Abstract
Cholesterol is known to be a major risk factor for coronary heart disease (CHD). Current treatments for elevated blood cholesterol include dietary management, regular exercise, and drug therapy with fibrates, bile acid sequestrants, and statins. Such therapies, however, are often suboptimal and carry a risk for serious side effects. This study shows that microencapsulated Lactobacillus plantarum 80 (pCBH1) cells can efficiently break down and remove bile acids, and establishes a basis for their use in lowering blood serum cholesterol. Results show that microencapsulated LP80 (pCBH1) is able to effectively break down the conjugated bile acids glycodeoxycholic acid (GDCA) and taurodeoxycholic acid (TDCA) with bile salt hydrolase (BSH) activities of 0.19 and 0.08 μmol DCA/mg CDW/h respectively. This article also summarizes the physiological interrelationship between bile acids and cholesterol and predicts the oral doses of microencapsulated Lactobacillus plantarum 80 (pCBH1) cells required for lowering cholesterol.
INTRODUCTION
Coronary heart disease (CHD) is the main cause of death in Canada, the US, and many other countries around the world [1, 2]. The World Health Organization (WHO) predicts that by the year 2020, up to 40% of all deaths will be related to cardiovascular diseases or disease of the heart [3]. Although cholesterol is an important basic building block for body tissues, elevated blood cholesterol is a well known major risk factor for CHD [4, 5]. Recent modalities for lowering blood cholesterol levels involve dietary management, behavior modification, regular exercise, and drug therapy [6, 7, 8, 9]. Pharmacologic agents such as fibrates, nicotinic acid, bile acid sequestrants, and statins are also available for the treatment of high cholesterol. Although these drugs effectively reduce cholesterol levels, they are expensive and are known to have severe side effects [10, 11, 12].
Bile salts are the water-soluble end products of cholesterol, and are synthesized in the liver. During normal enterohepatic circulation (EHC), the average bile salt pool of 4.0 g is secreted into the duodenum twice during each meal, or an average of 6–8 times per day for the purpose of forming mixed micelles with the products of lipid digestion [13, 14]. During intestinal transit, 90%–95% of secreted bile salts are absorbed in the terminal ileum and are returned to the liver via the portal vein [15]. The bile salt pool is replenished by hepatic synthesis of new bile from serum cholesterol. It has been shown that upon surgical, pharmacological, or pathological interruption of the EHC, bile salt synthesis is increased up to 15-fold, leading to an increased demand for cholesterol in the liver [13, 16].
Bile acid sequestrants (BAS) are a type of cholesterol-lowering therapy that interrupt the EHC and cause lowering of blood serum cholesterol through de novo synthesis of bile acids in the liver. BAS bind bile acids in the intestine and form insoluble complexes that are excreted in the feces. It has been shown that BAS alone can reduce cholesterol concentrations by 10% to 30% [17, 18]. However, the common BAS cholestyramine resin (Locholest, Questran), Colesevelam (WelChol), and Colestipol (Colestid) are known to exhibit major adverse effects [19].
Certain strains of bacteria act directly on bile acids in the gastrointestinal (GI) tract and are beneficial in reducing serum cholesterol levels [16, 20, 21]. Control of cholesterol through oral live bacterial cell therapy is based on the demonstration that naturally occurring bacteria such as Lactobacillus acidophilus, Lactobacillus bulgaricus, and Lactobacillus reuteri can significantly lower serum cholesterol levels due to BSH activities [16, 20, 21]. De Smet et al calculated that a daily intake of a realistic amount of highly BSH active Lactobacillus cells, in the form of yogurt, might lead to a significant reduction of cholesterol [16]. It has also been reported that the oral administration of this and other free bacteria can reduce serum cholesterol levels by 22% to 33% [16, 21]. While this was very promising, several limiting factors to the oral administration of free bacteria have been identified. For example, of those free bacteria ingested only 1% survive GI transit limiting the overall therapeutic effect [16]. Also, oral administration of live bacterial cells can cause a host immune response and can be retained in the intestine replacing the natural intestinal flora [21, 22]. Thus, concerns of safety and practicality have prevented the regular use of this promising therapy in clinical practice.
Artificial cell microencapsulation is a technique used to encapsulate biologically active materials in specialized ultra-thin semipermeable polymer membranes [23]. The polymer membrane protects encapsulated materials from harsh external environments, while at the same time allowing for the metabolism of selected solutes capable of passing into and out of the microcapsule. In this manner, the enclosed material can be retained inside and be separated from the external environment, making microencapsulation particularly useful for biomedical and clinical applications [24, 25, 26, 27, 28]. In the presentstudy we examine the potential of artificial cell microencapsulated genetically engineered Lactobacillus plantarum 80 (pCBH1) cells for bile acids deconjugation to lower cholesterol.
MATERIALS AND METHODS
Media and chemicals
The sodium salts of glycocholic acid (GCA), taurodeoxycholic (TDCA), glycodeoxycholic acid (GDCA), and deoxycholic acid (DCA) were supplied by SIGMA (St Louis, Mo). De Man-Rogosa-Sharpe (MRS) broth was obtained from Difco (Sparks, Md). The water was purified with an EASYpure Reverse Osmosis System and a NANOpure Diamond Life Science (UV/UF) ultrapure water system from Barnstead/Thermoline (Dubuque, Iowa). Methanol was HPLC-gradient from Fisher Scientific (Fair Lawn, NJ). All other chemicals were of analytical grade.
Bacterial strains and growth conditions
The bacterial strain used in this study is the bile salt hydrolytic (BSH) isogenic Lactobacillus plantarum 80 (pCBH1) strain. Overproduction of the BSH enzyme in LP80 (pCBH1) was obtained as described by Christiaens et al [29]. The BSH overproducing LP80 (pCBH1) strain carries the multicopy plasmid pCBH1 carrying the LP80 (pCBH1) chromosomal bsh gene and an erythromycin-resistance gene.
The Lactobacillus strains were grown in MRS broth at 37°C in a Sanyo MIR-162 bench top incubator. The MRS broth was supplemented with 100 μg/mL erythromycin from FisherBiotech (Fair Lawn, NJ) to select bacteria carrying the multicopy plasmid pCBH1.
Microencapsulation of Lactobacillus plantarum 80 (pCBH1)
50 ml of 1.5% low-viscosity alginate (Kelco, Chicago, Ill) solution was prepared and filtered through a 0.22 μm Sterivex-GS filter (Millipore, Bedford, Mass) into a sterile 60 mL syringe. LP80 (pCBH1) was grown at 37°C in MRS broth and prepared as a concentrated microorganism suspension by resuspension of microorganism in 10 mL of sterilized physiologic solution. The 10 mL concentrated microorganism suspension was added to the 50 ml low-viscosity alginate solution and mixed well. The alginate/microorganism mixture was immobilized through a 300 μm nozzle into a filtered solution of CaCl2 with an Inotech Encapsulator IER-20 (Inotech Biosystems International, Rockville, Md). The immobilized LP80 (pCBH1) alginate beads were washed in sterilized physiological solution (8.5 g/L NaCl), placed in a 1% solution of poly-L-lysine from Sigma (St Louis, Mo) for 10 minutes, washed in physiological solution, placed in 1% solution of low-viscosity alginate for 10 minutes, and washed in physiological solution a final time. This procedure was performed in a Microzone Biological Containment Hood (Microzone Corporation ON, Canada) to assure sterility. The microencapsulated LP80 (pCBH1) (Figure 1) was stored in 1.0 L minimal solution (10% MRS and 90% physiologic solution) at 4°C to mimic presale product storage.
Figure 1.

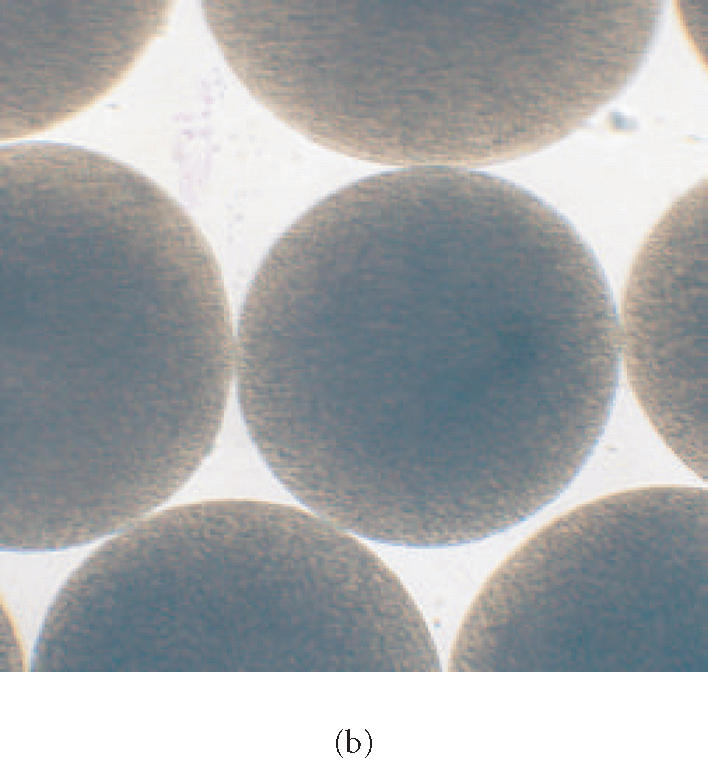
(a) Photomicrograph of Lactobacillus plantarum 80 (pCBH1) microcapsules at 77× magnification and (b) at 112× magnification.
BSH activity of Lactobacillus plantarum 80 (pCBH1) microcapsules
To investigate the BSH activity of the microencapsulated BSH overproducing LP80 (pCBH1), batch experiments were performed. Five grams of microencapsulated LP80 (pCBH1) was added to fresh MRS broth to which 10.0 mM GDCA and 5.0 mM TDCA were added. Samples were taken at regular time intervals during the 12-hours incubation to determine the bile salt concentration in the reaction vessels. The experiment was performed in triplicate. (HPLC calibration curves for GDCA and TDCA measurements were used; see Supplement 1)
Supplement 1.
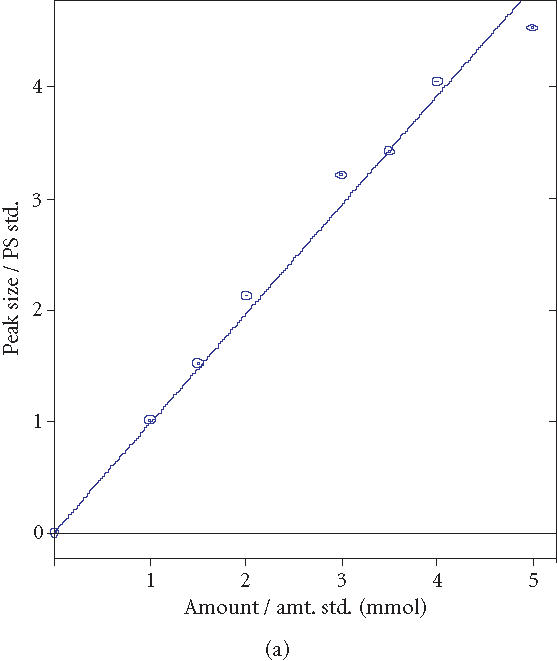
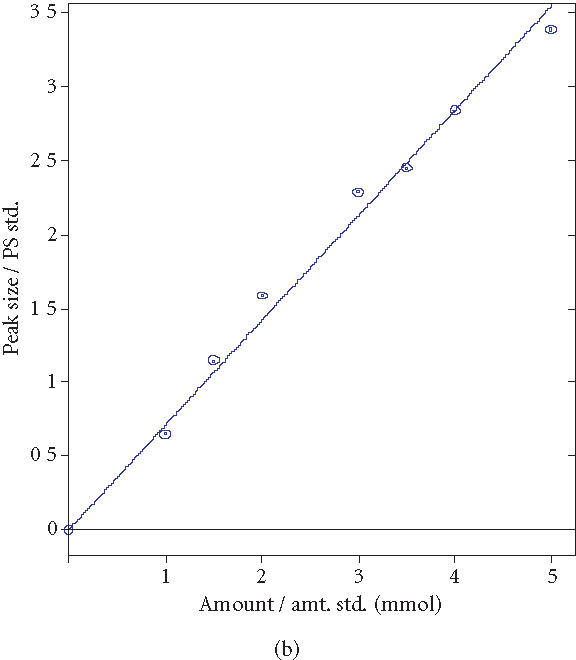
HPLC calibration curves for GDCA and TDCA measurements. (a) GDCA coeff. det. (r2): 0.987599. (b) TDCA coeff. det. (r2): 0.991610.
Bile salt hydrolase assay
A modification of the HPLC procedure described by Scalia [30] was used to determine BSH activity [31]. Analyses were performed on a reversed-phase C-18 column: LiChrosorb RP-18, 5 μm, 250 × 4.6 mm from HiChrom (Novato, Calif). The HPLC system was made up of two ProStar 210/215 solvent delivery modules, and a ProStar 320 UV/Vis Detector, and a ProStar 410 AutoSampler, and Star LC Workstation Version 6.0 software was used. The solvents used were HPLC-grade methanol (solvent A), and solvent B, which was acetate buffer prepared daily with 0.5 M sodium acetate, adjusted to pH 4.3 with o-phosphoric acid, and filtered through a 0.22-μm filter (Whatman, England). An isocratic elution of 70% solvent A and 30% solvent B was used at a flow rate of 1.0 mL/minute at room temperature. An injection loop of 20 μL was used, and the detection occurred at 205 nm within 25 minutes after injection of the bile salt extract.
Quarter-mL samples to be analyzed were acidified by the addition of 2.5 μL of 6 N HCl to stop any further enzymatic activity. A modification of the extraction procedure described by Cantafora was used [16, 31, 32]. From the 0.25-mL sample, bile salts were extracted using a solution of methanol (1:1;v:v). GCA was added as an internal standard at 4.0 mM. The samples were mixed vigorously for 10 minutes and centrifuged at 1000 g for 15 minutes. The supernatant was then filtered through a 0.22 μL syringe driven HPLC filter (Millipore, Japan) and the samples were analyzed directly after filtration.
RESULTS
Preparation of artificial cell microcapsules containing genetically engineered Lactobacillus plantarum 80 (pCBH1) cells and determination of bile acids by HPLC
Artificial cell microcapsules containing genetically engineered Lactobacillus plantarum 80 (pCBH1) cells (Figure 1) were prepared using the methods described above and were stored at 4°C for use in experiments. Sterile conditions and procedures were strictly adhered to during the process of microencapsulation. For all experiments, alginate was used as it is a commonly available immobilization product which is derived from seaweed and is known to be nontoxic [33]. Alginate provides the necessary medium for preparation of an APA membrane, which is well characterized and known for cell environmental isolation including immunoisolation [34].
Known quantities of GDCA and TDCA were added to MRS broth and 0.25-mL samples were analyzed using the modified HPLC bile salt hydrolase assay (Figure 2). Using 4.0 mM GCA internal standard, correlation of determinant factors (R 2) of 0.987599 for GDCA and 0.991610 for TDCA were obtained and used in all experiments.
Figure 2.
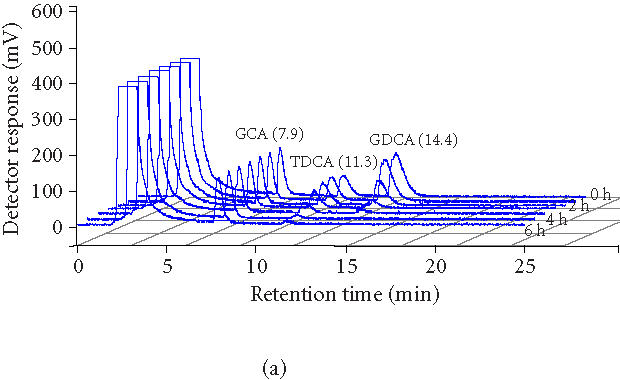
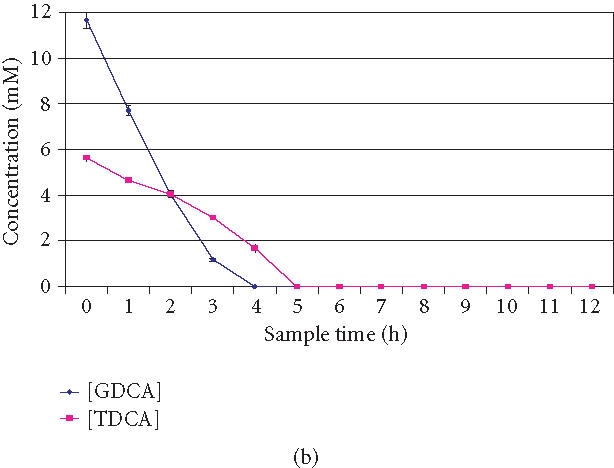
(a) Overlaid HPLC chromatograms of bile acids over time (0, 1, 2, 3, 4, 5, and 6 hours). Decreasing peak areas of TDCA and GDCA indicate BSH activity of Lactobacillus plantarum 80 (pCBH1) microcapsules. (b) BSH activity and GDCA and TDCA depleting efficiency of Lactobacillus plantarum 80 (pCBH1) microcapsules in in vitro experiment. The concentration of GDCA and TDCA bile acids are shown over time.
BSH activity of Lactobacillus plantarum 80 (pCBH1) microcapsules
The BSH activity of the microencapsulated LP80 (pCBH1) cells was determined to evaluate the potential for depleting high concentrations of bile acids. Five grams of microencapsulated LP80 (pCBH1) was incubated in MRS broth supplemented with 10.0 mM GDCA and 5.0 mM TDCA and sample was analyzed. The concentration of bile acids was monitored by analyzing media samples at regular intervals over 12 hours. Figure 2a shows superimposed HPLC chromatograms of bile acids in reaction media at 0 hour, 1 hour, 2 hours, 3 hours, 4 hours, 5 hours, and 6 hours. Decreasing peak areas of TDCA and GDCA bile acids indicate BSH activity of LP80 (pCBH1) microcapsules. The BSH activity of microencapsulated LP80 (pCBH1) towards glyco- and tauro-bile acids was also analyzed (Table 1).
Table 1.
Bile salt hydrolase (BSH) activity ( μmol DCA/mg CDW/h) of microencapsulated Lactobacillus plantarum 80 (pCBH1) towards glyco- and tauro-bile acids.
| Strain | BSH activity | ||
|---|---|---|---|
| (μmol DCA/mg CDW/h) | |||
| GDCA | TDCA | DCA | |
| Microencapsulated LP80 (pCBH1) | 0.19 | 0.08 | 0.27 |
The BSH activity of 0.26 g CDW of microencapsulated LP80 (pCBH1) was calculated based on the depletion of 0.2 mmol of GDCA in a 4-hour period, and the BSH activity towards TDCA was based on the breakdown of 0.1 mmol of TDCA in a 5-hour period. Also, this calculation was based on the in vitro depletion of bile acids with 0.26 g CDW LP80 (pCBH1) in 5.0 g alginate microcapsules in a complex mixture of the bile acids. Figure 2b shows the BSH activity of LP80 (pCBH1) microcapsules in the in vitro bile acid experiment over a 12 hours period. The concentration of GDCA and TDCA bile acids were found to decrease over time.
Figure 2b shows that the BSH activity of LP80 (pCBH1) began immediately and depleted GDCA at a greater initial rate. While TDCA also began to breakdown immediately, it did so at a slower rate than GDCA. The removal of GDCA, however, experienced concentration effects as it was depleted early and thus the breakdown of GDCA slowed as the experiment progressed and the BSH activity towards TDCA increased.
To investigate the fate of the products of deconjugation, experiment was performed using a calibration of increasing concentrations of TDCA, GDCA, and DCA. Figure 3a shows superimposed HPLC chromatograms of samples at 0 hour, 1 hour, 2 hours, 3 hours, 4 hours, 5 hours, and 6 hours. Decreasing peak areas of TDCA and GDCA bile acids indicate BSH activity of microencapsulated LP80 (pCBH1). We compared these results to earlier studies using immobilized beads containing LP80 (pCBH1) (Figure 3b). Decreasing peak areas of TDCA and GDCA bile acids indicate BSH activity of alginate beads containing immobilized LP80 (pCBH1). The peak detected just before the measured TDCA peak was diminished totally within 4 hours and corresponds to the calibration peak of DCA. The absence of a corresponding peak in the encapsulation results shows the clear advantage of using encapsulated cells.
Figure 3.
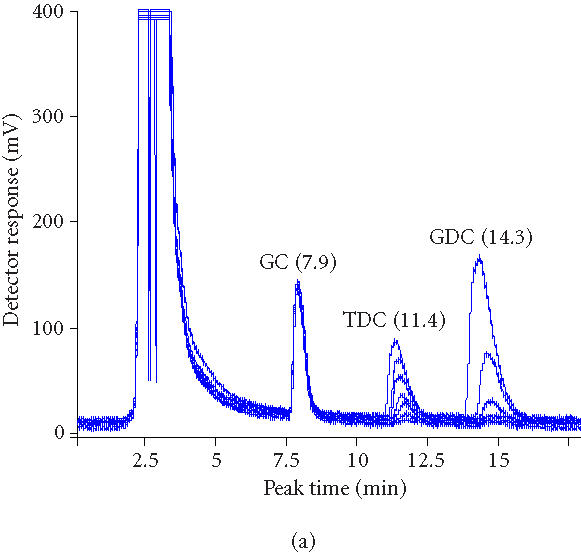
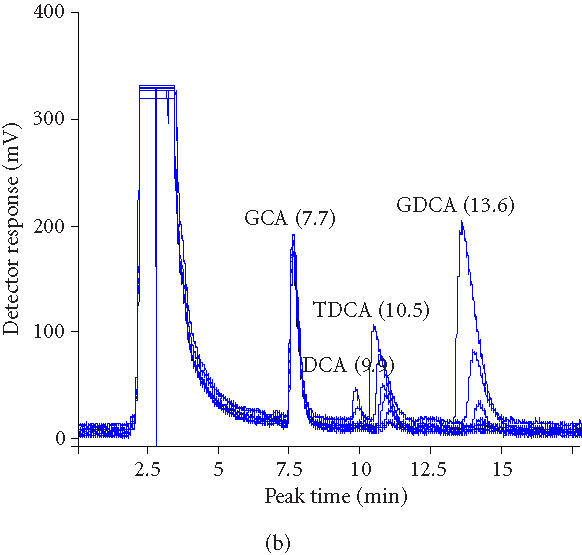
(a) Overlaid HPLC chromatograms of samples (0, 1, 2, 3, 4, 5, and 6 hours) from experiment in which microencapsulated LP80 (pCBH1) was used to deconjugate 10 mM GDCA and 5 mM TCDA. (b) Overlaid HPCL chromatograms of samples (0, 1, 2, 3, 4, 5, and 6 hours) from experiment in which immobilized LP80 (pCBH1) was used to deconjugate 10 mM GDCA and 5 mM TCDA.
DISCUSSION
Earlier studies have shown that free LP80 (pCBH1) cells can be use to break down bile acids in vitro [16]. We intended to prove the efficacy of the microencapsulated bacteria at breaking down tauro- and glyco-bile acids, and hence establish a basis for their use in lowering blood serum cholesterol when administered orally. Results obtained in this study clearly show that microencapsulated LP80 (pCBH1) was able to effectively break down physiologically relevant concentrations of bile acids in vitro. The BSH activity results show that 0.26 g CDW of microencapsulated LP80 (pCBH1) can breakdown 0.2 mmol of GDCA in a 4-hour period and 0.1 mmol of TDCA in a 5-hour period against an average hepatic bile salt secretion of 2.0 mmol/4 h [13, 35]. Thus, an oral administration of microcapsules, containing 1.85 g CDW of LP80 (pCBH1), would have the capacity to completely breakdown the total bile salt secretion in the average human during a 4-hour intestinal transit period. However, breaking down all secreted bile salts may not be the goal of therapy with microencapsulated LP80 (pCBH1), as some bile acids are required for intestinal absorption of fat through formation of mixed micelles. Even so, enzymatic breakdown of the bile salt pool in this way would have an impact on serum cholesterol due to the deconjugation of tauro- and glyco-bile acids and their resulting de-novo synthesis, from serum cholesterol, in the liver (Figure 4a).
Figure 4.
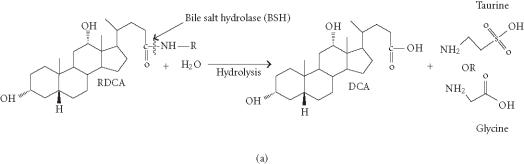
(a) Hydrolysis of conjugated bile salts by the bile salt hydrolase (BSH) enzyme overproduced by genetically engineered Lactobacillus plantarum 80 (pCBH1) [36]. R indicates the amino acid glycine or taurine. RDCA: glyco- or tauro-deoxycholic acid, DCA: deoxycholic acid. (b) Enterohepatic circulation of bile (EHC) [13, 14, 15, 35, 37].
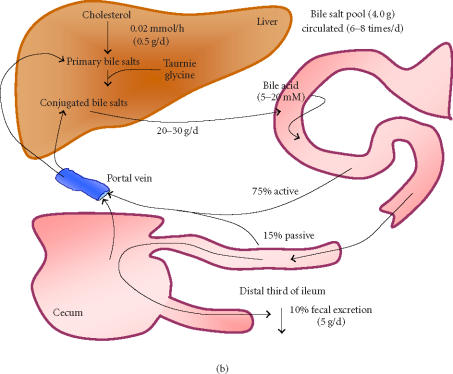
In the average human, conjugated bile salts are produced from cholesterol and taruine or glycine at a rate of 0.02 mmol/h in the liver [37]. During normal EHC, the average bile salt pool of 4.0 g is secreted into the duodenum twice during each meal, or an average of 6–8 times per day [13, 14] for lipid digestion (Figure 4b). Daily bile acid secretions approach 20–30 g [13, 14] or 20–60 mmol [38] sustaining an intestinal bile salt concentration of 5–20 mM [15]. During intestinal transit, 90%–95% of secreted bile salts are reabsorbed in the terminal ileum and are returned to the liver via the portal vein [15] (Figure 4b). About 75% of the bile acid secretion is reabsorbed in the conjugated form in an active sodium-dependent way [15]. The remaining 25% is hydrolyzed during intestinal transit and only 15% is reabsorbed in a passive way [15] (Figure 4b). Thus, about 60% of deconjugated bile acids are reabsorbed. Thus, one mmol of deconjugated bile salt, formed through breakdown by microencapsulated LP80 (pCBH1), is responsible for a 0.4-mmol faecal excretion [13]. It is consequently required for this amount to be replaced by newly synthesized bile salts from blood serum cholesterol. In this way, interruption of the EHC can result in an increased bile acid biosynthesis of up to 15-fold, from 0.02 mmol/h to 0.3 mmol/h [37]. One may calculate the effects that different dosages of microencapsulated LP80 (pCBH1) can have on blood serum cholesterol levels through interruption of the EHC in this way (Figure 5).
Figure 5.
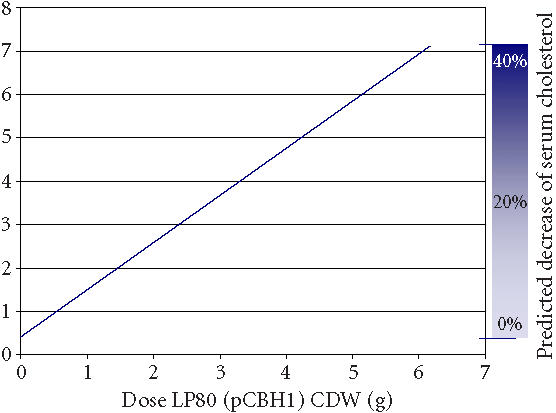
Predicted removal of cholesterol from blood serum in humans. The calculation is based on dose data from in vitro experimental results and physiologic data from the literature. The calculation assumes an average bile salt synthesis of 0.02 mmol/h, a daily bile salt secretion of 40 mmol (normal range: 20–60 mmol) [38], and that with interruption of the EHC bile acid synthesis increases up to 15-fold [15, 37].
Microencapsulated LP80 (pCBH1) was able to deconjugate GDCA and TDCA completely within 4 hours and 5 hours respectively (Figure 3a). Earlier, immobilized LP80 (pCBH1) was able to effectively break down GDCA and TDCA bile acids within 5 hours and 6 hours respectively (Figure 3b). However, the deconjugation product, deoxycholic acid (DCA), was detected (Figure 3b). This suggests that unlike immobilized cells, microencapsulated cells diminish the bioavailability of BSH-deconjugated bile acids totally (Figure 4). This finding may improve the therapeutic properties of microencapsulated LP80 (pCBH1) in several ways. For example, it addresses concerns over the production of large amounts of deconjugated bile salts and their association with an increased risk of developing colon cancer. Also, if bile salts are actually being deconjugated, precipitated, and then bound within the microcapsule, microencapsulated LP80 (pCBH1) may be capable of removing all bile acid from the GI lumen. This effect contrasts previous results, using free bacteria, where the authors predicted only an improved clearance (from 95% for conjugated to 60% for deconjugated) of bile acids from the EHC and not total clearance [16]. Further, elevated intraluminal concentrations of deconjugated bile acids in the colon, normally resulting in an increased secretion of electrolytes and water and causing diarrhea [37], would cease to present difficulty, as the deconjugated bile acids would be entirely precipitated and bound within the microcapsules and excreted in the stool.
It is now well known that statins increase the risk of myopathy in patients receiving large dosages and in patients with renal or hepatic impairment, serious infections, hypothyroidism, or advanced age [39]. In such patients, and in patients with an inadequate LDL lowering response to statins, it is widely accepted that combination therapy with a bile acid sequestrant or niacin should be considered [39, 40, 41, 42]. We submit that microencapsulated LP80 (pCBH1) may also prove to be an excellent choice of cholesterol-lowering agents for use in combination therapy with statins and other lipid-lowering therapies.
While this in vitro study shows the potential for cholesterol therapy with LP80 (pCBH1), there are number of additional factors that must be taken into consideration in proceeding with in-vivo experimentation. Important considerations may include dosage, frequency and timing of therapeutic administration, composition of microcapsule membrane, and potential side effects of by-products. There is reason, however, to believe that the deconjugate by-products are made less bioavailable, as shown by the results in Figure 3. While these factors present challenges in implementing this approach, we believe the potential risks to be very low.
This study has shown that microcapsules containing LP80 (pCBH1) have several advantages to the free or immobilized bacteria. Microencapsulation renders the potentially harmful products of BSH deconjugation, namely DCA, less bioavailable while at the same time avoid the problems with oral administration of free bacterial cells. However, further research is required to substantiate these results, in particular in-vivo affirmation of the cholesterol-lowering capacity of LP80 (pCBH1) is required before complete potential of this research can be comprehended.
ACKNOWLEDGMENT
We gratefully acknowledge the financial support from the Natural Sciences and Engineering Research Council (NSERC) of Canada.
References
- 1.American Heart Association. Heart Disease and Stroke Statistics-2002 Update. Dallas, Tex: American Heart Association; 2002. [Google Scholar]
- 2.Heart and Stroke Foundation of Canada. The Changing Face of Heart Disease and Stroke in Canada 2000. Canada: Heart and stroke foundation of Canada; 1999. [Google Scholar]
- 3.Aboderin I, Kalache A, Ben-Shlomo Y, et al. Geneva: World Health Organisation; 2002. Life Course Perspectives on Coronary Heart Disease, Stroke and Diabetes: Key Issues and Implications for Policy and Research. [Google Scholar]
- 4.Ridker P.M. Evaluating novel cardiovascular risk factors: can we better predict heart attacks? Ann Intern Med. 1999;130(11):933–937. doi: 10.7326/0003-4819-130-11-199906010-00018. [DOI] [PubMed] [Google Scholar]
- 5.Tall A.R. Plasma high density lipoproteins. Metabolism and relationship to atherogenesis. J Clin Invest. 1990;86(2):379–384. doi: 10.1172/JCI114722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dunn-Emke S, Weidner G, Ornish D. Benefits of a low-fat plant-based diet. Obes Res. 2001;9(11):731. doi: 10.1038/oby.2001.100. [DOI] [PubMed] [Google Scholar]
- 7.Lichtenstein A.H. Effects of diet and exercise on cholesterol levels. N Engl J Med. 1998;339(21):1552–1553. [PubMed] [Google Scholar]
- 8.Ornish D, Denke M. Dietary treatment of hyperlipidemia. J Cardiovasc Risk. 1994;1(4):283–286. doi: 10.1097/00043798-199412000-00001. [DOI] [PubMed] [Google Scholar]
- 9.Ornish D, Scherwitz L.W, Billings J.H, et al. Intensive lifestyle changes for reversal of coronary heart disease. JAMA. 1998;280(23):2001–2007. doi: 10.1001/jama.280.23.2001. [DOI] [PubMed] [Google Scholar]
- 10.Bliznakov E.G. Lipid-lowering drugs (statins), cholesterol, and coenzyme Q10. The baycol case—a modern Pandora’s box. Biomed Pharmacother. 2002;56(1):56–59. doi: 10.1016/s0753-3322(01)00150-0. [DOI] [PubMed] [Google Scholar]
- 11.Kolata G, Andrews E.L. Anticholesterol drug pulled after link to 31 deaths. New York Times Online. September 9, 2001 [Google Scholar]
- 12.Willman D. A gigantic headache for Bayer. New York Times Online. September 14, 2001 [Google Scholar]
- 13.Hofmann A.F. Current concepts of biliary secretion. Dig Dis Sci. 1989;34(suppl 12):16S–20S. doi: 10.1007/BF01536657. [DOI] [PubMed] [Google Scholar]
- 14.Binder H.J. Disorders of absorption. In: Braunwald E, Fauci A.S, Kasper D.L, Hauser S.L, Longo D.L, Jameson J.L, editors. Principles of Internal Medicine. 15th ed. McGraw-Hill; 2001. pp. 1665–1678. [Google Scholar]
- 15.Hofmann A.F. Handbook of Physiology. The Gastrointestinal System. Vol. IV. Bethesda, Md: American Physiological Society; 1989. Enterohepatic circulation of bile acids; pp. 567–596. [Google Scholar]
- 16.De Smet I, Van Hoorde L, De Saeyer M, Vande W.M, Verstraete W. In vitro study of bile salt hydrolase (bsh) activity of bsh isogenic Lactobacillus plantarum 80 strains and estimation of cholesterol lowering through enhanced bsh activity. Microbial Ecology in Health and Disease. 1994;7:315–329. [Google Scholar]
- 17.The Lipid Research Clinics Coronary Primary Prevention Trial Results. I. Reduction in Incidence of Coronary Heart Disease. JAMA. 1984;251(3):351–364. doi: 10.1001/jama.1984.03340270029025. [DOI] [PubMed] [Google Scholar]
- 18.Levine G.N, Keaney J.F. Jr, Vita J.A. Cholesterol reduction in cardiovascular disease. Clinical benefits and possible mechanisms. N Engl J Med. 1995;332(8):512–521. doi: 10.1056/NEJM199502233320807. [DOI] [PubMed] [Google Scholar]
- 19.Bristol-Myers Squibb . 1995 In Review: Worldwide Consumer Medicines. Company Annual Tech. Rep. http://www.bms.com/static/annual/95annu/data/cp95me.html. 1995. [Google Scholar]
- 20.Anderson J.W, Gilliland S.E. Effect of fermented milk (yogurt) containing Lactobacillus acidophilus L1 on serum cholesterol in hypercholesterolemic humans. J Am Coll Nutr. 1999;18(1):43–50. doi: 10.1080/07315724.1999.10718826. [DOI] [PubMed] [Google Scholar]
- 21.Taranto M.P, Medici M, Perdigon G, Ruiz Holgado A.P, Valdez G.F. Effect of Lactobacillus reuteri on the prevention of hypercholesterolemia in mice. J Dairy Sci. 2000;83(3):401–403. doi: 10.3168/jds.S0022-0302(00)74895-8. [DOI] [PubMed] [Google Scholar]
- 22.Chin J, Turner B, Barchia I, Mullbacher A. Immune response to orally consumed antigens and probiotic bacteria. Immunol Cell Biol. 2000;78(1):55–66. doi: 10.1046/j.1440-1711.2000.00883.x. [DOI] [PubMed] [Google Scholar]
- 23.Chang T.M, Prakash S. Artificial cells for bioencapsulation of cells and genetically engineered E coli. For cell therapy, gene therapy, and removal of urea and ammonia. Methods Mol Biol. 1997;63:343–358. doi: 10.1385/0-89603-481-X:343. [DOI] [PubMed] [Google Scholar]
- 24.Prakash S, Chang T.M. Microencapsulated genetically engineered live E coli DH5 cells administered orally to maintain normal plasma urea level in uremic rats. Nat Med. 1996;2(8):883–887. doi: 10.1038/nm0896-883. [DOI] [PubMed] [Google Scholar]
- 25.Prakash S, Chang T.M. Microencapsulated genetically engineered E coli DH5 cells for plasma urea and ammonia removal based on: 1. column bioreactor and 2. oral administration in uremic rats. Artif Cells Blood Substit Immobil Biotechnol. 1996;24(3):201–218. doi: 10.3109/10731199609117436. [DOI] [PubMed] [Google Scholar]
- 26.Prakash S, Chang T.M. Growth kinetics of genetically engineered E coli DH5 cells in artificial cell APA membrane microcapsules: preliminary report. Artif Cells Blood Substit Immobil Biotechnol. 1999;27(3):291–301. doi: 10.3109/10731199909117701. [DOI] [PubMed] [Google Scholar]
- 27.Prakash S, Chang T.M. Artificial cell microcapsules containing genetically engineered E coli DH5 cells for in-vitro lowering of plasma potassium, phosphate, magnesium, sodium, chloride, uric acid, cholesterol, and creatinine: a preliminary report. Artif Cells Blood Substit Immobil Biotechnol. 1999;27(5–6):475–481. doi: 10.3109/10731199909117722. [DOI] [PubMed] [Google Scholar]
- 28.Prakash S, Chang T.M. In vitro and in vivo uric acid lowering by artificial cells containing microencapsulated genetically engineered E coli DH5 cells. Int J Artif Organs. 2000;23:429–435. [PubMed] [Google Scholar]
- 29.Christiaens H, Leer R.J, Pouwels P.H, Verstraete W. Cloning and expression of a conjugated bile acid hydrolase gene from Lactobacillus plantarum by using a direct plate assay. Appl Environ Microbiol. 1992;58(12):3792–3798. doi: 10.1128/aem.58.12.3792-3798.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Scalia S. Simultaneous determination of free and conjugated bile acids in human gastric juice by high-performance liquid chromatography. J Chromatogr. 1988;431(2):259–269. doi: 10.1016/s0378-4347(00)83095-9. [DOI] [PubMed] [Google Scholar]
- 31.Jones M.L, Hongmei C, Ouyang W, Metz T, Prakash S. Method for bile acid determination by high performance liquid chromatography. Journal of Medical Sciences. 2003;23:277–280. [Google Scholar]
- 32.Cantafora A, Di Biase A, Alvaro D, Angelico M. Improved method for measuring the glycine and taurine conjugates of bile salts by high-performance liquid chromatography with tauro-7 alpha,12 alpha-dihydroxy-5 beta-cholanic acid as internal standard. J Chromatogr. 1987;386:367–370. doi: 10.1016/s0021-9673(01)94614-x. [DOI] [PubMed] [Google Scholar]
- 33.Prokop A, Hunkeler D, DiMari S, Haralson A, Wang T.G. In Advances in Polymer Science. Vol. 136. Berlin: Springer-Verlag; 1989. Water soluble polymers for immunoisolation I: complex coacervation and cytotoxicity; pp. 1–51. [Google Scholar]
- 34.Li R.H. Materials for immunoisolated cell transplantation. Adv Drug Deliv Rev. 1998;33(1–2):87–109. doi: 10.1016/s0169-409x(98)00022-2. [DOI] [PubMed] [Google Scholar]
- 35.McGilvery R.W. Biochemistry. A Functional Approach. Tokio: W.B. Saunders Company; 1983. pp. 555–571. [Google Scholar]
- 36.De Boever P, Verstraete W. Bile salt deconjugation by Lactobacillus plantarum 80 and its implication for bacterial toxicity. J Appl Microbiol. 1999;87(3):345–352. doi: 10.1046/j.1365-2672.1999.00019.x. [DOI] [PubMed] [Google Scholar]
- 37.Hofmann A.F. Bile acids: the good, the bad, and the ugly. News Physiol Sci. 1999;14:24–29. doi: 10.1152/physiologyonline.1999.14.1.24. [DOI] [PubMed] [Google Scholar]
- 38.Bongaerts G.P, Severijnen R.S, Tangerman A, Verrips A, Tolboom J.J. Bile acid deconjugation by lactobacilli and its effects in patients with a short small bowel. J Gastroenterol. 2000;35(11):801–804. doi: 10.1007/s005350070016. [DOI] [PubMed] [Google Scholar]
- 39.AP Association . New Product Bulletin: Advicor (niacin extended-release and lovostatin). 2002 [Google Scholar]
- 40.Brown B.G, Zhao X.Q, Chait A, et al. Simvastatin and niacin, antioxidant vitamins, or the combination for the prevention of coronary disease. N Engl J Med. 2001;345(22):1583–1592. doi: 10.1056/NEJMoa011090. [DOI] [PubMed] [Google Scholar]
- 41.Gupta E.K, Ito M.K. Lovastatin and extended-release niacin combination product: the first drug combination for the management of hyperlipidemia. Heart Dis. 2002;4(2):124–137. doi: 10.1097/00132580-200203000-00010. [DOI] [PubMed] [Google Scholar]
- 42.Kashyap M.L, McGovern M.E, Berra K, et al. Long-term safety and efficacy of a once-daily niacin/lovastatin formulation for patients with dyslipidemia. Am J Cardiol. 2002;89(6):672–678. doi: 10.1016/s0002-9149(01)02338-4. [DOI] [PubMed] [Google Scholar]