Abstract
mTOR, the mammalian target of rapamycin, is a conserved serine/threonine kinase which belongs to the phosphatidyl-linositol kinase-related kinase (PIKK) family. It has two complexes called mTORC1 and mTORC2. It is well established that mTOR plays important roles in cell growth, proliferation and differentiation. Over-activation of the mTOR pathway is considered to have a relationship with the development of many types of diseases, including polycystic ovary syndrome (PCOS) and ovarian cancer (OC). mTOR pathway inhibitors, such as rapamycin and its derivatives, can directly or indirectly treat or relieve the symptoms of patients suffering from PCOS or OC. Moreover, mTOR inhibitors in combination with other chemical-molecular agents may have extraordinary efficacy. This paper will discuss links between mTOR signaling and PCOS and OC, and explore the mechanisms of mTOR inhibitors in treating these two diseases, with conclusions regarding the most effective therapeutic approaches.
Keywords: mTOR signaling pathway, PCOS, ovarian cancer
Introduction
Polycystic Ovary Syndrome (PCOS) is a life-long endocrinopathy, with a heterogeneous collection of signs and symptoms that influences approximately 10% of women in reproductive age (Domecq et al., 2013). The molecular pathogenesis of PCOS, however, still remains unknown. Ovarian carcinoma (OC), another ovarian-related disease, is the leading cause of death for gynecologic malignancies in female (Muralidhar and Barbolina, 2015). Even though more than 80% OC patients will have a response after initial therapy, OC still recurs in the majority of patients. With the accumulation of an increasing knowledge at genetic and genomic level in the post-genomic era, it is time to find novel method to treat the two diseases by revisit their molecular mechanisms. The phosphatidylinositol 3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) signal pathway, which was found to be especially active in PCOS and OC, is considered to have close relationship with the pathogenesis of PCOS and OC. First-generation mTOR inhibitors have demonstrated to be effective in treating in several types of tumors. In addition, a growing number of studies try to target PI3K or AKT as second-generation mTOR inhibitors to explore whether there is clinical impact.
This paper will first briefly overview the knowledge of PCOS and OC, with a focus on the relationship between PI3K/AKT/mTOR signal pathway and two diseases, and then try to dissect the rational therapeutic protocols for targeting PI3K/AKT/mTOR pathway in PCOS and OC. Some relevant studies and preclinical data related to these two diseases and mTOR pathway are going to be evaluated. A number of challenges aiming at the development of PI3K/AKT/mTOR pathway inhibitors in PCOS and OC are discussed, including the risk of drug resistance and tactics for optimizing drug combination to inhibit mTOR signal pathway in PCOS and OC.
mTOR complexes
mTOR is a serine/threonine kinase. It belongs to phosphatidylinositol kinase-related kinase (PIKK) protein family, and has been shown to actively regulate cell growth, proliferation and differentiation. The gene and the protein of mTOR are relatively conservative during the process of evolution. mTOR-mediated signaling pathway plays a crucial role in the development of a number of diseases, such as type-2 diabetes and some cancers. Therefore, exploring the mechanism of mTOR is becoming more and more attractive.
mTOR is a kind of macro-molecular protein, it will form two different kinds of complexes when it combines with different proteins: rapamycin sensitive mTORC1 and rapamycin insensitive mTORC2. mTORC1 consists of mTOR, Raptor, mLST8 and PRAS40. It plays a critical role in cell growth and proliferation, protein synthesis, metabolism and autophagy, mitochondrial and ribosomal biogenesis through regulating RNA translation and nutrient metabolism. mTORC2 is composed of mTOR, Rictor, mSin1, Protor-1 and mLST8. The major roles of mTORC2 played include cytoskelatal. organization, cell survival and polarity (Johnson et al, 2013; Laplante and Sabatini, 2013; Lee, 2015). In 1991, Heitman et al demonstrated that mTORC1 and mTORC2 were encoded by different genes in yeast. mTORC1 and mTORC2 control a large variety of cellular processes via different pathways. However, it is difficult to clearly distinguish from the two complexes based on the current evidence. Additionally, there are more studies investigating mTORC1 than mTORC2 when searching the regulation of cellular homeostasis. In fact, mTORC1 induces translation initiation via translation of the 5’TOP mRNAs, inducing protein synthesis and ribosomal biogenesis. This mechanism provides a reasonable explain why the activity of mTORC1 is frequently found to be increased in tumor cells (Johnson et al, 2013; Laplante and Sabatini, 2013). There are researches supporting that mTORC1 activity would be suppressed by energy stress, endoplasmic reticulum (ER) stress, acid deprivation and hypoxia (Johnson et al, 2013; Laplante and Sabatini, 2013).
mTORC2 mediates cellular growth and survival via regulating the serum levels of glucocorticoid induced protein kinase 1 (SGK1) and AKT. mTORC2 can also regulate cytoskelatal organization and cell polarity by regulating RhoA and PKC-α (Sciarretta et al, 2014).
Upstream regulators of mTOR complexes
There are two mTOR signaling pathways, i.e, PI3K/AKT/mTOR signaling pathway and LKB1/AMPK/TSC/mTOR signaling pathway. The former pathway works through phosphorylation. AKT can, directly or indirectly, work on mTOR. Receptor tyrosine kinases (RKT) is activated when growth factors, such as vascular Endothelial Growth Factor (VEGF) and Insulin, combined with RKT located in cell surface during the process of regulating cell growth, and subsequently, activated RKT activates AKT (Badura et al, 2013). AKT could, directly or indirectly, stimulate the whole signaling pathway through TSC1/TSC2 complex (Huang and Houghton, 2003). Due to the fact that TSC1/TSC2 complex is a negative regulator, it can suppress the activation of mTOR by combining with the upstream molecule of PI3K/AKT/mTOR pathway. AKT, however, can inhibit TSC1/TSC2 complex through phosphorylation. AKT can also signal to mTORC1 which is TSC1/TSC2-independent through phosphorylation and causing dissociation raptor from PRAS40, a mTORC1 inhibitor (Sancak et al, 2007). TSC1/TSC2 is a critical upstream regulator of mTORC1. TSC1/TSC2 can transmit many of the upstream signals that influence on mTORC1, including growth factors, to activate the PI3K pathway (Laplante and Sabatini, 2012). Moreover, some factors, such as low oxygen and energy levels, can also in some degree act on mTORC1 through TSC1/TSC2 complex, which is controlled by LKB1/SKT1-AMPK pathway (Yip, 2015). In the latter pathway, AMPK acts as energy sensor, it can act directly on mTOR just like AKT. AMPK can phosphorylate raptor, inducing 14-3-3 (a kind of protein in yeast) binding to raptor and the allosteric inhibition of mTORC1 (Gwinn et al., 2008). Additionally, AMPK can activate TSC1/2 complex, increasing GTP enzyme activity and suppressing GTP-binding protein Rheb, a mTORC1 activator. Furthermore, glycogen synthase kinase 3β (GSK-3β) could also activate TSC1/2 complex. And it can suppress mTORC1 activity under energy stress circumstance (Inoki et al, 2006). Alternatively, regulated in development and DNA damage response 1 (REDD1/2) activates mTORC1 under low oxygen circumstance with no help of AMPK and GSK-3β (Brugarolas et al., 2004).
In addition, there are many growth factors that could promote mTORC2 activity, but the upstream regulatory mechanisms are poorly understood. It has been shown that mTORC2 could be activated by insulin, however, mTORC2 is relatively not sensitive to nutrient deprivation (Laplante and Sabatini, 2012). There another report indicated that the PI3K pathway could also activate mTORC2 (Zinzalla et al, 2011). Recently, Ashworth et al (Ashworth and Wu, 2014) pointed out that mTOR played a crucial role in full activation of PKB/AKT by two steps of phosphorylation. Firstly, Thr308 of the protein is phosphorylated by PDK1. In the second step residue Ser473 is phosphorylated by mTORC2. Then, the mTORC2 could indirectly promote mTORC1 activity through the activation of PKB/AKT. It has also been shown that the TSC1/2 complex could activate mTORC2. Besides, TSC1/2 complex could physically combine with mTORC2 instead of mTORC1 (Zinzalla et al, 2011). mTORC1 seems to suppress mTORC2 activity via phosphorylation of Rictor (Dibble et al, 2009), indicating that there is functionally interconnect between mTORC1 and mTORC2.
Downstream regulators of mTOR complexes
There are two downstream targets in the activated mTOR signaling pathway: the 70s ribosomal protein S6 kinase (S6K1) and the eukaryotic translation initiation factor 4E binding protein 1 (4E-BP1). S6K1 phosphorylates eukaryotic translation initiation factor 4B (eIF4B) and eukaryotic initiation factor 2 kinase (eEF2K), leading to further promoting translation initiation (Lyu et al, 2013). S6K1 can also be phosphorylated by mTORC1, resulting in the activation of translation initiation. But sometimes it also plays a negative role on translation initiation via the phosphorylated S6K1, attenuating PI3K signaling through suppressing the activity of insulin receptor substrate 1 (IRS1) (Yap et al, 2008). 4E-BP1 is a negative regulator and can inhibit translation initiation when it combines with eIF-4E 5’TOP. mTORC1 releases eIF-4E from the binding complex when it is phosphorylated through some stimulation. Released eIF-4E binds to eIF-4A, eIF-4G and eIF-4B, forming eIF-4F complex and promoting the translation initiation (Lipton and Sahin, 2014) (Figure 1).
Figure 1.
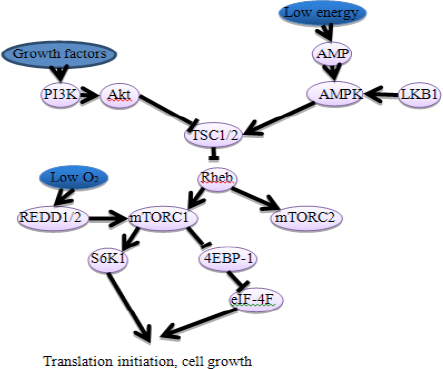
mTOR Signaling Pathway
mTOR signaling pathway in PCOS
PCOS, a common endocrine disorder syndrome, is regarded as the dominant factor for anovulatory infertility. Even through the mechanism of anovulation still remains unclear. It is reported that environmental and genetic factors play important roles in the origin and development of this disease. PCOS is mainly characterized by ovulatory dysfunction, hyperandrogenism, and insulin resistance. According to American society for reproductive medicine (ASRM) and Europe society for human reproduction and embryology (ESHRE), PCOS can be defined when it satisfies the two of the following three standards. i) ovulatory dysfunction or anovulation; ii) hyperandrogenism; and iii) there are polycystic ovaries when observed by ultrasonic diagnosis (Tsikouras et al, 2015). PCOS is also characterized by increasing level of Luteinizing hormone (LH) and Follicle stimulating hormone (FSH) ratio. PCOS patients have higher level of LH/FSH ratio than normal one, which is a common finding among women. Even so, the level of LH/FSH ratio still can not be considered as one of the diagnosis criteria (Hsu, 2015). The increased LH level will subsequently induce theca cell to produce androgens, while the deficiency of FSH level will weaken the ability of granulosa cells to turn androgen into estrogen, damaging follicle maturation and ovulation (McCartney et al, 2002). The majority of PCOS patients often accompany with some long-term complications to which have not attach much importance, such as type-2 diabetes, hyperlipidemia, obesity and glucose intolerance (Yang and Choi, 2015).
Relationship between PI3K/AKT/mTOR signal pathway and PCOS
According to Zhang et al’s (Zhang et al, 2012) experiment, PCOS (52 patients) had higher expression of phosphorylated AKT (p-AKT) as compared to the controls (32 non-PCOS patients). Interestingly, when PCOS patients were grouped as insulin resistance and non-insulin resistance based on homeostasis model assessment-insulin resistance index (HOMA-IR), p-AKT was found to have a higher expression in insulin resistance group than in non-insulin resistance group. This paper addressed that the PI3K/AKT signaling pathway was over-activated in endometrium with PCOS. And insulin resistance was probably a key factor for PI3K/AKT over-activating in endometrium with PCOS. Roa et al (Roa et al, 2009) indicated that mTOR played a crucial role in controlling puberty onset and LH secretion. mTOR could be activated by 1-leucine promoted LH secretion in pubertal female mice. However, when mTOR signaling pathway was suppressed by rapamycin, it would cause the inhibition of gonadotropic axis in puberty, decreasing estradiol and LH levels, dramaticlly delaying vaginal opening, and causing uterine and ovarian atrophy. mTOR promotes translation initiation by phosphorylating p70S6K kinase and 40S ribosomal protein S6 (Alam et al, 2004). Yaba and Demir (Yaba and Demir, 2012) indicating that mTORC1 and mTORC2 played an important role in the development of PCOS. The results suggested that mTOR increased ovarian follicular cell growth and proliferation in a mouse model of PCOS. As p-p70S6K showed a low level in POCS mouse model, it is possible that insufficient p70S6K activity led to the suppression of follicular development. In addition, Rictor, a component of mTORC2, had a high level expression in PCOS mouse model, suggesting that Rictor may play a role in the development of PCOS. Another research pointed out that mTOR could regulate follicle growth in vivo by working as a novel mitotic survival checkpoint. mTOR, which is inhibited by rapamycin, reduces follicle growth in vitro and granulosa cell proliferation (Yaba et al, 2008). Metformin could promote glucose transporters 4 (GLUT4) expression and suppress androgen receptors (AR) expression and block insulin receptor/PI3K/AKT/mTOR signal pathway on the endometrial hyperplasia tissues. The finding showed that changes of endometrial GLUT4 expression in PCOS patients were related to the androgen-dependent alteration of AR expression and changes of insulin receptor/PI3K/AKT/mTOR signal network (Li et al, 2015). It has been shown that insulin receptor substrate 1 (ISR-1), a signaling protein that binds insulin receptor to PI3K signaling cascade, is associated PI3K activity decreased in vivo in skelatal. muscle with PCOS. Besides, the skelatal. muscle from PCOS in vitro increased level of ISR-1 serine phosphorylation compared with controls. Allemand et al also guided that elevated phosphorylation of S6K and mTOR led to sub-cellular rearrangement of ISR-1, inhibiting ISR-1 activation via elevating its serine phosphorylation and finally resulting in insulin resistance (Allemand et al, 2009). WNT5α works as a pro-inflammatory factor in mural granulosa cells and was over expressed in PCOS patients as compared with controls. The elevated expression of WNT5α increased oxidative stress and inflammation mainly through PI3K/AKT/NF-κB (nuclear factor- κB) pathway, indicating a novel mechanism of chronic inflammatory in PCOS (Zhao et al, 2015). These findings provided a new visual for the relationship between mTOR signal pathway and PCOS. Meanwhile, the expressions of this pathway related molecules are also different in different PCOS symptoms, such as inflammation and insulin resistance.
mTOR inhibitors in the treatment of PCOS
In clinical application, rapamycin and its derivatives, Everolimus, Temsirolimus and Ridaforolimus are always considered as the first line mTOR inhibitors. As mentioned above, rapamycin can decrease the level of LH and is thus used to treat the PCOS patients (Roa et al, 2009). In Swaroop’s (Swaroop et al, 2015) study, PCOS patients with LH/FSH ratio at about 3.16 can be decreased to 1.61 through furocyst(a novel fenugreek seed extract) treatment. Inducing ovulation is regarded as the most fundamental method in treating PCOS patients. And it may be indirectly realized by clomiphene citrate (CC) that is an oral estrogen receptor modular with the ability to increase FSH level. CC could restore ovulation up to 80% after first line of treatment. However, the pregnant rate was only up to 35% (Tessaro et al, 2015). Temsirolimus is a new type of oral mTOR inhibitor and can be hydrolyzed to rapamycin after oral medication. As it has a low bioavailability, Temsirolimus can also be used to release the symptom of PCOS. Collectively, these therapeutic data demonstrated that changing the secretion level of LH or FSH was prone to relieve the symptom of PCOS. Very fortunately, the past decades have witnessed the development of derivatives of rapamycin and some small molecule inhibitors, such as PKI-587 (Mallon et al, 2011), PI-103 (Hong et al, 2011), et al.
PI3K inhibitors in the treatment of PCOS
Shah et al., (2016) performed a PCOS related experiment in pre-pubertal and female rats with metfromin and quercetin treatment included. The treatment strategy was carried out orally at the dose of 150mg/kg after 6 weeks. Ovary and uterine weights were decreased dramatically compared to the PCOS control group after treatment with quercetin. Treatment with quercetin revealed dramatical improvement in luteinizing hormone, testosterone and insulin. They also indicated that quercetin could inhibit PI3K activity in a docking experiment and decrease the expression of an ovary determining gene of CYP17A1. Therefore this study suggested that quercetin might have positive effect on PCOS patients through inhibiting PI3K signaling pathway. Sivalingam VN et al., (2014) demonstrated that metformin, an efficient oral anti-hyperglycaemic medicine, can be used alone or combining with clomiphene citrate to promote ovulation in female with PCOS through interacting with PI3K/Akt/mTOR signal pathway. Babies born from woman who treated with metformin were less likely to develop insulin resistance in their later life. Zhao et al., (2015) also showed that both LY-294002 (PI3K inhibitor) and BAY-117082 could obviously inhibit WNT5α-induced inflammatory in PCOS patients.
mTOR signaling pathway in OC
OC is the most lethal gynecologic malignancies around the world, which is partially attributed to the late stage diagnosis of the disease. The five-year survival rate of patients in the world with ovarian carcinoma is approximately 45% (Koizume and Miyagi, 2015). More than 90% of OC are epithelial origin, and the rest of ovarian tumors come from granulosa-theca cells or germ cells. Among all epithelial carcinoma, approximately 5% are mucinous, 65% are serous and the rest are either clear-cell or endometrioid (Mok et al, 2007). There are several obvious types of epithelial OC, i e, high or low grade serous ovarian, clear cell, endometrioid, mucinous and undifferentiated or transitional (Rescigno et al, 2013). Additionally, high grade serous ovarian is the most common form of OC. Tumor cells metastasis will be directly related to higher mortality associated with OC (Lengyel, 2010).
Relationship between PI3K/AKT/mTOR signal pathway and OC
Intrinsic activation of PI3K/AKT/mTOR pathway has been shown to initiate OC in mice, and inhibiting mTOR pathway in those models was found to deferred tumor growth and extend survival (Kinross et al, 2012). Gao et al showed that activated PI3K/AKT signal pathway contributed to tumorigenesis, inhibition mTOR’s activity with rapamycin led to G1 cell arrest. PI3K can transmit mitogenic signal via AKT and mTOR to p70S6K1, up-regulating the expression of cyclin-dependent kinase 4(CDK4) and cell cyclin-related protein, inducing cells develop towards G1 stage (Gao et al, 2004). Montero et al (Montero et al, 2012) proved that mTORC1 had more predominant role than mTORC2 in controlling ovarian tumor cell proliferation. Knockdown the Raptor (mTORC1’s component) and Rictor (mTORC2’s component) respectively, finding that knockdown the former had more effective inhibition effect than knockdown the latter. AB073614, a new candidate long non-coding RNA (lncRNA) that can induce the development of OC, exerts its function via targeting AKT and extracellular signal-regulated kinase 1/2 (ERK1/2)-mediated signaling pathway. When knockdown AB073614 site, it will cause the inhibition of cell invasion and proliferation, leading to G1 cell phase arrest and increasing cell apoptosis (Cheng et al, 2015). Zhu et al (Zhu et al, 2015) indicated that miR-661, a tumor promoter through targeting INPP5J gene, induced AKT pathway and promoted tumor cell proliferation. Activation of PI3K/AKT/mTOR signal pathway is closely associated with higher migration and invasion ability in human OC cells. Suppressing this pathway is very useful in treating tumor (Bai et al, 2015). All the above suggests that PI3K/AKT/mTOR signaling pathway plays a significant role in OC, indicating that mTOR inhibitors maybe is a good choice for treating OC.
mTOR inhibitors in the treatment of OC
mTOR inhibitors mono-therapy
Rapamycin is the prototype of mTOR inhibitors. Both rapamycin and its derivatives could combine with mTORC1 rather than mTORC2 to inhibit mTOR activity (Yuan et al, 2009). Rapamycin derivatives have less immuno-suppressive capacity than Rapamycin, but its derivatives could exert its function in treating a lot of tumor types and have also been tested in OC. Temsirolimus and Ridaforolimus are two of clinically useful rapamycin derivatives (Cheaib et al, 2015). In a phase II clinical trial of OC, intravenous injection of temsirolimus at the dose of 25mg showed 9.3% patients response and reached 6-mouth progression-free survival rate about 24%. This result fails to meet anticipatory efficacy and is possibly due to the dosage used (Behbakht et al, 2011). In another clinical trial, patients with OC were treated weekly by temsiroliums(10mg/m2) for three weeks, the response rate was increased up to approximately 20% among five cases. Patients who exert response gained 14 months progression-free period. In other words, weekly temsiroliums exerts a potential clinical therapeutic benefit for people with OC (Takano et al, 2011).
mTOR inhibitors combination therapy
Similar to many other therapeutic protocols that have either monotherapy or polytherapy, the combination therapy of mTOR inhibitors sometimes has more significant therapeutic efficiency than mono-therapy in treating OC patients. Schlosshauer et al (Schlosshauer et al, 2009) divided OC cell lines into three groups treated with rapamycin, SH-6(AKT inhibitor) and LY-294002 respectively, finding that the inhibition capacity of rapamycin was better than SH-6 and LY-294002 in colony formation, cell growth, cell invasion and cell apoptosis. However, when cell lines were further treated by rapamycin/carboplation, there was much stronger inhibition capacity than rapamycin mono-therapy. The other phase I studies of combination therapy in patients including a fraction of patients with advanced OC using everoliums conjunction with weekly paclitaxel (Campone et al, 2009), Ridaforolimus plus paclitaxel and carboplation (the response rate is approximately 32%) (Chon et al, 2014), temsirolimus associated to paclitaxel and carboplation(the response rate is approximately 50%) (Kollmannsberger et al, 2012). Combination therapy showed higher therapeutic effects than monotherapy.
Other inhibitors of PI3K pathway in the treatment of OC PI3K inhibitors
Gao et al (Gao et al, 2004) also demonstrated that inhibition of PI3K activation through LY-294002 suppressed ovarian carcinoma cell proliferation and promoted G1 cell cycle arrest. In addition, LY-294002 in combination with metformin could enhance the inhibition capacity of cell growth and induce cell apoptosis through suppressing PI3K/AKT pathway, including down-regulating the expression of AKT, mTOR and AMPK, and up-regulating cell cycle regulatory factors, such as p21, p27 and p53 (Li et al, 2012). Jdbahi et al (Jebahi et al, 2014) showed that NVP-BEZ235, a dual PI3K/mTOR pathway inhibitor, suppressed tumor cell proliferation without inducing cell apoptosis. And BEZ235 was more effective than rapamycin in inhibiting OC cell proliferation. Biochemically, BEZ235 entirely inhibited p4E-BP1, pAkt and pS6 (Montero et al, 2012). Another research indicated that wortmannin could bind to PI3Kγ, resulting in the 833rd site of amino acid’s irreversible modification and inhibiting the activation of PI3K. Additionally, wortmannin elevated the efficiency of cisplatin-induced tumor cell’s apoptosis. Therefore, combining wortmanin with cisplatin played an important role in tumor cell apoptosis (Ohta et al, 2006). Kim et al (Kim et al, 2007) pointed out that paclitaxel could down-regulate the activation of PI3K, inducing ovarian cancer cell apoptosis. Furthermore, inhibition of AKT through PI3K inhibitors, such as LY-294002 and Wortmannin, synergistically increased the efficiency of paclitaxel-induced cell apoptosis.
AKT inhibitors
Gallic acid, a kind of polyphenol found in vegetables and fruits, can down-regulate Akt phosphorylation, and has a potential in preventing and treating OC (He et al, 2016). WYE-132, a mTORC1/2 dual inhibitor, potently suppress the proliferation of OC cells. Furthermore, WY-132 could induce caspase-dependent apoptosis in tumor. Namely, WY-132 worked by inhibiting mTORC1/2 activation and the expression of mTOR-mediate gene (Zhang et al, 2016). Cyclin-dependent kinase 5 (CDK5), a cytoplasmic serine/threonine kinase, could significantly suppress the activation of AKT after knockdown. And CDK5 silencing could enhance paclitaxel sensitivity and suppresse tumor cell growth. Additionally, CDK5 knockdown also increases G1 phage cell arrest and tumor cell apoptosis no matter where there is paclitaxel or not. CDK5 knockdown combining with paclitaxel treatment produces dual inhibition of A2780 and HEYA8, enhancing G1 cell cycle arrest and caspase-3 dependent cell apoptotic in human ovarian cancer. Consequently, CDK5 is a potential target and bio-marker for molecular therapy (Zhang et al, 2015). Myricetin and galangin which can be isolated form fruits and vegetables can inhibit the secretion of vascular endothelial growth factor (VEGF) and decrease the level of p70S6K, p-Akt and hypoxia-inducible factor-1α(HIF-1α) proteins in OVCAR-3(a kind of OC cell) and A2780 cells. Indicating that myricetin and galangin can act as potential anti-angiogenic role in treating OC (Huang et al, 2015). RY-2f, a chemical synthesized isoflavone analog, can suppress tumor cell proliferation by inhibiting PI3K/AKT/mTOR signal pathway. Besides, RY-2f can also enhance the chemotherapeutic efficacy of cisplatin to promote tumor cell apoptosis (Liu et al, 2015).
Results
The better understanding of the pathophysiology of mTOR signal pathway did helped us in improving the clinical therapy to several specific diseases, but it rises more unsolved questions to be addressed at the same time. First, it is necessary and important for researchers to explore the specific function of mTORC2 in the occurrence of diseases. This could be done by knockdown or knockout one of components in mTORC2 in a specific disease model (Montero et al, 2012). Second, much effort needs to be made to explore the substrate of mTORC1/2 that mediate some concrete functions and the possible mechanisms in pathological circumstances such as stress and low oxygen. In addition, whether there is common function between mTORC1 and mTORC2 still remains unclear. It is likely that the mTORC1/2 complex could regulate one another under the special environment. Fourthly, the different functions mediated by different substrates of mTORC1/2 in different cellular progresses need to be addressed as well.
Not only is PI3K/AKT/mTOR pathway over-activated in PCOS, but also in OC patients. But conversely, there are few studies involving the LKB1/AMPK/TSC/mTOR signal pathway when researching OC. It is necessary to investigate whether there is relationship between LKB1/AMPK/TSC/mTOR pathway and OC. LY-294002, a PI3K inhibitor, can inhibit cell proliferation, while when it combines with paclitaxel or wortmanin, the efficiency will be significantly elevated owning to synergistic effect (Kim et al, 2007). In addition, rapamycin and carboplatin (Schlosshauer et al, 2009), wortmanin and cisplatin (Ohta et al, 2006), RY-2f and cisplatin (Liu et al., 2015), all those groups would increase the efficiency in treating OC tumor cells. Therefore, it will be very valuable to explore more effective drug groups with little side work. On the other hand, the dual inhibitors also have much more powerful inhibition capacity than normal inhibitors such as NVP-BEZ235 (Jebahi et al, 2014) and WYE-132 (Zhang et al, 2016). Furthermore, dual inhibitors need less drug resources compared with drug combination therapy methods, and maybe those dual inhibitors have less side work, suggesting that much effort can be made to investigate more effective dual inhibitors.
It is well known that the ratio of LH/FSH is regarded as one of the most important characteristics of PCOS, and it has been documented that treating PCOS patients using rapamycin (Roa et al, 2009), furocyst (Swaroop et al, 2015) and clomiphene citrate (Tessaro et al, 2015) are effective. However, the concrete mechanism of those therapeutic inhibitors needs to be investigated and addressed. What’s more, more function-similar drugs need to be found to relieve this kind of symptom. Researchers always concentrate much more energy on PI3K inhibitors when it comes to mTOR pathway inhibitors based on the present study, such as LY-294002(Zhao et al, 2015), quercetion (Shah and Patel, 2016) and metformin (Sivalingam et al, 2014). Why should researchers take AKT inhibitors into account? Such as MK-2206 (Tao et al, 2016) and AZD5363 (Zhang et al, 2016) have show some potential already. Therefore, much effort needs to be made to explore other cellular inhibitors. Even though there are two mTOR signal pathways, i e, PI3K/AKT/mTOR and LKB1/AMPK/TSC/mTOR signaling pathway, the information available about the latter pathway is scarce. Therefore, it is important to study the role of LKB1/AMPK/TSC/mTOR pathway in PCOS. LY-294002 can be used to release the inflammatory reactions, while quercetion and metformin are prone to ease the insulin resistance in PCOS. What will happen when treating PCOS patients with both LY-194002 and quercetion, could this method product the double efficacy or double side effect? Consequently, it also provides us another direction to classify the symptom of PCOS for the purpose of finding out the most suitable choice for each symptom, finally, to see whether if there are the most effective combination methods in treating PCOS patients.
Discussion
In summary, the ovary-related diseases of PCOS and OC are leading healthy issues that are harmful to female both physically or mentally. An increasing number of evidence suggested the relationship between mTOR signaling pathway and PCOS or OC. Activated mTOR signaling pathway promotes cell proliferation, differentiation and up-regulating protein expression. mTOR inhibitors, such as rapamycin, can, directly or indirectly, inhibit the activation of mTOR signaling pathway. Besides, some small molecule inhibitors have better efficacy as compared with traditional inhibitors especially when those inhibitors combined with each other, which brings new hope for PCOS and OC patients.
Until now, some mechanisms of mTOR signaling pathway remain unknown and need to be discovered. But based on current studies, there are many mTOR inhibitors with high efficacies in treating PCOS and OC. Furthermore, some dual inhibitors with high efficacies have been discovered recently. There is no doubt that mTOR related studies will provides better understanding on PCOS and OC and also will lead to more efficient therapeutics to these diseases.
Conflict of interest
The authors confirm that there is no conflict of interest in this paper content.
Funding
This work was supported by grants from the National Science Foundation of China (31371277) and the Natural Science Foundation of Hunan Province(2016JJ6126).
Acknowledgements
Dr Kai Li offered me great help especially in language grammar, thanks for his support.
References
- Alam H, Maizels ET, Park Y, et al. Follicle-stimulating hormone activation of hypoxia-inducible factor-1 by the phosphatidylinositol 3-kinase/AKT/Ras homolog enriched in brain (Rheb)/mammalian target of rapamycin (mTOR) pathway is necessary for induction of select protein markers of follicular differentiation. J Biol Chem. 2004;279:19431–40. doi: 10.1074/jbc.M401235200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allemand MC, Irving BA, Asmann YW, et al. Effect of testosterone on insulin stimulated IRS1 Ser phosphorylation in primary rat myotubes--a potential model for PCOS-related insulin resistance. PLoS One. 2009;4:e4274. doi: 10.1371/journal.pone.0004274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ashworth RE, Wu J. Mammalian target of rapamycin inhibition in hepatocellular carcinoma. World J Hepatol. 2014;6:776–82. doi: 10.4254/wjh.v6.i11.776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Badura S, Tesanovic T, Pfeifer H, et al. Differential effects of selective inhibitors targeting the PI3K/AKT/mTOR pathway in acute lymphoblastic leukemia. PLoS One. 2013;8:e80070. doi: 10.1371/journal.pone.0080070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bai H, Li H, Li W, et al. The PI3K/AKT/mTOR pathway is a potential predictor of distinct invasive and migratory capacities in human ovarian cancer cell lines. Oncotarget. 2015;6:25520–32. doi: 10.18632/oncotarget.4550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behbakht K, Sill MW, Darcy KM, et al. Phase II trial of the mTOR inhibitor, temsirolimus and evaluation of circulating tumor cells and tumor biomarkers in persistent and recurrent epithelial ovarian and primary peritoneal malignancies: a Gynecologic Oncology Group study. Gynecol Oncol. 2011;123:19–26. doi: 10.1016/j.ygyno.2011.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brugarolas J, Lei K, Hurley RL, et al. Regulation of mTOR function in response to hypoxia by REDD1 and the TSC1/TSC2 tumor suppressor complex. Genes Dev. 2004;18:2893–904. doi: 10.1101/gad.1256804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campone M, Levy V, Bourbouloux E, et al. Safety and pharmacokinetics of paclitaxel and the oral mTOR inhibitor everolimus in advanced solid tumours. Br J Cancer. 2009;100:315–21. doi: 10.1038/sj.bjc.6604851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheaib B, Auguste A, Leary A. The PI3K/Akt/mTOR pathway in ovarian cancer: therapeutic opportunities and challenges. Chin J Cancer. 2015;34:4–16. doi: 10.5732/cjc.014.10289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Z, Guo J, Chen L, et al. A long noncoding RNA AB073614 promotes tumorigenesis and predicts poor prognosis in ovarian cancer. Oncotarget. 2015;6:25381–9. doi: 10.18632/oncotarget.4541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chon HJP, Apte S, Lee JH. Phase I study of combination carboplatin, paclitaxel and ridaforolimus in patients with solid, endometrial and ovarian cancers. J Clin Oncol. 2014;32:2614. [Google Scholar]
- Dibble CC, Asara JM, Manning BD. Characterization of Rictor phosphorylation sites reveals direct regulation of mTOR complex 2 by S6K1. Mol Cell Biol. 2009;29:5657–70. doi: 10.1128/MCB.00735-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Domecq JP, Prutsky G, Mullan RJ, et al. Adverse effects of the common treatments for polycystic ovary syndrome: a systematic review and meta-analysis. J Clin Endocrinol Metab. 2013;98:4646–54. doi: 10.1210/jc.2013-2374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao N, Flynn DC, Zhang Z, et al. G1 cell cycle progression and the expression of G1 cyclins are regulated by PI3K/AKT/mTOR/p70S6K1 signaling in human ovarian cancer cells. Am J Physiol Cell Physiol. 2004;287:281–91. doi: 10.1152/ajpcell.00422.2003. [DOI] [PubMed] [Google Scholar]
- Gwinn DM, Shackelford DB, Egan DF, et al. AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol Cell. 2008;30:214–26. doi: 10.1016/j.molcel.2008.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heitman J, Movva NR, Hall MN. Targets for cell cycle arrest by the immunosuppressant rapamycin in yeast. Science. 1991;253:905–9. doi: 10.1126/science.1715094. [DOI] [PubMed] [Google Scholar]
- He Z, Chen AY, Rojanasakul Y, Rankin GO, Chen YC. Gallic acid, a phenolic compound, exerts anti-angiogenic effects via the PTEN/AKT/HIF-1alpha/VEGF signaling pathway in ovarian cancer cells. Oncol Rep. 2016;35:291–7. doi: 10.3892/or.2015.4354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong Z, Xiao M, Yang Y, et al. Arsenic disulfide synergizes with the phosphoinositide 3-kinase inhibitor PI-103 to eradicate acute myeloid leukemia stem cells by inducing differentiation. Carcinogenesis. 2011;32:1550–8. doi: 10.1093/carcin/bgr176. [DOI] [PubMed] [Google Scholar]
- Hsu MI. Clinical characteristics in Taiwanese women with polycystic ovary syndrome. Clin Exp Reprod Med. 2015;42:86–93. doi: 10.5653/cerm.2015.42.3.86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang S, Houghton PJ. Targeting mTOR signaling for cancer therapy. Curr Opin Pharmacol. 2003;3:371–7. doi: 10.1016/s1471-4892(03)00071-7. [DOI] [PubMed] [Google Scholar]
- Huang H, Chen AY, Rojanasakul Y, et al. Dietary compounds galangin and myricetin suppress ovarian cancer cell angiogenesis. J Funct Foods. 2015;15:464–75. doi: 10.1016/j.jff.2015.03.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoki K, Ouyang H, Zhu T, et al. TSC2 integrates Wnt and energy signals via a coordinated phosphorylation by AMPK and GSK3 to regulate cell growth. Cell. 2006;126:955–68. doi: 10.1016/j.cell.2006.06.055. [DOI] [PubMed] [Google Scholar]
- Jebahi A, Villedieu M, Petigny-Lechartier C, et al. PI3K/mTOR dual inhibitor NVP-BEZ235 decreases Mcl-1 expression and sensitizes ovarian carcinoma cells to Bcl-xL-targeting strategies, provided that Bim expression is induced. Cancer Lett. 2014;348:38–49. doi: 10.1016/j.canlet.2014.03.001. [DOI] [PubMed] [Google Scholar]
- Johnson SC, Rabinovitch PS, Kaeberlein M. mTOR is a key modulator of ageing and age-related disease. Nature. 2013;493:338–45. doi: 10.1038/nature11861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim SH, Juhnn YS, Song YS. Akt involvement in paclitaxel chemoresistance of human ovarian cancer cells. Ann N Y Acad Sci. 2007;1095:82–9. doi: 10.1196/annals.1397.012. [DOI] [PubMed] [Google Scholar]
- Kinross KM, Montgomery KG, Kleinschmidt M, et al. An activating Pi3kca mutation coupled with Pten loss is sufficient to initiate tumorigenesis in mice. J Clin Invest. 2012;122:553–7. doi: 10.1172/JCI59309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koizume S, Miyagi Y. Tissue factor-factor VII complex as a key regulator of ovarian cancer phenotypes. Biomark Cancer. 2015;7:1–13. doi: 10.4137/BIC.S29318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kollmannsberger C, Hirte H, Siu LL, et al. Temsirolimus in combination with carboplatin and paclitaxel in patients with advanced solid tumors: a NCIC-CTG, phase I, open-label dose-escalation study (IND 179) Ann Oncol. 2012;23:238–44. doi: 10.1093/annonc/mdr063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laplante M, Sabatini DM. mTOR signaling in growth control and disease. Cell. 2012;149:274–93. doi: 10.1016/j.cell.2012.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee d Y. Roles of mTOR Signaling in Brain Development. Exp Neurobiol. 2015;24:177–85. doi: 10.5607/en.2015.24.3.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lengyel E. Ovarian cancer development and metastasis. Am J Pathol. 2010;177:1053–64. doi: 10.2353/ajpath.2010.100105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li C, Liu VW, Chan DW, Yao KM, Ngan HY. LY294002 and metformin cooperatively enhance the inhibition of growth and the induction of apoptosis of ovarian cancer cells. Int J Gynecol Cancer. 2012;22:15–22. doi: 10.1097/IGC.0b013e3182322834. [DOI] [PubMed] [Google Scholar]
- Lipton JO, Sahin M. The neurology of mTOR. Neuron. 2014;84:275–91. doi: 10.1016/j.neuron.2014.09.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu M, Qi Z, Liu B, et al. RY-2f, an isoflavone analog, overcomes cisplatin resistance to inhibit ovarian tumorigenesis via targeting the PI3K/AKT/mTOR signaling pathway. Oncotarget. 2015;6:25281–94. doi: 10.18632/oncotarget.4634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X, Cui P, Jiang HY, et al. Reversing the reduced level of endometrial GLUT4 expression in polycystic ovary syndrome: a mechanistic study of metformin action. Am J Transl Res. 2015;7:574–86. [PMC free article] [PubMed] [Google Scholar]
- Lyu D, Yu W, Tang N, et al. The mTOR signaling pathway regulates pain-related synaptic plasticity in rat entorhinal-hippocampal pathways. Mol Pain. 2013;9:64. doi: 10.1186/1744-8069-9-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mallon R, Feldberg LR, Lucas J, et al. Antitumor efficacy of PKI-587, a highly potent dual PI3K/mTOR kinase inhibitor. Clin Cancer Res. 2011;17:3193–203. doi: 10.1158/1078-0432.CCR-10-1694. [DOI] [PubMed] [Google Scholar]
- McCartney CR, Eagleson CA, Marshall JC. Regulation of gonadotropin secretion: implications for polycystic ovary syndrome. Semin Reprod Med. 2002;20:317–26. doi: 10.1055/s-2002-36706. [DOI] [PubMed] [Google Scholar]
- Mok SC, Kwong J, Welch WR, et al. Etiology and pathogenesis of epithelial ovarian cancer. Dis Markers. 2007;23:367–76. doi: 10.1155/2007/474320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montero JC, Chen X, Ocana A, Pandiella A. Predominance of mTORC1 over mTORC2 in the regulation of proliferation of ovarian cancer cells: therapeutic implications. Mol Cancer Ther. 2012;11:1342–52. doi: 10.1158/1535-7163.MCT-11-0723. [DOI] [PubMed] [Google Scholar]
- Muralidhar GG, Barbolina MV. The miR-200 family: versatile players in epithelial ovarian cancer. Int J Mol Sci. 2015;16:16833–47. doi: 10.3390/ijms160816833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohta T, Ohmichi M, Hayasaka T, et al. Inhibition of phosphatidylinositol 3-kinase increases efficacy of cisplatin in in vivo ovarian cancer models. Endocrinology. 2006;147:1761–9. doi: 10.1210/en.2005-1450. [DOI] [PubMed] [Google Scholar]
- Rescigno P, Cerillo I, Ruocco R, et al. New hypothesis on pathogenesis of ovarian cancer lead to future tailored approaches. Biomed Res Int. 2013;2013:852839. doi: 10.1155/2013/852839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roa J, Garcia-Galiano D, Varela L, et al. The mammalian target of rapamycin as novel central regulator of puberty onset via modulation of hypothalamic Kiss1 system. Endocrinology. 2009;150:5016–26. doi: 10.1210/en.2009-0096. [DOI] [PubMed] [Google Scholar]
- Sancak Y, Thoreen CC, Peterson TR, et al. PRAS40 is an insulin-regulated inhibitor of the mTORC1 protein kinase. Mol Cell. 2007;25:903–15. doi: 10.1016/j.molcel.2007.03.003. [DOI] [PubMed] [Google Scholar]
- Schlosshauer PW, Li W, Lin KT, Chan JL, Wang LH. Rapamycin by itself and additively in combination with carboplatin inhibits the growth of ovarian cancer cells. Gynecol Oncol. 2009;114:516–22. doi: 10.1016/j.ygyno.2009.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sciarretta S, Volpe M, Sadoshima J. Mammalian target of rapamycin signaling in cardiac physiology and disease. Circ Res. 2014;114:549–64. doi: 10.1161/CIRCRESAHA.114.302022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah KN, Patel SS. Phosphatidylinositide 3-kinase inhibition: A new potential target for the treatment of polycystic ovarian syndrome. Pharm Biol. 2016;54:975–83. doi: 10.3109/13880209.2015.1091482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sivalingam VN, Myers J, Nicholas S, Balen AH, Crosbie EJ. Metformin in reproductive health, pregnancy and gynaecological cancer: established and emerging indications. Hum Reprod Update. 2014;20:853–68. doi: 10.1093/humupd/dmu037. [DOI] [PubMed] [Google Scholar]
- Swaroop A, Jaipuriar AS, Gupta SK, et al. Efficacy of a novel fenugreek seed extract (Trigonella foenum-graecum, Furocyst) in polycystic ovary syndrome (PCOS) Int J Med Sci. 2015;12:825–31. doi: 10.7150/ijms.13024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takano M, Kikuchi Y, Kudoh K, et al. Weekly administration of temsirolimus for heavily pretreated patients with clear cell carcinoma of the ovary: a report of six cases. Int J Clin Oncol. 2011;16:605–9. doi: 10.1007/s10147-010-0177-z. [DOI] [PubMed] [Google Scholar]
- Tao K, Yin Y, Shen Q, et al. Akt inhibitor MK-2206 enhances the effect of cisplatin in gastric cancer cells. Biomed Rep. 2016;4:365–68. doi: 10.3892/br.2016.594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tessaro I, Modina SC, Franciosi F, et al. Effect of oral administration of low-dose follicle stimulating hormone on hyperandrogenized mice as a model of polycystic ovary syndrome. J Ovarian Res. 2015;8:64. doi: 10.1186/s13048-015-0192-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsikouras P, Spyros L, Manav B, et al. Features of polycystic Ovary Syndrome in adolescence. J Med Life. 2015;8:291–6. [PMC free article] [PubMed] [Google Scholar]
- Yaba A, Bianchi V, Borini A, Johnson J. A putative mitotic checkpoint dependent on mTOR function controls cell proliferation and survival in ovarian granulosa cells. Reprod Sci. 2008;15:128–38. doi: 10.1177/1933719107312037. [DOI] [PubMed] [Google Scholar]
- Yaba A, Demir N. The mechanism of mTOR (mammalian target of rapamycin) in a mouse model of polycystic ovary syndrome (PCOS) J Ovarian Res. 2012;5:38. doi: 10.1186/1757-2215-5-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang YM, Choi EJ. Efficacy and safety of metformin or oral contraceptives, or both in polycystic ovary syndrome. Ther Clin Risk Manag. 2015;11:1345–53. doi: 10.2147/TCRM.S89737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yap TA, Garrett MD, Walton MI, et al. Targeting the PI3K-AKT-mTOR pathway: progress, pitfalls, and promises. Curr Opin Pharmacol. 2008;8:393–412. doi: 10.1016/j.coph.2008.08.004. [DOI] [PubMed] [Google Scholar]
- Yip PY. Phosphatidylinositol 3-kinase-AKT-mammalian target of rapamycin (PI3K-Akt-mTOR) signaling pathway in non-small cell lung cancer. Transl Lung Cancer Res. 2015;4:165–76. doi: 10.3978/j.issn.2218-6751.2015.01.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan R, Kay A, Berg WJ, Lebwohl D. Targeting tumorigenesis: development and use of mTOR inhibitors in cancer therapy. J Hematol Oncol. 2009;2:45. doi: 10.1186/1756-8722-2-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang HY, Zhang YF, Han YK, et al. Activation and significance of the PI3K/Akt pathway in endometrium with polycystic ovary syndrome patients. Zhonghua Fu Chan Ke Za Zhi. 2012;47:19–23. [PubMed] [Google Scholar]
- Zhang S, Lu Z, Mao W, et al. CDK5 regulates paclitaxel sensitivity in ovarian cancer cells by modulating AKT Activation, p21Cip1- and p27Kip1-mediated G1 cell cycle arrest and apoptosis. PLoS One. 2015;10:e0131833. doi: 10.1371/journal.pone.0131833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang D, Xia H, Zhang W, Fang B. The anti-ovarian cancer activity by WYE-132, a mTORC1/2 dual inhibitor. Tumour Biol. 2016;37:1327–36. doi: 10.1007/s13277-015-3922-0. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Zheng Y, Faheem A, et al. A novel AKT inhibitor, AZD5363, inhibits phosphorylation of AKT downstream molecules, and activates phosphorylation of mTOR and SMG-1 dependent on the liver cancer cell type. Oncol Lett. 2016;11:1685–92. doi: 10.3892/ol.2016.4111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y, Zhang C, Huang Y, et al. Up-regulated expression of WNT5a increases inflammation and oxidative stress via PI3K/AKT/NF-kappaB signaling in the granulosa cells of PCOS patients. J Clin Endocrinol Meta. 2015;100:201–11. doi: 10.1210/jc.2014-2419. [DOI] [PubMed] [Google Scholar]
- Zhu T, Yuan J, Wang Y, et al. MiR-661 contributed to cell proliferation of human ovarian cancer cells by repressing INPP5J expression. Biomed Pharmacother. 2015;75:123–8. doi: 10.1016/j.biopha.2015.07.023. [DOI] [PubMed] [Google Scholar]
- Zinzalla V, Stracka D, Oppliger W, Hall MN. Activation of mTORC2 by association with the ribosome. Cell. 2011;144:757–68. doi: 10.1016/j.cell.2011.02.014. [DOI] [PubMed] [Google Scholar]


