Abstract
Background/Objectives:
Neonatal sepsis is an important cause of morbidity and mortality in the pediatric age group in spite of several attempts at mitigating its effects. This article determines the prevalence of neonatal sepsis and the pathogens responsible for sepsis as well as risk factors and outcome at the Babcock University Teaching Hospital.
Methods:
A retrospective analysis of laboratory records of consecutive babies delivered within and outside our hospital suspected of having sepsis over a 1-year period.
Results:
The isolation rate was 34% from 100 neonates with the predominant pathogens being coagulase-negative staphylococci (CONS), Staphylococcus aureus, and Klebsiella pneumoniae. The risk factors for sepsis were age <3 days (P = 0.03) and prematurity (P < 0.001). The mortality rate was 12% with risk factors for mortality being birth weight <2500 g (P = 0.005), prematurity (P = 0.036), premature rupture of membranes (P = 0.007), and delivery outside a tertiary hospital (P = 0.007). Meropenem, ciprofloxacin, and amikacin showed the highest rates of in vitro efficacy.
Conclusion:
We highlight the prevalent pathogens in our local facility to be a combination of CONS, S. aureus, and K. pneumoniae with susceptibility patterns showing meropenem, ciprofloxacin, and amikacin to be our most effective antimicrobials in vitro.
Keywords: Coagulase-negative staphylococci, Klebsiella pneumoniae, low birth weight, neonatal sepsis, premature rupture of membranes, prematurity, Staphylocoques à coagulase négative, Klebsiella pneumoniae, faible poids de naissance, sepsis néonatal, prématurité, rupture prématurée des membranes
Résumé
Contexte/Objectifs:
La septicémie néonatale est une cause importante de morbidité et de mortalité dans le groupe d’âge pédiatrique, malgré plusieurs Atténuer ses effets. Cet article détermine la prévalence du sepsis néonatal et des pathogènes responsables de la septicémie, ainsi Les facteurs de risque et les résultats au Babcock University Teaching Hospital.
Méthodes:
Une analyse rétrospective des données de laboratoire des Les bébés livrés à l’intérieur et à l’extérieur de notre hôpital soupçonnés d’avoir une septicémie sur une période d’un an.
Résultats:
Le taux d’isolement était de 34% Les nouveau-nés dont les agents pathogènes prédominants sont les staphylocoques coagulases négatifs (CONS), Staphylococcus aureus et Klebsiella pneumoniae. Les facteurs de risque de sepsis étaient l’âge <3 jours (P = 0,03) et la prématurité (P <0,001). Le taux de mortalité était de 12% avec des facteurs de risque de mortalité (P = 0,005), la prématurité (P = 0,036), la rupture prématurée des membranes (P = 0,007) et la délivrance à l’extérieur d’un tertiaire Hôpital (P = 0,007). Meropenem, ciprofl oxacine et amikacine ont montré les plus hauts taux d’efficacité in vitro.
Conclusion:
Nous soulignons Pathogènes prédominants dans notre établissement local pour être une combinaison de CONS, S. aureus et K. pneumoniae avec des schémas de susceptibilité montrant Meropenem, ciprofloxacine et amikacine pour être nos antimicrobiens les plus efficaces in vitro.
Introduction
Neonatal sepsis is an important cause of morbidity and mortality in the pediatric age group in spite of several attempts at mitigating its effects. It is one of the leading causes of death in the first 28 days of life both in the developed and developing parts of the world. The salient clinical features include systemic signs of infection such as fever, hypothermia, tachycardia, failure to thrive, lethargy, irritability, listlessness as well as isolation of a bacterial pathogen from the bloodstream; however, in a substantial number of cases signs and symptoms are nonspecific at presentation.[1,2,3]
The neonatal period is the most vulnerable period of life due to susceptibility to infectious agents. Neonates are known to be deficient in humoral and cell-mediated immunity due to the relative immaturity of their immune systems and nonexposure to infectious agents, and in addition, studies have proven that they produce immunoglobulins at a lower rate when compared to adults.[4]
Data show that as much as 40% of under-five deaths globally occur in the neonatal period, resulting in 3 million newborn deaths each year. The vast majority of these deaths usually occur in low-income countries, and about 1 million of these deaths are attributed to infectious causes, including neonatal sepsis. The survivors of neonatal sepsis are vulnerable to both short- and long-term neurodevelopmental morbidity.[5]
Despite advances in chemotherapy, sepsis remains one of the most common diseases of the neonatal period and is still a significant cause of morbidity and mortality. It contributes up to 13%–15% of all deaths during the neonatal period, particularly in developing countries where sepsis contributes as much as half of neonatal deaths. In addition, as much as one-third of the survivors of neonatal sepsis may come down with neurological sequelae. Sepsis-related mortality is, however, largely preventable with rational antimicrobial therapy and aggressive supportive care.[6]
Neonatal sepsis may be classified according to the time of onset as either early onset (<3 days old) or late onset (older than 3 days). The distinction has clinical relevance, as the early onset variant is primarily due to bacteria acquired before and during delivery, or late onset sepsis is due to bacteria acquired after delivery (health acquired or environmental sources).[7]
It is caused by a variety of Gram-positive as well as Gram-negative bacteria and sometimes fungi. The spectrum of organisms that causes neonatal sepsis changes over times and varies from country to country and this is in turn due to local patterns of antibiotic use. The agents responsible for neonatal sepsis vary across geographical boundaries and with the time of onset of illness. In addition, the local ecology and niche of bacteria causing neonatal sepsis is constantly changing. In several developing countries, Gram-negative bacteria, for example, Klebsiella pneumoniae, remains the major source of infection, however, in the developed countries, Gram-positive organisms such as Streptococcus agalactiae have been identified as the most common cause of sepsis in neonates. With respect to the etiology of neonatal sepsis, some studies have shown Klebsiella species to be the predominant microorganism causing neonatal septicemia, especially late-onset sepsis.[8,9,10]
The diagnosis and management of sepsis are a great challenge facing clinical microbiologists and pediatricians. The diagnosis of sepsis in neonates is challenging due to nonspecific signs and symptoms. In addition, laboratory diagnosis until recently was time-consuming. This, therefore, necessitates the initiation of empirical antibiotic therapy pending a definitive diagnosis.[11]
Pathogens implicated in neonatal sepsis have gradually developed increased drug resistance to commonly used antibiotics as a result of selection pressure, which is an inevitable phenomenon in antimicrobial use and thus making treatment extremely difficult.[12,13]
Aim
This study is aimed at evaluating causative organisms of neonatal sepsis and their antibiotic sensitivity patterns.
Objectives
The objective of this study is to determine the prevalence of neonatal sepsis and the baseline susceptibility rates of prevalent pathogens causing such in our local environment and the outcome of neonatal sepsis.
Methods
Study location
The study was conducted at the Babcock University Teaching Hospital, Ilishan-Remo, Ogun State, Nigeria. Ilishan is located in Ogun State in Southwest Nigeria. The Babcock University Teaching Hospital is a 140-bed facility that caters for the immediate community and surrounding environs. The hospital has a 14-bed neonatal unit that caters for the local community.
Study design
It was a retrospective, cross-sectional study that enrolled newborns admitted to our neonatal unit with risk factors or clinical signs of sepsis and positive blood culture from August 2014 to August 2015.
Inclusion criteria
All the admitted inborn and outborn neonates during this study period were screened for sepsis based on clinical features. All infants satisfying the criteria for sepsis will be subjected to blood culture. Inclusion criteria for sepsis in neonates included fever, hypothermia, failure to thrive, bulging anterior fontanelle, respiratory distress, and seizures. Others include refusal of feeds, irritability, vomiting, lethargy as well as a full blood count picture showing leukopenia or leukocytosis as well as >10% band forms on a blood film.
Exclusion criteria
Neonates of parents who refuse to provide informed consent shall be excluded from the study.
Sample size
The sample size was calculated using the Kish formula with a prevalence of 15% resulting in a sample size of 120 patients.
Isolation of organisms
Blood culture was performed by the BACTEC 9050 system, which is a continuous self- monitoring setup that uses the production of CO2 by bacteria as a means of detecting growth.
Bacterial growth from the liquid broth was subcultured on enriched and selective media including blood, chocolate, and MacConkey and examined for growth after 24–48 h of incubation. Isolates obtained were identified by standard microbiological techniques, namely, Gram staining, colony characteristics, and biochemical properties including catalase, coagulase, for Gram-positive isolates. The Microbact 12A (Oxoid, UK) was used to confirm the identification of Gram-negative isolates.
Antimicrobial susceptibility testing
Antibiotic sensitivity testing was performed by the modified Kirby-Bauer disc diffusion method on Mueller-Hinton agar (Oxoid, UK) according to the recommendations of the Clinical and Laboratory Standard Institute (2013).
The antibiotics tested were ampicillin (10μg), oxacillin (1μg), amoxicillin-clavulanic acid (30μg), cefoxitin (30μg), cefotaxime (30μg), ceftriaxone (30μg), ceftazidime (30μg), imipenem (10μg), vancomycin (30μg), genticin (10μg), amikacin (30μg), erythromycin (15μg), and ciprofloxacin (5μg).
Data analysis/safety
Data on maternal, neonatal risk factors were obtained from the case records and entered into Epi Info 3.5.1 (Center for Disease Control Atlanta, GA, USA). P value was set at <0.05 while the Chi-square test was used to compare two groups. The odds ratio (OR) was calculated with the confidence interval (CI) set at 95%.
Results
There were 100 neonates data recruited in the study. The male to female ratio was 1.1:1 as there were 52 males to 48 females, most babies were <3 day old, n = 65 while the remaining 35 were older than 3 days. The mortality rate was 12%, and the isolation rate from the BACTEC blood culture system was 34%. Of the 88 patients who were discharged two had sequelae–hydrocephalus and cerebral palsy each. The majority of parents were in the low socioeconomic bracket, n = 48 (38.9%). Ten of the neonates had preterm premature rupture of membranes (PROMs) (10%), 17% were delivered after prolonged labor. All our babies were singleton deliveries. Sepsis was equal in incidence in both early and late-onset variants [Table 1].
Table 1.
Summary statistics of neonates
| Variable | n (%) |
|---|---|
| Age (days) | |
| <3 | 65 (65) |
| >3 | 35 (35) |
| Growth | |
| Positive | 34 (34.3) |
| Negative | 66 (65.7) |
| Outcome | |
| Died | 12 (12) |
| Discharged | 88 (88) |
| Gestation | |
| Preterm | 19 (35.2) |
| Term | 35 (64.8) |
| Birth weight | |
| >2500 | 72 (42.5) |
| <2500 | 28 (57.5) |
| Socioeconomic status | |
| High | 16 (14.9) |
| Middle | 36 (36.2) |
| Low | 48 (38.9) |
| Meconium stained amniotic fluid | |
| Present | 2 (2) |
| Absent | 98 (98) |
| Premature rupture of membranes | |
| Present | 12 (12) |
| Absent | 88 (88) |
| Prolonged labor | |
| Yes | 17 (17) |
| No | 83 (83) |
| Gender | |
| Male | 52 (52.5) |
| Female | 48 (47.5) |
The risk factors discovered in this study for mortality were birth weight <2500 g (P = 0.005, OR = 0.15, 95% CI = 0.04–0.6, χ2 = 2.83), prematurity (P = 0.036, OR = 4.32, 95% CI = 1.2–15.7, χ2 = 2.22), PROMs (P = 0.007, OR = 5.85, 95% CI = 1.6–20.9, χ2 = 2.71), and delivery outside a tertiary hospital (P = 0.007, OR = 5.85, 95% CI = 1.6–20.9, χ2 = 2.71) [Table 2].
Table 2.
Outcome of neonates with bacteriologic sepsis
| Variable | Outcome | OR | 95% CI | χ2 | P | |
|---|---|---|---|---|---|---|
| Died | Discharged | |||||
| Age (days) | ||||||
| <3 | 8 (12.1) | 58 (87.9) | 1.04 | 0.45-5.1 | 0.003 | 0.96 |
| >3 | 4 (11.8) | 30 (88.2) | ||||
| Birth weight (g) | ||||||
| >2500 | 4 (5.6) | 68 (94.4) | 0.15 | 0.04-0.6 | 10.11 | 0.004 |
| <2500 | 8 (28.6) | 20 (71.4) | ||||
| Birth status | ||||||
| Preterm | 8 (21.1) | 30 (78.9) | 3.87 | 1.1-13.9 | 4.76 | 0.036 |
| Term | 4 (6.5) | 58 (93.5) | ||||
| PROM | ||||||
| Present | 6 (46.2) | 13 (53.8) | 5.77 | 1.6-20.7 | 8.52 | 0.007 |
| Absent | 6 (10.5) | 75 (89.5) | ||||
| Parity | ||||||
| <3 | 8 (10.5) | 68 (89.5) | 0.62 | 0.17-2.3 | 0.99 | 0.32 |
| >3 | 4 (16.7) | 20 (83.3) | ||||
| Delivery | ||||||
| BUTH | 8 (42.1) | 13 (57.9) | 5.85 | 1.6-20.9 | 2.71 | 0.007 |
| Sepsis | ||||||
| Outside BUTH | 4 (5.1) | 75 (74.9) | 0.35 | 0.08-1.5 | 1.41 | 0.16 |
| Early onset | 9 (53) | 8 (47) | ||||
| Late onset | 13 (76) | 4 (34) | ||||
OR=Odds ratio, CI=Confidence interval, PROM=Premature rupture of membranes, BUTH=Babcock University Teaching Hospital
The risk factors for sepsis in this study was age <3 days (P = 0.03, OR = 2.6, 95% CI = 1.1–6.2, χ2 = 4.86) and prematurity (P = <0.001, OR = 17.3, 95% CI = 6.1–49.1, χ2 = 35.03) [Table 3].
Table 3.
Risk factors for neonatal sepsis
| Variable | Growth | OR | 95% CI | χ2 | P | |
|---|---|---|---|---|---|---|
| Negative | Positive | |||||
| Age (days) | ||||||
| <3 | 48 (73.8) | 17 (26.2) | 2.61 | 1.1-6.2 | 5.09 | 0.03 |
| >3 | 18 (51.4) | 17 (48.6) | ||||
| Anemia | ||||||
| Yes | 4 (57.1) | 3 (42.9) | 0.69 | 0.15-3.3 | 0.21 | 0.65 |
| No | 61 (65.6) | 32 (34.4) | ||||
| Birth status | ||||||
| Preterm | 42 (73.7) | 15 (26.3) | 17.3 | 6.1-49.1 | 35.03 | 0.001 |
| Term | 6 (13.9) | 37 (86.1) | ||||
| Booking status | ||||||
| Inborn | 39 (62.9) | 23 (37.1) | 1.06 | 0.46-2.43 | 0.69 | 0.89 |
| Out born | 27 (71.1) | 11 (28.9) | ||||
| Gender | ||||||
| Female | 32 (66.7) | 16 (33.3) | 0.72 | 0.31-1.65 | 0.62 | 0.43 |
| Male | 34 (65.4) | 18 (34.6) | ||||
| Prolonged labor | ||||||
| Yes | 11 (64.7) | 6 (35.3) | 0.93 | 0.31-2.79 | 0.02 | 0.91 |
| No | 55 (66.2) | 28 (33.8) | ||||
| PROM | ||||||
| Present | 6 (50) | 6 (50) | 0.44 | 0.13-1.49 | 1.78 | 0.19 |
| Absent | 61 (69.3) | 27 (31.7) | ||||
OR=Odds ratio, CI=Confidence interval, PROM=Premature rupture of membranes
The predominant organism was coagulase-negative staphylococci (CONS) n = 11, followed closely by Staphylococcus aureus n = 9, K. pneumoniae n = 8, and Pseudomonas aeruginosa n = 6. Others were Escherichia coli n = 3, Proteus mirabilis n = 2, and finally Acinetobacter baumannii n = 1 [Figure 1].
Figure 1.
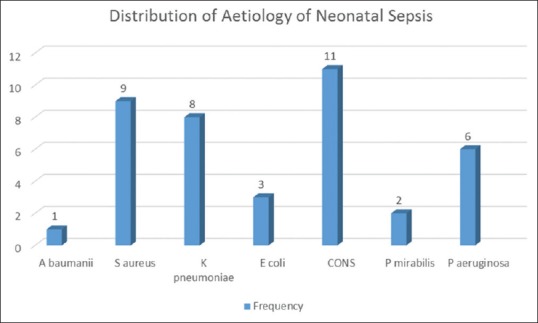
Distribution of organisms causing neonatal sepsis
Meropenem displayed the highest level of in vitro efficacy as susceptibility by pathogens was 94.1%; the next were ciprofloxacin 77.4, clindamycin 73.3%, and amikacin 68.9%. Piperacillin-tazobactam and cefepime showed 66.7% co-amoxiclav 53.9% susceptibility by organisms such as cefuroxime 58.3%, ceftazidime 50%, and ampicillin-sulbactam 12.5% [Figure 2].
Figure 2.
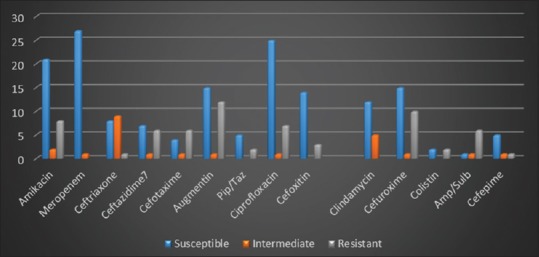
Antimicrobial susceptibility profiles of pathogens isolated in neonatal sepsis
Discussion
It is estimated that over a million deaths in neonates each year occur due to invasive infections such as bloodstream infections. The mortality rates in such instances tend to be high due to their poorly developed immune systems, and as a result, active surveillance of such infections are needed to curb this trend.[14]
Accurately defining the etiology of neonatal sepsis in each treatment facility is, therefore, important to optimize treatment outcomes and reduce mortality. This is because the microecological flora of each hospital is different. The risk factors identified in this study predominate in the early onset of sepsis; however, they are also found in late-onset sepsis. These include low birth weight, prematurity, and PROMs. This has also been elucidated by other studies which elucidate similar risk factors in neonates for sepsis.[15]
The effect of neonatal sepsis is felt globally as it is an important cause of neonatal morbidity and mortality with varied etiologies in different hospitals. Sepsis is a common serious problem in preterm infants in the Neonatal Intensive Care Units (NICUs). Diagnosis can be difficult because clinical manifestations are not specific and none of the available laboratory tests can be considered an ideal marker. For this reason, a combination of markers has been proposed; however, automated blood culture still remains the gold standard in its diagnosis.[16]
Among the 100 neonates recruited in this study, we had an isolation rate of 34% from a pool of patients with suspected sepsis. We discovered that the prevalence of early and late onset sepsis were both equal in our neonates which is not the norm in neonatal sepsis and as such our findings differ from those of the prior studies such as in Addis Ababa, Ethiopia where 81% were diagnosed as early onset and 19% late onset.[17]
The isolation rate of 34% is considered high in any environment, and these points to defects in infection control practices which are still weak in developing countries. Mugalu et al. in Uganda reported an isolation rate of 37% from 293 patients with a provisional diagnosis of sepsis which is similar to our figure. Researchers in Port Harcourt Nigeria also reported similar findings with an isolation rate of 33.1%.[18,19,20]
The diagnosis of neonatal sepsis is traditionally based on a combination of clinical presentations and automated blood culture such as the BACTEC 9050; however, in the recent past, there has been an increase in the use of nonspecific markers, including C-reactive protein and procalcitonin. It is, therefore, pertinent that neonatal sepsis be promptly and accurately diagnosed in resource challenged settings to improve on mortality rates. This necessitates the need to move toward automation to increase the yield from blood culture is evident in this study that employed the BACTEC 9050. The resins which bind antimicrobials are an added feature that increases isolation rates.[21]
These mortality rates can be driven down through the prompt recognition of babies at risk for sepsis. According to West and Tabansi of the University of Port Harcourt, the main risk factors for neonatal sepsis were inborn delivery, birth asphyxia, and prematurity. In their study, however, K. pneumoniae and S. aureus were the predominant organisms.[19]
Regarding the agents of sepsis, a similar study to ours in Egypt showed a similar pathogen profile with CONS being their predominant organism followed by K. pneumoniae with reported mortality rates of 51% and 42.9% for early and late onset sepsis, respectively. Their antimicrobial profile also showed that imipenem, ciprofloxacin, vancomycin, and amikacin performed creditably well, which was also seen in our antibiograms.[22]
Published studies also show a high incidence of CONS causing sepsis in other hospitals, however, with high rates of oxacillin resistance which is the reverse from our findings probably due to relatively lower antimicrobial usage in our facility, which in turn could be due to our fewer year of existence as a health-care facility.[23]
The recent trends from similar studies suggest that Gram-negative organisms such as K. pneumoniae which is quite prevalent in our environment display a high level of resistance to ampicillin, ceftazidime, and cefotaxime with lower rates of resistance to aminoglycosides, carbapenems, and ciprofloxacin.[24]
This is important because the default antimicrobial combination in use by our pediatricians is a combination of either cefuroxime and amikacin or ceftazidime and amikacin pending results of susceptibility profiles from the clinical microbiology laboratory.
Some studies, now, report high resistance to antibiotics used commonly for the empirical treatment of neonatal sepsis due to the widespread use of third-generation cephalosporins and lack of reliance on blood culture reports as well as inappropriate therapeutic regimens.[25]
In conjunction with prematurity, neonates who had PROM were 5.35 times more likely to die compared to who did not. The previous studies do show that PROM results in an increase in ascending infection into the birth canal and subsequently to the body in utero. PROM is an important risk factor for early-onset neonatal sepsis and preterm births, and in developing continues, it continues to be a major cause of mortality and morbidity among neonates.[26,27]
The association of low birthweight and prematurity with poor outcome has been demonstrated in other studies. The less mature preterm infant is at increased risk for developing complications of sepsis because of deficiencies in humoral and cellular immunity. Transplacental maternal antibodies primarily mediate humoral immunity; hence, preterm infants are less likely to receive as many immunoglobulins as term infants. Furthermore, T-cell function and phagocytic function are also deficient.[28]
These findings, therefore, portray prematurity as one of the most consistent significant cofactor associated with increased perinatal morbidity and mortality in NICUs. It has also been demonstrated that premature and low birthweight babies tend to be immune deficient and this explains why prematurity was identified as a risk factor for sepsis in this study as with other studies.[29]
In relation to the outcome of sepsis, previous data show the long-term effect of sepsis on neonates, especially those who weight <2500 g. This is also seen in some of our neonates who subsequently developed hydrocephalus and cerebral palsy.[30]
With respect to late onset sepsis which is largely controllable, the prevention of healthcare-associated infections through antimicrobial stewardship, minimal handling of neonates, the limited use of invasive devices and standardization of catheter care practices, and meticulous hand hygiene are important and cost-effective strategies for reducing the burden of neonatal sepsis in our NICU’s.[14]
Conclusion
This study has highlighted the prevalent pathogens in our local facility to be a combination of CONS and K. pneumoniae with susceptibility patterns showing meropenem, ciprofloxacin, and amikacin to be our most effective antimicrobials in vitro. The clinician should have a heightened sense of awareness in neonates with prematurity, PROM, and age <3 days. There is the need for widespread use of automated blood culture systems to enhance isolation rates, detect sepsis early, and thereby reduce mortality.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.Sanghvi KP, Tudehope DI. Neonatal bacterial sepsis in a neonatal Intensive Care Unit: A 5 year analysis. J Paediatr Child Health. 1996;32:333–8. doi: 10.1111/j.1440-1754.1996.tb02565.x. [DOI] [PubMed] [Google Scholar]
- 2.Ogunlesi TA, Ogunfowora OB. Predictors of mortality in neonatal septicemia in an underresourced setting. J Natl Med Assoc. 2010;102:915–21. doi: 10.1016/s0027-9684(15)30710-0. [DOI] [PubMed] [Google Scholar]
- 3.Dammann O, Kuban KC, Leviton A. Perinatal infection, fetal inflammatory response, white matter damage, and cognitive limitations in children born preterm. Ment Retard Dev Disabil Res Rev. 2002;8:46–50. doi: 10.1002/mrdd.10005. [DOI] [PubMed] [Google Scholar]
- 4.Shobowale EO, Ogunsola FT, Oduyebo OO, Ezeaka VI. A study on the outcome of neonates with sepsis at the Lagos University Teaching Hospital. Int J Med Biomed Res. 2015;4:46–9. [Google Scholar]
- 5.Shehab El-Din EM, El-Sokkary MM, Bassiouny MR, Hassan R. Epidemiology of neonatal sepsis and implicated pathogens: A study from Egypt. Biomed Res Int 2015. 2015 doi: 10.1155/2015/509484. 509484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.West BA, Peterside O. Sensitivity pattern among bacterial isolates in neonatal septicaemia in Port Harcourt. Ann Clin Microbiol Antimicrob. 2012;11:7. doi: 10.1186/1476-0711-11-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med. 2013;369:840–51. doi: 10.1056/NEJMra1208623. [DOI] [PubMed] [Google Scholar]
- 8.Vergnano S, Sharland M, Kazembe P, Mwansambo C, Heath PT. Neonatal sepsis: An international perspective. Arch Dis Child Fetal Neonatal Ed. 2005;90:220–4. doi: 10.1136/adc.2002.022863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jyothi P, Basavaraj MC, Basavaraj PV. Bacteriological profile of neonatal septicemia and antibiotic susceptibility pattern of the isolates. J Nat Sci Biol Med. 2013;4:306–9. doi: 10.4103/0976-9668.116981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Omoregie R, Egbe CA, Dirisu J, Ogefere HO. Microbiology of neonatal septicemia in a tertiary hospital in Benin City, Nigeria. Biomark Genomic Med. 2013;5:142–4. [Google Scholar]
- 11.Edwards MS, Baker CJ. Sepsis in the newborn. In: Gershon AA, Hotez PJ, Katz SL, editors. Krugman's Infectious Diseases of Children. Philadelphia, PA, USA: Mosby; 2004. p. 545. [Google Scholar]
- 12.Placzek MM, Whitelaw A. Early and late neonatal septicaemia. Arch Dis Child. 1983;58:728–31. doi: 10.1136/adc.58.9.728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Al-Zwaini EJ. Neonatal septicaemia in the neonatal care unit, Al-Anbar governorate, Iraq. East Mediterr Health J. 2002;8:509–14. [PubMed] [Google Scholar]
- 14.Shane AL, Stoll BJ. Neonatal sepsis: Progress towards improved outcomes. J Infect. 2014;68(Suppl 1):S24–32. doi: 10.1016/j.jinf.2013.09.011. [DOI] [PubMed] [Google Scholar]
- 15.Leal YA, Álvarez-Nemegyei J, Velázquez JR, Rosado-Quiab U, Diego-Rodríguez N, Paz-Baeza E, et al. Risk factors and prognosis for neonatal sepsis in Southeastern Mexico: Analysis of a four-year historic cohort follow-up. BMC Pregnancy Childbirth. 2012;12:48. doi: 10.1186/1471-2393-12-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Maria RB, Lígia MS, De Souza R. Late-onset sepsis: Epidemiology, evaluation, and outcome. Neoreviews. 2010;11:110–5. [Google Scholar]
- 17.Shitaye D, Asrat D, Woldeamanuel Y, Worku B. Risk factors and etiology of neonatal sepsis in Tikur Anbessa University Hospital, Ethiopia. Ethiop Med J. 2010;48:11–21. [PubMed] [Google Scholar]
- 18.Mugalu J, Nakakeeto MK, Kiguli S, Kaddu-Mulindwa DH. Aetiology, risk factors and immediate outcome of bacteriologically confirmed neonatal septicaemia in Mulago hospital, Uganda. Afr Health Sci. 2006;6:120–6. doi: 10.5555/afhs.2006.6.2.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.West BA, Tabansi PN. Prevalence of neonatal septicemia in the University of Port Harcourt Teaching Hospital Nigeria. Niger J Paediatr. 2014;41:33–7. [Google Scholar]
- 20.Ogunlesi TA, Ogunfowora OB, Osinupebi O, Olanrewaju DM. Changing trends in newborn sepsis in Sagamu, Nigeria: Bacterial aetiology, risk factors and antibiotic susceptibility. J Paediatr Child Health. 2011;47:5–11. doi: 10.1111/j.1440-1754.2010.01882.x. [DOI] [PubMed] [Google Scholar]
- 21.Flayhart D, Borek AP, Wakefield T, Dick J, Carroll KC. Comparison of BACTEC PLUS blood culture media to BacT/Alert FA blood culture media for detection of bacterial pathogens in samples containing therapeutic levels of antibiotics. J Clin Microbiol. 2007;45:816–21. doi: 10.1128/JCM.02064-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Eman MP, Mohammed AE, Mohammed RB, Ramadem H. Epidemiology of neonatal sepsis and implicated pathogens: A study from Egypt. Biomed Res Int 2015. 2015 doi: 10.1155/2015/509484. Article ID 509484. [Doi: 10.1155/2015/509484] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.van den Hoogen A, Gerards LJ, Verboon-Maciolek MA, Fleer A, Krediet TG. Long-term trends in the epidemiology of neonatal sepsis and antibiotic susceptibility of causative agents. Neonatology. 2010;97:22–8. doi: 10.1159/000226604. [DOI] [PubMed] [Google Scholar]
- 24.Aurangzeb B, Hameed A. Neonatal sepsis in hospital-born babies: Bacterial isolates and antibiotic susceptibility patterns. J Coll Physicians Surg Pak. 2003;13:629–32. [PubMed] [Google Scholar]
- 25.Khatua SP, Das AK, Chatterjee BD, Khatua S, Ghose B, Saha A. Neonatal septicemia. Indian J Pediatr. 1986;53:509–14. doi: 10.1007/BF02749537. [DOI] [PubMed] [Google Scholar]
- 26.Hasan MS, Mohammed B. Predictive values of risk factors in neonatal sepsis. Bangladesh Coll Physiol. 2011;2:187–95. [Google Scholar]
- 27.Alam MM, Saleem AF, Shaikh AS, Munir O, Qadir M. Neonatal sepsis following prolonged rupture of membranes in a tertiary care hospital in Karachi, Pakistan. J Infect Dev Ctries. 2014;8:67–73. doi: 10.3855/jidc.3136. [DOI] [PubMed] [Google Scholar]
- 28.Trotman H, Bell Y, Thame M, Nicholson AM, Barton M. Predictors of poor outcome in neonates with bacterial sepsis admitted to the University Hospital of the West Indies. West Indian Med J. 2006;55:80–4. doi: 10.1590/s0043-31442006000200003. [DOI] [PubMed] [Google Scholar]
- 29.Shah GS, Budhathoki S, Das BK, Mandal RN. Risk factors in early neonatal sepsis. Kathmandu Univ Med J (KUMJ) 2006;4:187–91. [PubMed] [Google Scholar]
- 30.Alshaikh B, Yusuf K, Sauve R. Neurodevelopmental outcomes of very low birth weight infants with neonatal sepsis: Systematic review and meta-analysis. J Perinatol. 2013;33:558–64. doi: 10.1038/jp.2012.167. [DOI] [PubMed] [Google Scholar]


