Abstract
Whether the specific blood transfer conductance for nitric oxide (NO) with hemoglobin (θNO) is finite or infinite is controversial but important in the calculation of alveolar capillary membrane conductance (DmCO) and pulmonary capillary blood volume (VC) from values of lung diffusing capacity for carbon monoxide (DLCO) and nitric oxide (DLNO). In this review, we discuss the background associated with θNO, explore the resulting values of DmCO and VC when applying either assumption, and investigate the mathematical underpinnings of DmCO and VC calculations. In general, both assumptions yield reasonable rest and exercise DmCO and VC values. However, the finite θNO assumption demonstrates increasing VC, but not DmCO, with submaximal exercise. At relatively high, but physiologic, DLNO/DLCO ratios both assumptions can result in asymptotic behavior for VC values, and under the finite θNO assumption, DmCO values. In conclusion, we feel that the assumptions associated with a finite θNO require further in vivo validation against an established method before widespread research and clinical use.
Keywords: Lung diffusing capacity, alveolar capillary membrane conductance, pulmonary capillary blood volume, exercise, in vivo validation
1. INTRODUCTION
There remains significant uncertainty as to the correct specific blood transfer conductance for nitric oxide (NO) with hemoglobin (θNO) when studying lung diffusing capacity even in light of the ERS task force findings (Zavorsky et al., 2017). Specifically, there is currently a debate as to whether to assume an infinite or finite θNO when calculating alveolar-capillary membrane conductance (DmCO) and pulmonary-capillary blood volume (VC) from measures of lung diffusing capacity for CO (DLCO) and NO (DLNO). While our laboratory has consistently held the original assumption that θNO is effectively infinite, other groups have begun applying a finite value for θNO of 4.5 mlCO/min/mmHg/mlblood measured via an in vitro study and supported by animal and human studies not designed to calculate an exact θNO value (Borland and Cox, 1991; Borland et al., 2010; Guenard et al., 2016; Zavorsky et al., 2014). Based on this limited work, a task force has recently recommended the use of a finite θNO; however, we believe these recommendations, including those concerning θNO and also the correct DmNO/DmCO ratio, are premature (Zavorsky et al., 2017). In this manuscript, we will begin by summarizing the evolution of, and the scientific reasoning behind, the original assumption that θNO is effectively infinite. Next, using both published and preliminary data from our laboratory, we will show in detail the effect of assuming an infinite θNO vs. using a finite value of 4.5 mlCO/min/mmHg/mlblood on calculation of DmCO and VC values in vivo in humans. We will do this for lung diffusing capacity data collected using different techniques (i.e. rebreathe and single-breath) and in different populations (i.e. healthy and heart failure, young and old age) at rest and during exercise. Furthermore, we will extend these findings to an investigation of the mathematical limits within which each assumption yields reasonable, physiologic values for DmCO and VC; i.e., positive values on the same order of magnitude as values previously reported in the literature. Finally, we will discuss the implications of our findings and offer insight into future research, as well as clinical practice, regarding the use of an infinite versus a finite θNO in the calculation of DmCO and Vc in vivo in humans. As we will show, both assumptions yield reasonable values for DmCO and VC at rest. However, the assumption that θNO is finite can yield values which do not increase with submaximal exercise; this is clearly a concern, as DmCO and VC would be anticipated to increase during the exercise levels included in these data. Additionally, using a finite θNO requires more assumptions during calculations (α/Krogh coefficient, θCO equation, θNO) than applying an infinite θNO (α/Krogh coefficient, θCO equation). As such, while the use of a finite θNO value may have merit, we urge caution in its application for the calculation of DmCO and Vc in humans until the method can be properly validated against the multiple O2 tension method. Overall, we feel that it is more important to interpret DmCO and VC as physiologic variables rather than anatomical measurements and to interpret changes over time or between groups rather than to focus on the specific method used.
1.1 Theory and evidence for an infinite vs. finite θNO
In our laboratory, we use two methods for the determination of DLCO: 1) a single breath technique, and 2) a rebreathe technique. The single breath technique for measuring DLCO was first described in 1909 (Krogh and Krogh, 1910; Krogh, 1915), but the methodology currently in common use was established in 1954 (Forster et al., 1954). This technique requires the participant to take a deep inspiration from residual volume to total lung capacity of a test gas containing approximately 10% helium, 0.3% CO, 21% oxygen, and balance nitrogen, followed by a short breath hold (typically 4 to 10 seconds), with a subsequent swift expiration. Simply put, the ratio of inspired to expired CO concentrations, after taking into account dead space and alveolar mixing, yields the diffusing capacity of the lungs for CO (DLCO). The rebreathe technique used in our laboratory to measure DLCO requires participants to rebreathe from a bag containing 9% Helium, 0.3% C18O, 35% O2, and balance nitrogen for 8 to 10 tidal breaths (Ceridon et al., 2010; Meyer et al., 1990; Sackner et al., 1975). The assumptions of the rebreathe technique are identical to that of the single breath method. However, the rebreathe technique often yields lower values for DmCO and VC; while this inconsistency is not completely understood, it is not the focus of the manuscript and hence, we will separate results for both techniques for clarity.
In 1957, Roughton and Forster extended this technique and established a method for determining DmCO and VC by measuring DLCO at multiple O2 concentrations (Roughton and Forster, 1957). In short, because CO and hemoglobin competitively bind hemoglobin, a higher partial pressure of oxygen (PO2) in the pulmonary capillaries yields a lower DLCO. Taking advantage of this, it is then possible to separate the contribution of resistances to gas transfer due to both the alveolar capillary membrane (Dm) and the pulmonary capillary blood volume (Vc) by graphing 1/DLCO against 1/θCO and fitting the resulting points. Specifically, DmCO will be equal to the inverse of the y-intercept and VC will be equal to the inverse of the slope of the regression line, according to the following equation developed by Roughton and Forster (Roughton and Forster, 1957):
However, this method is less than ideal, as it is not only time-consuming, but also requires the assumption that measurements of DLCO separated by multiple minutes represent an identical physiologic state unaffected by varying oxygen tensions, i.e. the same CO distribution throughout the lung during breath hold and the same cardiac output during the maneuver. Despite these shortcomings, this multiple-O2 tension method is considered the ‘gold standard’ for the calculation of DmCO and VC.
In the late 1980’s, a method was developed in which DLNO is measured simultaneously with DLCO by the addition of 40 ppm NO to the gas mixture and has quickly become the technique of choice as it removes the issues described above associated with the multiple O2 tension method (Borland and Higenbottam, 1989; Guenard et al., 1987). The theory behind this method is that the rate of reaction of NO with hemoglobin in vitro is extremely rapid, approximately two orders of magnitude greater than θCO (Borland and Higenbottam, 1989; Hakim et al., 1996).. Furthermore it is assumed that the extremely rapid reaction of NO with hemoglobin extends to the intact red cell in vivo. In this way, the resistance to NO transfer from the alveoli onto hemoglobin can be assumed to be only dependent on alveolar capillary membrane resistance and independent of the volume of pulmonary capillary blood present. This technique simplifies calculation of DmCO and VC, as it does not require tests to be performed at several oxygen tensions, therefore removing the necessity of assuming that the physiologic state is the same across multiple trials and also shortening the time required to obtain data (detailed calculations are described in sections 2.1–2 “Assumptions and Calculations”).
More recently, however, it has been argued that the assumption that θNO is infinite is not valid. Indeed, it has been demonstrated by multiple groups that the specific blood transfer conductance for NO with free hemoglobin is anywhere from 100 to over 1000 times faster than that with red blood cells, suggesting that the red blood cell does in fact have a meaningful resistance to the diffusion of NO. These studies have been completed in several animal species and using various methods (Azarov et al., 2011; Borland et al., 2006; Borland et al., 2010; Carlsen and Comroe, 1958; Deonikar and Kavdia, 2010; Liu et al., 1998; Vaughn et al., 2000). Accordingly, it has been argued by some researchers that θNO cannot, by definition, be infinite as a red cell resistance does in fact exist. Indeed, several in vitro studies find evidence for a finite specific blood transfer conductance for NO (Azarov et al., 2011; Sakai et al., 2008); however, these studies do not provide evidence as to the appropriate value for in vivo experiments. Furthermore, the in vivo value recently recommended for θNO by the ERS task force is based on only three papers. Each of these papers has limitations; either the studies were performed in vitro or in animals, or the study has a large degree of variability in the value obtained (for further discussion, see section 3.3 “Required assumptions/problems of each method”) (Borland et al., 2010; Carlsen and Comroe, 1958; Guenard et al., 2016). Importantly, we recognize that it is likely impossible to directly measure θNO in vivo in humans, and as such these studies are meaningful first steps in determining the value of θNO (currently suggested to be 4.5 mlCO/min/mmHg/mlblood). However, we feel that this θNO value needs to be validated and/or optimized with respect to a method that does not utilize NO as a means to gain confidence in its application\.
So, the question becomes: should θNO be considered infinite or finite in the calculation of DmCO and VC? In order to investigate the outcomes of both methods, we have calculated DmCO and VC while applying both a finite and infinite θNO for over 750 DLCO and DLNO measurements from our laboratory. What follows includes an overview of these calculations, details on the dataset used, and the outcomes of this analysis.
2. METHODS
2.1 Assumptions and Calculations - Infinite θNO
As we and others have demonstrated previously, the calculation of DmCO and VC when assuming that θNO is infinite is critically dependent on two key considerations. First, it is essential that the correct equation for calculating θCO is chosen. Second, the ratio of DmNO to DmCO (termed α) must be established. While there exists a theoretical value for α that is based on the molecular weights and solubilities in water of both NO and CO (termed the Krogh coefficient), numerous studies utilizing an infinite θNO have experimentally determined that α is actually greater than 1.97 (Magini et al., 2013; Tamhane et al., 2001). Accordingly, we have previously experimentally determined the best θCO equation and α value to be used, for both the rebreathe and single breath techniques (Ceridon et al., 2010; Coffman et al., 2016), by systematically comparing the resulting DmCO and VC values against those obtained via the original multiple O2 tension method. From both of these studies, our laboratory has concluded that the θCO equation described by Reeves and Park is ideal, while the optimal α value is dependent on the technique used; ~2.26 for rebreathe and ~4.40 for single breath. This difference can be understood by recognizing that the single breath method, in our laboratory and others, often yields DLNO values somewhat higher than the rebreathe method (Ceridon et al., 2010; Ceridon et al., 2011; Coffman et al., 2016; Snyder et al., 2007; Zavorsky et al., 2014; Zavorsky and Murias, 2006; Zavorsky et al., 2004). Of note, while this inconsistency in DLNO values between the two methods in not completely understood, it is not the focus of this manuscript. Importantly, the chosen α value for each method ensures that calculated DmCO values are in agreement with those obtained via the original multiple O2 tension method, which does NOT rely on measures of DLNO. Details of the calculation of DmCO and VC, under the assumption that θNO is effectively infinite, are as follows:
where α = 2.26 (rebreathe) or α = 4.40 (single breath)
where (Reeves and Park, 1992)
2.2 Assumptions and Calculations - Finite θNO
When calculating DmCO and VC using a finite θNO, three assumptions must be established. First, the correct θCO equation must be determined; Zavorksy et al. have chosen to use the equation reported by Guenard in 2016 (Forster, 1987; Guenard et al., 2016). Second, the α ratio must be determined; most groups that assume a finite θNO have chosen to use an approximation of the Krogh coefficient, equal to 2. This Krogh coefficient, and the α ratio used under the infinite θNO assumption, are similar, as they both convert DmNO to DmCO. The relationship of α and the Krogh coefficient and why α differs between the methods is discussed later (see Fig. 4, as well as section 3.3 “Required assumptions/problems of each method”). Third, the value for θNO must be chosen; θNO is currently set to a value of 4.5 mlCO/min/mmHg/mlblood (see section 3.3 “Required assumptions/problems of each method “ for more details) (Carlsen and Comroe 1958). Details of the calculation of DmCO and VC, under the assumption that θNO is finite, are as follows:
where (Guenard et al., 2016)
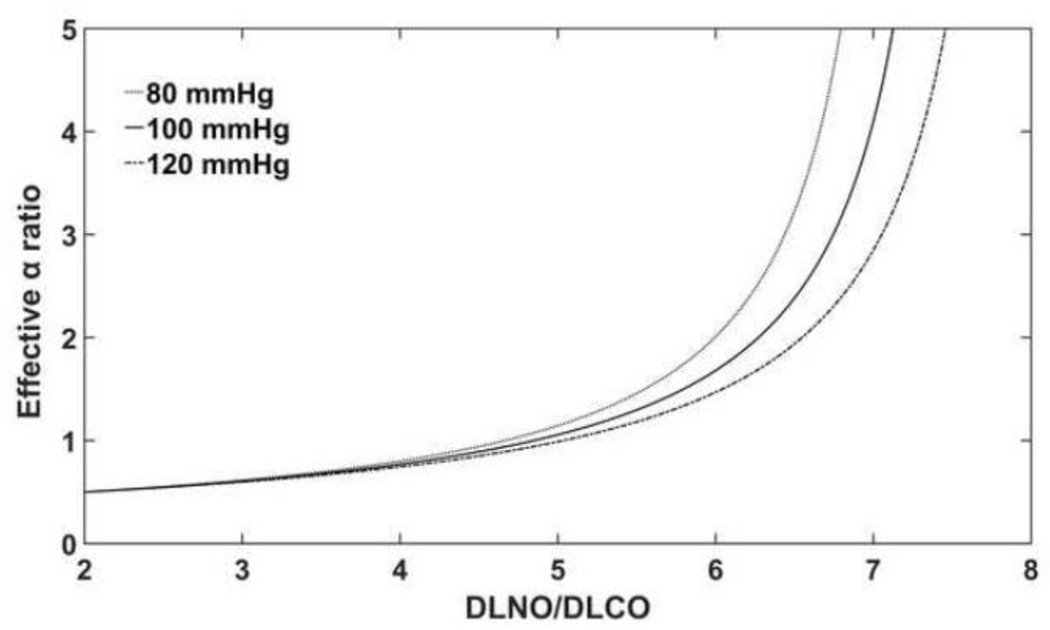
Figure 4. Effective α ratio for the conversion of DLNO to DmCO for the finite θNO assumption.
The effective α ratio, which converts DLNO directly to DmCO for the finite θNO assumption, was calculated for a range for DLNO/DLCO ratios. The resulting effective α ratio is lower than the Krogh Coefficient (1.97) because the finite θNO assumption calculations only factor in the theoretical red blood cell resistance to the transfer of NO. The resulting effective α ratio is also lower than the α ratio used under the infinite θNO assumption (2.26) for DLNO/DLCO ratios below ~6.5.
3. RESULTS
3.1 Effect of infinite vs. finite θNO assumption on actual data
In order to determine the effect of a finite vs. infinite θNO on calculated values of DmCO and VC, the data discussed next includes over 750 observations (an observation is a single, simultaneous measurement of DLCO and DLNO on a study participant) from our laboratory using both the rebreathe and single breath methods, from which we have calculated DmCO and VC using both the infinite and finite θNO assumptions described above (see sections 2.1–2 “Assumptions and Calculations”). Table 1 details the source of these data points; this includes data from healthy and heart failure participants, younger and older individuals, and rest as well as submaximal or incremental exercise trials. Therefore, these data span multiple variations in potential values of DLCO and DLNO, as well as DmCO and VC (Table 2).
Table 1.
Source of over 750 DLCO and DLNO observations
| Study ID | Age (y) | Height (cm) | Weight (kg) | BMI (kg/m2) | Exercise | Condition |
|---|---|---|---|---|---|---|
| Rebreathe | ||||||
| 1. | 66.5 ± 10.0 | 172.4 ± 10.3 | 82.2 ± 17.6 | 27.8 ± 4.4 | -- | HF + Control |
| 2. | 59.7 ± 10.6 | 174.5 ± 8.9 | 85.8 ± 17.6 | 28.1 ± 5.0 | -- | HF + Control |
| 3. | 25.9 ± 4.0 | 176.4 ± 11.7 | 71.3 ± 10.5 | 22.8 ± 2.1 | Incremental to 70% Wpeak | Healthy |
| 4. | 46.1 ± 20.6 | 176.2 ± 5.1 | 75.1 ± 7.4 | 24.2 ± 2.4 | Incremental to 90% Wpeak | Healthy |
| Average | 49.6 ± 19.6 | 175.3 ± 8.2 | 78.7 ± 14.1 | 25.6 ± 4.1 | -- | -- |
| Single Breath | ||||||
| 5. | 25.1 ± 2.4 | 180.7 ± 6.4 | 74.0 ± 10.1 | 22.6 ± 2.4 | Constant @ 80 Watts | Healthy |
| 6. | 26.9 ± 3.3 | 173.5 ± 7.4 | 65.4 ± 6.8 | 21.7 ± 1.5 | Constant @ 82 ± 27 Watts | Healthy |
| Average | 25.9 ± 2.9 | 177.7 ± 7.7 | 70.6 ± 9.9 | 22.3 ± 2.1 | -- | -- |
Values are reported as mean ± SD. BMI, body mass index; HF, heart failure; Wpeak, peak work rate.
Table 2.
DLCO/DLNO data and resulting DmCO and VC values calculated via both the infinite and finite θNO assumptions
| Infinite Method | Finite Method | P-value | ||
|---|---|---|---|---|
| Rebreathe | ||||
| DLCO | 26.5 ± 11.9 | |||
| Range | 5.9 – 72.1 | |||
| DLNO | 93.1 ± 40.0 | |||
| Range | 15.1 – 276.9 | |||
| DLNO/DLCO | 3.58 ± 0.59 | |||
| Range | 2.33 – 7.03 | |||
| DmCO | 41.2 ± 17.7 | 63.8 ± 31.1 | < 0.001 | |
| Range | 6.7 – 122.5 | 8.33 – 319.8 | ||
| VC | 85.8 ± 46.3 | 89.8 ± 43.3 | 0.111 | |
| Range | 18.1 – 497.2 | 20.1 – 253.3 | ||
| Single Breath | ||||
| DLCO | 40.7 ± 8.2 | |||
| Range | 29.3 – 68.6 | |||
| DLNO | 217.7 ± 36.8 | |||
| Range | 165.4 – 335.4 | |||
| DLNO/DLCO | 5.39 ± 0.43 | |||
| Range | 4.59 – 6.62 | |||
| DmCO | 49.5 ± 8.4 | 281.7 ± 81.5 | < 0.001 | |
| Range | 37.6 – 76.2 | 183.6 – 55.8 | ||
| VC | 223.2 ± 141.2 | 82.5 ± 18.7 | < 0.001 | |
| Range | 73.0 – 878.5 | 55.8 – 144.4 | ||
Values are reported as mean ± SD. Values include all rest, submaximal, and incremental exercise observations as well as data from both healthy individuals and heart failure patients. DLCO, lung diffusing capacity for carbon monoxide; DLNO, lung diffusing capacity for nitric oxide; DmCO, alveolar capillary membrane conductance; VC, pulmonary capillary blood volume.
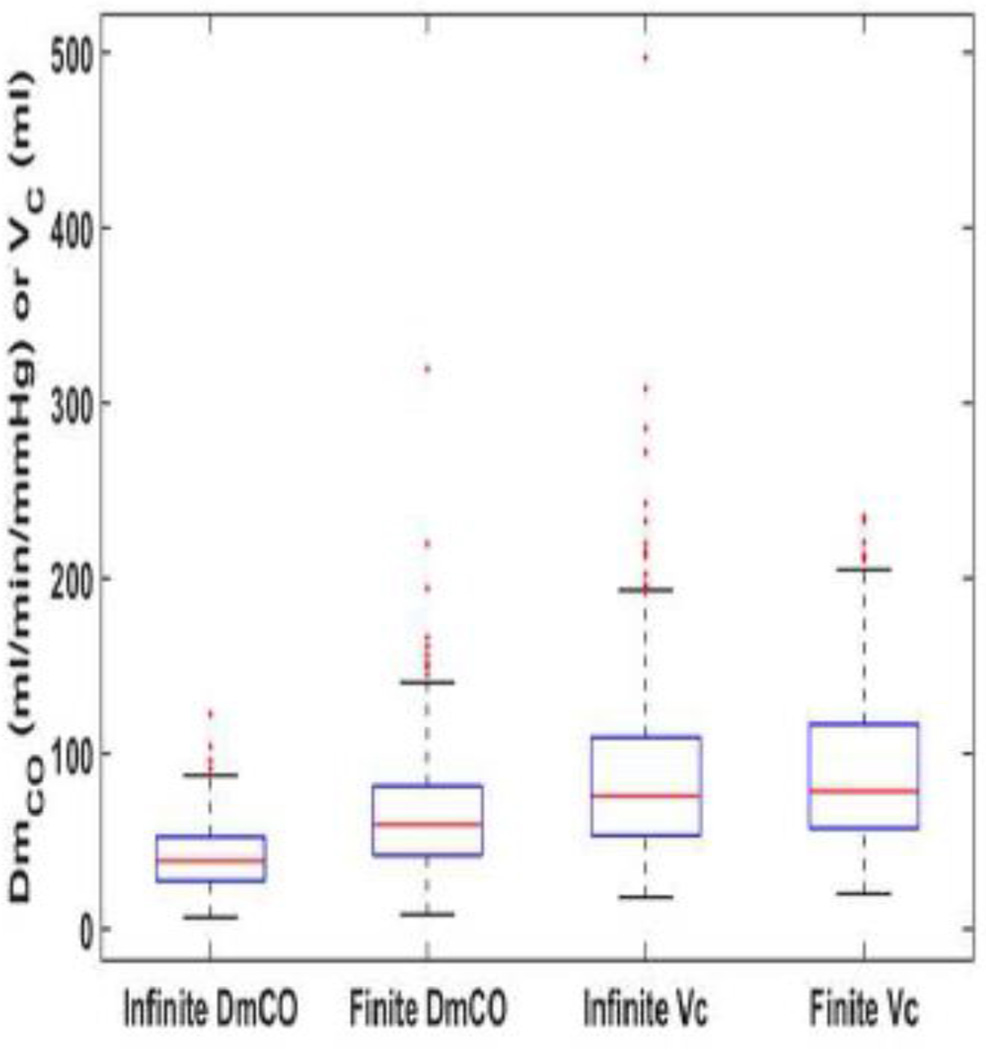
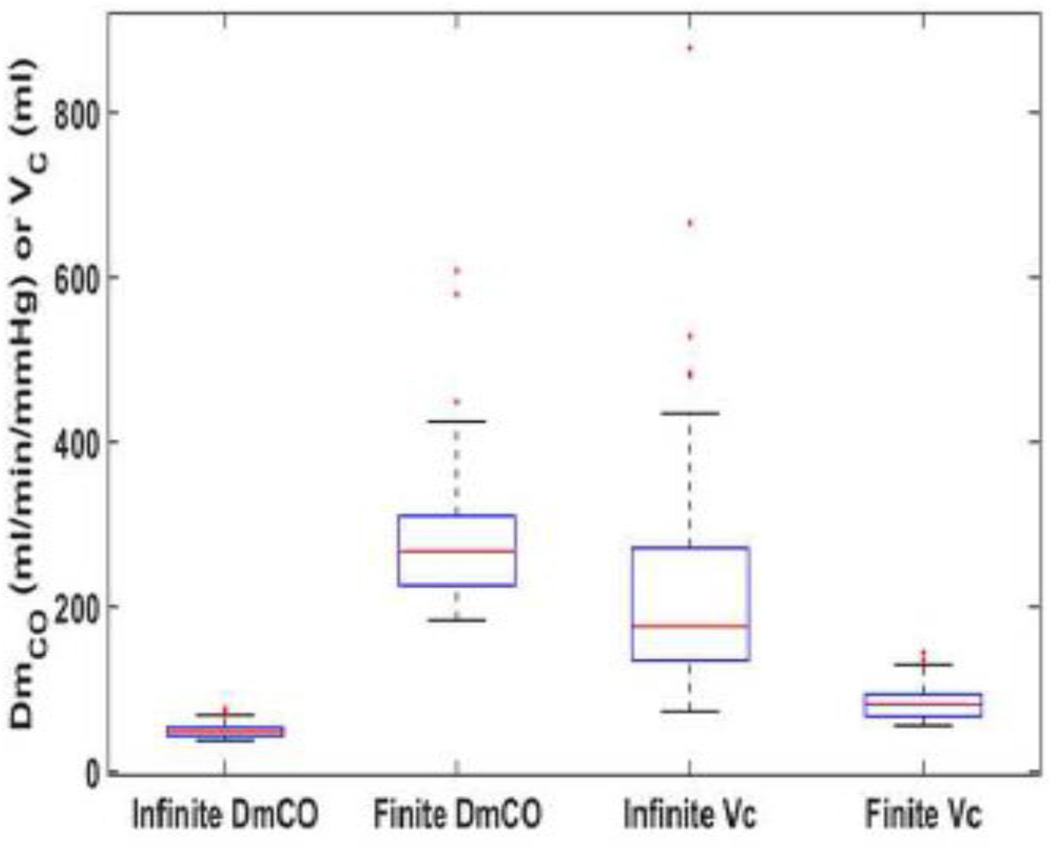
Figure 1 and 2, as well as Table 2, show the median and distribution for DmCO and VC values calculated assuming an infinite θNO and a finite θNO using a rebreathe (Table 2, Figure 1) or single breath (Table 2, Figure 2) technique. Using the rebreathe method (Figure 1), both the infinite and finite θNO assumptions yield DmCO and VC values that qualitatively appear to be grouped in a physiological range and incorporate very few outliers. However, the DmCO values are statistically higher using the finite vs. infinite assumption. Quantitatively, the means, standard deviations, and number of outliers (defined as ± 2.7 standard deviations from the mean), are as follows: for DmCO: Infinite DmCO, 41.2 ± 17.7 ml/min/mmHg, 4 outliers; Finite DmCO, 63.8 ± 31.1 ml/min/mmHg, 12 outliers (Infinite vs. Finite, p < 0.001); for VC: Infinite VC, 85.8 ± 46.3 ml, 14 outliers; Finite VC, 89.8 ± 43.3 ml, 6 outliers (Infinite vs. Finite, p = 0.111). Using the single breath method (Figure 2), the findings are similar; DmCO and VC under both an infinite and finite θNO assumption are grouped in a physiologic range with few outliers. However, the mean DmCO values are statistically higher, and the VC values are statistically lower, using the finite vs. infinite assumption. The means, standard deviations, and number of outliers for the single breath method are as follows: for DmCO: Infinite DmCO, 49.5 ± 8.4 ml/min/mmHg, 3 outliers; Finite DmCO, 281.7 ± 81.5 ml/min/mmHg, 3 outliers (p < 0.001); for VC: Infinite VC, 223.2 ± 141.2 ml, 5 outliers; Finite VC, 82.5 ± 18.7 ml, 2 outliers (p < 0.001).
Figure 1. DmCO and VC values calculated from rebreathe data using both the finite and infinite θNO assumptions.
Both assumptions yield reasonable values for DmCO and VC – i.e., positive and within range of previously reported values. Outliers (red +) are defined as ± 2.7 standard deviations from the mean. Finite DmCO values are significantly greater that infinite DmCO values (p < 0.001); VC values are not statistically different. DmCO, alveolar capillary membrane conductance; VC, pulmonary capillary blood volume.
Figure 2. DmCO and VC values calculated from single breath data using both the finite and infinite θNO assumptions.
Both assumptions yield reasonable values for DmCO and VC – i.e., positive and within range of previously reported values. Outliers (red +) are defined as ± 2.7 standard deviations from the mean. Finite DmCO values are significantly greater, and finite VC values are significantly lower, that the infinite assumption values (both p < 0.001). DmCO, alveolar capillary membrane conductance; VC, pulmonary capillary blood volume.
Such a difference in results between the finite and infinite assumption may complicate the ability to implement measures of DmCO and VC clinically. This is in contrast to the research environment, where we feel that the absolute values are of less importance, whereas the ability to observe changes between research groups or after an intervention is of the utmost importance. However, in order to implement DmCO and VC clinically, a single method to be implemented across the entire practice would simply need to be chosen. We discuss this idea further later (see section 4.3 “Conclusions”).
3.2 Exercise responses
In healthy humans, exercise is associated with an increase in cardiac output and pulmonary perfusion pressure that causes both recruitment of under-perfused pulmonary capillaries and distension of already perfused pulmonary blood vessels, as evidenced by an increase in DLCO, DmCO, and VC (La Gerche et al., 2010; Tamhane et al., 2001; Taylor et al., 2014). Therefore, it is crucial that measures of DmCO and Vc increase accordingly in response to exercise, regardless of whether θNO is assumed to be infinite or finite.
We have simultaneously assessed DLCO and DLNO using the single-breath technique at rest and during submaximal exercise in two separate studies in our laboratory (Coffman et al., 2016). In the first study, DLCO and DLNO were measured in duplicate at rest and during cycle exercise at 80W in 11 healthy subjects (Table 1, #5). In the second study, DLCO and DLNO were again measured in duplicate at rest and during cycle exercise at an intensity designed to elicit a doubling of resting cardiac output and at least ~70% of age predicted heart-rate maximum. This study was performed in 8 healthy subjects (Table 1, #6). For the present analyses, the data from both studies were pooled, yielding a mean submaximal workload of 80 W, range 40 – 130 W.
In order to determine if DmCO and VC significantly increased with submaximal exercise, we performed a student’s paired t-test on the data calculated under both infinite and finite θNO assumptions. Table 3 shows that, when assuming an infinite θNO, DmCO and VC increased significantly with submaximal exercise, as would be expected (DmCO at rest 45.6 ± 6.5 vs. exercise 53.7 ± 8.2 ml/min/mmHg, P < 0.001; VC at rest 162.6 ± 68.6 vs. exercise 288.9 ± 168.9 ml, P < 0.001). On the other hand, only VC significantly increased under the finite θNO assumption, whereas DmCO was statistically unchanged (DmCO at rest 279.1 ± 95.4 vs. exercise 284.5 ± 64.2 ml/min/mmHg, P = 0.498; VC at rest 72.7 ± 12.9 vs exercise 93.1 ± 18.3 ml, P < 0.001). This is clearly concerning, as DmCO is expected to rise in concert with Vc with increasing exercise intensity. Thus, the lack of response suggests a likely flaw with the determination of these values when assuming a finite θNO (Lewis et al., 1958; Tamhane et al., 2001).
Table 3.
Rest and submaximal exercise values for DmCO and VC using the single breath technique calculated via both the infinite and finite θNO assumptions
| Rest | Exercise (80 ± 17 W) | Absolute Change | % Change | P-value | ||
|---|---|---|---|---|---|---|
| Infinite Method | ||||||
| DmCO | 45.6 ± 6.5 | 53.7 ± 8.2 | 7.7 ± 3.6 | 16.9 ± 7.3 | < 0.001 | |
| Range | 37.6 – 63.7 | 42.0 – 76.2 | 1.4 – 18.8 | 2.9 – 31.4 | ||
| VC | 162.6 ± 68.6 | 288.9 ± 168.9 | 129.0 ± 160.7 | 92.0 ± 112.2 | < 0.001 | |
| Range | 73.0 – 427.8 | 109.7 – 878.5 | −119.5 – 725.1 | −52.1 – 472.4 | ||
| Finite Method | ||||||
| DmCO | 279.1 ± 95.4 | 284.5 ± 64.2 | 0.1 ± 76.9 | 4.79 ± 27.0 | 0.498 | |
| Range | 185.4 – 608.0 | 183.6 – 422.7 | −219.3 – 177.0 | −37.9 – 95.5 | ||
| VC | 72.7 ± 12.9 | 93.1 ± 18.3 | 20.3 ± 10.9 | 28.5 ± 16.4 | < 0.001 | |
| Range | 55.8 – 110.9 | 64.5 – 144.4 | −3.6 – 39.9 | −4.9 – 69.8 | ||
Values are reported as mean ± SD. DmCO, alveolar capillary membrane conductance; VC, pulmonary capillary blood volume.
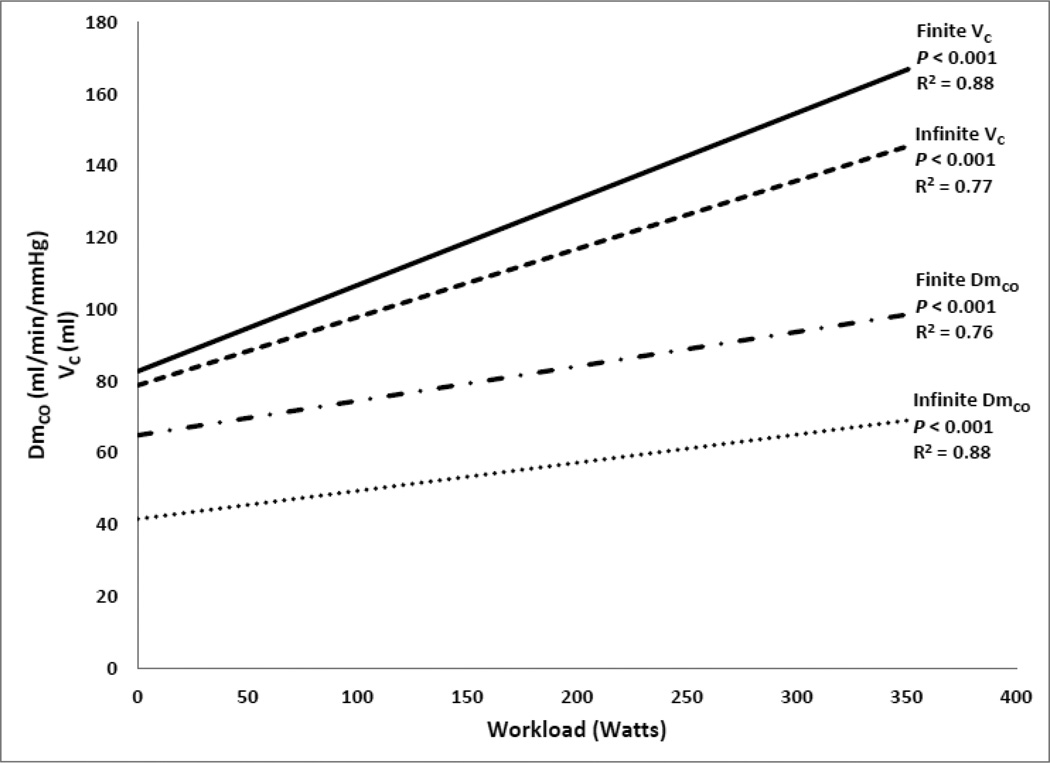
We have also simultaneously assessed DLCO and DLNO using the rebreathe technique at rest and during incremental exercise in two separate studies in our laboratory. In the first study, DLCO and DLNO were measured at rest and during cycle exercise at 0, 10, 15, 30, 50, and 70% of Wpeak (determined during a maximal exercise test at a prior study visit). This study was performed in 7 healthy subjects (Table 1, #3). In the second study, DLCO and DLNO were measured in duplicate at rest and during cycle exercise at 25, 50, 75, and 90% of Wpeak (determined during a maximal exercise test at a prior study visit). This study was performed in 31 healthy subjects (Table 1, #4). For the present analysis, the data from both studies were pooled, and DmCO or VC was then plotted as a function of workload. Because these data incorporate repeat measures, a linear mixed effects model was used to separate the individual and group effects on either DmCO or VC with exercise. The group effect was then plotted for both variables under the infinite and finite assumptions (Figure 3). Whether an infinite or a finite θNO is assumed, DmCO and VC significantly increased with increasing workload (all P < 0.001). All in all, this suggests that assuming both an infinite or finite θNO yields statistically significant increases in DmCO and VC with incremental exercise when using the rebreathe method, as would be anticipated.
Figure 3. Response of DmCO and VC to incremental exercise calculated from rebreathe data.
A linear mixed effects model was implemented to separate the individual and group effects on either DmCO or VC throughout incremental cycling exercise. The group effects for both the infinite and finite θNO assumptions are plotted as a function of workload. For both assumptions, DmCO and VC increased significantly throughout exercise (all p < 0.001). DmCO, alveolar capillary membrane conductance; VC, pulmonary capillary blood volume.
3.3 Required assumptions/problems of each method
Both the finite and infinite methods for calculating VC and DmCO from DLCO and DLNO rely on several values that likely impossible to measure directly in vivo in humans. The infinite method has two assumptions that must be optimized, the θCO coefficients and α, while the finite method has the additional term, θNO. Because these values cannot be directly measured, our laboratory has taken the approach of systematically verifying these terms using the gold-standard multiple O2 tension method, which does not rely on measures of DLNO. By contrast, this rigorous optimization does not appear to have been done for the finite method.
Both methods are very sensitive to the values chosen for the α ratio/Krogh coefficient, θNO, and θCO coefficients. While the determination of the optimal θCO coefficients is essential to the use of either method, the following section will focus on the α ratio/Krogh coefficient. When θNO and θCO are required in the theoretical discussion going forward, we will use the values that have either been determined as optimal by our group for the infinite assumption, or the values currently used in the literature for the finite assumption.
The Krogh coefficient used with the finite assumption and the α ratio used with the infinite assumption are very similar, as they both convert in DmNO to DmCO. Here, we compare the values used by each assumption and explore their relationship. The Krogh diffusion constant for CO versus NO is 1.97 in water. However, studies in our laboratory and others have suggested that a value greater than 1.97 should be used when converting DmNO to DmCO (Magini et al., 2013; Tamhane et al., 2001). For this reason, we are wary of the conclusion by the recent task force that a value of 1.97 should be utilized (Zavorsky et al., 2017). Below, we have calculated an ‘effective α’ for the finite method (see below) which can be used to directly convert DLNO to DmCO as the α ratio does for the infinite method. The value of this ‘effective α’ is shown in Figure 4 over a range DLNO/DLCO ratios.
This effective α is less than 1.97 for most DLNO/DLCO ratios using the finite method. The lower ‘effective α’ of the finite method represents the resistance to the transfer of NO through the red blood cell that is not taken into account by the infinite method. Some have argued that it is inappropriate to vary α as it is a chemical property based on the solubility of NO and CO. However, others have shown that α should be greater than 1.97 and have suggested that confounders such as uptake by the airway epithelium, conversion of NO to N2O, or differences in solubility of NO and CO in biological tissues may have a larger effect on DLNO than red blood cell resistance (Tamhane et al., 2001). Thus, we feel that it is appropriate for the α ratio to vary away from the Krogh coefficient when performing a systematic optimization.
4. DISCUSSION
The determination of θNO has been chosen based on three studies. First was an in vitro study from 1958 determining the second order rate constant of NO with red blood cells(Carlsen and Comroe, 1958). Later, the value of 4.5 mlCO/min/mmHg/mlblood was determined from this earlier study (Borland and Cox, 1991). A major methodological concern with this method is that the concentration of NO used was very high relative to that used for DLNO measurements, possibly altering the reaction kinetics and underestimating the rate constant observed in vivo (see CD Borland, this issue, Hypothesis: Why θNO could be finite in vitro but infinite in vivo). Second, a 2010 study in dogs where oxyglobin was exchanged with red blood cells found an increase in DLNO with progressively greater exchange (Borland et al., 2010). These results suggest that θNO is finite, but the study was not designed to calculate the actual value. Finally, a 2016 study in humans measured DLCO and DLNO while participants breathed 15% and 21% oxygen and attempted to estimate θNO; however, this study was not designed to precisely calculate θNO and therefore only suggests the continued use of the 4.5 mlCO/min/mmHg/mlblood value (Guenard et al., 2016). Based on only these three studies, a recent task force has recommended use of a finite θNO for calculation of DmCO and VC (Zavorsky et al., 2017). Furthermore, to the best of our knowledge, there have been no studies optimizing θNO to the multiple O2 tension method, a method which does not utilize NO, or any other method. Therefore, we suggest that further validation is necessary to determine the correct values for θNO, as well as θCO coefficients, before the finite method enters standard practice.
4.1 Theoretical breaking points for each assumption
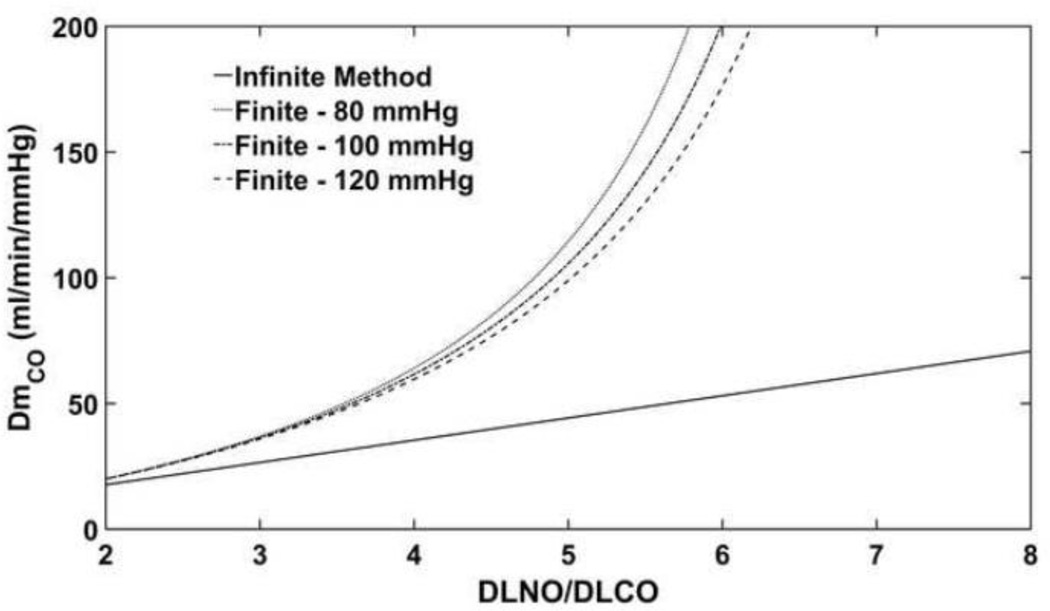
In our analysis, both the infinite and finite θNO assumptions yielded extreme outliers in a total of four instances (approximately 0.5% of all observations). Though we removed these outliers from further analysis, this finding led us to ask under which circumstances each assumption would fail to yield reasonable physiologic values for DmCO and VC. Figures 5–6 demonstrates the values that would be obtained for DmCO (Figure 5) and VC (Figure 6) over a range of DLNO/DLCO values for both methods assuming PO2 of 80 mmHg, 100 mmHg, and 120 mmHg, DLCO of 20 ml*min−1*mmHg−1 and a DLCO/DLNO ratio of up to 10.
Figure 5. Theoretical DmCO values over a range of DLNO/DLCO ratios.
DmCO was calculated under both the infinite and finite θNO assumptions using DLCO = 20 ml/min/mmHg. The finite calculation of DmCO is also dependent on PO2; values of 80, 100, and 120 mmHg are show here. While the infinite calculation of DmCO is stable over a large range of DLNO/DLCO ratios, the finite calculation of DmCO rapidly increases as the DLNO/DLCO ratio increases. DLCO, lung diffusing capacity for carbon monoxide; DLNO, lung diffusing capacity for nitric oxide; DmCO, alveolar capillary membrane conductance.
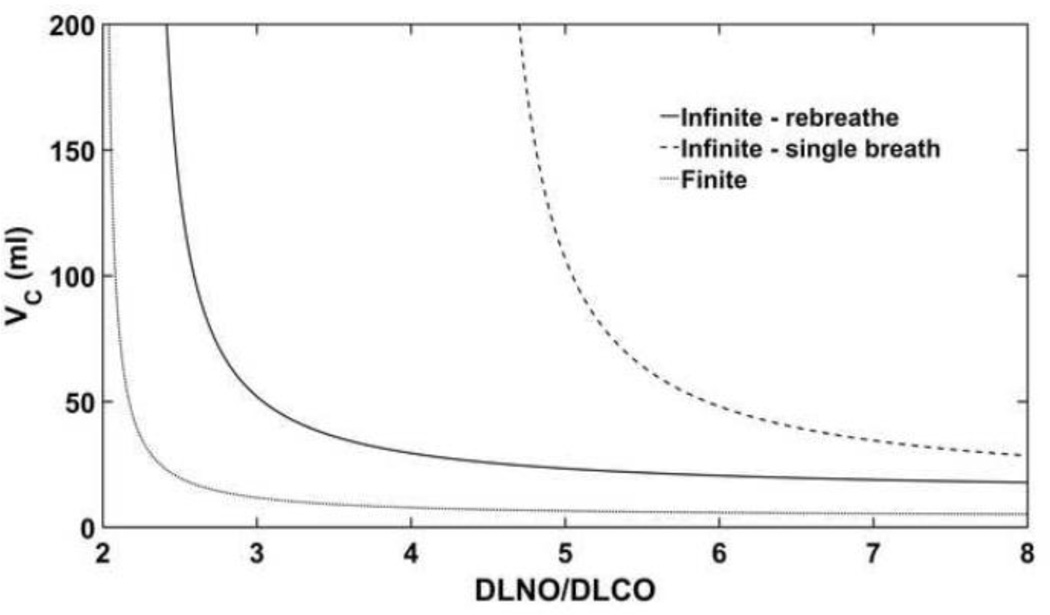
Figure 6. Theoretical VC values over a range of DLNO/DLCO ratios.
VC was calculated under both the infinite and finite θNO assumptions using DLCO = 20 ml/min/mmHg. The infinite calculation of VC is dependent on the technique used in our laboratory; hence, both rebreathe and single breath are shown here. Under both assumptions, calculation of VC is increases rapidly when the DLNO/DLCO ratio is equal to the α ratio/Krogh coefficient used. DLCO, lung diffusing capacity for carbon monoxide; DLNO, lung diffusing capacity for nitric oxide; VC, pulmonary capillary blood volume.
For DmCO, assuming an infinite θNO yields stable DmCO values for any DLNO/DLCO ratio. However, assuming a finite θNO causes an asymptote to occur, in this example around DLNO/DLCO equals 7, upon which DmCO values increase rapidly to a non-physiologic range. This asymptote occurs when DLNO/DLCO is equal to θNO/θCO (note that θCO is dependent on PO2). The asymptote location is very sensitive to the choice of θCO equation in the finite method and care must be taken to ensure that the asymptote does not occur in the physiological range of DLNO/DLCO ratio. For VC, both the infinite and finite θNO assumptions are stable unless DLNO/DLCO approaches α, where an asymptote occurs yielding rapidly increasing VC values which climb to a non-physiologic range. It is important to note that these asymptotes will shift depending on the specific values entered into the calculations.
All in all, Figures 5–6 demonstrates the mathematical constraints that are incorporated into the determination of DmCO and VC from values of DLCO and DLNO, regardless of the assumptions made as to the correct value of θNO. While DmCO and VC are themselves physiologic variables, the calculations that have been established to determine these variables incorporate an unavoidable complication. The DLNO/DLCO values at which each assumption yields an asymptote is potentially concerning in two cases. First, DmCO values calculated using a finite θNO under normal conditions are unlikely to encroach upon the asymptote, as a physiologic DLNO/DLCO value is generally around 4–5 (Hughes and van der Lee, 2013). However, in instances such as altitude, where PO2 is lower and the asymptote is therefore shifted to lower DLNO/DLCO ratios, the DLNO/DLCO ratio may fall within the range where DmCO rapidly increases to large, non-physiologic values. Second, VC values calculated using the infinite θNO assumption and the single breath method, where the optimal α value has been optimized at ~ 4.4, also causes the asymptote to occur at a physiologic range for DLNO/DLCO. However, as can be seen in Table 2, the single breath method tends to yield higher DLNO/DLCO ratios than that of the rebreathe method (presently, mean 5.39 ± 0.43 for single breath vs. 3.58 ± 0.59 for rebreathe), such that approaching the asymptote is not usually an issue. The observation that DLNO/DLCO is slightly higher using the single breath method is found in our laboratory and others (Ceridon et al., 2010; Ceridon et al., 2011; Coffman et al., 2016; Zavorsky and Lands, 2005; Zavorsky and Murias, 2006).
4.2 Key Points
To be clear, we agree that θNO is, in a strict biochemical sense, not infinite, as no biological process can occur instantaneously. Indeed, as we have highlighted above, there is experimental data which demonstrates a resistance to combination of NO with hemoglobin that resides in the red cell (Azarov et al., 2011; Sakai et al., 2008). In this sense, we concede that a finite θNO may be a more accurate representation of gas transfer from the environment to hemoglobin in blood. However, in the case of calculating DmCO and VC, the value of θNO has not been optimized relative to the gold standard multiple O2 method or in any other way. Therefore, the assumption of an infinite θNO relative to θCO is still appropriate. While our laboratory recognizes that considering θNO infinite may not be ideal, we argue two main points that cause our group to be wary of the use of a finite θNO, in its current form, at this point.
First, when we have applied the finite θNO value to our calculations of DmCO and VC using the single breath technique, we obtain values which do not increase as expected during exercise. This is concerning, as much of the research performed in our laboratory relies on accurate measures of the change in DmCO and VC during submaximal and maximal exercise bouts. Second, as is the case in any scientific field, new methodology must be validated against a gold-standard. While some groups may argue that there is no gold standard in the case of DmCO and VC, we feel it is important that the finite θNO calculations be validated against a method that does not utilize NO. We have performed such optimizations on our calculations, which assume an infinite θNO, using both the single breath and rebreathe techniques (Ceridon et al., 2010; Coffman et al., 2016). We feel it is important that those groups that are invested in the use of a finite θNO apply a similar method, where the θNO value would be systematically varied with respect to the other two required assumptions (the θCO equation and Krogh coefficient) of the finite θNO calculations. This methodology would produce optimized values for all three calculation parameter assumptions, thus adding confidence to the use of the finite method.
All in all, while the finite θNO calculations have merit and may move into standard practice in the future, we feel that two main issues remain with the use of a finite θNO in practice, including the lack of validation of the currently established θNO value as well as the non-physiological response to exercise.
Additionally, we cannot stress enough that while alveolar-capillary membrane conductance and pulmonary-capillary blood volume are anatomical phenomena, the calculations that have been established to determine these variables from measured values of DLNO and DLCO are entirely dependent on the mathematical relationships underlying the given assumptions. In this sense, perhaps it is better to consider DmCO and VC functional variables that can be used to observe changes over time, whether that be during exercise studies or clinically in disease, instead of true representations of anatomy.
4.3 Conclusions
The nuances of the calculations required for both assumptions can be discussed endlessly, but our ability to utilize DmCO and VC to contribute meaningfully to the field is the overriding goal. We have shown that the values themselves are very dependent on the mathematical relationships, and therefore are likely not entirely in agreement with the physiology regardless of the assumptions. However, by using a consistent method for calculation of DmCO and VC, it is entirely possible to observe changes within study participants or differences between study groups. If DmCO and VC are to eventually enter clinical practice, an assumption regarding θNO must be determined, in addition to a θCO equation and α ratio, and standardized across the practice. Once the optimal calculation parameters have been chosen, it will then be possible to systematically investigate appropriate cutoffs for the determination of disease states. At the current time, with a number of conflicting assumptions and methods, the results are simply too variable to determine how a particular result should be interpreted. In the research arena, the choice of calculation parameters is of little concern as long as the resulting data remain within the physiologic range (i.e. positive) and have appropriate response to stimuli, such as pulmonary-capillary blood volume increasing with exercise. While we respect the work of the recent ERS task force, we believe their findings are premature as no study has been designed to calculate an exact θNO in vivo in humans, and we are also uncomfortable with their conclusions regarding the correct DmNO/DmCO (a-) ratio. Additionally, the ERS task force has not presented any new evidence for use of a finite θNO beyond the evidence discussed above (Zavorsky et al., 2017). Therefore at this time, we suggest the continued use of the assumption that θNO is infinite until further optimization of the finite θNO method can be performed.
Highlights.
We discuss whether θNO is finite or infinite
We calculate DmCO and VC under both assumptions at rest and during exercise
We find that a finite θNO yields unexpected submaximal exercise results
We conclude that a finite θNO should be validated in vivo prior to further use
Acknowledgments
KEC and SCC are supported by Mayo Graduate School. KEC is supported by the National Heart, Lung, And Blood Institute of the National Institutes of Health grant F31HL131076. BJT is supported by a Fulbright Commission UK Distinguished Scholar Award and American Heart Association Grant AHA12-POST12070084. This study was funded by NIH grant HL71478.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- Azarov I, Liu C, Reynolds H, Tsekouras Z, Lee JS, Gladwin MT, Kim-Shapiro DB. Mechanisms of slower nitric oxide uptake by red blood cells and other hemoglobin-containing vesicles. The Journal of biological chemistry. 2011;286:33567–33579. doi: 10.1074/jbc.M111.228650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borland C, Dunningham H, Bottrill F, Vuylsteke A. Can a membrane oxygenator be a model for lung NO and CO trannsfer? Journal of applied physiology. 2006;100:1527–1538. doi: 10.1152/japplphysiol.00949.2005. [DOI] [PubMed] [Google Scholar]
- Borland CD, Cox Y. Effect of varying alveolar oxygen partial pressure on diffusing capacity for nitric oxide and carbon monoxide, membrane diffusing capacity and lung capillary blood volume. Clinical science. 1991;81:759–765. doi: 10.1042/cs0810759. [DOI] [PubMed] [Google Scholar]
- Borland CD, Dunningham H, Bottrill F, Vuylsteke A, Yilmaz C, Dane DM, Hsia CC. Significant blood resistance to nitric oxide transfer in the lung. Journal of applied physiology. 2010;108:1052–1060. doi: 10.1152/japplphysiol.00904.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borland CD, Higenbottam TW. A simultaneous single breath measurement of pulmonary diffusing capacity with nitric oxide and carbon monoxide. The European respiratory journal. 1989;2:56–63. [PubMed] [Google Scholar]
- Carlsen E, Comroe JH., Jr The rate of uptake of carbon monoxide and of nitric oxide by normal human erythrocytes and experimentally produced spherocytes. The Journal of general physiology. 1958;42:83–107. doi: 10.1085/jgp.42.1.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ceridon ML, Beck KC, Olson TP, Bilezikian JA, Johnson BD. Calculating alveolar capillary conductance and pulmonary capillary blood volume: comparing the multiple-and single-inspired oxygen tension methods. Journal of applied physiology. 2010;109:643–653. doi: 10.1152/japplphysiol.01411.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ceridon ML, Morris NR, Hulsebus ML, Olson TP, Lalande S, Johnson BD. Influence of bronchial blood flow and conductance on pulmonary function in stable systolic heart failure. Respiratory physiology & neurobiology. 2011;177:256–264. doi: 10.1016/j.resp.2011.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coffman KE, Taylor BJ, Carlson AR, Wentz RJ, Johnson BD. Optimizing the calculation of DM,CO and VC via the single breath single oxygen tension DLCO/NO method. Respiratory physiology & neurobiology. 2016;221:19–29. doi: 10.1016/j.resp.2015.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deonikar P, Kavdia M. An integrated computational and experimental model of nitric oxide-red blood cell interactions. Annals of biomedical engineering. 2010;38:357–370. doi: 10.1007/s10439-009-9823-x. [DOI] [PubMed] [Google Scholar]
- Forster RE. Diffusion of Gases Across the Alveolar Membrane, Comprehensive Physiology. John Wiley & Sons, Inc; 1987. [Google Scholar]
- Forster RE, Fowler WS, Bates DV, Van Lingen B. The absorption of carbon monoxide by the lungs during breath-holding. The Journal of clinical investigation. 1954;33:1135–1145. doi: 10.1172/JCI102987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guenard H, Varene N, Vaida P. Determination of lung capillary blood volume and membrane diffusing capacity in man by the measurements of NO and CO transfer. Respiration physiology. 1987;70:113–120. doi: 10.1016/s0034-5687(87)80036-1. [DOI] [PubMed] [Google Scholar]
- Guenard HJ, Martinot JB, Martin S, Maury B, Lalande S, Kays C. In vivo estimates of NO and CO conductance for haemoglobin and for lung transfer in humans. Respiratory physiology & neurobiology. 2016;228:1–8. doi: 10.1016/j.resp.2016.03.003. [DOI] [PubMed] [Google Scholar]
- Hakim TS, Sugimori K, Camporesi EM, Anderson G. Half-life of nitric oxide in aqueous solutions with and without haemoglobin. Physiological measurement. 1996;17:267–277. doi: 10.1088/0967-3334/17/4/004. [DOI] [PubMed] [Google Scholar]
- Hughes JM, Bates DV. Historical review: the carbon monoxide diffusing capacity (DLCO) and its membrane (DM) and red cell (Theta.Vc) components. Respiratory physiology & neurobiology. 2003;138:115–142. doi: 10.1016/j.resp.2003.08.004. [DOI] [PubMed] [Google Scholar]
- Hughes JM, van der Lee I. The TL,NO/TL,CO ratio in pulmonary function test interpretation. The European respiratory journal. 2013;41:453–461. doi: 10.1183/09031936.00082112. [DOI] [PubMed] [Google Scholar]
- Krogh A, Krogh M. On the Rate of Diffusion of Carbonic Oxide into the Lungs of Man1. Skandinavisches Archiv Für Physiologie. 1910;23:236–247. [Google Scholar]
- Krogh M. The diffusion of gases through the lungs of man. The Journal of physiology. 1915;49:271–300. doi: 10.1113/jphysiol.1915.sp001710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- La Gerche A, MacIsaac AI, Burns AT, Mooney DJ, Inder WJ, Voigt JU, Heidbuchel H, Prior DL. Pulmonary transit of agitated contrast is associated with enhanced pulmonary vascular reserve and right ventricular function during exercise. Journal of applied physiology. 2010;109:1307–1317. doi: 10.1152/japplphysiol.00457.2010. [DOI] [PubMed] [Google Scholar]
- Lewis BM, Lin TH, Noe FE, Komisaruk R. The measurement of pulmonary capillary blood volume and pulmonary membrane diffusing capacity in normal subjects; the effects of exercise and position. The Journal of clinical investigation. 1958;37:1061–1070. doi: 10.1172/JCI103687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X, Miller MJ, Joshi MS, Sadowska-Krowicka H, Clark DA, Lancaster JR., Jr Diffusion-limited reaction of free nitric oxide with erythrocytes. The Journal of biological chemistry. 1998;273:18709–18713. doi: 10.1074/jbc.273.30.18709. [DOI] [PubMed] [Google Scholar]
- Magini A, Apostolo A, Salvioni E, Italiano G, Veglia F, Agostoni P. Alveolar-capillary membrane diffusion measurement by nitric oxide inhalation in heart failure. European journal of preventive cardiology. 2013 doi: 10.1177/2047487313510397. [DOI] [PubMed] [Google Scholar]
- Meyer M, Schuster KD, Schulz H, Mohr M, Piiper J. Pulmonary diffusing capacities for nitric oxide and carbon monoxide determined by rebreathing in dogs. Journal of applied physiology. 1990;68:2344–2357. doi: 10.1152/jappl.1990.68.6.2344. [DOI] [PubMed] [Google Scholar]
- Reeves RB, Park HK. CO uptake kinetics of red cells and CO diffusing capacity. Respiration physiology. 1992;88:1–21. doi: 10.1016/0034-5687(92)90025-r. [DOI] [PubMed] [Google Scholar]
- Roughton FJ, Forster RE. Relative importance of diffusion and chemical reaction rates in determining rate of exchange of gases in the human lung, with special reference to true diffusing capacity of pulmonary membrane and volume of blood in the lung capillaries. Journal of applied physiology. 1957;11:290–302. doi: 10.1152/jappl.1957.11.2.290. [DOI] [PubMed] [Google Scholar]
- Sackner MA, Greeneltch D, Heiman MS, Epstein S, Atkins N. Diffusing capacity, membrane diffusing capacity, capillary blood volume, pulmonary tissue volume, and cardiac output measured by a rebreathing technique. The American review of respiratory disease. 1975;111:157–165. doi: 10.1164/arrd.1975.111.2.157. [DOI] [PubMed] [Google Scholar]
- Sakai H, Sato A, Masuda K, Takeoka S, Tsuchida E. Encapsulation of concentrated hemoglobin solution in phospholipid vesicles retards the reaction with NO, but not CO, by intracellular diffusion barrier. J Biol Chem. 2008;283:1508–1517. doi: 10.1074/jbc.M707660200. [DOI] [PubMed] [Google Scholar]
- Snyder EM, Beck KC, Turner ST, Hoffman EA, Joyner MJ, Johnson BD. Genetic variation of the beta2-adrenergic receptor is associated with differences in lung fluid accumulation in humans. Journal of applied physiology. 2007;102:2172–2178. doi: 10.1152/japplphysiol.01300.2006. [DOI] [PubMed] [Google Scholar]
- Tamhane RM, Johnson RL, Jr, Hsia CC. Pulmonary membrane diffusing capacity and capillary blood volume measured during exercise from nitric oxide uptake. Chest. 2001;120:1850–1856. doi: 10.1378/chest.120.6.1850. [DOI] [PubMed] [Google Scholar]
- Taylor BJ, Carlson AR, Miller AD, Johnson BD. Exercise-induced interstitial pulmonary edema at sea-level in young and old healthy humans. Respiratory physiology & neurobiology. 2014;191:17–25. doi: 10.1016/j.resp.2013.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaughn MW, Huang KT, Kuo L, Liao JC. Erythrocytes possess an intrinsic barrier to nitric oxide consumption. The Journal of biological chemistry. 2000;275:2342–2348. doi: 10.1074/jbc.275.4.2342. [DOI] [PubMed] [Google Scholar]
- Zavorsky GS, Hsia CCW, Hughes JMB, Borland CDR, Guenard HJ, Van der Lee I, Steenbruggen I, Naeije RCJ, Dinh-Xuan AT. Standardization and application of the single-breath determination of nitric oxide uptake in the lung. European Respiratory Journal. 2017 doi: 10.1183/13993003.00962-2016. [In press] [DOI] [PubMed] [Google Scholar]
- Zavorsky GS, Lands LC. Lung diffusion capacity for nitric oxide and carbon monoxide is impaired similarly following short-term graded exercise. Nitric oxide : biology and chemistry / official journal of the Nitric Oxide Society. 2005;12:31–38. doi: 10.1016/j.niox.2004.11.002. [DOI] [PubMed] [Google Scholar]
- Zavorsky GS, Milne EN, Lavorini F, Rienzi JP, Cutrufello PT, Kumar SS, Pistolesi M. Small changes in lung function in runners with marathon-induced interstitial lung edema. Physiological reports. 2014:2. doi: 10.14814/phy2.12056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zavorsky GS, Murias JM. A small amount of inhaled nitric oxide does not increase lung diffusing capacity. The European respiratory journal. 2006;27:1251–1257. doi: 10.1183/09031936.06.00146805. [DOI] [PubMed] [Google Scholar]
- Zavorsky GS, Quiron KB, Massarelli PS, Lands LC. The relationship between single-breath diffusion capacity of the lung for nitric oxide and carbon monoxide during various exercise intensities. Chest. 2004;125:1019–1027. doi: 10.1378/chest.125.3.1019. [DOI] [PubMed] [Google Scholar]