Abstract
Background
Prolonged storage of packed red blood cells (PRBCs) may increase morbidity and mortality, and patients having massive transfusion might be especially susceptible. We therefore tested the hypothesis that prolonged storage increases mortality in patients receiving massive transfusion after trauma or nontrauma surgery. Secondarily, we considered the extent to which storage effects differ for trauma and nontrauma surgery.
Methods
We considered surgical patients given more than 10 units of PRBC within 24 hours and evaluated the relationship between mean PRBC storage duration and in-hospital mortality using multivariable logistic regression. Potential nonlinearities in the relationship were assessed via restricted cubic splines. The secondary hypothesis was evaluated by considering whether there was an interaction between the type of surgery (trauma versus nontrauma) and the effect of storage duration on outcomes.
Results
305 patients were given a total of 8,046 units of PRBCs, with duration ranging from 8 to 36 days (mean ± SD: 22 ± 6 days). The odds ratio [95% confidence interval (CI)] for in-hospital mortality corresponding to a one-day in mean PRBC storage duration was 0.99 (0.95, 1.03, P = 0.77). The relationship did not differ for trauma and nontrauma patients (P = 0.75). Results were similar after adjusting for multiple potential confounders.
Conclusions
Mortality after massive blood transfusion was no worse in patients transfused with PRBC stored for long periods. Trauma and nontrauma patients did not differ in their susceptibility to prolonged PRBC storage.
1. Background
Over 14 million units of blood products are transfused in the United States (U.S.) annually [1]. Massive blood transfusions are given to 3–5% [2] of the civilian and 8–10% [3] of the military trauma patient population. Patients requiring massive blood transfusion are at high risk for adverse clinical outcomes largely because of their serious trauma, but also as a direct consequence of receiving numerous blood products. Transfusions of blood products are associated with several complications and current evidence suggests that transfusions independently increase the risk of morbidity and death in critically ill patients [4–8], with mortality increasing linearly as a function of the amount of blood transfused. For example, a recent trauma registry analysis found that major blood loss constitutes an important prognostic factor for the survival [9] and the authors of the PROMMTT trial [10] demonstrated an adjusted odd ratio of 6-hour mortality for patients receiving ≥4 units within 30 minutes of 2.1 (95% confidence interval: 1.2–3.5). Overall, adverse consequences of transfusions add about $17 billion to United States healthcare costs which exceeds the costs of blood acquisition and transfusion combined [11].
Koch and colleagues identified a strong association between storage of red cells more than 14 days and major complications and mortality after coronary artery bypass graft (CABG) surgery [1]. The United States Food and Drug Administration allows storage of PRBCs for up to 42 days; however, it is well established that stored red cells undergo substantial biochemical and morphological changes during this period. Important dysfunction includes reduced oxygen delivery resulting from progressive decrease in 2,3-diphosphoglycerate (2,3-DPG) concentrations [12], reduced PRBC-dependent vasodilation [13] (for review see [14, 15]), decreased cell membrane deformability [16, 17] which potentially obstructs capillary flow, consumption of clotting factors, and activation of intravascular coagulation through PRBC derived microvesicles [18]. Prolonged PRBC storage is also thought to increase the risk of severe infection after CABG surgery [19].
Other studies, though, do not identify harm from transfusion of older blood. For example, Edgren and colleagues [20] analysed the Scandinavian Donations and Transfusions (SCANDAT) database and found that the initial trend towards a seven-day risk of death reduction diminishes within the 2-year follow-up and that transfusion of older blood is likely to contribute to less than 5% to excess mortality. Saager and colleagues similarly reported no relationship between prolonged median storage duration and mortality in a retrospective analysis of data from noncardiac surgery patients [21]. More importantly, two large randomized trials, RECESS and ABLE, concluded that PRBC storage duration did not affect the outcome after CABG surgery [22] or in critically ill patients [23].
Routine surgical patients, if given blood, usually receive just one or two units. Trauma patients differ in often experiencing major blood loss and consequently requiring large amounts of blood products. Trauma patients, along with nontrauma patients, who are given large amounts of blood (i.e., ≥10 units) may thus be especially susceptible to injury from blood that has been long stored.
Consistent with this theory, Zallen and colleagues performed a small prospective database analysis in trauma patients and concluded that multiorgan failure was more likely when patients were transfused with blood stored for 30 days than when stored for 24 days [24]. Two similar small studies suggest that prolonged blood storage duration is associated with an increased need [25] and increased duration of critical care [26]. Furthermore Weinberg and colleagues [27] included 176 trauma patients who were given at least one unit of blood within 24 hours after hospital arrival. Those given six units or more of PRBC, with at least three units stored for more than 14 days, had an odds ratio of dying of 7.8 (95% CI 2.3–26.3), twice the odds ratio of patients who received blood stored for less than 14 days. Previous published reports focused mainly on the impact of different storage durations in relative small numbers of transfused PRBCs (i.e., 5–10 units) on clinical outcome [24, 26, 28, 29].
We therefore tested the hypothesis that mean storage duration of transfused PRBC is associated with in-hospital mortality in patients given more than 10 units of PRBC within 24 hours. We also compared the relationship between storage duration and mortality in trauma and nontrauma patients who required massive blood transfusion for elective surgery. Specifically, we tested the secondary hypothesis that the relationship between blood storage duration and mortality is similar in trauma and nontrauma patients given massive amounts of blood.
2. Patients and Methods
The University of Louisville Hospital is a level-one trauma centre with about 3,000 trauma admissions/year. After the approval by the institutional review board at the University of Louisville, we performed this retrospective observational cohort study. Our analysis was restricted to patients who were given more than 10 units of PRBCs within a 24-hour surgical intervention according to the Hospital Blood Bank database. We include neither patients who died within six hours after starting massive blood transfusion and suffered from irreversible lethal injuries (i.e., gunshot to the head, open brain injuries) nor patients with more than 20% incomplete data or inadequate documentation within the medical records into our analysis. In addition to eligibility criteria for the study, we excluded patients for whom storage duration information was unavailable for >10% of their transfused PRBC units.
We accessed the University Hospital Information Management system to retrieve baseline demographic, perioperative, and outcome variables as listed in Table 1. Trauma scores and injury schemes were retrieved from the hospital's trauma database and reviewed by an attending trauma surgeon (J.S.).
Table 1.
Summary of baseline patient characteristics by quartiles of patient-specific mean storage duration of transfused red blood cells. Statistics reported as a percentage, mean ± standard deviation, or median [first quartile, third quartile]. Medians and quartiles are reported when the sample skewness coefficient is >0.9 in absolute value.
| Factor | Level | First quartile (8.0–17.7 days) |
Second quartile (17.7–21.6 days) |
Third quartile (21.6–26.6 days) |
Fourth quartile (26.6–36.6 days) |
Percent missing |
|---|---|---|---|---|---|---|
| (N = 77) | (N = 76) | (N = 76) | (N = 76) | |||
|
| ||||||
| Year of admission | 2006 | 16 | 8 | 5 | 7 | 0.7 |
| 2007 | 13 | 11 | 11 | 5 | ||
| 2008 | 31 | 26 | 27 | 13 | ||
| 2009 | 9 | 17 | 23 | 41 | ||
| 2010 | 16 | 16 | 17 | 16 | ||
| 2011 | 16 | 22 | 17 | 17 | ||
|
| ||||||
| Age | 41 ± 18 | 45 ± 19 | 48 ± 18 | 47 ± 18 | ||
|
| ||||||
| Female sex | 32 | 33 | 28 | 29 | ||
|
| ||||||
| Body mass index (kg/m2) | 26 [22,29] | 26 [24,32] | 26 [23,32] | 25 [24,30] | 36.7 | |
|
| ||||||
| Patient blood type | A− | 8 | 11 | 12 | 7 | 0.7 |
| A+ | 19 | 30 | 33 | 37 | ||
| AB+ | 0 | 4 | 3 | 7 | ||
| B− | 1 | 3 | 4 | 1 | ||
| B+ | 8 | 7 | 8 | 12 | ||
| O− | 13 | 3 | 12 | 4 | ||
| O+ | 51 | 43 | 29 | 33 | ||
|
| ||||||
| Type of surgery | Trauma | 74 | 73 | 70 | 64 | 0.7 |
| Cancer | 10 | 7 | 11 | 12 | ||
| General | 6 | 9 | 11 | 12 | ||
| Vascular | 4 | 9 | 5 | 7 | ||
| Gynecological | 4 | 0 | 4 | 3 | ||
| Orthopedic | 1 | 1 | 0 | 3 | ||
|
| ||||||
| Heart disease | 13 | 20 | 17 | 18 | ||
|
| ||||||
| Pulmonary disease | 14 | 11 | 7 | 8 | ||
|
| ||||||
| Renal disease | 0 | 5 | 1 | 1 | ||
|
| ||||||
| Hypertension | 26 | 39 | 41 | 37 | ||
|
| ||||||
| Diabetes mellitus | 13 | 4 | 16 | 12 | ||
|
| ||||||
| Carcinoma | 10 | 16 | 12 | 16 | ||
|
| ||||||
| Liver disease | 9 | 16 | 12 | 13 | ||
|
| ||||||
| Tobacco use | 29 | 29 | 21 | 24 | ||
|
| ||||||
| Alcohol use | 12 | 11 | 11 | 8 | ||
|
| ||||||
| Illicit drug use | 9 | 7 | 5 | 8 | ||
|
| ||||||
| Initial heart rate (beats per minute) | 107 ± 35 | 105 ± 37 | 102 ± 35 | 103 ± 32 | 1.3 | |
|
| ||||||
| Initial systolic blood pressure (mmHg) | 108 [82, 122] | 100 [84, 123] | 101 [88, 123] | 111 [87, 130] | 0.7 | |
|
| ||||||
| Initial temperature (°C) | 36 [36, 37] | 36 [35, 37] | 36 [36, 37] | 36 [35, 37] | 14.1 | |
|
| ||||||
| ED crystalloids (L) | 2.6 [1.0, 4.0] | 2.0 [1.0, 2.6] | 1.5 [1.0, 2.3] | 2.0 [1.0, 2.5] | 32.1 | |
|
| ||||||
| Length of surgery (min) | 146 [100, 240] | 140 [99, 214] | 141 [94, 253] | 140 [104, 240] | 6.9 | |
|
| ||||||
| Total number of units transfused | 23 [17, 38] | 23 [16, 35] | 22 [17, 31] | 18 [15, 25] | ||
|
| ||||||
| Minimum RBC storage duration | 8 ± 3 | 11 ± 4 | 14 ± 5 | 19 ± 7 | ||
|
| ||||||
| Maximum RBC storage duration | 33 [20, 37] | 37 [32, 40] | 37 [34, 41] | 39 [36, 41] | ||
|
| ||||||
| Initial laboratory values | ||||||
| Hemoglobin (g/dL) | 10 [8, 12] | 11 [8, 12] | 11 [9, 12] | 10 [9, 12] | 4.3 | |
| Blood pH | 7.2 [7.1, 7.3] | 7.2 [7.0, 7.3] | 7.2 [7.1, 7.3] | 7.2 [7.1, 7.4] | 2.3 | |
| Platelet count (k/mcL) | 190 [120, 227] | 180 [94, 256] | 187 [111, 248] | 184 [100, 264] | 2.3 | |
| International normalized ratio | 1.5 [1.2, 1.9] | 1.5 [1.2, 1.9] | 1.5 [1.2, 1.8] | 1.4 [1.1, 2.0] | 4.9 | |
| Activated prothrombin time (s) | 16 [13, 20] | 15 [13, 20] | 15 [12, 20] | 15 [12, 20] | 5.2 | |
| Activated partial thromboplastin time (s) | 37 [29, 59] | 35 [27, 57] | 40 [27, 55] | 33 [26, 64] | 4.9 | |
|
| ||||||
| Subset of trauma patients | (N = 57) | (N = 55) | (N = 53) | (N = 48) | — | |
|
| ||||||
| Blunt injury | 65 | 65 | 64 | 58 | ||
| Penetrating injury | 25 | 25 | 32 | 27 | ||
| Other type of injury | 11 | 9 | 4 | 15 | ||
| Injury severity score | 30 ± 10 | 29 ± 14 | 28 ± 13 | 27 ± 11 | 2.8 | |
| Glasgow coma score | 10 ± 5 | 9 ± 6 | 10 ± 5 | 10 ± 5 | ||
Patients' medical records were reviewed from the time they arrived in the Emergency Department (ED, trauma patients) or the start of massive blood transfusion (nontrauma patients) until the time of discharge. We considered Emergency Department, Surgical and Anaesthesia, Laboratory, and Blood Bank records. Only initial surgical procedures were considered.
Information regarding storage duration of blood components, ABO blood type, and the time at which each product was released by the University of Louisville Hospital Blood Bank was collected from the same database. All PRBCs were leukoreduced by the blood supplier (usually the American Red Cross) and stored in AS-1 solution.
Preprocedure biochemistry and blood gas data were retrieved from either ED records or, if not available, from the initial intraoperative blood collection. Postprocedure biochemistry and haematological values were obtained from the final intraoperative or initial postanesthesia care unit blood samples.
2.1. Statistical Methods
Data analysis followed a stepwise approach: first, baseline potential confounding variables were assessed for balance using standard univariable numerical summaries across quartiles of observed mean PRBC storage duration.
To evaluate the principal hypothesis of the study, namely, that mean PRBC storage duration is associated with in-hospital mortality, we conducted both a univariable analysis and a (primary) multivariable analysis. Within our logistic regression models, patient mean PRBC storage duration was analysed as a continuous variable. Potential nonlinearities in the relationship between patient mean PRBC storage duration and mortality were assessed using a Chi-squared goodness of fit test, which compared a model that incorporated restricted cubic splines to a model that assumed a linear effect. The secondary hypotheses regarding the existence of differential relationships for trauma and nontrauma patients were evaluated by adding an interaction term between patient mean PRBC storage duration and an indicator for trauma to these models.
To construct the multivariable model, we used backward stepwise variable selection, starting with a “full” model that included the variables listed in Table 1 (excluding the indicator variables describing the type of trauma that occurred in lieu of the fact that we analysed trauma as a binary variable that was linearly dependent on these indicator variables) provided the variables were not >10% missing. Variable selection was implemented in a penalized fashion, based on Akaike's Information Criterion (AIC) [30]. By introducing a penalty proportional to each additional parameter in the model, the AIC encourages parsimonious models.
The potential confounding variables we considered were year of admission, age, female sex, patient blood type, along with a history of heart disease, pulmonary disease, renal disease, hypertension, diabetes mellitus, carcinoma, liver disease, tobacco use, alcohol use, or illicit drug use. We also considered preprocedural hemoglobins, heart rate, systolic blood pressure, blood pH, platelet count, international normalized ratio, prothrombin time, activated partial thromboplastin time, and number of transfused PRBC units. And finally, we also included duration of surgery. The multivariable models included only patients with complete information on these covariates.
R statistical software version 3.2.0 (The R Foundation for Statistical Computing, Vienna, Austria) for 64-bit Microsoft Windows operating system was used to perform all analyses. We used a Type I error rate of 5% for the evaluation of all hypotheses.
3. Results
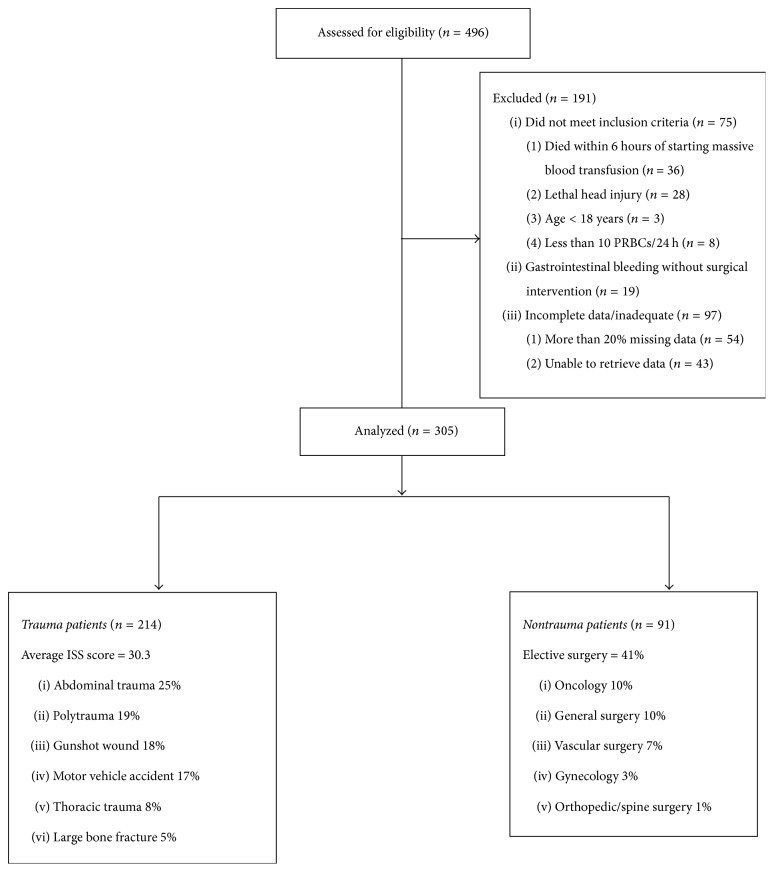
We identified 496 consecutive trauma and nontrauma patients who were given more than 10 units of PRBCs within 24 hours of surgery over a six-year period. 191 were excluded, mostly because patients died within six hours or because of missing data or failure to meet inclusion criteria. A detailed enrolment scheme is attached as Figure 1 and a summary of baseline characteristics for the four quartile groups of patient mean PRBC storage duration defined among the remaining 305 patients is given in Table 1. Baseline characteristics by type of surgery (trauma versus nontrauma) are presented in Table 2. With patients staying an average of 21 days (minimum: 1 day, maximum: 138 days) in the hospital, the overall in-hospital mortality was 101/305 (33.1%), including 23/90 (25.6%) of nontrauma patients and 76/213 (35.7%) of trauma patients.
Figure 1.
Enrolment scheme.
Table 2.
Summary of baseline patient characteristics by type of surgery. Statistics reported as a percentage, mean ± standard deviation, or median [first quartile, third quartile]. Medians and quartiles are reported when the sample skewness coefficient is >0.9 in absolute value.
| Factor | Level | Nontrauma | Trauma | Percent missing |
|---|---|---|---|---|
| (N = 91) | (N = 214) | |||
| Year of admission | 2006 | 2 | 12 | 0.7 |
| 2007 | 10 | 10 | ||
| 2008 | 29 | 23 | ||
| 2009 | 27 | 20 | ||
| 2010 | 16 | 16 | ||
| 2011 | 15 | 19 | ||
|
| ||||
| Age | 53 ± 15 | 42 ± 18 | ||
|
| ||||
| Female sex | 42 | 26 | ||
|
| ||||
| Body mass index (kg/m2) | 25 [23, 31] | 27 [24, 30] | 36.3 | |
|
| ||||
| Patient blood type | A− | 8 | 10 | 0.7 |
| A+ | 32 | 29 | ||
| AB+ | 4 | 3 | ||
| B− | 1 | 3 | ||
| B+ | 7 | 9 | ||
| O− | 12 | 6 | ||
| O+ | 36 | 40 | ||
|
| ||||
| Heart disease | 33 | 10 | ||
|
| ||||
| Pulmonary disease | 15 | 7 | ||
|
| ||||
| Renal disease | 5 | 0 | ||
|
| ||||
| Hypertension | 60 | 25 | ||
|
| ||||
| Diabetes mellitus | 22 | 7 | ||
|
| ||||
| Carcinoma | 36 | 4 | ||
|
| ||||
| Liver disease | 21 | 9 | ||
|
| ||||
| Tobacco use | 36 | 21 | ||
|
| ||||
| Alcohol use | 12 | 9 | ||
|
| ||||
| Illicit drug use | 3 | 9 | ||
|
| ||||
| Initial heart rate (beats per minute) | 91 [79, 110] | 112 [88, 132] | 1.3 | |
|
| ||||
| Initial systolic blood pressure (mmHg) | 116 ± 25 | 98 ± 36 | 0.7 | |
|
| ||||
| Initial temperature (°C) | 36 [36, 37] | 36 [35, 37] | 14.1 | |
|
| ||||
| ED crystalloids (L) | 0.6 [0.5, 1.8] | 2.0 [1.0, 3.0] | 32.1 | |
|
| ||||
| Length of surgery (min) | 214 [120, 400] | 130 [90, 206] | 6.9 | |
|
| ||||
| Total number of units transfused | 18 [14, 24] | 23 [16, 34] | ||
|
| ||||
| Minimum PRBC storage duration | 15 ± 7 | 12 ± 6 | ||
|
| ||||
| Maximum PRBC storage duration | 37 [34, 40] | 37 [32, 40] | ||
|
| ||||
| Initial laboratory values | ||||
| Hemoglobin (g/dL) | 10 [8, 12] | 11 [9, 12] | 4.3 | |
| Blood pH | 7.3 [7.2, 7.4] | 7.2 [7.1, 7.3] | 2.3 | |
| Platelet count (k/mcL) | 178 [97, 260] | 189 [112, 248] | 2.3 | |
| International normalized ratio | 1.3 [1.1, 1.6] | 1.5 [1.2, 2.0] | 4.9 | |
| Activated prothrombin time (s) | 14 [12, 17] | 16 [13, 20] | 5.2 | |
| Activated partial thromboplastin time (s) | 34 [27, 44] | 39 [28, 63] | 4.9 | |
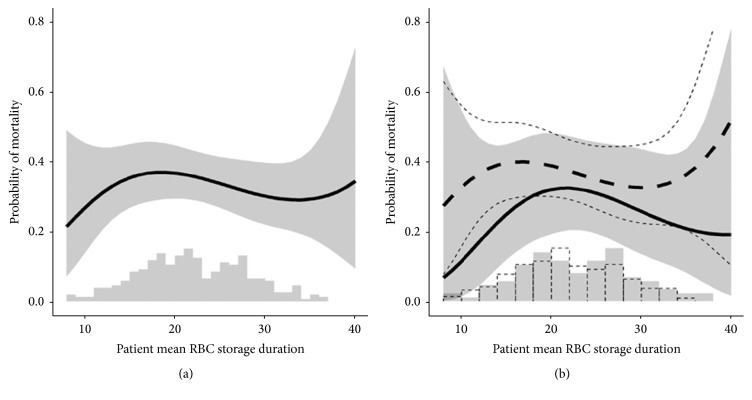
Overall, patient mean PRBC storage duration ranged from 8 to 37 days, with a mean ± standard deviation storage duration of 22 ± 6 days. Figure 2(a) displays a histogram, as well as the estimated (nonlinear) relationship with in-hospital mortality, based on the univariable logistic regression model. The restricted cubic spline term which characterized the nonlinear relationship did not significantly improve the model fit (P = 0.78, Chi-squared goodness of fit test), suggesting that a linear approximation sufficed. Based on this simpler model, the odds ratio (95% confidence interval) for in-hospital mortality corresponding to a one-day increase in patient mean PRBC storage duration was estimated at 0.99 (0.95, 1.03, P = 0.77, Chi-squared test for model coefficients). No significant evidence of differential relationships between trauma and nontrauma patients was found (P = 0.87 for the interaction between trauma and patient mean PRBC storage duration; see Figure 2(b)).
Figure 2.
(a) Univariable relationship between patient mean red blood cell (RBC) storage duration and in-hospital mortality. Results presented as estimate and pointwise 95% confidence interval. A histogram of the observed values of patient mean RBC storage duration underlies the plot. The P value represents a Chi-squared test of association for a univariable logistic regression model which incorporated restricted cubic splines to allow for nonlinearities. (b) Univariable relationship between patient mean red blood cell (RBC) storage duration and in-hospital mortality, separately for trauma (dashed lines) and nontrauma (continuous line/shaded region) patients. Results presented as group-specific estimates and pointwise 95% confidence intervals. Histograms of the observed values of patient mean RBC storage duration underlie the plot. The lack of significance indicated by the P value in the figure indicates that our data did not support the hypothesis that the nature of the relationship differed among the groups (i.e., test for the interaction between group and patient mean RBC storage duration).
Data from 259 patients were included in our multivariable modelling. The stepwise variable selection procedure identified age, renal disease, alcohol use, preprocedure blood pH, activated partial thromboplastin time, and number of units transfused as covariates for the final multivariable model. However, results were similar to those obtained from the univariable modelling. The adjusted odds ratio (95% CI) for patient mean PRBC storage duration was 0.98 (0.93, 1.04).
Secondary outcomes are summarized in Tables 3 and 4. There was no significant relationship between mean storage duration and in-hospital mortality. Patients who were given blood of the first and third quartile had a slightly (nonsignificantly) longer hospitalization, ICU stay, and mechanical ventilation than patients given blood from the second or fourth storage duration quartile.
Table 3.
Summary statistics of outcomes by quartiles of patient-specific mean storage duration of transfused red blood cells. Statistics reported as a percentage, mean ± standard deviation, or median [first quartile, third quartile]. Results reflect 305 patients included in the univariable analyses (see Methods). Medians and quartiles are reported when the sample skewness coefficient is >0.9 in absolute value.
| Outcome variable | First quartile (8.0–17.7 days) |
Second quartile (17.7–21.6 days) |
Third quartile (21.6–26.6 days) |
Fourth quartile (26.6–36.6 days) |
Percent missing |
|---|---|---|---|---|---|
| (N = 77) | (N = 76) | (N = 76) | (N = 76) | ||
| Primary outcome | |||||
| In-hospital mortality | 29 | 43 | 28 | 34 | |
| Secondary outcomes | |||||
| 30-day mortality | 29 | 43 | 25 | 34 | |
| Duration of hospitalization (d) | 21 [7, 34] | 15 [4, 30] | 20 [8, 32] | 14 [4, 26] | |
| Duration of ICU stay (h) | 312 [95, 484] | 182 [35, 408] | 235 [66, 528] | 192 [56, 338] | 1 |
| Duration of mechanical ventilation (h) | 126 [29, 304] | 104 [24, 318] | 124 [48, 359] | 72 [24, 213] | 1.3 |
| Disseminated intravascular coagulopathy | 29 | 14 | 21 | 25 | |
| Shock | 78 | 78 | 82 | 76 | |
| Sepsis | 19 | 21 | 20 | 12 | |
| Wound infection | 30 | 25 | 29 | 24 | |
| Arrhythmia | 34 | 38 | 37 | 34 | |
| Myocardial infarction | 1 | 4 | 4 | 5 | |
| Acute respiratory distress syndrome | 39 | 42 | 47 | 38 | |
| Acute kidney injury | 14 | 30 | 20 | 17 | |
| Pneumonia | 38 | 33 | 47 | 29 | |
| Pulmonary embolism | 3 | 0 | 8 | 1 | |
| Transfusion-related acute lung injury | 1 | 1 | 1 | 3 |
Table 4.
Independent association between patient-specific mean red blood cell storage duration and secondary outcomes. All estimates are adjusted for age, renal disease, liver disease, initial blood pH, initial antiprothrombin time, and initial activated partial thromboplastin time. Estimates are reported with 95% confidence intervals in parentheses. Analyses reflect the same 259 patients that were included in the multivariable modeling for the primary outcome of in-hospital mortality. The reported measures of association reflect a one-day increase in patient mean red blood cell storage duration.
| Outcome variable | Model type | Measure of association | Estimate | P value |
|---|---|---|---|---|
| Duration of hospitalization (d) | Linear regression | Slope (95% CI) | −0.27 (−0.68, 0.13) | 0.18 |
| Duration of ICU stay (h) | Linear regression | Slope (95% CI) | −1.79 (−9.82, 6.24) | 0.66 |
| Duration of mechanical ventilation (h) | Linear regression | Slope (95% CI) | −0.91 (−7.13, 5.30) | 0.77 |
| Disseminated intravascular coagulopathy | Logistic regression | Odds ratio (95% CI) | 0.98 (0.93, 1.03) | 0.42 |
| Shock | Logistic regression | Odds ratio (95% CI) | 1.00 (0.95, 1.05) | 0.97 |
| Sepsis | Logistic regression | Odds ratio (95% CI) | 0.96 (0.91, 1.02) | 0.19 |
| Wound infection | Logistic regression | Odds ratio (95% CI) | 0.97 (0.93, 1.02) | 0.28 |
| Arrhythmia | Logistic regression | Odds ratio (95% CI) | 0.98 (0.93, 1.02) | 0.30 |
| Myocardial infarction | Logistic regression | Odds ratio (95% CI) | 1.09 (0.96, 1.25) | 0.18 |
| Acute respiratory distress syndrome | Logistic regression | Odds ratio (95% CI) | 1.00 (0.96, 1.05) | >0.99 |
| Acute kidney injury | Logistic regression | Odds ratio (95% CI) | 1.00 (0.94, 1.05) | 0.94 |
| Pneumonia | Logistic regression | Odds ratio (95% CI) | 0.99 (0.94, 1.03) | 0.57 |
| Pulmonary embolism | Logistic regression | Odds ratio (95% CI) | 1.10 (0.96, 1.25) | 0.15 |
| Transfusion-related acute lung injury | Logistic regression | Odds ratio (95% CI) | 1.06 (0.90, 1.26) | 0.47 |
About 22% of all patients suffered from disseminated intravascular coagulation, independent of blood storage duration, and the incidence of myocardial infarction appeared to increase with storage duration. A third of all patients suffered from infections (i.e., surgical site infection or pneumonia), while almost 40% of all patients developed acute respiratory distress syndrome with no particular prevalence towards a particular mean storage duration.
4. Discussion
We included patients given massive transfusion, defined by transfusion of 10 or more units of PRBCs within 24 hours. Only about 5% of the estimated annual 50 million trauma cases in the U.S. require this much blood, but they account for 10 to 15% of all transfused blood products [2] and accrue more than 4 billion dollars of health care cost [31]. Blood transfusion is a strong independent predictor of mortality in trauma and nontrauma patients [32], especially in massively transfused patients [33]. Our results are consistent in that mortality was high in our massively transfused patients, many of whom did not even live six hours. Among those who lived and thus qualified for our analysis, a third died during their initial hospitalization.
Two recent meta-analyses evaluated the relationship between PRBC storage duration and long-term mortality, multiple organ failure, in-hospital infections, duration of mechanical ventilation, and respiratory failure requiring ventilator support [34, 35]. The analysis suggested that storage duration did not affect outcome, but the authors noted considerable heterogeneity concerning patient populations, diversity of interventions, and measurement of clinical outcome. In contrast, Wang and colleagues analysed 21 studies including a total of 409,966 patients concluding that older blood is associated with increased mortality [36].
Patients having cardiovascular/CABG surgery are well studied, but both prospective and retrospective data conflict regarding the potential harm of prolonged stored blood products [1, 37–40]. The most recent large prospective trials in cardiac surgery patients [22] and critically ill patients [23] demonstrate no difference in outcomes including the incidence of multiorgan dysfunction syndrome or mortality [22], the incidence of major illnesses, duration of hemodynamic instability, renal or ventilator support, length of stay in the hospital, or transfusion reactions [23]. However, patients in both studies were typically given just one or two units of red cells.
Trauma patients are likely to experience multiorgan failure [24, 41], infection [28], kidney failure [27], pneumonia [42, 43], deep vein thrombosis [41], and death [27, 41, 43, 44]. Risk is presumably largely related to tissue injury, but the need for large amounts of blood products contributes [33].
Massively transfused patients have been underrepresented in previous studies. For example, only 13% of patients in Edgren's database [20] received more than five units of PRBCs, the RECESS trial [22] included only 145 patients who received more than eight units, and only 156 trauma patients in the ABLE trial were given more than four units of blood [23]. Even in the recently published INFORM trial [45] including 30,000 patients randomized to transfusion of short-term or long-term stored PRBCs, the number of trauma patients and massive transfused patients was negligible similar to other studies including even fewer massively transfused patients [24, 28, 42, 43]. Our analysis is the first focusing exclusively on patients who received a minimum of at least ten units of PRBCs within 24 hours, representing transfusion of 8,046 units in just 305 patients. Nonetheless, there was no association between mean storage duration of PRBCs and in-hospital mortality. Nor was there a differential effect amongst trauma and nontrauma patients.
Stored PRBCs undergo progressive structural and conformational changes associated with proposed subsequent worsening of quality, function, and viability of PRBC after transfusion [17, 46, 47]. Assessing effects of blood product age in our patient population receiving at least 10 units of PRBCs is challenging as the majority of patients do not receive exclusively old nor young blood products, but a mixture of both.
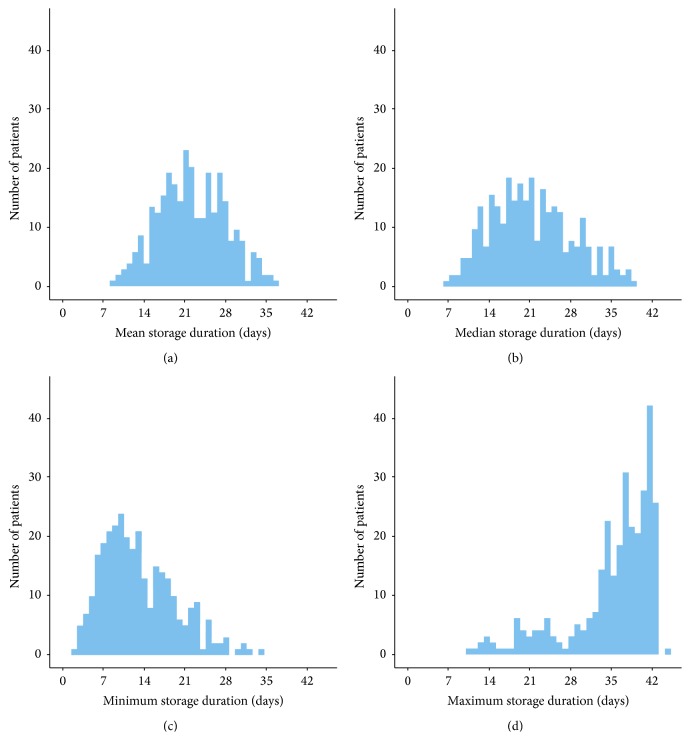
Unfortunately, no clear cut-off time has been defined after which changes during storage become clinically important with the result that various investigators have used arbitrary storage thresholds (i.e., 14 days [27], 21 days [22], or 28 days [41] of storage time) or based their analyses on the oldest unit [41]. As most blood banks in the U.S. and worldwide follow the inventory principle “first-in-first-out” to avoid outdating of stored blood products [23, 48], massively transfused patients typically receive PRBCs with a considerable range of storage durations because a single patient may use a considerable fraction of the matching units for an entire hospital, even in a major trauma centre like the University of Louisville Hospital. We therefore used mean storage time as a continuous variable, with a consequence that the difference between the first and fourth quartile of PRBC storage duration was only six days. However, histograms displaying the distribution of PRBC storage duration considering median, minimum, and maximum storage duration revealed a similar difference between patients receiving short or prolonged stored products (Figure 3). With the relative broad distribution of storage times of PRBCs/patient, it is not possible to determine whether a specific storage threshold exists (i.e., 35 days [49]) beyond which age will effect outcome in this particular patient population. While this relatively small range limits our ability to assess the specific effects of blood age on mortality, our conclusion that storage duration and mortality are unassociated probably applies broadly.
Figure 3.
Histograms of the observed values of patient RBC storage duration assembled taking (a) mean, (b) median, (c) minimum, and (d) maximum storage duration as dependent variable. The histogram indicates lack of discrimination between short and prolonged stored blood products.
Mortality within 6 hours after hospital admission increased is doubled in patients who receive 4 or more units of resuscitation fluid [50]. We excluded 36 patients who died within the first 6 hours after starting massive transfusion as most experienced such severe trauma (i.e., penetrating trauma to major vessels and organs with 50% of the patient during initial surgery) that the storage lesion of transfused red cells is unlikely to have caused their demise. In fact, some investigators postulate that some detrimental effects of prolonged red cell storage become evident more than two weeks after transfusion [1]. Our average observation time was 21 days (minimum: 1 day, maximum: 138 days) and therefore remains possible that we missed longer-term transfusion-related outcomes.
As in all observational studies—particularly, those involving emergency surgeries in trauma patients—missing data are a major concern. We reviewed the records of 498 patients who had massive blood transfusions. Despite all efforts, we were unable to retrieve complete data for many patients, a problem that has been noted in previous studies [51]. We therefore only included patients in whom more than 80% of all study-related data were available. A limitation of our analysis is that we cannot determine the effect of missing data in the patients we included, much less the effect of excluding patients who had much missing data.
In summary, our data suggest that the median storage time of PRBCs transfused is not associated with in-hospital mortality amongst patients given at least 10 units of PRBC within 24 hours.
Acknowledgments
Dr. Dalton's effort was supported by the Clinical and Translational Science Collaborative of Cleveland from the National Center for Advancing Translational Sciences (NCATS) component of the National Institutes of Health and NIH roadmap for Medical Research (KL2TR000440).
Disclosure
The contents of this publication are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Authors' Contributions
Daniel I. Sessler and Detlef Obal were responsible for study design and concept; Alexander Bautista, Theodore B. Wright, Janice Meany, Sunitha K. Kandadai, Benjamin Brown, Kareim Khalafalla, and Saeed Hashem were responsible for data collection; Jason W. Smith was responsible for assigning trauma scores and evaluation of trauma severity; Tayyeb M. Ayyoubi and Janice Meany were responsible for reviewing Blood Bank data; Jarrod E. Dalton and Detlef Obal were responsible for statistical analysis; Tayyeb M. Ayyoubi, Jarrod E. Dalton, Anupama Wadhwa, and Detlef Obal were responsible for data analysis; Alexander Bautista, Theodore B. Wright, Anupama Wadhwa, Daniel I. Sessler, and Detlef Obal were responsible for writing manuscript. Alexander Bautista and Theodore B. Wright contributed equally to this work.
References
- 1.Koch C. G., Li L., Sessler D. I., et al. Duration of red-cell storage and complications after cardiac surgery. New England Journal of Medicine. 2008;358(12):1229–1239. doi: 10.1056/NEJMoa070403. [DOI] [PubMed] [Google Scholar]
- 2.Como J. J., Dutton R. P., Scalea T. M., Edelman B. B., Hess J. R. Blood transfusion rates in the care of acute trauma. Transfusion. 2004;44(6):809–813. doi: 10.1111/j.1537-2995.2004.03409.x. [DOI] [PubMed] [Google Scholar]
- 3.Holcomb J. B., Jenkins D., Rhee P., et al. Damage control resuscitation: directly addressing the early coagulopathy of trauma. The Journal of trauma. 2007;62(2):307–310. doi: 10.1097/TA.0b013e3180324124. [DOI] [PubMed] [Google Scholar]
- 4.Cherkas D. Traumatic hemorrhagic shock: advances in fluid management. Emergency medicine practice. 2011;13(11):1–20. [PubMed] [Google Scholar]
- 5.Robinson III W. P., Ahn J., Stiffler A., et al. Blood transfusion is an independent predictor of increased mortality in nonoperatively managed blunt hepatic and splenic injuries. The Journal of trauma. 2005;58(3):437–445. doi: 10.1097/01.ta.0000153935.18997.14. [DOI] [PubMed] [Google Scholar]
- 6.Malone D. L., Dunne J., Tracy J. K., Putnam A. T., Scalea T. M., Napolitano L. M. Blood transfusion, independent of shock severity, is associated with worse outcome in trauma. The Journal of Trauma. 2003;54(5):898–907. doi: 10.1097/01.TA.0000060261.10597.5C. [DOI] [PubMed] [Google Scholar]
- 7.Taylor R. W., Manganaro L., O'Brien J., Trottier S. J., Parkar N., Veremakis C. Impact of allogenic packed red blood cell transfusion on nosocomial infection rates in the critically ill patient. Critical Care Medicine. 2002;30(10):2249–2254. doi: 10.1097/00003246-200210000-00012. [DOI] [PubMed] [Google Scholar]
- 8.Koch C. G., Li L., Duncan A. I., et al. Morbidity and mortality risk associated with red blood cell and blood-component transfusion in isolated coronary artery bypass grafting. Critical Care Medicine. 2006;34(6):1608–1616. doi: 10.1097/01.CCM.0000217920.48559.D8. [DOI] [PubMed] [Google Scholar]
- 9.Huber-Wagner S., Qvick M., Mussack T., et al. Massive blood transfusion and outcome in 1062 polytrauma patients: a prospective study based on the Trauma Registry of the German Trauma Society. Vox Sanguinis. 2007;92(1):69–78. doi: 10.1111/j.1423-0410.2006.00858.x. [DOI] [PubMed] [Google Scholar]
- 10.Holcomb J. B., Fox E. E., Wade C. E. The PRospective observational multicenter major trauma transfusion (PROMMTT) study. Journal of Trauma and Acute Care Surgery. 2013;75(1 , supplement 1):S1–S2. doi: 10.1097/TA.0b013e3182983876. [DOI] [PubMed] [Google Scholar]
- 11.Shander A., Hofmann A., Gombotz H., Theusinger O. M., Spahn D. R. Estimating the cost of blood: past, present, and future directions. Best Practice and Research: Clinical Anaesthesiology. 2007;21(2):271–289. doi: 10.1016/j.bpa.2007.01.002. [DOI] [PubMed] [Google Scholar]
- 12.Hamasaki N., Yamamoto M. Red blood cell function and blood storage. Vox Sanguinis. 2000;79(4):191–197. doi: 10.1159/000056729. [DOI] [PubMed] [Google Scholar]
- 13.Bennett-Guerrero E., Veldman T. H., Doctor A., et al. Evolution of adverse changes in stored RBCs. Proceedings of the National Academy of Sciences of the United States of America. 2007;104(43):17063–17068. doi: 10.1073/pnas.0708160104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cohen B., Matot I., Hemmings H. C. Aged erythrocytes: a fine wine or sour grapes? British Journal of Anaesthesia. 2013;111, supplement 1:i62–i70. doi: 10.1093/bja/aet405. [DOI] [PubMed] [Google Scholar]
- 15.Zimring J. C. Fresh versus old blood: are there differences and do they matter? American Society of Hematology. 2013;2013:651–655. doi: 10.1182/asheducation-2013.1.651. [DOI] [PubMed] [Google Scholar]
- 16.Frank S. M., Abazyan B., Ono M., et al. Decreased erythrocyte deformability after transfusion and the effects of erythrocyte storage duration. Anesthesia & Analgesia. 2013;116(5):975–981. doi: 10.1213/ANE.0b013e31828843e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Berezina T. L., Zaets S. B., Morgan C., et al. Influence of storage on red blood cell rheological properties. Journal of Surgical Research. 2002;102(1):6–12. doi: 10.1006/jsre.2001.6306. [DOI] [PubMed] [Google Scholar]
- 18.Zecher D., Cumpelik A., Schifferli J. A. Erythrocyte-derived microvesicles amplify systemic inflammation by thrombin-dependent activation of complement. Arteriosclerosis, Thrombosis, and Vascular Biology. 2014;34(2):313–320. doi: 10.1161/ATVBAHA.113.302378. [DOI] [PubMed] [Google Scholar]
- 19.Andreasen J. J., Dethlefsen C., Modrau I. S., et al. Storage time of allogeneic red blood cells is associated with risk of severe postoperative infection after coronary artery bypass grafting. European Journal of Cardio-Thoracic Surgery. 2011;39(3):329–334. doi: 10.1016/j.ejcts.2010.06.019. [DOI] [PubMed] [Google Scholar]
- 20.Edgren G., Kamper-Jørgensen M., Eloranta S., et al. Duration of red blood cell storage and survival of transfused patients. Transfusion. 2010;50(6):1185–1195. doi: 10.1111/j.1537-2995.2010.02583.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Saager L., Turan A., Dalton J. E., Figueroa P. I., Sessler D. I., Kurz A. Erythrocyte storage duration is not associated with increased mortality in noncardiac surgical patients: a retrospective analysis of 6,994 patients. Anesthesiology. 2013;118(1):51–58. doi: 10.1097/ALN.0b013e3182746ba4. [DOI] [PubMed] [Google Scholar]
- 22.Steiner M. E., Ness P. M., Assmann S. F., et al. Effects of red-cell storage duration on patients undergoing cardiac surgery. The New England Journal of Medicine. 2015;372(15):1419–1429. doi: 10.1056/NEJMoa1414219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lacroix J., Hébert P. C., Fergusson D. A., et al. Age of transfused blood in critically ill adults. The New England Journal of Medicine. 2015;372(15):1410–1418. doi: 10.1056/nejmoa1500704. [DOI] [PubMed] [Google Scholar]
- 24.Zallen G., Offner P. J., Moore E. E., et al. Age of transfused blood is an independent risk factor for postinjury multiple organ failure. The American Journal of Surgery. 1999;178(6):570–572. doi: 10.1016/s0002-9610(99)00239-1. [DOI] [PubMed] [Google Scholar]
- 25.Murrell Z., Haukoos J. S., Putnam B., Klein S. R. The effect of older blood on mortality, need for ICU care, and the length of ICU stay after major trauma. American Surgeon. 2005;71(9):781–785. doi: 10.1177/000313480507100918. [DOI] [PubMed] [Google Scholar]
- 26.Keller M. E., Jean R., LaMorte W. W., Millham F., Hirsch E. Effects of age of transfused blood on length of stay in trauma patients: a preliminary report. Journal of Trauma. 2002;53(5):1023–1025. doi: 10.1097/00005373-200211000-00037. [DOI] [PubMed] [Google Scholar]
- 27.Weinberg J. A., McGwin G., Jr., Griffin R. L., et al. Age of transfused blood: an independent predictor of mortality despite universal leukoreduction. The Journal of Trauma. 2008;65(2):279–284. doi: 10.1097/TA.0b013e31817c9687. [DOI] [PubMed] [Google Scholar]
- 28.Offner P. J., Moore E. E., Biffl W. L., Johnson J. L., Silliman C. C. Increased rate of infection associated with transfusion of old blood after severe injury. Archives of Surgery. 2002;137(6):711–717. doi: 10.1001/archsurg.137.6.711. [DOI] [PubMed] [Google Scholar]
- 29.Kiraly L. N., Underwood S., Differding J. A., Schreiber M. A. Transfusion of aged packed red blood cells results in decreased tissue oxygenation in critically injured trauma patients. Journal of Trauma—Injury, Infection and Critical Care. 2009;67(1):29–32. doi: 10.1097/TA.0b013e3181af6a8c. [DOI] [PubMed] [Google Scholar]
- 30.Akaike H. Akaike’s Information Criterion. Berlin, Germany: Springer; 2011. [Google Scholar]
- 31.Corso P., Finkelstein E., Miller T., Fiebelkorn I., Zaloshnja E. Incidence and lifetime costs of injuries in the United States. Injury Prevention. 2006;12(4):212–218. doi: 10.1136/ip.2005.010983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Turan A., Yang D., Bonilla A., et al. Morbidity and mortality after massive transfusion in patients undergoing non-cardiac surgery. Canadian Journal of Anesthesia. 2013;60(8):761–770. doi: 10.1007/s12630-013-9937-3. [DOI] [PubMed] [Google Scholar]
- 33.Johnson D. J., Scott A. V., Barodka V. M., et al. Morbidity and mortality after high-dose transfusion. Anesthesiology. 2016;124(2):387–395. doi: 10.1097/ALN.0000000000000945. [DOI] [PubMed] [Google Scholar]
- 34.Brunskill S. J., Wilkinson K. L., Doree C., Trivella M., Stanworth S. Transfusion of fresher versus older red blood cells for all conditions. Cochrane Database of Systematic Reviews. 2015;5(5) doi: 10.1002/14651858.cd010801.pub2.CD010801 [DOI] [PubMed] [Google Scholar]
- 35.Ng M. S. Y., Ng A. S. Y., Chan J., Tung J.-P., Fraser J. F. Effects of packed red blood cell storage duration on post-transfusion clinical outcomes: a meta-analysis and systematic review. Intensive Care Medicine. 2015;41(12):2087–2097. doi: 10.1007/s00134-015-4078-5. [DOI] [PubMed] [Google Scholar]
- 36.Wang D., Sun J., Solomon S. B., Klein H. G., Natanson C. Transfusion of older stored blood and risk of death: a meta-analysis. Transfusion. 2012;52(6):1184–1195. doi: 10.1111/j.1537-2995.2011.03466.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Vamvakas E. C., Carven J. H. Transfusion and postoperative pneumonia in coronary artery bypass graft surgery: effect of the length of storage of transfused red cells. Transfusion. 1999;39(7):701–710. doi: 10.1046/j.1537-2995.1999.39070701.x. [DOI] [PubMed] [Google Scholar]
- 38.Basran S., Frumento R. J., Cohen A., et al. The association between duration of storage of transfused red blood cells and morbidity and mortality after reoperative cardiac surgery. Anesthesia and Analgesia. 2006;103(1):15–20. doi: 10.1213/01.ane.0000221167.58135.3d. [DOI] [PubMed] [Google Scholar]
- 39.Wasser M. N. J. M., Houbiers J. G. A., D'Amaro J., et al. The effect of fresh versus stored blood on post-operative bleeding after coronary bypass surgery: a prospective randomized study. British Journal of Haematology. 1989;72(1):81–84. doi: 10.1111/j.1365-2141.1989.tb07656.x. [DOI] [PubMed] [Google Scholar]
- 40.Yap C.-H., Lau L., Krishnaswamy M., Gaskell M., Yii M. Age of transfused red cells and early outcomes after cardiac surgery. Annals of Thoracic Surgery. 2008;86(2):554–559. doi: 10.1016/j.athoracsur.2008.04.040. [DOI] [PubMed] [Google Scholar]
- 41.Spinella P. C., Carroll C. L., Staff I., et al. Duration of red blood cell storage is associated with increased incidence of deep vein thrombosis and in hospital mortality in patients with traumatic injuries. Critical Care. 2009;13(5, article R151) doi: 10.1186/cc8050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vandromme M. J., McGwin G., Weinberg J. A. Blood transfusion in the critically ill: does storage age matter? Scandinavian Journal of Trauma, Resuscitation and Emergency Medicine. 2009;17, article 35 doi: 10.1186/1757-7241-17-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Weinberg J. A., McGwin G., Marques M. B., et al. Transfusions in the less severely injured: does age of transfused blood affect outcomes? The Journal of Trauma: Injury, Infection, and Critical Care. 2008;65(4):794–798. doi: 10.1097/TA.0b013e318184aa11. [DOI] [PubMed] [Google Scholar]
- 44.Purdy F. R., Tweeddale M. G., Merrick P. M. Association of mortality with age of blood transfused in septic ICU patients. Canadian Journal of Anaesthesia. 1997;44(12):1256–1261. doi: 10.1007/BF03012772. [DOI] [PubMed] [Google Scholar]
- 45.Heddle N. M., Cook R. J., Arnold D. M., et al. Effect of short-term vs. long-term blood storage on mortality after transfusion. The New England Journal of Medicine. 2016;375(20):1937–1945. doi: 10.1056/NEJMoa1609014. [DOI] [PubMed] [Google Scholar]
- 46.D'almeida M. S., Jagger J., Duggan M., White M., Ellis C., Chin-Yee I. H. A comparison of biochemical and functional alterations of rat and human erythrocytes stored in CPDA-1 for 29 days: implications for animal models of transfusion. Transfusion Medicine. 2000;10(4):291–303. doi: 10.1046/j.1365-3148.2000.00267.x. [DOI] [PubMed] [Google Scholar]
- 47.Grimshaw K., Sahler J., Spinelli S. L., Phipps R. P., Blumberg N. New frontiers in transfusion biology: identification and significance of mediators of morbidity and mortality in stored red blood cells. Transfusion. 2011;51(4):874–880. doi: 10.1111/j.1537-2995.2011.03095.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.McDaniel L. M., Triulzi D. J., Cramer J., et al. Massive transfusion protocol activation does not result in preferential use of older red blood cells. Journal of Blood Transfusion. 2014;2014:5. doi: 10.1155/2014/328967.328967 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Goel R., Johnson D. J., Scott A. V., et al. Red blood cells stored 35 days or more are associated with adverse outcomes in high-risk patients. Transfusion. 2016;56(7):1690–1698. doi: 10.1111/trf.13559. [DOI] [PubMed] [Google Scholar]
- 50.Rahbar E., Fox E. E., Del Junco D. J., et al. Early resuscitation intensity as a surrogate for bleeding severity and early mortality in the PROMMTT study. Journal of Trauma and Acute Care Surgery. 2013;75(1, supplement 1):S16–S23. doi: 10.1097/TA.0b013e31828fa535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Trickey A. W., Fox E. E., Del Junco D. J., et al. The impact of missing trauma data on predicting massive transfusion. Journal of Trauma and Acute Care Surgery. 2013;75(1, supplement 1):S68–S74. doi: 10.1097/TA.0b013e3182914530. [DOI] [PMC free article] [PubMed] [Google Scholar]