Abstract
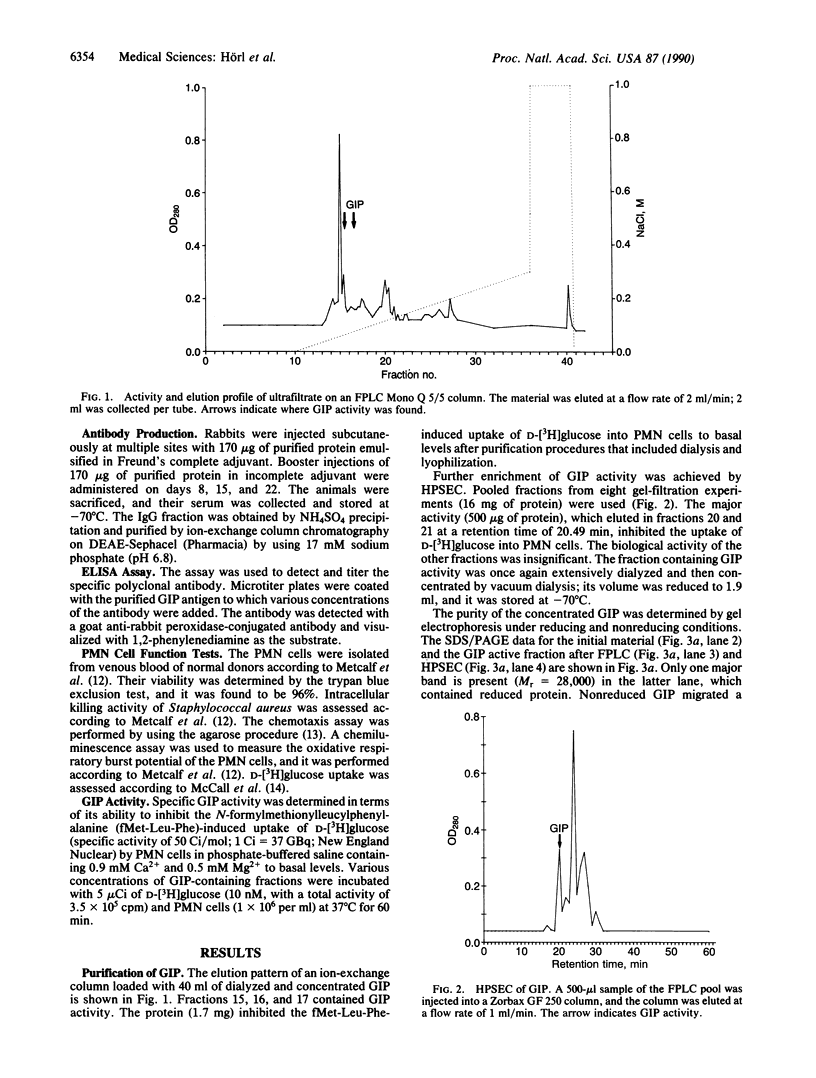
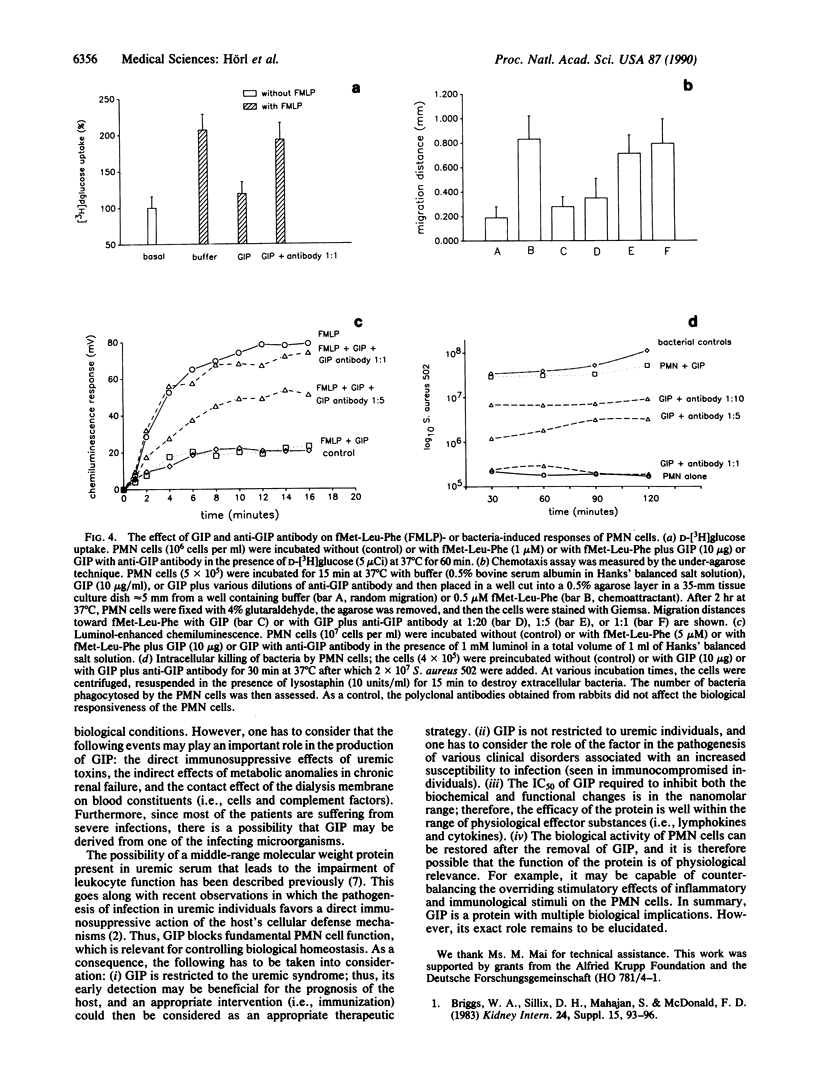
A granulocyte inhibitory protein was isolated and characterized from uremic serum by using ion-exchange column chromatography, high-performance size-exclusion chromatography, and immunochemical procedures. The purification process concentrated the protein 240-fold and to a purity of greater than 95%. An overall recovery of 45% was achieved; the purified protein had a specific activity of 104 units per mg of protein. The polypeptide had a molecular weight of approximately 28,000 and an isoelectric point of 4.0-4.5. Amino acid sequencing of the NH2 terminus revealed a single sequence (Asp-Ile-Val-Met-Thr-Gln-Ser-Pro-Gly-Thr-Leu-Ser-Val-Ser-Pro-Gly-Glu-Arg-Ala- Thr) that proved to be nonhomologous with other serum proteins that appear during an inflammatory state. The polypeptide inhibited the uptake of deoxyglucose, chemotaxis, oxidative metabolism, and intracellular bacterial killing by polymorphonuclear leukocytes. A specific rabbit polyclonal antibody raised against the protein nullified these inhibitory changes. We contend that the protein is responsible for the leukocyte dysfunction that is commonly seen in patients with uremia.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Hunkapiller M. W., Hewick R. M., Dreyer W. J., Hood L. E. High-sensitivity sequencing with a gas-phase sequenator. Methods Enzymol. 1983;91:399–413. doi: 10.1016/s0076-6879(83)91038-8. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Mansell M. A., Grimes A. J., Jones N. F. Leucocyte ATP and renal failure. Clin Sci (Lond) 1981 Jul;61(1):43–46. doi: 10.1042/cs0610043. [DOI] [PubMed] [Google Scholar]
- McCall C., Schmitt J., Cousart S., O'Flaherty J., Bass D., Wykle R. Stimulation of hexose transport by human polymorphonuclear leukocytes: a possible role for protein kinase C. Biochem Biophys Res Commun. 1985 Jan 16;126(1):450–456. doi: 10.1016/0006-291x(85)90626-6. [DOI] [PubMed] [Google Scholar]
- Metcoff J., Lindeman R., Baxter D., Pederson J. Cell metabolism in uremia. Am J Clin Nutr. 1978 Sep;31(9):1627–1634. doi: 10.1093/ajcn/31.9.1627. [DOI] [PubMed] [Google Scholar]
- Nelson R. D., Quie P. G., Simmons R. L. Chemotaxis under agarose: a new and simple method for measuring chemotaxis and spontaneous migration of human polymorphonuclear leukocytes and monocytes. J Immunol. 1975 Dec;115(6):1650–1656. [PubMed] [Google Scholar]
- Ritchey E. E., Wallin J. D., Shah S. V. Chemiluminescence and superoxide anion production by leukocytes from chronic hemodialysis patients. Kidney Int. 1981 Feb;19(2):349–358. doi: 10.1038/ki.1981.26. [DOI] [PubMed] [Google Scholar]
- Siriwatratananonta P., Sinsakul V., Stern K., Slavin R. G. Defective chemotaxis in uremia. J Lab Clin Med. 1978 Sep;92(3):402–407. [PubMed] [Google Scholar]
- Tolkoff-Rubin N. E., Rubin R. H. Uremia and host defenses. N Engl J Med. 1990 Mar 15;322(11):770–772. doi: 10.1056/NEJM199003153221109. [DOI] [PubMed] [Google Scholar]