among the vast number of organisms that live in mucus. In this study, we build upon our previous work showing that MUC5B mucins affect intraspecies interactions by promoting dispersal of bacteria and fungi (Caldara et al., 2012; Kavanaugh et al., 2014; Frenkel and Ribbeck, 2015). Here we develop an experimental model to probe the influence of gel-forming human salivary mucins on dual-species bacterial competition to understand how this matrix affects the viability of competing bacteria.
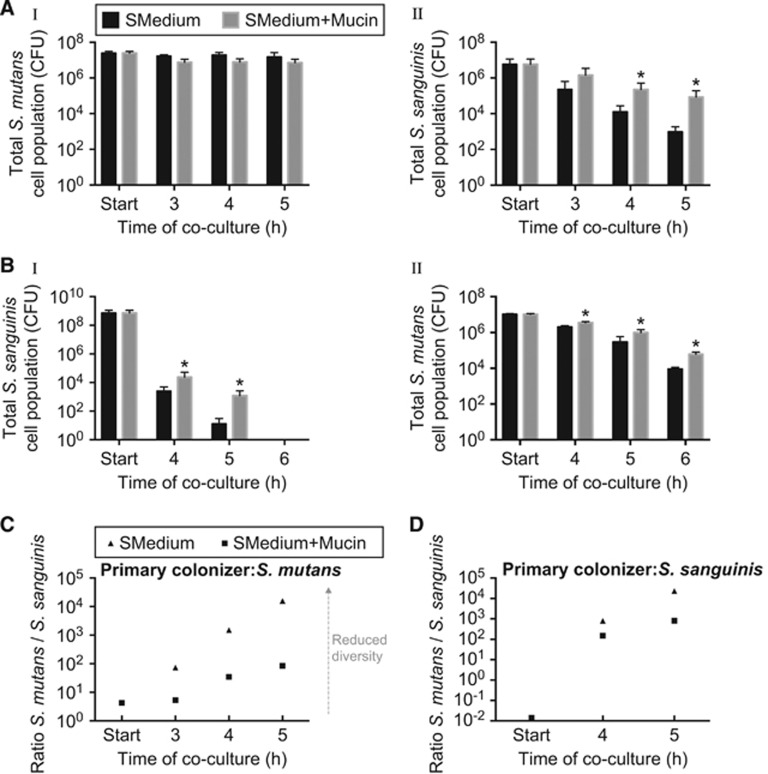
The dual-species model is composed of Streptococcus sanguinis JFP36 and S. mutans UA159, which compete in the oral cavity through the production of hydrogen peroxide and antimicrobial peptides called mutacins, respectively (Kreth et al., 2005, 2008; Ge et al., 2008; Senty Turner et al., 2009). In this model, the two species were inoculated sequentially to more closely mimic the natural environment of the oral cavity where surfaces are generally coated by microbes before other species attempt to colonize. When S. mutans was the primary colonizer, and MUC5B mucins were not present in the growth medium, viability of the secondary colonizer (S. sanguinis) rapidly declined, suggesting that S. mutans outcompetes S. sanguinis (Figure 1A (I, II)). In contrast, when MUC5B was present in the growth medium, the total number of viable S. sanguinis cells increased by 18- and 88-fold after 4 and 5 h of co-culture, respectively, compared with the control without mucin (Figure 1A (II)). The same protective effect by MUC5B was observed when S. sanguinis was the primary colonizer. In this case, the addition of MUC5B to medium enhanced survival of both S. sanguinis and S. mutans (Figure 1B (I, II)). After 4 and 5 h of co-culture in the presence of MUC5B, the number of viable S. sanguinis cells increased by 9- and 94-fold, respectively, relative to the control (Figure 1B (I)). S. mutans CFU increased by 2-, 3-, and 7-fold at 4, 5 and 6 h, respectively (Figure 1B (II)). Of note is that, although MUC5B significantly enhanced S. sanguinis viability, there was an overall reduction in S. sanguinis CFU due to self-killing, which could be caused by increasing hydrogen peroxide concentrations. The same reduction in viability was observed when S. sanguinis was grown in monoculture, indicating that the killing was not due to S. mutans (Supplementary Figure 1). Methylcellulose, a gel-forming polymer that is commonly used to mimic the viscosity of mucus, did not have the same protective effect on S. mutans or S. sanguinis in both of these experimental models; the enhanced survival is likely not due to the addition of a polymer, which could increase viscosity or cause osmotic stress (Supplementary Figure 2) (Ivic et al., 2002; Smith et al., 2009). Together, these results show that MUC5B significantly enhances bacterial diversity by increasing survival of at least one bacterial population compared with the control without MUC5B (Figures 1C and D).
Figure 1.
MUC5B promotes S. mutans and S. sanguinis coexistence, which leads to increased bacterial diversity. S. mutans and S. sanguinis viability in a dual-species experimental model containing control medium (half-strength BHI with 1% sucrose; SMedium) and control medium containing 0.4% MUC5B mucin. Viability was studied when S. mutans was the primary colonizer (A (I)) and S. sanguinis was the secondary colonizer (A (II)) and the reverse scenario where S. sanguinis was the primary colonizer (B (I)) and S. mutans was the secondary colonizer (B (II)). (C, D) the ratios of viable S. mutans and S. sanguinis when S. mutans was the primary colonizer (c) and when S. sanguinis was the primary colonizer (D) as an indication of species diversity. *, statistically significant increase relative to the control with half-strength BHI containing 1% sucrose determined by Student’s t-test (P<0.05). Experiments were performed in triplicate and error bars represent s.d. of CFU between replicates.
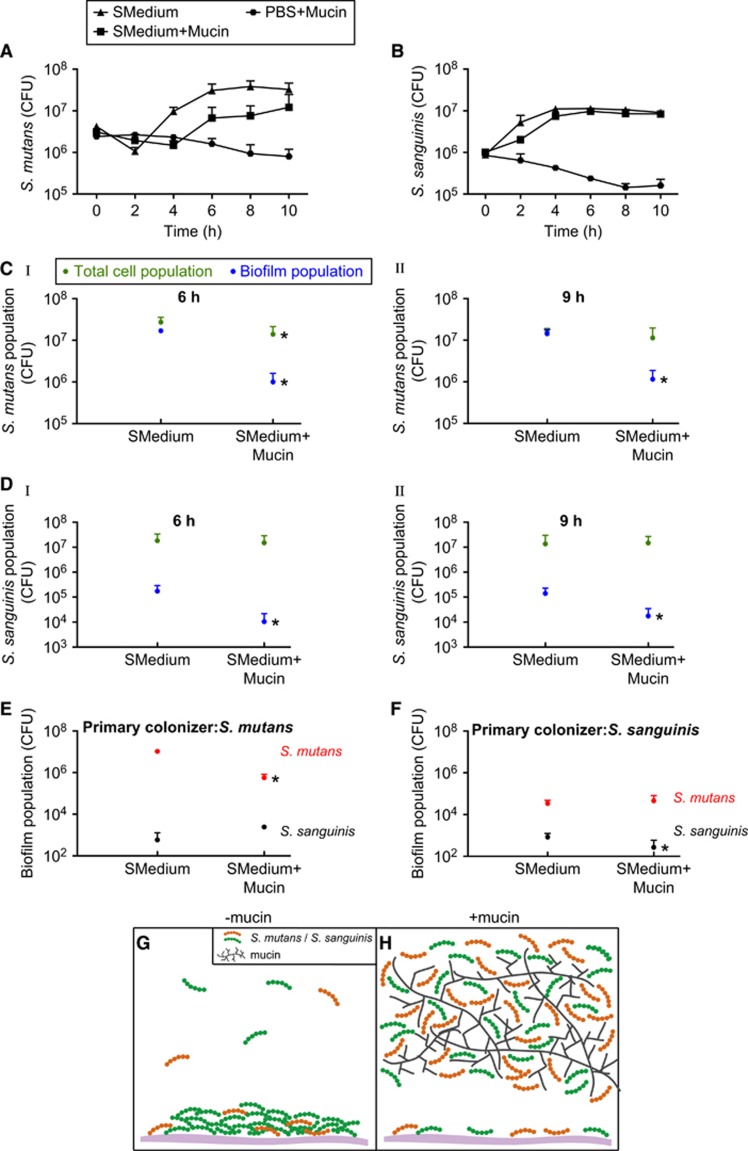
To better understand how MUC5B influences bacterial viability, we studied several aspects of S. mutans and S. sanguinis growth in the presence of MUC5B. First, we determined that the observed protective effect of MUC5B was not due to increased bacterial growth; MUC5B slightly reduces or has no effect on S. mutans and S. sanguinis growth rates (Figures 2A and B). In addition, S. mutans and S. sanguinis did not grow in PBS containing mucin, indicating that MUC5B is not used as a nutrient source under the conditions studied (Figures 2A and B). Another way MUC5B could increase bacterial survival is by altering the cells’ mode of growth, which can influence interspecies competition and cell properties such as gene regulation and cell-cell communication (O’Toole and Kolter, 1998; Pratt and Kolter, 1998; Prigent-Combaret et al., 1999; Kearns et al., 2005; Oliveira et al., 2015; Schluter et al., 2015). Our data show that MUC5B efficiently reduces S. mutans and S. sanguinis biofilm formation in single-species cultures at 6 h and 9 h: S. mutans biofilm formation was reduced by 17- and 12-fold, and S. sanguinis biofilm formation decreased 16- and 8-fold compared with the control without mucin (Figures 2C (I, II) and D (I, II)). Strikingly, the total S. mutans and S. sanguinis cell populations were unchanged at all time points in the presence of MUC5B (except for a slight decrease at 6 h, but the number of cells in the biofilm at this time still account for only 13% of total cells) (Figures 2C (I, II) and D (I, II)). This result implies that, in the presence of MUC5B, the vast majority of cells shifted into the planktonic state. Because the single cell bacterial form can be less competitive than the surface-attached state, this movement of cells away from the biofilm could be a mechanism to reduce interspecies competition. After 4 h of co-culture in the dual-species model, we found that MUC5B also reduced biofilm formation of the primary colonizer in the mixed biofilm by 19-fold for S. mutans and 3-fold for S. sanguinis relative to the control without polymer (Figures 2E and F). As shown in Figures 1A (I) and B (I), the total cell population in each of these cases was unaffected (S. mutans) or increased (S. sanguinis) by MUC5B, indicating a decrease in the relative proportion of biofilm cells. In the case of the secondary colonizer, there was also an overall reduction in the proportion of biofilm cells for both S. mutans and S. sanguinis; the total number of viable cells increased in the presence of MUC5B (Figures 1A (II) and B (II)), yet there was only a slight increase or no change in biofilm formation in the presence of MUC5B relative to the control without polymer (Figures 2E and F). Taken together, these results indicate that MUC5B could enhance bacterial coexistence and, ultimately, bacterial diversity, by shifting competing species away from the biofilm and into the less competitive planktonic state.
Figure 2.
MUC5B reduces S. mutans and S. sanguinis surface attachment by shifting cells into the planktonic state. S. mutans (A) and S. sanguinis (B) growth in control medium (half-strength BHI with 1% sucrose; SMedium), control medium containing 0.4% MUC5B mucin, and PBS containing 0.4% MUC5B mucin. (C (I, II)) S. mutans biofilm population and total cell population in mono-species cultures containing control medium and control medium with 0.4% MUC5B mucin at 6 h (C (I)) and 9 h (C (II)). (D (I, II)) S. sanguinis biofilm population and total cell population in mono-species cultures containing control medium and control medium with 0.4% MUC5B mucin at 6 h (D (I)) and 9 h (D (II)). (E, F) S. mutans and S. sanguinis biofilm formation in control medium and control medium containing 0.4% MUC5B mucin in a dual-species model after 4 h of co-culture when S. mutans was the primary colonizer (E) and when S. sanguinis was the primary colonizer (F). (G, H) schematic illustrating a summary of conclusions. In the absence of MUC5B (G), biofilm formation of S. mutans and S. sanguinis increases and bacterial coexistence decreases compared with biofilm formation and species coexistence in the presence of mucin (H). *, statistically significant decrease relative to the control with half-strength BHI containing 1% sucrose determined by Student’s t test (P<0.05). Experiments were performed in triplicate and error bars represent s.d. of CFU between replicates.
In this work, we use a dual-species bacterial model containing human MUC5B salivary mucin to understand how this prevalent environmental factor influences bacterial viability. Our results show that: (1) MUC5B promotes S. mutans and S. sanguinis coexistence, and (2) MUC5B shifts cells from the biofilm into the planktonic state (Figures 2G and H). By promoting the single-cell (planktonic) state, MUC5B could alter cell-cell interactions, toxin production, or other mechanisms of competition. Although this model is not as complex as the oral cavity microbiome, these findings are among the first to indicate that mucus and its primary structural component, mucins, could influence bacterial survival in a multispecies environment. Further studies are needed, however, to understand if the observed increase in bacterial survival and reduction in surface colonization in the presence of mucin are due to an indirect influence of mucin, such as altered transport of secreted factors, or a direct impact on bacterial physiology, which could change gene regulation.
Acknowledgments
The authors thank Julia Co, Nicole Kavanaugh and Gerardo Cárcamo-Oyarce for their helpful advice and guidance. This work was generously supported by a F30 NIDCR fellowship 1F30DE024917-01A1 (ESF), a 2013 Preterm Birth Research Grant from the Burroughs Wellcome Fund (KR) and a core center grant P30-ES002109 from the National Institute of Environmental Health Sciences, National Institutes of Health (KR).
Footnotes
Supplementary Information accompanies this paper on The ISME Journal website (http://www.nature.com/ismej)
The authors declare no conflict of interest.
Supplementary Material
References
- Bansil R, Turner BS. (2006). Mucin structure, aggregation, physiological functions and biomedical applications. Curr Opin Colloid Interface Sci 11: 164–170. [Google Scholar]
- Caldara M, Friedlander RS, Kavanaugh NL, Aizenberg J, Foster KR, Ribbeck K. (2012). Mucin biopolymers prevent bacterial aggregation by retaining cells in the free-swimming state. Curr Biol 22: 2325–2330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Derrien M, van Passel MW, van de Bovenkamp JH, Schipper RG, de Vos WM, Dekker J. (2010). Mucin-bacterial interactions in the human oral cavity and digestive tract. Gut Microbes 1: 254–268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frenkel ES, Ribbeck K. (2015). Salivary mucins protect surfaces from colonization by cariogenic bacteria. Appl Environ Microbiol 81: 332–338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ge X, Kitten T, Chen Z, Lee SP, Munro CL, Xu P. (2008). Identification of Streptococcus sanguinis genes required for biofilm formation and examination of their role in endocarditis virulence. Infect Immun 76: 2551–2559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ivic A, Onyeaka H, Girling A, Brewis IA, Ola B, Hammadieh N et al. (2002). Critical evaluation of methylcellulose as an alternative medium in sperm migration tests. Hum Reprod 17: 143–149. [DOI] [PubMed] [Google Scholar]
- Kavanaugh NL, Zhang AQ, Nobile CJ, Johnson AD, Ribbeck K. (2014). Mucins suppress virulence traits of Candida albicans. mBio 5: e01911–e01914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kearns DB, Chu F, Branda SS, Kolter R, Losick R. (2005). A master regulator for biofilm formation by Bacillus subtilis. Mol Microbiol 55: 739–749. [DOI] [PubMed] [Google Scholar]
- Kreth J, Merritt J, Shi W, Qi F. (2005). Competition and Coexistence between Streptococcus mutans and Streptococcus sanguinis in the dental biofilm. J Bacteriol 187: 7193–7203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreth J, Zhang Y, Herzberg MC. (2008). Streptococcal antagonism in oral Biofilms: Streptococcus sanguinis and Streptococcus gordonii interference with Streptococcus mutans. J Bacteriol 190: 4632–4640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveira NM, Martinez-Garcia E, Xavier J, Durham WM, Kolter R, Kim W et al. (2015). Biofilm formation as a response to ecological competition. PLoS Biol 13: e1002191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Toole GA, Kolter R. (1998). Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signalling pathways: a genetic analysis. Mol Microbiol 28: 449–461. [DOI] [PubMed] [Google Scholar]
- Pratt LA, Kolter R. (1998). Genetic analysis of Escherichia coli biofilm formation: roles of flagella, motility, chemotaxis and type I pili. Mol Microbiol 30: 285–293. [DOI] [PubMed] [Google Scholar]
- Prigent-Combaret C, Vidal O, Dorel C, Lejeune P. (1999). Abiotic surface sensing and biofilm-dependent regulation of gene expression in Escherichia coli. J Bacteriol 181: 5993–6002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schluter J, Nadell CD, Bassler BL, Foster KR. (2015). Adhesion as a weapon in microbial competition. ISME J 9: 139–149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Senty Turner L, Das S, Kanamoto T, Munro CL, Kitten T. (2009). Development of genetic tools for in vivo virulence analysis of Streptococcus sanguinis. Microbiology 155: 2573–2582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith DJ, Gaffney EA, Gadêlha H, Kapur N, Kirkman-Brown JC. (2009). Bend propagation in the flagella of migrating human sperm, and its modulation by viscosity. Cell Motil Cytoskeleton 66: 220–236. [DOI] [PubMed] [Google Scholar]
- Tabak LA. (1995). In defense of the oral cavity: structure, biosynthesis, and function of salivary mucins. Annu Rev Physiol 57: 547–564. [DOI] [PubMed] [Google Scholar]
- Tabak LA, Levine MJ, Mandel ID, Ellison SA. (1982). Role of salivary mucins in the protection of the oral cavity. J Oral Pathol 11: 1–17. [DOI] [PubMed] [Google Scholar]
- Worlitzsch D, Tarran R, Ulrich M, Schwab U, Cekici A, Meyer KC et al. (2002). Effects of reduced mucus oxygen concentration in airway Pseudomonas infections of cystic fibrosis patients. J Clin Invest 109: 317–325. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.