Abstract
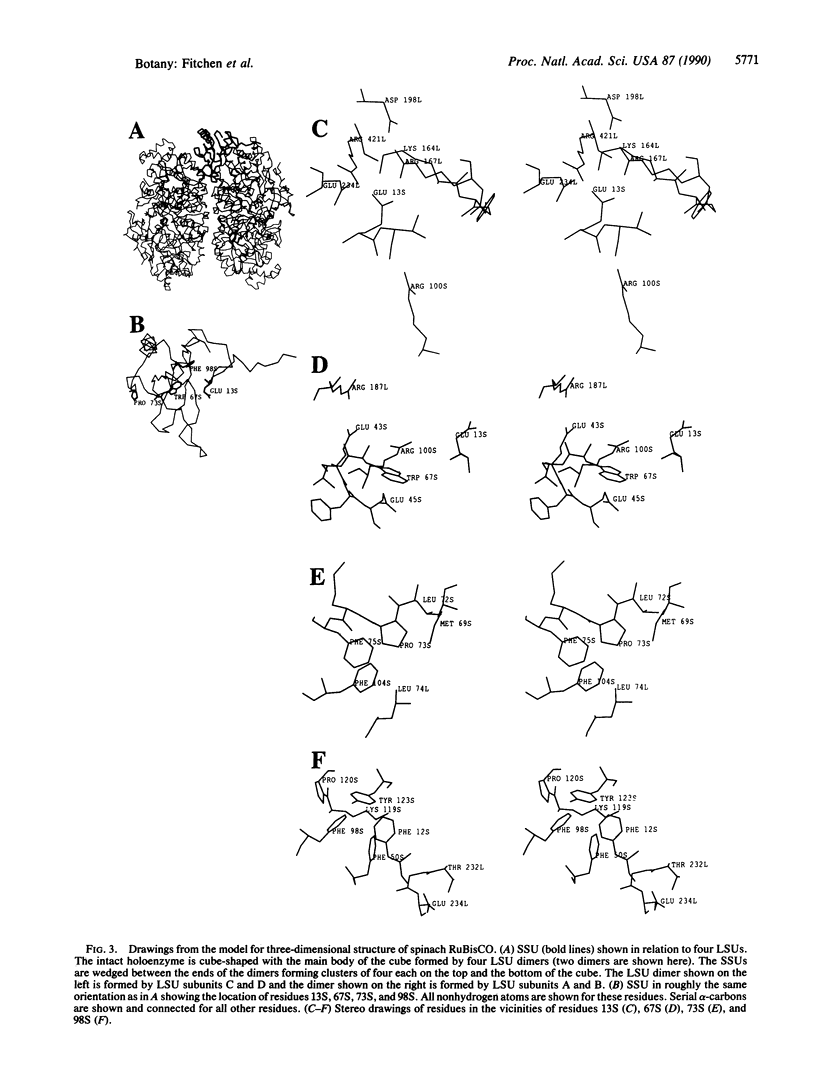
To explore the role of individual residues in the small subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase (EC 4.1.1.39), small subunits with single amino acid substitutions in three regions of relative sequence conservation were produced by directed mutagenesis of the rbcS gene from Anabaena 7120. These altered small subunits were cosynthesized with large subunits (from an expressed Anabaena rbcL gene) in Escherichia coli. Mutants were analyzed for effects on quaternary structure and catalytic activity. Changing Glu-13S (numbering used is that of the spinach enzyme) to Val, Trp-67S to Arg, Pro-73S to His, or Tyr-98S to Asn prevented accumulation of stable holoenzyme. Interpretation of these results using a model for the three-dimensional structure of the spinach enzyme based on x-ray crystallographic data suggests that our small subunit mutants containing substitutions at positions 13S and 67S probably do not assemble because of mispairing or nonpairing of charged residues on the interfacing surfaces of the large and small subunits. The failure of small subunits substituted at positions 73S or 98S to assemble correctly may result from disruption of intersubunit or intrasubunit hydrophobic pockets, respectively.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andersen K., Caton J. Sequence analysis of the Alcaligenes eutrophus chromosomally encoded ribulose bisphosphate carboxylase large and small subunit genes and their gene products. J Bacteriol. 1987 Oct;169(10):4547–4558. doi: 10.1128/jb.169.10.4547-4558.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews T. J. Catalysis by cyanobacterial ribulose-bisphosphate carboxylase large subunits in the complete absence of small subunits. J Biol Chem. 1988 Sep 5;263(25):12213–12219. [PubMed] [Google Scholar]
- Andrews T. J., Lorimer G. H. Catalytic properties of a hybrid between cyanobacterial large subunits and higher plant small subunits of ribulose bisphosphate carboxylase-oxygenase. J Biol Chem. 1985 Apr 25;260(8):4632–4636. [PubMed] [Google Scholar]
- Boczar B. A., Delaney T. P., Cattolico R. A. Gene for the ribulose-1,5-bisphosphate carboxylase small subunit protein of the marine chromophyte Olisthodiscus luteus is similar to that of a chemoautotrophic bacterium. Proc Natl Acad Sci U S A. 1989 Jul;86(13):4996–4999. doi: 10.1073/pnas.86.13.4996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chou P. Y., Fasman G. D. Prediction of the secondary structure of proteins from their amino acid sequence. Adv Enzymol Relat Areas Mol Biol. 1978;47:45–148. doi: 10.1002/9780470122921.ch2. [DOI] [PubMed] [Google Scholar]
- Elthon T. E., McIntosh L. Identification of the alternative terminal oxidase of higher plant mitochondria. Proc Natl Acad Sci U S A. 1987 Dec;84(23):8399–8403. doi: 10.1073/pnas.84.23.8399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goloubinoff P., Gatenby A. A., Lorimer G. H. GroE heat-shock proteins promote assembly of foreign prokaryotic ribulose bisphosphate carboxylase oligomers in Escherichia coli. Nature. 1989 Jan 5;337(6202):44–47. doi: 10.1038/337044a0. [DOI] [PubMed] [Google Scholar]
- Gurevitz M., Somerville C. R., McIntosh L. Pathway of assembly of ribulosebisphosphate carboxylase/oxygenase from Anabaena 7120 expressed in Escherichia coli. Proc Natl Acad Sci U S A. 1985 Oct;82(19):6546–6550. doi: 10.1073/pnas.82.19.6546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hemmingsen S. M., Woolford C., van der Vies S. M., Tilly K., Dennis D. T., Georgopoulos C. P., Hendrix R. W., Ellis R. J. Homologous plant and bacterial proteins chaperone oligomeric protein assembly. Nature. 1988 May 26;333(6171):330–334. doi: 10.1038/333330a0. [DOI] [PubMed] [Google Scholar]
- Herrero A., Elhai J., Hohn B., Wolk C. P. Infrequent cleavage of cloned Anabaena variabilis DNA by restriction endonucleases from A. variabilis. J Bacteriol. 1984 Nov;160(2):781–784. doi: 10.1128/jb.160.2.781-784.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones T. A., Thirup S. Using known substructures in protein model building and crystallography. EMBO J. 1986 Apr;5(4):819–822. doi: 10.1002/j.1460-2075.1986.tb04287.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knight S., Andersson I., Brändén C. I. Reexamination of the Three-Dimensional Structure of the Small Subunit of RuBisCo from Higher Plants. Science. 1989 May 12;244(4905):702–705. doi: 10.1126/science.244.4905.702. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Larson E., Howlett B., Jagendorf A. Artificial reductant enhancement of the Lowry method for protein determination. Anal Biochem. 1986 Jun;155(2):243–248. doi: 10.1016/0003-2697(86)90432-x. [DOI] [PubMed] [Google Scholar]
- Lee B., Richards F. M. The interpretation of protein structures: estimation of static accessibility. J Mol Biol. 1971 Feb 14;55(3):379–400. doi: 10.1016/0022-2836(71)90324-x. [DOI] [PubMed] [Google Scholar]
- Miziorko H. M., Lorimer G. H. Ribulose-1,5-bisphosphate carboxylase-oxygenase. Annu Rev Biochem. 1983;52:507–535. doi: 10.1146/annurev.bi.52.070183.002451. [DOI] [PubMed] [Google Scholar]
- Mulligan R. M., Houtz R. L., Tolbert N. E. Reaction-intermediate analogue binding by ribulose bisphosphate carboxylase/oxygenase causes specific changes in proteolytic sensitivity: the amino-terminal residue of the large subunit is acetylated proline. Proc Natl Acad Sci U S A. 1988 Mar;85(5):1513–1517. doi: 10.1073/pnas.85.5.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myers R. M., Lerman L. S., Maniatis T. A general method for saturation mutagenesis of cloned DNA fragments. Science. 1985 Jul 19;229(4710):242–247. doi: 10.1126/science.2990046. [DOI] [PubMed] [Google Scholar]
- Nakamaye K. L., Eckstein F. Inhibition of restriction endonuclease Nci I cleavage by phosphorothioate groups and its application to oligonucleotide-directed mutagenesis. Nucleic Acids Res. 1986 Dec 22;14(24):9679–9698. doi: 10.1093/nar/14.24.9679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nierzwicki-Bauer S. A., Curtis S. E., Haselkorn R. Cotranscription of genes encoding the small and large subunits of ribulose-1,5-bisphosphate carboxylase in the cyanobacterium Anabaena 7120. Proc Natl Acad Sci U S A. 1984 Oct;81(19):5961–5965. doi: 10.1073/pnas.81.19.5961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabita F. R., Small C. L. Expression and assembly of active cyanobacterial ribulose-1,5-bisphosphate carboxylase/oxygenase in Escherichia coli containing stoichiometric amounts of large and small subunits. Proc Natl Acad Sci U S A. 1985 Sep;82(18):6100–6103. doi: 10.1073/pnas.82.18.6100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vieira J., Messing J. Production of single-stranded plasmid DNA. Methods Enzymol. 1987;153:3–11. doi: 10.1016/0076-6879(87)53044-0. [DOI] [PubMed] [Google Scholar]
- Voordouw G., De Vries P. A., Van den Berg W. A., De Clerck E. P. Site-directed mutagenesis of the small subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase from Anacystis nidulans. Eur J Biochem. 1987 Mar 16;163(3):591–598. doi: 10.1111/j.1432-1033.1987.tb10908.x. [DOI] [PubMed] [Google Scholar]
- Wasmann C. C., Ramage R. T., Bohnert H. J., Ostrem J. A. Identification of an assembly domain in the small subunit of ribulose-1,5-bisphosphate carboxylase. Proc Natl Acad Sci U S A. 1989 Feb;86(4):1198–1202. doi: 10.1073/pnas.86.4.1198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanisch-Perron C., Vieira J., Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33(1):103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]




