Abstract
Osteoporosis is a common skeletal disorder, resulting from an imbalance in bone resorption relative to formation. Bone morphogenetic protein (BMP) is a key regulator in bone formation and osteoblastic differentiation. Hence, compounds that promote BMP expression may be suitable candidates for osteoporosis treatment. This study examined the effects of the traditional Chinese medicinal agent, Kuei-Lu-Er-Xian-Jiao (KLEXJ), on BMP-2 production in osteoblasts. We found that KLEXJ extract promoted osteoblastic differentiation marker ALP activity and increased BMP-2 production; pretreatment with PI3K and Akt inhibitors, or small interfering RNA (siRNA), reduced these effects. KLEXJ also enhanced PI3K and Akt phosphorylation. Treatment of osteoblastic cells with NF-κB inhibitors (TPCK or PDTC) markedly inhibited KLEXJ-enhancement of ALP activity and BMP-2 production. KLEXJ also significantly promoted p65 phosphorylation, while treatment with PI3K and Akt inhibitors antagonized KLEXJ-enhanced p65 phosphorylation. Thus, KLEXJ enhances ALP activity and BMP-2 production of osteoblasts through the PI3K/Akt/ NF-κB signaling pathway and hence may be suitable in the treatment of osteoporosis.
Keywords: Kuei-Lu-Er-Xian-Jiao, Osteoblasts, BMP-2, ALP activity China Medical University
Abbreviations
- ALP
alkaline phosphatase
- BMP
bone morphogenetic protein
- KLEXJ
Kuei-Lu-Er-Xian-Jiao
- TCM
Traditional Chinese Medicine
- FBS
fetal bovine serum
- qPCR
quantitative real-time polymerase chain reaction
1. Introduction
Bone is a mineralized organ containing several types of cells, including osteoblasts (bone-forming cells) and osteoclasts (bone-resorbing cells), which subject bone to a continuous renewal and repair process during the life of each individual by the process of bone remodelling [1, 2]. Bone resorption and osteogenic functions must be in balance in order to maintain a constant bone mass [3, 4]. Compounds that promote osteoblastic proliferation or enhance differentiation of osteoblasts result in increased bone formation [5–7]. At this time, teriparatide®, the recombinant 1-34 fragment of human parathyroid hormone (rhPTH1-34), is the first bone formation agent to be approved for the treatment of osteoporosis [8, 9].
Up until now, the detailed molecular mechanism of osteoporosis has remained unclear, albeit the process is probably correlated with reduced availability or activity of growth factors. For example, bone morphogenetic proteins (BMPs), [10] were first discovered due to their ability to promote bone formation in rodents. The protein structure of BMPs resembles that of the transforming growth factor-β superfamily [11]. It is known that BMP-2 plays a critical role in osteoblastic diffemtiation and bone formation by increasing osteopontin, collagen and proteoglycan production, as well as promoting alkaline phosphatase (ALP) activity [12]. Previous research has also linked osteoporosis with specific polymorphisms in the BMP-2 gene, confirming an association with osteoporosis [13].
Traditional Chinese Medicine (TCM) is a popular component of health care in Taiwan that provides one therapeutic option for osteoporosis treatment. Emerging studies indicate that TCM promotes bone formation and prevents bone loss in the ovariectomized rat model [14, 15]. The TCM drug Kuei-Lu-Er-Xian-Jiao (KLEXJ) is a multicomponent Chinese herbal supplement that has been used for treatment of degenerative joint diseases without adverse effects for over 2, 000 years [16, 17]. However, its role in osteoblastic function remains largely unknown. We report that KLEXJ extract increases osteoblastic differentiation marker ALP activity and BMP-2 production in osteoblasts, while simultaneously mediating the PI3 K/Akt-NF-κB pathway. Our findings suggest that KLEXJ may be useful in the treatment of osteoporosis.
2. Experimental section
2.1. Materials
Kuei-Lu-Er-Xian-Jiao (KLEXJ) contains Testudinis Plastrum (species: Chinemys reevesii; Animal part: plastrum); Cervi cornu (species: Cervus elaphus; animal part: antler); Radix Ginseng (species: Panax ginseng C. A. Meyer; plant part: root) and Lycii fructus (species: Lycium barbarum; plant part: fruit) and was prepared as follows: Testudinis Plastrum and Cervi cornu were stewed for 7 days, after which Radix Ginseng and Lycii fructus were added into the mixture. A 6.25 g extract was derived from the ratio between the 4 components, consisting of about 5 g of Testudinis Plastrum, 10 g of Cornu cervi, 0.55 g of Radix Ginseng, 1.1 g of Lycii fructus, which was provided by the LiAn Biotechnology Pharmaceutical Company (Tainan; Taiwan). Li-An Biotechnology Pharmaceutical Company was awarded the Good Manufacturing Practice certification in Taiwan (Drug license number-013857, issued by the Department of Health, Taiwan). Rabbit monoclonal antibodies specific for BMP-2, p85, Akt, p65, p-p85, p-Akt, p-p65 and b-actin, as well as anti-mouse and anti-rabbit IgG-conjugated horseradish peroxidase, were all purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). The BMP-2 ELISA kit was obtained from Biosource Technology (Nivelles, Belgium). TRIzol reagent, Lipofectamine 2000, and the MMLV RT kit were obtained from Invitrogen (Carlsbad, CA, USA). The control, p85 and Akt siRNA were obtained from Dharmacon Research (Lafayette, CO, USA). The TaqMan assay kit was obtained from Thermo Fisher Scientific (Grand Island, NY, USA). LY294002 and other pharmacological inhibitors were purchased from Sigma-Aldrich (St. Louis, MO, USA).
2.2. Cell culture
The mouse osteoblast cell line MC3T3-E1 was obtained from American Type Culture Collection (Manassas, VA, USA). Cells were maintained in humidified air containing 5% CO2 at 37°C with a-minimum essential medium (MEM), 10% fetal bovine serum (FBS), 100 units/ml penicillin and 100 mg/ml streptomycin (Gibco-BRL Life technologies; Grand Island, NY, USA).
2.3. ALP activity assay
Osteoblasts were treated with KLEXJ for 24 h and then resolved with 0.2% Nonidet P-40. The medium was collected and ALP activity was examined by a commercial ALP activity detection kit (Sigma-Aldrich, St. Louis, MO, US) following manufacturer’s instructions.
2.4. Western blotting
Cellular lysates were prepared as our prior study [18–20]. Proteins were resolved by SDS-polyacrylamide gel electrophoresis and then transferred to polyvinyldifluoride membranes. The blot membranes were blocked with 4% non-fat milk for 1 h at room temperature, followed by incubation with primary antibodies at 4°C for overnight. After washing three times, the blots were incubated with anti-rabbit or anti-mouse HRP-conjugated secondary antibodies for 1 h at room temperature. Finally, the blots were visualized by enhanced chemiluminescence, using a Fujifilm LAS-3000 chemiluminescence detection system (Fujifilm; Tokyo, Japan).
2.5. Quantitative real-time polymerase chain reaction (qPCR)
Total RNA was extracted from MC3T3-E1 cells using TRIzol reagent. Messenger RNA was reversely transcribed to complementary DNA using an MMLV RT kit, and qPCR was then performed using the Taqman assay kit [21].
2.6. Statistical analysis
Data are presented as mean ± standard error of mean (SEM). Statistical analysis of both samples used the Student’s t test. Statistical comparisons of more than two groups were performed by oneway analysis of variance with Bonferroni’s post-hoc test; p < 0.05 was considered significant.
3. Results
3.1. KLEXJ enhances ALP activity and BMP-2 production in osteoblasts
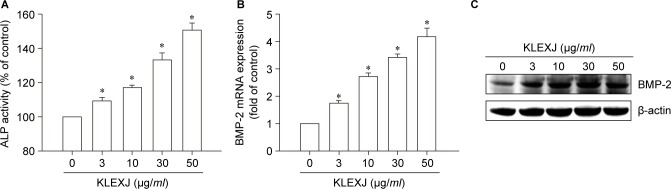
Differentiated osteoblasts express high ALP activity, rendering ALP activity a key marker for osteoblastic formation [22, 23]. When we examined the role of KLEXJ in ALP activity, we found that incubation of osteoblasts with KLEXJ significantly augmented ALP activity (Fig. 1A). As BMP-2 has been reported to play a key role in osteoblastic differentiation [10], we next examined whether KLEXJ promotes osteoblastic differentiation by regulating BMP-2 expression. We found that incubation with KLEXJ stimulated BMP-2 mRNA expression and protein production, in a concentration-dependent manner (Fig. 1B&C). These combined findings indicate that KLEXJ promotes osteoblastic differentiation marker ALP activity and BMP-2 production in osteoblasts.
Fig. 1.
KLEXJ extract enhances ALP activity and BMP-2 expression in osteoblasts. (A) Osteoblasts were treated with KLEXJ for 48 h and ALP activity was examined by a commercial ALP assay kit. (B&C) Osteoblasts were treated with KLEXJ for 24 h and BMP-2 expression was examined by qPCR and Western blot analysis. Results are expressed as mean ± S.E.M.*, p < 0.05 compared with control.
3.1. KLEXJ enhances ALP activity and BMP-2 production through the PI3K/Akt signaling pathway
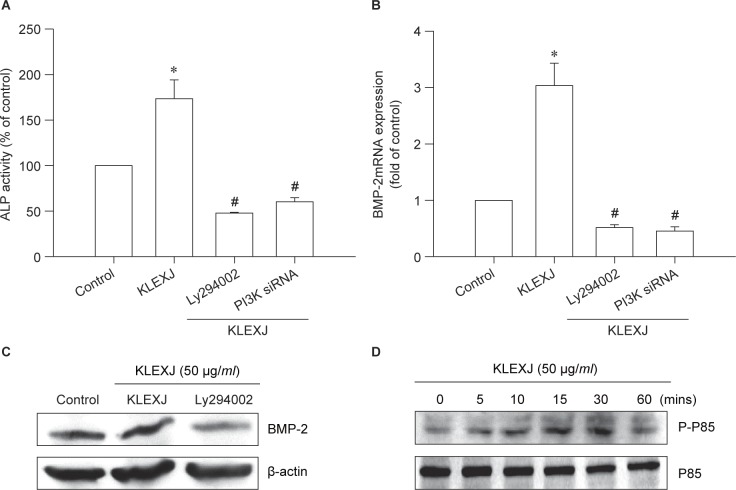
PI3K activation has been described as mediating bone formation and differentiation [24, 25]. The effects of KLEXJ were tested on the PI3K pathway. Incubation with the PI3K inhibitor Ly294002 or transfection with PI3K siRNA markedly abolished KLEXJ-induced ALP activity and BMP-2 expression (Fig. 2A-C). Treatment of osteoblasts with KLEXJ promoted phosphorylation of PI3K in a time-dependent manner (Fig. 2D). Thus, KLEXJ enhances BMP-2 production in osteoblasts via PI3K activation.
Fig. 2.
KLEXJ extract promotes ALP activity and BMP-2 expression through the PI3K pathway. (A) Osteoblasts were pretreated with Ly294002 (10 μM) for 30 min or transfected with p85 siRNA for 24 h, followed by stimulation with KLEXJ for 48 h; ALP activity was examined with a commercial ALP assay kit. (B&C) Osteoblasts were pretreated with Ly294002 (10 μM) for 30 min or transfected with p85 siRNA for 24 h, followed by stimulation with KLEXJ for 24 h; BMP-2 expression was examined by qPCR and Western blot analysis. (D) Osteoblasts were incubated with KLEXJ for indicated time intervals and p85 phosphorylation was examined by Western blot analysis. Results are expressed as mean ± S.E.M.*, p < 0.05 compared with control. #, p < 0 .05 compared with KLEXJ-treated group.
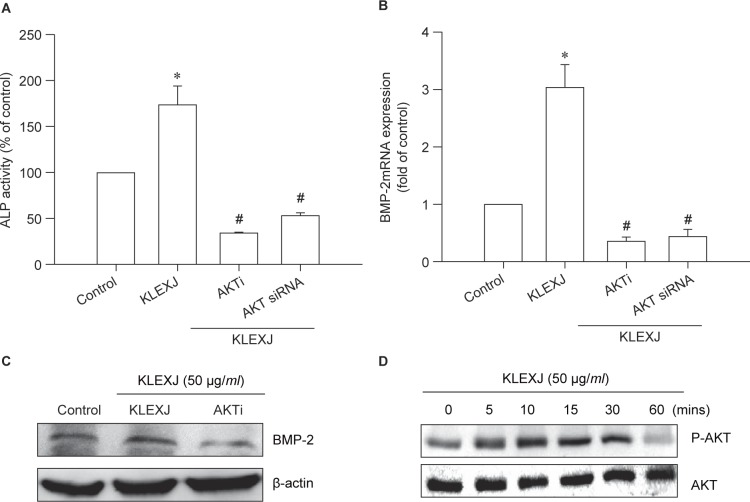
Akt is a downstream pathway in PI3K signaling and plays an important role in osteoblastic function [14, 26]. We therefore studied whether KLEXJ also activates the Akt signaling pathway. We found that an Akt inhibitor or Akt siRNA abolished KLEXJ-induced ALP activity and BMP-2 production (Fig. 3A-C). In addition, Akt phosphorylation was increased after KLEXJ stimula tion (Fig. 3D), suggesting that KLEXJ enhances ALP activity and BMP-2 production in osteoblasts through the Akt pathway.
Fig. 3.
KLEXJ extract promotes ALP activity and BMP-2 expression through the Akt pathway. (A) Osteoblasts were pretreated with an Akt inhibitor (10 μM) for 30 min or transfected with Akt siRNA for 24 h, followed by stimulation with KLEXJ for 48 h. ALP activity was examined using a commercial ALP assay kit. (B&C) Osteoblasts were pretreated with an Akt inhibitor (10 μM) for 30 min or transfected with Akt siRNA for 24 h, followed by stimulation with KLEXJ for 24 h. BMP-2 expression was examined by qPCR and Western blot analysis. (D) Osteoblasts were incubated with KLEXJ for indicated time intervals and Akt phosphorylation was examined by Western blot analysis. Results are expressed as mean ± S.E.M.*, p < 0.05 compared with control. #, p < 0.05 compared with KLEXJ-treated group.
3.2. KLEXJ increases ALP activity and BMP-2 production in osteoblasts via the NF-κB pathway
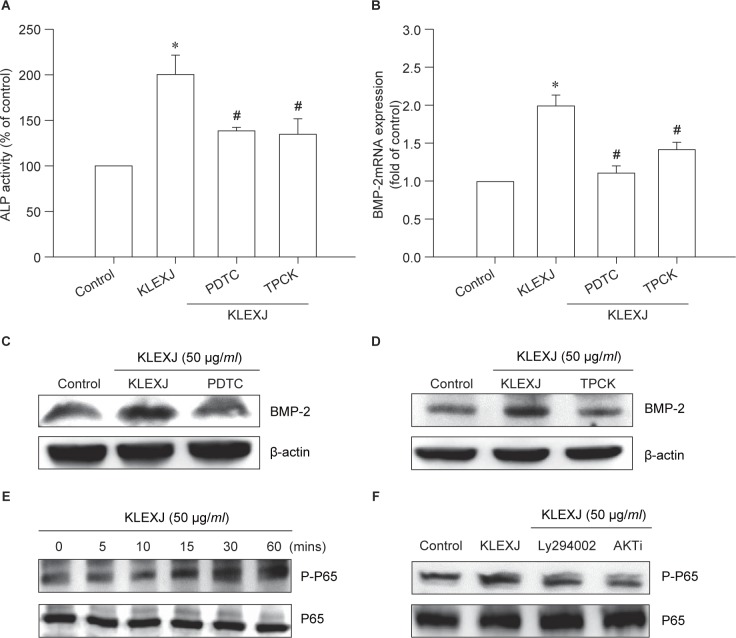
NF-κB activation has been indicated to regulate BMP-2 expression and is implicated in bone formation [27, 28]. We assessed whether NF-κB activation is mediated via KLEXJ increasing BMP-2 production. Pretreatment with NF-κB inhibitors (TPCK and PDTC) reversed KLEXJ-induced ALP activity and increases in BMP-2 expression (Fig. 4A-D). Furthermore, KLEXJ increased phosphorylation of p65 in a time-dependent manner (Fig. 4E). In contrast, KLEXJ-induced activation of p65 was antagonized by pretreatment with Ly294002 and an Akt inhibitor (Fig. 4F). Hence, NF-κB is the downstream molecule of PI3K/Akt.
Fig. 4.
KLEXJ promotes ALP activity and BMP-2 expression through the NF-κB pathway. (A) Osteoblasts were pretreated with TPCK (10 μM) or PDTC (10 μM) for 30 min, followed by stimulation with KLEXJ for 48 h. ALP activity was examined using a commercial ALP assay kit. (B-D) Osteoblasts were pretreated with TPCK (10 μM) or PDTC (10 μM) for 30 min, followed by stimulation with KLEXJ for 24 h and BMP-2 expression was examined by qPCR and Western blot analysis. (E) Osteoblasts were incubated with KLEXJ for indicated time intervals and p65 phosphorylation was examined by Western blot analysis. (F) Osteoblasts were pretreated with Ly294002 or an Akt inhibitor, followed by stimulation with KLEXJ. p65 phosphorylation was examined by Western blot analysis. Results are expressed as mean ± S.E.M. *, P < 0. 05 compared with control. #, p < 0.05 compared with KLEXJ-treated group.
4. Discussion
Kuei-Lu-Er-Xian-Jiao (KLEXJ), a TCM formula, is widely used in traditional medicine for osteoporosis treatment and has been reported to reduce osteoarthritis progress [16]. However, the detailed effects of KLEXJ in bone cells are unclear. To the best of our knowledge, this study is the first analysis of the role played by KLEXJ extract in osteoblasts. Our results demonstrate that
KLEXJ extract induces ALP activity (an important osteoblastic differentiation marker). In addition, we suggest that BMP-2 acts as a target molecule of KLEXJ-induced signaling that requires the PI3K/Akt/NF-κB pathway.
The detailed molecular mechanisms of osteoporosis remain unknown, but they are believed to correlate with decreased availability or activity of bone growth factors: e.g., BMPs that play key roles in bone remodeling and formation [29, 30]. The present study found that KLEXJ extract increases production of BMP-2. In contrast, incubation of osteoblasts with KLEXJ increased osteoblastic differentiation marker expression (ALP activity). These results imply that KLEXJ promotes bone formation by up-regulating expression of BMP-2.
PI3K activation is a potential signaling pathway that regulates bone formation [31, 32]. Here, we report that both a PI3K inhibitor and a siRNA antagonized KLEXJ-promoted activity of ALP and production of BMP-2. Incubation of osteoblasts with KLEXJ increased phosphorylation of PI3K, suggesting that PI3K activation plays a crucial role in KLEXJ-increased bone differentiation and BMP-2 expression.
Akt activation is reportedly mediates ALP activity during osteoblastic cell differentiation [33, 34]. In this study, we found that KLEXJ promotes phosphorylation of Akt, while an Akt inhibitor or siRNA diminishes KLEXJ-induced potentiation of ALP activity and BMP-2 production in osteoblasts, which suggests that Akt activation plays a critical role in KLEXJ-promoted osteoblastic function.
The BMP-2 5’ promoter region contains the ERE, AP-1 and Sp1 binding sites, which regulate BMP-2 expression [35]. In this study, we found that the NF-κB inhibitors PDTC and TPCK antagonized KLEXJ-induced increases in ALP activity and BMP-2 expression in osteoblasts. In addition, treatment with KLEXJ enhanced p65 NF-κB phosphorylation, suggesting that NF-κB activation plays a critical role in KLEXJ-promoted bone formation and BMP-2 production. Further evidence that activation of NF-κB plays a key role in TCM-mediated BMP-2 production is shown by the TCM formula Si-Wu-Tang, which also increases BMP-2 expression through NF-kB activation [14]; moreover, the Chinese herbal medicine Cistanche deserticola extract up-regulates BMP-2, which involves the NF-kB pathway [23]. In this study, we found that PI3K and Akt inhibitors diminished KLEXJ-induced increases in p65 phosphorylation. Therefore, the PI3K/ Akt pathway is mediated by KLEXJ-promoted NF-κB activation.
5. Conclusions
Drugs that are synthesised from natural products have a key role in pharmaceutical care [36–38] and have proven to be critical sources of potential products for osteoporosis treatment [39, 40]. We have shown that KLEXJ boosts ALP activity and BMP-2 production in osteoblasts via the PI3K/Akt-NF-κB pathway and hence may be suitable in the treatment of osteoporosis.
Conflict of interest
None of the authors have any financial or personal relationships with other people or organizations that could inappropriately influence this work.
Acknowledgments
This study was supported by grants from the Ministry of Science and Technology of Taiwan (MOST-103-2320-B-029-002 and 104-2320-B-029-002) and China Medical University Hospital (DMR-105-009).
References
- 1. Manolagas SC, Jilka RL. Bone marrow, cytokines, and bone remodeling Emerging insights into the pathophysiology of osteoporosis. N Engl J Med. 1995; 332: 305–11. [DOI] [PubMed] [Google Scholar]
- 2. Chen PC, Cheng HC, Yang SF, Lin CW, Tang CH. The CCN family proteins: modulators of bone development and novel targets in bone-associated tumors. Biomed Res Int. 2014; 2014: 437096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Goltzman D. Discoveries, drugs and skeletal disorders. Nat Rev Drug Discov. 2002; 1: 784–96. [DOI] [PubMed] [Google Scholar]
- 4. Wong SK, Chin KY, Suhaimi FH, Ahmad F, Ima-Nirwana S. The Relationship between Metabolic Syndrome and Osteoporosis: A Review. Nutrients. 2016; 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Ducy P, Schinke T, Karsenty G. The osteoblast: a sophisticated fibroblast under central surveillance. Science. 2000; 289: 1501–4. [DOI] [PubMed] [Google Scholar]
- 6. Lane NE, Kelman A. A review of anabolic therapies for osteoporosis. Arthritis Res Ther. 2003; 5: 214–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Wu JB, Fong YC, Tsai HY, Chen YF, Tsuzuki M, Tang CH. Naringin-induced bone morphogenetic protein-2 expression via PI3K, Akt, c-Fos/c-Jun and AP-1 pathway in osteoblasts. Eur J Pharmacol. 2008; 588: 333–41. [DOI] [PubMed] [Google Scholar]
- 8. Fox J. Developments in parathyroid hormone and related peptides as bone-formation agents. Curr Opin Pharmacol. 2002; 2: 338–44. [DOI] [PubMed] [Google Scholar]
- 9. Lindsay R, Krege JH, Marin F, Jin L, Stepan JJ. Teriparatide for osteoporosis: importance of the full course. Osteoporos Int. 2016; 27: 2395–410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Rider CC, Mulloy B. Bone morphogenetic protein and growth differentiation factor cytokine families and their protein antagonists. Biochem J. 2010; 429: 1–12. [DOI] [PubMed] [Google Scholar]
- 11. Thatcher JD. The TGF-beta signal transduction pathway. Sci Signal. 2010; 3: tr4. [DOI] [PubMed] [Google Scholar]
- 12. Hong CC, Yu PB. Applications of small molecule BMP inhibitors in physiology and disease. Cytokine Growth Factor Rev. 2009; 20: 409–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Styrkarsdottir U, Cazier JB, Kong A, Rolfsson O, Larsen H, Bjarnadottir E, et al Linkage of osteoporosis to chromosome 20p12 and association to BMP2. PLoS Biol. 2003; 1: E69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Wu CM, Chen PC, Li TM, Fong YC, Tang CH. Si-Wu-tang extract stimulates bone formation through PI3K/Akt/NF-kappaB signaling pathways in osteoblasts. BMC Complement Altern Med. 2013; 13: 277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Li TM, Huang HC, Su CM, Ho TY, Wu CM, Chen WC, et al Cistanche deserticola extract increases bone formation in osteoblasts. J Pharm Pharmacol. 2012; 64: 897–907. [DOI] [PubMed] [Google Scholar]
- 16. Tsai CC, Chou YY, Chen YM, Tang YJ, Ho HC, Chen DY. Effect of the herbal drug guilu erxian jiao on muscle strength, articular pain, and disability in elderly men with knee osteoarthritis. Evid Based Complement Alternat Med. 2014; 2014: 297458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Lee SC, Chang SJ, Tsai LY. Effects of traditional Chinese medicines on serum lipid profiles and homocysteine in the ovariectomized rats. Am J Chin Med. 2004; 32: 541–50. [DOI] [PubMed] [Google Scholar]
- 18. Tang CH, Hsu CJ, Fong YC. The CCL5/CCR5 axis promotes interleukin-6 production in human synovial fibroblasts. Arthritis Rheum. 2010; 62: 3615–24. [DOI] [PubMed] [Google Scholar]
- 19. Chien SY, Huang CY, Tsai CH, Wang SW, Lin YM, Tang CH. Interleukin-1beta induces fibroblast growth factor 2 expression and subsequently promotes endothelial progenitor cell angiogenesis in chondrocytes. Clin Sci (Lond). 2016; 130: 667–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Yang WH, Chang AC, Wang SW, Wang SJ, Chang YS, Chang TM, et al Leptin promotes VEGF-C production and induces lymphangiogenesis by suppressing miR-27b in human chondrosarcoma cells. Sci Rep. 2016; 6: 28647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Huang CY, Chang AC, Chen HT, Wang SW, Lo YS, Tang CH. Adiponectin promotes VEGF-C-dependent lymphangiogenesis by inhibiting miR-27b through a CaMKII/AMPK/p38 signaling pathway in human chondrosarcoma cells. Clin Sci (Lond). 2016; 130: 1523–33. [DOI] [PubMed] [Google Scholar]
- 22. Tang CH, Hsu TL, Lin WW, Lai MZ, Yang RS, Hsieh SL, et al Attenuation of bone mass and increase of osteoclast formation in decoy receptor 3 transgenic mice. J Biol Chem. 2007; 282: 2346–54. [DOI] [PubMed] [Google Scholar]
- 23. Tang CH, Yang RS, Chien MY, Chen CC, Fu WM. Enhancement of bone morphogenetic protein-2 expression and bone formation by coumarin derivatives via p38 and ERK-dependent pathway in osteoblasts. Eur J Pharmacol. 2008; 579: 40–9. [DOI] [PubMed] [Google Scholar]
- 24. Li H, Li T, Fan J, Fan L, Wang S, Weng X, et al miR-216a rescues dexamethasone suppression of osteogenesis, promotes osteoblast differentiation and enhances bone formation, by regulating c-Cbl-mediated PI3K/AKT pathway. Cell Death Differ. 2015; 22: 193545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Su CM, Lee WL, Hsu CJ, Lu TT, Wang LH, Xu GH, et al Adiponectin Induces Oncostatin M Expression in Osteoblasts through the PI3K/Akt Signaling Pathway. Int J Mol Sci. 2016; 17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Chen HT, Tsou HK, Chang CH, Tang CH. Hepatocyte growth factor increases osteopontin expression in human osteoblasts through PI3K, Akt, c-Src, and AP-1 signaling pathway. PLoS One. 2012; 7: e38378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Tan TW, Huang YL, Chang JT, Lin JJ, Fong YC, Kuo CC, et al CCN3 increases BMP-4 expression and bone mineralization in osteoblasts. J Cell Physiol. 2012; 227: 2531–41. [DOI] [PubMed] [Google Scholar]
- 28. Kageyama A, Matsui H, Ohta M, Sambuichi K, Kawano H, Notsu T, et al Palmitic acid induces osteoblastic differentiation in vascular smooth muscle cells through ACSL3 and NF-kappaB, novel targets of eicosapentaenoic acid. PLoS One. 2013; 8: e68197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wang J, Guo J, Liu J, Wei L, Wu G. BMP-Functionalised Coatings to Promote Osteogenesis for Orthopaedic Implants. Int J Mol Sci. 2014; 15: 10150–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Bandyopadhyay A, Yadav PS, Prashar P. BMP signaling in development and diseases: a pharmacological perspective. Biochem Pharmacol. 2013; 85: 857–64. [DOI] [PubMed] [Google Scholar]
- 31. Chen CY, Su CM, Huang YL, Tsai CH, Fuh LJ, Tang CH. CCN1 induces oncostatin M production in osteoblasts via integrin-dependent signal pathways. PLoS One. 2014; 9: e106632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Pramojanee SN, Phimphilai M, Chattipakorn N, Chattipakorn SC. Possible roles of insulin signaling in osteoblasts. Endocr Res. 2014; 39: 144–51. [DOI] [PubMed] [Google Scholar]
- 33. Lai CH, Wu YW, Yeh SD, Lin YH, Tsai YH. Effects of 6-Hydroxy-flavone on Osteoblast Differentiation in MC3T3–E1 Cells. Evid Based Complement Alternat Med. 2014; 2014: 924560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Marie PJ. Signaling pathways affecting skeletal health. Curr Osteoporos Rep. 2012; 10: 190–8. [DOI] [PubMed] [Google Scholar]
- 35. Hou CH, Hou SM, Tang CH. Ultrasound increased BMP-2 expression via PI3K, Akt, c-Fos/c-Jun, and AP-1 pathways in cultured osteoblasts. J Cell Biochem. 2009; 106: 7–15. [DOI] [PubMed] [Google Scholar]
- 36. Yin MC. Development of natural antitumor agents. BioMedicine. 2013; 3: 105. [Google Scholar]
- 37. Su KP. Nutrition, psychoneuroimmunology and depression: the therapeutic implications of omega-3 fatty acids in interferon-alpha-induced depression. BioMedicine. 2015; 5: 21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Yao HT, Yang YC, Chang CH, Yang HT, Yin MC. Protective effects of (-)-epigallocatechin-3-gallate against acetaminophen-induced liver injury in rats). BioMedicine. 2015; 5: 15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Zhai YK, Guo X, Pan YL, Niu YB, Li CR, Wu XL, et al A systematic review of the efficacy and pharmacological profile of Herba Epimedii in osteoporosis therapy. Pharmazie. 2013; 68: 713–22. [PubMed] [Google Scholar]
- 40. Keiler AM, Zierau O, Kretzschmar G. Hop extracts and hop substances in treatment of menopausal complaints. Planta Med. 2013; 79: 576–9 [DOI] [PubMed] [Google Scholar]