Abstract
Background
Autosomal dominant polycystic kidney disease (ADPKD) is a genetic ciliopathy disease characterized by progressive formation and enlargement of cysts in multiple organs. The kidneys are particularly affected and patients may eventually develop end-stage renal disease (ESRD). We hypothesize that bone marrow mesenchymal stromal cells (BMMSCs) are renotropic and may improve kidney function via anti-apoptotic, anti-fibrotic, and anti-inflammatory effects. In this study, we aim to assess the safety and tolerability of a BMMSC infusion in ADPKD patients.
Methods
We performed a single-arm phase I clinical trial with a 12-month follow-up. This study enrolled six eligible ADPKD patients with an estimated glomerular filtration rate (eGFR) of 25–60 ml/min/1.73 m2. Patients received autologous cultured BMMSCs (2 × 106 cells/kg) through the cubital vein according to our infusion protocol. We investigated safety issues and kidney function during the follow-up visits, and compared the findings to baseline and 1 year prior to the intervention.
Results
There were no patients lost to follow-up. We observed no cell-related adverse events (AE) and serious adverse events (SAE) after 12 months of follow-up. The mean eGFR value of 33.8 ± 5.3 ml/min/1.73 m2 1 year before cell infusion declined to 26.7 ± 3.1 ml/min/1.73 m2 at baseline (P = 0.03) and 25.8 ± 6.2 ml/min/1.73 m2 at the 12-month follow-up visit (P = 0.62). The mean serum creatinine (SCr) level of 2 ± 0.3 mg/dl 1 year before the infusion increased to 2.5 ± 0.4 mg/dl at baseline (P = 0.04) and 2.5 ± 0.6 mg/dl at the 12-month follow-up (P = 0.96). This indicated significant changes between the differences of these two periods (12 months before infusion to baseline, and 12 months after infusion to baseline) in SCr (P = 0.05), but not eGFR (P = 0.09).
Conclusions
This trial demonstrated the safety and tolerability of an intravenous transplantation of autologous BMMSCs. BMMSC efficacy in ADPKD patients should be investigated in a randomized placebo-controlled trial with a larger population, which we intend to perform.
Trial registration
ClinicalTrials.gov, NCT02166489. Registered on June 14, 2014.
Electronic supplementary material
The online version of this article (doi:10.1186/s13287-017-0557-7) contains supplementary material, which is available to authorized users.
Keywords: Autosomal dominant polycystic kidney disease, Bone marrow mesenchymal stromal cells, Chronic kidney disease
Background
Autosomal dominant polycystic kidney disease (ADPKD) is a genetic ciliopathy disease characterized by progressive formation and enlargement of cysts in multiple organs. The kidneys are particularly affected and patients may eventually progress to end-stage renal disease (ESRD). ADPKD is the most common genetic renal disease [1] that affects approximately 4 to 7 million individuals [2]. Mutations in polycystins 1 and 2 impair homeostasis and intracellular calcium signaling, which subsequently cause cyst formation, inflammation, and fibrosis in the kidneys [3, 4].
Despite recent advances in the identification of ADPKD pathogenesis, current treatments have been unable to completely cure or control disease progression. Tolvaptan, a vasopressin II receptor antagonist (V2RA), is the only drug that has passed a phase III clinical trial. Though it has the ability to reduce progression of height-adjusted total kidney volume (htTKV), it is associated with numerous adverse effects [5].
Transplantation of mesenchymal stromal cells (MSCs) is a recent approach to treat renal disease [6, 7]. MSCs are plastic-adherent cells that must at least express CD73, CD90, and CD105 and lack expression of CD45, CD34, CD14 and human leukocyte antigen (HLA)-DR. They have the potential for in vitro differentiation to osteoblasts, adipocytes, and chondroblasts [8]. Several studies report their ability to treat renal diseases by different mechanisms [9]. Anti-apoptotic [10], anti-fibrotic [11], and anti-inflammatory [12] capabilities of MSCs [7] provide the basis for their administration to control the progression of polycystic kidney disease (PKD). Franchi et al. have reported that transplanted MSCs restored kidney function in a PKD model and improved damaged vasculature of these kidneys [13]. Previously we showed the promising efficacy of transplanted MSCs in experimental models of chronic kidney disease (CKD) and acute kidney injury (AKI) [14–16]. We had indicated that MSC-derived conditioned medium induced renal tubular cell regeneration after nephrotoxicity induction [14], then we showed the bone marrow MSC (BMMSC) lowered serum creatinine (SCr) and urea levels and accelerated regulatory T cells (Tregs) in a monkey AKI model [15]. In a recent study, our results indicated that MSCs resulted in decreased CKD progression in a monkey model [16]. Carvalhosa et al. reported that cells lining cysts in ADPKD consisted of polycystic CD133+ progenitor [17]. Our unpublished data showed that in vitro co-culture of MSCs and tubular CD133+ progenitor cells from ADPKD patients resulted in reduced cyst formation and proliferation potential.
We hypothesize that MSCs are renotropic. Therefore, this study aims to assess the safety and tolerability of MSC infusion in ADPKD patients.
Methods
Study design
We performed a single-arm phase I clinical trial at one trial center with a 12-month follow-up period. We investigated safety issues and kidney function during the follow-up visits, and compared the findings to baseline and 1 year prior to intervention. Table 1 shows the study assessment schedule. We evaluated 27 ADPKD patients for potential participation in this trial. We had diagnosed all these patients according to unified imaging diagnostic criteria [18]. Patients were under our observation for at least 1 year prior to study enrollment. Their blood pressures (BP) were well controlled. From these potential participants, we recruited six eligible ADPKD patients who provided written informed consent for study participation. This study enrolled patients from June 2014 to January 2015. All patients have been on a special diet that included decreased salt, protein, and caffeine consumption, and high water intake, recommended by our nutritionist. We strongly recommended that patients keep track and not change their medications, supplements, or make any lifestyle changes. This trial was registered with clinical trial number NCT02166489.
Table 1.
Assessment schedule
| SV | BV1 | V1 | BV2 | V2 | V3 | V4 | V5 | V6 | V7 | |
|---|---|---|---|---|---|---|---|---|---|---|
| Day | -56 | -49 | -42 | 0 | 1 | 30 | 90 | 180 | 270 | 360 |
| Time window | NA | ±3d | ±7d | ±1d | ±1d | ±7d | ±7d | ±7d | ±7d | ±7d |
| Informed consent | x | |||||||||
| Physical examination | x | x | x | x | x | x | x | x | ||
| Vital signs | x | x | x | x | x | x | x | x | x | |
| Medical history | x | |||||||||
| Laboratory assessments | x | x | x | x | x | x | x | |||
| CBC | x | x | x | x | x | x | ||||
| BG | x | x | x | x | x | x | ||||
| NA, K, Ca, P | x | x | x | x | x | x | ||||
| BUN | x | x | x | x | x | x | ||||
| Uric acid | x | x | x | x | x | x | ||||
| Serum creatinine | x | x | x | x | x | x | x | |||
| eGFR | x | x | x | x | x | x | x | |||
| Albumin | x | x | x | |||||||
| TG, Chol, LDL,HDL | x | x | x | x | x | x | ||||
| FBS | x | x | x | x | x | x | x | |||
| HbA1c | x | x | ||||||||
| TSH, PTH | x | x | ||||||||
| ESR, CRP | x | x | x | x | x | x | ||||
| ALT, ALK-p, AST | x | x | x | x | x | x | ||||
| Dipstick proteinuria | x | x | x | x | x | x | ||||
| HIV 1,2, HCV, HBV, HTLV | x | |||||||||
| Eligibility criteria assessment | x | x | ||||||||
| Kidney sonography | x | x | ||||||||
| Kidney DTPA scan | x | x | ||||||||
| BM harvesting | x | |||||||||
| MSC infusion | x | |||||||||
| Follow-up visit | x | x | x | x | x | |||||
| AE assessment a | x | x | x | x | x | x | ||||
| SAE assessment a | x | x | x | x | x | x |
SV screen visit, BV1 baseline visit before bone marrow aspiration (BMA), BV2 baseline visit before infusion day, V visit, d day, CBC complete blood count, BUN blood urea nitrogen, eGFR estimated glomerular filtration rate, TG triglycerides, Chol cholesterol, LDL low-density lipoprotein, HDL high-density lipoprotein, FBS fasting blood sugar, HbA1c hemoglobin A1c, TSH thyroid-stimulating hormone, PTH parathyroid hormone, ESR erythrocyte sedimentation rate, CRP C-reactive protein, ALT alanine aminotransferase, AlK-p alkaline phosphatase, AST aspartate aminotransferase, HIV human immunodeficiency virus, HCV hepatitis C virus, HBV hepatitis B virus, HTLV human T-lymphotropic virus, DTPA diethylenetriaminepentaacetic acid, BM bone marrow, MSC mesenchymal stromal cell, AE adverse event, SAE serious adverse event
aAE and SAE were assessed during the follow-up visit and when reported by patients
We evaluated both safety and tolerability of the cell infusion according to physical examination, adverse event (AE) assessments, and laboratory changes. Potential efficacy included changes in kidney length (KL) measured by ultrasound imaging, estimated glomerular filtration rate (eGFR) by the modification of diet in renal disease (MDRD) study formula, and glomerular filtration rate (GFR) by diethylenetriaminepentaacetic acid (DTPA) scan.
We abstracted data that pertained to the “1 year prior to infusion” point from patients’ medical files in the clinic. We scheduled patient follow-up visits for clinical and laboratory assessments at specified intervals of 1, 3, 6, 9, and 12 months after the cell infusion.
Enrollment criteria
Inclusion criteria were: (1) both genders; (2) ADPKD confirmed by sonography imaging or genetic testing; (3) age 18 to 60 years; (4) eGFR 25–60 mL/min/1.73 m2; (5) ability to understand and willingness to sign a consent form. Exclusion criteria consisted of: (1) pregnancy or breastfeeding; (2) positive history of associated cardiovascular disease; (3) diabetes that required medical intervention; (4) other systemic diseases that involved the kidneys such as cancers, autoimmune diseases, blood diseases, and liver diseases; (5) hospitalization due to illness 2 months prior to study entry; (6) life expectancy less than 2 years; and (7) allergies to the cell culture ingredients.
Primary and secondary endpoints
Primary endpoints consisted of the numbers, type, and severity of AEs related to the cell infusion. We recorded AEs and serious adverse events (SAE) during follow-up visits and whenever patients reported any symptoms. We recorded the type and grade of AEs according to the Common Terminology Criteria for Adverse Events (CTCAE) version 4.0. We sent these reports to the Data Safety Monitoring Board (DSMB) of the study. Secondary endpoints consisted of changes in eGFR (as an existing surrogate endpoint) [19] from baseline to 12 months after the cell infusion.
Kidney ultrasound imaging and kidney DTPA scan
Additional file 1 lists this information.
Bone marrow aspiration (BMA) procedure, cell preparation, and culture
Once the patients’ laboratory viral tests were normal (Additional file 1: Table S1), an oncologist performed a bone marrow aspiration (BMA) under local anesthesia (2% lidocaine solution) and an intravenous sedation that consisted of 0.1 mg/kg midazolam and 25–50 mg intravenous fentanyl. The BMA was obtained from the posterior superior iliac crest with the patient placed in the lateral decubitus position. Samples were placed inside a cold box and sent to the clean room. The oncologist discharged all patients 4 h after the procedure. We telephoned patients 2 days later to ask about any complications related to the BMA procedure. None of the patients reported any complications. Bone marrow (BM) mononuclear cells were plated at 1 × 106 cells/cm2 in a 150-cm2 culture flask with alpha modified Eagle medium (α-MEM; 22571-020, Gibco BRL, Karlsruhe, Germany) supplemented with 100 IU penicillin and 100 IU streptomycin (15070-063, Gibco), 10% Hyclone defined fetal bovine serum (FBS; SH30070.03, Thermo Fisher Scientific, Waltham, MA, USA), and 1% L-glutamine (25030-024, Gibco). Phase-contrast microscopy of the BMMSC cultures demonstrated a low heterogeneous population that predominately consisted of long, spindle-shaped cells. For each patient, we characterized the cells for immunophenotypic properties that included surface marker expressions for MSCs - phycoerythrin (PE)-conjugated CD105, CD44, CD73 (Becton Dickinson, Franklin Lakes, NJ, USA) and fluorescein isothiocyanate (FITC)-conjugated CD90 (Dako, Glostrup, Denmark) along with those that were negative for hematopoietic stem cell (HSC) surface markers PE-conjugated CD34 and FITC-conjugated CD45. Negative isotype controls included nonspecific mouse IgG1-FITC/IgG1-PE and IgG2a-FITC that were substituted for the primary antibody. All samples were analyzed by flow cytometry (BD FACS Caliber, BD Biosciences, San Jose, CA, USA) and win-MDI2.9 software. Additional file 2: Figure S1 show MSC characterization of the enrolled patients. The cells were tested for possible microbial contamination prior to infusion. We used quality control tests (QCT) in accordance with the recommendations for cell and tissue therapy promotion and validation tests of the Iranian Health Ministry Pharmacopoeia Commission and the Department of Health and Human Services, Food and Drug Organization (FDO). Tests included the sample microbial test, limulus amebocyte lysate (LAL) gel clot assay, mycoplasma detection, and karyotype analysis.
Passages 1 to 3 of the cultured BMMSCs were washed with phosphate-buffered saline and trypsinized with trypsin/EDTA (0.05%; 25300-062, Gibco). The cells were diluted in 10 ml of normal saline and loaded into 10 ml sterile syringes. For each patient, we prepared approximately 1–2 × 106 cells/kg, which were delivered to the institute’s operating room in a cold box at a temperature of approximately 4 °C.
Cell infusion
We infused autologous cultured MSCs (1–2 × 106/kg) through the cubital vein according to our infusion protocol.
Statistical analysis
Data are written as mean ± standard deviation (SD). Following normal distribution of parametric data, we used the paired t test to compare variables at two time points (baseline and 12 months after cell infusion). We compared variables at three time points (12 months before infusion, baseline, and 12 months after infusion) by the repeated measures ANOVA test. A two-sided P value of <0.05 was considered statistically significant. We used SPSS for statistical analyses (Version 16, SPSS Inc., Chicago, IL, USA).
Results
Patient characteristics
All six patients completed the trial. No patients were lost to follow-up. Table 2 lists the patients’ characteristics and demographic data at the time of enrollment. Additional file 1: Table S1 shows the cell parameters. All patients’ BP was under tight control prior to enrollment. Patients’ antihypertensive medications did not change during the trial. There were five patients with children. None of these patients underwent any procedures to prevent the transmission of the disease gene to their children. One patient has not attempted to have any children. None experienced external renal complications due to PKD prior to enrollment. We did not observe any external renal complications from PKD during the 12 months of follow-up. Although one patient had autosomal dominant polycystic liver disease (ADPLD), we did not observe any changes in the size of her liver by ultrasonography at the 12-month follow-up.
Table 2.
Patient characteristics and demographic data at the time of enrollment
| Patient number | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| Sex (m/f) | F | M | M | M | F | F |
| Age (years) | 45 | 37 | 39 | 28 | 51 | 54 |
| Race | Caucasian | Caucasian | Caucasian | Caucasian | Caucasian | Caucasian |
| Educational level | Graduate studies | Completed high school | Graduate studies | Graduate studies | Completed high school | Completed high school |
| Marital status | Married | Married | Married | Married | Married | Married |
| Number of children | 2 | 1 | 2 | 0 | 2 | 2 |
| Employment | Full-time employment | Full-time employment | Part-time employment | Full-time employment | Retired | Homemaker |
| Diagnosis due to | Incidental imaging | Screening | Screening | Screening | Screening | Screening |
| BMI (kg/m2) | 29.38 | 26.45 | 26.18 | 35.51 | 24.34 | 23.41 |
| Blood group | O+ | A- | A+ | O+ | B+ | B+ |
| BP sitting position (mm Hg) | 135/80 | 115/79 | 110/70 | 131/70 | 133/73 | 127/83 |
| eGFR (ml/min/1.73 m2) | 25 | 32 | 25 | 25 | 25 | 29 |
| GFR DTPA SCAN | 23 | 41.6 | 24 | 28 | 26 | 32.83 |
| Serum creatinine (mg/dl) | 2.3 | 2.3 | 2.8 | 3.0 | 2.5 | 1.9 |
| Dipstick proteinuriaa | Trace | Neg | Trace | Neg | Neg | + |
| Previous symptoms or complication of ADPKD (yes/no) | ||||||
| Cyst infection | N | N | N | N | N | N |
| Urinary infection | Y | N | N | Y | N | N |
| Flank pain | N | N | N | Y | Y | N |
| Macrohematuria | N | N | N | Y | Y | N |
| Other | N | N | N | N | N | N |
| Past medical history (yes/no) | ||||||
| DM | N | N | N | N | N | N |
| HTN | Y | Y | Y | Y | Y | Y |
| ADPLD | N | N | N | N | N | Y |
| Other (name) | L4, L5 laminectomy, kidney stone | Varicocelectomy | Low back pain, kidney stone | Incarcerated spinal disc, canal stenosis, kidney stone, fatty liver grade I | Hypothyroidism, kidney stone, C/S surgery | Hysterectomy |
| Diagnosis of HTN, age | 40 | 32 | 19 | 22 | 36 | 44 |
| Duration of CKD at enrollment (estimation by month) | 62 | 132 | 36 | 14 | 120 | 120 |
| Smoking history (yes/no) | N | N | N | N | N | N |
| Alcohol consumption history (yes/no) | N | N | N | N | N | N |
| Family history of ADPKD (yes/no) | N | Y | Y | Y | Y | Y |
| Under controlled diet (yes/no)b | Y | Y | Y | Y | Y | Y |
| Drug list (name/dose) | Metoral 50 mg/daily calcitrol I daily PD-poetin each 10 days | Gemfibrozil 300 mg/daily losartan 25 mg/daily amlodipine 5 mg/daily allopurinol 100 mg/daily calcium I daily | Enalapril 5 mg/daily losartan 25 mg/daily | Losartan 25 mg/daily atenolol 50 mg/daily diltiazem 60 mg/daily prazocin 5 mg/daily allopurinol 100 mg/daily, nephrovit I daily, vit D3 I biweekly | Losartan 25 mg ½ daily atorvastatin 20 mg/daily allopurinol 100 mg ½ daily levothyroxine ½ daily calcitrol in 5 days of week | Losartan 50 mg bid amilodipine 50 mg/daily allopurinol 100 mg/daily |
BMI body mass index, BP blood pressure, eGFR estimated glomerular filtration rate, DTPA diethylenetriaminepentaacetic acid, ADPKD autosomal dominant polycystic kidney disease, DM diabetes mellitus, HTN hypertension, ADPLD autosomal dominant polycystic liver disease, CKD chronic kidney disease, NA not applicable, N no, Y yes
a Neg: 0 mg/dl; Trace: 15–30 mg/dl; +: 30–100 mg/dl; ++: 100–300 mg/d; +++: 300–1000 mg/dl; ++++: >1000 mg/dl
b Under controlled diet is defined as: no: on normal diet; yes: patients have been under the recommendation of nutritionist by taking low protein, low salt diet, with low caffeine and high intake of water
Safety and tolerability
We discharged all patients 2 h after the cell infusion and observed no AEs during this time. The bone marrow puncture site healed within 72 h and the intravenous infusion site healed within 24 h without any related complications. In total, we recorded 1 SAE and 37 AEs during the 12-month follow-up in all patients (Additional file 1: Table S2).
We sent the related AE reports to the DSMB of the trial as scheduled. The DSMB raised the issue that vomiting and dizziness AEs occurred shortly after the DTPA kidney scan in the majority of patients at the baseline and follow-up visits. They recommended that we cancel the DTPA kidney scans for the rest of the trial.
According to DSMB comments, all other AEs were unrelated to the intervention and probably attributed to patients’ underlying disease progression or other medical conditions. All AEs resolved spontaneously or by the use of over-the-counter medications.
One patient experienced an SAE. Patient number 3 who had a positive history for low back pain took a single dose of a celecoxib tablet (200 mg) and one acetaminophen tablet (500 mg) due to severe back pain 3 weeks after his cell infusion. His blood urea nitrogen (BUN) and SCr levels elevated 24 h after celecoxib and acetaminophen. We advised him against using these types of medications. Instead, we prescribed physiotherapy for pain relief. After 1 week, his BUN and SCr values returned to previous levels. We referred him to the orthopedic surgeon who performed a lumbosacral magnetic resonance imaging (MRI) scan without contrast. According to the MRI report, the patient had L4/L5 disc demyelination and a central disc protrusion with significant canal and left foraminal stenosis. He underwent surgery 1 week later. After surgery, his pain reduced and cleared completely within 10 days.
Primary endpoint
We did not detect any SAEs or AEs related to the cell infusion in any of the patients during the 12-month follow-up. The results showed no significant differences in laboratory parameters, which we evaluated for safety issues after 1, 3, 6, 9 (Additional file 1: Table S3) and 12 months compared to baseline (Table 3).
Table 3.
Laboratory parameters at baseline and 12-month follow-up
| Patient parameters | Normal range | Baseline (Mean ± SD) | 12-month (Mean ± SD) | Difference (Mean ± SD) | P value (Paired t test) |
|---|---|---|---|---|---|
| Leukocytes (/ul) | 4000–10000 | 5700 ± 885.4 | 6468.3 ± 2153.2 | 768.3 ± 1406.9 | 0.24 |
| Hemoglobin (g/dl) | 12–16 | 12.8 ± 1.7 | 13 ± 1.2 | 0.2 ± 0.8 | 0.55 |
| HCT (%) | 38–47 | 37.4 ± 3.8 | 39.5 ± 3.4 | 2.1 ± 2.9 | 0.15 |
| MCV (fl) | 80–96 | 88.8 ± 9.4 | 89.5 ± 3.3 | 0.7 ± 7.8 | 0.82 |
| Platelets (*103/ul) | 150–450 | 211.7 ± 56.8 | 187.3 ± 42 | -24.3 ± 46 | 0.25 |
| FBS (mg/dl) | 60–105 | 96.8 ± 10 | 87.4 ± 11.9 | -6.2 ± 7.2 | 0.13 |
| HbA1c (%) | 4–6 | 5.2 ± 0.3 | 5.1 ± 0.40 | 0 ± 0.1 | 0.3 |
| Sodium (mEq/l) | 134–148 | 141.8 ± 1.9 | 141.3 ± 2 | -0.5 ± 1.2 | 0.36 |
| Potassium (mEq/l) | 3.5–5.5 | 4.4 ± 0.5 | 4.3 ± 0.5 | -0.1 ± 0.4 | 0.41 |
| Calcium (mg/dl) | 8.6–10 | 9.3 ± 0.6 | 9.4 ± 0.3 | 0 ± 0.8 | 0.79 |
| Phosphorus (mg/dl) | 2.6–4.5 | 3.7 ± 0.6 | 3.7 ± 0.5 | 0 ± 0.5 | 0.89 |
| TSH (MIU/ml) | 0.2–5.0 | 2.3 ± 1.1 | 2.4 ± 1.3 | 0.1 ± 1.7 | 0.86 |
| PTH (pg/ml) | 15–65 | 140.3 ± 71 | 112 ± 48.3 | -29.2 ± 55.2 | 0.25 |
| ESR 1 h (mm/h) | 3–20 | 22.2 ± 18.8 | 15.5 ± 10.3 | -6.7 ± 9.9 | 0.16 |
| CRP (mg/l) | 0–10 | 5.1 ± 8.7 | 0.3 ± 0.9 | -4.7 ± 8.5 | 0.23 |
| Albumin (g/dl) | 3.5–5.2 | 4.3 ± 0.3 | 4.4 ± 0.5 | 0.1 ± 0.6 | 0.68 |
| Uric acid (mg/l) | 2.6–6 | 6.5 ± 2.1 | 7.2 ± 1.3 | 0.7 ± 1.7 | 0.38 |
| ALT(U/l) | 0–40 | 24.3 ± 9.8 | 19.3 ± 6.6 | -5.0 ± 11.5 | 0.34 |
| AST (U/l) | 0–31 | 29.7 ± 18.5 | 20.5 ± 5.8 | -9.1 ± 17.4 | 0.25 |
| Alkaline phosphatase (U/l) | 0–240 | 231.2 ± 62.5 | 155.8 ± 53.8 | -75.3 ± 83.9 | 0.07 |
| Total cholesterol (mg/dl) | <200 desirable | 177 ± 44.2 | 172.5 ± 42 | -4.5 ± 23.7 | 0.66 |
| Triglycerides (mg/dl) | <200 desirable | 191.7 ± 150.7 | 192.5 ± 108.2 | 0.8 ± 62.8 | 0.97 |
| LDL cholesterol (mg/dl) | <100 low risk | 99.5 ± 34 | 96 ± 30.2 | -3.5 ± 18.8 | 0.67 |
| HDL cholesterol (mg/dl) | >55 no risk | 39.2 ± 11 | 41.3 ± 12.3 | 2.2 ± 9.8 | 0.61 |
| Dipstick proteinuriaa | Negative | 1.7 ± 0.8 | 1.3 ± 0.5 | -0.3 ± 1 | 0.46 |
ul microliter, g/dl grams per deciliter, HCT hematocrit, MCV mean corpuscular volume, fl femtoliters, FBS fasting blood sugar, HbA1c hemoglobin A1c, mg/dl milligram per deciliter, mEq/l milliequivalents per liter, TSH thyroid-stimulating hormone, MIU/ml milli-international units per milliliter, PTH parathyroid hormone, pg/ml pictogram per milliliter, ESR erythrocyte sedimentation rate, mm/h millimeters per hour, CRP C-reactive protein, mg/l milligram per liter, ALT alanine aminotransferase, U/lit units per liter, AST aspartate aminotransferase, LDL low-density lipoprotein, HDL high-density lipoprotein
a Negative or 1 = 0 mg/dl; 2 = 15–30 mg/dl; 3 = 30–100 mg/dl; 4 = 100–300 mg/d; 5 = 300–1000 mg/dl; 6 > 1000 mg/dl
Secondary endpoint
The single-dose autologous MSC infusion did not induce any significant changes in eGFR, nor reductions in SCr at 12 months compared to baseline in all patients (Table 4).
Table 4.
Changes in renal function parameters and blood pressure (BP) from 1 year before mesenchymal stromal cell (MSC) infusion, baseline and the 12-month follow up
| Patient parameters | Normal range | -12 | Baseline | 12-month | P value (RMA) | Dif 1 (BV-1yp) | P value Dif 1 | Dif 2 (12 m-BV) | P value Dif 2 | Dif 3 (dif2- dif1) | P valve Dif 3 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| SBP (mmHg) | <120 | 123.8 ± 9.6 | 125.2 ± 10.3 | 126.8 ± 5.7 | 0.55 | 1.3 ± 6.8 | 0.65 | 1.6 ± 5.0 | 0.46 | NA | NA |
| DBP (mmHg) | 60-80 | 77.7 ± 3.5 | 75.8 ± 5.6 | 78.8 ± 3.3 | 0.43 | -1.8 ± 4.5 | 0.36 | 3 ± 6 | 0.27 | NA | NA |
| BUN (mg/dl) | 7–20.6 | 28.5 ± 8.6 | 27.8 ± 10.5 | 29.5 ± 7.1 | 0.92 | -0.6 ± 12.7 | 0.90 | 1.7 ± 4.3 | 0.38 | NA | NA |
| SCr (mg/dl) | 0.4–1.4 | 2 ± 0.3 | 2.5 ± 0.4 | 2.5 ± 0.6 | 0.02b | 0.5 ± 0.4 | 0.04b | 0 ± 0.3 | 0.96 | 0.5 ± 0.5 | 0.05b |
| eGFRa | 90–120 | 33.8 ± 5.3 | 26.7 ± 3.1 | 25.8 ± 6.2 | 0.01b | -7.2 ± 6 | 0.03b | -0.8 ± 3.9 | 0.62 | -6.3 ± 7.5 | 0.09 |
| Rt KL (cm) | 10–12 | NA | 19.1 ± 2 | 19.5 ± 2 | NA | NA | NA | 0.4 ± 0.6 | 0.13 | NA | NA |
| Lt KL (cm) | 10–12 | NA | 18.3 ± 2.4 | 18.7 ± 2.6 | NA | NA | NA | 0.4 ± 0.4 | 0.06 | NA | NA |
BV baseline visit, 1yp 1 year prior to intervention, 12 m 12 month after baseline, RMA repeated measures ANOVA, Dif difference, SPB systolic blood pressure, mmHg millimeter of mercury, NA not applicable, DBP diastolic blood pressure, BUN blood urea nitrogen, mg/dl milligram per deciliter SCr serum creatinine, eGFR estimated glomerular filtration rate, Rt right, KL kidney length, Lt left
a MDRD study formula (ml/min/1.73 m2)
b Significant
eGFR, sCr, and blood pressure (BP)
Patients had a mean eGFR value of 33.8 ± 5.3 ml/min/1.73 m2 1 year before the cell infusion, which declined to 26.7 ± 3.1 ml/min/1.73 m2 at baseline (P = 0.03) and 25.8 ± 6.2 ml/min/1.73 m2 at the 12-month visit (P = 0.62). Figure 1 shows the patients’ eGFR changes. The mean SCr level of 2 ± 0.30 mg/dl within the same period increased to 2.5 ± 0.4 mg/dl at baseline (P = 0.04) and 2.5 ± 0.6 mg/dl at the 12-month follow-up (P = 0.96). There was a significant change between the differences of these two periods (-12, baseline and +12, baseline) in SCr (P = 0.05) but not in eGFR (P = 0.09; Table 4).
Fig. 1.
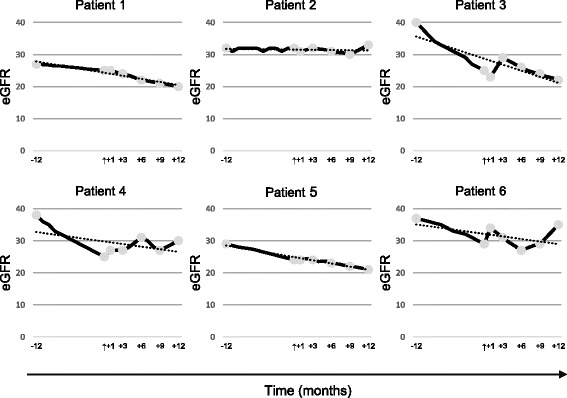
Estimated glomerular filtration rate (eGFR) changes in autosomal dominant polycystic kidney disease (ADPKD) patients from 1 year prior to mesenchymal stromal cell (MSC) infusion up to 12 months after the infusion
Systolic (P = 0.55) and diastolic (P = 0.43) BP did not significantly change 12 months after the cell infusion (Table 4). We observed no significant changes in renal function parameters and BP during the year after intervention (Additional file 1: Table S4).
Kidney length (KL)
There was a slight increase in the median left KL at baseline (18.3 ± 2.4 cm) to 12 months after the cell infusion (18.7 ± 2.6 cm; P = 0.06). The median right KL at baseline was 19.1 ± 2 cm which increased slightly to 19.5 ± 2 cm at 12 months (P = 0.13; Table 4).
Discussion
To the best of our knowledge, this is the first clinical trial that has shown the safety and tolerability of a single BMMSC intravenous infusion in ADPKD patients.
However, the mean eGFR level noticeably decreased from 1 year before intervention to baseline (P = 0.03). We observed a slight, nonsignificant decrease for eGFR at 12-month follow-up that could not be considered exactly due to the intervention.
We observed enlargement of both kidneys in all patients at 12 months after the cell infusion that was probably due to usual cyst growth. We used KL to evaluate disease progression and, to some extent, represent the size of the kidney [20, 21]. However, KL is not as appropriate as HtTKV as proposed by the Consortium for Radiologic Imaging Studies of Polycystic Kidney Disease (CRISP) [22].
The HALT study showed a crucial role for hypertension (HTN) control in management of ADPKD [23]. Accordingly, we tightly controlled HTN in our patients prior to their enrollment. This did not change during the follow-up period.
Franchi et al. performed the only preclinical study that used allogeneic MSCs in a PKD rat model. They showed the beneficial effect of a single intravenous infusion for improvements in systolic HTN and fibrosis. Although the infusion did not affect cyst size and number, it had a significant improvement on cortical and parenchymal vasculature density, which resulted in better tubular function and improvements in creatinine clearance seen in the intervention group in the autosomal recessive polycystic kidney disease (ARPKD) model [13]. They also reported the safety of the MSC infusion with no mortality related to their protocol, which supported our safety results.
There is a growing body of clinical research which indicates that a systematic MSC infusion is safe under different conditions [24], including renal diseases [7, 9]. No AEs such as acute infusion toxicity, organ complications, infections, death or malignancies associated with systematic infusion of these cells have been reported in human studies [24]. In concordance with their results, our study showed the safety of an intravenous MSC infusion following comprehensive assessment of AEs, SAEs, and clinical and paraclinical parameters. There are also reports of potential efficacy and safety of MSC infusions in patients with different etiologies of CKD [9, 25–27]. However, there is a lack of data demonstrates the impact of MSCs on PKD.
ADPKD progression is associated with several pathways such as apoptosis [28], fibrosis [2, 29, 30], inflammation [2, 31, 32], cyst proliferation, renin–angiotensin system (RAS) activation [33, 34], and kidney vasculature impairment [13]. MSCs may affect these pathways through different mechanisms, as briefly discussed in the following.
Recent experimental studies reported that MSCs predominantly improved kidney tubular regeneration by secretion of growth factors, cytokines, and chemokines which decrease apoptosis, inflammation, and fibrosis [7], and subsequently ameliorate kidney function (creatinine clearance, eGFR, and HTN) and structure in CKD models [7, 9]. The potential capabilities of MSCs [35] are not limited to antifibrotic and anti-inflammatory effects on CKD models [11, 12] and its beneficial impact on apoptosis in an AKI model [10, 36] which could be useful for PKD.
Rapamycin decreases the proliferation of CD133+ and CD24+ cells and cyst formation in vitro through inhibition of the mechanistic target of rapamycin (mTOR) in PKD [17]. In our in vitro study, we observed that MSCs could reduce the proliferative potential and cyst formation of CD133+ progenitor cells from ADPKD patients (unpublished data).
The mitogen-activated protein kinase (MAPK) family is involved in proliferation, apoptosis, and fibrosis in PKD. MSC-derived conditioned medium has been shown to inhibit p38 mitogen-activated protein kinases (p38) and extracellular signal-regulated kinase (ERK) activity (MAPK family) as well as induce antiapoptotic effects in an AKI rat model [37].
Various studies illustrated that downregulation of pro-inflammatory cytokines reduced cyst formation and inflammation in ADPKD [38–40]. MSCs have the potential to decrease both the activity of the nuclear factor kappa B (NF-ĸB) pathway [11] and pro-inflammatory cytokines, which eventually lessen inflammation in a CKD model [12].
Following ADPKD progression, renovascular architecture disrupts where there is the vessel network around the cyst [13, 41]. Vascular endothelial growth factor (VEGF) is an antiapoptotic molecule [42] that can normalize the blood vessel network surrounding the cysts, BUN, SCr, and the cyst disorder as well as reduce macrophage pericystic infiltration in an ARPKD model [43]. VEGF C has been shown to decrease in an ARPKD mice model [43]. Human MSCs are a source of VEGF and may be useful for appropriate angiogenesis [44].
RAS inhibition improves BP and TKV, and prevents the spread of cyst formation in experimental animal models and humans [23, 45]. MSCs reduce renin, angiotensin-converting enzyme (ACE), and angiotensin II type 1 (AT1) receptor expression, and lead to reductions in BP, inflammation, and fibrosis [46, 47]. The MSC inhibitory effect on RAS is more stable than ACE inhibitors [47].
Taken together, following the safety and tolerability results of our study, the next step is to test the efficacy of MSC in ADPKD patients.
Limitations
The current trial limitations included our inability to demonstrate the efficacy due to the study design that consisted of a small sample size, lack of a control group, short follow-up period, the single cell infusion, and not using htTKV as a surrogate endpoint.
Conclusions
This trial demonstrated that an intravenous infusion of autologous MSCs was safe and well tolerated in ADPKD patients. We could not assess the efficacy due to the trial design. Our findings should be investigated in a randomized placebo-controlled trial with a larger population, which we intend to perform.
Additional files
Method of kidney ultrasound imaging. DTPA kidney scan method. Cell parameters for the study patients. Table S2. Safety assessment of the mesenchymal stromal cell (MSC) infusion in 1 year following baseline visit of study patients. Table S3. Laboratory parameters at baseline, 1, 3, 6, 9, and 12 months of follow-up. Table S4. Changes in renal function parameters and blood pressure (BP) 1 year before, and at 1, 3, 6, 9, and 12 months after mesenchymal stromal cell (MSC) infusion. (DOCX 70 kb)
Characterization of the patients’ bone mesenchymal stromal cells (MSCs). (DOCX 781 kb)
Acknowledgments
We gratefully express our appreciation to Ms. Dastvan for proofreading this article, Ms. Salimiyan for assistance with data gathering, Dr. Moininia for bone marrow aspiration, and Dr. Dastgheib, Dr. Ghasemi and Ms. Khoshdelrad for their valuable comments, in addition to Dr. Amini for assistance with statistical analyses. We are grateful to members of the Department of Regenerative Medicine for their expertise and feedback. We would particularly like to thank the patients and their families for participating in this study.
Funding
This trial was supported by a grant from Royan Institute and the Royan Charity Association for Health Research.
Availability of data and materials
The data that support the findings of this study are available from the enrolled patients but restrictions apply to the availability of these data, which were used under license for the current study. Hence, they are not publicly available. However, the data are available from the authors upon reasonable request and with permission of the enrolled patients.
Authors’ contributions
AM, NA, and RM conceived and designed the original protocol. All authors were involved in amending the protocol. AM and SSH coordinated the study. NJ and TB performed the cell processing. AM, BE, and SSH were involved in patient recruitment and follow-up visits. Data entry was performed by SSH and SEH, and SSH performed the cell infusion. SSH cleaned and analyzed the data. SSH wrote the first draft of the manuscript. HB oversaw the manuscript preparation and revised the first draft. NA supervised the study. All authors contributed to subsequent and final drafts. All authors read and approved the final manuscript.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
All patients gave written informed consent for publication.
Ethics approval and consent to participate
We conducted this study in accordance with the Declaration of Helsinki. The Institutional Review Board (IRB) and local Ethics Committee (Royan Research Ethics Committee, number: 91000316) approved this study. The DSMB and the trial monitor reviewed the entire trial to ensure participants’ safety. All patients gave written informed consent for participation.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviations
- ACE
Angiotensin-converting enzyme
- ADPKD
Autosomal dominant polycystic kidney disease
- ADPLD
Autosomal dominant polycystic liver disease
- AE
Adverse events
- AKI
Acute kidney injury
- ARPKD
Autosomal recessive polycystic kidney disease
- AT1
Angiotensin II receptor type 1
- BM
Bone marrow
- BMA
Bone marrow aspiration
- BMMSCs
Bone marrow mesenchymal stromal cells
- BP
Blood pressure
- BUN
Blood urea nitrogen
- CKD
Chronic kidney disease
- CRISP
Consortium for Radiologic Imaging Studies of Polycystic Kidney Disease
- CTCAE
Common Terminology Criteria for Adverse Events
- DSMB
Data Safety Monitoring Board
- DTPA
Diethylenetriaminepentaacetic acid
- eGFR
Estimated glomerular filtration rate
- ERK
Extracellular signal-regulated kinase
- ESRD
End-stage renal disease
- FBS
Fetal bovine serum
- FDO
Food and Drug Organization
- FITC
Fluorescein isothiocyanate
- GFR
Glomerular filtration rates
- h
Hour
- HLA
Human leukocyte antigen
- HSC
Hematopoietic stem cell
- HTN
Hypertension
- htTKV
Height-adjusted total kidney volume
- KL
Kidney length
- LAL
Limulus amebocyte lysate
- MAPK
Mitogen-activated protein kinase
- MDRD
Modification of diet in renal disease
- MRI
Magnetic resonance imaging
- MSCs
Mesenchymal stromal cells
- mTOR
Mechanistic target of rapamycin
- NF-ĸB pathway
Nuclear factor kappa B - a prototypical pro-inflammatory signaling pathway
- p38
p38 mitogen-activated protein kinases
- PE
Phycoerythrin
- PKD
Polycystic kidney disease
- QCT
Quality control tests
- RAS
Renin–angiotensin system
- SAE
Serious adverse events
- SCr
Serum creatinine
- SD
Standard deviation
- Tregs
Regulatory T cells
- V2RA
Vasopressin II receptor antagonist
- VEGF
Vascular endothelial growth factor
- α-MEM
alpha modified Eagle medium
Footnotes
Electronic supplementary material
The online version of this article (doi:10.1186/s13287-017-0557-7) contains supplementary material, which is available to authorized users.
Contributor Information
Atieh Makhlough, Phone: +98-9111510564, Email: makhlough_a@yahoo.com.
Soroosh Shekarchian, Phone: +98-2122339949, Email: soroosh.shekarchian@royaninstitute.org.
Reza Moghadasali, Phone: +98-21235625060, Email: rezamoghadasali@yahoo.com.
Behzad Einollahi, Phone: +98-2181262073, Email: einollahi@yahoo.com.
Seyedeh Esmat Hosseini, Phone: +98-2122516465, Email: esmat.hosseini_110@yahoo.com.
Neda Jaroughi, Phone: +98-2122436301, Email: bluebox7777@gmail.com.
Tina Bolurieh, Phone: +98-2122436301, Email: tbolurieh@yahoo.com.
Hossein Baharvand, Phone: +98-2122306485, Email: baharvand@royaninstitute.org.
Nasser Aghdami, Phone: +98-2122436300, Email: nasser.aghdami@royaninstitute.org.
References
- 1.Iglesias CG, Torres VE, Offord KP, Holley KE, Beard CM, Kurland LT. Epidemiology of adult polycystic kidney disease, Olmsted County, Minnesota: 1935-1980. Am J Kidney Dis. 1983;2(6):630–9. doi: 10.1016/S0272-6386(83)80044-4. [DOI] [PubMed] [Google Scholar]
- 2.Torra R. Autosomal dominant policystic kidney disease, more than a renal disease. Minerva Endocrinol. 2014;39(2):79–87. [PubMed] [Google Scholar]
- 3.Chang MY, Ong AC. New treatments for autosomal dominant polycystic kidney disease. Br J Clin Pharmacol. 2013;76(4):524–35. doi: 10.1111/bcp.12136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bastos AP, Onuchic LF. Molecular and cellular pathogenesis of autosomal dominant polycystic kidney disease. Braz J Med Biol Res. 2011;44(7):606–17. doi: 10.1590/S0100-879X2011007500068. [DOI] [PubMed] [Google Scholar]
- 5.Torres VE, Chapman AB, Devuyst O, Gansevoort RT, Grantham JJ, Higashihara E, Perrone RD, Krasa HB, Ouyang J, Czerwiec FS, et al. Tolvaptan in patients with autosomal dominant polycystic kidney disease. N Engl J Med. 2012;367(25):2407–18. doi: 10.1056/NEJMoa1205511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Suzuki E, Fujita D, Takahashi M, Oba S, Nishimatsu H. Adult stem cells as a tool for kidney regeneration. World J Nephrol. 2016;5(1):43–52. doi: 10.5527/wjn.v5.i1.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Morigi M, Rota C, Remuzzi G. Mesenchymal stem cells in kidney repair. Methods Mol Biol. 2016;1416:89–107. doi: 10.1007/978-1-4939-3584-0_5. [DOI] [PubMed] [Google Scholar]
- 8.Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315–7. doi: 10.1080/14653240600855905. [DOI] [PubMed] [Google Scholar]
- 9.Hickson LJ, Eirin A, Lerman LO. Challenges and opportunities for stem cell therapy in patients with chronic kidney disease. Kidney Int. 2016;89(4):767–78. doi: 10.1016/j.kint.2015.11.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bruno S, Grange C, Collino F, Deregibus MC, Cantaluppi V, Biancone L, Tetta C, Camussi G. Microvesicles derived from mesenchymal stem cells enhance survival in a lethal model of acute kidney injury. PLoS One. 2012;7(3) doi: 10.1371/journal.pone.0033115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wu HJ, Yiu WH, Li RX, Wong DW, Leung JC, Chan LY, Zhang Y, Lian Q, Lin M, Tse HF, et al. Mesenchymal stem cells modulate albumin-induced renal tubular inflammation and fibrosis. PLoS One. 2014;9(3) doi: 10.1371/journal.pone.0090883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Semedo P, Correa-Costa M, Antonio Cenedeze M, Maria Avancini Costa Malheiros D, Antonia dos Reis M, Shimizu MH, Seguro AC, Pacheco-Silva A, Saraiva Camara NO. Mesenchymal stem cells attenuate renal fibrosis through immune modulation and remodeling properties in a rat remnant kidney model. Stem Cells. 2009;27(12):3063–73. doi: 10.1002/stem.214. [DOI] [PubMed] [Google Scholar]
- 13.Franchi F, Peterson KM, Xu R, Miller B, Psaltis PJ, Harris PC, Lerman LO, Rodriguez-Porcel M. Mesenchymal stromal cells improve renovascular function in polycystic kidney disease. Cell Transplant. 2015;24(9):1687–98. doi: 10.3727/096368914X684619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Moghadasali R, Mutsaers HA, Azarnia M, Aghdami N, Baharvand H, Torensma R, Wilmer MJ, Masereeuw R. Mesenchymal stem cell-conditioned medium accelerates regeneration of human renal proximal tubule epithelial cells after gentamicin toxicity. Exp Toxicol Pathol. 2013;65(5):595–600. doi: 10.1016/j.etp.2012.06.002. [DOI] [PubMed] [Google Scholar]
- 15.Moghadasali R, Azarnia M, Hajinasrollah M, Arghani H, Nassiri SM, Molazem M, Vosough A, Mohitmafi S, Najarasl M, Ajdari Z, et al. Intra-renal arterial injection of autologous bone marrow mesenchymal stromal cells ameliorates cisplatin-induced acute kidney injury in a rhesus Macaque mulatta monkey model. Cytotherapy. 2014;16(6):734–49. doi: 10.1016/j.jcyt.2014.01.004. [DOI] [PubMed] [Google Scholar]
- 16.Moghadasali R, Hajinasrollah M, Argani H, Nassiri SM, Najarasl M, Sodeifi N, Baharvand H, Aghdami N. Autologous transplantation of mesenchymal stromal cells tends to prevent progress of interstitial fibrosis in a rhesus Macaca mulatta monkey model of chronic kidney disease. Cytotherapy. 2015;17(11):1495–505. doi: 10.1016/j.jcyt.2015.06.006. [DOI] [PubMed] [Google Scholar]
- 17.Carvalhosa R, Deambrosis I, Carrera P, Pasquino C, Rigo F, Ferrari M, Lasaponara F, Ranghino A, Biancone L, Segoloni G, et al. Cystogenic potential of CD133+ progenitor cells of human polycystic kidneys. J Pathol. 2011;225(1):129–41. doi: 10.1002/path.2920. [DOI] [PubMed] [Google Scholar]
- 18.Pei Y, Obaji J, Dupuis A, Paterson AD, Magistroni R, Dicks E, Parfrey P, Cramer B, Coto E, Torra R, et al. Unified criteria for ultrasonographic diagnosis of ADPKD. J Am Soc Nephrol. 2009;20(1):205–12. doi: 10.1681/ASN.2008050507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hartung EA. Biomarkers and surrogate endpoints in kidney disease. Pediatr Nephrol. 2016;31(3):381–91. doi: 10.1007/s00467-015-3104-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Vlajkovic S, Cukuranovic R, Bjelakovic MD, Ilic G, Jaksic T, Cukuranovic J. Relative length of human kidney as more precise measuring of normal kidney. Med Pregl. 2010;63(11-12):784–7. doi: 10.2298/MPNS1012784V. [DOI] [PubMed] [Google Scholar]
- 21.Bhutani H, Smith V, Rahbari-Oskoui F, Mittal A, Grantham JJ, Torres VE, Mrug M, Bae KT, Wu Z, Ge Y, et al. A comparison of ultrasound and magnetic resonance imaging shows that kidney length predicts chronic kidney disease in autosomal dominant polycystic kidney disease. Kidney Int. 2015;88(1):146–51. doi: 10.1038/ki.2015.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bae KT, Tao C, Wang J, Kaya D, Wu Z, Bae JT, Chapman AB, Torres VE, Grantham JJ, Mrug M, et al. Novel approach to estimate kidney and cyst volumes using mid-slice magnetic resonance images in polycystic kidney disease. Am J Nephrol. 2013;38(4):333–41. doi: 10.1159/000355375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Schrier RW. Blood pressure in early autosomal dominant polycystic kidney disease. N Engl J Med. 2015;372(10):976–7. doi: 10.1056/NEJMc1500332. [DOI] [PubMed] [Google Scholar]
- 24.Lalu MM, McIntyre L, Pugliese C, Fergusson D, Winston BW, Marshall JC, Granton J, Stewart DJ. Canadian Critical Care Trials Group. Safety of cell therapy with mesenchymal stromal cells (SafeCell): a systematic review and meta-analysis of clinical trials. PLoS One. 2012;7(10) doi: 10.1371/journal.pone.0047559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tan J, Wu W, Xu X, Liao L, Zheng F, Messinger S, Sun X, Chen J, Yang S, Cai J, et al. Induction therapy with autologous mesenchymal stem cells in living-related kidney transplants: a randomized controlled trial. JAMA. 2012;307(11):1169–77. doi: 10.1001/jama.2012.316. [DOI] [PubMed] [Google Scholar]
- 26.El-Ansary M, Saadi G, Abd El-Hamid SM. Mesenchymal stem cells are a rescue approach for recovery of deteriorating kidney function. Nephrology (Carlton) 2012;17(7):650–7. doi: 10.1111/j.1440-1797.2012.01622.x. [DOI] [PubMed] [Google Scholar]
- 27.Lee H, Park JB, Lee S, Baek S, Kim H, Kim SJ. Intra-osseous injection of donor mesenchymal stem cell (MSC) into the bone marrow in living donor kidney transplantation; a pilot study. J Transl Med. 2013;11:96. doi: 10.1186/1479-5876-11-96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Riella C, Czarnecki PG, Steinman TI. Therapeutic advances in the treatment of polycystic kidney disease. Nephron Clin Pract. 2014;128(3-4):297–302. doi: 10.1159/000368244. [DOI] [PubMed] [Google Scholar]
- 29.Omori S, Hida M, Fujita H, Takahashi H, Tanimura S, Kohno M, Awazu M. Extracellular signal-regulated kinase inhibition slows disease progression in mice with polycystic kidney disease. J Am Soc Nephrol. 2006;17(6):1604–14. doi: 10.1681/ASN.2004090800. [DOI] [PubMed] [Google Scholar]
- 30.Sugiyama N, Kohno M, Yokoyama T. Inhibition of the p38 MAPK pathway ameliorates renal fibrosis in an NPHP2 mouse model. Nephrol Dial Transplant. 2012;27(4):1351–8. doi: 10.1093/ndt/gfr550. [DOI] [PubMed] [Google Scholar]
- 31.Nagao S, Yamaguchi T, Kusaka M, Maser RL, Takahashi H, Cowley BD, Grantham JJ. Renal activation of extracellular signal-regulated kinase in rats with autosomal-dominant polycystic kidney disease. Kidney Int. 2003;63(2):427–37. doi: 10.1046/j.1523-1755.2003.00755.x. [DOI] [PubMed] [Google Scholar]
- 32.Okumura Y, Sugiyama N, Tanimura S, Nishida M, Hamaoka K, Kohno M, Yokoyama T. ERK regulates renal cell proliferation and renal cyst expansion in inv mutant mice. Acta Histochem Cytochem. 2009;42(2):39–45. doi: 10.1267/ahc.08040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Torres VE, Donovan KA, Scicli G, Holley KE, Thibodeau SN, Carretero OA, Inagami T, McAteer JA, Johnson CM. Synthesis of renin by tubulocystic epithelium in autosomal-dominant polycystic kidney disease. Kidney Int. 1992;42(2):364–73. doi: 10.1038/ki.1992.297. [DOI] [PubMed] [Google Scholar]
- 34.Saigusa T, Dang Y, Bunni MA, Amria MY, Steele SL, Fitzgibbon WR, Bell PD. Activation of the intrarenal renin-angiotensin-system in murine polycystic kidney disease. Physiol Rep. 2015;3(5) doi: 10.14814/phy2.12405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Patel DM, Shah J, Srivastava AS. Therapeutic potential of mesenchymal stem cells in regenerative medicine. Stem Cells Int. 2013;2013:496218. doi: 10.1155/2013/496218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhou Y, Xu H, Xu W, Wang B, Wu H, Tao Y, Zhang B, Wang M, Mao F, Yan Y, et al. Exosomes released by human umbilical cord mesenchymal stem cells protect against cisplatin-induced renal oxidative stress and apoptosis in vivo and in vitro. Stem Cell Res Ther. 2013;4(2):34. doi: 10.1186/scrt194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Qi S, Wu D. Bone marrow-derived mesenchymal stem cells protect against cisplatin-induced acute kidney injury in rats by inhibiting cell apoptosis. Int J Mol Med. 2013;32(6):1262–72. doi: 10.3892/ijmm.2013.1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Li X, Magenheimer BS, Xia S, Johnson T, Wallace DP, Calvet JP, Li R. A tumor necrosis factor-alpha-mediated pathway promoting autosomal dominant polycystic kidney disease. Nat Med. 2008;14(8):863–8. doi: 10.1038/nm1783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ta MH, Harris DC, Rangan GK. Role of interstitial inflammation in the pathogenesis of polycystic kidney disease. Nephrology (Carlton) 2013;18(5):317–30. doi: 10.1111/nep.12045. [DOI] [PubMed] [Google Scholar]
- 40.Lee EJ, Song SA, Mun HW, Yoo KH, Choi SY, Park EY, Park JH. Blockade of interleukin-8 receptor signalling inhibits cyst development in vitro, via suppression of cell proliferation in autosomal polycystic kidney disease. Nephrology (Carlton) 2014;19(8):471–8. doi: 10.1111/nep.12261. [DOI] [PubMed] [Google Scholar]
- 41.Xu R, Franchi F, Miller B, Crane JA, Peterson KM, Psaltis PJ, Harris PC, Lerman LO, Rodriguez-Porcel M. Polycystic kidneys have decreased vascular density: a micro-CT study. Microcirculation. 2013;20(2):183–9. doi: 10.1111/micc.12022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ilic D, Almeida EA, Schlaepfer DD, Dazin P, Aizawa S, Damsky CH. Extracellular matrix survival signals transduced by focal adhesion kinase suppress p53-mediated apoptosis. J Cell Biol. 1998;143(2):547–60. doi: 10.1083/jcb.143.2.547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Huang JL, Woolf AS, Kolatsi-Joannou M, Baluk P, Sandford RN, Peters DJ, McDonald DM, Price KL, Winyard PJ, Long DA. Vascular endothelial growth factor C for polycystic kidney diseases. J Am Soc Nephrol. 2016;27(1):69–77. doi: 10.1681/ASN.2014090856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kagiwada H, Yashiki T, Ohshima A, Tadokoro M, Nagaya N, Ohgushi H. Human mesenchymal stem cells as a stable source of VEGF-producing cells. J Tissue Eng Regen Med. 2008;2(4):184–9. doi: 10.1002/term.79. [DOI] [PubMed] [Google Scholar]
- 45.Zafar I, Tao Y, Falk S, McFann K, Schrier RW, Edelstein CL. Effect of statin and angiotensin-converting enzyme inhibition on structural and hemodynamic alterations in autosomal dominant polycystic kidney disease model. Am J Physiol Renal Physiol. 2007;293(3):F854–9. doi: 10.1152/ajprenal.00059.2007. [DOI] [PubMed] [Google Scholar]
- 46.Oliveira-Sales EB, Maquigussa E, Semedo P, Pereira LG, Ferreira VM, Camara NO, Bergamaschi CT, Campos RR, Boim MA. Mesenchymal stem cells (MSC) prevented the progression of renovascular hypertension, improved renal function and architecture. PLoS One. 2013;8(11) doi: 10.1371/journal.pone.0078464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gregorini M, Corradetti V, Rocca C, Pattonieri EF, Valsania T, Milanesi S, Serpieri N, Bedino G, Esposito P, Libetta C, et al. Mesenchymal stromal cells prevent renal fibrosis in a rat model of unilateral ureteral obstruction by suppressing the renin-angiotensin system via HuR. PLoS One. 2016;11(2) doi: 10.1371/journal.pone.0148542. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Method of kidney ultrasound imaging. DTPA kidney scan method. Cell parameters for the study patients. Table S2. Safety assessment of the mesenchymal stromal cell (MSC) infusion in 1 year following baseline visit of study patients. Table S3. Laboratory parameters at baseline, 1, 3, 6, 9, and 12 months of follow-up. Table S4. Changes in renal function parameters and blood pressure (BP) 1 year before, and at 1, 3, 6, 9, and 12 months after mesenchymal stromal cell (MSC) infusion. (DOCX 70 kb)
Characterization of the patients’ bone mesenchymal stromal cells (MSCs). (DOCX 781 kb)
Data Availability Statement
The data that support the findings of this study are available from the enrolled patients but restrictions apply to the availability of these data, which were used under license for the current study. Hence, they are not publicly available. However, the data are available from the authors upon reasonable request and with permission of the enrolled patients.


