Abstract
An inactivated virus vaccine was prepared from a pathogenic isolate of feline immunodeficiency virus containing a mutation that eliminated an endocytic sorting signal in the envelope glycoprotein, increasing its expression on virions. Cats immunized with inactivated preparations of this modified virus exhibited strong titers of antibody to Env by enzyme-linked immunosorbent assay. Evidence of protection following challenge demonstrated the potential of this approach to lentiviral vaccination.
Although several animal model systems have been developed to test lentivirus vaccines (18) and numerous immunogens have been evaluated, the mechanisms underlying protective immunity and the essential components of an effective vaccine remain unclear. Feline immunodeficiency virus (FIV) infection of the domestic cat is a useful model since it represents an immunosuppressive lentivirus in its natural host (18, 23). Vaccine-induced protection against FIV infection has been achieved using several immunogens, but protection has been restricted mainly to viruses of low virulence (8), with both inactivated virus vaccines and molecularly derived vaccines having been ineffective at protecting cats against homologous challenge with a pathogenic isolate (9, 11). Previous studies have indicated that Env is an important component of efficacious vaccines, to stimulate cytotoxic T cells (CTL) and virus-neutralizing antibodies (VNA) (5, 10). To attempt to improve immunogenicity, our strategy was to prepare a vaccine from a pathogenic clone of FIV-GL8 engineered to express high levels of Env by means of a mutation preventing Env endocytosis. In this way we aimed to reproduce high levels of Env in a native conformation.
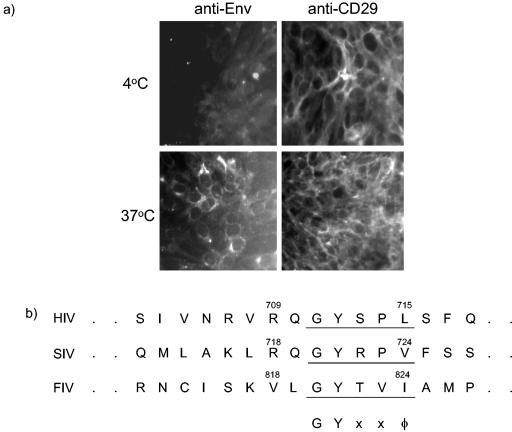
First we confirmed that FIV Env was endocytosed from the cell surface, as had been demonstrated for simian immunodeficiency virus Env (20). Immunofluorescence microscopy of FIV-infected CrFK cells incubated at 4°C with the anti-FIV Env monoclonal antibody vpg71.2 (22) demonstrated only low surface expression compared with that of control antibody recognizing CD29 (4B4). However, when cells were incubated with antibody at 37°C, a marked increase in fluorescence was noted in intracellular sites (Fig. 1a). These data indicated that at 37°C, Env was transiently expressed on the cell surface and then internalized. In contrast, no change in the level of fluorescence was seen on the cells incubated with the control antibody at 4 or 37°C.
FIG. 1.
(a) Immunofluorescence of FIV-infected CrFK cells incubated with antibodies detecting either FIV Env or CD29 for 1 h at either 4 or 37°C. (b) Conservation of the endocytosis motif between feline and primate lentiviruses. SIV, simian immunodeficiency virus. (c) Relative Env contents in the GL8WT (▪) and GL8YI (□) clones were determined by measuring the ability of virions to bind anti-FIV antibody. Equal amounts of gradient-purified virions of the two clones were adsorbed onto G. nivalis lectin-coated microwells and then probed for the ability to bind immunoglobulin G from cat sera diluted 1:100. Cat sera from a GL8-infected cat and a PET-infected cat and a serum pool from five uninfected control cats were used. The experiment was repeated twice, with comparable results.
The observation that FIV Env was rapidly endocytosed from the surfaces of infected cells, similar to what occurred with other lentivirus Envs (17), led us to identify the tyrosine-containing endocytosis motif GYTVI, located between positions 820 and 824 of the env gene of the GL8Mya molecular clone (9), corresponding to the GYXXφ motif conserved in all simian and human immunodeficiency virus (HIV) Envs (3, 4) (Fig. 1b). To test the effect of eliminating this motif in FIV env, we constructed the mutant GL8YI based on the GL8Mya molecular clone by PCR-mediated mutagenesis, incorporating the mutation Y821I. Stocks of GL8YI were prepared after transfection in 3T3 cells with Superfect transfection reagent (QIAGEN, Valencia, Calif.), followed by recovery into Mya-1 cells (19). Evaluation of Env surface expression by fluorescence-activated cell sorting on Mya-1 cells infected with either GL8YI or the wild-type GL8 clone (GL8WT) revealed that although GL8YI and GL8WT produced similar levels of FIV p24 as measured by enzyme-linked immunosorbent assay (ELISA) (data not shown), surface expression of Env was markedly greater for cells infected with GL8YI (49%) than for those infected with GL8WT (2.6%). Furthermore, we compared the Env contents of GL8WT and GL8YI by adsorbing equal amounts of the two viruses to Galanthus nivalis lectin-coated microwells and comparing the abilities of the adsorbed virions to bind FIV immune sera, using the method described previously (6). Although similar amounts of p24 were present by immunoblotting (data not shown), GL8YI bound considerably more antibody than GL8WT, indicating a higher Env content in the mutated virions (Fig. 1c). Subsequently, an inactivated virus vaccine was prepared from paraformaldehyde-treated culture fluids of GL8YI-infected Mya-1 cells as described previously (12).
Eight 11-week-old kittens were randomly divided into two groups of four. One group of kittens (V1 to V4) was immunized subcutaneously at 0, 3, and 7 weeks with 250 μg of inactivated GL8YI virus in a solution containing 0.5 ml of phosphate-buffered saline and 0.5 ml of MF 59.0 citrate adjuvant. The controls (C1 to C4) received 0.5 ml of phosphate-buffered saline and 0.5 ml of MF 59.0 citrate adjuvant (kindly provided by Chiron Corp.) at the same times. At week 10, 3 weeks following the third inoculation, no virus could be isolated from peripheral blood mononuclear cells (PBMC), confirming that the inactivated virus vaccine did not contain any residual infectivity for cats.
FIV gp140 Env, consisting of the entire surface unit and the ectodomain of the transmembrane domain, was used as the antigen in an ELISA to measure anti-Env antibodies in cat sera. The env gene of the FIV-GL8 molecular clone GL8414 (9) was cloned by PCR into pPPI4 (2) with KasI and BstBI, with the 3′ primer containing a C-terminal D7324 epitope tag (1). An antigen capture ELISA was developed based on one described previously (1) in which supernatant containing FIV Env (approximately 1 μg/ml) derived from 293T cell transfections was captured by using sheep antibody raised against the D7324 epitope tag (Cliniqa, Fallbrook, Calif.). Bound antibody was detected using goat anti-cat immunoglobulin G Fc alkaline phosphatase conjugate (Accurate Chemical and Scientific, Westbury, N.Y.) at a 1:5,000 dilution, and the signal was amplified and developed as described previously (1). GL8YI vaccinates developed anti-Env antibodies, detectable by ELISA in all vaccinates (Table 1), with the highest titer occurring in vaccinate V4.
TABLE 1.
Anti-Env antibody responses and neutralizing activity of day-of-challenge vaccine and control sera
| Source of serum | Anti-Env titer as determined by ELISA | VNA titer on indicated cells used for sera adsorptiona
|
||
|---|---|---|---|---|
| None | MBM | Mya-1 | ||
| Vaccinated cats | ||||
| V1 | 14,000 | <8 | 128 | 128 |
| V2 | 5,000 | <8 | 16 | 64 |
| V3 | 10,000 | <8 | <8 | <8 |
| V4 | 80,000 | <8 | 16 | <8 |
| Control cats | ||||
| C1 | <1,000 | <8 | <8 | <8 |
| C2 | <1,000 | <8 | <8 | <8 |
| C3 | <1,000 | <8 | <8 | <8 |
| C4 | <1,000 | <8 | <8 | <8 |
VNA titers were measured in untreated sera as well as cell-adsorbed sera as indicated and are expressed as the reciprocal of the highest serum dilution that gave 50% inhibition of reverse transcriptase production by 10 50% tissue culture infectious doses of the GL8 clone mixed with the corresponding dilution of a pool of 10 normal cat sera. The experiment was repeated twice, with comparable results.
To test whether the anti-Env antibodies were neutralizing, VNA were measured against 10 50% tissue culture infectious doses of the GL8 molecular clone using MBM cells as previously described (6), with or without prior adsorption of test sera with MBM or Mya-1 cells. Vaccinates V1, V2, and V4 developed VNA against the GL8 challenge virus, provided that the sera were absorbed with either MBM or Mya-1 cells prior to testing. It has been reported that the presence of antibodies to substrate cells can mask the detection of VNA in vaccinated-cat sera (6), and in this study absorption of sera with Mya-1 cells eliminated this ability to mask detection in two of four sera, whereas absorption of sera with the substrate MBM cells revealed neutralizing antibodies in three of four sera (Table 1).
To assess whether immunization with GL8YI would protect against virulent challenge, all eight cats were challenged intraperitoneally with 10 50% infectious doses of virus derived from the GL8 molecular clone at week 10. The challenge virus was prepared from the GL8414 (9) molecular clone of FIV by transfection of the murine fibroblast cell line 3T3 with Superfect transfection reagent (QIAGEN). Seventy-two hours posttransfection, supernatants were harvested, filtered through a 0.45-μm-pore-size filter, and used to infect the interleukin 2-dependent feline T-cell line Q201 (21). The infected cultures were monitored visually for cytopathicity and for the production of FIV p24 by ELISA (PetCheck FIV antigen ELISA; IDEXX Corp., Portland, Maine). Supernatants were collected at peak cytopathicity and p24 production, filtered (pore size, 0.45 μm), dispensed into 1-ml aliquots, and stored at −70°C. Previously we had demonstrated that infection of cats with a similar dose of this challenge stock led to a high proviral burden that was maintained throughout the acute phase of infection. Coincident with the sharp rise in proviral load was a marked decline in the CD4/CD8 ratio (13), consistent with the pathogenic potential of the GL8 molecular clone challenge stock.
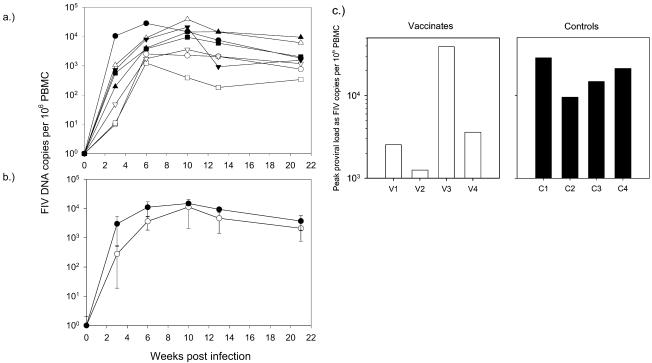
Cultures of PBMC isolated from samples from all cats taken 3 weeks postchallenge were positive by FIV p24 antigen ELISA after 7 days in culture, indicating that all of the vaccinated and control cats became infected following challenge. However, we noted that a smaller proportion of PBMC was infected in three vaccinates than in the remaining vaccinate (cat V3) and the control cats (data not shown), and so, to provide more quantitative information on the viral burdens following challenge, we examined proviral loads in PBMC at intervals until 21 weeks postchallenge by real-time PCR (7) using oligonucleotides designed to detect a variety of FIV A-subtype isolates as described previously (14-16) (Fig. 2a). The mean proviral load was consistently lower in the vaccinates than in the controls, although the difference did not reach statistical significance because of the high proviral load of vaccinate V3 (Fig. 2b). Furthermore, the peak proviral loads in PBMC of three of four vaccinates were markedly lower than those of the controls (Fig. 2c). Supporting evidence that the virulent challenge was controlled in three of four vaccinates was provided by the analysis of tissues sampled postmortem, 21 weeks after challenge (data not shown). The highest proviral burden in splenocytes was in vaccinate V3, consistent with the high proviral load in the PBMC of this cat throughout the study. In contrast, the proviral loads of the remaining vaccinates were lower than those of the controls in both splenocytes and PBMC, although again these differences were not statistically significant. Proviral DNA was detected in cells isolated from the mesenteric lymph nodes of all of the control cats but only a single vaccinate (cat V3). The high proviral loads detected in cat V3 were inconsistent with those in the remaining three vaccinates, providing no evidence of even partial protection against the challenge; indeed the viral and proviral loads measured in this cat usually exceeded the loads in the unvaccinated, control cats, suggesting that the infection may have been enhanced. Taken together, these data indicate that a larger vaccine trial is merited to test the applicability of this approach to lentiviral vaccines in general.
FIG. 2.
Proviral loads in vaccinated (open symbols) and control (filled symbols) cats measured at intervals postchallenge by real-time PCR. (a) Proviral loads of the individual cats (values for V3 are represented by triangles); (b) mean loads ± standard errors of the means for the vaccinates (○) and the controls (•); (c) peak proviral loads in PBMC from vaccinated (□) and control (▪) cats.
The mutated-virus preparation did express increased levels of Env, and immunization induced anti-Env responses in four of four cats and VNA in three of four cats. The pilot challenge study led to encouraging results, and although vaccination did not prevent infection with the pathogenic GL8 molecular clone, the course of infection was modified in the cats that developed VNA, which exhibited decreased viral burdens compared to those of the controls. Vaccinate V3, which developed very high viral and proviral loads following challenge, did not develop VNA. It is tempting to speculate, therefore, that the VNA detected in the remaining three vaccinates played a role in reducing the viral burden. Thus, the data indicate that Env is an important constituent of an effective lentiviral vaccine and encourage further studies to optimize immune responses against virulent isolates, since the GL8 molecular clone is pathogenic and achieves high virus loads similar to those described for other pathogenic strains (our unpublished observations). It is possible that the quality of the humoral immune response generated against Env was suboptimal, since the vaccine appeared to induce VNA inefficiently. Therefore, future studies might address methods of improving the quality of the VNA responses since the generation of protective immune responses in the domestic cat against challenge with the GL8 isolate would provide encouragement for the testing of similar strategies to increase the quantity of Env on HIV type 1 or HIV type 2 virions.
Acknowledgments
M.J.H. was supported by the United Kingdom Medical Research Council.
REFERENCES
- 1.Binley, J. M., P. J. Klasse, Y. Cao, I. Jones, M. Markowitz, D. D. Ho, and J. P. Moore. 1997. Differential regulation of the antibody responses to Gag and Env proteins of human immunodeficiency virus type 1. J. Virol. 71:2799-2809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Binley, J. M., R. W. Sanders, B. Clas, N. Schuelke, A. Master, Y. Guo, F. Kajumo, D. J. Anselma, P. J. Maddon, W. C. Olson, and J. P. Moore. 2000. A recombinant human immunodeficiency virus type 1 envelope glycoprotein complex stabilized by an intermolecular disulfide bond between the gp120 and gp41 subunits is an antigenic mimic of the trimeric virion-associated structure. J. Virol. 74:627-643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Boge, M., S. Wyss, J. S. Bonifacino, and M. Thali. 1998. A membrane-proximal tyrosine-based signal mediates internalization of the HIV-1 envelope glycoprotein via interaction with the AP-2 clathrin adaptor. J. Biol. Chem. 273:15773-15778. [DOI] [PubMed] [Google Scholar]
- 4.Bowers, K., A. Pelchen-Matthews, S. Honig, P. J. Vance, L. Creary, B. S. Haggarty, J. Romano, W. Ballensiefen, J. A. Hoxie, and M. Marsh. 2000. The simian immunodeficiency virus envelope glycoprotein contains multiple signals that regulate its cell surface expression and endocytosis. Traffic 1:661-674. [DOI] [PubMed] [Google Scholar]
- 5.Flynn, J. N., P. Keating, M. J. Hosie, M. Mackett, E. B. Stephens, J. A. Beatty, J. C. Neil, and O. Jarrett. 1996. Env-specific CTL predominate in cats protected from FIV infection by vaccination. J. Immunol. 157:3658-3665. [PubMed] [Google Scholar]
- 6.Giannecchini, S., D. Del Mauro, D. Matteucci, and M. Bendinelli. 2001. AIDS vaccination studies using an ex vivo feline immunodeficiency virus model: reevaluation of neutralizing antibody levels elicited by a protective and a nonprotective vaccine after removal of antisubstrate cell antibodies. J. Virol. 75:4424-4429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Heid, C. A., J. A. Stevens, K. J. Livak, and P. M. Williams. 1996. Real time quantitative PCR. Genome Res. 6:986-994. [DOI] [PubMed] [Google Scholar]
- 8.Hesselink, W., P. Sondermeijer, H. Pouwels, E. Verblakt, and C. Dhore. 1999. Vaccination of cats against feline immunodeficiency virus (FIV): a matter of challenge. Vet. Microbiol. 69:109-110. [DOI] [PubMed] [Google Scholar]
- 9.Hosie, M. J., T. H. Dunsford, D. Klein, B. J. Willett, C. Cannon, R. Osborne, J. Macdonald, N. Spibey, N. Mackay, O. Jarrett, and J. C. Neil. 2000. Vaccination with inactivated virus but not viral DNA reduces viral load following challenge with a heterologous and virulent isolate of feline immunodeficiency virus. J. Virol. 74:9403-9411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hosie, M. J., and J. N. Flynn. 1996. Feline immunodeficiency virus vaccination: characterization of the immune correlates of protection. J. Virol. 70:7561-7568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hosie, M. J., R. Osborne, G. Reid, J. C. Neil, and O. Jarrett. 1992. Enhancement after feline immunodeficiency virus vaccination. Vet. Immunol. Immunopathol. 35:191-198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hosie, M. J., R. Osborne, J. K. Yamamoto, J. C. Neil, and O. Jarrett. 1995. Protection against homologous but not heterologous challenge induced by inactivated feline immunodeficiency virus vaccines. J. Virol. 69:1253-1255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hosie, M. J., B. J. Willett, D. Klein, T. H. Dunsford, C. Cannon, M. Shimojima, J. Neil, and O. Jarrett. 2002. Evolution of replication efficiency following infection with a molecularly cloned feline immunodeficiency virus of low virulence. J. Virol. 76:6062-6072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Klein, D., P. Janda, R. Steinborn, M. Muller, B. Salmons, and W. H. Gunzburg. 1999. Proviral load determination of different feline immunodeficiency virus isolates using real-time polymerase chain reaction: influence of mismatches on quantification. Electrophoresis 20:291-299. [DOI] [PubMed] [Google Scholar]
- 15.Klein, D., C. Leutenegger, P. Bahula, R. Hofmann-Lehmann, R. Gold, B. Salmons, H. Lutz, and W. H. Guenzburg. 2001. Influence of preassay and sequence variations on viral load determination by a multiplex real-time reverse transcriptase-polymerase chain reaction for feline immunodeficiency virus. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 26:8-20. [DOI] [PubMed] [Google Scholar]
- 16.Leutenegger, C., D. Klein, R. Hofmann-Lehmann, C. Mislin, U. Hummel, J. Boni, F. Boretti, W. H. Guenzburg, and H. Lutz. 1999. Rapid FIV provirus quantitation by PCR using the TaqMan fluorigenic real time detection system. J. Virol. Methods 78:105-116. [DOI] [PubMed] [Google Scholar]
- 17.Marsh, M., A. Pelchen-Matthews, and J. A. Hoxie. 1997. Roles for endocytosis in lentiviral replication. Trends Cell Biol. 7:1-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Miller, R. J., J. S. Cairns, S. Bridges, and N. Sarver. 2000. Human immunodeficiency virus and AIDS: insights from animal lentiviruses. J. Virol. 74:7187-7195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Miyazawa, T. M., T. Furuya, S. Itagaki, Y. Tohya, E. Takahashi, and T. Mikami. 1989. Establishment of a feline T-lymphoblastoid cell line highly sensitive for replication of feline immunodeficiency virus. Arch. Virol. 108:131-135. [DOI] [PubMed] [Google Scholar]
- 20.Sauter, M. M., A. Pelchen-Matthews, R. Bron, M. Marsh, C. C. Labranche, P. J. Vance, J. Romano, B. S. Haggarty, T. K. Hart, W. M. Lee, and J. A. Hoxie. 1996. An internalization signal in the simian immunodeficiency virus transmembrane protein cytoplasmic domain modulates expression of envelope glycoproteins on the cell surface. J. Cell Biol. 132:795-811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Willett, B., M. J. Hosie, T. Dunsford, J. C. Neil, and O. Jarrett. 1991. Productive infection of helper T lymphocytes with FIV is accompanied by reduced expression of CD4. AIDS 5:1469-1475. [DOI] [PubMed] [Google Scholar]
- 22.Willett, B. J., M. J. Hosie, J. C. Neil, J. D. Turner, and J. A. Hoxie. 1997. Common mechanism of infection by lentiviruses. Nature 385:587. [DOI] [PubMed] [Google Scholar]
- 23.Willett, B. J., J. N. Flynn, and M. J. Hosie. 1997. FIV infection of the domestic cat: an animal model for AIDS. Immunol. Today 18:182-189. [DOI] [PubMed] [Google Scholar]