Abstract
Pur-alpha is a highly conserved sequence specific DNA and RNA binding protein with established roles in DNA replication, RNA translation, cell cycle regulation, and maintenance of neuronal differentiation. Prior studies have shown that mice lacking Pur-alpha (−/−) display decreased neurogenesis and impaired neuronal differentiation. We sought to examine for the first time, the behavioral phenotype and brain histopathology of mice that are heterozygous (+/−) for Pur-alpha. Standardized behavioral phenotyping revealed a decreased escape response to touch, limb and abdominal hypotonia, and gait abnormalities in heterozygous Pur-alpha (+/−) mice, compared to wild-type (+/+) littermates. Footprint pattern analyses showed wider-based steps, increased missteps and more outwardly rotated hindpaws in heterozygous Pur-alpha (+/−) mice, suggestive of cerebellar pathology. Barnes maze and novel object location testing revealed significant memory deficits in heterozygous Pur-alpha mice, suggestive of hippocampal pathology. Quantitative immunohistochemical assays of the vermal region of the cerebellum and CA1-3 regions of the hippocampus revealed reduced numbers of neurons in general, as well as reduced numbers of Pur-alpha+-immunopositive neurons and dendrites in heterozygous Pur-alpha mice, compared to wild type littermates. Past studies have implicated mutations in Pur-alpha in several diseases of brain development and neurodegeneration. When combined with these new findings, the Pur-alpha heterozygous knockout mice may provide an animal model in which to study mechanisms of and treatments for Pur-alpha related cognitive deficiencies and neuropathology.
Keywords: Animals, Mice, Knockout, SHIRPA, Brain pathology, PURA, Puralpha
Introduction
The Pur-alpha protein is required for neuronal development and differentiation. Pur-alpha binds to single stranded DNA and RNA in a sequence-specific manner (binding to GGN repeats within GC rich regions) and is a member of a protein family strongly conserved from bacteria through humans (Gallia et al., 2000). It is a multifunctional protein essential for DNA replication and gene transcription (see (Gallia et al., 2000, Johnson et al., 2006, Johnson et al., 2013)) and is a critical component in the compartmentalized translation of RNA ((Ohashi et al., 2002, Johnson et al., 2006). Pur-alpha is a widely expressed ubiquitous protein that exhibits both developmental and tissue-specific regulation (Khalili et al., 2003, Hokkanen et al., 2012, Mishra et al., 2013). For example, while Pur-alpha protein levels are quite low in brain during embryogenesis, its levels quickly rise after the first postnatal week in the mouse and reach their peak levels at 18 days postnatal. Pur-alpha is then maintained at relatively high levels in the adult brain (Khalili et al., 2003). In addition to its nuclear function, Pur-alpha is detected in the cytoplasm of neurons, particularly in dendrites at branch points. Several studies have detected Pur-alpha in complex with polyribosomes and heterogeneous nuclear ribonucleoproteins (hnRNP) proteins (Kanai et al., 2004).
Pur-alpha protein plays an important role in genome maintenance and has specialized functions for regulating gene expression in specific organs. In the CNS, its role in mRNA transport and translation have led to the elucidation that Pur-alpha is a major contributor in this regard, particularly in neurons (Li et al., 2001, Ohashi et al., 2002, Johnson, 2003, Kanai et al., 2004, Johnson et al., 2006, Jin et al., 2007). Mutations of Pur-alpha may be involved in several diseases of brain development and neurodegeneration. For example, Fragile X Tremor Ataxia Syndrome (FXTAS), a neurodegenerative syndrome with progressive intention tremor, gait ataxia and cognitive decline, is characterized by ubiquitin-positive intranuclear inclusions throughout the brain in which Pur-alpha is present, as well as a marked dropout of cerebellar Purkinje cells (Hagerman et al., 2005, Jin et al., 2007). A trinucleotide CGG expansion repeat located in the 5′-untranslated region (UTR) of the Fragile X Mental Retardation gene (FMR1), leading to chromosomal instability and loss of expression of the encoded FMR protein (FMRP), is the underlying cause of the Fragile X Syndrome (Darnell et al., 2001, Darnell et al., 2009). FMRP regulates mRNA transport in neurons and has been found in complex with mRNAs together with Pur-alpha and non-coding RNA molecules in dendrites thus suggesting their role in regulating RNA transport (Kobayashi et al., 2000, Kanai et al., 2004, Johnson et al., 2006). Next a role for Pur-alpha has been demonstrated in the regulation of amyloid precursor protein (APP), which is located on chromosome 21 and implicated in the pathogenesis of Down’s syndrome at both transcriptional and post-transcriptional levels (Darbinian et al., 2008, Wiseman et al., 2015). Recent studies have also identified de novo mutations in the PURA gene, which encodes Pur-alpha, that are associated with neonatal hypotonia, encephalopathy with or without seizures, neurodevelopmental delays, learning disabilities, and poor coordination or ataxias in several young human subjects (Hunt et al., 2014, Lalani et al., 2014, Tanaka et al., 2015). Dysregulation of Pur-alpha has also been shown in C9Orf72-related Frontotemporal lobe dementia in association with Amyotrophic Lateral Sclerosis, presumably resulting from accumulation of intracellular inclusion bodies that contain Pur-alpha and RNA complexes, and RNA toxic gain-in-function effects (Daigle et al., 2016). Pur-alpha may even be involved in enhancing HIV-1 transcription, with increasing concentrations of Pur-alpha leading to increased transcription of HIV-1 (Chepenik et al., 1998).
Our lab has developed knockout mice that are either homozygous (−/−) or heterozygous for Pur-alpha (+/−). Pur-alpha deficient (−/−) mice develop in our lab show severe neurological problems, such as severe tremor and spontaneous seizures, and expire by 4 weeks of age (Khalili et al., 2003). Heterozygous mice lacking one allele for the Pur-alpha gene (+/−) produce less Pur-alpha protein in whole-cell brain extracts and live to adulthood, although they display developmental delays and undergo occasional spontaneous seizures during routine handling (Khalili et al., 2003, Mishra et al., 2013). These latter results combined with those described above in association with mutations or dysregulation in Pur-alpha suggest that chronic reduction of Pur-alpha leads to neuropathogenic effects. However, the overall behavioral phenotype and brain histopathology have yet to be examined in Pur-alpha (+/−) mice.
Thus, our goals here were to examine for the first time, whether Pur-alpha (+/−) mice display altered sensorimotor reflexes, locomotive or cognitive dysfunction, and brain neuropathology. Specifically, we examined the hippocampus, cerebellum, amygdala, and prefrontal, frontal and parietal cortices for morphological and neurohistological changes following a comprehensive neurological screen, gait analysis and cognitive memory assays. We hypothesized that mice heterozygous for the Pur-alpha knockout allele would show memory deficits concomitant with neurodegeneration in the hippocampus, as well as locomotor ataxias and other gait abnormalities concomitant with neurodegeneration in the cerebellum.
Methods
Animals
The Temple University Institutional Animal Care and Use Committee approved all experiments, and in accordance with the guidelines laid down by the National Institute of Health. Mice that were heterozygous for Pur-alpha (+/−) were bred at Temple University by crossing C57/BL6 females with heterozygous Pur-alpha (+/−) knockout males. Homozygous deletion of Pur-alpha is lethal during late development; therefore mice are maintained as heterozygous (Khalili et al., 2003). Twenty-four wild-type C57/BL6 mice (+/+; 15 females and 9 males) and 24 heterozygous Pur-alpha knock-out (+/−; 12 females and 12 males) littermates underwent behavioral testing from 7 weeks to 11 months of age. Progeny were screened by PCR analysis using primer sets to distinguish, based on size of the amplicon, between the wild-type gene and knockout which contains a neomycin cassette inserted into the Pur-alpha gene (Khalili et al., 2003). After weaning at day 21, mouse pups were handled for two weeks before onset of experiments. Mice were group-housed with same-sexed siblings on a 12:12 light dark cycle with food and water ad libitum. Mice were allowed at least one hour to acclimate before behavioral testing, after being moved to the animal behavioral testing facility. Experimenters were blind to animal genotype during all analyses.
Primary behavioral phenotype assessment including open field assays
Characterization of the neural behavioral phenotype was performed on 18 mice per group (wild-type: 12 females and 9 males; heterozygous Pur-alpha (+/−): 9 females and 9 males) using the Irwin Observation Test Battery (Irwin, 1968) with Paylor’s and Crawley’s refinements (Crawley and Paylor, 1997, Paylor et al., 2006, Crawley, 2007). This standardized set of assays is also termed the SHIRPA (an acronym of SmithKline Beecham, Harwell, Imperial College, Royal London Hospital, phenotype assessment).
General health screen
A general physical examination of the mice was first conducted in home cages. All mice were weighed and presence of any superficial anatomical abnormalities, whisker condition (missing, sparse, short versus normal), coat quality, palpebral closure, presence of nasal and lacrimal discharge, respiration, and head, body, limbs and tail morphology were recorded and scored (0=normal or absent, and 1 = abnormal or present, as appropriate for the observation) (Crawley and Paylor, 1997, Paylor et al., 2006, Crawley, 2007). Any unusual patterns of locomotion, hyperactivity to handling or fighting in the home cage were also recorded.
Assay of neurologic reflexes
Next, the same18 mice/group mice underwent evaluation of their neurologic reflexes (Crawley and Paylor, 1997, Paylor et al., 2006, Crawley, 2007). For this, mice were removed from their home cage and placed on a disposable pad. Whisker response, pinna reflex and eye blink were tested by gently brushing the whiskers, ears, or eyelids of awake and freely moving mice with a sterile cotton swab (score: 0= no response; 1= reflexive response). Escape to touch was performed by approaching the mouse with a bent index finger (response score: 1 = mild, 2= moderate, and 3 = vigorous). Positional passivity was scored with increasing intensity of restraints, followed by assessment of abdominal and limb tone (score: −1 = low, 0= normal, +1 = high tone). Trunk curl, hind-limb extension and visual placing reflexes were assessed by gently restraining the animal by the tail and then lowering the mouse to the testing surface rapidly. Righting and toe pinch reflex assays were performed last. Acoustic startle was recorded in the same 18 mice/group as above in response to a training clicker (<70 dB) and scored: 0= no response, 0.5 reduced response, and 1= startle/Preyer reflex and momentary freezing. An additional 6 mice/group (n=3/gender/genotype; average of 5 months of age) were tested in response to a loud handclap (≥120 dB) using similar scoring.
Open-field activities
Mice (n=18/gp) were transferred in 27.3 cm × 27.3 cm clear open field boxes. The first 3 minutes of activity were scored for immediate transfer arousal and short-term spontaneous activity and behaviors (Crawley and Paylor, 1997, Paylor et al., 2006, Crawley, 2007). Vocalizations and biting were noted on occurrence. Spontaneous activity was tracked using a digital camera attached to the ceiling and ANY-Maze software to assess average distance moved, speed and the percent time spent in center versus the periphery. Locomotive abilities were qualitatively assessed as the animal walked around the open field box (scored: normal = 0; abnormal = 1). The equipment was cleaned with 70% isopropanol after each mouse.
Motor function
Gait abilities were assessed further in 4 wild-type (+/+) and 5 heterozygous (+/−) Pur-alpha mice using footprint pathway methods. The mice were trained to walk along a 50 cm long, 12 cm wide, paper-covered runway towards an enclosed box (Barlow and others 1996; Carter and others 1999; Clarke and Still 1999). Foot drags and missteps were recorded from the inked footprint patterns, as was angle of forepaw and hindpaw rotation, stride length, forepaw and hindpaw base width and front/hind footprint overlap.
Learning and Memory Tests
Novel Object Location memory
A spatial memory task was used in which mice were trained briefly to the location of three objects in open field boxes in 17 animals per group: 8 female and 9 male wild-type (+/+) mice; 11 female and 6 male heterozygous (+/−) Pur-alpha mice. The objects used were equal in shape and size (65 cm high × 60 cm long × 30 cm deep) and were plastic objects with no natural significance to mice. The mice were habituated to an empty open field arena in two 5-min trials, separated by a 3 min inter-trial interval. Twenty-four hours later, mice were permitted to explore three new objects placed into a diagonal arrangement in three 5-min acquisition trials, also with a 3 min inter-trial interval. Between each acquisition trial, mice were removed and the objects cleaned. One hour later, memory retention was probed by returning the mouse to the arena in which two objects had been moved to two new locations, and given 3 minutes to explore the objects. Nose-point tracking was used to define exploration of an object (Benice and Raber, 2008). The magnitude of reaction to spatial changes of familiar objects was determined, as described previously (Delcour et al., 2012).
Novel Object Recognition memory
In this memory task, the three objects differed in shape in order to assay ability to recognize objects. At the retention interval of 1 hour, memory was probed by returning the mouse to an arena in which two objects had been replaced with two “new” objects, and the third object was replaced with an identical version of the “familiar” object. Duration of contacts (i.e., exploration time) was measured for both “novel” and “familiar” objects during the 3 min testing session. Nose-point tracking was again used. The magnitude of reaction to novel objects was determined, as described previously (Delcour et al., 2012).
Barnes Maze
Twelve wild-type (+/+) and 11 heterozygous Pur-alpha mice (+/−) underwent Barnes Maze testing for spatial memory: 7 male and 5 female wild-type mice, and 5 male and 6 female heterozygous mice. A commercially available maze was used (Stoelting Co.). Dark curtains were hung to form a 177 cm × 177 cm square around the maze to exclude extraneous cues. Four spatial cues consisting of simple geometric figures (plus, square, circle, triangle) were hung on the four curtains. Mice were transported to testing room and allowed 1–2 hours to acclimate. The first habituation trial lasted for 1 minute, before the experimenter used their hands to guide the mouse to the target hole. If the mouse did not enter on its own volition, a gentle tail-pull was used to encourage entry into the escape tunnel. The escape tunnel was then covered and the mouse rested for one minute before moving the mouse to a holding cage for a 3 minute inter-trial interval during which time the maze was cleaned with 70% isopropanol. Acquisition training began one hour later, with mice being placed on the maze and permitted to explore for 5 minutes. If the mouse entered the escape tunnel, the trial was terminated, the target hole covered and the mouse permitted one minute of rest in the target hole. If the mouse did not exit the maze within the trial, the experimenter guided the mouse to the target hole as before and the mouse was kept there for 1 minute. Four acquisition trials were performed per day, for 4 days, for a total of 16 acquisition trials. The first memory retention trial occurred 24 hours later (on the fifth day) and again on day 30. For this, the escape tunnel was removed from the target hole and replaced with a shallow tray. The ability of the mice in finding the target hold was tested in a 5 minute trial. Trials were recorded using a CCD monochrome camera and analyzed using ANY-Maze tracking software. Using nose-point tracking, average speed around the maze and distance traveled and latency to entrance of the animal’s head into the target hole, blank holes, or neighboring holes were recorded per trial. The search strategy for each mouse to the target hole was scored from ANY-Maze captured videos as spatial, random or serial (Rosenfeld and Ferguson, 2014).
Immunochemistry in brain sections and quantification method
Brains were collected after behavioural testing from 10 wild-type (+/+) and 10 heterozygous Pur-alpha mice (+/−), when the mice were 11 months of age. One side of each brain was harvested for histological assessment (the other half was reserved for separate biochemical experiments not included here). The brain half to be assayed histologically was immersion fixed in formalin fixative and paraffin embedded. Brains were sectioned into 4 micrometer thick mid-sagittal or coronal serial sections and mounted onto charged slides (Thermo Fisher Scientific, Waltham, MA, USA). Half of the brains per group were sectioned longitudinally, and the other half coronally (n=5/group/genotype). Sections on slides were processed for immunocytochemical analysis using the streptavidin-biotin-peroxidase method to detect Pur-alpha- and neurofilament-labelled cell staining (Vecta Elite ABC Peroxidase Kit, Vector Laboratories, Inc., Burlingame, CA) (Khalili et al., 2003). Briefly, a monoclonal mouse anti-Pur-alpha antibody developed in the laboratory of Dr. Edward M. Johnson (clone 10B12) and a mouse anti-total neurofilament antibody (SMI33; Biolegend, San Diego, CA) were used. Sections were counterstained with haematoxylin.
Specificity of the anti-Pur-alpha antibody has been previously described (Khalili et al., 2003). We also performed no primary antibody control staining in which serum was substituted for the primary antibody, followed by secondary antibodies. No labeling was observed as a result of incubation of tissues with serum and then secondary antibodies alone.
The cerebellum and hippocampus were assayed in mid-sagittal longitudinal sections; amygdala, prefrontal, frontal and parietal cortices were assayed in coronal sections. Heights of the prefrontal, frontal and parietal cortices were assayed as previously described (Delcour et al., 2012). Zonal arrangement of hippocampal and prefrontal, frontal and parietal cortices were examined qualitatively. Numerical densities of the total neuronal population (neurofilament-labelled cells), Pur-alpha immunostained neuronal population and Pur-alpha immunostained dendritic population were estimated using stereological optical dissector methods, a Nikon E800 microscope equipped with a motorized stage, and an image analysis software controlled computer (Bioquant Life Science, Nashville, TN). The region of interest was circumscribed at 10.5 × magnification using a 1× objective on “live” digitized microscope images on a monitor, and the volume of reference estimated. Neurons and dendrites were counted in this region using a 40× oil objective and the optical dissector sampling method, as described previously (Delcour et al., 2012). Ten optical dissectors were counted in 5–10 separate regions in the volume of interest (dependent on size of structure measured). For the cerebellum, the density of Purkinje cell bodies and dendrites were quantified in the mid-line vermis.
Statistics
Graph-Pad Prism was used for all statistical analyses. Two-way repeated measures ANOVAs were used to analyse behavioral tests using the factors: genotype and behavioral assay (Figures 1A, 2C), genotype and gender (Figures 1B and C, 3B, Table 1 results), genotype and limb (Fig. 2B), genotype and hole visited (Fig. 4), or genotype and time (Fig. 5A–E). Two-way ANOVAs were used to analyse the brain histopathology with the factors genotype and structure analysed (total number of neurons, and number of Pur-alpha immunostained neurons or dendrites). Fisher least significant difference tests were used for post-hoc analyses in most cases, although Mann-Whitney U (non-parametric) tests were used as the post-hoc tests for the neuronal and dendritic counts due to the small sample size for each brain region assayed. P values of < 0.05 were considered significant. Results were expressed as mean ± SEM.
Figure 1.
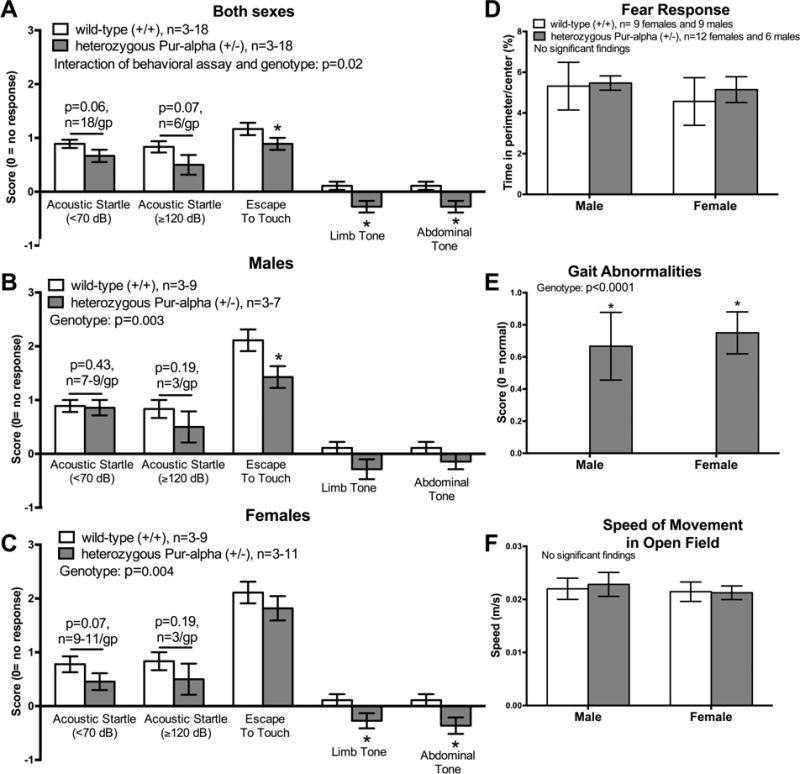
Select results (neural reflexes and open field activities) from a general health screen. (A) Several neural reflexes were decreased in heterozygous Pur-alpha (+/−) mice (both sexes mixed), compared to wild-type (+/+) littermates (both sexes mixed) (two-way ANOVA showed a difference between the genotypes, p<0.0001). Posthoc analysis showed a significantly decreased escape behavior, limb tone and abdominal tone in the heterozygous mice, as well as a trend towards a reduction in acoustic startle to two different sudden noises. (B, C) Similar results were observed after dividing the groups by sex. (D–F) In the open field assay, both male and female heterozygous Pur-alpha (+/−) mice showed similar amounts of time spent in the perimeter versus the center of the arena and similar mean speeds, yet increased gait abnormalities. *p<0.05, compared to wild-type (+/+) littermates. Mean ± SEM shown.
Figure 2.
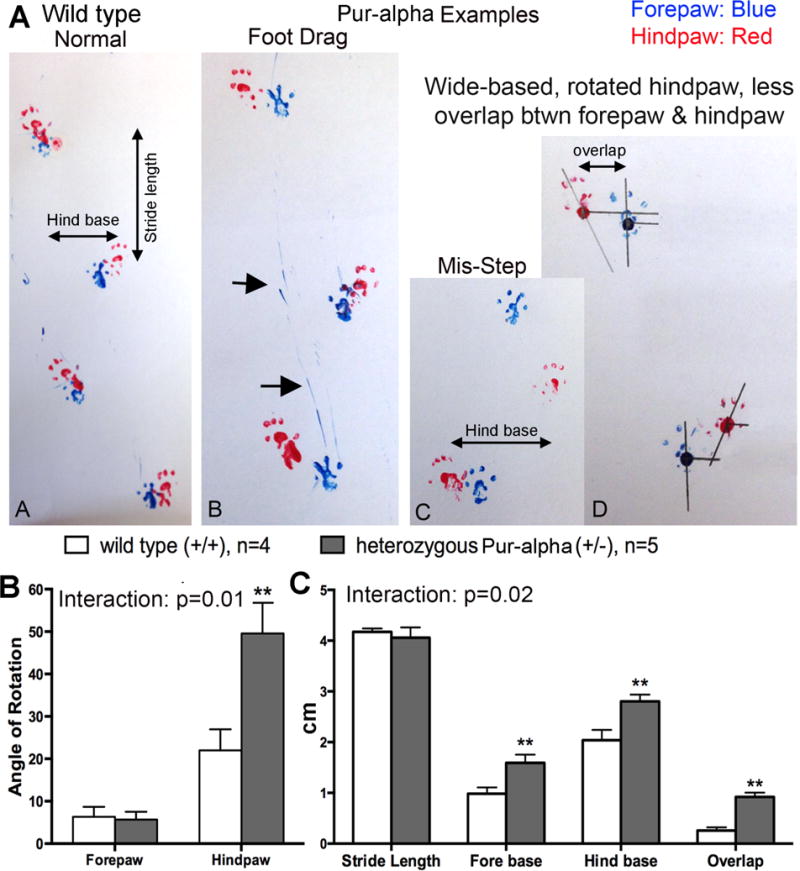
Gait abnormalities were evident in heterozygous Pur-alpha (+/−) mice during forward walking. (A) Representative figures show that heterozygous Pur-alpha (+/−) mice have more foot drags, missteps, outwardly positioned hindpaws, and wider-based steps, compared qualitatively to wild-type (+/+) mice. (B) Footprint pattern analysis showed that hindpaws of heterozygous Pur-alpha mice were more rotated than wild-type (+/+) littermates (specifically, hindpaws showed an increased angle during walking so that they pointed away from the forward direction of the mouse). (C) Footprint pattern analysis also showed that heterozygous Pur-alpha mice had wider fore base and hind base gaits, and less overlap between forepaws and hindpaws during forward walking. **p<0.01, compared to wild-type (+/+) littermates. Mean ± SEM shown.
Figure 3.
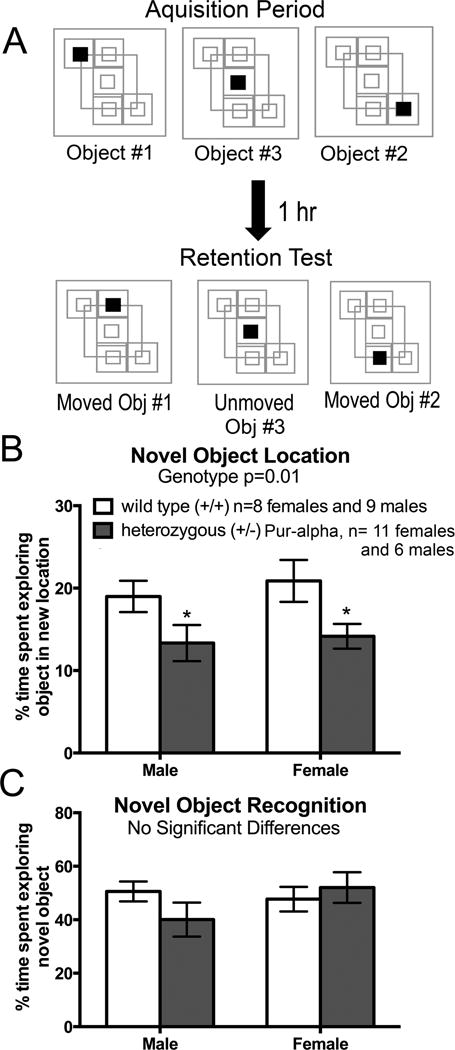
Assessment of memory abilities in novel object location and novel object recognition assays in wild-type mice (+/+) and heterozygous Pur-alpha (+/−) mice. (A) Cartoons showing the design of the novel object recognition assay of the location of three objects in an open field chamber. Mice were allowed to explore the location of 3 objects during a 3 minute acquisition assay. For the retention test that occurred 1 hour later, two objects were moved and one object remained in the same location. (B) Both male and female heterozygous Pur-alpha mice showed similar decreases in percent time exploring moved objects relative to unmoved objects, compared to age-matched male and female wild-type (+/+) littermates. This is indicative of a deficit in spatial location memory in the heterozygous Pur-alpha mice. (C) Neither group showed deficits in novel object recognition. *p<0.05, compared to wild-type (+/+) littermates. Mean ± SEM shown.
Table 1.
Barnes Maze results on Retention day 5 (24 hours after final acquisition trial) analyzed with factors sex and genotype.
| Wt (+/+) Males (n=7) |
Wt (+/+) Females (n=5) |
Het Pur-alpha (+/−) Males (n=5) |
Het Pur-alpha (+/−) Females (n=6) |
Significant 2-way ANOVA findings | |
|---|---|---|---|---|---|
| Average velocity | 0.08+0.003 | 0.07+0.003 | 0.05+0.002* | 0.05+0.008* | Genotype p = 0.01 |
| Total distance | 14.62+1.99 | 14.63+1.84 | 22.09+1.85* | 21.63+1.25* | Genotype p = 0.0008 |
| Distance to head entering target hole | 0.87+0.02 | 0.61+0.13 | 1.77+0.53* | 1.19+0.30 | Genotype p = 0.02 |
| Distance to head entering blank hole | 0.50+0.8 | 0.73+0.13 | 0.43+0.07 | 0.36+0.02* | Genotype p = 0.01 |
| Primary latency to head entering target hole | 15.34+4.61 | 6.04+1.16 | 29.88+9.89 | 23.60+10.08 | Genotype p = 0.04 |
p<0.05, compared to same sex wild type (+/+) mice; Sex not significant for any test.
Figure 4.
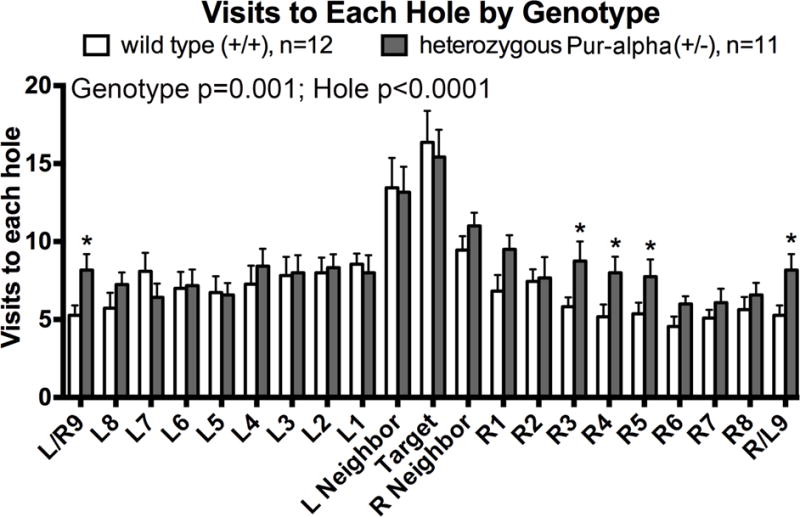
Assessment of memory abilities in a Barnes maze test at 24 hours after the last day of acquisition training. (A) Heterozygous Pur-alpha (+/−) mice showed increased numbers of visits to several blank holes (*:p<0.05), compared to age-matched wild-type (+/+) littermates. *p<0.05, compared to wild-type (+/+) littermates. Mean ± SEM shown.
Figure 5.
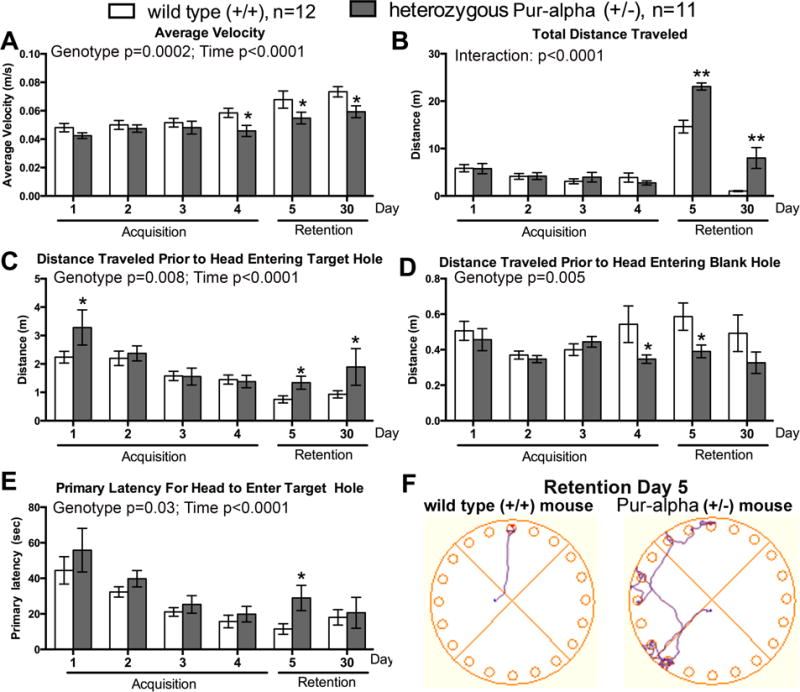
Further assessment of memory abilities in Barnes maze tests. Animals were trained for 4 days to learn the maze (4 trials/day for a total of 16 trials) before being tested in retention tests at 24 hours (day 5 of testing) and 30 days after the last day of acquisition training. (A) Heterozygous Pur-alpha (+/−) mice showed slower average velocity around the Barnes maze on the last day of acquisition training, and at both retention test time points, than wild-type (+/+) littermates. (B,C) Heterozygous Pur-alpha mice showed longer total distance traveled and greater distance prior to their head entering the target hole, at both retention test time points. (D) Heterozygous Pur-alpha mice had shorter distances prior to their heads entering a blank hole during the last day of acquisition training and 24 hour (day 5) retention test. (E) Heterozygous Pur-alpha mice had an overall longer latency before the head entered the target hole during the 24 hour (day 5) retention test. (F) Representative cartoons showing that wild-type (+/+) mice had a higher prevalence for a direct spatial search strategy as shown on left, while heterozygous Pur-alpha mice showed more random, serial, or a mix (as shown on right) during the Day 5 Retention Test. *:p<0.05 and **p<0.01, compared to wild-type (+/+) littermates. Mean ± SEM shown.
Results
No differences by weight by adulthood, but increased spontaneous early death of heterozygous Pur-alpha (+/−) mice, compared to wild-type (+/+) mice
Mice did not differ by weight between the two genotypes after week 7 or throughout the course of the 11 month experiment. No differences in superficial anatomical abnormalities, whisker condition, coat quality, morphology or other general physical characteristics were observed between the two groups. However, the prevalence of spontaneous early deaths in the Pur-alpha colony over its 16 year history was higher in the heterozygous Pur-alpha (+/−) mice than wild-type mice, with 129 early deaths recorded in the heterozygous Pur-alpha (+/−) mice (7.14 %), versus only 8 early deaths recorded in the wild-type (+/+) mice (0.44%), out of a total of 1805 mice.
Heterozygous Pur-alpha (+/−) mice show decreased escape to touch and hypotonia
With regard to neuro-reflexive functions, significant decreases in an escape response to touch and tone in the limbs and abdomen were observed in heterozygous Pur-alpha (+/−) mice (p<0.05 in post hoc test), compared to wild-type (+/+) mice (Fig. 1A; the two-way ANOVA showed a significant difference in the interaction of behavioral assay and genotype, p=0.02). The reduced escape to touch matched qualitative observations of reduced anxiety during handling in the heterozygous Pur-alpha (+/−) mice, compared to wild-type mice (data not shown). There was a trend towards a reduced acoustic startle response to a training clicker noise of < 70dB in heterozygous Pur-alpha (+/−) mice (p=0.06 post hoc test result; Fig 1A). Therefore, the acoustic startle response to a loud handclap (≥120 dB) was tested in six additional animals per group and again, a trend towards a reduced response was observed in heterozygous Pur-alpha (+/−) mice of both sexes (p<0.07 post hoc test result; Fig. 1A). Similar results were observed after dividing the groups by sex, although a stronger trend towards a reduced acoustic startle was observed in female heterozygous Pur-alpha (+/−) mice compared to wild-type mice (Fig. 1B,C). The heterozygous Pur-alpha (+/−) mice displayed no differences in time spent in the perimeter of the open field arena (a fear response), compared to wild-type mice (Fig. 1D). However, both male and female heterozygous Pur-alpha (+/−) mice displayed gait abnormalities during this general health screen (p<0.05 each sex), compared to wild-type mice that showed none (Fig. 1E; two-way ANOVA showed a significant difference between genotypes, p<0.0001). These gait differences did not affect speed of movement in the open field arena (Fig. 1F).
Heterozygous Pur-alpha (+/−) mice show increased missteps, outwardly rotated hindpaws and wide-based gait
We next used footprint pattern analyses to further explore the gait differences in a smaller cohort of mice (Fig. 2). Heterozygous Pur-alpha (+/−) mice displayed increased incidence of foot drags, missteps, wide-based steps and outwardly rotated hindpaws when walking forward, compared qualitatively to wild-type littermates (Fig. 2A). Footprint pattern analysis showed increased rotation in the hindpaws of heterozygous Pur-alpha (+/−) mice (p<0.05), compared to wild-type littermates (Fig. 2B; the two-way ANOVA showed a significant difference in the interaction of genotype and limb, p=0.01). Footprint pattern analysis also showed wider-based steps in both forepaws and hindpaws, and less overlap in forepaws and hindpaws during forward walking in heterozygous Pur-alpha (+/−) mice (p<0.01 each), compared to wild-type littermates (Fig. 2B,C; the two-way ANOVA showed a significant difference in the interaction of genotype and behavioral assay, p=0.02).
Heterozygous Pur-alpha (+/−) mice show deficits in spatial memory, but preservation of novel object recognition
In the novel object location task that tests an animal’s ability to detect “where” features, heterozygous Pur-alpha (+/−) mice displayed deficits at the 1 hour memory retention testing point (Fig. 3A,B). Specifically, both male and female heterozygous Pur-alpha (+/−) mice spent less time exploring the “moved” versus “unmoved” objects (p<0.05 each), compared to male and female wild-type mice (Fig. 3A,B; two-way ANOVA showed a significant difference by genotype, p=0.005). No sex differences were observed in either group.
In contrast, the novel object recognition task that tests an animal’s ability to detect “what” features revealed no significant differences in either group’s ability to recognize novel objects at the 1 hour retention testing point (Fig. 3C). No sex differences were observed in either group.
In the Barnes Maze assay, heterozygous Pur-alpha (+/−) mice also displayed memory deficits, in the form of increased numbers of blank holes visited, compared to wild-type littermates (Fig. 4). The two-way ANOVA revealed significant differences between the genotypes (p=0.001) and hole visited (p<0.0001), while the posthoc tests showed increased visits to several blank holes by Pur-alpha (+/−) mice, as indicated in Fig. 4. Despite the ability to walk at normal speeds in the open field test (Fig. 1F), the heterozygous Pur-alpha (+/−) mice had slower speeds around the maze on the 4th day of acquisition training, at the 24 hour retention trial (day 5) and at the 30 day retention test, compared to wild-type littermates (Fig. 5A). Part of the reason for the slower velocity was that heterozygous Pur-alpha (+/−) mice traveled longer distances around the maze before their head entered the target hole on both retention day tests (days 5 and 30; Fig. 5B,C). The heterozygous Pur-alpha (+/−) mice traveled shorter distances before investigating a blank hole during the fourth acquisition trial and 24 hour retention trial (day 5), compared to wild-type littermates (Fig. 5D), and showed a similar trend during the 30 day retention test. They also showed an increase in the primary latency before their head entered the target hole at the 24 hour retention trial (day 5) (Fig. 5E), although both genotypes had similar latencies during the 30 day retention test. The heterozygous Pur-alpha (+/−) mice had a higher prevalence for random or serial search strategies on 24 hour retention trial (day 5), than the wild-type mice that more commonly used a spatial search strategy (Fig. 5F). For succinctness here, specifics of ANOVA and significant posthoc findings are reported in individual panels of Figure 5. Table 1 shows that there were no sex differences observed in either group regarding Barnes Maze results.
Heterozygous Pur-alpha (+/−) mice show no gross morphological differences in several brain regions compared to wild-type mice
No differences in overall size of the brains were visible between the two groups at tissue collection or after sectioning of the brains. No differences in height of several regions of the cerebral cortex (prefrontal, frontal cortex, and parietal cortices were examined) were detected (data not shown), nor did there appear to be altered zonal arrangements in these same cortices between the two groups. Nor were gross morphological differences observed in the hippocampus or cerebellum between the two groups (Fig. 6A–H).
Figure 6.
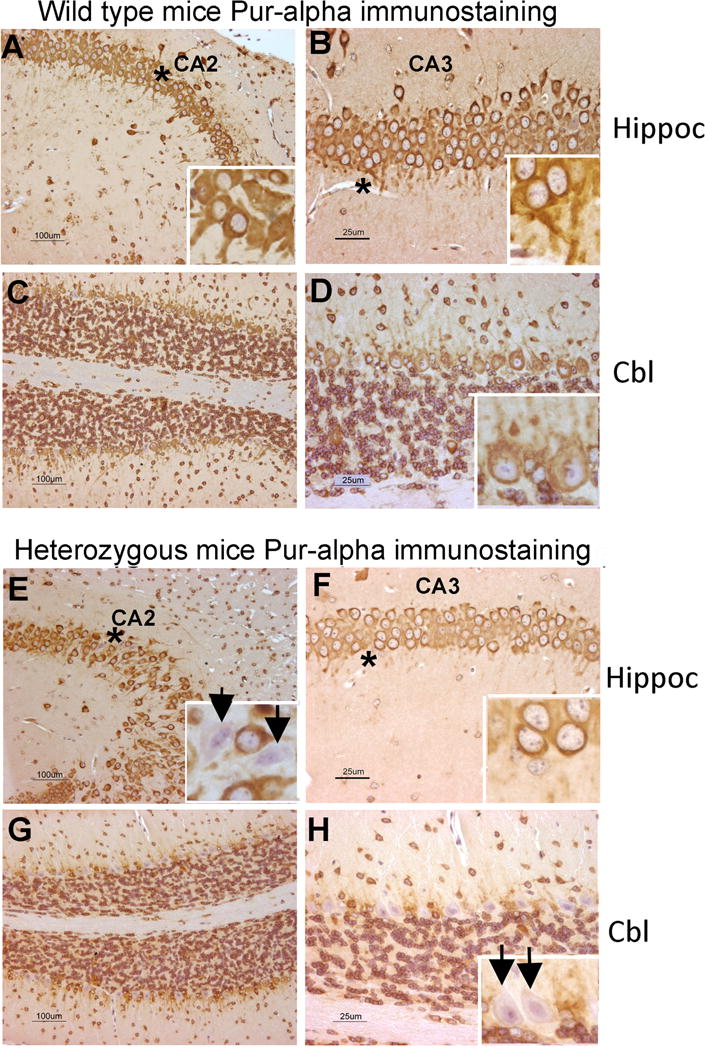
Reduced Pur-alpha immunostained neurons and dendrites in hippocampus and cerebellum of mature (11 months of age) heterozygous Pur-alpha (+/−) mice, compared qualitatively to wild-type (+/+) mice littermates. (A–D). Low power and higher power photomicrographs from hippocampal (Hippoc) CA2 and CA3 regions and vermal cerebellum (Cbl) region of wild-type (+/+) mice. Insets in A and B show neurons with cytoplasm intensely stained for Pur-alpha as well as long intensely stained dendrites. (E–H) Low power and higher power photomicrographs from hippocampal (Hippoc) CA2 and CA3 regions and cerebellum and vermal cerebellum (Cbl) region of heterozygous Pur-alpha (+/−) littermates. Arrows in the insets of panels E and H indicate hippocampal neurons and Purkinje cells lacking Pur-alpha staining, respectively. Panel F and its inset shows that neurons in the CA3 region of a heterozygous Pur-alpha mouse have little to no Pur-alpha stained dendrites. Scale bars in A, C, E and G = 100 micrometers; Scale bars in B, D, F and H = 25 micrometers.
Heterozygous Pur-alpha (+/−) mice show reduced numbers of neurons and dendrites in the hippocampus, cerebellum and amygdala
In contrast to above, differences in neuronal density, dendritic density and length, and numbers of neurons and dendrites stained for Pur-alpha were apparent between the two groups in the cerebellum and hippocampus. In the wild-type (+/+) mice, most neurons and dendrites in each of these regions showed clear immunostaining for Pur-alpha (Fig. 6A–D. In the cerebellum (Fig. 6G,H; Fig. 7A), there were reduced numbers of Purkinje cell bodies and dendrites immunostained for Pur-alpha in heterozygous Pur-alpha (+/−) mice (p<0.01 each), compared to wild-type (+/+) littermates. The total number of Purkinje cell bodies in heterozygous Pur-alpha (+/−) mice (immunostained for Pur-alpha and hematoxylin stained only) was also reduced (p<0.05), compared to wild-type (+/+) littermates (Fig. 7; the two-way ANOVA showed a significant interaction between genotype and structure of p=0.006. Such frank differences were not observed in the cerebellum granule cell layer of heterozygous Pur-alpha (+/−) mice, compared to wild-type (+/+) littermates (data not shown). In the CA1-3 regions of the hippocampus (Fig. 6E,F; Fig. 7B), we observed reduced numbers of neuronal cell bodies overall, and reduced numbers of Pur-alpha+ neuronal cell bodies and dendrites in heterozygous Pur-alpha (+/−) mice (p<0.05 and p<0.01, respectively), compared to wild-type (+/+) littermates.
Fig. 7.
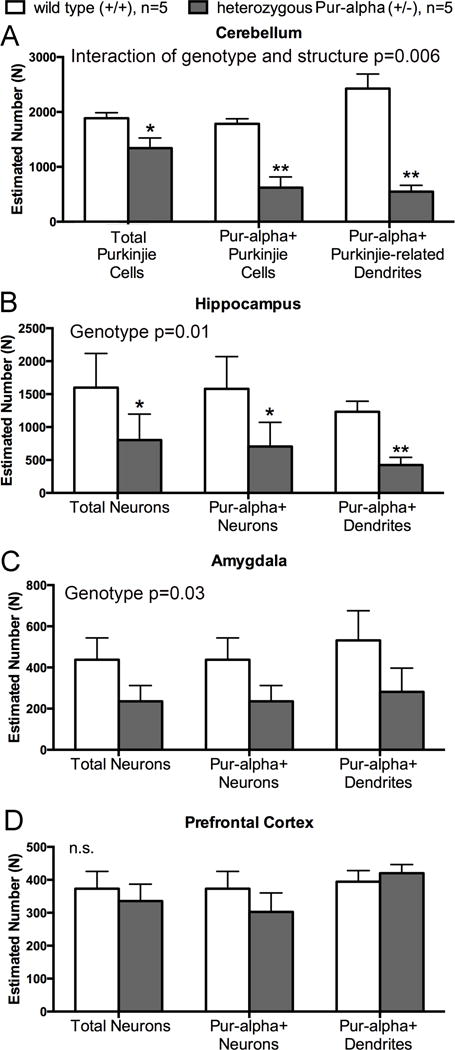
Reduced numbers of Pur-alpha immunostained neurons and dendrites in hippocampus, cerebellum and amygdala of mature (11 months of age) heterozygous Pur-alpha (+/−) mice, compared to wild-type (+/+) mice littermates. (A) Estimated total number of Purkinje cell bodies, Pur-alpha immunostained (+) Purkinje cell bodies, and Pur-alpha + Purkinje cell dendrites in the cerebellum. (B–D) Estimated total number of neuronal cell bodies, Pur-alpha-immunopositive neuronal cell bodies, and Pur-alpha-immunopositive dendritic elements in the hippocampus CA1-3 region, amygdala and prefrontal cortex. Results of two-way ANOVA shown at upper right in each panel; significant posthoc findings shown as *p<0.05 and **p<0.01, compared to wild-type (+/+) littermates. Mean ± SEM shown.
A small reduction of Pur-alpha-immunopositive neurons and dendrites were observed in the amygdala after two-way ANOVA analysis by genotype (p=0.03), although there were no significant post-hoc findings (Fig. 7C). However, the prefrontal cortex showed no significant changes between the two genotypes (Fig. 7D).
Discussion
Here we present for the first time an extensive assessment of the behavioral phenotype and brain histopathology of mice that are heterozygous for the Pur-alpha allele. The heterozygous Pur-alpha (+/−) mice showed a tendency to spontaneous early deaths. They also show limb and abdominal hypotonia, ataxic gait abnormalities suggestive of cerebellar pathology, and cognitive memory deficits suggestive of hippocampal pathology. The heterozygous Pur-alpha (+/−) mice also showed reduced anxiety, a reduced escape to touch, and a trend towards reduced acoustic startle responses suggestive of amygdala pathology. Histological assays of the vermal region of the cerebellum, CA2 and 3 regions of the hippocampus, and amygdala revealed reduced neurons in general in heterozygous Pur-alpha (+/−) mice, as well as reduced numbers of neurons and dendrites that were immunoreactive for the Pur-alpha protein in these same brain regions.
Pur-alpha is a member of a protein family that is strongly conserved across species (Johnson, 2003). It plays multiple roles in DNA replication and gene transcription (Gallia et al., 2000, Johnson, 2003). Prior studies have shown that Pur-alpha is essential for proper early postnatal development of the mouse brain (Khalili et al., 2003, Zeng et al., 2005, Johnson et al., 2006, Hokkanen et al., 2012). We have previously reported that homozygous Pur-alpha (−/−) mice show no evidence of Pur-alpha protein production in whole brain extracts, and in immunohistochemically-stained cerebral cortex, cerebellum and hippocampus (Khalili et al., 2003, Mishra et al., 2013). They also showed significantly reduced numbers of neurons, reduced synaptic connections, abnormal dendritic development in their hippocampus and cerebellum relative to wild type (+/+) mice, and reduced numbers of cells in bronchial epithelium, thymus and spleen (Khalili et al., 2003), as well as abnormal dendritic development in their cerebellum and hippocampus (Johnson et al., 2006). Unfortunately, the homozygous Pur-alpha (−/−) mice developed in our lab do not gain weight normally, show increased incidence of tremors and seizures, and die typically by 1 month of age (Khalili et al., 2003). Another group has also developed a Pur-alpha deficient (−/−) knockout mouse model (Hokkanen et al., 2012). These mice display a persistent continuous tremor after 2 weeks of age, a significant loss of Purkinje cell density by 5 months of age, megacephaly and expiration by 6 months of age (the latter two considerably later than our homozygous Pur-alpha (−/−) mice) (Hokkanen et al., 2012), though it should be noted that these animals contain a large deletion of sequences flanking the PURA coding region and this, as well as possible off-target effects, may account in part for the differences observed between the two models.
Due to the early lethality of the knockout mice and the haploinsufficiency in Pur-alpha levels in heterozygous animals (Khalili et al., 2003, Mishra et al., 2013), we decided to pursue behavioral studies in mice that are heterozygous (+/−) for the Pur-alpha protein. We have previously reported that heterozygous Pur-alpha (+/−) mice show a 25% reduction in the Pur-alpha protein from wild-type levels at postnatal day 5 and a 50% from wild-type levels by adulthood (Khalili et al., 2003, Mishra et al., 2013), and a loss in coordinated RhoA expression during development, albeit less of a loss than in homozygous Pur-alpha (−/−) mice (Mishra et al., 2013). They also display delays in weight gain during development, although they eventually recover so that by adulthood they are nearly indistinguishable in body weight and morphology from wild-type (+/+) littermates (Khalili et al., 2003). However, Pur-alpha (+/−) mice have occasional spontaneous seizures during routine handling (Khalili et al., 2003). We further report here an increased prevalence of spontaneous early deaths in heterozygous Pur-alpha (+/−) mice, compared to wild-type (+/+) mice, over a 16 year history of 0.44% versus 7.14%.
We have previously reported that heterozygous Pur-alpha (+/−) mice have a slight waddling gait (Khalili et al., 2003). We extended those findings here to show that heterozygous Pur-alpha (+/−) mice display gait ataxias, such as wider-based steps, less overlap in forepaws and hindpaws, and increased foot drags and missteps, as well as more outwardly rotated hindpaws during forward walking. Such gait changes are typically associated with cerebellar degeneration (Ivanov et al., 2004, Takeuchi et al., 2012), matching our findings of neuronal loss in the cerebellar vermis of heterozygous Pur-alpha (+/−) mice. These mice also showed limb and abdomen hypotonia during SHIRPA testing, changes also typically attributed to the cerebellum. The increased foot drags and outward hindpaw rotation during forward walking could also be related to peripheral nerve degeneration (Sereda et al., 1996), the possibility of which we are exploring in a separate study, although similar gait changes have been observed in association with cerebellar dysfunction (Ivanov et al., 2004, Takeuchi et al., 2012). Protein-altering de novo mutations in PURA, which encodes the Pur-alpha protein, have recently been described in several young human subjects (Hunt et al., 2014, Lalani et al., 2014, Tanaka et al., 2015). Three of four subjects diagnosed with PURA syndrome presented with hypotonia, poor coordination or wide-based gait and had Pura-alpha mutations that included two different frameshifts, one in frame deletion and one missense mutation (Hunt et al., 2014). In another study, eleven human subjects with 5q31.3 microdeletion syndrome and de novo mutations in PURA had significant hypotonia and developmental delays; most had seizures and abnormal EEGs (Lalani et al., 2014). In a third recent study, six subjects with several novel variants in PURA also shared similar clinical features of hypotonia, developmental delays and seizures or seizure-like movements (Tanaka et al., 2015). Thus, the phenotype of the Pur-alpha (+/−) mice studied here display several striking parallels with some of the young human subjects harboring PURA mutations and may be useful for further mechanistic studies in this regard.
Heterozygous Pur-alpha (+/−) mice also showed significant cognitive deficits, specifically spatial memory deficits. This included short-term memory deficits (1 hour) in a novel object location memory test, evidenced by reduced reaction times to a new location of the moved objects. A lower reaction time in this test may result from impairments in encoding object spatial configuration or disturbances in short-term memory processing, a pattern of cognitive deficit that is consistent with hippocampal dysfunction (Benice et al., 2006, Murai et al., 2007, Delcour et al., 2012). In Barnes Maze testing, the heterozygous Pur-alpha (+/−) mice had slower velocities and traveled longer distances before finding the target hole during the 4th acquisition trial and both retention test days. This was despite their ability to walk at normal speeds in an earlier open field test (the latter indicating that their gait abnormality did not affect their speed). They also visited more blank holes during the 5 day retention test than wild-type littermates (+/+), further indicative of spatial memory deficits. At the 30 day retention trial, the wild-type mice also show signs of forgetting the location of the target hole, reducing the difference between the two genotypes in the primary latency for the head entering the target hole. The heterozygous Pur-alpha (+/−) did not develop the typical direct spatial search strategy, but retained random or serial search strategies (or a mix), also highlighting their difficulties in navigating to a specific location using spatial cues (Bach et al., 1995, Lieb et al., 1999, O’Leary and Brown, 2013). Like the novel object location memory test results, these findings are indicative of hippocampal pathology, since an intact hippocampus is needed for both storage and retrieval of spatial location memories (Eichenbaum et al., 2007, Murai et al., 2007, Farovik et al., 2011). Such pathology was evident in the CA2 and CA3 regions of the hippocampus as reduced numbers of neurons overall, reduced numbers of Pur-alpha immunostained neurons and dendrites, and shorter Pur-alpha immunostained dendrites. The lack of significant histopathological findings in the prefrontal cortex and the preservation of novel object recognition abilities suggests that prefrontal cortical function is spared in the heterozygous Pur-alpha (+/−) mice (Wais, 2011).
Past studies have implicated Pur-alpha in the mediation of neurodegeneration in the context of HIV-1 infection, particularly through its association with the key viral regulatory Tat protein (for review see Johnson et al, 2013). Humans with HIV show astrogliosis, myelin pallor within white matter tracts, and deficits in synaptic transmission in areas of the brain involving the dopaminergic system, including the prefrontal cortex and hippocampus (Gelman et al., 2012, Gelman, 2015). All of these have been described in the Pur-alpha mouse model (Khalili et al, 2003). Similarly, deficits in learning and memory, executive functioning, and speed of informational processing are common in HIV-1 associated neurological disease (HAND), even in patients on antiretroviral therapy (Heaton et al., 2011). Absent in our Pur-alpha mouse model are any signs of overt inflammatory effects, which play a critical role in HIV-1 infection and in more severe stages of HAND, but that are minimal in patients with mild CNS disease. Thus, Pur-alpha heterozygous knockout mice with their deficits in learning, memory and speed of informational processing, as shown with Barnes maze testing, may provide a well-defined animal model in which to study mechanisms of HIV-1 associated neurological disorders, including cognitive deficiencies.
The reduced anxiety, reduced escape to touch, and reduced response to acoustic startle observed in the heterozygous Pur-alpha (+/−) mice are behaviors typically indicative of docility or reduced fear. These behaviors may be the result of the decreased numbers of neurons overall and decreased numbers of Pur-alpha immunostained neurons and dendrites in the amygdala (Trogrlic et al., 2011), although deficits in escape to touch have also been associated with Purkinje cell loss in the cerebellum (Chintala et al., 2009). Jacquelin et al found similar reductions in escape to touch (as well as ataxic motor and other behavioral changes) in homozygous Dab1(scm) mouse mutants that had cell ectopias in the cerebellar cortex, hippocampus, and neocortex, compared to non-ataxic controls on postnatal day 22 (Jacquelin et al., 2012). The Dab1(scm) mutants showed hypometabolic neuronal activity in cerebellar cortical layers and efferent deep cerebellar nuclei, estimated by cytochrome oxidase histochemistry on cerebellar sections, relative to non-ataxic mice, despite normal metabolism in both regular and ectopic Purkinje cells. The reduced response to acoustic startle was an unexpected finding. It is interesting that one human subject with PURA syndrome lacked awareness of danger (Hunt et al., 2014), a behavior finding that could be similar to the reduced anxiety, acoustic startle and reduced escape to touch responses seen in our heterozygous Pur-alpha (+/−) mice. However, since the magnitude of the reduced acoustic startle responses was small and did not reach significance in the heterozygous Pur-alpha (+/−) mice, further studies are needed to determine their source, and cognitive or biological significance.
Limitations of this study include not prolonging the novel object location retention test to longer time frames than 1 hour. However, Murai et al have shown that wild-type (+/+) mice were able to discriminate an object in a novel location at < 2 hours after the final acquisition trial, but not at > 2 hours (Murai et al., 2007). Therefore, we utilized the Barnes Maze to test for long-term memory deficits, with good success. Sex differences were not examined in every assay due to limited numbers of mice and/or tissues in subsets of assays. A longitudinal assessment could also be employed in future studies to examine the effects of development and aging in mice that are heterozygous for a regulator of amyloid precursor (that is, Pur-alpha) (Darbinian et al., 2008). Lastly, additional studies are needed to determine the reasons for the reduced acoustic startle reflexes in the heterozygous Pur-alpha (+/−) mice.
In conclusion, mice heterozygous for Pur-alpha have impaired gait and limb tone as well as several memory deficits, which may relate to observed deficits in the cerebellum and hippocampus, respectfully. Based on our previous results where we observed neuronal loss in the cortex, cerebellum and hippocampus, and cell loss in many non-neural tissues of homozygous Pur-alpha (−/−) mice (Khalili et al., 2003, Mishra et al., 2013), we anticipate other brain regions and organs may be effected in heterozygous Pur-alpha (+/−) mice during development, although this is still under investigation. These results are encouraging and support further research using Pur-alpha heterozygous knockout mice to study mechanisms or treatments of several neurodegenerative and neurocognitive disorders, such as Fragile X Tremor Ataxia Syndrome, PURA Syndrome and neuroAIDS. For example, peripheral neuropathology is a clinical problem observed in patients with HIV, which may result from the virus itself, co-morbidities, or the use of some antiretroviral therapies. We are pursuing that topic in a separate study.
Acknowledgments
Research reported in this publication was supported by the National Institute of Mental Health of the National Institutes of Health under Award Number P30 MH092177. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
References
- Bach ME, Hawkins RD, Osman M, Kandel ER, Mayford M. Impairment of spatial but not contextual memory in CaMKII mutant mice with a selective loss of hippocampal LTP in the range of the theta frequency. Cell. 1995;81:905–915. doi: 10.1016/0092-8674(95)90010-1. [DOI] [PubMed] [Google Scholar]
- Benice TS, Raber J. Object recognition analysis in mice using nose-point digital video tracking. J Neurosci Methods. 2008;168:422–430. doi: 10.1016/j.jneumeth.2007.11.002. [DOI] [PubMed] [Google Scholar]
- Benice TS, Rizk A, Kohama S, Pfankuch T, Raber J. Sex-differences in age-related cognitive decline in C57BL/6J mice associated with increased brain microtubule-associated protein 2 and synaptophysin immunoreactivity. Neuroscience. 2006;137:413–423. doi: 10.1016/j.neuroscience.2005.08.029. [DOI] [PubMed] [Google Scholar]
- Chepenik LG, Tretiakova AP, Krachmarov CP, Johnson EM, Khalili K. The single-stranded DNA binding protein, Pur-alpha, binds HIV-1 TAR RNA and activates HIV-1 transcription. Gene. 1998;210:37–44. doi: 10.1016/s0378-1119(98)00033-x. [DOI] [PubMed] [Google Scholar]
- Chintala S, Novak EK, Spernyak JA, Mazurchuk R, Torres G, Patel S, Busch K, Meeder BA, Horowitz JM, Vaughan MM, Swank RT. The Vps33a gene regulates behavior and cerebellar Purkinje cell number. Brain Res. 2009;1266:18–28. doi: 10.1016/j.brainres.2009.02.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crawley JN. What’s wrong with my mouse?: behavioral phenotyping of transgenic and knockout mice. Hoboken, N.J.: Wiley-Interscience; 2007. [Google Scholar]
- Crawley JN, Paylor R. A proposed test battery and constellations of specific behavioral paradigms to investigate the behavioral phenotypes of transgenic and knockout mice. Hormones and behavior. 1997;31:197–211. doi: 10.1006/hbeh.1997.1382. [DOI] [PubMed] [Google Scholar]
- Daigle JG, Krishnamurthy K, Ramesh N, Casci I, Monaghan J, McAvoy K, Godfrey EW, Daniel DC, Johnson EM, Monahan Z, Shewmaker F, Pasinelli P, Pandey UB. Pur-alpha regulates cytoplasmic stress granule dynamics and ameliorates FUS toxicity. Acta Neuropathol. 2016;131:605–620. doi: 10.1007/s00401-015-1530-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darbinian N, Cui J, Basile A, Del Valle L, Otte J, Miklossy J, Sawaya BE, Amini S, Khalili K, Gordon J. Negative regulation of AbetaPP gene expression by pur-alpha. J Alzheimers Dis. 2008;15:71–82. doi: 10.3233/jad-2008-15106. [DOI] [PubMed] [Google Scholar]
- Darnell JC, Fraser CE, Mostovetsky O, Darnell RB. Discrimination of common and unique RNA-binding activities among Fragile X mental retardation protein paralogs. Hum Mol Genet. 2009;18:3164–3177. doi: 10.1093/hmg/ddp255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darnell JC, Jensen KB, Jin P, Brown V, Warren ST, Darnell RB. Fragile X mental retardation protein targets G quartet mRNAs important for neuronal function. Cell. 2001;107:489–499. doi: 10.1016/s0092-8674(01)00566-9. [DOI] [PubMed] [Google Scholar]
- Delcour M, Olivier P, Chambon C, Pansiot J, Russier M, Liberge M, Xin D, Gestreau C, Alescio-Lautier B, Gressens P, Verney C, Barbe MF, Baud O, Coq JO. Neuroanatomical, sensorimotor and cognitive deficits in adult rats with white matter injury following prenatal ischemia. Brain Pathol. 2012;22:1–16. doi: 10.1111/j.1750-3639.2011.00504.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eichenbaum H, Yonelinas AP, Ranganath C. The medial temporal lobe and recognition memory. Annu Rev Neurosci. 2007;30:123–152. doi: 10.1146/annurev.neuro.30.051606.094328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farovik A, Place RJ, Miller DR, Eichenbaum H. Amygdala lesions selectively impair familiarity in recognition memory. Nat Neurosci. 2011;14:1416–1417. doi: 10.1038/nn.2919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallia GL, Johnson EM, Khalili K. Puralpha: a multifunctional single-stranded DNA- and RNA-binding protein. Nucleic Acids Res. 2000;28:3197–3205. doi: 10.1093/nar/28.17.3197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelman BB. Neuropathology of HAND With Suppressive Antiretroviral Therapy: Encephalitis and Neurodegeneration Reconsidered. Curr HIV/AIDS Rep. 2015;12:272–279. doi: 10.1007/s11904-015-0266-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelman BB, Lisinicchia JG, Chen T, Johnson KM, Jennings K, Freeman DH, Jr, Soukup VM. Prefrontal dopaminergic and enkephalinergic synaptic accommodation in HIV-associated neurocognitive disorders and encephalitis. J Neuroimmune Pharmacol. 2012;7:686–700. doi: 10.1007/s11481-012-9345-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagerman RJ, Ono MY, Hagerman PJ. Recent advances in fragile X: a model for autism and neurodegeneration. Curr Opin Psychiatry. 2005;18:490–496. doi: 10.1097/01.yco.0000179485.39520.b0. [DOI] [PubMed] [Google Scholar]
- Heaton RK, Franklin DR, Ellis RJ, McCutchan JA, Letendre SL, Leblanc S, Corkran SH, Duarte NA, Clifford DB, Woods SP, Collier AC, Marra CM, Morgello S, Mindt MR, Taylor MJ, Marcotte TD, Atkinson JH, Wolfson T, Gelman BB, McArthur JC, Simpson DM, Abramson I, Gamst A, Fennema-Notestine C, Jernigan TL, Wong J, Grant I, Group C, Group H HIV-associated neurocognitive disorders before and during the era of combination antiretroviral therapy: differences in rates, nature, and predictors. J Neurovirol. 2011;17:3–16. doi: 10.1007/s13365-010-0006-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hokkanen S, Feldmann HM, Ding H, Jung CK, Bojarski L, Renner-Muller I, Schuller U, Kretzschmar H, Wolf E, Herms J. Lack of Pur-alpha alters postnatal brain development and causes megalencephaly. Hum Mol Genet. 2012;21:473–484. doi: 10.1093/hmg/ddr476. [DOI] [PubMed] [Google Scholar]
- Hunt D, Leventer RJ, Simons C, Taft R, Swoboda KJ, Gawne-Cain M, study DDD. Magee AC, Turnpenny PD, Baralle D. Whole exome sequencing in family trios reveals de novo mutations in PURA as a cause of severe neurodevelopmental delay and learning disability. J Med Genet. 2014;51:806–813. doi: 10.1136/jmedgenet-2014-102798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irwin S. Comprehensive observational assessment: Ia. A systematic, quantitative procedure for assessing the behavioral and physiologic state of the mouse. Psychopharmacologia. 1968;13:222–257. doi: 10.1007/BF00401402. [DOI] [PubMed] [Google Scholar]
- Ivanov SV, Ward JM, Tessarollo L, McAreavey D, Sachdev V, Fananapazir L, Banks MK, Morris N, Djurickovic D, Devor-Henneman DE, Wei MH, Alvord GW, Gao B, Richardson JA, Minna JD, Rogawski MA, Lerman MI. Cerebellar ataxia, seizures, premature death, and cardiac abnormalities in mice with targeted disruption of the Cacna2d2 gene. Am J Pathol. 2004;165:1007–1018. doi: 10.1016/S0002-9440(10)63362-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacquelin C, Strazielle C, Lalonde R. Neurologic function during developmental and adult stages in Dab1(scm) (scrambler) mutant mice. Behav Brain Res. 2012;226:265–273. doi: 10.1016/j.bbr.2011.09.020. [DOI] [PubMed] [Google Scholar]
- Jin P, Duan R, Qurashi A, Qin Y, Tian D, Rosser TC, Liu H, Feng Y, Warren ST. Pur alpha binds to rCGG repeats and modulates repeat-mediated neurodegeneration in a Drosophila model of fragile X tremor/ataxia syndrome. Neuron. 2007;55:556–564. doi: 10.1016/j.neuron.2007.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson EM. The Pur protein family: clues to function from recent studies on cancer and AIDS. Anticancer Res. 2003;23:2093–2100. [PubMed] [Google Scholar]
- Johnson EM, Daniel DC, Gordon J. The pur protein family: genetic and structural features in development and disease. J Cell Physiol. 2013;228:930–937. doi: 10.1002/jcp.24237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson EM, Kinoshita Y, Weinreb DB, Wortman MJ, Simon R, Khalili K, Winckler B, Gordon J. Role of Pur alpha in targeting mRNA to sites of translation in hippocampal neuronal dendrites. J Neurosci Res. 2006;83:929–943. doi: 10.1002/jnr.20806. [DOI] [PubMed] [Google Scholar]
- Kanai Y, Dohmae N, Hirokawa N. Kinesin transports RNA: isolation and characterization of an RNA-transporting granule. Neuron. 2004;43:513–525. doi: 10.1016/j.neuron.2004.07.022. [DOI] [PubMed] [Google Scholar]
- Khalili K, Del Valle L, Muralidharan V, Gault WJ, Darbinian N, Otte J, Meier E, Johnson EM, Daniel DC, Kinoshita Y, Amini S, Gordon J. Puralpha is essential for postnatal brain development and developmentally coupled cellular proliferation as revealed by genetic inactivation in the mouse. Mol Cell Biol. 2003;23:6857–6875. doi: 10.1128/MCB.23.19.6857-6875.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi S, Agui K, Kamo S, Li Y, Anzai K. Neural BC1 RNA associates with pur alpha, a single-stranded DNA and RNA binding protein, which is involved in the transcription of the BC1 RNA gene. Biochem Biophys Res Commun. 2000;277:341–347. doi: 10.1006/bbrc.2000.3683. [DOI] [PubMed] [Google Scholar]
- Lalani SR, Zhang J, Schaaf CP, Brown CW, Magoulas P, Tsai AC, El-Gharbawy A, Wierenga KJ, Bartholomew D, Fong CT, Barbaro-Dieber T, Kukolich MK, Burrage LC, Austin E, Keller K, Pastore M, Fernandez F, Lotze T, Wilfong A, Purcarin G, Zhu W, Craigen WJ, McGuire M, Jain M, Cooney E, Azamian M, Bainbridge MN, Muzny DM, Boerwinkle E, Person RE, Niu Z, Eng CM, Lupski JR, Gibbs RA, Beaudet AL, Yang Y, Wang MC, Xia F. Mutations in PURA cause profound neonatal hypotonia, seizures, and encephalopathy in 5q31.3 microdeletion syndrome. Am J Hum Genet. 2014;95:579–583. doi: 10.1016/j.ajhg.2014.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Koike K, Ohashi S, Funakoshi T, Tadano M, Kobayashi S, Anzai K, Shibata N, Kobayashi M. Pur alpha protein implicated in dendritic RNA transport interacts with ribosomes in neuronal cytoplasm. Biol Pharm Bull. 2001;24:231–235. doi: 10.1248/bpb.24.231. [DOI] [PubMed] [Google Scholar]
- Lieb K, Brucker S, Bach M, Els T, Lucking CH, Greenlee MW. Impairment in preattentive visual processing in patients with Parkinson’s disease. Brain. 1999;122(Pt 2):303–313. doi: 10.1093/brain/122.2.303. [DOI] [PubMed] [Google Scholar]
- Mishra M, Del Valle L, Otte J, Darbinian N, Gordon J. Pur-alpha regulates RhoA developmental expression and downstream signaling. J Cell Physiol. 2013;228:65–72. doi: 10.1002/jcp.24105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murai T, Okuda S, Tanaka T, Ohta H. Characteristics of object location memory in mice: Behavioral and pharmacological studies. Physiology & behavior. 2007;90:116–124. doi: 10.1016/j.physbeh.2006.09.013. [DOI] [PubMed] [Google Scholar]
- O’Leary TP, Brown RE. Optimization of apparatus design and behavioral measures for the assessment of visuo-spatial learning and memory of mice on the Barnes maze. Learn Mem. 2013;20:85–96. doi: 10.1101/lm.028076.112. [DOI] [PubMed] [Google Scholar]
- Ohashi S, Koike K, Omori A, Ichinose S, Ohara S, Kobayashi S, Sato TA, Anzai K. Identification of mRNA/protein (mRNP) complexes containing Puralpha, mStaufen, fragile X protein, and myosin Va and their association with rough endoplasmic reticulum equipped with a kinesin motor. J Biol Chem. 2002;277:37804–37810. doi: 10.1074/jbc.M203608200. [DOI] [PubMed] [Google Scholar]
- Paylor R, Spencer CM, Yuva-Paylor LA, Pieke-Dahl S. The use of behavioral test batteries, II: effect of test interval. Physiology & behavior. 2006;87:95–102. doi: 10.1016/j.physbeh.2005.09.002. [DOI] [PubMed] [Google Scholar]
- Rosenfeld CS, Ferguson SA. Barnes maze testing strategies with small and large rodent models. J Vis Exp. 2014:e51194. doi: 10.3791/51194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sereda M, Griffiths I, Puhlhofer A, Stewart H, Rossner MJ, Zimmerman F, Magyar JP, Schneider A, Hund E, Meinck HM, Suter U, Nave KA. A transgenic rat model of Charcot-Marie-Tooth disease. Neuron. 1996;16:1049–1060. doi: 10.1016/s0896-6273(00)80128-2. [DOI] [PubMed] [Google Scholar]
- Takeuchi E, Sato Y, Miura E, Yamaura H, Yuzaki M, Yanagihara D. Characteristics of gait ataxia in delta2 glutamate receptor mutant mice, ho15J. PLoS One. 2012;7:e47553. doi: 10.1371/journal.pone.0047553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka AJ, Bai R, Cho MT, Anyane-Yeboa K, Ahimaz P, Wilson AL, Kendall F, Hay B, Moss T, Nardini M, Bauer M, Retterer K, Juusola J, Chung WK. De novo mutations in PURA are associated with hypotonia and developmental delay. Cold Spring Harb Mol Case Stud. 2015;1:a000356. doi: 10.1101/mcs.a000356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trogrlic L, Wilson YM, Newman AG, Murphy M. Context fear learning specifically activates distinct populations of neurons in amygdala and hypothalamus. Learn Mem. 2011;18:678–687. doi: 10.1101/lm.2314311. [DOI] [PubMed] [Google Scholar]
- Wais PE. Hippocampal signals for strong memory when associative memory is available and when it is not. Hippocampus. 2011;21:9–21. doi: 10.1002/hipo.20716. [DOI] [PubMed] [Google Scholar]
- Wiseman FK, Al-Janabi T, Hardy J, Karmiloff-Smith A, Nizetic D, Tybulewicz VL, Fisher EM, Strydom A. A genetic cause of Alzheimer disease: mechanistic insights from Down syndrome. Nat Rev Neurosci. 2015;16:564–574. doi: 10.1038/nrn3983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng LH, Okamura K, Tanaka H, Miki N, Kuo CH. Concomitant translocation of Puralpha with its binding proteins (PurBPs) from nuclei to cytoplasm during neuronal development. Neurosci Res. 2005;51:105–109. doi: 10.1016/j.neures.2004.09.009. [DOI] [PubMed] [Google Scholar]


