ABSTRACT
Increasing the resistance of plant-fermenting bacteria to lignocellulosic inhibitors is useful to understand microbial adaptation and to develop candidate strains for consolidated bioprocessing. Here, we study and improve inhibitor resistance in Clostridium phytofermentans (also called Lachnoclostridium phytofermentans), a model anaerobe that ferments lignocellulosic biomass. We survey the resistance of this bacterium to a panel of biomass inhibitors and then evolve strains that grow in increasing concentrations of the lignin phenolic, ferulic acid, by automated, long-term growth selection in an anaerobic GM3 automat. Ultimately, strains resist multiple inhibitors and grow robustly at the solubility limit of ferulate while retaining the ability to ferment cellulose. We analyze genome-wide transcription patterns during ferulate stress and genomic variants that arose along the ferulate growth selection, revealing how cells adapt to inhibitors through changes in gene dosage and regulation, membrane fatty acid structure, and the surface layer. Collectively, this study demonstrates an automated framework for in vivo directed evolution of anaerobes and gives insight into the genetic mechanisms by which bacteria survive exposure to chemical inhibitors.
IMPORTANCE Fermentation of plant biomass is a key part of carbon cycling in diverse ecosystems. Further, industrial biomass fermentation may provide a renewable alternative to fossil fuels. Plants are primarily composed of lignocellulose, a matrix of polysaccharides and polyphenolic lignin. Thus, when microorganisms degrade lignocellulose to access sugars, they also release phenolic and acidic inhibitors. Here, we study how the plant-fermenting bacterium Clostridium phytofermentans resists plant inhibitors using the lignin phenolic, ferulic acid. We examine how the cell responds to abrupt ferulate stress by measuring changes in gene expression. We evolve increasingly resistant strains by automated, long-term cultivation at progressively higher ferulate concentrations and sequence their genomes to identify mutations associated with acquired ferulate resistance. Our study develops an inhibitor-resistant bacterium that ferments cellulose and provides insights into genomic evolution to resist chemical inhibitors.
KEYWORDS: clostridia, evolution, genomics
INTRODUCTION
Fermentation of lignocellulosic biomass by bacteria like Clostridium phytofermentans is central to the function of soil, aquatic, and intestinal microbiomes. In addition, industrial fermentation of lignocellulosic biomass into fuels and chemicals could contribute significantly to global energy needs without impacting food production (1). Plant biomass is primarily composed of a macromolecular network of polysaccharides linked with lignin, a polymer of phenylpropanoid subunits with aromatic rings of various degrees of methoxylation (2). Thus, when microorganisms hydrolyze lignocellulose to access sugars, they also liberate the following three main types of inhibitors: aliphatic acids, furans, and solubilized phenolics. The relative amounts of inhibitors depend on the species and condition of the plant matter (3), but hydrolysates generally contain inhibitors at concentrations that impede the growth of microorganisms (4) by damaging the cell membrane, metabolic enzymes, and nucleic acids (5). The most abundant aliphatic acids are generally acetate, particularly in acetylxylan-rich hardwoods (6), and formate from furan breakdown. The main furans are furfural and hydroxymethylfurfural (5-HMF), which are formed by the dehydration of pentose and hexose sugars, respectively. The most potent inhibitors released during biomass hydrolysis are generally phenolics released from lignin (7).
The resistance of model, sugar-fermenting bacteria, such as Escherichia coli, to biomass inhibitors has been well studied for aliphatic acids (8, 9), furans (7), and phenolics (10). However, much less is known about resistance in bacteria like C. phytofermentans that hydrolyze and ferment lignocellulose, even though plant inhibitors are important to the ecology of these species. Moreover, development of inhibitor-resistant microorganisms that directly metabolize biomass is needed for consolidated bioprocessing in a single reactor, which is generally regarded as the most economical configuration for microbial transformation of biomass into value-added chemicals (11).
Here, we study and increase resistance to plant-derived inhibitors in C. phytofermentans, an anaerobic bacterium in Clostridium cluster XIVa that expresses dozens of carbohydrate-active enzymes to degrade lignocellulosic biomass into hexoses and pentoses, which it then ferments to ethanol, H2, and acetate (12). We initially define the effects of a panel of biomass inhibitors, including phenolics, furans, and aliphatic acids, on C. phytofermentans growth. Among these compounds, we focus on ferulic acid, a guaiacyl lignin precursor that is one of the most abundant phenolic inhibitors in woods, grasses, and agriculturally important crops (13). We examine the transcriptional response to ferulate stress by quantifying genome-wide mRNA expression changes. We apply long-term, anaerobic growth selection in a GM3 device (14) to isolate a series of increasingly ferulate-resistant strains. We examined the phenotypes of clones from along the selection and sequenced their genomes to identify positively selected genomic point mutations, small insertions and deletions (indels), and large structural rearrangements. Finally, we discuss how these results improve our understanding of the genetic basis of how bacteria evolve to resist chemical inhibitors.
RESULTS
Native C. phytofermentans inhibitor resistance.
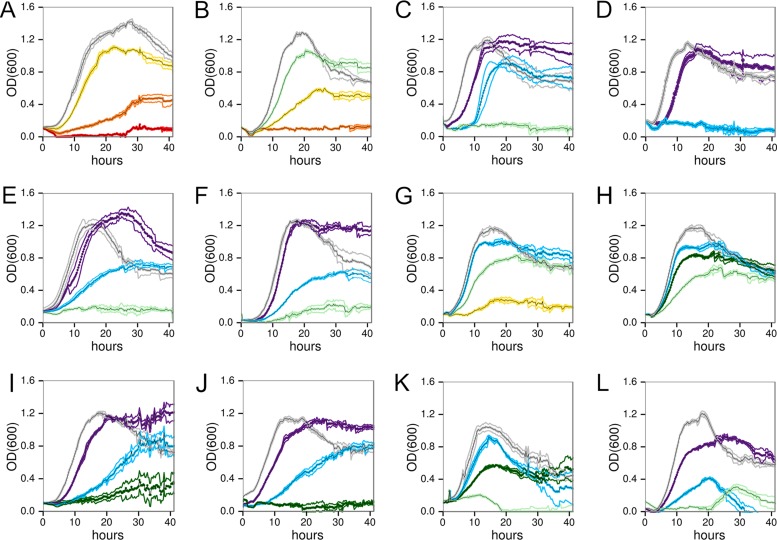
We measured the growth of C. phytofermentans in various concentrations of 12 lignocellulosic inhibitors (see Table S1 in the supplemental material) to gain a general understanding of the relative effects of aliphatic acids, furans, and phenolics (Fig. 1; see also Fig. S1 in the supplemental material). Both aliphatic acids reduce growth; acetate (Fig. 1A) was less toxic than formate (Fig. 1B) on a mass per volume basis, but both acids had similar effects in terms of molarity (Fig. S1). At low furan concentrations, we observed normal growth rates after an extended lag phase (Fig. 1C and D), similar to other bacteria that reduce and detoxify furans (15, 16). Growth lags are proposed to be due to alcohol dehydrogenase (ADH) reducing furan, causing NADH depletion and acetaldehyde accumulation (17). Additionally, the ADH protein Cphy1179 shares 28% amino acid identity with a furfural-reducing, Zn-dependent ADH (18). However, if C. phytofermentans detoxifies furans, this mechanism is abruptly overwhelmed at concentrations above 2 g liter−1 5-HMF and 1 g liter−1 furfural.
FIG 1.
C. phytofermentans growth in GS2 glucose medium containing different concentrations of acetate (A), formate (B), 5-hydroxymethylfurfural (C), furfural (D), coumarate (E), ferulate (F), vanillate (G), 4-hydroxybenzoic acid (H), vanillin (I), benzaldehyde (J), syringaldehyde (K), and catechol (L). Colors show inhibitor concentrations (g liter−1): gray (0), purple (1), blue (2), dark green (3), light green (5), yellow (10), orange (20), and red (30). Data show mean cell density (OD600) of 4 cultures ± standard deviation (SD).
We examined the toxicities of two types of phenolic acids: hydrocinnamic acids (p-coumarate and ferulate) and hydroxybenzoic acids (vanillate and 4-hydroxybenzoic acid). We found that hydrocinnamic acids (Fig. 1E and F) are more toxic than hydroxybenzoic acids (Fig. 1G and H), which supports that the propionic group on the benzene ring in hydrocinnamic acids enhances toxicity, likely by affecting how the molecules partition into the membrane. Moreover, we found that phenolic acids are typically less toxic than the corresponding aldehydes (Fig. 1I to K) and catechol (Fig. 1L). For example, vanillate (Fig. 1G) is much less toxic than vanillin (Fig. 1I), and 4-hydroxybenzoic acid (Fig. 1H) is similarly less toxic than benzaldehyde (Fig. 1J). The enhanced toxicity of aldehydes is likely due to their reactivity, resulting in formation of adducts with nucleophilic sites on DNA, proteins, and other macromolecules (19).
Genome-wide mRNA expression during ferulate stress.
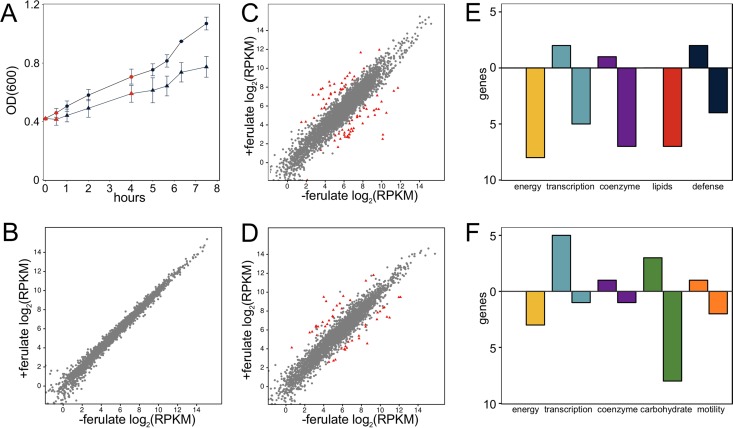
We quantified genome-wide mRNA expression changes at two time (t) points (t = 0.5 h and t = 4 h) following supplementation of mid-log cultures with 2 g liter−1 ferulate, which reduced growth (Fig. 2A) similar to that of the initial growth screen (Fig. 1F). Three to five million read pairs were aligned to the genome for each culture (see Table S3A in the supplemental material) to calculate gene expression levels (Table S3B). The number of differentially expressed genes (Table S3C to E) increased from 0 genes before ferulate addition (Fig. 2B) to 78 genes after 30 min (Fig. 2C) and then declined to 47 genes after 4 h (Fig. 2D). The most abundant functional categories of differentially expressed genes at a t of 0.5 h relate to the repression of energy production, coenzyme metabolism, and lipids (Fig. 2E). The coenzyme-associated genes enable siroheme biosynthesis, which is repressed in clostridia in response to redox stress (20). Lipid genes include the fab gene cluster (cphy0516-cphy0523) for fatty acid biosynthesis, which was strongly repressed at a t of 0.5 h (Fig. 2E) and recovered by a t of 4 h. While cultures continued active growth after sampling, many of the differences between cultures with or without ferulate at a t of 4 h indicate that the treatment without ferulate had depleted nutrients in the medium, triggering expression of genes to assimilate alternative carbohydrates (Fig. 2F).
FIG 2.
C. phytofermentans growth and gene expression during ferulate stress. (A) Growth in log-phase cultures in medium either lacking ferulate (−ferulate, circles) or containing 2 g liter−1 ferulate (+ferulate, triangles). Points are mean cell density (OD600) of duplicate cultures ± SD with red points showing times sampled for RNA-seq: t = 0 h (immediately before dilution), t = 0.5 h, and t = 4 h. (B to D) mRNA expression from cultures at t = 0 h (B), t = 0.5 h (C), and t = 4 h (D). Differentially expressed genes are identified by red triangles; unchanged genes are gray circles. (E and F) Five most abundant COG functional categories (64) of differentially expressed genes at t = 0.5 h (E) and t = 4 h (F). Positive y axis is upregulated genes and negative y axis is repressed genes.
Gene expression at a t of 0.5 h shows that abrupt ferulate stress induces expression of genes encoding the efflux pump cphy1055-cphy1056, which is similar to E. coli mdlAB conferring resistance to organic solvents (21). Many of the genes upregulated at a t of 0.5 h are colocated in two genomic regions. The first region encodes tad (tight adherence) cphy0029-cphy0040 genes for Flp-type type IV pilus assembly. Type IV pili are widespread in clostridia (22) for adhesion to solid substrates to form protective biofilms (23), reflecting how ferulate represses motility genes in Clostridium beijerinckii (24). The other cluster cphy1838-cphy1845 includes genes for the flavin mononucleotide (FMN)-binding protein WrbA (25) and two NADPH:FMN reductases. NADPH:FMN reductase inactivation confers ferulate resistance in C. beijerinckii by an unknown mechanism (26). While this appears to be in opposition to our data showing that NADPH:FMN reductases are upregulated by ferulate, both results support the importance of FMN-mediated oxidoreduction in ferulate resistance. This island also includes genes encoding an acetyltransferase and Cphy1845 that shares 41% amino acid identity and metal coordination with E. coli YhhW, which cleaves the plant phenolic quercetin (27). C. phytofermentans may thus upregulate genes to transform or detoxify plant phenolics, similar to some ruminal clostridia (28).
Selection and physiology of ferulate-resistant strains.
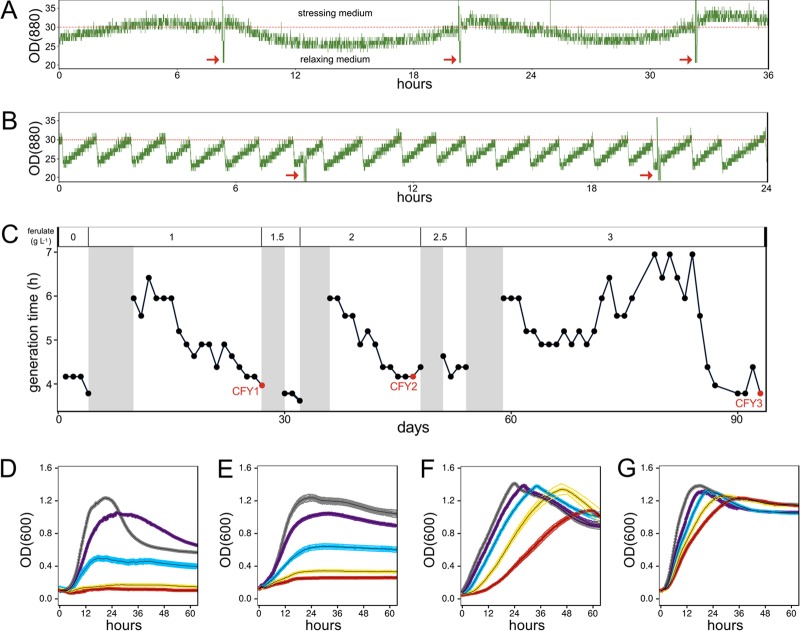
We selected C. phytofermentans strains with increased ferulate resistance by cultivation in a GM3 automat, a dual-chamber, continuous-culture device that automates delivery of fresh medium and transfers the evolving cell suspension between twin growth chambers to prevent biofilm formation. During acclimation to increased ferulate in medium-swap mode, cell densities oscillated for 2 to 5 days because high densities triggered pulses of stressing medium (high ferulate) that reduced culture density, which in turn resulted in delivery of relaxing medium (low ferulate) that enabled recovery (Fig. 3A). As such, the ferulate-based selection in medium-swap mode is modulated by the ratio of relaxing and stressing medium. Once cell densities stabilized in the stressing medium, the growth rate at the higher ferulate concentration was improved in turbidostat mode (Fig. 3B). We initiated the growth selection with a stressing medium containing 1 g liter−1 ferulate, the highest concentration at which we could establish a stable wild-type (WT) culture in the GM3. After 93 days (∼500 generations) of continuous, log-phase growth selection with incrementally higher ferulate, the culture grew with the same 3.75-h generation time in the 3 g liter−1 ferulate medium as that of the WT in the absence of ferulate (Fig. 3C). Clones isolated along the growth selection are progressively more ferulate resistant in batch culture (Fig. 3D to G); while no growth was observed above 2 g liter−1 ferulate in the WT strain (Fig. 3D), CFY3 clones grow robustly at the ferulate solubility limit (6 g liter−1) (Fig. 3G). We assessed the ferulate resistance of 2 clones from each of the CFY1 (CFY1A and CFY1B) and CFY2 (CFY2C and CFY2D) time points and 4 clones from the CFY3 time point (CFY3E to CFY3H). The duplicate CFY1 and CFY2 clones showed similar ferulate resistance, but CFY3H is much less ferulate resistant than the 3 other clones (see Fig. S2 in the supplemental material), showing that cells in the GM3 culture are heterogeneous with respect to ferulate resistance.
FIG 3.
Growth improvement of C. phytofermentans GM3 strains in ferulate medium. (A) Cells were acclimated to increased ferulate using medium-swap mode, a chemostat with dilutions of stressing medium if density exceeds a threshold and with relaxing medium otherwise. (B) Growth rate was improved using turbidostat mode in which the culture was diluted each time it reached the threshold. (A and B) Dashed red lines show cell density threshold (OD880 of 30), and red arrows show when the growth chamber was sterilized. (C) C. phytofermentans growth rate over 93 day GM3 experiment in medium with increasing ferulate concentrations (shown above plot). Shaded areas are periods of medium-swap with fixed 6-h generation time. Black lines show average daily generation time (h) during turbidostat growth selection. Red points are sample times for physiology and genome sequences (CFY1, CFY2, CFY3). (D to G) Batch culture growth (OD600) of wild-type (D) and clones CFY1A (E), CFY2C (F), and CFY3E (G) in GS2 glucose medium containing either 0 (gray), 1 (purple), 2 (blue), 4 (yellow), or 6 (red) g liter−1 ferulate. Data show mean cell density (OD600) of 4 cultures ± SD.
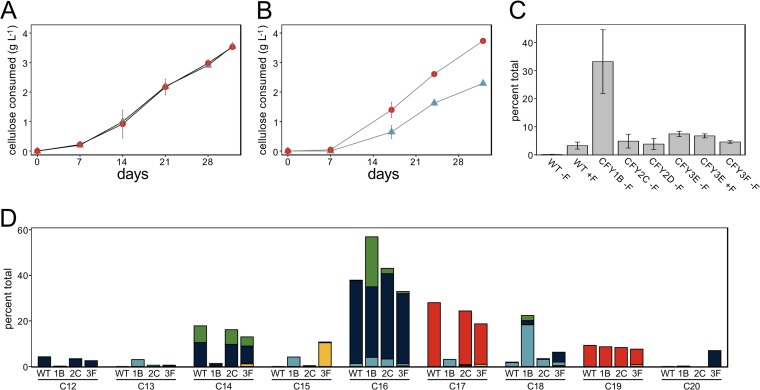
We examined whether selection for ferulate resistance in glucose medium resulted in physiological changes impacting cellulose fermentation and resistance to other inhibitors. CFY3 strains degrade cellulose similar to that in the WT (Fig. 4A) and show accelerated cellulose degradation in medium supplemented with ferulate (Fig. 4B), supporting that the evolved strains are potentially improved candidates for fermentation of lignocellulose. Moreover, the evolved resistance mechanisms extend to other biomass inhibitors, as CFY3 strains are also more resistant to vanillate and acetate (see Fig. S3 in the supplemental material), albeit with considerable variability between strains. We also used mass spectrometry to investigate if ferulate was consumed or transformed in WT and CFY3 cultures, revealing that the ferulate concentration was unaltered with no products corresponding to reduced, demethoxylated, or decarboxylated ferulate (see Fig. S4 in the supplemental material). Thus, even though C. phytofermentans upregulates potential phenol-degrading enzymes in response to ferulate, the cell adapted to ferulate by reinforcing the cell or excluding this molecule rather than detoxifying it.
FIG 4.
Growth physiology and membrane fatty acid composition of C. phytofermentans WT and GM3 strains. (A and B) Rate of cellulose degradation by CFY3E (red circles) and WT (blue triangles) in medium lacking ferulate (A) and medium supplemented with 2 g liter−1 ferulate (B). (C) Plasmalogen content expressed as percentage of total fatty acids of WT and GM3 strains grown in the presence (+F) or absence (−F) of ferulate. (D) Cellular fatty acid profiles of log-phase WT, CFY1B, CFY2C, and CFY3F cultures in medium without ferulate. Fatty acids are classified by acyl chain length (C12 to C20) and whether acyl chains were saturated (dark blue), unsaturated (light blue), hydroxylated (green), cyclopropane (red), or branched (yellow). (A to C) Data show mean of duplicate cultures ± SD.
As the toxicity of aromatic molecules is often associated with disruption of the cell membrane, we profiled fatty acids (FAs) to determine if ferulate resistance is associated with altered membrane phospholipids (see Table S4 in the supplemental material). We found that when WT was exposed to ferulate, the plasmalogen (vinyl ether phospholipid) content in the membranes increased 18-fold. Moreover, CFY strains retained elevated plasmalogens even in the absence of ferulate (Fig. 4C). In particular, the CFY1B plasmalogen content in the medium without ferulate was 185-fold higher than that of the WT. Related clostridia similarly increase plasmalogens in response to aliphatic alcohol stress (29, 30), likely to fine tune membrane fluidity and protect from redox-mediated damage (31). The distribution of FA chain lengths in WT cells (Fig. 4D) is similar to that of other clostridia but with fewer unsaturated FAs and more cyclopropanes (32), both of which reduce membrane fluidity to protect from solvent stress (33). While the addition of ferulate had little immediate effect on the FA chains of WT cells (see Fig. S5A and B in the supplemental material), the CFY strains showed altered FAs relative to those of the WT in the absence of ferulate (Fig. 4D). The CFY1B FA profile was the most perturbed with increased hydroxylated C16 and unsaturated fatty acids, largely C18:1, which is associated with increased ethanol tolerance in E. coli (34). CFY3F shifted to branched FA (especially C15) and longer chain lengths (C18, C20), which increase membrane rigidity (10) to potentially combat the membrane-fluidizing effects of ferulate. C. phytofermentans fatty acids are decorated with a diversity of phospho, glyco, and amino head groups (Fig. S5C). While we did not detect changes in these head groups in the WT response to ferulate or in the CFY strains, we consider it likely that they participate in the response to solvents, similar to some other bacteria (10).
Genomes of ferulate-resistant isolates.
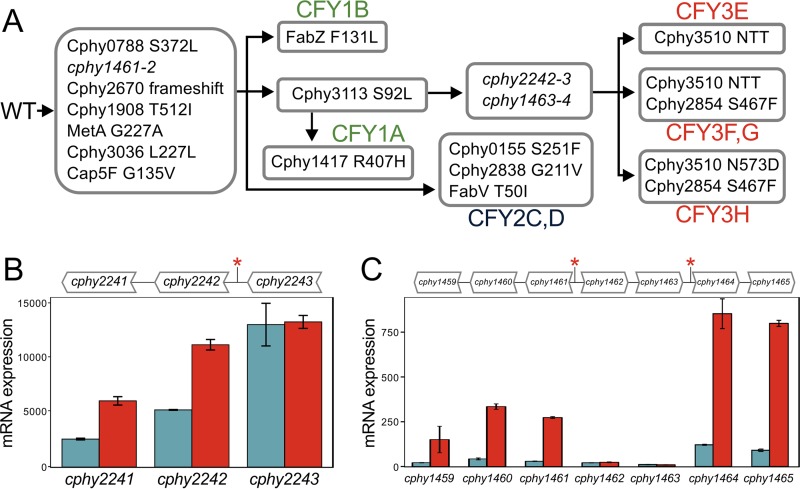
We sequenced the genomes of eight CFY1 to CFY3 clones, giving between 106- and 705-fold coverage (see Table S5A in the supplemental material) to identify DNA variants relative to the wild type (Table S5B). Seven single-nucleotide variations (SNVs) and short insertions/deletions (indels) are present in all of the CFY genomes (Fig. 5A), which likely fixed in the population during an early selective sweep. These variants caused nonsynonymous changes in 5 proteins, including a homolog of Cap5F (Cphy3503), a protein for biosynthesis of capsular polysaccharides (35) that is associated with biofilm formation (36) and stress resistance (37). Strains subsequently accrued strain-specific mutations consistent with the population exploring alternative mutational pathways to improve ferulate resistance, particularly by modifying sensor kinases that can transduce signals associated with ferulate stress, fatty acid biosynthesis, and the surface layer (S-layer) (Fig. 5A). For example, the CFY1 and CFY2 strains incurred coding variants in 3 genes putatively encoding fatty acid biosynthesis proteins: Cphy3113 for anaerobic synthesis of unbranched fatty acids (38), the fatty acid dehydratase FabZ (Cphy0520), and the reductase FabV (Cphy1286) for the final step in fatty acid elongation. The genomes of CFY3E to CFY3G (high resistance) and CFY3H (low resistance) differ by variants in Cphy3510, the most highly expressed protein in the proteome that is proposed to form the S-layer (39). The S-layer is a protein lattice that provides mechanical stabilization, sites for extracellular protein attachment, and a selective barrier for molecules (40).
FIG 5.
Small-scale genome differences in C. phytofermentans GM3 strains. (A) Accumulation of single-nucleotide variants and small indels in the genomes of clones isolated from the CFY1 to CFY3 time points. (B and C) mRNA expression of the cphy2241-cphy2243 operon (B) and the cphy1459-cphy1465 genes (C) in WT (blue) and CFY3E (red) strains. Genes are shown above plots with asterisks denoting positions of DNA changes. Expression was measured by qRT-PCR and quantified as 2−ΔCT normalized to 16S rRNA expression; bars show means of triplicate measurements ± SD.
Intergenic changes that arose in the CFY genomes affect the expression levels of adjacent genes. For example, a 15-bp sequence between the first two genes of the ABC glucose transporter operon (cphy2241-cphy2243) was duplicated in the CFY3 strains (see Fig. S6A and B in the supplemental material). The repeated sequence forms an inverted repeat (IR) similar to repeated extragenic palindrome (REP) sequences, a widespread mechanism in bacteria to tune gene expression by modulating the stability of different mRNA segments within an operon (41). Duplication of this putative REP increases the mRNA secondary structure of the cphy2243-cphy2242 intergenic region (Fig. S6B), supporting functions similar to those of REP that increase expression by forming stable stem-loop structures that protect mRNA from ribonucleases (42, 43). Similarly, we found that mRNA expression of the two genes downstream of the insertion was elevated (Fig. 5B), which may have increased fitness because the GM3 growth selections were done in glucose medium. The mRNA expression of genes in two colocated operons with upstream point mutations was upregulated in the CFY strains (Fig. 5C). The A-to-G transition upstream of cphy1464 created a TG dinucleotide 2 bp upstream of the Pribnow hexamer (Fig. S6A) that enhances transcription in other bacteria (44, 45) and is present in the consensus −10 promoter sequence in C. phytofermentans (46). We propose that the upregulated operons cphy1459-cphy1461 and cphy1464-cphy1465 either enable increased production of malonyl coenzyme A (malonyl-CoA) for fatty acid biosynthesis or neutralize intracellular pH in response to ferulic acid stress through production of ammonium and lactate (7) and bicarbonate buffering (Fig. S6C).
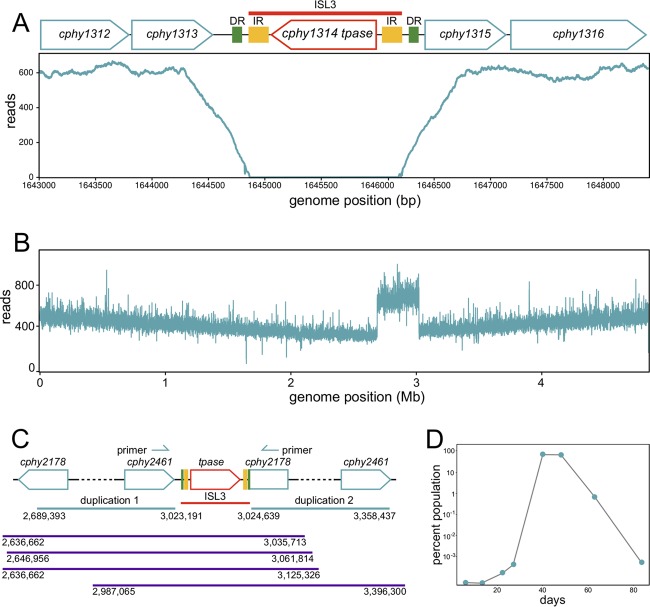
Adaptive function can be imparted by structural changes to the genome resulting from recombination and transposition of insertion sequences (IS elements). C. phytofermentans encodes 31 IS elements (see Table S6 in the supplemental material), including 12 ISL3 comprised of 2 isoforms—8 ISL3-1 elements and 4 ISL3-2 elements. IS elements inactivate genes through their transposition and act as the substrates for homologous recombination. In addition, the IRs of all 12 ISL3 contain a 5′-TTGACA-3′ sequence matching an outward-facing, consensus −35 box from this organism (46) (Table S6), suggesting that ISL3 could activate expression of adjacent genes (47). An ISL3-1 was precisely deleted in all CFY genomes as evidenced by reduced read coverage (Fig. 6A) as well as BioNano optical mapping and Sanger sequencing (see Fig. S7A and B in the supplemental material), showing that ISL3 are active in C. phytofermentans. Further, the CFY2C and CFY2D strains share a 333-kb duplication from cphy2178 to cphy2461 (276 genes) (Fig. 6B), which we showed by PCR exists as a tandem duplication joined by a novel ISL3-2 insertion (Fig. 6C; Fig. S7C). We did not observe any extrachromosomal DNA by pulsed-field gel in the CFY2C strain, supporting that the cphy2178-ISL3-cphy2461 fragment did not excise as a circular molecule. Further, BioNano sequencing of DNA molecules greater than 400 kb spanning the junctions of the duplicated region localizes the rearrangement as a genomic, tandem duplication (Fig. 6C). We quantified the relative abundance of cells bearing this duplication by quantitative PCR of the cphy2178-ISL3-cphy2461 fragment (Fig. 6D). The duplication arose between day 13 and 20 and overtook the population to comprise 68% of cells by day 40; supporting it was the subject of positive selection. Subsequently, this variant declined in the population, representing 1% of cells at day 63, as it was gradually replaced by mutants with higher fitness; it was not present in any of the CFY3 genomes.
FIG 6.
ISL3-associated genome changes in GM3 strains. (A) Read coverage showing deletion of the ISL3-1 element including the cphy1314 transposase gene (CFY1A shown). (B) Read coverage in CFY2C and CFY2D (shown) reveals the duplication of a 333-kb region from cphy2178-cphy2461 (genome position 2689393 to 3023191). (C) The cphy2178-cphy2461 region is a tandem duplication joined by an ISL3-2 element. Positions of qPCR primers and single-molecule restriction fragments of >400 kb from BioNano optical mapping that span the duplicated region are shown. (D) Relative abundance of the cphy2178-ISL3-cphy2461 junction in the GM3 culture from day 6 to 83 as measured by qPCR.
DISCUSSION
When plant-fermenting bacteria like C. phytofermentans degrade lignocellulosic biomass to access sugars, they also release various biomass-derived inhibitors, including ferulic acid. The abilities of these bacteria to survive exposures to these inhibitors thus influence both their ecology and industrial potential. Fatty-acid (FA) biosynthesis genes, such as cphy0520 (fabZ) and cphy3113, were associated with both the transcriptional and evolutionary response to ferulate. In particular, the FA profiles were massively perturbed in CFY1B, which has a strain-specific variant in FabZ, a dehydratase for unsaturated fatty acid synthesis. FabZ inactivation in CFY1B is consistent with the accumulation of hydroxylated C16 and decrease of C17 cyclopropanes, which are synthesized from C16:1. Our results also show important differences in the cellular processes implicated in the mRNA response to short-term stress and the DNA changes enabling long-term resistance. Faced with an abrupt increase in ferulate, the cell slows growth and upregulates transcription of genes for efflux pumps, biofilm formation, and flavoproteins, including two NADPH:FMN reductases that are associated with ferulate stress in C. beijerinckii (26). Over longer time periods, natural selection of strains with robust growth in the presence of ferulate resulted in DNA changes associated with metabolism, gene regulation, and the cell surface (S-layer).
In Gram-negative bacteria, the outer membrane protects against influx of toxic compounds. Gram-positive bacteria lack this outer membrane and instead have a thick peptidoglycan wall that cannot exclude solvents. Consequently, Gram-positive bacteria are generally more sensitive to hydrophobic solvents (10). The S-layer is a lattice, often composed of a single protein, that covers many Gram-positive bacteria. It functions as a permeability barrier (40), potentially excluding ferulate, and stabilizes the cell through noncovalent linkage to cell wall polysaccharides using threonine residues (48). A three-residue (asparagine-threonine-threonine) insertion near the C terminus of the S-layer protein Cphy3510 was the sole mutation exclusively present in the three highly resistant CFY3 strains (CFY3E to CFY3G); the CFY3 strain with lower resistance (CFY3H) had an N573D variant in Cphy3510. S-layer proteins differ greatly among bacteria, and neither of the Cphy3510 variants are in known domains; however, our results support that modification of the S-layer may be an effective strategy to improve inhibitor resistance.
In addition to minor genomic changes (SNVs and indels) that alter gene expression or protein activity, both CFY2 genomes contain a tandem duplication of a 333-kb region joined by a novel ISL3-2 insertion, supporting that this large genome rearrangement was positively selected during ferulate selection. Tandem duplications of large chromosomal regions have been detected in other bacteria (41) and can improve fitness by increasing gene dosage. A tandem duplication of regions joined by an IS element in E. coli was proposed to have arisen following insertion of IS elements into each copy of the duplication; when the IS elements recombined with each other, the intervening region was deleted to leave a single, central IS element (49). The duplication observed in CFY2 strains arose early in the experiment, perhaps because stress induced IS element activity. Strains containing the duplication rapidly took over the population, supporting that it enhanced fitness, then gradually declined to represent 1 in 105 cells at the end of the experiment (Fig. 6D), likely because this strain was outcompeted by others with higher fitness.
Our approach uses continuous, directed evolution as a framework for real-time study of natural selection by analyzing the succession of microbial strains with progressively higher fitness. Genome analysis of these strains using both high-coverage short reads and long-range optical mapping reveals both the small and large genomic changes that underlie a complex phenotype. When coupled with transcriptome sequencing (RNA-seq) to study the transcriptional response to abrupt change, this approach gives a portrait of how the cell adapts to a given perturbation on different time scales. These results can be applied to prioritize genes to engineer bacterial stress resistance. For example, abrupt ferulate stress could be mitigated by overexpressing efflux pumps and flavoproteins, whereas long-term ferulate resistance could be improved by altering the primary surface layer protein and membrane biosynthesis (fab genes) to favor longer fatty acids.
MATERIALS AND METHODS
Cell cultivation.
C. phytofermentans ISDg (ATCC 700394) was cultured anaerobically in GS2 medium (50). The growth of batch cultures containing inhibitors (see Table S1 in the supplemental material) was measured in 100-well microtiter plates (Bioscreen 9502550) containing 400 μl of GS2 medium with 3 g liter−1 glucose supplemented with a given inhibitor neutralized to pH 7. Wells were inoculated with 1:10 volume cells grown to log phase in a medium without inhibitor. The plates were sealed in the anaerobic chamber (2% H2, 98% N2) by press-fitting adhesive sheets (Qiagen 1018104) (51) and incubated at 37°C in a Thermo Scientific Bioscreen C. The cell densities (optical density at 600 nm [OD600]) were measured every 15 min with 30 s of shaking before each reading. Cellulose cultures were inoculated into GS2 containing 10 g liter−1 cellulose (0.5- by 5-cm strips of Whatman filter paper 1001-090, >98% cellulose content). Cellulose degradation was measured as the dry mass of cellulose remaining in culture by collecting the remaining cellulose on 11-μm-pore-size filters by vacuum filtration and drying it overnight at 65°C (52).
Ferulate-resistant C. phytofermentans clones were selected using a GM3 automat (14), a dual-chamber continuous-culture device that maintained anaerobic conditions by flushing cultures with 100% N2 gas. A 50-ml culture was maintained at 30°C with optical density readings every 30 s and was transferred between growth chambers every 12 h to clean the empty chamber with 5 N sodium hydroxide. Cells were acclimated to increased ferulate using medium-swap mode, a modified chemostat (6-h generation time) with dilutions every 30 min of stressing medium (high ferulate) if the cell density exceeded the density threshold (measured as an OD880 of 30, which is equivalent to an OD600 of 0.4) and relaxing medium (low ferulate) otherwise. Once cell densities stabilized at a constant cell density in the stressing medium for 24 h, the GM3 was run as a turbidostat using the stressing medium until the culture reattained a 3.75-h generation time, similar to that of the WT strain in medium without ferulate. In turbidostat mode, 20% of the culture volume was replaced with fresh medium each time the cell density reached an OD880 of 30. Initially, the stressing medium contained 1 g liter−1 ferulate, the highest concentration at which a WT culture can be established in the GM3, and the relaxing medium lacked ferulate. The medium-swap/turbidostat approach was iterated by incrementing the stress medium by 0.5 g liter−1 ferulate and replacing the relaxing medium with the previous stressing medium. Samples from the GM3 culture were plated to isolate colonies, called CFY1, CFY2, and CFY3 clones, at the end of the turbidostat selections in 1, 2, and 3 g liter−1 ferulate, respectively.
RNA-seq.
Log-phase cultures (OD600 of 0.8) of WT C. phytofermentans ISDg were diluted with 1 volume medium either lacking ferulate or containing 4 g liter−1 ferulate (2 g liter−1 final concentration). Samples for RNA and cell densities were taken from duplicate cultures for each treatment immediately before ferulate addition and 0.5 and 4 h afterward. Total RNA was extracted using TRI reagent (Sigma 93289), and 20 μg RNA was treated with 4 U Turbo DNase (Ambion AM2238) for 30 min at 37°C. RNA was purified by Zymoclean (Zymo Research R1015) to capture RNAs of >200 bp. Five micrograms of total RNA was depleted of rRNA by Ribo-Zero (Illumina MRZMB126), yielding 200 to 400 ng RNA, and purified by Zymo Concentrator-5 (total capture) into 10 μl of water. cDNA libraries were prepared from 100 ng RNA using the TruSeq stranded mRNA kit (Illumina 15031047) and sequenced on an Illumina HiSeq 2000 sequencer with paired-end 150-bp reads. Reads were aligned to the C. phytofermentans ISDg genome (NCBI accession number NC_010001.1) using Bowtie 2 (53). Gene expression was calculated as reads per kilobase of gene per million (RPKM) using the easyRNASeq bioconductor package (54). Differential expression was defined as a greater than 4-fold change in expression and a DESeq (55) P value of <0.01 after Bonferroni correction for multiple testing of the 3,902 genes in the genome.
Genome sequencing.
Genomes were sequenced for clones isolated from the GM3 samples: CFY1 (2 clones, CFY1A and CFY1B), CFY2 (2 clones, CFY2C and CFY2D), and CFY3 (4 clones, CFY3E to CFY3H). Genomic DNA (15 to 20 μg) was extracted from 3-ml cultures using the Sigma GenElute bacterial genomic DNA kit (NA2110). DNA (100 to 250 ng) was fragmented by Covaris E220 (Covaris, Inc., Woburn, MA, USA) to a 600-bp mean fragment size. The DNA was end-repaired, 3′ A-tailed, and ligated to Illumina compatible adapters using the NEBNext DNA sample prep master mix set 1 (New England BioLabs E6040). Ligation products were purified with 1 volume solid-phase reversible immobilization (SPRI) beads (Beckman Coulter A63880) and amplified by 12 cycles of PCR using the Kapa Hifi HotStart NGS library amplification kit (Kapa Biosystems KK2611) with P5/P7 primers. PCR products were purified (0.8 volume SPRI beads) and run on a 2% agarose gel, and DNA (700 to 800 bp) was excised and purified using the NucleoSpin extract II DNA purification kit (Macherey-Nagel 740609). cDNA libraries were sequenced using 300-bp paired-end reads on an Illumina MiSeq instrument. Reads were quality filtered by Picard (https://github.com/broadinstitute/picard) and aligned to the C. phytofermentans ISDg reference genome (NCBI accession number NC_010001.1) using Bowtie 2 (53). Sequence variants (single nucleotide polymorphisms [SNPs], indels) in the CFY strains relative to the reference genome were identified using GATK (56) as described previously (57). Structural variations were detected using the breseq split-read analysis tool (58). Insertion sequences (IS) were identified using ISfinder (59).
Optical genome mapping.
High-molecular-weight DNA of strain CFY2C was extracted in agar plugs, which were solubilized with 0.4 U of GELase (Epicentre G09200) and dialyzed for 45 min. DNA was treated using IrysPrep reagent kit (BioNano Genomics) to prepare nicked, labeled, repaired, and stained (NLRS) DNA. Briefly, 300 ng of DNA was nicked with 10 U Nt.BspQI (NEB R0644S) for 2 h at 37°C. Nicked DNA was incubated for 1 h at 72°C with fluorescently labeled Alexa 546-dUTP and Taq polymerase (NEB M0273). Nicks were ligated using Taq ligase (NEB M0208) with deoxynucleoside triphosphates (dNTPs). DNA was counterstained with YOYO-1 (Life Technologies). NLRS DNA was loaded into IrysChips (BioNano Genomics), and data were collected on the Irys instrument (BioNano Genomics) until reaching ≥1,000-fold coverage of molecules ≥100 kb.
CFY2C DNA molecules were filtered using BioNano IrysView software (version 2.5.1) retaining molecules ≥100 kb with at least 6 label sites, yielding 32,359 molecules with an N50 of 172 kb. The NCBI assembly (NCBI accession number NC_010001) was in silico digested with BspQI (5′-GCTCTTC-3′) and used to align and assemble CFY2C molecules using the BioNano assembly pipeline (Pipeline version 4618; RefAligner and Assembler version 4704) with the parameters used for small genomes. Molecules ≥400 kb were aligned against the NCBI assembly with a tandem duplication of bp 2689393 to 3023191 joined by an ISL3-2 element in order to identify molecules spanning the duplicated zone.
Quantitative PCR.
We measured mRNA expression by quantitative reverse transcription-PCR (qRT-PCR) as described previously (60). Briefly, RNA was extracted from log-phase WT and CFY3E cultures as for RNA-seq. RNA was reverse transcribed (Applied Biosystems 4368814), and mRNA expression was quantified by quantitative PCR (qPCR) (KAPA KK4621) with primers in Table S2 in the supplemental material. Expression values are means of triplicate measurements of duplicate cultures calculated by the threshold cycle method as 2−ΔCT (61), normalized to 16S rRNA levels and multiplied by a scaling factor of 106. To calculate the relative abundance of DNA variants in the GM3 cultures, genomic DNA was extracted as for genome sequencing from samples taken directly from the GM3 at 8 time points. The abundance of a DNA variant was measured by qPCR (KAPA KK4621) relative to 16S (primers in Table S2). The abundance of the DNA variant in the mixed population was calculated as 2−ΔΔCT relative to a CFY clone that bears the variant in 100% of cells.
Mass spectrometry and chromatography.
Ferulate concentrations were compared in WT and CFY3E cultures after 5 days of growth in GS2 medium containing 6 g liter−1 ferulate by liquid chromatography-electrospray ionization-mass spectrometry (LC-ESI-MS) and tandem mass spectrometry (MS/MS) using a Dionex TCC-3000RS chromatographic system (Thermo Fisher Scientific) coupled to an Orbitrap Elite mass spectrometer (Thermo Electron Corporation) equipped with a heated electrospray ionization (HESI) source. High-pressure liquid chromatography (HPLC) separation was performed on a 5-μm, 4.6- by 150-mm SeQuant ZIC-pHILIC column (Merck) at 40°C with a flow rate of 0.5 ml min−1 and a mobile phase of 10 mM (NH4)2CO3 pH 9.9 (phase A) and acetonitrile (phase B). Elution was conducted using the following gradient conditions: 2 min at 80% phase B, 20-min linear gradient from 80% to 40% of phase B, 8 min at 40% phase B, 5-min increase to 80% phase B, and 15 min of 80% phase B. The mass spectrometer was operated in ESI negative ion mode using a −4.5 kV ion spray, a 275°C capillary temperature, and a mass resolution of 60,000. Sheath gas, auxiliary gas, and sweep gas flow rates were set to 60, 10, and 2 arbitrary units, respectively. Mass spectra were analyzed using Xcalibur version 2.2 (Thermo Fisher Scientific).
Cellular fatty acids were analyzed in WT and CFY3E cultures with and without ferulate and CFY1B, CFY2C, CFY2D, and CFY3F cultures without ferulate. Late log-phase cells were collected by centrifugation from cultures grown in medium either with 2 g liter−1 ferulate or lacking ferulate. Fatty acid methyl esters (FAME) were obtained from 100 mg of cells by saponification, methylation, and extraction (62) and were identified using the DSMZ identification service (Braunschweig, Germany). Briefly, FAME mixtures were separated using the Sherlock microbial identification system (MIS) (Microbial ID, USA): an Agilent model 6890N gas chromatograph with a 5% phenyl-methyl silicone capillary column (0.2 mm by 25 m), a flame ionization detector, and an automatic sampler (Agilent model 7683A). Peaks were integrated and fatty acid names and percentages calculated using Sherlock MIS standard software (Microbial ID, USA). Plasmalogens were quantified as dimethyl acetyl fatty acids. Polar lipids were extracted from 100 mg cells using a chloroform–methanol–0.3% aqueous NaCl mixture (1:2:0.8, vol/vol/vol) by stirring overnight. Cells were centrifuged, and the polar lipids were recovered in the chloroform phase by adjusting the chloroform–methanol–0.3% aqueous NaCl mixture to 1:1:0.9 (vol/vol/vol). Polar lipids were resolved by two-dimensional (2D) silica gel thin-layer chromatography; dimension 1 was chloroform–methanol–water (65:25:4, vol/vol/vol), and dimension 2 was chloroform–methanol–acetic acid–water (80:12:15:4, vol/vol/vol/vol). Total lipids were detected using molybdatophosphoric acid, and specific functional groups were identified using spray reagents specific for defined functional groups (63).
Accession number(s).
All data underlying the findings are fully available without restriction. Sequencing files in FASTQ format are available in the European Nucleotide Archive under study accession numbers ERP018602 (RNA-seq) and ERP018603 (whole-genome sequencing).
Supplementary Material
ACKNOWLEDGMENTS
This work was funded by the Genoscope-CEA, a CNRS Chaire d'Excellence (to A.C.T.), and the Agence Nationale de la Recherche Grant ANR-16-CE05-0020 (to A.C.T.).
We thank P. Marlière for the GM3, E. Darii for mass spectrometry, and the Genoscope-CEA sequencing platform for DNA and RNA sequencing.
We declare no competing financial interests.
Footnotes
Supplemental material for this article may be found at https://doi.org/10.1128/AEM.00289-17.
REFERENCES
- 1.Metzger JO, Hüttermann A. 2009. Sustainable global energy supply based on lignocellulosic biomass from afforestation of degraded areas. Naturwissenschaften 96:279–288. doi: 10.1007/s00114-008-0479-4. [DOI] [PubMed] [Google Scholar]
- 2.Klinke HB, Thomsen AB, Ahring BK. 2004. Inhibition of ethanol-producing yeast and bacteria by degradation products produced during pre-treatment of biomass. Appl Microbiol Biotechnol 66:10–26. doi: 10.1007/s00253-004-1642-2. [DOI] [PubMed] [Google Scholar]
- 3.Galbe M, Zacchi G. 2007. Pretreatment of lignocellulosic materials for efficient bioethanol production. Adv Biochem Eng Biotechnol 108:41–65. [DOI] [PubMed] [Google Scholar]
- 4.Zaldivar J, Martinez A, Ingram LO. 1999. Effect of selected aldehydes on the growth and fermentation of ethanologenic Escherichia coli. Biotechnol Bioeng 65:24–33. doi:. [DOI] [PubMed] [Google Scholar]
- 5.Mills TY, Sandoval NR, Gill RT. 2009. Cellulosic hydrolysate toxicity and tolerance mechanisms in Escherichia coli. Biotechnol Biofuels 2:26. doi: 10.1186/1754-6834-2-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jönsson LJ, Alriksson B, Nilvebrant N-O. 2013. Bioconversion of lignocellulose: inhibitors and detoxification. Biotechnol Biofuels 6:16. doi: 10.1186/1754-6834-6-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ibraheem O, Ndimba BK. 2013. Molecular adaptation mechanisms employed by ethanologenic bacteria in response to lignocellulose-derived inhibitory compounds. Int J Biol Sci 9:598–612. doi: 10.7150/ijbs.6091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Axe DD, Bailey JE. 1995. Transport of lactate and acetate through the energized cytoplasmic membrane of Escherichia coli. Biotechnol Bioeng 47:8–19. doi: 10.1002/bit.260470103. [DOI] [PubMed] [Google Scholar]
- 9.Roe AJ, McLaggan D, Davidson I, O'Byrne C, Booth IR. 1998. Perturbation of anion balance during inhibition of growth of Escherichia coli by weak acids. J Bacteriol 180:767–772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Weber FJ, de Bont JA. 1996. Adaptation mechanisms of microorganisms to the toxic effects of organic solvents on membranes. Biochim Biophys Acta 1286:225–245. doi: 10.1016/S0304-4157(96)00010-X. [DOI] [PubMed] [Google Scholar]
- 11.Lynd LR, van Zyl WH, McBride JE, Laser M. 2005. Consolidated bioprocessing of cellulosic biomass: an update. Curr Opin Biotechnol 16:577–583. doi: 10.1016/j.copbio.2005.08.009. [DOI] [PubMed] [Google Scholar]
- 12.Warnick TA, Methé BA, Leschine SB. 2002. Clostridium phytofermentans sp. nov., a cellulolytic mesophile from forest soil. Int J Syst Evol Microbiol 52:1155–1160. doi: 10.1099/00207713-52-4-1155. [DOI] [PubMed] [Google Scholar]
- 13.Skerker JM, Leon D, Price MN, Mar JS, Tarjan DR, Wetmore KM, Deutschbauer AM, Baumohl JK, Bauer S, Ibáñez AB, Mitchell VD, Wu CH, Hu P, Hazen T, Arkin AP. 2013. Dissecting a complex chemical stress: chemogenomic profiling of plant hydrolysates. Mol Syst Biol 9:674. doi: 10.1038/msb.2013.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mutzel R, Marliere P. June 2000. Method and device for selecting accelerated proliferation of living cells in suspension. WIPO patent WO/2000/034433.
- 15.Ezeji T, Qureshi N, Blaschek HP. 2007. Butanol production from agricultural residues: impact of degradation products on Clostridium beijerinckii growth and butanol fermentation. Biotechnol Bioeng 97:1460–1469. doi: 10.1002/bit.21373. [DOI] [PubMed] [Google Scholar]
- 16.Miller EN, Jarboe LR, Yomano LP, York SW, Shanmugam KT, Ingram LO. 2009. Silencing of NADPH-dependent oxidoreductase genes (yqhD and dkgA) in furfural-resistant ethanologenic Escherichia coli. Appl Environ Microbiol 75:4315–4323. doi: 10.1128/AEM.00567-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Palmqvist E, Almeida JS, Hahn-Hägerdal B. 1999. Influence of furfural on anaerobic glycolytic kinetics of Saccharomyces cerevisiae in batch culture. Biotechnol Bioeng 62:447–454. doi:. [DOI] [PubMed] [Google Scholar]
- 18.Kang C, Hayes R, Sanchez EJ, Webb BN, Li Q, Hooper T, Nissen MS, Xun L. 2012. Furfural reduction mechanism of a zinc-dependent alcohol dehydrogenase from Cupriavidus necator JMP134. Mol Microbiol 83:85–95. doi: 10.1111/j.1365-2958.2011.07914.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.LoPachin RM, Gavin T. 2014. Molecular mechanisms of aldehyde toxicity: a chemical perspective. Chem Res Toxicol 27:1081–1091. doi: 10.1021/tx5001046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sander K, Wilson CM, Rodriguez M, Klingeman DM, Rydzak T, Davison BH, Brown SD. 2015. Clostridium thermocellum DSM 1313 transcriptional responses to redox perturbation. Biotechnol Biofuels 8:211. doi: 10.1186/s13068-015-0394-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Foo JL, Jensen HM, Dahl RH, George K, Keasling JD, Lee TS, Leong S, Mukhopadhyay A. 2014. Improving microbial biogasoline production in Escherichia coli using tolerance engineering. mBio 5:e01932-14. doi: 10.1128/mBio.01932-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Melville S, Craig L. 2013. Type IV pili in Gram-positive bacteria. Microbiol Mol Biol Rev 77:323–341. doi: 10.1128/MMBR.00063-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tomich M, Fine DH, Figurski DH. 2006. The TadV protein of Actinobacillus actinomycetemcomitans is a novel aspartic acid prepilin peptidase required for maturation of the Flp1 pilin and TadE and TadF pseudopilins. J Bacteriol 188:6899–6914. doi: 10.1128/JB.00690-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lee S, Lee JH, Mitchell RJ. 2015. Analysis of Clostridium beijerinckii NCIMB 8052's transcriptional response to ferulic acid and its application to enhance the strain tolerance. Biotechnol Biofuels 8:68. doi: 10.1186/s13068-015-0252-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Grandori R, Khalifah P, Boice JA, Fairman R, Giovanielli K, Carey J. 1998. Biochemical characterization of WrbA, founding member of a new family of multimeric flavodoxin-like proteins. J Biol Chem 273:20960–20966. doi: 10.1074/jbc.273.33.20960. [DOI] [PubMed] [Google Scholar]
- 26.Liu J, Guo T, Shen X, Xu J, Wang J, Wang Y, Liu D, Niu H, Liang L, Ying H. 2016. Engineering Clostridium beijerinckii with the Cbei_4693 gene knockout for enhanced ferulic acid tolerance. J Biotechnol 229:53–57. doi: 10.1016/j.jbiotec.2016.04.052. [DOI] [PubMed] [Google Scholar]
- 27.Adams M, Jia Z. 2005. Structural and biochemical analysis reveal pirins to possess quercetinase activity. J Biol Chem 280:28675–28682. doi: 10.1074/jbc.M501034200. [DOI] [PubMed] [Google Scholar]
- 28.Schoefer L, Mohan R, Schwiertz A, Braune A, Blaut M. 2003. Anaerobic degradation of flavonoids by Clostridium orbiscindens. Appl Environ Microbiol 69:5849–5854. doi: 10.1128/AEM.69.10.5849-5854.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kolek J, Patáková P, Melzoch K, Sigler K, Řezanka T. 2015. Changes in membrane plasmalogens of Clostridium pasteurianum during butanol fermentation as determined by lipidomic analysis. PLoS One 10:e0122058. doi: 10.1371/journal.pone.0122058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Timmons MD, Knutson BL, Nokes SE, Strobel HJ, Lynn BC. 2009. Analysis of composition and structure of Clostridium thermocellum membranes from wild-type and ethanol-adapted strains. Appl Microbiol Biotechnol 82:929–939. doi: 10.1007/s00253-009-1891-1. [DOI] [PubMed] [Google Scholar]
- 31.Nagan N, Zoeller RA. 2001. Plasmalogens: biosynthesis and functions. Prog Lipid Res 40:199–229. doi: 10.1016/S0163-7827(01)00003-0. [DOI] [PubMed] [Google Scholar]
- 32.Johnston NC, Goldfine H. 1983. Lipid composition in the classification of the butyric acid-producing clostridia. J Gen Microbiol 129:1075–1081. [DOI] [PubMed] [Google Scholar]
- 33.Zhao Y, Hindorff LA, Chuang A, Monroe-Augustus M, Lyristis M, Harrison ML, Rudolph FB, Bennett GN. 2003. Expression of a cloned cyclopropane fatty acid synthase gene reduces solvent formation in Clostridium acetobutylicum ATCC 824. Appl Environ Microbiol 69:2831–2841. doi: 10.1128/AEM.69.5.2831-2841.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ingram LO, Vreeland NS, Eaton LC. 1980. Alcohol tolerance in Escherichia coli. Pharmacol Biochem Behav 13(Suppl):S191–S195. doi: 10.1016/S0091-3057(80)80030-X. [DOI] [PubMed] [Google Scholar]
- 35.Kneidinger B, O'Riordan K, Li J, Brisson J-R, Lee JC, Lam JS. 2003. Three highly conserved proteins catalyze the conversion of UDP-N-acetyl-d-glucosamine to precursors for the biosynthesis of O antigen in Pseudomonas aeruginosa O11 and capsule in Staphylococcus aureus type 5. Implications for the UDP-N-acetyl-l-fucosamine biosynthetic pathway. J Biol Chem 278:3615–3627. doi: 10.1074/jbc.M203867200. [DOI] [PubMed] [Google Scholar]
- 36.Branda SS, Vik S, Friedman L, Kolter R. 2005. Biofilms: the matrix revisited. Trends Microbiol 13:20–26. doi: 10.1016/j.tim.2004.11.006. [DOI] [PubMed] [Google Scholar]
- 37.Ophir T, Gutnick DL. 1994. A role for exopolysaccharides in the protection of microorganisms from desiccation. Appl Environ Microbiol 60:740–745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Isabella VM, Clark VL. 2011. Identification of a conserved protein involved in anaerobic unsaturated fatty acid synthesis in Neiserria gonorrhoeae: implications for facultative and obligate anaerobes that lack FabA. Mol Microbiol 82:489–501. doi: 10.1111/j.1365-2958.2011.07826.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tolonen AC, Haas W, Chilaka AC, Aach J, Gygi SP, Church GM. 2011. Proteome-wide systems analysis of a cellulosic biofuel-producing microbe. Mol Syst Biol 7:461. doi: 10.1038/msb.2010.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sára M, Sleytr UB. 1987. Molecular sieving through S layers of Bacillus stearothermophilus strains. J Bacteriol 169:4092–4098. doi: 10.1128/jb.169.9.4092-4098.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Darmon E, Leach DRF. 2014. Bacterial genome instability. Microbiol Mol Biol Rev 78:1–39. doi: 10.1128/MMBR.00035-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Newbury SF, Smith NH, Robinson EC, Hiles ID, Higgins CF. 1987. Stabilization of translationally active mRNA by prokaryotic REP sequences. Cell 48:297–310. doi: 10.1016/0092-8674(87)90433-8. [DOI] [PubMed] [Google Scholar]
- 43.Xu C, Huang R, Teng L, Jing X, Hu J, Cui G, Wang Y, Cui Q, Xu J. 2015. Cellulosome stoichiometry in Clostridium cellulolyticum is regulated by selective RNA processing and stabilization. Nat Commun 6:6900. doi: 10.1038/ncomms7900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Graves MC, Rabinowitz JC. 1986. In vivo and in vitro transcription of the Clostridium pasteurianum ferredoxin gene. Evidence for “extended” promoter elements in Gram-positive organisms. J Biol Chem 261:11409–11415. [PubMed] [Google Scholar]
- 45.Helmann JD. 1995. Compilation and analysis of Bacillus subtilis sigma A-dependent promoter sequences: evidence for extended contact between RNA polymerase and upstream promoter DNA. Nucleic Acids Res 23:2351–2360. doi: 10.1093/nar/23.13.2351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Boutard M, Ettwiller L, Cerisy T, Alberti A, Labadie K, Salanoubat M, Schildkraut I, Tolonen AC. 2016. Global repositioning of transcription start sites in a plant-fermenting bacterium. Nat Commun 7:13783. doi: 10.1038/ncomms13783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zafarullah M, Charlier D, Glansdorff N. 1981. Insertion of IS3 can “turn-on” a silent gene in Escherichia coli. J Bacteriol 146:415–417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Fagan RP, Fairweather NF. 2014. Biogenesis and functions of bacterial S-layers. Nat Rev Microbiol 12:211–222. doi: 10.1038/nrmicro3213. [DOI] [PubMed] [Google Scholar]
- 49.Raeside C, Gaffé J, Deatherage DE, Tenaillon O, Briska AM, Ptashkin RN, Cruveiller S, Médigue C, Lenski RE, Barrick JE, Schneider D. 2014. Large chromosomal rearrangements during a long-term evolution experiment with Escherichia coli. mBio 5:e01377-14. doi: 10.1128/mBio.01377-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cavedon K, Leschine SB, Canale-Parola E. 1990. Cellulase system of a free-living, mesophilic clostridium (strain C7). J Bacteriol 172:4222–4230. doi: 10.1128/jb.172.8.4222-4230.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Boutard M, Cerisy T, Nogue P-Y, Alberti A, Weissenbach J, Salanoubat M, Tolonen AC. 2014. Functional diversity of carbohydrate-active enzymes enabling a bacterium to ferment plant biomass. PLoS Genet 10:e1004773. doi: 10.1371/journal.pgen.1004773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tolonen AC, Chilaka AC, Church GM. 2009. Targeted gene inactivation in Clostridium phytofermentans shows that cellulose degradation requires the family 9 hydrolase Cphy3367. Mol Microbiol 74:1300–1313. doi: 10.1111/j.1365-2958.2009.06890.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Langmead B, Salzberg SL. 2012. Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359. doi: 10.1038/nmeth.1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Delhomme N, Padioleau I, Furlong EE, Steinmetz LM. 2012. easyRNASeq: a bioconductor package for processing RNA-seq data. Bioinformatics 28:2532–2533. doi: 10.1093/bioinformatics/bts477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Anders S, Huber W. 2010. Differential expression analysis for sequence count data. Genome Biol 11:R106. doi: 10.1186/gb-2010-11-10-r106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Van der Auwera GA, Carneiro MO, Hartl C, Poplin R, Del Angel G, Levy-Moonshine A, Jordan T, Shakir K, Roazen D, Thibault J, Banks E, Garimella KV, Altshuler D, Gabriel S, DePristo MA. 2013. From FastQ data to high confidence variant calls: the Genome Analysis Toolkit best practices pipeline. Curr Protoc Bioinformatics 43:11.10.1–11.10.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tolonen AC, Zuroff TR, Ramya M, Boutard M, Cerisy T, Curtis WR. 2015. Physiology, genomics, and pathway engineering of an ethanol-tolerant strain of Clostridium phytofermentans. Appl Environ Microbiol 81:5440–5448. doi: 10.1128/AEM.00619-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Barrick JE, Colburn G, Deatherage DE, Traverse CC, Strand MD, Borges JJ, Knoester DB, Reba A, Meyer AG. 2014. Identifying structural variation in haploid microbial genomes from short-read resequencing data using breseq. BMC Genomics 15:1039. doi: 10.1186/1471-2164-15-1039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Siguier P, Gourbeyre E, Chandler M. 2014. Bacterial insertion sequences: their genomic impact and diversity. FEMS Microbiol Rev 38:865–891. doi: 10.1111/1574-6976.12067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tolonen AC, Cerisy T, El-Sayyed H, Boutard M, Salanoubat M, Church GM. 2015. Fungal lysis by a soil bacterium fermenting cellulose. Environ Microbiol 17:2618–2627. doi: 10.1111/1462-2920.12495. [DOI] [PubMed] [Google Scholar]
- 61.Schmittgen TD, Livak KJ. 2008. Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3:1101–1108. doi: 10.1038/nprot.2008.73. [DOI] [PubMed] [Google Scholar]
- 62.Miller LT. 1982. Single derivatization method for routine analysis of bacterial whole-cell fatty acid methyl esters, including hydroxy acids. J Clin Microbiol 16:584–586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Daniels L, Hanson RS, Phillips JA. 2007. Chemical analysis, p 462–503. In Reddy CA, Beveridge TJ, Breznak JA, Marzluf GA, Schmidt TM, Snyder LR (ed), Methods for general and molecular microbiology, 3rd ed ASM Press, Washington, DC. [Google Scholar]
- 64.Tatusov RL, Galperin MY, Natale DA, Koonin EV. 2000. The COG database: a tool for genome-scale analysis of protein functions and evolution. Nucleic Acids Res 28:33–36. doi: 10.1093/nar/28.1.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.