Abstract
Emerging evidence suggests that glycemic variability may be a more reliable measure of glycemic control than mean HbA1c in type 2 diabetes mellitus. This study aimed to determine if HbA1c variability is associated with cardiovascular events in type 2 diabetic patients and if different renal functions affect such association. This longitudinal study enrolled 8259 diabetic patients from the Kaohsiung Medical University Research Database in 2009 and were followed-up until 2015. Intra-individual HbA1C variability was defined as the standard deviation (SD) of HbA1c and cardiovascular events were defined as hospitalization for coronary artery disease, unstable angina, myocardial infarction, stroke, peripheral artery disease, and cardiovascular death. The patients were grouped into two based on their estimated glomerular filtration rate (eGFR) ≥ 60 or < 60 min/ml/1.73m2. In a mean follow-up period of 6.3 years, cardiovascular events were recorded in 8.9% of the patients. In an adjusted Cox model, high HbA1c SD (hazard ratio, 1.290; 95% confidence interval, 1.008–1.650; p = 0.043), but not mean HbA1c, was associated with significantly increased risk of cardiovascular events in patients with eGFR ≥ 60 min/ml/1.73m2. This association was not seen in patients with eGFR < 60 min/ml/1.73m2. In this study, greater HbA1c variability is associated with increased risk of cardiovascular among patients with preserved renal function, but not in those with moderate to advanced chronic kidney disease.
Introduction
There is a growing number of people with diabetes mellitus (DM) worldwide. People with diabetes have a two- to three-fold increased risk of developing cardiovascular disease, which remains the major cause of death in such patients [1, 2]. Several studies have demonstrated the importance of time-averaged mean levels of glycemia, as measured by glycosylated hemoglobin (HbA1c), which has since been considered the “gold standard” for measuring glycemic control [3, 4]. However, recent research has pointed that plasma glucose variability, regardless of hyperglycemia level, may confer additional risk for the development of micro- and macrovascular diabetic complications [5–7].
Among studies on type 2 DM with preserved renal function, glucose variability is positively associated with the development of diabetic retinopathy, neuropathy, and nephropathy [8–11], but its effects on macrovascular events remain uncertain. Wan et al. evaluated the association of HbA1c variability with cardiovascular events in patients with type 2 DM and found that high variability was associated with increased cardiovascular events [12]. On the other hand, the cross-sectional analysis within the Italian Renal Insufficiency and Cardiovascular Events (RIACE) multi-center study showed no impact of HbA1c variability [11]. Thus, current literature is conflicting and limited, especially on different renal functions.
Even less is known on how glycemic control affects prognosis in diabetic patients with late stage chronic kidney disease (CKD), especially since these patients are often excluded from clinical trials. Few studies have evaluated the association of HbA1c variability with cardiovascular events in diabetic patients with moderate-to-advanced CKD. This study therefore aimed to assess whether HbA1c variability is associated with cardiovascular events in type 2 DM patients. The study also tested whether different renal functions affect such an association.
Subjects and methods
Setting
This study used data from the Kaohsiung Medical University Hospital Research Database (KMUHRD). Kaohsiung Medical University Hospital (KMUH), established in 1957, is a medical center with 1600 beds, 1200 general beds, 144 intensive care beds, and about 6000 clinical visits per day in 2015. The KMUHRD includes data for approximately 2,000,000 patients who attended KMUH in the period 2009–2015, with predominance from southern Taiwan.
The KMUHRD offers a comprehensive database with coverage on ambulatory care, hospital admissions, dental services, drug-dispensing records, and biochemical data. The hospital records include detailed information on primary and secondary diagnoses, procedures, and dates of hospital admission and discharge. All diagnoses are coded according to the International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM). The drug-dispensing histories contain data on the dispensed drug, prescriber type, dispensing date, dispensed amount, prescribed dose regimens, and duration of use (prescription length).
The database is managed by the KMUH Division of Medical Statistics and Bioinformatics. For confidentiality and in accordance with the Personal Information Protection Act, all personal identifiers were removed and the authorized researchers only performed data linkage, processing, and statistical analyses with specified computers in an independent 24-hr monitored room using encrypted identifiers. All data analysts were required to sign an agreement. Only tables and figures from statistical analysis were allowed to be released after inspection.
Study population
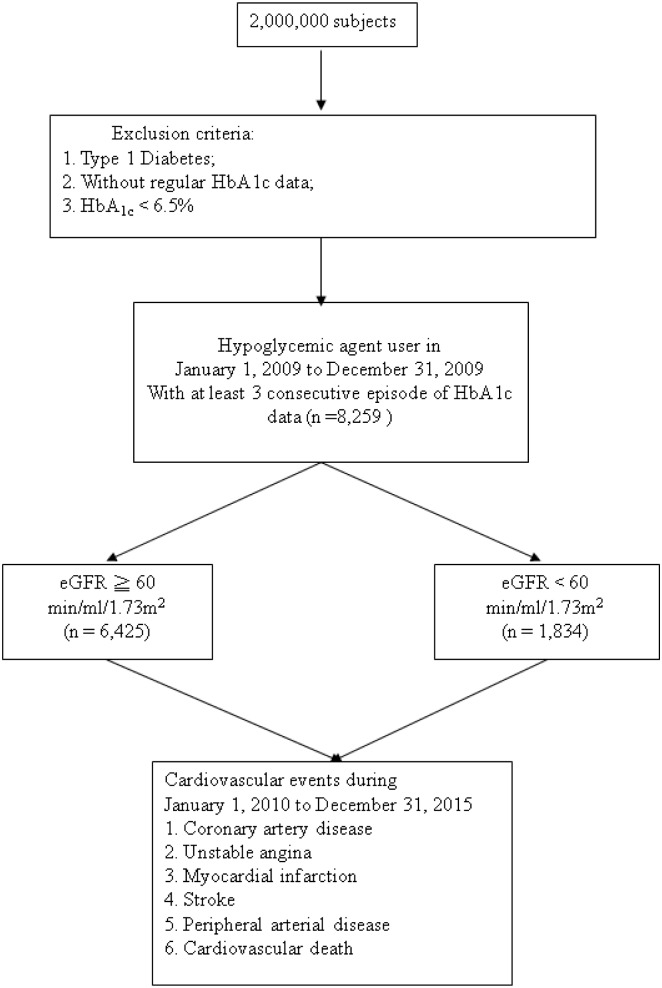
The entry date was between January 1, 2009 and December 31, 2009. All patients with type 2 diabetes (ICD-9-CM code 250.1–250.9) on hypoglycemic agents and with an HbA1c level of ≥ 6.5% were enrolled and followed-up from January 1, 2010 until December 31, 2015. Data on the occurrence of cardiovascular events, including coronary artery disease (ICD-9-CM code 414), myocardial infarction (ICD-9-CM code 410–412), ischemic stroke (ICD-9-CM code 435–438), and peripheral arterial disease (ICD-9-CM code 443 and 25070) on follow-up were also obtained from the KMUHRD. The exclusion criteria were type 1 diabetes, mortality, DM diagnosis of after a cardiovascular event, use of insulin dependent and independent agents, and HbA1c < 6.5%. The study patients were then stratified into two groups based on their estimated glomerular filtration rate (eGFR) ≥ 60 and < 60 min/ml/1.73m2 (Fig 1). We classified our patients with evidence of kidney damage lasting for more than 3 months into CKD stage 3, 4, and 5, based on estimated eGFR level (mL/min/1.73 m2) of 30 to 59, 15 to 29, and < 15, respectively.
Fig 1. Flow chart of study participants for the study to evaluate the effect of SD of HbA1c on cardiovascular events in type 2 diabetes.
The data recorded before entry into the study were age, sex, duration of diabetes, hypertension (ICD-9-CM code 401 and 405), hyperlipidemia (ICD-9-CM code 272), nephropathy (ICD-9-CM code 580–589), retinopathy (ICD-9-CM code 250.50), and neuropathy (ICD-9-CM code 249.60 and 250.60). Urine albumin and creatinine were measured on a spot urine sample by an autoanalyzer (COBAS Integra 400 plus; Roche Diagnostics, North America) and nephropathy was defined as the ratio of urine albumin to creatinine of ≥ 30 mg/g.
Definition of study endpoint
The study endpoint was defined as cardiovascular events, including coronary artery disease, stroke, peripheral arterial disease, and cardiovascular death. Coronary artery disease was defined as a history of angina and ischemic electrocardiogram change, ST-elevation myocardial infarction, non ST-elevation myocardial infarction, unstable angina or having undergone coronary bypass surgery or angioplasty.
Ethics statement
The study protocol was approved by the Institutional Review Board of Kaohsiung Medical University Hospital (KMUHIRB-E(I)-20160032). Written informed consent was obtained from the patients, and all clinical investigations were conducted according to the principles expressed in the Declaration of Helsinki. The patients also consented to the publication of the clinical details.
HbA1c variability
The intra-personal mean and standard deviation (SD) of HbA1c was calculated for each patient, and the latter was considered an index of HbA1c variability. The estimated HbA1c variability was based on at least three HbA1c measurements.
Statistical analysis
All statistical analyses were performed using the SPSS 19.0 for Windows (SPSS Inc. Chicago, USA). Data were expressed as percentages or mean ± standard deviation. Between-group differences were assessed using the chi-square test for categorical variables and the independent t-test for continuous variables. The association between HbA1c variability and cardiovascular events was assessed using multivariate Cox proportional hazard analysis. Age, sex, and significant variables in the univariate analysis were used in the multivariate analysis. The Kaplan-Meier analysis was used to determine the survival curve for cardiovascular events. Statistical significance was set at p<0.05.
Results
Comparisons of baseline data between patients with eGFR ≥ and those with < 60 min/ml/1.73m2 were show (Table 1). The patients’ mean age was 62.0 ± 11.9 years. Compared to patients with eGFR ≥ 60 min/ml/1.73m2, patients with eGFR < 60 min/ml/1.73m2 were older and had higher prevalence rates of hypertension, dyslipidemia, diabetic retinopathy, neuropathy, and nephropathy. They had higher HbA1c SD, triglyceride, and total cholesterol levels, but lower mean HbA1c, high-density lipoprotein (HDL) cholesterol, and eGFR. They also had higher percentage use of angiotensin converting enzyme inhibitors (ACEIs) and/or angiotensin II receptor blockers (ARBs), aspirin, and insulin. In addition, patients with eGFR < 60 min/ml/1.73m2 had less follow-up days and a higher frequency of cardiovascular events (p < 0.001).
Table 1. Comparison of clinical characteristics according to eGFR ≧ or < 60 min/ml/1.73m2.
| Characteristics | eGFR ≧ 60 min/ml/1.73m2 (n = 6425) | eGFR < 60 min/ml/1.73m2 (n = 1834) | All (n = 8259) | p |
|---|---|---|---|---|
| Age (year) | 60.3 ± 11.4 | 68.6 ± 10.9 | 62.0 ± 11.9 | < 0.001 |
| Male gender (%) | 51.5 | 54.0 | 52.0 | 0.056 |
| Hypertension (%) | 69.4 | 84.9 | 72.8 | < 0.001 |
| Dyslipidemia (%) | 71.4 | 61.8 | 69.3 | < 0.001 |
| Retinopathy (%) | 3.6 | 11.4 | 5.3 | < 0.001 |
| Neuropathy (%) | 12.2 | 17.4 | 13.4 | < 0.001 |
| Nephropathy (%) | 3.9 | 10.7 | 5.4 | < 0.001 |
| DM duration > 5 years (%) | 91.7 | 92.6 | 91.9 | 0.215 |
| Laboratory parameters | ||||
| SD HbA1C (%) | 0.81 ± 0.56 | 0.93 ± 0.65 | 0.84 ± 0.58 | < 0.001 |
| Mean HbA1C (%) | 7.5 ± 1.2 | 7.4 ± 1.2 | 7.5 ± 1.2 | 0.017 |
| Triglyceride (mg/dL) | 146.7 ± 136.8 | 161.6 ± 114.3 | 154.7 ± 206.0 | < 0.001 |
| Total cholesterol (mg/dL) | w176.8 ± 41.0 | 181.0 ± 48.1 | 178.2 ± 44.9 | 0.001 |
| HDL-cholesterol (mg/dL) | 41.3 ± 12.4 | 38.6 ± 12.4 | 40.7 ± 12.5 | < 0.001 |
| LDL-cholesterol (mg/dL) | 102.8 ± 33.1 | 102.2 ± 36.3 | 102.6 ± 34.0 | 0.578 |
| eGFR (mL/min/1.73 m2) | 96.0 ± 23.5 | 39.9 ± 14.8 | 84.5 ± 35.4 | < 0.001 |
| Medications | ||||
| ACEI and/or ARB use (%) | 62.8 | 80.5 | 66.6 | < 0.001 |
| Aspirin use (%) | 29.1 | 42.7 | 32.1 | < 0.001 |
| Statin and/or fibrate use (%) | 69.6 | 70.8 | 69.9 | 0.341 |
| Insulin use (%) | 18.1 | 35.3 | 22.1 | < 0.001 |
| Days of follow-up (days) | 2330.4 ± 426.7 | 2165.6 ± 650.1 | 2294.3 ± 489.1 | < 0.001 |
| CV events (%) | 6.5 | 17.6 | 8.9 | < 0.001 |
Abbreviations. eGFR, estimated glomerular filtration rate; DM, diabetes mellitus; SD, standard deviation; HDL, high-density lipoprotein; LDL, low-density lipoprotein; ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin II receptor blocker; CV, cardiovascular.
In a mean follow-up period of 6.3 years, cardiovascular events were recorded in 8.9% of the patients. These included hospitalization for coronary artery disease (n = 876), stroke (n = 100), peripheral artery disease (n = 54), and cardiovascular death (n = 27).
Risk of cardiovascular events in patients with eGFR ≥ 60 min/ml/1.73m2
Cox proportional hazard regression analysis was used to evaluate the association between SD of HbA1c and cardiovascular events in patients with eGFR ≥ 60 min/ml/1.73m2 (Table 2). By univariate regression analysis, old age, male sex, hypertension, diabetic retinopathy and neuropathy, high HbA1c SD, high mean HbA1c, high triglyceride, low HDL-cholesterol, low eGFR, ACEI and/or ARB use, aspirin use, statin and/or fibrate use, and insulin use were associated with significantly higher risk of cardiovascular events.
Table 2. Risk factors for cardiovascular events using Cox proportional hazards model in patients with eGFR ≧ 60 min/ml/1.73m2.
| Univariable | Multivariable | |||
|---|---|---|---|---|
| Parameters | HR (95% CI) | p | HR (95% CI) | p |
| Age (per 1 year) | 1.044 (1.035–1.053) | < 0.001 | 1.043 (1.030–1.057) | < 0.001 |
| Male gender | 1.288 (1.063–1.561) | 0.010 | 1.427 (1.112–1.832) | 0.005 |
| Hypertension | 1.829 (1.441–2.322) | < 0.001 | 1.010 (0.744–1.372) | 0.947 |
| Dyslipidemia | 1.167 (0.939–1.451) | 0.164 | – | – |
| Retinopathy | 1.739 (1.170–2.586) | 0.006 | 1.136 (0.636–2.028) | 0.666 |
| Neuropathy | 1.462 (1.133–1.887) | 0.003 | 0.988 (0.703–1.388) | 0.945 |
| Nephropathy | 0.902 (0.539–1.510) | 0.694 | – | – |
| DM duration > 5 years | 1.167 (0.805–1.691) | 0.415 | – | – |
| Laboratory parameters | ||||
| SD HbA1C (per 1%) | 1.427 (1.242–1.640) | < 0.001 | 1.290 (1.008–1.650) | 0.043 |
| Mean HbA1C (per 1%) | 1.115 (1.036–1.199) | 0.004 | 0.941 (0.825–1.074) | 0.365 |
| Triglyceride (per 1 mg/dL) | 1.001 (1.001–1.001) | < 0.001 | 1.001 (1.000–1.001) | 0.010 |
| Total cholesterol (per 1 mg/dL) | 1.001 (0.998–1.003) | 0.499 | – | – |
| HDL-cholesterol (per 1 mg/dL) | 0.983 (0.973–0.993) | 0.001 | 0.992 (0.982–1.004) | 0.182 |
| LDL-cholesterol (per 1 mg/dL) | 0.998 (0.995–1.001) | 0.281 | – | – |
| eGFR (per 1 mL/min/1.73 m2) | 0.986 (0.982–0.991) | < 0.001 | 0.999 (0.993–1.005) | 0.775 |
| Medications | ||||
| ACEI and/or ARB use | 2.483 (1.956–3.152) | < 0.001 | 1.818 (1.316–2.510) | < 0.001 |
| Aspirin use | 4.701 (3.856–5.732) | < 0.001 | 3.716 (2.893–4.773) | < 0.001 |
| Statin and/or fibrate use | 1.861 (1.462–2.367) | < 0.001 | 1.623 (1.172–2.247) | 0.004 |
| Insulin use | 1.757 (1.421–2.174) | < 0.001 | 1.342 (0.995–1.811) | 0.054 |
Values expressed as Hazard Ratios and 95% confidence interval (CI).
By multivariate analysis, old age (hazard ratio [HR], 1.043; 95% confidence interval [CI], 1.030–1.057; p < 0.001), male sex (HR, 1.427; 95% CI, 1.112–1.832; p = 0.005), high HbA1c SD (HR, 1.290; 95% CI, 1.008–1.650; p = 0.043), high triglyceride (HR, 1.001; 95% CI, 1.000–1.001; p = 0.010), ACEI and/or ARB use (HR, 1.818; 95% CI, 1.316–2.510; p < 0.001), aspirin use (HR, 3.716; 95% CI, 2.893–4.773; p < 0.001), and statin and/or fibrate use (HR, 1.623; 95% CI, 1.172–2.247; p = 0.004) were associated with significantly increased risk of cardiovascular events.
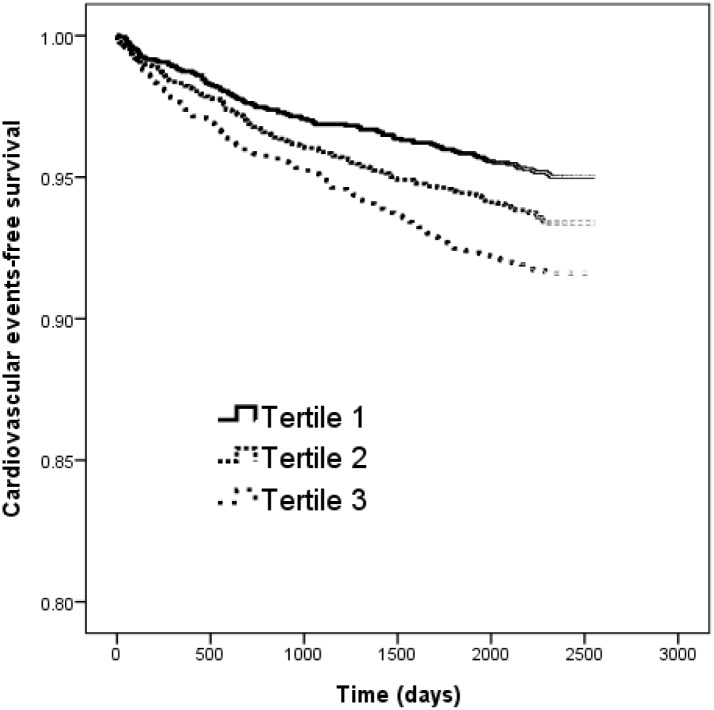
The Kaplan-Meier curves for cardiovascular event-free survival (log-rank p < 0.001) of study patients with eGFR ≥ 60 min/ml/1.73m2 according to tertiles of SD of HbA1C (<0.48 and ≥ 0.48 to < 0.90 and ≥ 0.90%) revealed that tertile 2 (HR, 1.341; 95% CI, 1.047–1.705; p = 0.023) and tertile 3 (HR, 1.724; 95% CI, 1.355–2.191; p < 0.001) patients had worse cardiovascular events-free survival than those in tertile 1 (Fig 2).
Fig 2. Kaplan-Meier analysis of cardiovascular events-free survival according to tertile of SD of HbA1c (log-rank p < 0.001) in patients with eGFR ≧ 60 min/ml/1.73m2.
Diabetic patients with tertile 2 and tertile 3 of HbA1c SD had a worse cardiovascular events-free survival than those with tertile 1 of HbA1c SD.
We further performed the analysis of relation of SD of HbA1C for separate cardiovascular events in patients with eGFR ≧ 60 min/ml/1.73m2, and found that high HbA1c SD (HR, 1.353; 95% CI, 1.024–1.787; p = 0.033) was still associated with significantly increased risk of coronary artery disease after multivariate analysis (Table 3).
Table 3. Relation of SD of HbA1C for separate cardiovascular events using Cox proportional hazards model in patients with eGFR ≧ 60 min/ml/1.73m2.
| Univariable | Multivariable | |||
|---|---|---|---|---|
| Parameters | HR (95% CI) | p | HR (95% CI) | p |
| Coronary artery disease | 1.415 (1.212–1.652) | < 0.001 | 1.353 (1.024–1.787) | 0.033 |
| Stroke | 1.140 (0.781–1.664) | 0.496 | 1.606 (0.894–2.887) | 0.113 |
| Peripheral artery disease | 2.054 (1.326–3.182) | 0.001 | 1.253 (0.477–3.296) | 0.647 |
Values expressed as Hazard Ratios and 95% confidence interval (CI). Adjusted for age, gender, a history of hypertension, retinopathy and neuropathy, SD of HbA1C, mean HbA1C, triglyceride, HDL-cholesterol, eGFR, and medications use, including ACEI and/or ARB, aspirin, statin and/or fibrate, and insulin.
Risk of cardiovascular events in patients with eGFR < 60 min/ml/1.73m2
The Cox proportional hazard regression was used to evaluate the association between SD of HbA1c and cardiovascular events in patients with eGFR < 60 min/ml/1.73m2 (Table 4). By univariate regression analysis, old age, female sex, hypertension, diabetic retinopathy and nephropathy, high HbA1c SD, high triglyceride, low HDL-cholesterol, low eGFR, ACEI and/or ARB use, aspirin use, statin and/or fibrate use, and insulin use were associated with significantly greater risk of cardiovascular events.
Table 4. Risk factors for cardiovascular events using Cox proportional hazards model in patients with eGFR < 60 min/ml/1.73m2.
| Univariable | Multivariable | |||
|---|---|---|---|---|
| Parameters | HR (95% CI) | p | HR (95% CI) | p |
| Age (per 1 year) | 1.009 (0.998–1.019) | 0.102 | 1.007 (0.992–1.022) | 0.378 |
| Male gender | 0.858 (0.687–1.070) | 0.174 | 0.729 (0.534–0.994) | 0.046 |
| Hypertension | 1.825 (1.246–2.675) | 0.002 | 1.613 (0.889–2.925) | 0.115 |
| Dyslipidemia | 1.146 (0.909–1.445) | 0.249 | – | – |
| Retinopathy | 1.799 (1.344–2.406) | < 0.001 | 1.420 (0.915–2.203) | 0.118 |
| Neuropathy | 0.999 (0.745–1.339) | 0.995 | – | – |
| Nephropathy | 0.590 (1.169–2.162) | 0.003 | 1.294 (0.818–2.045) | 0.271 |
| DM duration > 5 years | 0.826 (0.553–1.235) | 0.352 | – | – |
| Laboratory parameters | ||||
| SD HbA1C (per 1%) | 1.211 (1.041–1.410) | 0.013 | 1.014 (0.803–1.280) | 0.909 |
| Mean HbA1C (per 1%) | 1.071 (0.978–1.173) | 0.140 | – | – |
| Triglyceride (per 1 mg/dL) | 1.001 (1.001–1.002) | 0.001 | 1.000 (0.999–1.002) | 0.648 |
| Total cholesterol (per 1 mg/dL) | 1.001 (0.998–1.003) | 0.540 | – | – |
| HDL-cholesterol (per 1 mg/dL) | 0.981 (0.968–0.994) | 0.004 | 0.977 (0.964–0.992) | 0.002 |
| LDL-cholesterol (per 1 mg/dL) | 1.000 (0.996–1.003) | 0.810 | – | – |
| eGFR (per 1 mL/min/1.73 m2) | 0.976 (0.969–0.983) | < 0.001 | 0.983 (0.972–0.993) | 0.001 |
| Medications | ||||
| ACEI and/or ARB use | 1.562 (1.132–2.155) | 0.007 | 1.584 (0.998–2.516) | 0.051 |
| Aspirin use | 2.455 (1.953–3.085) | < 0.001 | 2.882 (2.081–3.990) | < 0.001 |
| Statin and/or fibrate use | 1.493 (1.145–1.948) | 0.003 | 1.342 (0.890–2.023) | 0.160 |
| Insulin use | 1.885 (1.510–2.352) | < 0.001 | 1.417 (1.020–1.969) | 0.038 |
Values expressed as Hazard Ratios and 95% confidence interval (CI).
In the multivariate analysis, female sex (HR, 0.729; 95% CI, 0.534–0.994; p = 0.046), low HDL-cholesterol (HR, 0.977; 95% CI, 0.964–0.992; p = 0.002), low eGFR (HR, 0.983; 95% CI, 0.972–0.993; p = 0.001), aspirin use (HR, 2.882; 95% CI, 2.081–3.990; p < 0.001), and insulin use (HR, 1.417; 95% CI, 1.020–1.969; p = 0.038) were associated with significantly higher risk of cardiovascular events. However, HbA1c SD was not.
We further performed the analysis of relation of SD of HbA1C for cardiovascular events in patients with different CKD stage, and found that no significant association was noted in each CKD stage after multivariate analysis (Table 5).
Table 5. Relation of SD of HbA1C for cardiovascular events using Cox proportional hazards model in patients with different CKD stage.
| Univariable | Multivariable | |||
|---|---|---|---|---|
| Parameters | HR (95% CI) | p | HR (95% CI) | p |
| CKD stage 3 | 1.323 (1.085–1.614) | 0.006 | 1.086 (0.800–1.474) | 0.598 |
| CKD stage 4 | 0.977 (0.711–1.363) | 0.892 | 1.215 (0.782–1.888) | 0.387 |
| CKD stage 5 | 1.086 (0.804–1.466) | 0.591 | 1.619 (0.902–2.908) | 0.107 |
Values expressed as Hazard Ratios and 95% confidence interval (CI). Adjusted for age, gender, a history of hypertension, retinopathy and nephropathy, SD of HbA1C, triglyceride, HDL-cholesterol, eGFR, and medications use, including ACEI and/or ARB, aspirin, statin and/or fibrate, and insulin.
Discussion
This study investigated the association between HbA1c variability and cardiovascular events in patients with type 2 DM over a follow-up period of 6.3 years. The results show that patients with greater HbA1c variability have increased risk of cardiovascular events in diabetic patients with eGFR ≥ 60 min/ml/1.73m2, but not in those with eGFR < 60 min/ml/1.73m2.
The first important finding here is that among diabetic patients with higher eGFR, greater HbA1c variability is associated with increased risk for cardiovascular events, independent of mean HbA1c levels. In a subsequent post hoc analysis, the Diabetes Control and Complications Trial (DCCT) data investigated the relationship between the risk of progression of retinopathy and HbA1c levels after 5 to 9 years of follow-up [13]. Their results revealed that the patients who received intensive treatment had a significantly lower risk of the progression of retinopathy than those with conventional treatment despite comparable HbA1c levels. The authors concluded that the mean level of HbA1c may not be the key factor determining the degree of hyperglycemia, and that other risk factors independent of HbA1c level may increase the risk of complications from diabetes due to additional metabolic burden [13]. Such findings may be partly due to the fact that studies have used HbA1c as a marker of glucose control, which fails to reflect glucose variability and the risk associated with extreme glucose change over an extended time period. The association between HbA1c variability and the development of cardiovascular events may be due to several mechanisms. In an in vitro study, Risso et al. reported that fluctuations in glycemia have a more harmful effect on endothelial cells than a persistently high level of glucose. They also found that apoptosis was significantly higher in human umbilical vein endothelial cells incubated in media in which the glucose concentration was alternately set at 5 or 20 mmol/l every other day for 14 days compared to a fixed high concentration of glucose [14]. Furthermore, intermittently high glucose levels enhance the formation of markers of oxidative stress, leading to increased cellular apoptosis [15]. In an in vivo study, Horva´th et al. reported that rats intermittently treated with glucose had a significantly worse endothelial function than those that were untreated despite a lower total exposure to glucose. Furthermore, the authors also hypothesized that the poly(ADP-ribose) polymerase pathway had been activated [16]. Human studies also showed that acute glucose fluctuations strongly correlated with triggering oxidative stress, urinary levels of 8-iso PGF2α, and more endothelial function damage leading to higher levels of oxidative stress markers than constantly high glucose [17, 18]. Some studies reveal that glucose variability is associated with the risk of hypoglycemia [19, 20], which, in turn, may contribute to increased progression of cardiovascular disease and mortality by inducing inflammation, blood coagulation abnormality, sympatho-adrenal response, and endothelial dysfunction [21, 22]. In the present study, the impact of HbA1c variability on the risks of cardiovascular events in diabetic patients with preserved renal function remained significant after adjustments in the mean HbA1c. This suggests that HbA1c variability may provide additional valuable information as a potential predictor of adverse cardiovascular outcomes.
Another important finding here is that the significant association between HbA1c variability and cardiovascular events is not observed in patients with moderate-to-advanced CKD. This implies that the predictor power of HbA1c variability on the risk of cardiovascular events among patients with CKD may be relatively lower. The prognostic role of HbA1c variability in such patients is unclear because of impaired glucose metabolism in CKD, while the HbA1c level may be altered by anemia or by the use of an erythropoiesis-stimulating agent. In patients with CKD, HbA1c formation is reduced because the fragile red blood cell (RBC) has a shortened lifespan by 30–70% and carbamylated hemoglobin molecules in a uremic environment become resistant to glycosylation [23]. Young RBCs have a lower glycosylation rate compared to old RBCs, thereby altering HbA1c formation [24]. Thus, although aggressive glycemic control may be beneficial for early cardiovascular disease prevention, there is paucity of data on outcomes supporting tight glycemic control in patients with CKD and HbA1c variability may be a more useful indicator in preserved renal function for predicting clinical outcomes [25].
This study has several limitations. First, as an observational study, the number and frequency of HbA1c measurement varied between individual patients. To minimize this, patients with fewer than three HbA1c measurements during the follow-up period and those with a follow-up period shorter than 6 months were excluded. Second, we used DM duration > 5 years instead of continuous diabetes duration. The diabetes duration were always underestimated in the case of diabetes due to underdiagnosis. Referred to most of the studies, categorical division was used for DM duration and the diabetes complications were started as early as 5 years after the diagnosis of diabetes. So we preferred to follow most of the studies reported by using categorical division of ≦ and > 5 years in diabetes duration. In addition, post hoc analysis of the Action to Control Cardiovascular Risk in Diabetes (ACCORD) [26] and other trials suggest that cardiac autonomic neuropathy may be a predisposing factor to cardiovascular events. However, the patients here were not evaluated for heart rate variability and as such, the effect of cardiac autonomic neuropathy on cardiovascular events could not be assessed. Nonetheless, a diagnosis of diabetic neuropathy according to the ICD-9-CM code is included in the analysis. Lastly, the effect of medications on cardiovascular events was not evaluated because this study was not a clinical trial aimed at investigating the effects of medication. There is insufficient data on cumulative exposure duration and defined daily dose. In this study, the positive correlation between medications use and cardiovascular events may be due to selection bias.
In conclusion, greater HbA1c variability is associated with increased risk of cardiovascular among patients with eGFR ≥ 60 min/ml/1.73m2, but not in patients with eGFR < 60 min/ml/1.73m2. These findings support the potential role of glucose variability, characterized by SD of HbA1c, as a predictor of cardiovascular events, independent of mean HbA1c level, in patients with type 2 DM with preserved renal function. Clinicians should be cautious about fluctuations along with the direction of changes for preventing the development of adverse cardiovascular outcomes.
Supporting information
(XLSX)
Data Availability
The data underlying this study is from the Kaohsiung Medical University Hospital Research Database (KMUHRD). Due to restrictions placed on the data by the Personal Information Protection Act of Taiwan, the minimal data set cannot be made publicly available. Data may be available upon request to interested researchers. Please send data requests to: Yi-Hsin Yang, PhD, Director, Division of Medical Statistics and Bioinformatics, Department of Medical Research, Kaohsiung Medical University Hospital, Taiwan. Email: yihsya@kmu.edu.tw.
Funding Statement
The authors received no specific funding for this work.
References
- 1.Seshasai SR, Kaptoge S, Thompson A, Di Angelantonio E, Gao P, Sarwar N, et al. Diabetes mellitus, fasting glucose, and risk of cause-specific death. New Engl J Med 2011;364:829–841. 10.1056/NEJMoa1008862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sarwar N, Gao P, Seshasai SR, Gobin R, Kaptoge S, Di Angelantonio E, et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: A collaborative meta-analysis of 102 prospective studies. Lancet 2010;375:2215–2222. 10.1016/S0140-6736(10)60484-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hanefeld M. Postprandial hyperglycaemia: Noxious effects on the vessel wall. Int J Clin Pract 2002;Suppl 129:45–50. [PubMed] [Google Scholar]
- 4.Stratton IM, Adler AI, Neil HA, Matthews DR, Manley SE, Cull CA, et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (ukpds 35): Prospective observational study. BMJ 2000;321:405–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hirsch IB, Brownlee M. Should minimal blood glucose variability become the gold standard of glycemic control? J Diabetes Complications 2005;19:178–181. 10.1016/j.jdiacomp.2004.10.001 [DOI] [PubMed] [Google Scholar]
- 6.Brownlee M, Hirsch IB. Glycemic variability: A hemoglobin a1c-independent risk factor for diabetic complications. JAMA 2006;295:1707–1708. 10.1001/jama.295.14.1707 [DOI] [PubMed] [Google Scholar]
- 7.Hanefeld M, Temelkova-Kurktschiev T. Control of post-prandial hyperglycemia—an essential part of good diabetes treatment and prevention of cardiovascular complications. Nutr Metab Cardiovasc Dis 2002;12:98–107. [PubMed] [Google Scholar]
- 8.Jun JE, Jin SM, Baek J, Oh S, Hur KY, Lee MS, et al. The association between glycemic variability and diabetic cardiovascular autonomic neuropathy in patients with type 2 diabetes. Cardiovasc Diabetol 2015;14:70 10.1186/s12933-015-0233-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sugawara A, Kawai K, Motohashi S, Saito K, Kodama S, Yachi Y, et al. Hba(1c) variability and the development of microalbuminuria in type 2 diabetes: Tsukuba kawai diabetes registry 2. Diabetologia 2012;55:2128–2131. 10.1007/s00125-012-2572-7 [DOI] [PubMed] [Google Scholar]
- 10.Lin CC, Chen CC, Chen FN, Li CI, Liu CS, Lin WY, et al. Risks of diabetic nephropathy with variation in hemoglobin a1c and fasting plasma glucose. Am J Med 2013;126:1017 e1–10. [DOI] [PubMed] [Google Scholar]
- 11.Hsu CR, Chen YT, Sheu WH. Glycemic variability and diabetes retinopathy: A missing link. J Diabetes Complications 2015;29:302–306. 10.1016/j.jdiacomp.2014.11.013 [DOI] [PubMed] [Google Scholar]
- 12.Wan EY, Fung CS, Fong DY, Lam CL. Association of variability in hemoglobin a1c with cardiovascular diseases and mortality in chinese patients with type 2 diabetes mellitus—a retrospective population-based cohort study. J Diabetes Complications 2016;30:1240–1247. 10.1016/j.jdiacomp.2016.05.024 [DOI] [PubMed] [Google Scholar]
- 13.Lachin JM, Genuth S, Nathan DM, Zinman B, Rutledge BN. Effect of glycemic exposure on the risk of microvascular complications in the diabetes control and complications trial—revisited. Diabetes 2008;57:995–1001. 10.2337/db07-1618 [DOI] [PubMed] [Google Scholar]
- 14.Risso A, Mercuri F, Quagliaro L, Damante G, Ceriello A. Intermittent high glucose enhances apoptosis in human umbilical vein endothelial cells in culture. Am J Physiol Endocrinol Metab 2001;281:E924–930. [DOI] [PubMed] [Google Scholar]
- 15.Quagliaro L, Piconi L, Assaloni R, Martinelli L, Motz E, Ceriello A. Intermittent high glucose enhances apoptosis related to oxidative stress in human umbilical vein endothelial cells: The role of protein kinase c and nad(p)h-oxidase activation. Diabetes 2003;52:2795–2804. [DOI] [PubMed] [Google Scholar]
- 16.Horvath EM, Benko R, Kiss L, Muranyi M, Pek T, Fekete K, et al. Rapid 'glycaemic swings' induce nitrosative stress, activate poly(ADP-ribose) polymerase and impair endothelial function in a rat model of diabetes mellitus. Diabetologia 2009;52:952–961. 10.1007/s00125-009-1304-0 [DOI] [PubMed] [Google Scholar]
- 17.Monnier L, Mas E, Ginet C, Michel F, Villon L, Cristol JP, et al. Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA 2006;295:1681–1687. 10.1001/jama.295.14.1681 [DOI] [PubMed] [Google Scholar]
- 18.Ceriello A, Esposito K, Piconi L, Ihnat MA, Thorpe JE, Testa R, et al. Oscillating glucose is more deleterious to endothelial function and oxidative stress than mean glucose in normal and type 2 diabetic patients. Diabetes 2008;57:1349–1354. 10.2337/db08-0063 [DOI] [PubMed] [Google Scholar]
- 19.Murata GH, Hoffman RM, Shah JH, Wendel CS, Duckworth WC. A probabilistic model for predicting hypoglycemia in type 2 diabetes mellitus: The Diabetes Outcomes in Veterans Study (DOVES). Arch Intern Med 2004;164:1445–1450. 10.1001/archinte.164.13.1445 [DOI] [PubMed] [Google Scholar]
- 20.Skyler JS, Bergenstal R, Bonow RO, Buse J, Deedwania P, Gale EA, et al. Intensive glycemic control and the prevention of cardiovascular events: Implications of the accord, advance, and va diabetes trials: A position statement of the american diabetes association and a scientific statement of the american college of cardiology foundation and the american heart association. J Am Coll Cardiol 2009;53:298–304. [DOI] [PubMed] [Google Scholar]
- 21.Desouza CV, Bolli GB, Fonseca V. Hypoglycemia, diabetes, and cardiovascular events. Diabetes Care 2010;33:1389–1394. 10.2337/dc09-2082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Saisho Y. Glycemic variability and oxidative stress: A link between diabetes and cardiovascular disease? Int J Mol Sci 2014;15:18381–18406. 10.3390/ijms151018381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ly J, Marticorena R, Donnelly S. Red blood cell survival in chronic renal failure. Am J Kidney Dis 2004;44:715–719. [PubMed] [Google Scholar]
- 24.Uzu T, Hatta T, Deji N, Izumiya T, Ueda H, Miyazawa I, et al. Target for glycemic control in type 2 diabetic patients on hemodialysis: Effects of anemia and erythropoietin injection on hemoglobin a(1c). Ther Apher Dial 2009;13:89–94. 10.1111/j.1744-9987.2009.00661.x [DOI] [PubMed] [Google Scholar]
- 25.Menon V, Sarnak MJ. The epidemiology of chronic kidney disease stages 1 to 4 and cardiovascular disease: A high-risk combination. Am J Kidney Dis 2005;45:223–232. [DOI] [PubMed] [Google Scholar]
- 26.Gerstein HC, Miller ME, Genuth S, Ismail-Beigi F, Buse JB, Goff DC Jr, et al. Long-term effects of intensive glucose lowering on cardiovascular outcomes. New Engl J Med 2011;364:818–828. 10.1056/NEJMoa1006524 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(XLSX)
Data Availability Statement
The data underlying this study is from the Kaohsiung Medical University Hospital Research Database (KMUHRD). Due to restrictions placed on the data by the Personal Information Protection Act of Taiwan, the minimal data set cannot be made publicly available. Data may be available upon request to interested researchers. Please send data requests to: Yi-Hsin Yang, PhD, Director, Division of Medical Statistics and Bioinformatics, Department of Medical Research, Kaohsiung Medical University Hospital, Taiwan. Email: yihsya@kmu.edu.tw.