Abstract
Background
Serum procalcitonin (PCT) is a useful biomarker to tailor the duration of antibiotics in respiratory infections. The objective of this study was to determine whether PCT levels could tailor postoperative antibiotic therapy in patients operated for peritonitis.
Method
Patients with peritonitis were randomized postoperatively. The control group received antibiotics for a defined duration according to institutional guidelines. In the study group, antibiotics were stopped based on serum PCT levels. Patients were stratified into three categories: (1) gastrointestinal perforation, (2) perforated appendicitis, and (3) postoperative complication. Primary outcome was duration of antibiotics.
Results
We included 162 patients; 83 and 79 patients in the control group and study group, respectively. In the subgroup of patients with peritonitis due to gastrointestinal perforation, we found 7 days of antibiotics in the PCT group versus 10 days in the control group (p value 0.065). There was no difference in infectious complications, mortality, median length of hospital stay, and necessity to restart antibiotics.
Conclusion
No significant differences were found in duration of antibiotics when applying PCT guidance. However, in the subgroup of primary perforation of the gastrointestinal tract, there was a difference in duration of antibiotics in favor of the PCT group without obtaining significance, as the study was not powered for subgroup analysis. Further studies including only this subgroup should be performed.
1. Introduction
Successful management of peritonitis due to perforation of the gastrointestinal tract remains challenging, even in the era of modern surgery. Incidences of infectious and noninfectious complications remain high, as is the mortality rate [1]. Rapid diagnosis, efficient surgical intervention, and potent antibiotics are the most relevant cornerstones of any treatment algorithm. Perioperative broad spectrum antibiotics are commonly used, allowing clearance of aerobic and anaerobic bacteria. While antibiotics are empirically targeted at the usual digestive flora, there is poor consensus on the duration of antibiotic treatment, and most institutions have adopted their own guidelines. Nevertheless, appropriate shortening of the treatment duration may be a crucial aspect limiting antibiotic resistance, costs, duration of hospital stay, and improving patient's outcomes [2].
The use of biomarkers to assess the treatment efficacy on infections is commonly known, but a specific parameter to monitor the duration of antibiotics has been lacking. Procalcitonin (PCT), a precursor of calcitonin, is amplified as part of the physical response to bacterial infections [3, 4]. In animal models as well as in clinical settings, the correlation of PCT and the severity of sepsis have been shown to be superior to other biological markers, such as C-reactive protein, interleukin-1, or interleukin-6 [4–6]. Furthermore, there is solid evidence that the treatment of community-acquired pneumonia and ventilator-associated pneumonia can effectively be monitored by repeated measurement of serum PCT levels, and the duration of antibiotics can significantly be shortened [7–11]. Therefore, we hypothesized that the duration of antibiotic treatments based on current guidelines represents an “overtreatment,” and a tailored approach could shorten antibiotic therapy without increasing infectious complications in patients with gastrointestinal perforations.
The aim of this study was to assess the usefulness of PCT to guide the duration of antibiotic use in surgical patients with peritonitis due to gastrointestinal perforation.
2. Patients and Methods
2.1. Setting and Study Population
The current study is a prospective randomized trial performed at a university hospital. The duration of antibiotic therapy in patients treated according to institution guidelines with defined antibiotic duration (control group, Table 1) was compared to patients in whom antibiotic treatment was guided by serum PCT levels (procalcitonin (PCT) group). The study protocol was approved by the local ethical committee, registered (www.clinicaltrials.gov, NCT01155739), and all patients gave written informed consent.
Table 1.
Institutional guidelines for duration of antibiotic therapy, based on the Surgical Infection Society guidelines [15–17].
| Indication | Duration | First choice antibiotics |
|---|---|---|
| Perforated appendicitis with peritonitis | 5 days | Amoxicillin/clavulanic acid Allergy: ciprofloxacin + metronidazole |
| Peritonitis due to gastrointestinal perforation | 10 days | Amoxicillin/clavulanic acid. If severe infection: imipenem or piperacillin/tazobactam Allergy: ciprofloxacin + metronidazole |
| Peritonitis due to gastrointestinal perforation, acquired in-hospital | 10 days | Imipenem or piperacillin/tazobactam Allergy: ciprofloxacin + metronidazole |
2.2. Inclusion Criteria
All patients > 18 years who underwent abdominal surgery and with an established diagnosis of peritonitis were potentially eligible. All types of peritonitis caused by gastrointestinal perforations were included (gastric, purulent, fecal, or fibrinous content) either restricted to one quadrant or generalized to all four quadrants. After inclusion into the study, patients were stratified into three categories based upon the diagnosis: (1) peritonitis related to a gastrointestinal perforation (e.g., diverticulitis, perforated gastric or duodenal ulcer, intestinal ischemia, iatrogenous perforation, intestinal perforation in trauma patients, and perforated malignancy), (2) peritonitis due to perforated appendicitis, and (3) peritonitis due to a postoperative complication.
2.3. Exclusion Criteria
Patients with any kind of immunosuppression (e.g., long-term corticosteroids, chemotherapy, organ transplantation, and HIV with <200 CD4 cells), patients with medullary cancer of the thyroid or severe hepatocellular insufficiency (high procalcitonin values), and spontaneous peritonitis in patients with ascites were excluded. In addition, patients who refused to sign informed consent were not included.
2.4. Study Intervention and Randomization
Inclusion of patients was done postoperatively, after the diagnosis of peritonitis was established and its origin determined by surgery. Patients were randomly assigned to the control or PCT group through sealed, opaque envelopes. There was no stratification of randomization based on the three subgroups. Randomization was blinded for the patient, while physicians were aware of the respective group. In both groups, empiric antibiotic therapy was started preoperatively or at induction of anesthesia, and serum PCT levels were determined at the day of surgery or postoperative day 1. In the control group, duration of antibiotic application was according to institutional guidelines (Table 1), but of note, duration was prolonged if clinically indicated, for example, in case of persisting fever, increasing infectious laboratory values, or infectious complications (abscess, positive blood cultures).
In the PCT group, antibiotics were continued until postoperative day 3, without consideration of the respective serum PCT levels. On the third postoperative day, serum PCT values were measured and antibiotics were stopped if serum PCT values < 1 μg/l; if values were >1 μg/l, antibiotics were continued. Serum PCT values were determined every following 48 h, and in case of serum PCT values < 0.25 μg/l or a decrease of 80% compared to the baseline value, antibiotics were then stopped. The daily physical examination of the patient was taken into account when stopping the antibiotics. In case of a clinical suspicion of an ongoing infection, antibiotics could be kept or reintroduced independently of the respective serum PCT levels or other diagnostic measures.
2.5. Measurement of PCT
Serum PCT measurements were performed with the Electrochemiluminescence immunoassay (ECLIA) on the automated Roche Elecsys® immunoassay analyzers and were obtained in approximately two hours.
2.6. Outcome Measures
The primary outcome measure was the duration of antibiotics in days. This includes the initial treatment, until discontinuation of antibiotics. If antibiotics were restarted, this was not included in the duration of antibiotics. Secondary outcome measures were (1) duration of hospitalization, (2) incidence of 30-day infectious complications, (3) 30-day mortality, and (4) incidence of reintroduction of antibiotics after having been ceased.
To compare groups, age, gender, comorbidities, ASA score, and Mannheim Peritonitis Index [12] were documented in all patients.
2.7. Statistical Analysis
The sample size was calculated based on a 33% reduction in antibiotic duration (10 days in control group, 6.7 days in study group, standard deviation 7.55 days). With a power of 80% and an α-error of 0.05 using a two-tailed test, 166 patients (83 per group) were necessary.
Categorical data are presented as numbers with percentages; numerical data are presented as medians with interquartile ranges. Univariate analysis was performed using a Fisher's exact test in case of categorical data, and a Mann–Whitney U test in case of numerical data. Data were analyzed as intention-to-treat. Statistical analysis was performed with SPSS version 22.
3. Results
From June 2009 to September 2012, 162 patients with peritonitis were included and randomized between procalcitonin and control groups (CONSORT diagram Figure 1). Due to dropouts after initial inclusion, the power calculation was initially not obtained and the 10 last were included end 2013–beginning 2014 for this reason.
Figure 1.
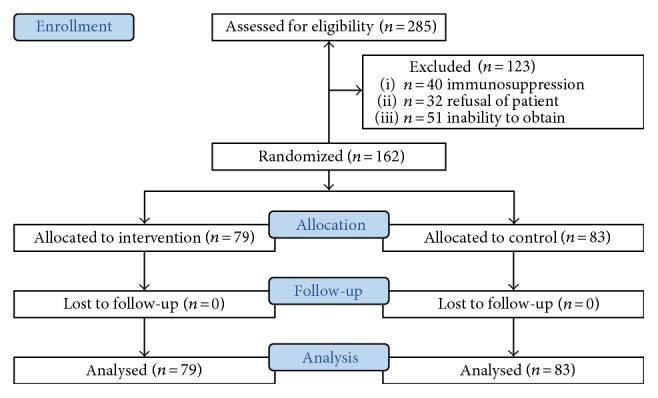
CONSORT diagram of included patients.
The baseline characteristics of all patients are shown in Table 2. There were 64 patients in the group with peritonitis related to a gastrointestinal perforation (group 1), 73 patients with peritonitis due to acute appendicitis (group 2), and 25 patients with peritonitis due to a postoperative complication (group 3).
Table 2.
Baseline characteristics.
| Procalcitonin group (n = 79) |
Control group (n = 83) |
p value | |
|---|---|---|---|
| Median age (range), years | 56 (36–73) | 57 (36–71) | 0.776 |
| Males, n (%) | 46 (58.2%) | 47 (57%) | 0.875 |
| ASA 1-2, n (%) | 46 (58.2%) | 60 (72.3%) | 0.07 |
| ASA 3-4 | 33 (41.8%) | 23 (27.7%) | |
| Comorbidities, n (%) | |||
| (i) Diabetes mellitus | 4 (5.1%) | 4 (4.8%) | 1.00 |
| (ii) Pulmonary | 13 (16.5%) | 7 (8.4%) | 0.15 |
| (iii) Cardiac | 29 (36.7%) | 34 (41%) | 0.63 |
| (iv) Renal dysfunction | 9 (11.4%) | 8 (9.6%) | 0.80 |
| Needing ICU hospitalization, n (%) | 10 (12.7%) | 10 (12.0%) | 1.00 |
| Mannheim Peritonitis score (points) | |||
| (i) All | 19 (11–25) | 17 (12–26) | 1.00 |
| (ii) Subgroup 1 | 20 (16–26) | 21 (15.5–27.5) | |
| (iii) Subgroup 2 | 15 (10.8–21.3) | 15 (10–21) | |
| (iv) Subgroup 3 | 24.5 (17.8–29) | 28 (21–38) | |
| Peritonitis | 0.91 | ||
| (i) Trouble exsudat | 5 (6.3%) | 7 (8.4%) | |
| (ii) Gastric/duodenal content | 4 (5.1%) | 5 (6.0%) | |
| (iii) Fibrinous | 3 (3.8%) | 3 (3.6%) | |
| (iv) Purulent | 53 (67.1%) | 52 (62.7%) | |
| (v) Fecal | 14 (17.7%) | 16 (19.3%) | |
| Peritonitis quadrants | 0.73 | ||
| 1 | 33 (41.8%) | 28 (33.7%) | |
| 2 | 10 (12.7%) | 14 (16.7%) | |
| 3 | 3 (3.8%) | 2 (2.4%) | |
| 4 | 33 (41.8%) | 39 (47.0%) | |
| Group 1 peritonitis due to GI perforation | 31 | 33 | 0.95 |
| (i) Diverticulitis | 14 | 9 | |
| (ii) Perforated ulcer | 4 | 11 | |
| (iii) Perforation stent/colonoscopy/biopsy | 6 | 5 | |
| (iv) Ischemia–necrosis | 6 | 2 | |
| (v) High-velocity trauma | 0 | 2 | |
| (vi) Perforated tumor | 1 | 1 | |
| (vii) Other | 0 | 3 | |
| Group 2 peritonitis due to appendicitis | 34 | 39 | 0.61 |
| Group 3 postoperative peritonitis | 14 | 11 | 0.43 |
| (i) Anastomotic leakage | 6 | 5 |
ASA: American Society of Anesthesiologists physical status classification system; ICU: intensive care unit.
The overall mortality rate was 4.3%, whereby these patients had a significant higher Mannheim Peritonitis Index (median 29 (range 23–34) versus median 17 (11.75–24), p value 0.005).
3.1. Primary Endpoint
When comparing all patients, there was no difference in duration of antibiotics between the PCT and control group (Table 3). The results for the subgroup analysis for patients with peritonitis related to a gastrointestinal perforation (group 1) separately show a trend towards a reduced duration of antibiotics in the PCT arm, without obtaining significance (median 7 versus 10 days, p value 0.065, Table 3).
Table 3.
Primary outcome measure: duration of antibiotics (median with interquartile range, days).
| Duration antibiotic treatment | Procalcitonin (n = 79) |
Control (n = 83) |
p value |
|---|---|---|---|
| (i) All patients | 8 (5–16) | 10 (6–12) | 0.714 |
| (ii) Subgroup 1: GI perforation | 7 (5–12) | 10 (8.5–12) | 0.065 |
| (iii) Subgroup 2: appendicitis | 8 (5.5–13.5) | 8 (5–11) | 0.573 |
| (iv) Subgroup 3: postoperative | 18.5 (6.75–29.5) | 13 (11–18) | 0.403 |
3.2. Secondary Endpoints
There were no significant differences in infectious complications or death, as depicted in Table 4. Median duration of hospital stay was similar between both groups. Results of the secondary endpoints are shown for all patients together and for the subgroup analysis of group 1 separately since this group had a trend for significant difference for the primary endpoint.
Table 4.
Secondary outcome measures.
| Procalcitonin | Control | p value | ||||
|---|---|---|---|---|---|---|
| All (n = 79) |
Subgroup 1 (n = 31) | All (n = 83) |
Subgroup 1 (n = 33) | All | Subgroup 1 | |
| Duration of hospitalization, d | 10 (5–24) | 10 (6–24) | 8 (4–16) | 10 (6–20) | 0.188 | 0.809 |
| Reintroduction of antibiotics | 6 (7.7%) | 3 (10%) | 5 (6.1%) | 1 (3%) | 0.762 | 0.340 |
| Reoperation | 24 (30.4%) | 6 (19.4%) | 20 (24.1%) | 7 (21.1%) | 0.383 | 1.00 |
| Relavage abdominal cavity | 10 (12.7%) | 2 (6.5%) | 7 (8.4%) | 3 (9.1%) | 0.447 | 1.00 |
| Rehospitalization | 5 (6.6%) | 1 (3.6%) | 5 (6.1%) | 2 (6.1%) | 1.00 | 1.00 |
| Positive blood cultures | 2 (2.5%) | 0 | 3 (3.6%) | 2 (6.1%) | 1.00 | 0.493 |
| Intra-abdominal abscess | 14 (17.7%) | 3 (9.7%) | 14 (16.9%) | 3 (9.1%) | 1.00 | 1.00 |
| Wound infection | 17 (21.5%) | 7 (22.6%) | 15 (18.1%) | 11 (33.3%) | 0.694 | 0.410 |
| Septic shock | 4 (5.1%) | 1 (3.2%) | 4 (4.8%) | 3 (9.1%) | 1.00 | 0.614 |
| Anastomotic leakage | 3 (3.8%) | 1 (3.2%) | 3 (3.6%) | 2 (6.1%) | 1.00 | 1.00 |
| Urinary infection | 3 (3.8%) | 2 (6.5%) | 1 (1.2%) | 0 | 0.358 | 0.231 |
| Pneumonia | 2 (2.5%) | 0 | 4 (4.8%) | 3 (9.1%) | 0.682 | 0.239 |
| Mortality | 3 (3.8%) | 3 (9.7%) | 4 (4.9%) | 3 (9.4%) | 1.00 | 1.00 |
Duration of antibiotics and duration of hospitalization are medians with interquartile range (days). All other parameters are numbers with percentages.
Figure 2 shows the relation of the initial level of procalcitonin to later complications. The median level of the initial PCT value (on day of surgery or the next day) in the group with no further complications was 2.2 μg/l (interquartile range 0.5–7.5); whereas it was 4.6 μg/l (interquartile range 1.3–14.7) in the group having one or more infectious complications in the postoperative phase. A Mann–Whitney U test shows that this difference is statistically different with a p value of 0.07.
Figure 2.
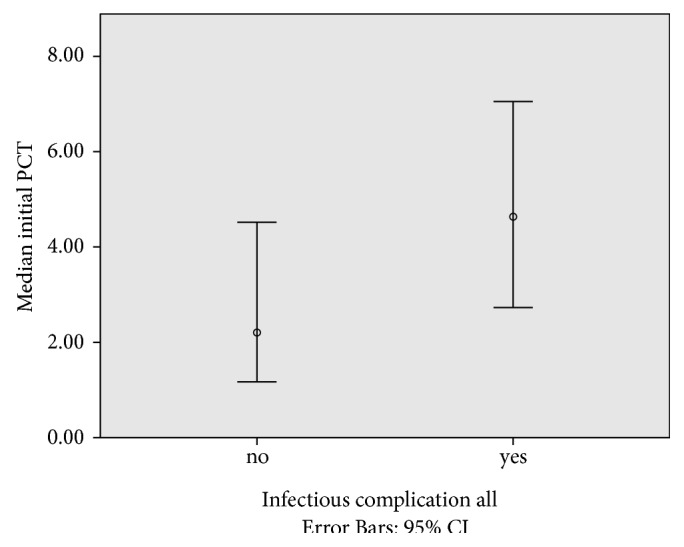
Relation initial PCT value and the occurring of postoperative infectious complications. x-axis show the group of patients with (yes) and without (no) an infectious postoperative complication. y-axis shows the median PCT value on day of surgery or postoperative day 1 in μg/l (error bars 95% CI).
Figure 3 shows the scatter plot of the initial level of procalcitonin to needed duration of antibiotic therapy. The Spearman's rank correlation coefficient is r = 0.4, with a p value > 0.01.
Figure 3.
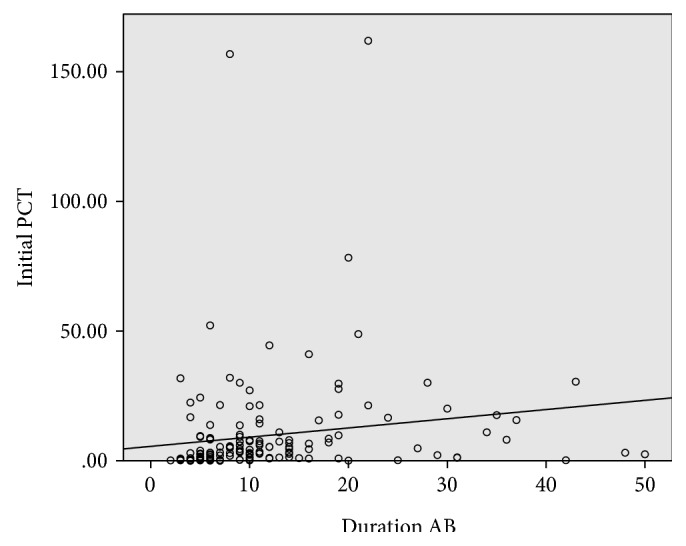
Relation initial PCT value and postoperative antibiotic therapy. Scatter plot of duration of antibiotic therapy in days on x-axis versus the median PCT value on day of surgery or postoperative day 1 in μg/l. The Spearman's rank correlation coefficient r = 0.4, with a p value > 0.01.
4. Discussion
This randomized controlled study assessed the role of a predefined algorithm using serum PCT levels to tailor the duration antibiotic therapies after surgery for peritonitis. To this end, 162 patient were randomized to receive either a PCT-guided therapy or a standard antibiotic treatment according to institutional guidelines. The main finding was that there were no differences of the duration of antibiotic treatment between both groups. In patients with an already short antibiotic treatment, for example, localized peritonitis after appendicitis, the algorithm failed to further shorten the duration of antibiotic use. Patients with peritonitis due to a postoperative complication (mostly anastomotic leakages) revealed the longest antibiotic therapies, but no reduction was achieved using the PCT algorithm. However, patients admitted to the hospital with peritonitis caused by primary gastrointestinal perforations were apt to the PCT algorithm, with a decreased median length of antibiotic treatment of three days, without obtaining statistical significance. This study was not powered for subgroup analysis; we speculate that no statistical significance was obtained due to insufficient power (type II error) due to a small patient subgroup. Further analysis of this subgroup showed no increased rate of infectious complications in the shorted antibiotics group compared to patients with a prolonged standard treatment time. The shortening of the antibiotic treatment duration may be a crucial aspect limiting antibiotic resistance, costs, duration of hospital stay, and improving patient's outcomes in this subgroup.
Interestingly, a higher initial value of PCT on postoperative day 0 or 1 seems to have a relation with a longer duration of antibiotic therapy and gives a higher risk of postoperative infectious complications.
This study was based on the positive results obtained with PCT guidance in pneumonia. Randomized controlled studies were set up in patients with suspicion of community-acquired pneumonia, and PCT values were used to initiate and to the duration of antibiotics [7, 8]. Initiation and duration was discouraged when finding PCT values less than 0.25 μg/l and encouraged when greater than 0.25 μg/l. These randomized studies showed that prescription of antibiotics on admission, total antibiotic exposure, and duration of antibiotic treatment was reduced in the PCT guidance group, compared with patients treated according to conventional guidelines. Corresponding results were obtained in ventilator-acquired pneumonia for the duration of antibiotics [9].
Our results are in line with the study by Huang et al. who performed a case-control study using a PCT algorithm to stop antibiotics in patients with secondary peritonitis [13]. They also showed a reduction of duration of antibiotic treatment by a median of 3 days, without observing an increased rate of adverse events. Another study on PCT after colorectal surgery found that PCT < 1.5 ng/ml on postoperative days 1 to 3 had a strong negative predictive value for systemic infectious complications [14].
There are some limitations of the current study that should be addressed. First, the duration of antibiotic treatment for peritonitis is not internationally standardized. Second, since an algorithm for discontinuation of antibiotics cannot completely replace clinical patient evaluations, treating physicians could violate the algorithm when clinically suspecting the need to continue antibiotic treatment. Last, the complete analysis is concordant with the prestudy power calculation but seems to include a too large variety of origins of peritonitis making the variance of duration of antibiotics too large for statistical significance. A subgroup analysis of a homogeneous group of patients approaches a level of significance without obtaining a statistical difference between groups. Since these are the results of a subgroup analysis, the lack of significance can be explained by a lower number of patients in the subgroup as anticipated.
In conclusion, a PCT algorithm could be used to guide antibiotic treatment in case of gastrointestinal perforation and allows a potential reduction in antibiotic duration. In particular, patients admitted with primary perforation of the GI tract may represent a suitable patient target group, whereas patients with an a priori limited time of antibiotic treatment do not benefit.
Conflicts of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
References
- 1.Brun-Buisson C., Doyon F., Carlet J., et al. Incidence, risk factors, and outcome of severe sepsis and septic shock in adults. A multicenter prospective study in intensive care units. French ICU Group for severe sepsis. Journal of the American Medical Association. 1995;274(12):968–974. doi: 10.1001/jama.1995.03530120060042. [DOI] [PubMed] [Google Scholar]
- 2.Nathan C., Cars O. Antibiotic resistance—problems, progress, and prospects. The New England Journal of Medicine. 2014;371(19):1761–1763. doi: 10.1056/NEJMp1408040. [DOI] [PubMed] [Google Scholar]
- 3.Becker K. L., Nylen E. S., White J. C., Muller B., Snider R. H., Jr. Clinical review 167: procalcitonin and the calcitonin gene family of peptides in inflammation, infection, and sepsis: a journey from calcitonin back to its precursors. The Journal of Clinical Endocrinology and Metabolism. 2004;89(4):1512–1525. doi: 10.1210/jc.2002-021444. [DOI] [PubMed] [Google Scholar]
- 4.Schuetz P., Christ-Crain M., Muller B. Biomarkers to improve diagnostic and prognostic accuracy in systemic infections. Current Opinion in Critical Care. 2007;13(5):578–585. doi: 10.1097/MCC.0b013e3282c9ac2a. [DOI] [PubMed] [Google Scholar]
- 5.Clec'h C., Ferriere F., Karoubi P., et al. Diagnostic and prognostic value of procalcitonin in patients with septic shock. Critical Care Medicine. 2004;32(5):1166–1169. doi: 10.1097/01.CCM.0000126263.00551.06. [DOI] [PubMed] [Google Scholar]
- 6.Harbarth S., Holeckova K., Froidevaux C., et al. Diagnostic value of procalcitonin, interleukin-6, and interleukin-8 in critically ill patients admitted with suspected sepsis. American Journal of Respiratory and Critical Care Medicine. 2001;164(3):396–402. doi: 10.1164/ajrccm.164.3.2009052. [DOI] [PubMed] [Google Scholar]
- 7.Christ-Crain M., Stolz D., Bingisser R., et al. Procalcitonin guidance of antibiotic therapy in community-acquired pneumonia: a randomized trial. American Journal of Respiratory and Critical Care Medicine. 2006;174(1):84–93. doi: 10.1164/rccm.200512-1922OC. [DOI] [PubMed] [Google Scholar]
- 8.Long W., Deng X., Zhang Y., Lu G., Xie J., Tang J. Procalcitonin guidance for reduction of antibiotic use in low-risk outpatients with community-acquired pneumonia. Respirology. 2011;16(5):819–824. doi: 10.1111/j.1440-1843.2011.01978.x. [DOI] [PubMed] [Google Scholar]
- 9.Stolz D., Smyrnios N., Eggimann P., et al. Procalcitonin for reduced antibiotic exposure in ventilator-associated pneumonia: a randomised study. The European Respiratory Journal. 2009;34(6):1364–1375. doi: 10.1183/09031936.00053209. [DOI] [PubMed] [Google Scholar]
- 10.Bouadma L., Luyt C. E., Tubach F., et al. Use of procalcitonin to reduce patients’ exposure to antibiotics in intensive care units (PRORATA trial): a multicentre randomised controlled trial. Lancet. 2010;375(9713):463–474. doi: 10.1016/S0140-6736(09)61879-1. [DOI] [PubMed] [Google Scholar]
- 11.Schuetz P., Müller B., Christ-Crain M., et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. The Cochrane Database of Systematic Reviews. 2012;9(4, CD007498):1297–1371. doi: 10.1002/ebch.1927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Linder M. M., Wacha H., Feldmann U., Wesch G., Streifensand R. A., Gundlach E. The Mannheim peritonitis index. An instrument for the intraoperative prognosis of peritonitis. Zeitschrift fur Alle Gebiete der Operativen Medizen. 1987;58(2):84–92. Der Chirurg. [PubMed] [Google Scholar]
- 13.Huang T. S., Huang S. S., Shyu Y. C., et al. A procalcitonin-based algorithm to guide antibiotic therapy in secondary peritonitis following emergency surgery: a prospective study with propensity score matching analysis. PloS One. 2014;9(3, e90539) doi: 10.1371/journal.pone.0092811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chromik A. M., Endter F., Uhl W., Thiede A., Reith H. B., Mittelkotter U. Pre-emptive antibiotic treatment vs 'standard' treatment in patients with elevated serum procalcitonin levels after elective colorectal surgery: a prospective randomised pilot study. Langenbeck's Archives of Surgery / Deutsche Gesellschaft fur Chirurgie. 2006;391(3):187–194. doi: 10.1007/s00423-005-0009-1. [DOI] [PubMed] [Google Scholar]
- 15.Solomkin J. S., Mazuski J. E., Bradley J. S., et al. Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the surgical infection Society and the Infectious Diseases Society of America. Clinical Infectious Diseases. 2010;50(2):133–164. doi: 10.1086/649554. [DOI] [PubMed] [Google Scholar]
- 16.Mazuski J. E., Sawyer R. G., Nathens A. B., et al. The surgical infection Society guidelines on antimicrobial therapy for intra-abdominal infections: evidence for the recommendations. Surgical Infections. 2002;3(3):175–233. doi: 10.1089/109629602761624180. [DOI] [PubMed] [Google Scholar]
- 17.Mazuski J. E., Sawyer R. G., Nathens A. B., et al. The surgical infection Society guidelines on antimicrobial therapy for intra-abdominal infections: an executive summary. Surgical Infections. 2002;3(3):161–173. doi: 10.1089/109629602761624171. [DOI] [PubMed] [Google Scholar]


