Abstract
Apolipoprotein CIII (apoCIII) has been reported to be tightly associated with triglyceride metabolism and the susceptibility to coronary artery disease (CAD). Besides, apoCIII has also been found to affect the anti-apoptotic effects of HDL. However, the effect of apoCIII on HDL-mediated cholesterol efflux, the crucial function of HDL, has not been reported. A hospital-based case-control study was conducted to compare the apoCIII distribution in lipoproteins between CAD patients and nonCAD controls and to explore the relationship between HDL-associated apoCIII (apoCIIIHDL) and HDL-mediated cholesterol efflux. One hundred forty CAD patients and nighty nine nonCAD controls were included. Plasma apoCIII, apoCIIIHDL and cholesterol efflux capacity was measured. The apoCIIIHDL ratio (apoCIIIHDL over plasma apoCIII) was significantly higher in CAD patients than that in control group (0.52 ± 0.24 vs. 0.43 ± 0.22, P = 0.004). Both apoCIIIHDL and apoCIIIHDL ratio were inversely correlated with cholesterol efflux capacity (r = −0.241, P = 0.0002; r = −0.318, P < 0.0001, respectively). Stepwise multiple regression analysis revealed that the apoCIIIHDL ratio was an independent contributor to HDL-mediated cholesterol efflux capacity (standardized β = −0.325, P < 0.001). This study indicates that the presence of apoCIII in HDL may affect HDL-mediated cholesterol efflux capacity, implying the alternative role of apoCIII in the atherogenesis.
Introduction
Many epidemiological studies demonstrated that high density lipoprotein (HDL)-cholesterol (HDL-C) was reduced in patients with coronary artery disease (CAD) and that HDL-C level was inversely correlated with CAD incidence1, 2, which led to the development of raising HDL-C as a therapeutic approach to prevent CAD. However, results from randomized controlled trails3, 4 and Mendelian randomization studies5 failed to prove the protective effects of HDL-C. Scientific interest has been gradually shifted from raising HDL-C to improving HDL function. HDL protects against CAD via multiple mechanisms and the most important one is reverse cholesterol transport (RCT)6. Previous study showed that cholesterol efflux from macrophages, a crucial step of RCT, had a strong inverse association with carotid intima-media thickness and CAD likelihood independent of HDL-C level7. Our observation also confirmed the reverse correlation between cholesterol efflux and carotid intima-media thickness in patients with chronic kidney diseases8. Recent prospective cohort study revealed that HDL-mediated cholesterol efflux capacity, instead of HDL-C level, was an independent predictor of CAD incidence9. Unraveling the molecular determinants of HDL-mediated cholesterol efflux capacity may help the development of therapeutic interventions to improve HDL function10. Our previous work showed that tryptophan oxidation in apoAI was responsible for the myeloperoxidase-mediated loss of apoAI function and impairment of cholesterol efflux capacity11. Animal experiments also revealed that increased proinflammatory protein components in HDL, such as serum amyloid A (SAA), attenuated HDL cholesterol efflux capacity and that SAA genetic ablation restored HDL function12, 13.
Apolipoprotein CIII (apoCIII), a critical modulator of triglyceride metabolism, mainly presents on HDL particles and triglyceride-rich lipoproteins (TRLs) including chylomicron (CM) and very low-density lipoprotein (VLDL)14. Genetic studies revealed that loss-of-function (LOF) mutations in the apoCIII gene were associated with reduced triglyceride concentration and decreased CAD incidence15, 16. Another prospective cohort study showed that the percentage of apoCIII-containing HDL particle was correlated with CAD risk17. In vitro experiments suggested that the deterioration of the HDL anti-apoptotic effects in CAD patients was associated with HDL proteome remodeling including apoCIII increase in HDL particles18. However, the effect of apoCIII on HDL-mediated cholesterol efflux has not been reported. In this study, we found that apoCIIIHDL and the apoCIIIHDL ratio were increased in CAD patients compared to controls and that the apoCIIIHDL ratio was an independent contributor to HDL-mediated cholesterol efflux capacity. These findings indicate that apoCIII redistribution may result in HDL dysfunction and CAD progression.
Results
Characteristics of subjects
Demographic and biochemical characteristics of participants were shown in Table 1. FFA and hsCRP were significantly higher, while TC and HDL-C, LDL-C, apoAI were significantly lower in CAD patients compared to nonCAD controls. The percentage of diabetes, statin therapy and smoking was significantly higher in CAD group with comparison to the controls. Other parameters had no statistically significant differences between two groups.
Table 1.
Baseline characteristics of all the subjects.
| Variables | CAD (n = 140) | nonCAD (n = 99) | P |
|---|---|---|---|
| Male (%) | 66.4 | 54.5 | NS |
| Age (years) | 63.10 ± 8.42 | 62.81 ± 8.01 | NS |
| BMI (kg/m2) | 24.43 ± 3.72 | 24.12 ± 3.02 | NS |
| TG (mmol/L) | 1.49 ± 0.87 | 1.47 ± 0.88 | NS |
| TC (mmol/L) | 3.75 ± 0.92 | 4.16 ± 1.02 | 0.002 |
| HDL-C (mmol/L) | 0.99 (0.85–1.19) | 1.16 (0.91–1.33) | 0.002 |
| LDL-C (mmol/L) | 2.27 ± 0.76 | 2.56 ± 0.81 | 0.006 |
| apoAI (g/L) | 1.08 ± 0.21 | 1.17 ± 0.24 | 0.002 |
| apoB (g/L) | 0.84 ± 0.25 | 0.89 ± 0.26 | NS |
| hsCRP (mg/L) | 4.44 (1.21–12.33) | 2.27 (0.96–6.62) | 0.032 |
| FFA (mmol/L) | 0.46 (0.31–0.69) | 0.40 (0.25–0.56) | 0.014 |
| Diabetes (%) | 24.29 | 7.07 | 0.0004 |
| Statin (%) | 59.29 | 11.11 | <0.0001 |
| Smoking (%) | 47.86 | 28.28 | 0.003 |
| Drinking (%) | 25.71 | 15.15 | NS |
Values are expressed as mean ± SD or median (interquartile range). CAD indicates coronary artery disease; BMI, body mass index; TG, triglyceride; TC, total cholesterol; HDL-C, high density lipoprotein-cholesterol; LDL-C, low density lipoprotein-cholesterol; apoAI, apolipoprotein AI; apoB, apolipoprotein B; hsCRP, high sensitivity C reactive protein; FFA, free fatty acid.
ApoCIII levels in CAD group
Plasma apoCIII and apoCIIIHDL were measured via ELISA method. No significant difference was seen in plasma apoCIII between CAD group [9.40 (6.56, 12.28) mg/dl] and non-CAD group [9.42 (6.80, 13.86) mg/dl, P = 0.575]. Both apoCIIIHDL and the apoCIIIHDL ratio, the quotient of apoCIIIHDL over plasma apoCIII, were significantly higher in CAD group than that in nonCAD group [apoCIIIHDL: 4.42 (3.12, 6.13) mg/dl vs. 3.59 (2.48, 5.26) mg/dl, P = 0.007, Fig. 1; the apoCIIIHDL ratio: 0.52 ± 0.24 vs. 0.43 ± 0.22, P = 0.004, Fig. 2]. No significant differences in plasma apoCIII, apoCIIIHDL and apoCIIIHDL ratio were observed between statin users and non-statin users, diabetes patients and non-diabetes patients, smokers and non-smokers (Table 2).
Figure 1.
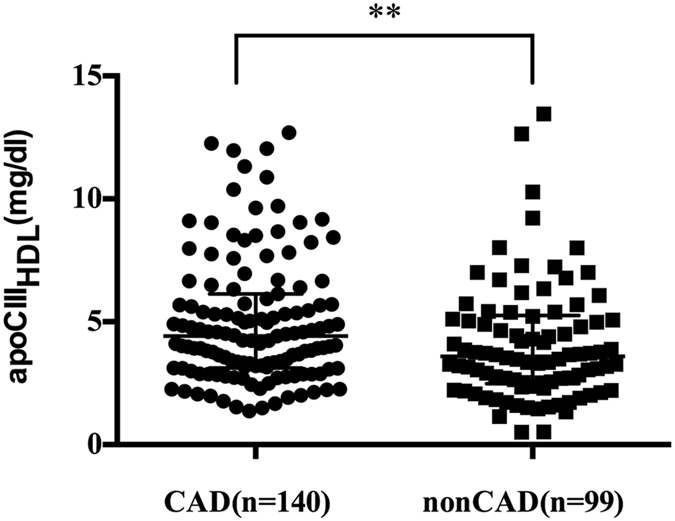
ApoCIIIHDL from CAD (n = 140) and nonCAD (n = 99) patients. Data are expressed as median ± interquartile range. ApoCIIIHDL indicates apolipoprotein CIII in apoB-depleted plasma; apoB, apolipoprotein B; CAD, coronary artery disease. **P < 0.01.
Figure 2.
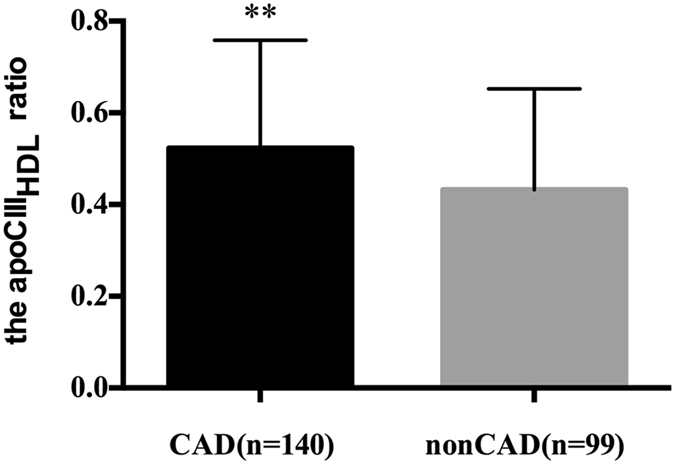
The apoCIIIHDL ratio (apoCIIIHDL over plasma apoCIII) between CAD (n = 140) and nonCAD (n = 99) patients. Data are expressed as mean ± SD. ApoCIIIHDL indicates apolipoprotein CIII in apoB-depleted plasma; apoB, apolipoprotein B; CAD, coronary artery disease. **P < 0.01.
Table 2.
Comparison of apoCIII concentration and HDL-mediated cholesterol efflux capacity between different groups.
| DM | Statin | Smoking | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Yes | No | P | Yes | No | P | Yes | No | P | |
| Plasma apoCIII (mg/dl) | 10.61 (7.73–12.95) | 9.20 (6.34–13.08) | 0.21 | 9.60 (6.77–12.93) | 9.04 (6.53–13.50) | 0.44 | 9.20 (6.62–14.59) | 9.46 (6.56–12.86) | 0.99 |
| apoCIIIHDL (mg/dl) | 4.87 (3.07–5.92) | 3.92 (2.76–5.69) | 0.21 | 4.56 (3.33–6.61) | 3.65 (2.60–5.35) | 0.08 | 3.79 (2.74–5.69) | 4.15 (3.04–5.69) | 0.32 |
| apoCIIIHDL ratio | 0.474 ± 0.189 | 0.488 ± 0.241 | 0.98 | 0.523 ± 0.256 | 0.461 ± 0.213 | 0.15 | 0.462 ± 0.226 | 0.501 ± 0.237 | 0.12 |
| CEC (%) | 12.2 ± 3.2 | 13.6 ± 2.6 | 0.02 | 12.8 ± 2.9 | 13.8 ± 2.6 | 0.03 | 13.3 ± 2.9 | 13.5 ± 2.6 | 0.60 |
DM indicates diabetes mellitus; apoCIII, apolipoprotein C-III; apoCIIIHDL, apolipoprotein CIII in apoB-depleted plasma; CEC, cholesterol efflux capacity.
Correlation analysis of apoCIII and triglyceride
To examine the relationship between apoCIII and triglyceride, correlation analyses were performed. As previously mentioned, plasma apoCIII and apoCIIIHDL were log-transformed to be normally distributed. The results showed that plasma apoCIII concentration was positively associated with plasma triglyceride levels (r = 0.330, P < 0.0001, Fig. 3). Within-group analysis showed that the correlation between plasma apoCIII and triglyceride remained significant in CAD and nonCAD groups (Table 3). However, no significant relationship was found between apoCIIIHDL (log transformed) and plasma triglyceride in within-group analysis or combined analysis (Table 4).
Figure 3.
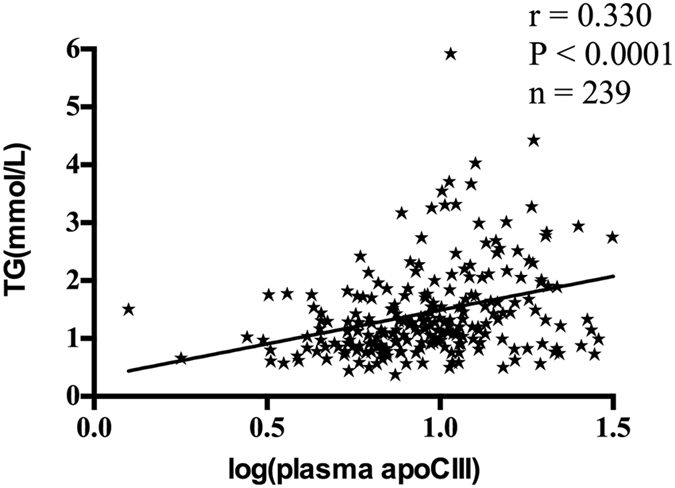
The correlation between plasma apoCIII (log transformed) and triglyceride in all the subjects (n = 239). ApoCIII indicates apolipoprotein CIII; TG, triglyceride.
Table 3.
Pearson’s correlation analyses between log-transformed plasma apoCIII and clinical and biochemical parameters.
| All subjects (n = 239) | CAD (n = 140) | nonCAD (n = 99) | ||||
|---|---|---|---|---|---|---|
| r | P | r | P | r | P | |
| TG | 0.330 | <0.0001 | 0.371 | <0.0001 | 0.276 | 0.008 |
| TC | 0.208 | 0.002 | 0.261 | 0.002 | 0.136 | 0.193 |
| LDL-C | 0.207 | 0.002 | 0.264 | 0.002 | 0.127 | 0.224 |
| apoB | 0.249 | 0.0002 | 0.282 | 0.001 | 0.195 | 0.065 |
| hsCRP (log-transformed) | 0.059 | 0.384 | 0.016 | 0.853 | 0.136 | 0.198 |
CAD indicates coronary artery disease; TG, triglyceride; TC, total cholesterol; LDL-C, low-density lipoprotein-cholesterol; apoB, apolipoprotein B; hsCRP, high-sensitivity C reactive protein.
Table 4.
Pearson’s correlation analyses between log-transformed apoCIIIHDL and clinical and biochemical parameters.
| All subjects (n = 239) | CAD (n = 140) | nonCAD (n = 99) | ||||
|---|---|---|---|---|---|---|
| r | P | r | P | r | P | |
| plasma apoCIII (log-transformed) | 0.607 | <0.0001 | 0.688 | <0.0001 | 0.555 | <0.0001 |
| TG | −0.025 | 0.705 | 0.103 | 0.234 | −0.064 | 0.542 |
| CEC | −0.241 | 0.0002 | −0.158 | 0.068 | −0.247 | 0.018 |
| CK | 0.163 | 0.022 | 0.214 | 0.021 | −0.077 | 0.495 |
| UA | 0.186 | 0.005 | 0.224 | 0.01 | 0.202 | 0.051 |
| HDL-C (log-transformed) | 0.083 | 0.214 | 0.042 | 0.627 | 0.199 | 0.055 |
| hsCRP (log-transformed) | 0.041 | 0.547 | −0.097 | 0.273 | 0.158 | 0.135 |
CAD indicates coronary artery disease; apoCIII, apolipoprotein CIII; TG, triglyceride; CEC, cholesterol efflux capacity; CK, Creatine phosphokinase; UA, uric acid; HDL-C, high density lipoprotein-cholesterol; hsCRP, high sensitivity C reactive protein.
Correlation analysis of apoCIII and other parameters associated with CAD
Plasma apoCIII (log transformed) was positively correlated with TC (r = 0.208, P = 0.002) and LDL-C (r = 0.207, P = 0.002) and apoB (r = 0.249, P = 0.0002) in all the subjects (Table 3). The correlation remained significant in patients with CAD but not in nonCAD subjects (Table 3). ApoCIIIHDL (log transformed) was positively correlated with plasma apoCIII (r = 0.607, P < 0.0001), but negatively correlated with cholesterol efflux capacity (r = −0.241, P = 0.0002) in all the subjects (Table 4). In order to investigate the relationship between apoCIII and inflammation, correlation analysis of apoCIII to hsCRP, a typical inflammation marker, was performed. The results showed that plasma apoCIII, apoCIIIHDL and the apoCIIIHDL ratio did not have significant relationship with hsCRP (Tables 3 and 4).
HDL-mediated Cholesterol efflux capacity
In order to measure HDL-mediated cholesterol efflux capacity, apoB-depleted plasma obtained by heparin-manganese method was adopted. Cholesterol efflux capacity was significantly lower in CAD patients than that in nonCAD patients (12.5 ± 2.8% vs. 14.7 ± 2.0%, P < 0.0001, Fig. 4). To investigate the impacts of statin use, smoking and diabetes status on the cholesterol efflux capacity, subjects were divided into groups accordingly. We found that cholesterol efflux capacity in subjects without diabetes was significantly higher than that in subjects with diabetes (13.6 ± 2.6% vs. 12.2 ± 3.2%, P = 0.021, Table 2). Besides, non-statin users also present significantly higher cholesterol efflux capacity than statin users (13.8 ± 2.6% vs. 12.8 ± 2.9%, P = 0.026, Table 2). However, smoking had no effects on cholesterol efflux capacity (smokers vs. non-smokers 13.3 ± 2.9% vs. 13.5 ± 2.6%, P = 0.597, Table 2).
Figure 4.
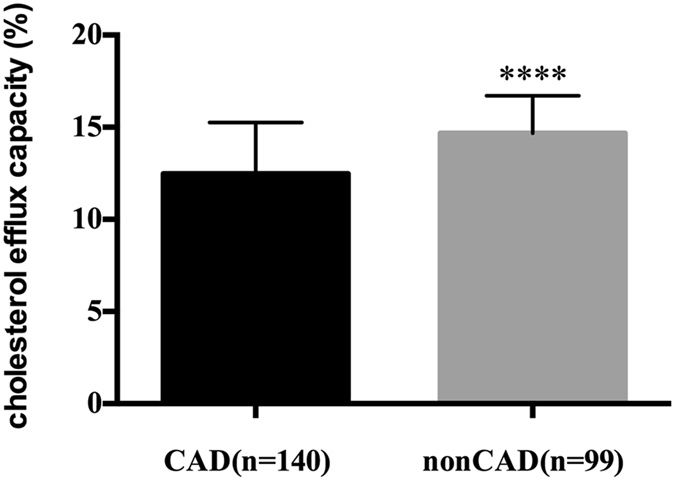
Cholesterol efflux capacity in CAD (n = 140) and nonCAD (n = 99) patients. Data are expressed as mean ± SD. CAD indicates coronary artery disease. ****P < 0.0001.
Relationship between apoCIII and cholesterol efflux capacity
No research has been reported to investigate the relationship between HDL-associated apoCIII and HDL-mediated cholesterol efflux capacity. In our research, cholesterol efflux capacity was inversely correlated with BMI (r = −0.147, P = 0.028), log-transformed apoCIIIHDL (r = −0.241, P = 0.0002), the apoCIIIHDL ratio (r = −0.318, P < 0.0001, Fig. 5) and log-transformed hsCRP (r = −0.248, P = 0.0002). The positive correlation with HDL-C (r = 0.443, P < 0.0001) and apoAI (r = 0.541, P < 0.0001) has been validated (Table 5). In order to exclude confounding effects of HDL-C on the correlation of HDL-mediated cholesterol efflux capacity to apoCIIIHDL and the apoCIIIHDL ratio, partial correlation analysis was performed after controlling HDL-C level. The inverse correlation remained significant for apoCIIIHDL (r = −0.314, P < 0.001) and for apoCIIIHDL ratio (r = −0.423, P < 0.001). Besides, in order to check for the effects of inflammation, partial correlation analysis was conducted after controlling hsCRP. HDL-mediated cholesterol efflux capacity was still inversely correlated with apoCIIIHDL (r = −0.230, P = 0.001) and for apoCIIIHDL ratio (r = −0.348, P < 0.001) after controlling hsCRP.
Figure 5.
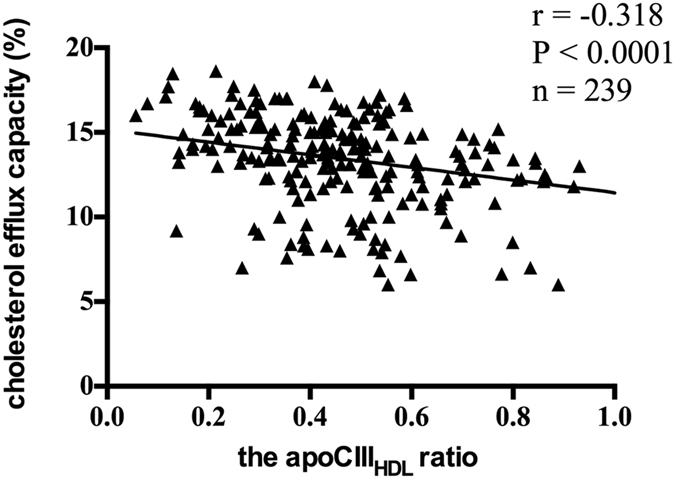
The correlation of the apoCIIIHDL ratio and cholesterol efflux capacity in all the subjects (n = 239). ApoCIIIHDL indicates apolipoprotein CIII in apoB-depleted plasma.
Table 5.
Pearson’s correlation analyses between HDL-mediated cholesterol efflux capacity and clinical and biochemical parameters.
| All subjects (n = 239) | CAD (n = 140) | nonCAD (n = 99) | ||||
|---|---|---|---|---|---|---|
| r | P | r | P | r | P | |
| BMI | −0.147 | 0.028 | −0.160 | 0.064 | −0.103 | 0.328 |
| plasma apoCIII (log-transformed) | 0.069 | 0.302 | 0.023 | 0.791 | 0.134 | 0.204 |
| apoCIIIHDL (log-transformed) | −0.241 | 0.0002 | −0.158 | 0.068 | −0.247 | 0.018 |
| apoCIIIHDL ratio | −0.318 | <0.0001 | −0.199 | 0.021 | −0.409 | <0.0001 |
| hsCRP (log-transformed) | −0.248 | 0.0002 | −0.205 | 0.019 | −0.237 | 0.025 |
| apoAI | 0.541 | <0.0001 | 0.585 | <0.0001 | 0.442 | <0.0001 |
| HDL-C (log-transformed) | 0.443 | <0.0001 | 0.448 | <0.0001 | 0.373 | 0.0003 |
CAD indicates coronary artery disease; BMI, body mass index; apoCIII, apolipoprotein CIII; hsCRP, high sensitivity C reactive protein; apoAI, apolipoprotein AI; HDL-C, high-density lipoprotein-cholesterol.
Multiple regression analysis
In addition to HDL-C and hsCRP, other factors may also confound the relationship between apoCIII distribution in HDL and HDL-mediated cholesterol efflux. A stepwise multiple regression model was fitted after adjustment for diabetes status, statin use and the variables that were significantly correlated with HDL-mediated cholesterol efflux capacity in univariate analysis, including BMI, apoCIIIHDL ratio, hsCRP, apoAI and HDL-C. Log-transformed values were used for the variables skewed distributed, including hsCRP and HDL-C. The regression analysis revealed that the apoCIIIHDL ratio, apoAI and diabetes status were independent risk factors for HDL-mediated cholesterol efflux capacity (Table 6).
Table 6.
Stepwise multiple regression analysis detecting independent contributors to HDL-mediated cholesterol efflux capacity in all the subjects.
| Factors | β | Standardized β | P |
|---|---|---|---|
| BMI | <0.001 | −0.058 | 0.274 |
| apoCIIIHDL ratio | −0.038 | −0.325 | <0.001 |
| apoAI | 0.067 | 0.558 | <0.001 |
| hsCRP (log-transformed) | −0.002 | −0.053 | 0.348 |
| HDL-C (log-transformed) | −0.016 | −0.065 | 0.541 |
| Diabetes | −0.012 | −0.159 | 0.003 |
| Statin | −0.005 | −0.097 | 0.065 |
BMI indicates body mass index; apoCIIIHDL indicates apolipoprotein CIII in apoB-depleted plasma; apoAI, apolipoprotein AI; hsCRP, high sensitivity C reactive protein; HDL-C, high density lipoprotein-cholesterol.
R square: 0.452, P < 0.001.
Discussion
In this study, we found that apoCIIIHDL and the apoCIIIHDL ratio were significantly higher in CAD patients compared to nonCAD controls. Besides, they were inversely correlated with HDL-mediated cholesterol efflux capacity. Moreover, increased apoCIIIHDL ratio was an independent contributor to impaired HDL-mediated cholesterol efflux capacity.
Our experiments showed that plasma apoCIII was significantly correlated with triglyceride, which was consistent with the established role of apoCIII in triglyceride metabolism14. Recent genetic epidemiological studies found that LOF mutation in apoCIII gene was associated with decreased triglyceride level and reduced CAD risk15, 16. On the other hand, gain of function mutant of apoCIII gene, T-455C variant, was found to be associated with elevated apoCIII and triglyceride levels and increased CAD probability in metabolic syndrome (MS) patients19. However, disappointing results from randomized controlled trials of fenofibrate and omega-3 fatty acids challenged the role of triglyceride in CAD risk20, 21. These controversial findings gave rise to the hypothesis that triglyceride-related effects on CAD incidence might be mediated by the direct atherogenic effects of apoCIII22. However, our results did not show significant difference in plasma apoCIII between CAD and nonCAD group, but apoCIII in HDL particles was significantly increased in CAD patients compared to controls, which was in accordance with previous studies23, 24. These findings indicated that apoCIII distribution in lipoproteins, instead of total apoCIII concentration, might paly an important role in CAD propensity. In circulation, apoCIII mainly resides on the surface of lipoproteins and apoCIII is transferable between TRLs and HDL14, 23. In our study, the apoCIIIHDL ratio was significantly higher in CAD group than that in nonCAD group, which indicated that more apoCIII was transferred from apoB-containing lipoproteins to HDL in CAD patients. Previous study revealed that two HDL subtypes, HDL with apoCIII and HDL without apoCIII, were oppositely associated with CAD risk17. Besides, per standard deviation increase of apoCIII-containing HDL particle was associated with an 18% higher risk for CAD17. But the mechanism by which apoCIII damaged the protective effects of HDL on CAD risk was still not clearly defined. Current study suggested that accumulation of apoCIII in HDL particles might impair HDL-mediated cholesterol efflux capacity, the crucial athero-protective effect of HDL. HDL anti-apoptotic property was also attenuated by the presence of apoCIII in HDL as demonstrated by Riwanto et al.18. Being a reasonable index of relative enrichment of apoCIII in HDL, the apoCIIIHDL ratio might be regarded as a significant biomarker for HDL dysfunction and CAD susceptibility. Results from Chin-Shan Community Cardiovascular Cohort Study suggested that HDL-apoCIII to VLDL-apoCIII ratio was a reliable marker to predict CAD23, which was in agreement with our findings. But the measurement of HDL-apoCIII to VLDL-apoCIII ratio involved sophisticated lipoprotein isolation and therefore it was less clinically applicable.
Inflammation was related to the alteration of HDL function. Our early work showed that myeloperoxidase-mediated oxidation could modify tryptophan in apoAI and thus impair the cholesterol acceptor activity of apoAI11. In addition to the apoAI modification, HDL might be subjected to structure remodeling following inflammation. For example, increased SAA was found in HDL particle in response to the acute inflammation stress12, 13. Increased SAA in HDL could interact with proteoglycans on the cell surface and lead to HDL trapping in the extracellular matrix, thereby disrupting HDL-mediated cholesterol efflux12, 13. ApoCIII was reported to be involved in inflammation response. Increasing apoCIII content was accompanied by increasing apoCIII sialylation and apoCIII sialylation further promoted the proinflammatory properties of apoCIII25. ApoCIII could induce the adhesion of monocytes to endothelial cells and enhance the pro-inflammatory properties of VLDL and LDL particles26, 27. It was still unclear how the enrichment of apoCIII in HDL affected HDL-mediated cholesterol efflux. In this study, the correlation between apoCIIIHDL ratio and HDL-mediated cholesterol efflux remained significant after controlling for CRP, a marker of inflammation, suggesting that the effect of apoCIII on HDL-mediated cholesterol efflux might be independent of its pro-inflammatory effects.
Cholesterol efflux is a process in which apoAI or HDL accepts cholesterol from peripheral cells via several membrane transporters: ATP-binding cassette A1 (ABCA1), ATP-binding cassette G1 (ABCG1) and scavenger receptor family B type I (SR-BI)28. ApoAI accepts cellular cholesterol and phospholipids transported by ABCA1 to form nascent HDL particles29. HDL particles depend on ABCG1 and SR-BI to accept peripheral cellular cholesterol and mediate cholesterol efflux. SR-BI can mediate bi-directional cholesterol flux, including cholesterol efflux from peripheral cells to HDL and cholesterol uptake from HDL by hepatocytes30. ApoCIII can bind to hepatic SR-BI and inhibit hepatocytes to intake cholesterol from HDL and LDL14, 31. Based on these findings, we hypothesized that elevated apoCIII in HDL could competitively bind to SR-BI and interrupt the cholesterol unloading from HDL, thereby impairing HDL-mediated cholesterol efflux.
In the current study, patients with diabetes exhibited significantly lower cholesterol efflux capacity compared to non-diabetic subjects. The impairment of HDL-mediated cholesterol efflux capacity was potentially related to the increased HDL-apoCIII level in diabetic patients, although the increase of HDL-apoCIII did not reach statistical significance in the study. But more evidence was needed to elucidate the definite role of HDL-apoCIII in the impairment of cholesterol efflux capacity in diabetic subjects. Other factors have been reported to contribute to the reduced cholesterol efflux, including the increased apoAI nitrosylation32 and elevated SAA concentration33 in HDL particles from DM patients. Another interesting finding was that statin users had significantly lower cholesterol efflux capacity compared to the non-statin users. This phenomenon might be confounded by the higher CAD prevalence in statin users (88.3%) compared to that in non-statin users (39.3%) as CAD patients usually exhibited impaired cholesterol efflux capacity. In addition, previous study showed that HDL-apoCIII was elevated after statin treatment in CAD patients24 and this might help explain the reduced cholesterol efflux capacity in statin users. However, the effects of statin therapy on HDL-mediated cholesterol efflux were inconsistent in different researches28. Concerning the positive correlation of HDL-apoCIII and uric acid, no clear evidence has been found. The detailed effects of uric acid on apoCIII distribution and CAD progression still need more investigation.
To the best of our knowledge, no experiments have been conducted to observe the relationship between HDL-associated apoCIII and HDL-mediated cholesterol efflux capacity in the CAD progression. In the current study, we showed that the apoCIIIHDL ratio was an independent contributor to HDL-mediated cholesterol efflux capacity, which provided evidence for the role of apoCIII in HDL dysfunction and a possible explanation for the direct atherogenic effects of apoCIII. However, apoCIII may be just a bystander of other constituents remodeling of HDL in CAD progression. Whether the correlation of HDL-associated apoCIII and HDL dysfunction is causative is still not clear. Further in vitro and animal studies are warranted to investigate the causal relationship between apoCIII and HDL-mediated cholesterol efflux.
Methods
Subjects
We recruited 239 subjects from the Department of Cardiovascular Medicine of Second Xiangya Hospital, Central South University. To evaluate the relationship of HDL-apoCIII with cholesterol efflux capacity, we studied 2 groups: CAD group (n = 140) and nonCAD group (n = 99). In this study, CAD meant acute coronary syndrome (ACS). ACS included ST-segment elevated myocardial infarction (STEMI), non ST-segment elevated myocardial infarction (NSTEMI) and unstable angina. ACS diagnosis was based on the clinical symptoms and signs, ischemic electrocardiographic abnormalities, and coronary angiography showing ≥ 50% stenosis in at least one main coronary artery. The exclusion criteria included: a history of renal failure, chronic hepatic diseases, high fever, or bacterial/viral infection, autoimmune disease, arthritis, malignancies, severe diabetes and hypertension, and other severe medical illnesses. All the subjects provided written informed consent. The study was approved by the Medical Ethics Committee of the Xiangya Second Hospital of Central South University and was conducted in accordance with approved guidelines and regulations.
Clinical and biochemical measurements
Patient information, including age, gender, smoking and drinking history, and statin therapy history, was recorded. The details of anthropometric measurements (weight, height, body mass index) were assessed after overnight fasting for at least 10 hours. Peripheral blood samples were obtained from patients’ brachial veins. Subjects fasted for at least 10 hours before blood collection and then blood routine, urine routine, concentrations of lipid parameters, including total cholesterol (TC), triglyceride (TG), low-density lipoprotein cholesterol (LDL-C), HDL-C, apoAI, apoB, free fatty acid (FFA), were evaluated via standard laboratory procedures. Concentrations of high-sensitivity C-reactive protein (hsCRP) were measured with a latex particle, enhanced immunoturbidimetric assay. For the subsequent experiments, fresh plasma was obtained by centrifugation at 3000 r/min at 4 °C for 10 minutes. The plasma was aliquoted and stored at −80 °C freezer until analysis.
ApoB-depleted plasma preparation
According to previous experiments34, 540 ul heparin sodium solution (280 mg/ml, Aladdin, H104201) and 10 ml manganese chloride solution (1.06 mol/L, Aladdin, M112542) was mixed. 1 ml plasma was incubated for 30 minutes at 4 °C with 100 ul mixed solution, and then centrifuged at 1500 g for 30 minutes. Supernatant was collected. If supernatant was still turbid (especially samples from patients with hypertriglyceridemia), plasma was centrifuged at 12000 g for 10 minutes again. Previous study revealed that heparin sodium/manganese chloride precipitation had no effects on HDL size as well as cholesterol efflux measurement35, and therefore this method was chosen to prepare apoB-depleted plasma in the study.
Measurement of plasma apoCIII and apoCIIIHDL
Plasma apoCIII and apoCIIIHDL was determined by a sandwich enzyme-linked immunosorbent assay (ELISA). ApoCIIIHDL was referred to as apoCIII in apoB-depleted plasma according to previous literature19, 36. Commercial ApoCIII ELISA kits (Abcam, ab154131) were used to quantify the concentrations. All the measurement of plasma apoCIII and apoCIIIHDL were performed in duplicate for each sample. The coefficient of variation for intra- and inter-assay variation was 2.9% and 3.4%, respectively.
Cholesterol efflux capacity measurement
Cholesterol efflux experiments were performed according to established procedures11, 37. THP-1 human monocytes (ATCC) were grown in RPMI1640 medium (Gibco, 22400089), supplemented with 10% heat-inactivated FBS, 1% penicillin/streptomycin until differentiation into macrophages by the addition of phorbol myristate acetate (100 ng/ml, Sigma, P1585). Subsequently, differentiated THP-1 macrophages were loaded with 50 ug/mL acetylated LDL (Peking Union-Biology Co.Ltd) and 1 uCi/mL [3 H] cholesterol for 24 hours. Macrophages were then washed twice with PBS (Gibco, 10010023) and equilibrated for 24 hours in RPMI1640 medium containing 2% bovine serum albumin. Cells were then washed with PBS again and apoB-depleted plasma from individual patients was diluted in medium (2.5%, vol/vol). After 16 hours, the supernatant was collected and centrifuged to remove cellular debris. The cells were washed twice with PBS, and then incubated for at least 30 min at room temperature with 0.1 mol/L NaOH solution. The radioactivity within the supernatant and cells was determined by liquid scintillation counting. Wells incubated with RPMI1640 but without added apoB-depleted plasma were used as blanks, and these values were subtracted from the respective experimental values. Efflux is given as the percentage of counts recovered from the medium in relation to the total counts present on the plate (sum of medium and cells). All efflux experiments were performed in duplicate for each sample with intra- and inter-assay coefficients of variation of 4.1% and 7.9%, respectively.
Statistical analysis
Statistical analysis was performed with Statistical Package for Social Sciences version 22.0 and plots were made with GraphPad Prism V.6.0 (GraphPad Software, Inc, La Jolla, California, USA). Clinical data are expressed as mean ± standard deviation (normally distributed continuous data) or median with interquartile range (skewed distributed continuous data). Comparisons between categorical data were performed with Chi Squared tests, while continuous variables were assessed by unpaired t test (for normal distribution) or nonparametric test (for skewed distribution). To evaluate the associations between variables, Pearson correlation analysis was used. Stepwise multiple linear regression analysis was performed to determine the variables with independent significant association with cholesterol efflux capacity. These variables included all potential ones that might have significant relationship with cholesterol efflux capacity in univariate analyses. In the correlation and regression analysis, logarithmatic-transformed values were used for the variables skewed distributed. A two tailed P value < 0.05 was considered statistically significant.
Acknowledgements
This project was supported by grants from National Natural Science Foundation of China (No. 81370393 and No. 81670426 to D.P.); supported by the Fundamental Research Funds for the Central Universities of Central South University (No. 2016zzts545 to M.L.).
Author Contributions
Conception and design: D.P. and M.L.; Experiments performance: M.L., A.L., T.W. and D.H.; Analysis and interpretation of the data: M.L. and D.P.; Article drafting and revision: M.L., D.P., S. Wang. and S. Wu. All authors reviewed the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Emerging Risk Factors C, et al. Major lipids, apolipoproteins, and risk of vascular disease. JAMA. 2009;302:1993–2000. doi: 10.1001/jama.2009.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boekholdt SM, et al. Levels and changes of HDL cholesterol and apolipoprotein A-I in relation to risk of cardiovascular events among statin-treated patients: a meta-analysis. Circulation. 2013;128:1504–1512. doi: 10.1161/CIRCULATIONAHA.113.002670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Investigators A-H, et al. Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy. N Engl J Med. 2011;365:2255–2267. doi: 10.1056/NEJMoa1107579. [DOI] [PubMed] [Google Scholar]
- 4.Keene D, Price C, Shun-Shin MJ, Francis DP. Effect on cardiovascular risk of high density lipoprotein targeted drug treatments niacin, fibrates, and CETP inhibitors: meta-analysis of randomised controlled trials including 117411 patients. BMJ. 2014;349:g4379–g4379. doi: 10.1136/bmj.g4379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jansen H, Samani NJ, Schunkert H. Mendelian randomization studies in coronary artery disease. Eur Heart J. 2014;35:1917–1924. doi: 10.1093/eurheartj/ehu208. [DOI] [PubMed] [Google Scholar]
- 6.Rosenson RS, et al. Cholesterol efflux and atheroprotection: advancing the concept of reverse cholesterol transport. Circulation. 2012;125:1905–1919. doi: 10.1161/CIRCULATIONAHA.111.066589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Khera AV, et al. Cholesterol efflux capacity, high-density lipoprotein function, and atherosclerosis. N Engl J Med. 2011;364:127–135. doi: 10.1056/NEJMoa1001689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wu S, et al. Impaired cholesterol efflux capacity is related to increased carotid intima media thickness in patients with end-stage renal disease. Int J Cardiol. 2015;187:456–458. doi: 10.1016/j.ijcard.2015.03.362. [DOI] [PubMed] [Google Scholar]
- 9.Rohatgi A, et al. HDL cholesterol efflux capacity and incident cardiovascular events. N Engl J Med. 2014;371:2383–2393. doi: 10.1056/NEJMoa1409065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ronsein GE, Vaisar T. Inflammation, remodeling, and other factors affecting HDL cholesterol efflux. Curr Opin Lipidol. 2017;28:52–59. doi: 10.1097/MOL.0000000000000382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Peng DQ, et al. Apolipoprotein A-I tryptophan substitution leads to resistance to myeloperoxidase-mediated loss of function. Arterioscler Thromb Vasc Biol. 2008;28:2063–2070. doi: 10.1161/ATVBAHA.108.173815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Vaisar T, et al. Inflammatory remodeling of the HDL proteome impairs cholesterol efflux capacity. J Lipid Res. 2015;56:1519–1530. doi: 10.1194/jlr.M059089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Han CY, et al. Serum amyloid A impairs the antiinflammatory properties of HDL. J Clin Invest. 2016;126:266–281. doi: 10.1172/JCI83475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Norata GD, Tsimikas S, Pirillo A, Catapano AL. Apolipoprotein C-III: From Pathophysiology to Pharmacology. Trends Pharmacol Sci. 2015;36:675–687. doi: 10.1016/j.tips.2015.07.001. [DOI] [PubMed] [Google Scholar]
- 15.Tg, et al. Loss-of-function mutations in APOC3, triglycerides, and coronary disease. N Engl J Med. 2014;371:22–31. doi: 10.1056/NEJMoa1307095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jorgensen AB, Frikke-Schmidt R, Nordestgaard BG, Tybjaerg-Hansen A. Loss-of-function mutations in APOC3 and risk of ischemic vascular disease. N Engl J Med. 2014;371:32–41. doi: 10.1056/NEJMoa1308027. [DOI] [PubMed] [Google Scholar]
- 17.Jensen MK, Rimm EB, Furtado JD, Sacks FM. Apolipoprotein C-III as a Potential Modulator of the Association Between HDL-Cholesterol and Incident Coronary Heart Disease. J Am Heart Assoc. 2012;1:jah3–e000232. doi: 10.1161/JAHA.111.000232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Riwanto M, et al. Altered activation of endothelial anti- and proapoptotic pathways by high-density lipoprotein from patients with coronary artery disease: role of high-density lipoprotein-proteome remodeling. Circulation. 2013;127:891–904. doi: 10.1161/CIRCULATIONAHA.112.108753. [DOI] [PubMed] [Google Scholar]
- 19.Olivieri O, et al. Apolipoprotein C-III, metabolic syndrome, and risk of coronary artery disease. J Lipid Res. 2003;44:2374–2381. doi: 10.1194/jlr.M300253-JLR200. [DOI] [PubMed] [Google Scholar]
- 20.Investigators OT, et al. n-3 fatty acids and cardiovascular outcomes in patients with dysglycemia. N Engl J Med. 2012;367:309–318. doi: 10.1056/NEJMoa1203859. [DOI] [PubMed] [Google Scholar]
- 21.Group AS, et al. Effects of combination lipid therapy in type 2 diabetes mellitus. N Engl J Med. 2010;362:1563–1574. doi: 10.1056/NEJMoa1001282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Luo M, Peng D. The emerging role of apolipoprotein C-III: beyond effects on triglyceride metabolism. Lipids Health Dis. 2016;15:184. doi: 10.1186/s12944-016-0352-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chang PY, et al. Identification of the HDL-ApoCIII to VLDL-ApoCIII ratio as a predictor of coronary artery disease in the general population: the Chin-Shan Community Cardiovascular Cohort (CCCC) study in Taiwan. Lipids Health Dis. 2012;11:162. doi: 10.1186/1476-511X-11-162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Xiong X, et al. The association of HDL-apoCIII with coronary heart disease and the effect of statin treatment on it. Lipids Health Dis. 2015;14:127. doi: 10.1186/s12944-015-0129-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hiukka A, et al. ApoCIII-enriched LDL in type 2 diabetes displays altered lipid composition, increased susceptibility for sphingomyelinase, and increased binding to biglycan. Diabetes. 2009;58:2018–2026. doi: 10.2337/db09-0206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kawakami A, et al. Apolipoprotein CIII-induced THP-1 cell adhesion to endothelial cells involves pertussis toxin-sensitive G protein- and protein kinase C alpha-mediated nuclear factor-kappaB activation. Arterioscler Thromb Vasc Biol. 2007;27:219–225. doi: 10.1161/01.ATV.0000249620.68705.0d. [DOI] [PubMed] [Google Scholar]
- 27.Kawakami A, et al. Apolipoprotein CIII induces expression of vascular cell adhesion molecule-1 in vascular endothelial cells and increases adhesion of monocytic cells. Circulation. 2006;114:681–687. doi: 10.1161/CIRCULATIONAHA.106.622514. [DOI] [PubMed] [Google Scholar]
- 28.Brownell N, Rohatgi A. Modulating cholesterol efflux capacity to improve cardiovascular disease. Curr Opin Lipidol. 2016;27:398–407. doi: 10.1097/MOL.0000000000000317. [DOI] [PubMed] [Google Scholar]
- 29.Wang S, Gulshan K, Brubaker G, Hazen SL, Smith JD. ABCA1 mediates unfolding of apolipoprotein AI N terminus on the cell surface before lipidation and release of nascent high-density lipoprotein. Arterioscler Thromb Vasc Biol. 2013;33:1197–1205. doi: 10.1161/ATVBAHA.112.301195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Phillips MC. Molecular mechanisms of cellular cholesterol efflux. J Biol Chem. 2014;289:24020–24029. doi: 10.1074/jbc.R114.583658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xu S, et al. Apolipoproteins of HDL can directly mediate binding to the scavenger receptor SR-BI, an HDL receptor that mediates selective lipid uptake. J Lipid Res. 1997;38:1289–1298. [PubMed] [Google Scholar]
- 32.Chen X, et al. Nitrated apolipoprotein AI/apolipoprotein AI ratio is increased in diabetic patients with coronary artery disease. Atherosclerosis. 2016;245:12–21. doi: 10.1016/j.atherosclerosis.2015.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tsun JG, et al. Impact of serum amyloid A on cellular cholesterol efflux to serum in type 2 diabetes mellitus. Atherosclerosis. 2013;231:405–410. doi: 10.1016/j.atherosclerosis.2013.10.008. [DOI] [PubMed] [Google Scholar]
- 34.Warnick GR, Albers JJ. A comprehensive evaluation of the heparin-manganese precipitation procedure for estimating high density lipoprotein cholesterol. J Lipid Res. 1978;19:65–76. [PubMed] [Google Scholar]
- 35.Davidson WS, et al. The effects of apolipoprotein B depletion on HDL subspecies composition and function. J Lipid Res. 2016;57:674–686. doi: 10.1194/jlr.M066613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Marcoux C, Tremblay M, Fredenrich A, Davignon J, Cohn JS. Lipoprotein distribution of apolipoprotein C-III and its relationship to the presence in plasma of triglyceride-rich remnant lipoproteins. Metabolism. 2001;50:112–119. doi: 10.1053/meta.2001.19452. [DOI] [PubMed] [Google Scholar]
- 37.Annema W, et al. Myeloperoxidase and serum amyloid A contribute to impaired in vivo reverse cholesterol transport during the acute phase response but not group IIA secretory phospholipase A(2) J Lipid Res. 2010;51:743–754. doi: 10.1194/jlr.M000323. [DOI] [PMC free article] [PubMed] [Google Scholar]


