Abstract
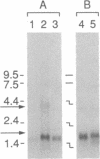
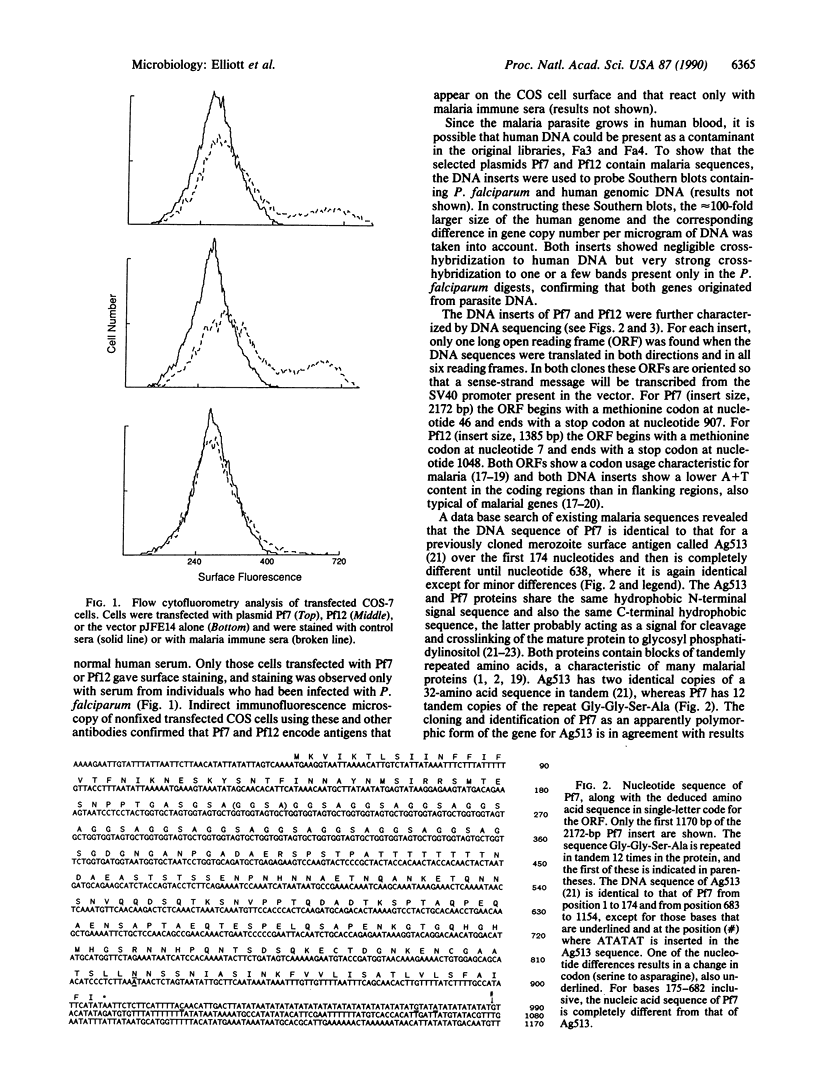
Two genes encoding membrane antigens of Plasmodium falciparum were isolated by transient expression in mammalian cells and selection with human immune sera from African adults exposed to P. falciparum malaria. COS-7 cells were transfected with a plasmid expression library constructed from P. falciparum genomic DNA, and cells expressing reactive malaria antigens on their surface were enriched by adherence to antibody-coated dishes. One of the genes isolated is distinctive in that it does not contain repeat sequences typical of many malarial genes cloned by immunoscreening of bacterial expression libraries. The second gene apparently encodes a polymorphic version of the P. falciparum merozoite surface antigen Ag513, since the two sequences are identical in the 5' and 3' coding regions but diverge completely in the center. The COS-7 expression system provides an alternate means for cloning genes encoding malarial membrane antigens by using those antibodies in complex immune sera that bind membrane-associated, nondenatured molecules.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anders R. F., Smythe J. A. Polymorphic antigens in Plasmodium falciparum. Blood. 1989 Nov 1;74(6):1865–1875. [PubMed] [Google Scholar]
- Aruffo A., Seed B. Molecular cloning of a CD28 cDNA by a high-efficiency COS cell expression system. Proc Natl Acad Sci U S A. 1987 Dec;84(23):8573–8577. doi: 10.1073/pnas.84.23.8573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aruffo A., Seed B. Molecular cloning of two CD7 (T-cell leukemia antigen) cDNAs by a COS cell expression system. EMBO J. 1987 Nov;6(11):3313–3316. doi: 10.1002/j.1460-2075.1987.tb02651.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bleackley R. C., Caplan B., Havele C., Ritzel R. G., Mosmann T. R., Farrar J. J., Paetkau V. Translation of lymphocyte mRNA into biologically-active Interleukin 2 in oocytes. J Immunol. 1981 Dec;127(6):2432–2435. [PubMed] [Google Scholar]
- Caras I. W., Weddell G. N. Signal peptide for protein secretion directing glycophospholipid membrane anchor attachment. Science. 1989 Mar 3;243(4895):1196–1198. doi: 10.1126/science.2466338. [DOI] [PubMed] [Google Scholar]
- Dower W. J., Miller J. F., Ragsdale C. W. High efficiency transformation of E. coli by high voltage electroporation. Nucleic Acids Res. 1988 Jul 11;16(13):6127–6145. doi: 10.1093/nar/16.13.6127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Epping R. J., Goldstone S. D., Ingram L. T., Upcroft J. A., Ramasamy R., Cooper J. A., Bushell G. R., Geysen H. M. An epitope recognised by inhibitory monoclonal antibodies that react with a 51 kilodalton merozoite surface antigen in Plasmodium falciparum. Mol Biochem Parasitol. 1988 Feb;28(1):1–10. doi: 10.1016/0166-6851(88)90173-9. [DOI] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983 Jul 1;132(1):6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- Ferguson M. A., Williams A. F. Cell-surface anchoring of proteins via glycosyl-phosphatidylinositol structures. Annu Rev Biochem. 1988;57:285–320. doi: 10.1146/annurev.bi.57.070188.001441. [DOI] [PubMed] [Google Scholar]
- Hoopes B. C., McClure W. R. Studies on the selectivity of DNA precipitation by spermine. Nucleic Acids Res. 1981 Oct 24;9(20):5493–5504. doi: 10.1093/nar/9.20.5493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard R. J., Barnwell J. W., Rock E. P., Neequaye J., Ofori-Adjei D., Maloy W. L., Lyon J. A., Saul A. Two approximately 300 kilodalton Plasmodium falciparum proteins at the surface membrane of infected erythrocytes. Mol Biochem Parasitol. 1988 Jan 15;27(2-3):207–223. doi: 10.1016/0166-6851(88)90040-0. [DOI] [PubMed] [Google Scholar]
- Hyde J. E., Sims P. F. Anomalous dinucleotide frequencies in both coding and non-coding regions from the genome of the human malaria parasite Plasmodium falciparum. Gene. 1987;61(2):177–187. doi: 10.1016/0378-1119(87)90112-0. [DOI] [PubMed] [Google Scholar]
- Kemp D. J., Coppel R. L., Anders R. F. Repetitive proteins and genes of malaria. Annu Rev Microbiol. 1987;41:181–208. doi: 10.1146/annurev.mi.41.100187.001145. [DOI] [PubMed] [Google Scholar]
- Okayama H., Berg P. A cDNA cloning vector that permits expression of cDNA inserts in mammalian cells. Mol Cell Biol. 1983 Feb;3(2):280–289. doi: 10.1128/mcb.3.2.280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramasamy R. Studies on glycoproteins in the human malaria parasite Plasmodium falciparum. Identification of a myristilated 45kDa merozoite membrane glycoprotein. Immunol Cell Biol. 1987 Oct;65(Pt 5):419–424. doi: 10.1038/icb.1987.48. [DOI] [PubMed] [Google Scholar]
- Sandri-Goldin R. M., Goldin A. L., Levine M., Glorioso J. C. High-frequency transfer of cloned herpes simplex virus type 1 sequences to mammalian cells by protoplast fusion. Mol Cell Biol. 1981 Aug;1(8):743–752. doi: 10.1128/mcb.1.8.743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saul A., Battistutta D. Codon usage in Plasmodium falciparum. Mol Biochem Parasitol. 1988 Jan 1;27(1):35–42. doi: 10.1016/0166-6851(88)90022-9. [DOI] [PubMed] [Google Scholar]
- Scherf A., Hilbich C., Sieg K., Mattei D., Mercereau-Puijalon O., Müller-Hill B. The 11-1 gene of Plasmodium falciparum codes for distinct fast evolving repeats. EMBO J. 1988 Apr;7(4):1129–1137. doi: 10.1002/j.1460-2075.1988.tb02922.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seed B. An LFA-3 cDNA encodes a phospholipid-linked membrane protein homologous to its receptor CD2. 1987 Oct 29-Nov 4Nature. 329(6142):840–842. doi: 10.1038/329840a0. [DOI] [PubMed] [Google Scholar]
- Seed B., Aruffo A. Molecular cloning of the CD2 antigen, the T-cell erythrocyte receptor, by a rapid immunoselection procedure. Proc Natl Acad Sci U S A. 1987 May;84(10):3365–3369. doi: 10.1073/pnas.84.10.3365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smythe J. A., Coppel R. L., Brown G. V., Ramasamy R., Kemp D. J., Anders R. F. Identification of two integral membrane proteins of Plasmodium falciparum. Proc Natl Acad Sci U S A. 1988 Jul;85(14):5195–5199. doi: 10.1073/pnas.85.14.5195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takebe Y., Seiki M., Fujisawa J., Hoy P., Yokota K., Arai K., Yoshida M., Arai N. SR alpha promoter: an efficient and versatile mammalian cDNA expression system composed of the simian virus 40 early promoter and the R-U5 segment of human T-cell leukemia virus type 1 long terminal repeat. Mol Cell Biol. 1988 Jan;8(1):466–472. doi: 10.1128/mcb.8.1.466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber J. L. Analysis of sequences from the extremely A + T-rich genome of Plasmodium falciparum. Gene. 1987;52(1):103–109. doi: 10.1016/0378-1119(87)90399-4. [DOI] [PubMed] [Google Scholar]
- Weber J. L. Molecular biology of malaria parasites. Exp Parasitol. 1988 Aug;66(2):143–170. doi: 10.1016/0014-4894(88)90087-2. [DOI] [PubMed] [Google Scholar]
- von Heijne G. A new method for predicting signal sequence cleavage sites. Nucleic Acids Res. 1986 Jun 11;14(11):4683–4690. doi: 10.1093/nar/14.11.4683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Heijne G. Signal sequences. The limits of variation. J Mol Biol. 1985 Jul 5;184(1):99–105. doi: 10.1016/0022-2836(85)90046-4. [DOI] [PubMed] [Google Scholar]