Abstract
Defects in meiotic recombination in many organisms result in arrest because of activation of a meiotic checkpoint(s). The proximal defect that triggers this checkpoint in mammalian germ cells is not understood, but it has been suggested to involve either the presence of DNA damage in the form of unrepaired recombination intermediates or defects in homologous chromosome pairing and synapsis independent of DNA damage per se. To distinguish between these possibilities in the female germ line, we compared mouse oocyte development in a mutant that fails to form the double-strand breaks (DSBs) that initiate meiotic recombination (Spo11-/-) to mutants with defects in processing DSBs when they are formed (Dmc1-/- and Msh5-/-), and we examined the epistasis relationships between these mutations. Absence of DSB formation caused a partial defect in follicle formation, whereas defects in DSB repair caused earlier and more severe meiotic arrest, which could be suppressed by eliminating DSB formation. Therefore, our analysis reveals that there are both DNA-damage-dependent and -independent responses to recombination errors in mammalian oocytes. By using these findings as a paradigm, we also examined oocyte loss in mutants lacking the DNA-damage checkpoint kinase ATM. The absence of ATM caused defects in folliculogenesis that were similar to those in Dmc1 mutants and that could be suppressed by Spo11 mutation, implying that oocyte death in Atm-deficient animals is a response to defective DSB repair.
Keywords: Dmc1, Msh5, Spo11
Meiotic recombination promotes formation of physical links (chiasmata) between homologous chromosomes that ensure their faithful segregation at the first meiotic division. Defects in recombination can generate gametes with aneuploid or damaged genomes, so cells actively monitor recombination to coordinate its completion with progression through meiosis (1-3).
The mechanism of meiotic recombination is currently best understood in Saccharomyces cerevisiae (4). Recombination initiates with DNA double-strand breaks (DSBs) formed by the SPO11 protein. DNA strand exchange proteins (including DMC1 and RAD51) act on these DSBs to catalyze strand invasion into intact homologous duplexes, giving rise ultimately to mature recombinant products. In S. cerevisiae, dmc1 mutants arrest during prophase I (5). This arrest can be ascribed to accumulation of unrepaired recombination intermediates because arrest requires DSB formation by SPO11 (5, 6). A similar DNA-damage-dependent checkpoint appears to operate in rad51-deficient oocytes in Caenorhabditis elegans and Drosophila melanogaster (7, 8).
It is likely that DSBs initiate meiotic recombination in mammals as well. A mouse ortholog of SPO11 is required for meiotic recombination and chromosome synapsis (9, 10), and physical and/or cytological analyses provide evidence for DSBs during meiotic prophase (9-13). As in other organisms, recombination defects in mammals result in arrest and/or programmed cell death by means of apoptosis before the first meiotic division (reviewed in refs. 3, 14, and 15). In male mice, several very different molecular defects cause spermatocytes to arrest at approximately the same point in late zygotene to midpachytene. Examples include Spo11-/- (defective in the initiation of recombination), Dmc1-/- (defective in the repair of SPO11-induced DSBs), and Sycp3-/- (lacking a structural component of the synaptonemal complex) (3, 14, 15). Arrest and apoptosis in these mutants are assumed to result from activation of a checkpoint during meiotic prophase, but the trigger for this checkpoint is not known. One possible trigger is defects in homologous chromosome synapsis (e.g., refs. 9 and 16), which are similar in each of these mutants despite different underlying molecular defects. However, other alternatives cannot be ruled out, including persistent unrepaired DSBs or other chromosomal abnormalities, such as a failure in sex-body formation.
The situation in females has been less extensively characterized, partly because of the greater technical demands for studying meiosis in oocytes compared with spermatocytes. Nevertheless, it is clear that mammalian oocytes also respond to recombination defects by arresting development and undergoing programmed cell death (3). As in males, it is not known precisely what molecular defect triggers oocyte loss, although synaptic defects have been proposed in several instances (e.g., ref. 17). However, apparently unlike the situation in males, different recombination defects may effect different responses. Specifically, oocytes appear to progress further through meiosis in DSB-defective mutants such as Spo11-/- and, apparently, Mei1-/- (9, 10, 18), as compared with mutants in which DSBs are made but inefficiently repaired, such as Dmc1-/- (19, 20).
To account for these apparent differences, we proposed that there are at least the following two qualitatively different responses to recombination defects in mouse oocytes: a DNA-damage-dependent response triggered by persistent unrepaired DSBs, and a distinct response that is independent of DNA damage (9). In this article, we provide formal genetic proof for this hypothesis through characterization of oocytes from animals carrying a mutation that eliminates DSB formation (Spo11-/-) vs. mutations that affect repair of DSBs as soon as they have formed (Dmc1-/- and Msh5-/-). By using this analysis as the paradigm, we also show that the severe oocyte loss in Atm-deficient animals is a DSB-dependent response.
Materials and Methods
Mice. Spo11, Dmc1, Msh5, Atm, and Mlh1 null mice have been generated (9, 19, 21-23). All mice were of C57BL6 × 129 mixed background. To minimize variability due to strain background, experimental animals were compared with controls from the same litter (where possible) or from other litters from the same matings. For simplicity, the term “wild type” in the text and figures refers to both homozygous (+/+) and heterozygous (+/-) mice, because no differences were observed between them where analyzed. Complete data are provided in Tables 1 and 2, which are published as supporting information on the PNAS web site. Genotyping was performed by PCR analysis of tail-tip DNA (refs. 9, 19, and 24, and see Supporting Materials and Methods, which is published as supporting information on the PNAS web site).
Histological Analysis. Ovaries from up to 5-days-postpartum (dpp) mice were fixed in 4% paraformaldehyde and embedded in paraffin for c-KIT immunohistochemistry. Sections (8 μm) were processed as follows: antigen was unmasked by treating slides for 3 × 5 min in 0.01 M citrate buffer (pH 6.0, 100°C) by using an H2800 microwave processor (Energy Beam Sciences, Agawam, MA). Rabbit polyclonal anti-c-KIT antibody (Ab-1, Calbiochem) was used at 2.5 μg/ml and detected by using the Vectastain Elite ABC kit (Vector Laboratories). Ovaries from 8-dpp and older mice were fixed in Bouin's solution and embedded in paraffin, according to standard techniques. Sections (8 μm) were stained by periodic acid Schiff and hematoxylin by using routine methods.
To determine oocyte and follicle numbers, serial sections from each ovary were ordered on glass microscope slides. For mice up to 5 dpp, the number of c-KIT-positive oocytes was counted in every fifth section with random start, as described (25). The total for each ovary was then estimated by multiplying the counted number by five. Follicle counts in 4-dpp and older ovaries were determined in every other section; the reported numbers are the sums from the counted sections. For 8-dpp mice, counts were first determined individually for primordial, primary, small preantral, and antral follicles, as described (25), and then the numbers for each were summed. Only follicles containing an oocyte with a clearly visible nucleus were scored, so as not to double-count follicles. For each point in the graphs in Figs. 1, 2, 3, ovaries from one to seven females were scored (see Tables 1 and 2).
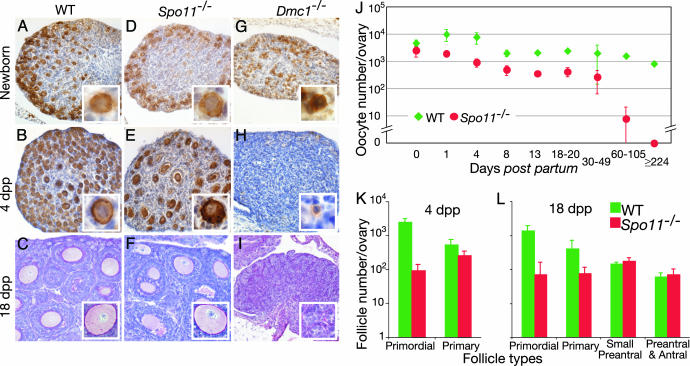
Fig. 1.
Oocyte loss in Spo11-/- and Dmc1-/- ovaries. Nearly normal numbers of diplotene oocytes and primary follicles are found in neonatal Spo11-/- ovaries but not in Dmc1-/-. Diplotene oocytes were detected by anti-c-KIT staining (brown) of newborn (A, D, and G) and 4-dpp (B, E, and H) wild-type (A and B), Spo11-/- (D and E), and Dmc1-/- (G and H) ovaries. Insets show the appearance of oocytes and primary follicles. (C, F, and I) Antral and small preantral follicles were present in normal numbers in Spo11-/- 18-dpp ovaries but absent from Dmc1-/- ovaries. Periodic acid Schiff staining of 18-dpp wild-type (C), Spo11-/- (F), and Dmc1-/- (I) ovaries. Insets highlight the appearance of oocytes, where present. (J) Total oocyte numbers in wild-type and Spo11-/- ovaries from mice of the indicated ages. (K and L) Follicle counts in ovaries of mice at 4 (K) and 18 (L) dpp, revealing depletion of primordial follicles in Spo11-/- mice.
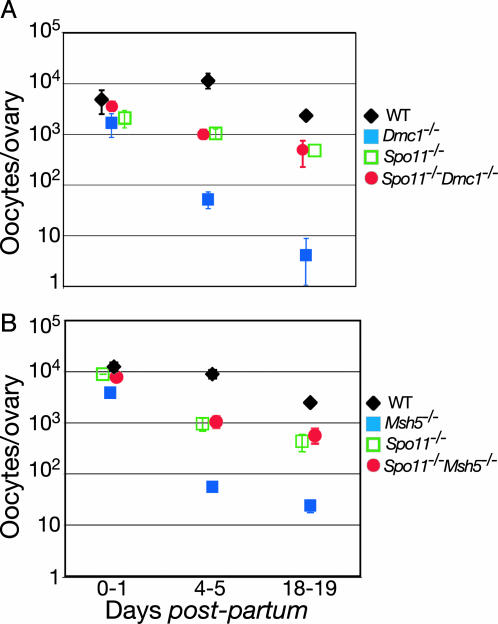
Fig. 2.
Spo11 mutation suppresses more severe oocyte loss in Dmc1-/- and Msh5-/-.(A) Total oocyte counts in the indicated Spo11 and Dmc1 single and double mutants at various ages, with age-matched wild-type controls from the same breeding. (B) Total oocyte counts in the indicated Spo11 and Msh5 single and double mutants at various ages, with age-matched wild-type controls from the same breeding.
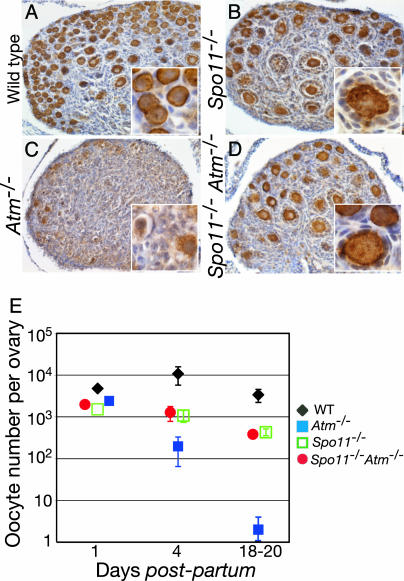
Fig. 3.
Spo11 mutation rescues oocyte numbers in Atm-/- mutants. (A-D) Anti-c-KIT-stained ovary sections from 4-dpp mice of the indicated genotypes. (E) Total oocyte counts in the indicated Spo11 and Atm single and double mutants, with age-matched wild-type controls from the same breeding.
Results
Oocyte Depletion in Spo11-/- Ovaries: A DNA-Damage-Independent Response. In females, germ cells complete the early stages of meiotic prophase I in the fetal gonad and enter a stage referred to as dictyate arrest immediately before or immediately after birth (26). Based on c-KIT expression as a marker of diplotene and dictyate stage oocytes (27), we have shown that Spo11-/- ovaries contained ≈60% of the normal number of oocytes at birth (Fig. 1 A and D) but that oocyte numbers in the mutant later became further reduced relative to controls, resulting in premature ovarian failure (9).
To determine more precisely when postnatal oocyte loss occurred, we compared ovarian development of wild-type and Spo11-/- mice. During the first 4 days after birth, oocytes in wild-type females become surrounded by a few flattened granulosa cells, forming the primordial follicles that represent the resting pool of germ cells that will be recruited for further development during the reproductive life of the animal (28). At this time, an initial synchronous wave of follicular recruitment occurs for some of the oocytes. These oocytes increase in size and the surrounding flattened granulosa cells become cuboidal and proliferate, forming the primary follicle (Fig. 1 B and Inset). This first wave of follicle growth occurred on time in Spo11-/- ovaries, as demonstrated by the formation of primary follicles (Fig. 1 E and Inset). However, a portion of the oocytes was lost during this period, such that by 4 dpp, only 16% of the normal number of c-KIT-positive oocytes was observed (Fig. 1J). Most of this decrease was in the pool of primordial follicles: Spo11-/- ovaries showed a 27-fold decrease in the number of primordial follicles, whereas the number of primary follicles was decreased by only 2-fold (Fig. 1K).
By 10–12 dpp, a cohort of secondary-stage follicles develops; oocytes in these small preantral follicles are surrounded by two or more layers of granulosa cells. Subsequently, antral-stage follicles form between 14 and 24 dpp, when a fluid-filled cavity (the antrum) develops between the layers of somatic cells (29). To study the progression in follicle development in Spo11-/- females, we examined 18-dpp ovaries because all of the different follicular development stages are present in wild-type mice at this age. In Spo11-/- ovaries, the numbers of preantral and antral follicles were similar to those in wild-type ovaries, but the primordial and primary follicle numbers were reduced ≈20- and ≈5-fold, respectively (Fig. 1 C, F, and L; see also Fig. 5 A and C, which is published as supporting information on the PNAS web site). In older animals, the total number of oocytes was reduced further in Spo11-/- ovaries (≈100-fold by 60-100 dpp; Fig. 1J), such that only a few oocytes remained. In older animals, Spo11-/- ovaries were devoid of oocytes (224 dpp; Fig. 1J)
A straightforward interpretation of these patterns is that Spo11-/- oocytes survive to the diplotene stage in numbers reduced <2-fold from normal but then have a significant defect at or before primordial follicle formation, such that only ≈15-20% of the normal number of follicles forms. Despite this reduction, initial recruitment of follicles to grow occurs at nearly normal levels, resulting in nearly complete depletion of the available oocytes within the first two to three months of age. Similar effects on the population of growing oocytes and follicles have been described in other cases in which the primordial pool size is reduced (e.g., ref. 30). Because Spo11 mutants do not form the DSBs that initiate meiotic recombination (9, 10, 13), we can conclude that oocyte depletion in Spo11-/- females is independent of DNA damage arising from unrepaired recombination intermediates.
Dmc1-/- Oocytes Are Eliminated Before Follicle Formation: A DNA-Damage-Dependent Response. Disruption of the Dmc1 gene in mice causes chromosome synapsis defects and sterility (19, 20). In Dmc1-/- females, oocytes at various stages of prophase I were present in prenatal ovaries, but ovaries of newborn and adult animals were devoid of follicles (20). It was suggested that the synaptic defects or other chromosomal aberrations are the trigger for the oocyte loss (19, 20). However, because Spo11-/- mice have a less severe ovarian phenotype despite having similar chromosome structure defects as Dmc1-/- mice, we suggested instead that Dmc1-/- oocytes are eliminated as a direct response to the presence of unrepaired DSBs (9). To distinguish between these possibilities, we compared oocyte loss in Spo11-/- and Dmc1-/- mice and tested the epistasis relationship between the two mutations.
As a first step, we defined the timing of oocyte loss in Dmc1-/- females more precisely by analyzing c-KIT expression in ovary sections from newborn and 4-dpp animals. Similar to Spo11-/- mice, Dmc1-/- newborn ovaries contained 57% of the normal number of c-KIT-positive oocytes (Figs. 1G and 2 A). However, compared with Spo11-/-, the Dmc1-/- oocytes at this age appeared to be smaller and contained condensed nuclei, suggesting that they were degenerating (Fig. 1G Inset). By 4 dpp, when essentially all of the oocytes in wild-type ovaries were enclosed in follicles (Fig. 1B), Dmc1-/- ovaries contained very few oocytes (Figs. 1H and 2 A), and the few c-KIT-positive cells that were still present did not form follicles but instead were clearly degenerating (Fig. 1H Inset). Ovaries from 18-dpp Dmc1-/- mice were nearly devoid of oocytes, and the few that were present were rarely enclosed in follicles (Figs. 1I and 2 A). Thus, c-KIT-positive oocytes are present in newborn Dmc1-/- ovaries, but unlike in Spo11-/- mice, they are essentially eliminated at or before dictyate arrest and follicle formation.
If the more severe ovarian phenotype in Dmc1-/- relative to Spo11-/- reflects a response to unrepaired DSBs, then the ability of Dmc1-/- oocytes to form follicles should be rescued by Spo11 mutation to the level found in Spo11-/- mice. To test this prediction, we crossed Spo11+/- Dmc1+/- double heterozygotes and analyzed ovaries of single- and double-mutant animals at different ages after birth. In contrast to Dmc1-/- mutants, ovaries from Spo11-/-Dmc1-/- mice from the same breeding were indistinguishable from those in Spo11-/- single mutants (Fig. 5). Double mutants had similar numbers of oocytes as Spo11-/- mice, and the kinetics of oocyte loss were similar in Spo11-/-Dmc1-/- and Spo11-/- ovaries during the examined period (Fig. 2 A). Follicles containing morphologically normal oocytes had progressed to various stages of development by 18 dpp in the double mutant, but with a deficit in the numbers of primordial follicles, as in the Spo11 single mutant (Fig. 5). These results demonstrate that Spo11 mutation is epistatic to Dmc1 mutation, and they strongly implicate persistent DNA damage as the cause of the more severe ovarian phenotype in Dmc1-/- mice.
DNA-Damage-Dependent Response in Msh5-/- Ovaries. Yeast Msh5 is a meiosis-specific MutS homolog involved in recombination, although its biochemical function is not clearly defined (31). In mice, Msh5-deficient mutants are sterile as a result of severe gametogenic failure (21, 32). Arrest during meiosis has been attributed to activation of a checkpoint triggered by defects in homologous chromosome synapsis (21, 32). However, given that only a small number of oocytes survive to form follicles (21), the Msh5-/- oocyte phenotype appears to be more similar to Dmc1-/- than to Spo11-/-. Therefore, we tested whether Msh5-/- oocyte loss reflects a DNA-damage-dependent response by characterizing oocyte development in Msh5-/- single and Spo11-/-Msh5-/- double mutants.
As in Dmc1-/- mice, oocyte numbers in Msh5-/- ovaries were greatly reduced by 4-5 dpp, to 0.6% of wild-type numbers and 5.9% of Spo11-/- numbers (Fig. 2B). However, we noted the following morphological difference from Dmc1-/- oocytes: the few oocytes remaining at 4-5 dpp in Msh5-/- ovaries were normal in appearance and had formed follicles (Fig. 6, which is published as supporting information on the PNAS web site). Similar to Dmc1-/- mice, the dramatic depletion of oocytes in Msh5-/- ovaries was suppressed by introduction of a Spo11 mutation; oocyte numbers and follicle formation in the Spo11-/- Msh5-/- double mutant were indistinguishable from the Spo11-/- mutant (Figs. 2B and 6). Thus, the severe oocyte loss in Msh5-/- ovaries appears to be driven by a response to persistent DSBs.
As a counterexample for the Spo11-Msh5 epistasis relationship, we also examined ovarian development in Msh5-/-Mlh1-/- double mutants. Mlh1 mutation causes a relatively late defect in meiotic recombination, such that chromosome synapsis occurs but crossovers fail to mature (23, 33-35). For purposes of this study, the key point is that the late recombination mutant (Mlh1-/-) is superficially similar to the early recombination mutant (Spo11-/-) in that significant numbers of oocytes form follicles in both. However, despite this similarity, if the severe oocyte loss in Msh5-/- were caused by persistent DNA damage, the late-acting Mlh1 mutation should not rescue Msh5-/- oocyte numbers. Indeed, we found that ovaries from adult Mlh1-/-Msh5-/- females were devoid of follicles, indistinguishable from Msh5-/- single-mutant littermates (Fig. 7, which is published as supporting information on the PNAS web site), further supporting the interpretation that Msh5-/- oocyte loss is due to a DNA-damage-dependent response.
Atm-/- Oocyte Loss Depends on DSB Formation. The above results provide a set of standard criteria by which to evaluate the causes of oocyte loss in mice carrying other engineered mutations. We applied this analysis to a mutation affecting the DNA-damage checkpoint protein ATM, whose loss causes defects in meiotic progression but whose molecular role in meiotic chromosome dynamics is uncertain. Atm encodes a serine/threonine kinase that activates cell-cycle checkpoints in somatic cells in response to DSBs (36). In addition to somatic phenotypes, Atm-/- mice are sterile (22, 37, 38). Mutant spermatocytes have chromosome synapsis defects and undergo apoptosis during meiotic prophase I, and ovaries in 11 dpp Atm-/- animals are devoid of oocytes (24, 38). The trigger for cell death is not known, but it is often ascribed to a checkpoint response to defects in chromosome synapsis (e.g., refs. 38-40). However, the severe oocyte-loss phenotype reported for Atm-/- females seems to be more similar to that seen in Dmc1-/- than to Spo11-/-, suggesting instead that Atm-/- oocytes are eliminated in response to the presence of persistent DSBs.
To distinguish between these possibilities, we examined the kinetics of Atm-/- oocyte depletion and the epistasis relationship of the Atm and Spo11 mutations. Ovaries from a newborn Atm-/- female contained 49% of the normal number of c-KIT-positive oocytes (Fig. 3E). However, these oocytes were rapidly lost, such that only 1.8% of the normal numbers of oocytes were present at 4 dpp (Fig. 3 C and E). The few remaining c-KIT-positive oocytes at this age usually showed condensed chromatin and a degenerative morphology (Fig. 3C Inset), similar to Dmc1-/- oocytes (Fig. 1H). In contrast, oocyte depletion and follicle formation in Spo11-/-Atm-/- double mutants was indistinguishable from those in Spo11-/- mutant littermates (Fig. 3 B, D, and E), demonstrating that Spo11 mutation is epistatic to Atm-/-. These results imply that the severe oocyte loss in Atm-/- mice is a response to unrepaired DSBs generated by SPO11.
Discussion
Meiotic cells often arrest or abort development when recombination defects are encountered, revealing the existence of cellular systems for monitoring the progress of recombination (e.g., refs. 2 and 6). These monitoring processes presumably protect against formation of gametes with aneuploid or damaged genomes. Often, different organisms (and sometimes even different sexes in the same organism) respond differently to the same molecular defects in recombination, making it difficult to extrapolate findings from one organism to another.
To understand how mammalian oocytes respond to recombination defects, we compared oocyte and follicular development in mouse lines with targeted mutations of components of the meiotic recombination pathway. We found that Spo11-/- females show a significant defect in follicle formation relative to wild type. Because Spo11-/- mice do not form the DSBs that initiate meiotic recombination (9, 10, 13), the oocyte progression defect in the Spo11-/- mutant must reflect a DNA-damage-independent response. However, Dmc1-/- and Msh5-/- oocytes show a more profound developmental defect, with an absence or near absence of follicle formation, respectively. Because these mutations presumably cause defects in the repair of meiotic DSBs, their more severe phenotype is likely to be caused by a response to persistent DNA damage. Epistasis relationships between these mutants and Spo11-/- confirm this interpretation. Thus, we can infer that distinct DNA-damage-dependent and -independent mechanisms are responsible for death of oocytes at or before follicle formation and that oocyte loss is exacerbated in the presence of unrepaired DSBs. A summary of these results is shown in Fig. 4.
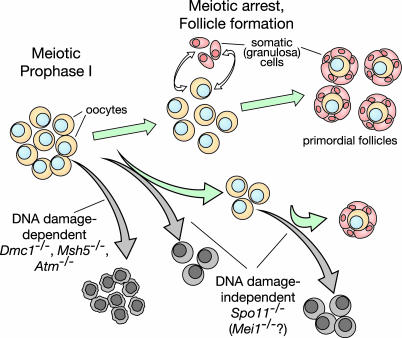
Fig. 4.
Schematic representation of DNA-damage-dependent and -independent oocyte loss. Stages of oocyte development in wild type are indicated, starting at early meiotic prophase in the fetal ovary. Meiotic arrest plus communication with somatic cells give rise to primordial follicles. Subsequent growth of follicles and oocytes is followed by resumption of meiosis, completion of the first division, and ovulation (not shown). Increased oocyte loss (gray arrows) in the Spo11 mutant is apparent during prenatal development and immediately after birth during primordial follicle formation. More severe DNA-damage-dependent oocyte loss occurs at or before follicle formation in Dmc1-, Atm-, and Msh5-deficient animals. See text for further details.
DNA-Damage-Independent Oocyte Loss in Spo11-/- Mice. Increased oocyte depletion is apparent during at least the following two stages in the Spo11-/- mutant: during prenatal development, revealed as a ≈2-fold deficit of c-KIT-positive oocytes at birth, and to a greater extent in the early postnatal period during follicle formation, revealed as a ≥20-fold deficit of primordial follicles in young females (Fig. 4). The trigger for these losses is not known, but it is clear that losses are not a response to unrepaired recombination intermediates. The failure to form chiasmata is presumably also not the proximal defect because oocyte loss at both stages occurs long before prometaphase.
Even in normal animals during prenatal development, oocyte loss occurs continuously (41). Although the causes for this normal attrition are not understood, the rate of oocyte loss is increased in mice with abnormal karyotypes (e.g., refs. 17 and 42). Such mice also display synaptic failures, so it has been suggested that defects in synapsis provoke programmed cell death in the female germ line at or before birth (17). Such a scenario might explain the prenatal oocyte depletion in Spo11-/- females, because synaptonemal-complex formation is highly aberrant in this mutant (9). However, it is true that if synaptic defects were responsible for the initial oocyte loss in Spo11-/- females, then it would be noteworthy that the extent of oocyte loss was quantitatively no greater than that detected in females with a single abnormal chromosome, even though Spo11-/- females have a global synaptic defect.
In principle, for the even stronger defect in primordial follicle formation during the early postnatal period in Spo11-/- mutants, this accelerated oocyte loss could arise from a regulatory response (checkpoint) that provides a quality-control function, perhaps in response to chromosome structure defects. However, if so, some Spo11-/- oocytes appear to be able to escape this checkpoint, even though all oocytes have abnormal chromosome structures. Alternatively, it is possible that the increased loss is not a programmed regulatory response but instead reflects physiological defects that are an indirect consequence of absence of SPO11. For example, defects in chromosome dynamics earlier in meiotic prophase might cause alterations of gene expression later, such that many oocytes are unable to provide or respond to necessary signals to/from granulosa cells. These findings do not allow us to distinguish between these possibilities.
DNA-Damage-Dependent Responses in Dmc1 and Msh5 Mutants. In yeast mutants lacking DMC1, DSBs with single-stranded tails persist and strand invasion of homologous chromosomes is defective (5). The same situation is likely to be true in the absence of mouse DMC1. Although the biochemical role of MSH5 is not clearly defined, it is known to be required for proper repair of recombination intermediates in yeast (e.g., ref. 43). The persistence of γ-H2AX staining (13) and RAD51 foci (21) in Msh5-/- spermatocytes indicates that the same requirement is true in mouse meiosis as well.
In female mice, the following more severe oocyte defects were observed in Dmc1-/- and Msh5-/- than in Spo11-/-: fewer (if any) oocytes were able to form follicles, and in newborn Dmc1-/- ovaries, most of c-KIT-positive oocytes showed clear signs of degeneration. Because Spo11 deficiency was fully epistatic to Dmc1-/- and Msh5-/- for oocyte development, these findings imply that oocytes undergo programmed cell death in response to unrepaired recombination intermediates. The phenotypic difference between Dmc1 and Msh5 mutations in females [i.e., a few morphologically normal follicles in the Msh5 mutant (ref. 21 and Fig. 6)] suggests that the DSB repair defect caused by MSH5 loss may be overcome in some oocytes, which is not possible when DMC1 is absent. A less severe phenotype for Msh5 versus Dmc1 or Rad51 mutation has also been observed in yeast and worms (31, 43-45).
A similar epistatic relationship between mutations that block DSB formation and mutations defective in DSB repair factors (e.g., DMC1, RAD51, and RAD51 paralogs) has been demonstrated in other organisms (5, 7, 8, 46, 47). However, the same relationship was not necessarily anticipated in mice, given that spermatocytes of Spo11-/- mice are eliminated with similar timing as spermatocytes in Dmc1-/- and Msh5-/- mice (i.e., early in meiotic prophase I at the late zygotene/early pachytene stage) (9, 10, 19-21, 32). Thus, compared with females, males have an earlier DNA-damage-independent response to meiotic chromosome defects. Either a DNA-damage-dependent response does not occur, or it occurs with a similar timing as the DNA-damage-independent response.
Many of the DNA-damage checkpoint-signaling pathways that function in mitotically dividing cells function also in the meiotic context in many organisms (2, 6). In mice, meiotic roles for some of these proteins are inferred from immunolocalization studies (48-51). Notably, the fact that Spo11-/- females have less severe meiotic progression defects than Dmc1-/- or Msh5-/- indicates that mammalian oocytes monitor the progression of recombination when it begins rather than simply responding to the absence of recombination per se.
DNA-Damage-Dependent Responses in Atm Mutants. Checkpoint-signaling proteins, such as ATM and its relative ATR, clearly function as mediators of cellular responses to DNA damage, triggering cell-cycle arrest and coordinating repair processes when genomic aberrations are encountered (36). However, recent evidence (52) indicates that ATM and/or ATR also promote proper execution of basic chromosomal events, in addition to responding to aberrations. Taking the properties of the Dmc1-/- and Msh5-/- mutant oocytes in this study as the paradigm, our observation that Spo11 deficiency is also epistatic to Atm deficiency indicates that oocyte death in Atm-/- animals is largely a response to unrepaired, or incorrectly repaired, DSBs. This finding seems counter to what would be expected if ATM were to function simply as a signaling protein to halt cell-cycle progression in response to unrepaired DSBs. Instead, our results provide evidence that ATM plays a more direct role in promoting meiotic recombination.
This conclusion explains the observation that chromosome-associated complexes of RAD51 and DMC1 proteins are altered in Atm-deficient mouse spermatocytes (24, 53). ATM has been proposed to promote DSB repair and homologous recombination in cultured vertebrate cells, although its role is unclear (36, 54). Moreover, yeast and Drosophila mutants that are deficient for the ATR homologs MEC1 and MEI-41, respectively, have altered patterns of meiotic recombination, consistent with direct roles in controlling the processing of meiotic DSBs for these proteins as well (55-57). Further studies to delineate the precise molecular functions of these kinases in DSB processing will be the key to understanding the meiotic defects in these mutants.
Supplementary Material
Acknowledgments
We thank Qingwen Wang (Sloan-Kettering Institute, New York) for providing some of the mice used in this study, and Katia Manova and the Molecular Cytology Core Facility for help with histology. We also thank John Schimenti (The Jackson Laboratory) and Raju Kucherlapati (Harvard University, Cambridge, MA) for providing mutant mice. This work was supported in part by the Lalor Foundation (M.D.G. and M.B.), an American-Italian Cancer Foundation Fellowship (to M.B.), and National Institutes of Health Grant HD40916 (to M.J. and S.K.).
Author contributions: M.D.G., F.B., S.K., and M.J. designed research; M.D.G., M.B., and F.B. performed research; M.D.G., F.B., S.K., and M.J. analyzed data; and M.D.G., F.B., S.K., and M.J. wrote the paper.
This paper was submitted directly (Track II) to the PNAS office.
Abbreviations: dpp, days postpartum; DSB, double-strand break.
References
- 1.Zickler, D. & Kleckner, N. (1999) Annu. Rev. Genet. 33, 603-754. [DOI] [PubMed] [Google Scholar]
- 2.Roeder, G. S. & Bailis, J. M. (2000) Trends Genet. 16, 395-403. [DOI] [PubMed] [Google Scholar]
- 3.Hunt, P. A. & Hassold, T. J. (2002) Science 296, 2181-2183. [DOI] [PubMed] [Google Scholar]
- 4.Keeney, S. (2001) Curr. Top. Dev. Biol. 52, 1-53. [DOI] [PubMed] [Google Scholar]
- 5.Bishop, D. K., Park, D., Xu, L. & Kleckner, N. (1992) Cell 69, 439-456. [DOI] [PubMed] [Google Scholar]
- 6.Lydall, D., Nikolsky, Y., Bishop, D. K. & Weinert, T. (1996) Nature 383, 840-843. [DOI] [PubMed] [Google Scholar]
- 7.Gartner, A., Milstein, S., Ahmed, S., Hodgkin, J. & Hengartner, M. O. (2000) Mol. Cell 5, 435-443. [DOI] [PubMed] [Google Scholar]
- 8.Staeva-Vieira, E., Yoo, S. & Lehmann, R. (2003) EMBO J. 22, 5863-5874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Baudat, F., Manova, K., Yuen, J. P., Jasin, M. & Keeney, S. (2000) Mol. Cell 6, 989-998. [DOI] [PubMed] [Google Scholar]
- 10.Romanienko, P. J. & Camerini-Otero, R. D. (2000) Mol. Cell 6, 975-987. [DOI] [PubMed] [Google Scholar]
- 11.Zenvirth, D., Richler, C., Bardhan, A., Baudat, F., Barzilai, A., Wahrman, J. & Simchen, G. (2003) Chromosoma 111, 369-376. [DOI] [PubMed] [Google Scholar]
- 12.Qin, J., Richardson, L. L., Jasin, M., Handel, M. A. & Arnheim, N. (2004) Mol. Cell. Biol. 24, 1655-1666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mahadevaiah, S. K., Turner, J. M. A., Baudat, F., Rogakou, E. P., de Boer, P., Blanco-Rodriguez, J., Jasin, M., Keeney, S., Bonner, W. M. & Burgoyne, P. S. (2001) Nat. Genet. 27, 271-276. [DOI] [PubMed] [Google Scholar]
- 14.Cohen, P. E. & Pollard, J. W. (2001) Bioessays 23, 996-1009. [DOI] [PubMed] [Google Scholar]
- 15.de Rooij, D. G. & de Boer, P. (2003) Cytogenet. Genome Res. 103, 267-276. [DOI] [PubMed] [Google Scholar]
- 16.Odorisio, T., Rodriguez, T. A., Evans, E. P., Clarke, A. R. & Burgoyne, P. S. (1998) Nat. Genet. 18, 257-261. [DOI] [PubMed] [Google Scholar]
- 17.Burgoyne, P. S., Mahadevaiah, S. & Mittwoch, U. (1985) J. Reprod. Fertil. 75, 647-652. [DOI] [PubMed] [Google Scholar]
- 18.Libby, B. J., Reinholdt, L. G. & Schimenti, J. C. (2003) Proc. Natl. Acad. Sci. USA 100, 15706-15711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pittman, D. L., Cobb, J., Schimenti, K. J., Wilson, L. A., Cooper, D. M., Brignull, E., Handel, M. A. & Schimenti, J. C. (1998) Mol. Cell 1, 697-705. [DOI] [PubMed] [Google Scholar]
- 20.Yoshida, K., Kondoh, G., Matsuda, Y., Habu, T., Nishimune, Y. & Morita, T. (1998) Mol. Cell 1, 707-718. [DOI] [PubMed] [Google Scholar]
- 21.Edelmann, W., Cohen, P. E., Kneitz, B., Winand, N., Lia, M., Heyer, J., Kolodner, R., Pollard, J. W. & Kucherlapati, R. (1999) Nat. Genet. 21, 123-127. [DOI] [PubMed] [Google Scholar]
- 22.Barlow, C., Hirotsune, S., Paylor, R., Liyanage, M., Eckhaus, M., Collins, F., Shiloh, Y., Crawley, J. N., Ried, T., Tagle, D. & Wynshaw-Boris, A. (1996) Cell 86, 159-171. [DOI] [PubMed] [Google Scholar]
- 23.Edelmann, W., Cohen, P. E., Kane, M., Lau, K., Morrow, B., Bennett, S., Umar, A., Kunkel, T., Cattoretti, G., Chaganti, R., et al. (1996) Cell 85, 1125-1134. [DOI] [PubMed] [Google Scholar]
- 24.Barlow, C., Liyanage, M., Moens, P. B., Tarsounas, M., Nagashima, K., Brown, K., Rottinghaus, S., Jackson, S. P., Tagle, D., Ried, T. & Wynshaw-Boris, A. (1998) Development (Cambridge, U.K.) 125, 4007-4017. [DOI] [PubMed] [Google Scholar]
- 25.Morita, Y., Perez, G. I., Maravei, D. V., Tilly, K. I. & Tilly, J. L. (1999) Mol. Endocrinol. 13, 841-850. [DOI] [PubMed] [Google Scholar]
- 26.Elvin, J. A. & Matzuk, M. M. (1998) Rev. Reprod. 3, 183-195. [DOI] [PubMed] [Google Scholar]
- 27.Manova, K., Nocka, K., Besmer, P. & Bachvarova, R. F. (1990) Development (Cambridge, U.K.) 110, 1057-1069. [DOI] [PubMed] [Google Scholar]
- 28.Peters, H. (1969) Acta Endocrinol. 62, 98-116. [DOI] [PubMed] [Google Scholar]
- 29.Matzuk, M. M. & Lamb, D. J. (2002) Nat. Cell Biol. 4, Suppl., s41-s49. [DOI] [PubMed] [Google Scholar]
- 30.Baker, T. G., Challoner, S. & Burgoyne, P. S. (1980) J. Reprod. Fertil. 60, 449-456. [DOI] [PubMed] [Google Scholar]
- 31.Hollingsworth, N. M., Ponte, L. & Halsey, C. (1995) Genes Dev. 9, 1728-1739. [DOI] [PubMed] [Google Scholar]
- 32.de Vries, S. S., Baart, E. B., Dekker, M., Siezen, A., de Rooij, D. G., de Boer, P. & te Riele, H. (1999) Genes Dev. 13, 523-531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Baker, S. M., Plug, A. W., Prolla, T. A., Bronner, C. E., Harris, A. C., Yao, X., Christie, D. M., Monell, C., Arnheim, N., Bradley, A., et al. (1996) Nat. Genet. 13, 336-342. [DOI] [PubMed] [Google Scholar]
- 34.Eaker, S., Cobb, J., Pyle, A. & Handel, M. A. (2002) Dev. Biol. 249, 85-95. [DOI] [PubMed] [Google Scholar]
- 35.Woods, L. M., Hodges, C. A., Baart, E., Baker, S. M., Liskay, M. & Hunt, P. A. (1999) J. Cell Biol. 145, 1395-1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shiloh, Y. (2003) Nat. Rev. Cancer 3, 155-168. [DOI] [PubMed] [Google Scholar]
- 37.Elson, A., Wang, Y., Daugherty, C. J., Morton, C. C., Zhou, F., Campos-Torres, J. & Leder, P. (1996) Proc. Natl. Acad. Sci. USA 93, 13084-13089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Xu, Y., Ashley, T., Brainerd, E. E., Bronson, R. T., Meyn, M. S. & Baltimore, D. (1996) Genes Dev. 10, 2411-2422. [DOI] [PubMed] [Google Scholar]
- 39.Scherthan, H., Jerratsch, M., Dhar, S., Wang, Y. A., Goff, S. P. & Pandita, T. K. (2000) Mol. Cell. Biol. 20, 7773-7783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hamer, G., Kal, H. B., Westphal, C. H., Ashley, T. & de Rooij, D. G. (2004) Biol. Reprod. 70, 1206-1212. [DOI] [PubMed] [Google Scholar]
- 41.McClellan, K. A., Gosden, R. & Taketo, T. (2003) Dev. Biol. 258, 334-348. [DOI] [PubMed] [Google Scholar]
- 42.Burgoyne, P. S. & Baker, T. G. (1981) J. Reprod. Fertil. 61, 207-212. [DOI] [PubMed] [Google Scholar]
- 43.Borner, G. V., Kleckner, N. & Hunter, N. (2004) Cell 117, 29-45. [DOI] [PubMed] [Google Scholar]
- 44.Ross-Macdonald, P. & Roeder, G. S. (1994) Cell 79, 1069-1080. [DOI] [PubMed] [Google Scholar]
- 45.Kelly, K. O., Dernburg, A. F., Stanfield, G. M. & Villeneuve, A. M. (2000) Genetics 156, 617-630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Abdu, U., Gonzalez-Reyes, A., Ghabrial, A. & Schupbach, T. (2003) Genetics 165, 197-204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ghabrial, A. & Schupbach, T. (1999) Nat. Cell Biol. 1, 354-357. [DOI] [PubMed] [Google Scholar]
- 48.Keegan, K. S., Holtzman, D. A., Plug, A. W., Christenson, E. R., Brainerd, E. E., Flaggs, G., Bentley, N. J., Taylor, E. M., Meyn, M. S., Moss, S. B., et al. (1996) Genes Dev. 10, 2423-2437. [DOI] [PubMed] [Google Scholar]
- 49.Freire, R., Murguia, J. R., Tarsounas, M., Lowndes, N. F., Moens, P. B. & Jackson, S. P. (1998) Genes Dev. 12, 2560-2573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Moens, P. B., Tarsounas, M., Morita, T., Habu, T., Rottinghaus, S. T., Freire, R., Jackson, S. P., Barlow, C. & Wynshaw-Boris, A. (1999) Chromosoma 108, 95-102. [DOI] [PubMed] [Google Scholar]
- 51.Perera, D., Perez-Hidalgo, L., Moens, P. B., Reini, K., Lakin, N., Syvaoja, J. E., San-Segundo, P. A. & Freire, R. (2004) Mol. Biol. Cell 15, 1568-1579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Cha, R. S. & Kleckner, N. (2002) Science 297, 602-606. [DOI] [PubMed] [Google Scholar]
- 53.Barlow, A. L., Benson, F. E., West, S. C. & Hulten, M. A. (1997) EMBO J. 16, 5207-5215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Foray, N., Priestley, A., Alsbeih, G., Badie, C., Capulas, E. P., Arlett, C. F. & Malaise, E. P. (1997) Int. J. Radiat. Biol. 72, 271-283. [DOI] [PubMed] [Google Scholar]
- 55.Baker, B. S. & Carpenter, A. T. (1972) Genetics 71, 255-286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kato, R. & Ogawa, H. (1994) Nucleic Acids Res. 22, 3104-3112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Grushcow, J. M., Holzen, T. M., Park, K. J., Weinert, T., Lichten, M. & Bishop, D. K. (1999) Genetics 153, 607-620. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.