Abstract
Carcinoembryonic antigen (CEA)–targeted imaging and therapeutic agents are being tested in clinical trials. If CEA overexpression in malignant tissue corresponds with elevated serum CEA, serum CEA could assist in selecting patients who may benefit from CEA-targeted agents. This study aims to assess the relationship between serum CEA and CEA expression in pancreatic (n = 20) and rectal cancer tissues (n = 35) using histopathology. According to local laboratory standards, a serum CEA >3 ng/mL was considered elevated. In pancreatic cancer patients a significant correlation between serum CEA and percentage of CEA-expressing tumor cells was observed (P = .04, ρ = .47). All 6 patients with homogeneous CEA expression in the tumor had a serum CEA >3 ng/mL. Most rectal cancer tissues (32/35) showed homogeneous CEA expression, independent of serum CEA levels. This study suggests that selection of pancreatic cancer patients for CEA-targeted agents via serum CEA appears adequate. For selection of rectal cancer patients, serum CEA levels are not informative.
Keywords: Pancreatic ductal adenocarcinoma, CEACAM5, fluorescence imaging, pancreas, rectum, biomarker
Introduction
Carcinoembryonic antigen (CEA) is a glycoprotein produced by gastrointestinal tissue under control of the cell adhesion molecule 5 (CEACAM5) gene.1 CEA is anchored on the cell membrane via glycosylphosphatidylinositol, which makes it vulnerable for shedding into the lumen, where it is cleared via the feces. In many cancer types, including pancreatic ductal adenocarcinoma (PDAC) and (colo)rectal cancer (CRC), CEA is found to be overexpressed, playing a role in cell recognition, adhesion, angiogenesis, and tumor suppression.2–4 A substantial part of CEA, shedded from tumor cells, derives into the bloodstream, and enhanced serum CEA is therefore used as sensitive biomarker indicating growth or recurrence of CRC.5
Since the recognition of serum CEA as a valuable cancer biomarker, multiple CEA-targeted agents have been developed. Therapeutic antibodies directed towards CEA-overexpressing tumors are currently tested in various phase I–III trials,6,7 but also anti-CEA–based radioimmunotherapy and CEA-targeted vaccines are investigated.8,9 In addition to the rapidly expanding field of CEA-targeted anticancer therapeutics, several imaging agents have become available for clinical testing. These agents can be labeled with radioisotopes for positron emission tomography or single-photon emission computed tomography imaging,10,11 with near-infrared (NIR) fluorescent dyes,12,13 or they can provide both molecular imaging and targeted therapy of CEA-expressing tumors.14
Especially in early phase clinical trials investigating CEA-targeted tumor imaging, it would be beneficial to identify patients with homogeneous CEA overexpression in the tumor prior to inclusion. For example, patient selection is pivotal in the recently initiated phase I study (Netherlands Trial Register ID: 5673) assessing the safety and feasibility of SGM-101, a monoclonal CEA-targeted antibody conjugated to a NIR fluorescent dye, utilized for intraoperative detection of PDAC and CRC. Selecting eligible patients with PDAC and rectal cancer for CEA-targeted applications might be feasible using serum CEA levels. Since CEA expression on normal epithelium is confined to the apical surface of polarized cells,2 it can be hypothesized that CEA in serum of cancer patients originates mostly from shedding by CEA-overexpressing malignant cells that lose their polarity. The aim of this study was to investigate if elevated serum CEA levels can predict overexpression of CEA on pancreatic and rectal cancer tissue.
Methods
This study was approved by the Institutional Ethics Committee of the Leiden University Medical Center (LUMC) and performed following the Code for Proper Secondary Use of Human Tissue, Dutch Federation of Medical Scientific Society. Tissue samples of 20 patients with PDAC and 35 patients with rectal adenocarcinoma, undergoing surgery at the LUMC between 2013 and 2016, were retrospectively collected. Patients were selected on availability of preoperative serum CEA values. Based on hematoxylin-eosin–stained (HE) slides, a single representative tumor containing formalin-fixed paraffin-embedded (FFPE) tissue block from each patient was chosen by a board-certified pathologist (A.F.S.). Serum CEA was measured using routine methodology (Elecsys; Roche Diagnostics) and according to local laboratory standards, serum CEA levels >3 ng/mL were considered elevated.
Immunohistochemistry
Immunohistochemical staining for CEA was performed on FFPE tissue. Tissue samples were stained using a monoclonal mouse antibody against CEACAM5 (clone number CI-P83-1; Santa Cruz Biotechnology). Validation of the staining protocol was performed using human colon cancer tissue as a positive control. Sections (thickness: 4 µm) were cut from paraffin blocks and mounted on adhesive slides (Starfrost). Slides were deparaffinized using xylene and rehydrated in decreasing concentrations of ethanol. Subsequently, slides were rinsed with distilled water, and endogenous peroxidase activity was blocked with 0.3% hydrogen peroxide (Merck Millipore) for 20 minutes. Antigen retrieval was performed in DAKO PT Link with target retrieval solution pH 6.0 at 95°C for 10 minutes. After rinsing with phosphate-buffered saline (PBS) the primary antibody was incubated overnight at room temperature and afterwards rinsed with PBS. Incubation of the secondary antibody (Envision anti-mouse HRP; Dako) was performed for 30 minutes at room temperature, followed by rinsing with PBS. Antibody binding was then visualized using 3,3′-diaminobenzidine (DAKO). Sections were counterstained with hematoxylin (Klinipath), rinsed in tap water, dehydrated, and mounted with Pertex. All slides were scanned using the Philips Ultra Fast Scanner 1.6 RA.
Scoring system
CEA expression in cancer tissue was determined by immunohistochemical staining (brown) and scored for staining intensity (0: none, 1: weak, 2: moderate, 3: strong) and the fraction of positive tumor cells (1: <10%, 2: 10%–50%, 3: 50%–80%, 4 > 80%). Homogeneity was defined when >80% of tumor cells stained positive for CEA (=score 4). All tissue specimens were scored by 2 independent observers (A.F.S. and L.S.F.B.); disagreements were resolved by consensus after reviewing the relevant slides.
Statistical analysis
Statistical analysis was performed using SPSS version 23.0 software (SPSS, IBM Corporation). The concordance of serum CEA and CEA expression on tumor tissue and of pathological T (pT) stage and serum CEA was assessed using Spearmans’ test, defined as the correlation coefficient. The correlation between groups, based on an elevated or normal level of serum CEA and between smokers and nonsmokers, was calculated using the Mann-Whitney U test. A χ2 test was used to compare homogeneity in PDAC tissues. In all tests, results were considered statistically significant at the level of P < .05.
Results
Patient and tumor characteristics
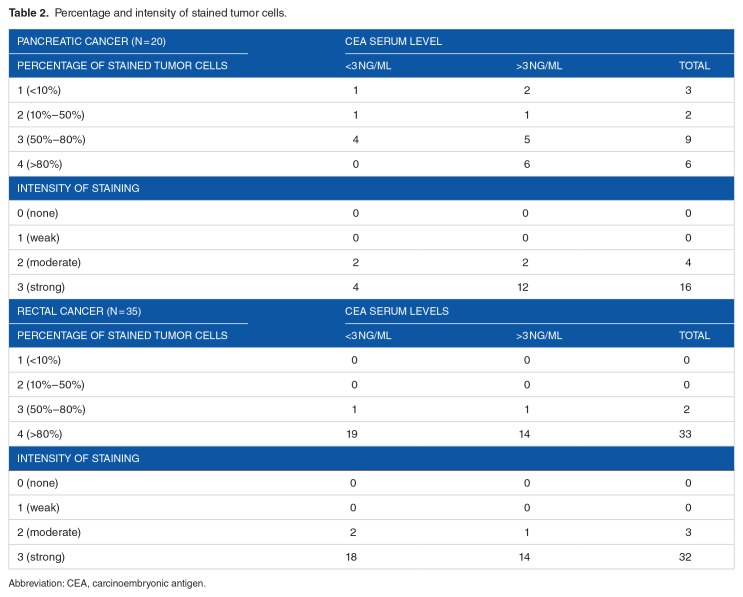
Patient and tumor characteristics are summarized in Table 1. In the pancreatic cancer cohort, 3 patients participated in the PREOPANC study protocol; 2 of them received preoperative chemoradiotherapy (CRT).15 In the rectal cancer cohort, 8 patients participated in the RAPIDO study protocol.16 All serum CEA measurements were performed prior to the start of CRT. Median preoperative serum CEA of smoking patients with PDAC was 3.8 (range: 1.2-6.9), compared with 3.7 (range: 0.6-23.8) for non-smokers (P = .50). For rectal cancer patients, median preoperative serum CEA of smoking patients was 2.9 (range: 1.0-16.9), compared with 2.7 for non-smokers (range: 1.3–9.9; P = .68). No significant correlation was found between pT stage and preoperative serum CEA (P = .61, ρ = .09 for PDAC patients and P = .95, ρ = .02 for rectal cancer patients).
Table 1.
Patient and tumor characteristics.
CEA expression and correlation to serum CEA level
PDAC cohort
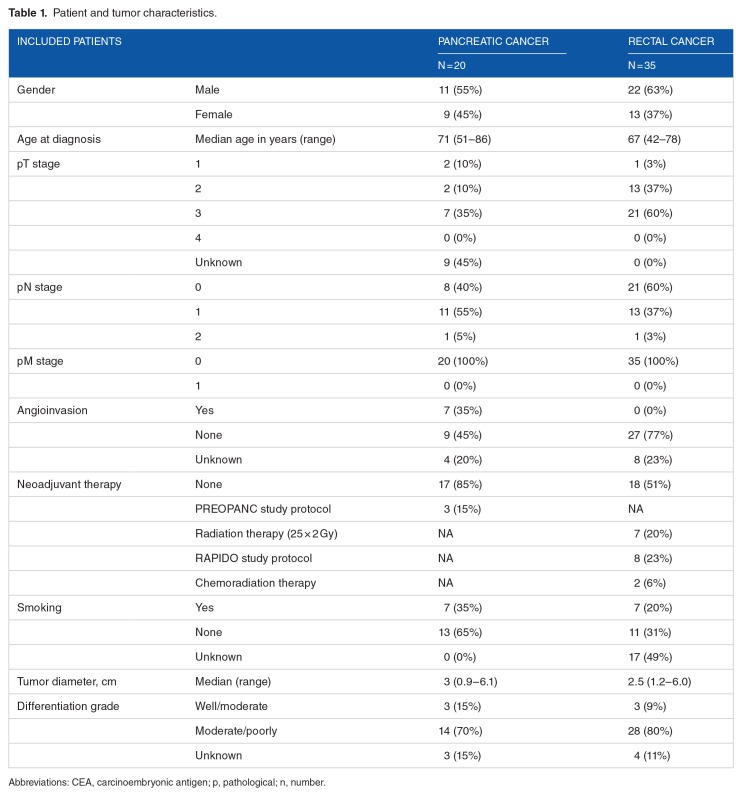
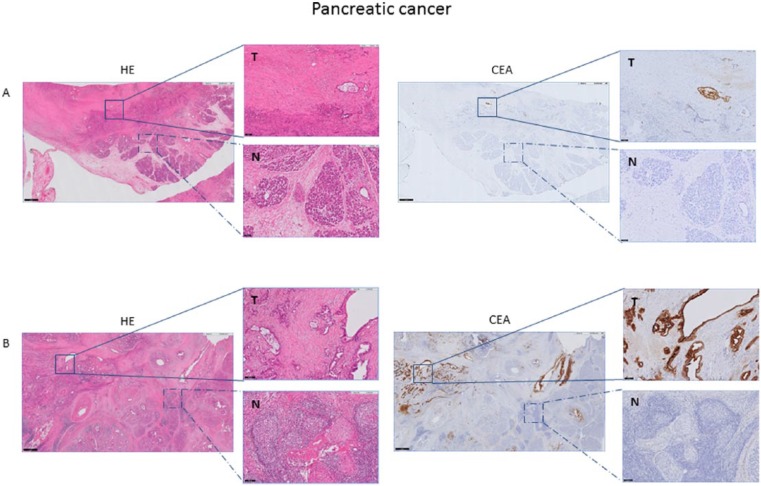
The majority of tumors showed a heterogeneous CEA expression pattern, whereas only 6 of 20 tumors showed homogeneous CEA expression (>80%). CEA expression was mostly seen on the luminal surface of neoplastic glands and necrotic tissue; low levels of immunoreactivity were seen in the stromal tissue, while normal acini did not express CEA (Figure 1). A significant correlation was found between preoperative serum CEA levels and percentage of CEA-expressing tumor cells (P = .04, ρ = .47). This could not be shown for intensity of the staining (P = .21, ρ = .29). 15/20 tissues showed >50% CEA-expressing tumor cells and 16 of 20 tissues showed a high intensity of CEA expression (Table 2). When dividing the patients in groups of normal (n = 6) and elevated serum CEA (n = 14), no significant correlation between groups and percentage and intensity of stained tumor cells was found (P = .15 and P = .34, respectively). However, all patients with tumors showing homogeneous CEA expression (n = 6) had elevated serum CEA levels (χ2 = 5.06, P = .03).
Figure 1.
Representative example of HE and CEA staining of pancreatic cancer tissue. CEA expression of tumor (T) tissue compared with adjacent normal (N) pancreatic tissue, derived from a patient with a preoperative serum CEA level <3ng/mL (A) and >3ng/mL (B) magnification 1x and 10x). The magnified image in (A) shows moderate CEA expression on the luminal side of neoplastic glands, while tumor tissue in image (B) shows an intense, circumferential CEA expression. The adjacent normal acini do not stain positive. CEA indicates carcinoembryonic antigen; HE, hematoxylin-eosin.
Table 2.
Percentage and intensity of stained tumor cells.
Rectal cancer cohort
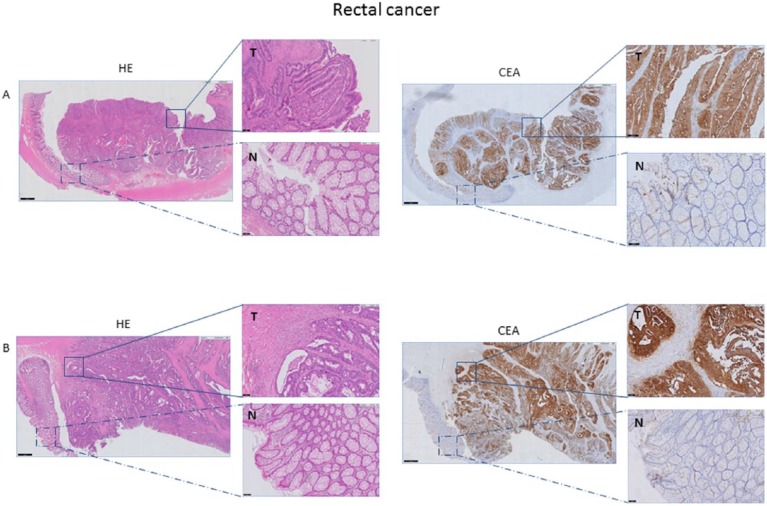
Almost all tumors (32/35, 91%) showed homogeneous CEA expression. CEA expression was found on luminal sides of malignant cells and in the majority of tumors, circumferential CEA staining was shown. Normal epithelium showed low levels of CEA immunostaining, which was far less than the CEA expression in cancer cells (Figure 2). No significant correlation was found between preoperative serum CEA and percentage and intensity of CEA-expressing tumor cells (P = .58, ρ = .10 and P = .17, ρ = .24 respectively). When dividing patients in groups of normal (n = 20) and elevated serum CEA (n = 15), again no significant correlation between groups and the percentage and intensity of CEA-expressing tumor cells was found (Table 2).
Figure 2.
Representative example of HE and CEA staining of rectal cancer tissue. CEA expression of tumor tissue (T) compared with adjacent normal (N) rectal tissue, derived from a patient with a preoperative serum CEA level <3gn/mL (A) and >3 ng/mL (B) magnification 1x and 10x). Both tumor tissues (A and B) show an intense, circumferential CEA expression, independent of the preoperative serum CEA level. Normal epithelium shows weak expression. CEA indicates carcinoembryonic antigen; HE, hematoxylin-eosin.
Discussion
This study shows a significant correlation between serum CEA and the percentage of CEA-expressing tumor cells in PDAC tissue. Moreover, serum CEA levels were elevated in all PDAC patients with homogeneous CEA expression in the tumor (>3 ng/mL, χ2 = 5.06, P = .03). These results suggest therefore that especially patients with elevated serum CEA levels could benefit from CEA-targeted agents. Importantly, normal acini did not show any expression of CEA. Personalized oncological care, to ensure that cancer patients benefit from targeted imaging and therapeutics, is already performed for other targets. For example, HER2-targeted drugs (i.e., trastuzumab) are recommended when >30% of tumor cells show a complete, circumferential and intense membrane staining.17
PDAC patient selection is justified as not all tissues express CEA. In this study, only 6 of 20 tissues showed homogeneous CEA expression and 15 of 20 tissues showed expression in >50% of neoplastic pancreatic cells. Previous studies reported CEA expression in approximately 60% to 75% of PDAC tissues.3,18 Serum CEA measurements were performed using the Elecsys CEA assay (Roche Diagnostics), which is previously described to be specific for CEACAM5 as well as for NCA-2, the nonspecific cross reacting antigen-2.19 NCA-2 is a truncated form of CEACAM5 with identical amino acid sequence, and therefore CEACAM5 was used as Several other CEACAM molecules have been previously described, each with a slightly different function.21,22 Gebauer et al3 investigated CEACAM1, CEACAM5, and CEACAM6 as suitable biomarkers for PDAC in a tissue microarray (TMA; n = 252). No significant correlation between elevated serum levels, eg, >4.7 ng/mL, and tissue expression of one of these CEACAM subtypes could be shown. Yet, a significant correlation between CEACAM5 and CEACAM6 tissue expression and decreased overall and disease-free survival was demonstrated. The partly different results compared with this study might be attributable to varying cut-off values for defining elevated serum CEA or differences in the staining or scoring protocol. Importantly, no correlation between serum CEA and homogeneity of CEA expression in PDAC tissues could be studied in this TMA study. No strict threshold of CEA tissue expression to ensure optimal applicability of CEA-targeted imaging or therapeutic agents has yet been defined. However, patients with homogeneous CEA expression are expected to benefit most from those agents.
In contrast to PDAC patients, no significant correlation between serum CEA and CEA tissue expression was found in the rectal cancer cohort. A possible cause for this finding might be the difference in vasculature and permeability of the two tumor types. Rectal tumors are generally well- vascularized and show a relatively good response to chemo(radio)therapy (CRT).23 PDAC shows poor blood supply and high amounts of stroma, possibly explaining the disappointing response to chemotherapy and the low 5-year survival rates of less than 5%.24 It can be hypothesized that CEA derived from rectal tumors accesses the bloodstream easier than CEA from PDAC, leading to higher CEA serum levels. However, no significant correlation between preoperative serum CEA and pT stage of tumors was found in this study. Previous reports have indicated that several other factors, including tumor necrosis, smoking, benign liver disease, and the location of the CEA receptor on the cell membrane could also affect serum CEA levels.25,26
Park et al27 showed that CEA serum concentrations were significantly higher in patients with CEA-positive tumors than CEA-negative tumors, but this correlation could not be reproduced in other studies.28–30 It is nowadays accepted that rectal and colon cancer should be categorized as two different types of malignancies and treated differently. This study differs from others as only patients with rectal cancer were included. Nearly all tissues showed an intense and homogeneous expression compared with low expression on normal epithelium, independently of the neoadjuvant therapy given nor the preoperative serum CEA level. Although a relatively low number of patients with rectal cancer were included, serum CEA levels, assessed prior to the start of preoperative CRT, are concordant with literature.31 Thus, all the patients with rectal cancer, independent of their serum CEA level, might benefit from CEA-targeted agents.
A limitation of this small, retrospective study is that different intervals elapsed between measurement of the CEA serum concentrations and resection. Over- or underrating serum CEA levels, as a consequence of the timing of CEA measurement, could have influenced the outcome of the study. Moreover, it is possible that selection bias has occurred because not all serum CEA levels were measured in all subsequent patients between 2013 and 2016. Both of these issues could be avoided by designing a prospective study in which serum CEA is measured directly prior to surgery. In addition, the effect of neoadjuvant therapy on serum CEA level and CEA expression on rectal and pancreatic cancer tissues should be studied further.
In conclusion, a significant correlation between serum CEA levels and percentage of CEA-expressing tumor cells in PDAC tissue was shown. PDAC patients that might benefit from CEA-targeted imaging or therapeutic agents can adequately be selected using serum CEA levels (>3 ng/mL). This finding can be used in the process of personalized cancer care. Selection based on serum CEA levels seems not useful in patients with rectal cancer, as almost all show an intense, homogeneous CEA expression.
Acknowledgments
The authors gratefully acknowledge H.A.J.M. Prevoo and S. Bhairosingh for their support and immunohistochemical expertise.
Footnotes
PEER REVIEW: Five peer reviewers contributed to the peer review report. Reviewers’ reports totaled 716 words, excluding any confidential comments to the academic editor.
FUNDING: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Dutch Cancer Society grant UL2010-4732.
DECLARATION OF CONFLICTING INTERESTS: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
LSFB, CESH, HJMH, and MJMV conceived and designed the experiments. LSFB, FAV, and AFS analyzed the data. LSFB, FAV, CESH, HJMH, and MJMV wrote the first draft of the manuscript. AFS, CFMS, CJHV, JB, and ALV contributed to the writing of the manuscript. All the authors agree with manuscript results and conclusions. LSFB, FAV, CFMS, AFS, and ALV jointly developed structure and arguments for the paper. CESH, HJMH, MJMV, CJHV, AFS, JB, and ALV made critical revisions and approved final version. All authors reviewed and approved the final manuscript.
REFERENCES
- 1.Nap M, Mollgard K, Burtin P, Fleuren GJ. Immunohistochemistry of carcino-embryonic antigen in the embryo, fetus and adult. Tumour Biol. 1988;9:145–153. doi: 10.1159/000217555. [DOI] [PubMed] [Google Scholar]
- 2.Hammarstrom S. The carcinoembryonic antigen (CEA) family: structures, suggested functions and expression in normal and malignant tissues. Semin Cancer Biol. 1999;9:67–81. doi: 10.1006/scbi.1998.0119. [DOI] [PubMed] [Google Scholar]
- 3.Gebauer F, Wicklein D, Horst J, et al. Carcinoembryonic antigen-related cell adhesion molecules (CEACAM) 1, 5 and 6 as biomarkers in pancreatic cancer. PLoS ONE. 2014;9:e113023. doi: 10.1371/journal.pone.0113023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li M, Li JY, Zhao AL, et al. Comparison of carcinoembryonic antigen prognostic value in serum and tumour tissue of patients with colorectal cancer. Colorectal Dis. 2009;11:276–281. doi: 10.1111/j.1463-1318.2008.01591.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Locker GY, Hamilton S, Harris J, et al. ASCO 2006 update of recommendations for the use of tumor markers in gastrointestinal cancer. J Clin Oncol. 2006;24:5313–5327. doi: 10.1200/JCO.2006.08.2644. [DOI] [PubMed] [Google Scholar]
- 6.Bacac M, Fauti T, Sam J, et al. A novel carcinoembryonic antigen T-cell bispe-cific antibody (CEA TCB) for the treatment of solid tumors. Clin Cancer Res. 2016;22:3286–3297. doi: 10.1158/1078-0432.CCR-15-1696. [DOI] [PubMed] [Google Scholar]
- 7.Boonstra MC, de Geus SW, Prevoo HA, et al. Selecting targets for tumor imaging: an overview of cancer-associated membrane proteins. Biomark Cancer. 2016;8:119–133. doi: 10.4137/BIC.S38542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sahlmann CO, Homayounfar K, Niessner M, et al. Repeated adjuvant anti-CEA radioimmunotherapy after resection of colorectal liver metastases: safety, feasibility, and long-term efficacy results of a prospective phase 2 study. Cancer. 2017;123:638–649. doi: 10.1002/cncr.30390. [DOI] [PubMed] [Google Scholar]
- 9.Duggan MC, Jochems C, Donahue RN, et al. A phase I study of recombinant (r) vaccinia-CEA(6D)-TRICOM and rFowlpox-CEA(6D)-TRICOM vaccines with GM-CSF and IFN-α-2b in patients with CEA-expressing carcinomas. Cancer Immunol Immunother. 2016;65:1353–1364. doi: 10.1007/s00262-016-1893-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bodet-Milin C, Faivre-Chauvet A, Carlier T, et al. Immuno-PET using anticar-cinoembryonic antigen bispecific antibody and 68Ga-labeled peptide in metastatic medullary thyroid carcinoma: clinical optimization of the pretargeting parameters in a first-in-human trial. J Nucl Med. 2016;57:1505–1511. doi: 10.2967/jnumed.116.172221. [DOI] [PubMed] [Google Scholar]
- 11.Schoffelen R, van der Graaf WT, Sharkey RM, et al. Pretargeted immuno-PET of CEA-expressing intraperitoneal human colonic tumor xenografts: a new sensitive detection method. EJNMMI Res. 2012;2:5. doi: 10.1186/2191-219X-2-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Boonstra MC, Tolner B, Schaafsma BE, et al. Preclinical evaluation of a novel CEA-targeting near-infrared fluorescent tracer delineating colorectal and pancreatic tumors. Int J Cancer. 2015;137:1910–1920. doi: 10.1002/ijc.29571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Vahrmeijer AL, Hutteman M, van der Vorst JR, van de Velde CJ, Frangioni JV. Image-guided cancer surgery using near-infrared fluorescence. Nat Rev Clin Oncol. 2013;10:507–518. doi: 10.1038/nrclinonc.2013.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Knutson S, Raja E, Bomgarden R, et al. Development and evaluation of a fluorescent antibody-drug conjugate for molecular imaging and targeted therapy of pancreatic cancer. PLoS ONE. 2016;11:e0157762. doi: 10.1371/journal.pone.0157762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Versteijne E, van Eijck CH, Punt CJ, et al. Preoperative radiochemotherapy versus immediate surgery for resectable and borderline resectable pancreatic cancer (PREOPANC trial): study protocol for a multicentre randomized controlled trial. Trials. 2016;17:127. doi: 10.1186/s13063-016-1262-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nilsson PJ, van Etten B, Hospers GA, et al. Short-course radiotherapy followed by neo-adjuvant chemotherapy in locally advanced rectal cancer—the RAPIDO trial. BMC Cancer. 2013;13:279. doi: 10.1186/1471-2407-13-279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wolff AC, Hammond ME, Hicks DG, et al. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. Arch Pathol Lab Med. 2014;138:241–256. doi: 10.5858/arpa.2013-0953-SA. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.de Geus SW, Boogerd LS, Swijnenburg RJ, et al. Selecting tumor-specific molecular targets in pancreatic adenocarcinoma: paving the way for image-guided pancreatic surgery. Mol Imaging Biol. 2016;18:807–819. doi: 10.1007/s11307-016-0959-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hanada H, Mugii S, Takeoka K, et al. Early detection of colorectal cancer metastasis and relapse by recognizing nonspecific cross-reacting antigen 2 in commercial carcinoembryonic antigen assays. Clin Chem. 2009;55:1747–1748. doi: 10.1373/clinchem.2008.121897. [DOI] [PubMed] [Google Scholar]
- 20.Schee K, Flatmark K, Holm R, Boye K, Paus E. Investigation of nonspecific cross-reacting antigen 2 as a prognostic biomarker in bone marrow plasma from colorectal cancer patients. Tumour Biol. 2012;33:73–83. doi: 10.1007/s13277-011-0247-5. [DOI] [PubMed] [Google Scholar]
- 21.Simeone DM, Ji B, Banerjee M, et al. CEACAM1, a novel serum biomarker for pancreatic cancer. Pancreas. 2007;34:436–443. doi: 10.1097/MPA.0b013e3180333ae3. [DOI] [PubMed] [Google Scholar]
- 22.Kuespert K, Pils S, Hauck CR. CEACAMs: their role in physiology and patho-physiology. Curr Opin Cell Biol. 2006;18:565–571. doi: 10.1016/j.ceb.2006.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Renehan AG, Malcomson L, Emsley R, et al. Watch-and-wait approach versus surgical resection after chemoradiotherapy for patients with rectal cancer (the OnCoRe project): a propensity-score matched cohort analysis. Lancet Oncol. 2016;17:174–183. doi: 10.1016/S1470-2045(15)00467-2. [DOI] [PubMed] [Google Scholar]
- 24.Ilic M, Ilic I. Epidemiology of pancreatic cancer. World J Gastroenterol. 2016;22:9694–9705. doi: 10.3748/wjg.v22.i44.9694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wagener C, Muller-Wallraf R, Nisson S, Groner J, Breuer H. Localization and concentration of carcinoembryonic antigen (CEA) in gastrointestinal tumors: correlation with CEA levels in plasma. J Natl Cancer Inst. 1981;67:539–547. [PubMed] [Google Scholar]
- 26.Hamada Y, Yamamura M, Hioki K, Yamamoto M, Nagura H, Watanabe K. Immunohistochemical study of carcinoembryonic antigen in patients with colorectal cancer. Correlation with plasma carcinoembryonic antigen levels. Cancer. 1985;55:136–141. doi: 10.1002/1097-0142(19850101)55:1<136::aid-cncr2820550121>3.0.co;2-p. [DOI] [PubMed] [Google Scholar]
- 27.Park JW, Chang HJ, Kim BC, Yeo HY, Kim DY. Clinical validity of tissue car-cinoembryonic antigen expression as ancillary to serum carcinoembryonic antigen concentration in patients curatively resected for colorectal cancer. Colorectal Dis. 2013;15:e503–e511. doi: 10.1111/codi.12304. [DOI] [PubMed] [Google Scholar]
- 28.Cosimelli M, De Peppo F, Castelli M, et al. Multivariate analysis of a tissue CEA, TPA, and CA 19.9 quantitative study in colorectal cancer patients. A preliminary finding. Dis Colon Rectum. 1989;32:389–397. doi: 10.1007/BF02563690. [DOI] [PubMed] [Google Scholar]
- 29.Nakagoe T, Sawai T, Ayabe H, et al. Prognostic value of carcinoembryonic antigen (CEA) in tumor tissue of patients with colorectal cancer. Anticancer Res. 2001;21:3031–3036. [PubMed] [Google Scholar]
- 30.Nazato DM, Matos LL, Waisberg DR, Souza JR, Martins LC, Waisberg J. Prognostic value of carcinoembryonic antigen distribution in tumor tissue of colorectal carcinoma. Arq Gastroenterol. 2009;46:26–31. doi: 10.1590/s0004-28032009000100010. [DOI] [PubMed] [Google Scholar]
- 31.Probst CP, Becerra AZ, Aquina CT, et al. Watch and wait?—elevated pretreatment CEA is associated with decreased pathological complete response in rectal cancer. J Gastrointest Surg. 2016;20:43–52. doi: 10.1007/s11605-015-2987-9. discussion 52. [DOI] [PubMed] [Google Scholar]