Abstract
We use Arabidopsis thaliana as a model to investigate coordination of cell proliferation and cell elongation in the three components that develop side by side in the seed. Two of these, the embryo and its nurturing annex, the endosperm, are placed under zygotic control and develop within the seed integument placed under maternal control. We show that integument cell proliferation and endosperm growth are largely independent from each other. By contrast, prevention of cell elongation in the integument by the mutation transparent testa glabra2 (ttg2) restricts endosperm and seed growth. Conversely, endosperm growth controlled by the HAIKU (IKU) genetic pathway modulates integument cell elongation. Combinations of TTG2 defective seed integument with reduction of endosperm size by iku mutations identify integument cell elongation and endosperm growth as the primary regulators of seed size. Our results strongly suggest that a cross talk between maternal and zygotic controls represents the primary regulator of the coordinated control of seed size in Arabidopsis.
INTRODUCTION
In flowering plants and gymnosperms the embryo develops inside the maternal reproductive organs. Hence, some mechanisms must have evolved to allow coordination of embryo development with morphological and physiological changes in the maternally controlled environment. In flowering plants, a double fertilization takes place (Faure et al., 2002). The ovule contains the two female gametes, the egg cell and the central cell, enclosed in the embryo sac (Christensen et al., 1997). The pollen tube delivers two male gametes. One male gamete fertilizes the egg cell and produces the embryo. Fertilization of the central cell by the other male gamete produces the endosperm, responsible for the transfer of maternally derived nutrients to the embryo (Berger, 2003). The endosperm and the embryo are enclosed in the maternally derived integument, which produces the seed coat. Seed growth requires coordination of cell proliferation and cell elongation of the three components of the seed. Only a few data are available concerning the nature of interactions between the maternal integument and the zygotic products (Chaudhury and Berger, 2001).
In Arabidopsis thaliana, seed development can be divided into two distinct phases. The first phase is characterized by the active proliferation and growth of the endosperm (Boisnard-Lorig et al., 2001). This results in a large increase of size. By contrast, growth of the embryo occurs primarily during the second phase at the expense of the endosperm (Jürgens and Mayer, 1994). The final seed size is mainly attained during the initial phase. Hence, seed growth is mostly coupled to the growth of endosperm and of the integument. During early endosperm development, nuclear division is not followed by cytokinesis and produces a syncytium (Boisnard-Lorig et al., 2001). The multinucleate endosperm cell enlarges as pseudosynchronous nuclear divisions take place until the 8th mitotic cycle when cellularization begins (Sørensen et al., 2002). Increase in size of the syncytial endosperm is prevented by haiku (iku) mutations, leading to precocious endosperm cellularization, reduced embryo proliferation, and decreased seed size (Garcia et al., 2003). Primary reduction of endosperm size in iku/iku seeds causes reduced cell proliferation in the embryo. The iku mutations are sporophytic recessive. Thus, the quarter of seeds produced by selfed iku/+ plants that consist of iku/iku endosperm surrounded by iku/+ integument show a reduced seed size. The sporophytic recessive nature of iku mutations implies that they affect directly the development of the embryo, the endosperm, or both. Although iku mutations affect primarily endosperm growth, the overall seed size is decreased, including the size of the integument. This results from reduction of cell elongation in iku/+ integuments. Because iku mutations are recessive, their effect on integuments cannot be direct, and reduction of integuments elongation is a consequence of the effect of iku mutations on endosperm growth. These observations provide evidence for communication from the endosperm to the integument. Autonomous seed development in fis class mutants further supports evidence for such communication (Ohad et al., 1996; Chaudhury et al., 1997). Central cells with fis mutation undergo development into an endosperm-like structure. Autonomous fis endosperm development is accompanied by enlargement and partial differentiation of seed integuments, which are heterozygous for fis mutations. Because fis mutations are recessive and cannot affect fis/+ seed integument directly, induction of seed integuments development might be a response to autonomous endosperm development. Evidence of communication from the endosperm to the integument led us to speculate on the existence of a feedback control from the integument on endosperm growth. There is some indirect evidence for such maternal sporophytic effects, defined by the control of the development of the filial generation by the genetic characteristics of the mother. Certain transparent testa (tt) mutants defective for tannin synthesis in the seed coat are also affected for seed size (Debeaujon et al., 2000, 2003), and crosses between Arabidopsis ecotypes with seeds of a distinct size produce seeds of the size of the maternal ecotype (Alonso-Blanco et al., 1999). However, in these reports, the cellular processes involved in the control of seed size were not investigated. In this study, we designed genetic experiments to determine the relative roles of integument cell elongation and integument cell proliferation in the maternal control of endosperm growth. This study reveals a new maternal sporophytic control of the endosperm. We further provide evidence for a feedback control on maternal integument elongation by endosperm growth, which altogether control seed size.
RESULTS
The ttg2 Mutation Affects Integument Cell Elongation and Has a Maternal Effect on Endosperm and Seed Growth
The mutant transparent testa glabra2 (ttg2) (Johnson et al., 2002) is defective for proanthocyanidin synthesis and mucilage deposition in the seed coat and produces yellow seeds in comparison with wild-type brown seeds (Figures 1A and 1B). TTG2 encodes a transcription factor of the WRKY family expressed strongly in the seed integument and at low level in the endosperm (Johnson et al., 2002). Seed size is reduced in seeds produced by ttg2/ttg2 plants after selfing (Figures 1A and 1B). Specifically, seed length is reduced by 15% in ttg2/ttg2 seeds (Table 1). Reduction of size in ttg2/ttg2 developing seeds is first observed after the late globular embryo stage (Figures 2A and 2B) and is correlated with reduction of endosperm size (Figures 2A and 2B). This reduction is coupled to precocious endosperm cellularization at the endosperm stage VIII in 70% of the ttg2/ttg2 seeds (n = 58; Figures 2A and 2B, insets), whereas cellularization occurs at the endosperm stage IX in the wild type (Sørensen et al., 2002). Seed size reduction of ttg2/ttg2 seeds is also coupled to a reduction of cell elongation in the integument (Figures 1E and 1F, Table 1). Although the ttg2 mutation affects growth of the endosperm and of the seed integument as in iku2/iku2 seed (Garcia et al., 2003), the reduction of integument cell elongation and of endosperm size is less severe in ttg2 than in iku2 mutants (Figures 1F and 1G, Table1). Endosperm homozygous for iku mutation cause seed length reduction by 30% (i.e., twice more than the reduction caused by ttg2/ttg2 integuments) (Figures 1B and 1C, Table 1). Because TTG2 is expressed both in the seed integument and in endosperm, we performed genetic tests to determine whether the reduction of ttg2/ttg2 seed size could be attributed to the loss-of-function of TTG2 in seed integument, in endosperm, or in both. In contrast with plants carrying the sporophytic recessive iku mutations, ttg2/ttg2 plants that pollinated with wild-type pollen only produce seeds of sizes smaller than wild-type seeds, resulting from a reduction of integument cell elongation (Table 1) and endosperm growth (Figure 2B) during early steps of seed development. The cross between wild-type ovules and ttg2 pollen produces seeds of wild-type size, which shows that ttg2 is not a dominant sporophytic mutation acting on endosperm (Table 1). Hence, because ttg2 is only effective when contributed by the female reproductive organ and because it affects the following generation, ttg2 effect on seed size has at least a maternal effect of sporophytic or of gametophytic origin. In the latter case, half of female gametophytes produced by ttg2/+ plants carry ttg2 and would produce seeds of reduced size when fertilized with wild-type pollen. This was not the case (Table 1), and ttg2/+ plants fertilized with wild-type pollen produced only seeds of wild-type size. Thus, because seed size, integument size, and endosperm size were only reduced when seed integument is homozygous for ttg2, we concluded that ttg2 is sporophytic recessive with a maternal effect on endosperm growth and seed size. Moreover, seeds produced by ttg2/+ plants fertilized with pollen from ttg2/ttg2 plant are uniformly of wild-type size, although half of them are homozygous for ttg2 in endosperm. Hence, reduction of endosperm size in ttg2/ttg2 seed does not result from a partial contribution of a sporophytic recessive action of ttg2 in endosperm, and the effect of ttg2 on endosperm size is strictly sporophytic maternal.
Figure 1.
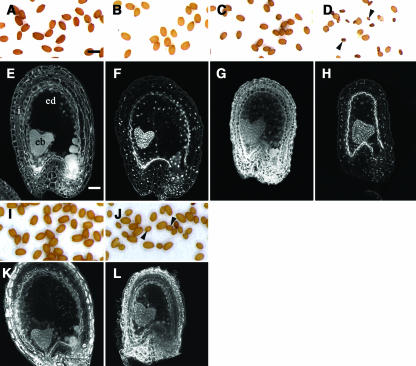
Influence of ttg2 Mutation, iku Mutations, and p35S:KRP2 Construct and Their Combinations on Endosperm Development and Seed Size.
In the wild-type Landsberg erecta (Ler), mature seed size is uniform (A). Confocal section showing the organization of the wild-type seed at the heart embryo stage with the integument (i) surrounding the endosperm (ed) and the embryo (eb) (E). We observe the effect on seed size ([B] to [D]) and on endosperm development ([F] to [H]) of the ttg2 mutation ([B] and [F]), of the iku2 mutation ([C] and [G]), and of the combination of ttg2 and iku2 ([D] and [H]). In all genetic backgrounds showed, embryogenesis is not affected up to the heart embryo stage, after which, a high proportion of ttg2/ttg2; iku2/iku2 seeds abort ([D], arrowheads). (I) and (K) show material obtained from a cross between p35S:KRP2/+ ovule and wild-type Ler pollen. (J) shows seeds resulting from the cross between p35S:KRP2/+; iku1/+ plants with iku1/iku1 pollen. Arrowheads show seeds with iku1/iku1 endosperm and embryo with a size close to that of iku2/iku2 seeds (C). (L) shows a confocal section of such seeds at the embryo heart stage. Bars = 500 μm for (A) to (D), (I), and (F) and 38 μm for (E) to (H), (K), and (L).
Table 1.
Combination of the Effect of ttg2 and iku2 Mutations on Integument Cell Length and Seed Size
| Seed Genotypea | Endosperm/Embryo Genotypeb | Integument Genotypeb | Average Number of Integument Cellsc | Average Integument Cell Length (μm)c | Dry Seed Length (μm) | Dry Seed Width (μm) |
|---|---|---|---|---|---|---|
| Wild-type Ler | id | id | 38.7 ± 2.2 (232) | 17.6 ± 1.3 (232)A | 455 ± 24 (57)A | 272 ± 15 (57)A |
| ttg2-1/ttg2-1 | id | id | 40.8 ± 1.3 (204) | 14.1 ± 1.2 (204)B | 389 ± 21 (57) | 268 ± 15 (57)A |
| ttg2-1/ttg2-1 × wild-type Lerd | ttg2-1/+ | ttg2-1/ttg2-1 | 41.7 ± 2.1 (423) | 14.8 ± 0.7 (423)A,B | 405 ± 24 (57)A | 277 ± 22 (57)A |
| Wild-type Ler × ttg2-1/ttg2-1d | ttg2-1/+ | Wild-type Ler | nd | nd | 453 ± 34 (57)A | 309 ± 29 (57)B |
| ttg2-1/+ × wild-type Lere | 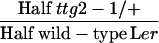 |
ttg2-1/+ | 39.5 ± 1.6 (395) | 17.9 ± 0.4 (395) | 453 ± 33 (57)A | 310 ± 20 (57)B |
| ttg2-1/+ × ttg2/ttg2de | 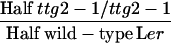 |
ttg2-1/+ | nd | nd | 455 ± 24 (57) | 276 ± 19 (57)A |
| iku2/iku2 | id | id | 41.5 ± 2.8 (581) | 12.1 ± 0.9 (581)C | 332 ± 25 (57)B | 235 ± 23 (57)C |
| iku2/iku2; ttg2-1/ttg2-1f | id | id | 41.5 ± 3.2 (623) | 10.2 ± 1.2 (623)D | 309 ± 26 (57)C | 227 ± 27 (57)C,D |
| iku2/iku2; ttg2-1/ttg2-1 × iku2/iku2d,e | iku2/iku2; ttg2-1/+ | iku2/iku2; ttg2-1/ttg2-1 | 43.3 ± 1.5 (390) | 10.0 ± 1.1 (390)D | 301 ± 31 (57)C | 219 ± 23 (57)D |
| iku2/iku2; ttg2-1/+ × iku2/iku2; ttg2-1/ttg2-1d | 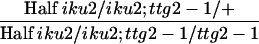 |
iku2/iku2; ttg2-1/+ | 40.7 ± 3.2 (818) | 12.1 ± 0.9 (818)C | 325 ± 27 (57)B | 235 ± 27 (57)C |
Trait values are reported as the mean ± 1 sd with n in parentheses. Subscript letters describe similar trait values, except in the case of average number of integument cells, where no significant differences were observed. Analysis is described in Methods.
id indicates that endosperm/embryo or integument genotype are identical to the genotype mentioned in seed genotype.
nd indicates that the value has not been determined for this parameter.
Crosses are always represented with the female genotype first × male genotype.
In cases of different endosperm/embryo genotype in the seed population, a single and uniform seed size population was observed.
Size of surviving seeds has been measured.
Figure 2.
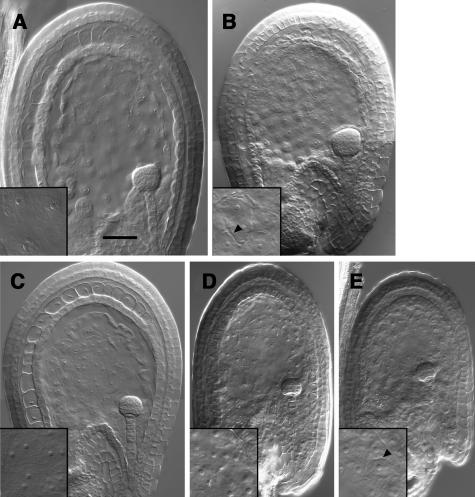
Early Endosperm Phenotype of ttg2 and iku2 Mutations and Their Combination.
Whole-mount cleared seeds observed with differential interference contrast (DIC) microscopy of seed at the triangular embryo stage in wild-type Ler containing uncellularized endosperm (A) and in ttg2/ttg2 containing precociously cellularized endosperm of reduced size (arrowhead shows endosperm cell wall) (B). Cleared seeds at the mid-globular embryo stage in wild-type Ler containing uncellularized endosperm (C), in iku2/iku2 containing uncellularized endosperm of reduced size (D), and in a iku2/iku2; ttg2/ttg2 double mutant seed containing very precociously cellularized endosperm with an increased reduction in size (arrowhead shows endosperm cell wall) (E). Bar = 50 μm for all photographs. Magnification of the inset is twice the magnification of the main micrograph.
Genetic Interaction between ttg2 and iku2 Mutations
To investigate interactions between the IKU and TTG2 pathways controlling endosperm growth and integument cell elongation, respectively, we generated double homozygous ttg2/ttg2; iku2/iku2 plants. Reduction of size in iku2/iku2 developing seeds is first observed at the globular embryo stage and is correlated with reduction of endosperm size (Garcia et al., 2003; Figure 2D). This reduction is followed by precocious endosperm cellularization at the endosperm stage VIII, whereas cellularization occurs at the endosperm stage IX in the wild type (Sørensen et al., 2002). Seed size reduction of iku2/iku2 seeds is also coupled to a reduction of cell elongation in the integument by 30% (Table 1; Garcia et al., 2003). Developing seeds produced by the double mutant are of extremely reduced size in comparison with iku2/iku2 and ttg2/ttg2 seeds (Figures 1B to 1D, Table 1). The combination of ttg2 and iku2 mutations prevents integument cell elongation and growth of endosperm more severely than in each single mutant (Figures 1F to 1H). This leads to arrest of endosperm growth as early as the early globular embryo stage like observed in iku2, whereas ttg2/ttg2 endosperm arrest growth prematurely at the embryo late globular stage (Figure 2). Up to the dermatogen embryo stage, nuclei proliferation does not appear affected in the syncytial endosperm of the double mutant. In ttg2/ttg2; iku2/iku2 seeds, extreme reduction of endosperm growth is followed by endosperm cellularization at the endosperm stage VII (Figures 2C and 2E) (i.e., one mitotic cycle earlier than in iku/iku endosperm [Figures 2D and 2E; Garcia et al., 2003] and two mitotic stages earlier than in the wild-type [Sørensen et al., 2002]). In conclusion, the double homozygous mutant displays a cumulative phenotype combining the maternal effects of ttg2 with the zygotic effect of iku2.
To test the maternal sporophytic contribution of ttg2 in the iku2/iku2 background, we performed crosses of the double mutant iku2/iku2; ttg2/ttg2 with iku2/iku2 pollen. Seeds produced have a phenotype comparable to double homozygous seeds (Table 1), which shows that the seed phenotype resulting from the two mutations is attributable to the addition of the maternal effect of ttg2 to the zygotic effect of iku2. To confirm that the iku2/iku2 background does not reveal a sporophytic recessive effect of ttg2, plants carrying iku2/iku2; ttg2/+ were pollinated with iku2/iku2; ttg2/ttg2 plants. All resulting seeds feature a phenotype comparable to iku2/iku2 seeds (Table 1). These results are consistent with a strict maternal sporophytic recessive effect of ttg2 on endosperm and seed growth in iku2/iku2 background.
In addition to the extreme reduction of seed size, the combination of iku2/iku2 with ttg2/ttg2 leads to increased seed abortion (Figure 1D), probably resulting from an arrest of embryo development earlier than the heart stage. Moreover, half of the viable seeds produced by iku2/iku2; ttg2/ttg2 germinate in the fruit during the desiccation phase, a phenomenon called vivipary (Figure 3D). Such phenomenon does not occur in the wild type, where seeds desiccate and enter dormancy, preventing germination in absence of appropriate conditions. The double mutant seedlings produced could be rescued by in vitro culture. Hence, the combination of ttg2 with iku2 produced a neomorphic effect because vivipary was not detected in either single mutant (Figures 3B and 3C). This synergy is distinct from the additive effect of each single mutation on seed size.
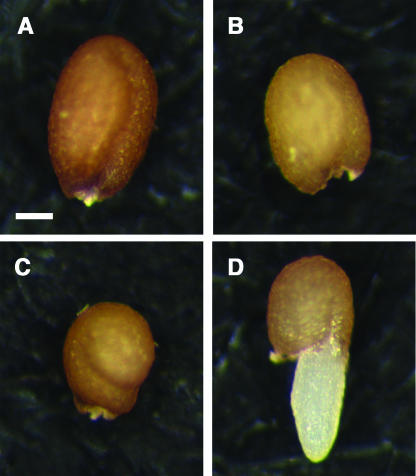
Figure 3.
Seed Neomorphic Phenotype in the iku2/iku2; ttg2/ttg2 Double Mutant.
Dry seeds of wild-type Ler (A), ttg2/ttg2 (B), iku2/iku2 (C), and double mutant iku2/iku2; ttg2/ttg2 showing precocious germination (D). Bar = 100 μm for (A) to (D).
Effect on Endosperm and Seed Size of Reduction of Cell Proliferation in the Integuments
Because a balance between cell elongation and cell proliferation has been demonstrated in leaves (Tsukaya, 2003), we looked for evidence of a cross talk between integument cell proliferation and elongation in relation to endosperm growth. We observed that integument development initiates with a phase of cell proliferation followed by a phase of cell elongation before maturation. To quantify the degree of cell proliferation during early integument development, we quantified the population of cells that undergo the transition from the G2 phase to mitosis with the reporter construct pCycB1,2:β-glucuronidase (GUS) (Schnittger et al., 2002) (Figure 4). Cell proliferation is observed in the integument of mature ovules before fertilization. The beginning of seed integument development is characterized by an increase in mitotic activity after fertilization followed by a sharp decline leading to a complete absence of cell division 4 d after pollination (Figures 4A to 4D).
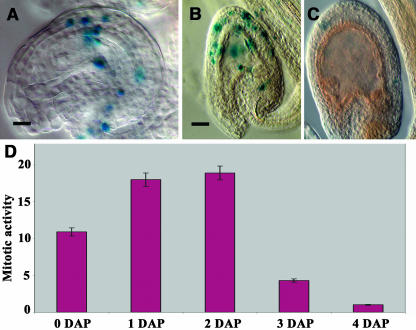
Figure 4.
Evaluation of the Mitotic Activity in the Maternal Seed Integument by Expression of the pCycB1;2:GUS Construct.
This experiment has been done on pistils pollinated 12 h after emasculation.
(A) to (C) GUS staining of cleared developing seeds carrying pCycB1;2:GUS construct harvested at 0, 2, and 4 d after pollination (DAP), respectively. Bars = 20 μm for (A) and 40 μm for (B) and (C).
(D) Variations of the number of integument cells expressing the pCycB1;2:GUS construct from 0 to 4 DAP. (0 DAP corresponds to the emasculation time 12 h before pollination.)
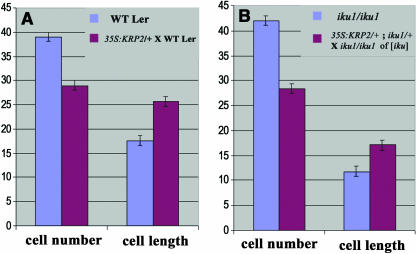
The phase of sustained integument cell proliferation correlates with an initial phase of growth in endosperm, and we hypothesized that a decreased cell proliferation in the integument may constrain endosperm growth and consequentially reduce seed size. We used the line p35S:KRP2 in which the KIP RELATED PROTEIN2 (KRP2) is overexpressed in the vegetative tissues, including the seed integuments (De Veylder et al., 2001; Vandepoele et al., 2002). As shown in other organs, KRP2 overexpression reduces the number of cells in the seed integument (Table 2, Figure 5A). Surprisingly, all seeds produced by crosses of p35S:KRP2/+ plants with wild-type pollen reach a size similar to wild-type seed size (Table 2). We observe as well that both wild-type and p35S:KRP2/+ endosperm surrounded by a reduced number of integument cells reach wild-type size (Figures 1I and 1K). We conclude that a significant reduction of integument cell number has a limited effect on endosperm growth and seed size. This results apparently from compensation of integument cell number reduction by an increased integument cell elongation (Figure 5A, Table 2).
Table 2.
Effect of a Reduced Cell Proliferation in the Integuments on Integument Cell Length and Seed Size
| Seed Genotypea | Endosperm/Embryo Genotypeb | Integument Genotypeb | Average Number of Integument Cellsc | Average Integument Cell Length (μm)c | Dry Seed Length (μm)c | Dry Seed Width (μm)c |
|---|---|---|---|---|---|---|
| Wild-type Ler | id | id | 38.7 ± 2.2 (232)B | 17.9 ± 1.2 (232)B | 455 ± 24 (57)A | 272 ± 15 (57)A |
| iku1/iku1 | id | id | 40.7 ± 2.2 (448)B | 12.5 ± 0.5 (448) | 329 ± 32 (57) | 236 ± 22 (57)B |
| p35S:KRP2/+ × wild-type Lerd,e | 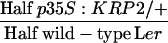 |
p35S:KRP2/+ | 24.6 ± 3.3 (123)A | 25.9 ± 3.1 (123)A | 431 ± 25 (57) | 281 ± 35 (57)A |
| p35S:KRP2/+; iku1/+ × iku1/iku1e | 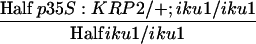 |
p35S:KRP2/+; iku1/+ | 28.4 ± 1.7 (202)A | 16.5 ± 4.5 (202)B | 302 ± 24 (57) | 238 ± 20 (57)B |
 |
nd | nd | nd | nd | ||
| Wild-type Col | id | id | 28.0 ± 2.0 (140)A | 26.0 ± 1.2 (140)A | 453 ± 46 (57)A | 277 ± 26 (57)A |
Trait values are reported as the mean ± 1 sd with n (population size) in parentheses. Subscript letters classify genotypes with no significant differences between them.
id indicates that endosperm/embryo or integument genotype are identical to the genotype mentioned in seed genotype.
nd indicates that the value has not been determined for this parameter.
In the case of different endosperm genotype in the seed population, a single and uniform seed size population was observed.
Crosses are always represented with the female genotype first × male genotype.
Figure 5.
Reduction of Cell Proliferation in the Integuments Is Compensated for by an Increased Integument Cell Elongation Irrespective of the Presence of the iku1 Mutation.
(A) Comparison of the cell number in the integument and the integument cell length between wild-type Ler seeds (blue bar) and a cross between p35S:KRP2/+ ovule and wild-type Ler pollen (red bar). Reduction of cell proliferation in the integument by overexpression of KRP2 in the integument is counterbalanced by integument cell elongation (A), leading to endosperm and seed sizes close to the wild type (Figures 1I and 1K).
(B) Comparison of the cell number in the integument between iku1/iku1 seeds (blue bar) and seeds containing iku1/iku1 endosperm selected from a cross between p35S:KRP2/+; iku1/+ ovule and iku1/iku1 pollen (blue bar; [iku] means of iku phenotype). Compensation of the decreased integument cell proliferation by an increased integument cell elongation is observed as well when endosperm growth is reduced by iku1 (B), leading to endosperm and seed sizes close to iku1 (Figures 1J and 1L).
(A) and (B) Compare the number of cells in the innermost layer (endothelium) of the integument with their length measured using confocal sections similar to those presented in Figure 1.
We further tested whether compensation of cell number reduction in the integument by cell elongation could be related to endosperm growth. The iku mutations limit endosperm growth and indirectly reduce cell elongation in the integument. Reduced integument cell elongation may result from the absence of an instructive IKU-dependent signal from the endosperm necessary for the initiation of integument cell elongation. According to this hypothesis, the reduction of cell integument number around an iku/iku endosperm should lead to an additive effect, reducing both cell number and cell length, finally producing seeds smaller than iku/iku seeds. Alternatively, the iku/iku endosperm may only limit the degree of integument cell elongation (permissive signal), and the integument cells adjust their elongation to the endosperm size. According to this second hypothesis, reduction of the number of integument cells around an iku/iku endosperm should be compensated by integument cell elongation. To test these alternative hypotheses, we pollinated iku1/+; p35S:KRP2/+ plants with iku1/iku1 pollen, hence producing 50% of seeds with iku1/iku1 endosperm surrounded by a reduced number of integument cells as a result of gain-of-function of KRP2 in the integument. According to the first hypothesis, an additive effect of both pathways should lead to ∼30% reduction of the size of p35S:KRP2/+; iku1/iku1 seed. This was not observed (Figures 1C and 1J, Table 2). Remarkably, the reduction of the number of integument cells by 33% is still largely compensated for by a 31% increase in cell elongation in p35S:KRP2/+; iku1/iku1 seed, leading to a size close to that of iku1/iku1 seed (Figures 1G, 1L, and 5B, Table 2). This result is in agreement with the second hypothesis stating that seed integument adjusts cell elongation to an overall size defined by the endosperm and controlled in part by the IKU pathway.
Seed size is known to vary between natural genetic backgrounds in Arabidopsis (Alonso-Blanco et al., 1999). We thus compared seed size between Columbia and Ler background and found that they are similar. Surprisingly, the Columbia integument cell number is reduced in comparison with Ler. The same integument and seed size is achieved because the average integument cell length is higher in Columbia than in Ler, thus allowing compensation (Table 2). This observation strongly suggests that compensation of integument cell number by cell elongation also accounts for natural genetic variations.
DISCUSSION
TTG2 Is Involved in Maternal Control of Seed Size
In this study, we have demonstrated the maternal effect of the ttg2 mutation on endosperm and seed growth. Thus, ttg2 represents one of the few examples of maternal sporophytic control of seed development in Arabidopsis (Ray et al., 1996; Gifford et al., 2003), and our results provide a demonstration of maternal sporophytic control of endosperm development and seed growth in Arabidopsis. The shape of seeds with ttg2 integument is also more round than wild-type seed. Such maternal control of seed shape has been reported in other mutants affecting the shape of the seed coat, such as ats (Léon-Kloosterziel et al., 1994) and acr4 (Gifford et al., 2003), and it is likely that these mutations also affect endosperm growth. We propose that the ttg2 mutation affects primarily cell elongation in the integument. Other tt mutants defective for tannin synthesis in the seed coat are also affected for seed size (Debeaujon et al., 2000). Because TTG2 controls several steps of tannin synthesis, it is likely that reduction of seed size by tt mutations is downstream of TTG2 in the TTG pathway. Some products of the tannin synthesis pathway may accumulate in the cell wall and change its competence to elongate. When TTG function is reduced, the wall may either become more rigid than the wild type with a reduced capacity to elongate or may acquire prematurely a loss of competence to elongate. As a consequence, endosperm growth becomes limited in space. Alternatively, products resulting for altered proanthocyanidin synthesis could diffuse into endosperm and affect its growth. Such a reduction of endosperm growth results in a reduction of cell proliferation in the embryo after the heart stage and ultimately in a smaller embryo and a small mature seed, as shown in iku (Garcia et al., 2003) and in interploid crosses (Scott et al., 1998).
Interactions between Maternal and Zygotic Controls of Seed Size
As previously reported for the zygotic iku mutations (Garcia et al., 2003), the maternal ttg2-mediated reduction of endosperm growth is accompanied by precocious endosperm cellularization, one mitotic cycle earlier than in the wild type. The similarities between the two mutant phenotypes suggested that they participate in the same genetic pathway. We observed an additive reduction of integument cell elongation and of endosperm growth and seed size when iku2 and ttg2 mutations are combined. This indicates that each mutation acts in distinct genetic pathways but have common effectors. Moreover, even in the iku2 background, ttg2 has a strict maternal sporophytic effect. Thus, integument cell elongation and endosperm growth are controlled in synergy by the sporophytic action of IKU2 on endosperm and by the sporophytic maternal action of TTG2 in the seed integument.
The additive phenotype observed in the double mutant ttg2/ttg2; iku2/iku2 also affects endosperm cellularization timing more dramatically than in each mutant. In parallel, reduction of the endosperm volume is markedly more pronounced in the double mutant phenotype than in single mutants. This correlation between endosperm size and cellularization timing corroborates the importance of a threshold nucleocytoplasmic ratio necessary to trigger endosperm cellularization (Sørensen et al., 2002). Because nuclei proliferation does not seem to be altered in ttg2/ttg2; iku2/iku2 endosperm, the threshold is reached prematurely and cellularization occurs earlier than in the iku2, ttg2, or wild-type endosperm.
Extreme reduction of endosperm size likely accounts for higher levels of seed abortion in ttg2/ttg2; iku2/iku2 plants, as observed in the case of extreme maternal genome dosage excess (12m:1p) in the endosperm (Scott et al., 1998). ttg2/ttg2; iku2/iku2 seeds reaching the mature stage display a more important reduction of seed size than iku2/iku2 seeds. This double mutant also displays a neomorphic viviparous phenotype. Previous studies showed the major role of seed coat integrity in the maintenance of seed dormancy (Léon-Kloosterziel et al., 1994; Debeaujon et al., 2000). We propose that the increased reduction of integument elongation by combination of iku2 and ttg2, added to inappropriate seed coat maturation resulting from ttg2, generates a weaker seed coat leading to vivipary. Alternatively, vivipary may be perturbed as in the abi3 and lec mutants (Raz et al., 2001), which deregulate maturation of the embryo.
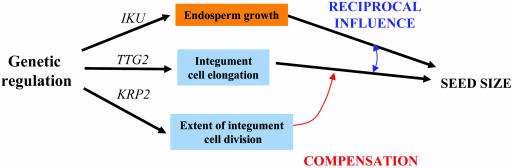
An Integrated Model for the Control of Seed Size
Surprisingly, in Arabidopsis, the initiation of cell proliferation in the seed integument does not appear to depend on fertilization. We further observed that to achieve the size of the integument, dictated by the size of syncytial endosperm, integument cells regulate elongation and not cell proliferation. The final cell number in the integument is balanced by cell elongation and does not influence the size of the seed. Such compensatory effects have been observed at the scale of the organ and of the organism in Arabidopsis (Wang et al., 2000; De Veylder et al., 2001; Jasinski et al., 2002; Tsukaya, 2003) and in animal species (Conlon and Raff, 1999). Independent manipulation of cell elongation and cell proliferation in the integument and their genetic combinations with iku mutations show that integument cell elongation plays the key role in the coordination of size between the endosperm and the integument. This early coordination later influences endosperm cellularization and embryo cell proliferation and determines the final seed size (Figure 6). We propose that the IKU-dependent pathway does not trigger integument cell elongation but rather controls the degree of cell elongation necessary to coordinate the size of the integument with endosperm growth. Such a control could be mediated by IKU-dependent regulation of osmotic pressure in the endosperm, providing a source of tension onto integument cells. This tension would sustain integument cell elongation as shown in other cell types (Cosgrove, 1993). According to this model, TTG2 would modulate the competence of integument cells to elongate (Figure 6). Hence, in the plant seed, cell elongation could integrate interplay between turgor in the syncytial endosperm and tension sensed by the surrounding integument cells.
Figure 6.
Model of Seed Size Control in Arabidopsis.
Seed size determination involves the zygotic control of endosperm growth by IKU class genes in addition to the maternal modulation of integument cell elongation by TTG2. These two pathways are integrated by a cross talk (blue arrow) and determine the final potential size of the seed. The integument cell number is regulated during the early phase of integument cell proliferation. However, as integument cell elongation compensates the cell number (red arrow) to accommodate to the endosperm size, integument cell proliferation does not have a strong influence on seed size.
Phenotypes similar to that of iku/iku seeds have been described in cases of genome dosage imbalance in the endosperm (Scott et al., 1998). Thus, iku seeds resemble seeds obtained by fertilization of wild-type ovules by male gametes with hypomethylated DNA as well as seeds containing an excess of maternal dosage in endosperm (Adams et al., 2000; Luo et al., 2000; Garcia et al., 2003). Such effects strongly suggest that epigenetic controls may regulate parental imprints of genes involved in the control of endosperm growth. Such genes could participate to the IKU pathway.
We observed a striking natural variation in the number of integument cells between Arabidopsis Columbia and Ler ecotypes, although the final seed size does not significantly change as a result of compensation by cell elongation (Table 2). The modulation of cell elongation was thus used by natural selection to produce seeds with a specific size. A maternal control of seed size has been observed when crossing different ecotypes of Arabidopsis producing different seed sizes (Alonso-Blanco et al., 1999). Our results suggest that this maternal control is attributable to maternal integument elongation properties. Thus, we propose that cell elongation was targeted by natural selection as a process that may be more amenable to tinkering (Alon, 2003) than the complex molecular innovations necessary for modulation of cell proliferation.
METHODS
Plant Material and Growth Conditions
The wild-type ecotypes Ler and Columbia, as well as the ttg2 mutant line, were provided by the Nottingham Arabidopsis Stock Centre. All mutant lines examined in Tables 1 and 2 were in the Ler background. Plants were grown at 20°C in a growth chamber with a 12-h-day/12-h-night cycle until they formed rosettes. Flowering was then induced at 22°C with a 16-h-day/8-h-night cycle in a greenhouse.
All crosses were performed using hand pollination 12 h after emasculation. The number of integument cells per seeds and the average integument cell length have been determined on longitudinal confocal sections of fixed seeds 6 DAP, corresponding to the embryo heart stage. Measurements have been performed on the innermost integument cell layer, also called the endothelium.
Microscopy and Image Processing
Endosperm and embryo development was observed in cleared seeds using DIC optics as described previously (Garcia et al., 2003). All figures were composed with Adobe Photoshop 5.5 (Adobe Systems, San Jose, CA). For Tables 1 and 2, the average number of integument cells per seeds and the average integument cell length were determined on longitudinal confocal sections of fixed seeds 6 DAP, similar to sections presented in Figure 1, on the innermost integument cell layer also called the endothelium.
GUS Assays
Siliques at different stages of development were incised along the septum and fixed in ice-cold acetone 80% for 1 h. After three washes with phosphate buffer (100 mM, pH 7.5), the tissues were vacuum infiltrated in a prestaining solution (100 mM phosphate buffer, pH 7.5, 4 mM potassium ferricyanide, and 4 mM potassium ferrocyanide) for 15 min. A second infiltration with a staining solution (100 mM phosphate buffer, pH 7.5, 2 mM potassium ferricyanide, 2 mM potassium ferrocyanide, 0.1% Triton X-100, and 1 mM X-gluc) was performed under vacuum for 15 min. The GUS enzymatic reaction was incubated overnight at 37°C.
Seeds were dissected out from the siliques and cleared using a derivative of Hoyer's medium. The histochemical localization of GUS activity was analyzed on cleared seeds with a Nikon (Tokyo, Japan) microscope using DIC optics. Images were captured with an Axiocam MRc digital camera (Zeiss, Jena, Germany) coupled to the microscope.
Statistical Analyses
Results in Tables 1 and 2 are reported as mean values ± one standard deviation from the mean. Significant differences among the genotypes in Tables 1 and 2 were determined by either one-way analysis of variance or the nonparametric Kruskall-Wallis test. First, for each measured variable, the entire data set was tested for normalcy by the Kolmogorov-Smirnov test against a generated normal distribution of equivalent mean, n, and standard deviation. Heterogeneity of variance was determined by Bartlett's test. Data sets fulfilling both requirements were tested by analysis of variance (P < 0.0001 in all cases except for average number of cells in Table 1), followed by Scheffé's F to determine significant differences between the various genotypes. All other data sets were analyzed using Kruskal-Wallis (P < 0.0001 in all cases), followed by Games-Howell as a post hoc test. For consistency and accuracy, all data sets were examined again by Games-Howell at a more stringent confidence level (99%), and this did not change the results. In the cases where two genotypes were compared independently, a Mann-Whitney U-test was employed. All calculations were performed using StatView 5.0.1 (SAS Institute).
Acknowledgments
We thank C. Dumas for hosting our team in the Unité Mixte de Recherche 5667. We are deeply indebted to Arp Schnittger for his gift of the marker pCycB1;2:GUS and to D. Inzé for the gift of the p35S:KRP2 line. We thank Mathieu Ingouff and Vanessa Vernoud for critical reading of the manuscript. We thank the Nottingham Arabidopsis Seed Centre for providing the line ttg2-1. This research was supported by the Institut National de la Recherche Agronomique, the European Molecular Biology Organization Young Investigator Program, and the Action Concertée Incitative Jeune supported by the Ministère de la Recherche et de la Technologie.
The author responsible for distribution of materials integral to the findings presented in this article in accordance with the policy described in the Instructions for Authors (www.plantcell.org) is: Frédéric Berger (fred@tll.org.sg).
Article, publication date, and citation information can be found at www.plantcell.org/cgi/doi/10.1105/tpc.104.027136.
References
- Adams, S., Vinkenoog, R., Spielman, M., Dickinson, H.G., and Scott, R.J. (2000). Parent-of-origin effects on seed development in Arabidopsis thaliana require DNA methylation. Development 127, 2493–2502. [DOI] [PubMed] [Google Scholar]
- Alon, U. (2003). Biological networks: The tinkerer as an engineer. Science 301, 1866–1867. [DOI] [PubMed] [Google Scholar]
- Alonso-Blanco, C., Blankestijn, D.E., Vries, H., Hanhart, C.J., and Koornneef, M. (1999). Natural allelic variation at seed size loci in relation to other life history traits of Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 96, 4710–4717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berger, F. (2003). Endosperm: The crossroad of seed development. Curr. Opin. Plant Biol. 6, 42–50. [DOI] [PubMed] [Google Scholar]
- Boisnard-Lorig, C., Colon-Carmona, A., Bauch, M., Hodge, S., Doerner, P., Bancharel, E., Dumas, C., Haseloff, J., and Berger, F. (2001). Dynamic analyses of the expression of the histone::YFP fusion protein in Arabidopsis show that syncytial endosperm is divided in mitotic domains. Plant Cell 13, 495–509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaudhury, A.M., and Berger, F. (2001). Maternal controls of seed development. Semin. Cell Dev. Biol. 12, 381–386. [DOI] [PubMed] [Google Scholar]
- Chaudhury, A.M., Ming, L., Miller, C., Craig, S., Dennis, E.S., and Peacock, W.J. (1997). Fertilization-independent seed development in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 94, 4223–4228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christensen, C.A., King, E.J., Jordan, R.J., and Drews, G.N. (1997). Megagametogenesis in Arabidopsis wild type and the Gf mutant. Sex. Plant Reprod. 10, 49–64. [Google Scholar]
- Conlon, I., and Raff, M. (1999). Size control in animal development. Cell 96, 235–244. [DOI] [PubMed] [Google Scholar]
- Cosgrove, D.J. (1993). How do plant cell walls extend? Plant Physiol. 102, 1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Veylder, L., Beeckman, T., Beemster, G.S.T., Krols, L., Terras, F., Landrieu, I., Van Der Schueren, E., Maes, S., Naudts, M., and Inzé, D. (2001). Functional analysis of cyclin-dependent kinase inhibitors of Arabidopsis. Plant Cell 13, 1653–1667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debeaujon, I., Léon-Kloosterziel, K.M., and Koorneef, M. (2000). Influence of the testa on seed dormancy, germination and longevity in Arabidopsis. Plant Physiol. 122, 403–413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debeaujon, I., Nesi, N., Perez, P., Devic, M., Grandjean, O., Caboche, M., and Lepiniec, L. (2003). Proanthocyanidin-accumulating cells in Arabidopsis testa: Regulation of differentiation and role in seed development. Plant Cell 15, 2514–2531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faure, J.E., Rotman, N., Fortuné, P., and Dumas, C. (2002). Fertlilization in Arabidopsis thaliana wild type: Developmental stages and time course. Plant J. 30, 481–488. [DOI] [PubMed] [Google Scholar]
- Garcia, D., Saingery, V., Chambrier, P., Mayer, U., Jürgens, G., and Berger, F. (2003). Arabidopsis haiku mutants reveal new controls of seed size by endosperm. Plant Physiol. 131, 1661–1670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gifford, M.L., Dean, S., and Ingram, G.C. (2003). The Arabidopsis ACR4 gene plays a role in cell layer organisation during ovule integument and sepal margin development. Development 130, 4249–4258. [DOI] [PubMed] [Google Scholar]
- Jasinski, S., Riou-Khamlichi, C., Roche, O., Perennes, C., Bergounioux, C., and Glab, N. (2002). The CDK inhibitor NtKIS1a is involved in plant development, endoreduplication and restores normal development of cyclin D3; 1-overexpressing plants. J. Cell Sci. 115, 973–982. [DOI] [PubMed] [Google Scholar]
- Johnson, C.S., Kolevski, B., and Smyth, D.R. (2002). TRANSPARENT TESTA GLABRA2, a trichome and seed coat development gene of Arabidopsis, encodes a WRKY transcription factor. Plant Cell 14, 1359–1375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jürgens, G., and Mayer, U. (1994). Arabidopsis. In Embryos: Colour Atlas of Development, J.B.L. Bard, ed (London: Wolfe Publishing), pp.7–21.
- Léon-Kloosterziel, K.M., Keijzer, C.J., and Koornneef, M. (1994). A seed shape mutant in Arabidopsis that is affected in integument development. Plant Cell 6, 385–392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo, M., Bilodeau, P., Dennis, E.S., Peacock, W.J., and Chaudhury, A. (2000). Expression and parent-of-origin effects for FIS2, MEA, and FIE in the endosperm and embryo of developing Arabidopsis seeds. Proc. Natl. Acad. Sci. USA 97, 10637–10642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohad, N., Margossian, L., Hsu, Y.C., Williams, C., Repetti, P., and Fischer, R.L. (1996). A mutation that allows endosperm development without fertilization. Proc. Natl. Acad. Sci. USA 93, 5319–5324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ray, S., Golden, T., and Ray, A. (1996). Maternal effects of the short integument mutation on embryo development in Arabidopsis. Dev. Biol. 180, 365–369. [DOI] [PubMed] [Google Scholar]
- Raz, V., Bergervoet, J.H.W., and Koornneef, M. (2001). Sequential steps for developmental arrest in Arabidopsis seeds. Development 128, 243–252. [DOI] [PubMed] [Google Scholar]
- Schnittger, A., Schöbinger, U., Stierhof, Y.D., and Hülskamp, M. (2002). Ectopic B-type cyclin expression induces mitotic cycles in endoreduplicating Arabidopsis trichomes. Curr. Biol. 12, 415–420. [DOI] [PubMed] [Google Scholar]
- Scott, R.J., Spielman, M., Bailey, J., and Dickinson, H.G. (1998). Parent-of-origin effects on seed development in Arabidopsis thaliana. Development 125, 3329–3341. [DOI] [PubMed] [Google Scholar]
- Sørensen, M.B., Mayer, U., Lukowitz, W., Chambrier, P., Jürgens, G., Lepiniec, L., and Berger, F. (2002). Cellularisation in the endosperm of Arabidopsis thaliana is coupled to mitosis and share multiple components with cytokinesis. Development 129, 5667–5676. [DOI] [PubMed] [Google Scholar]
- Tsukaya, H. (2003). Organ shape and size: A lesson from studies of leaf morphogenesis. Curr. Opin. Plant Sci. 6, 57–62. [DOI] [PubMed] [Google Scholar]
- Vandepoele, K., Raes, J., De Veylder, L., Rouzé, P., Rombauts, S., and Inzé, D. (2002). Genome-wide analysis of core cell cycle genes in Arabidopsis. Plant Cell 14, 903–916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, H., Zhou, Y., Gilmer, S., Whitwill, S., and Fowke, L.C. (2000). Expression of the plant cyclin-dependent kinase inhibitor ICK1 affects cell division, plant growth and morphology. Plant J. 24, 613–623. [DOI] [PubMed] [Google Scholar]