Abstract
Allogeneic hematopoietic cell transplantation (HCT) from siblings or unrelated donors (URD) during complete remission (CR) may improve leukemia-free survival (LFS) in FLT3+ acute myeloid leukemia (AML) that has poor prognosis due to high relapse rates. Umbilical cord blood (UCB) HCT outcomes are largely unknown in this population. We found that compared with sibling HCT, relapse risks were similar after UCB (n=126), (HR 0.86, p=0.54) and URD (n=91) (HR 0.81, p=0.43). UCB HCT was associated with statistically higher non-relapse mortality compared with sibling HCT (HR 2.32, p=0.02), but not vs. URD (HR 1.72, p=0.07). All three cohorts had statistically not significant 3-year LFS: 39% (95% CI 30–47) after UCB, 43% (95% CI 30–54) after sibling, and 50% (95% CI 40–60) after URD. Chronic GVHD rates were significantly lower after UCB compared with either sibling (HR 0.59, p=0.03) or URD (HR 0.49, p=0.001). Adverse factors for LFS included high leukocyte count at diagnosis and HCT during CR2. UCB is a suitable option for adults with FLT3+AML in the absence of an HLA-matched sibling and its immediate availability may be particularly important for FLT3+ AML where early relapse is common thus allowing HCT in CR1 when outcomes are best.
Keywords: AML, FLT3, umbilical cord blood HCT, unrelated donor, sibling, relapse, survival
Introduction
FMS-like tyrosine kinase (FLT3), a receptor tyrosine kinase (TK), is present in early hematopoietic progenitors and influences the survival, proliferation and differentiation of hematopoietic cells. Mutation in the FLT3 gene (FLT3+) has been reported in acute myeloid leukemia (AML). The internal tandem duplication (FLT3-ITD, 15% to 35%) and missense point mutations (5% to 10%) in the TK domain (TKD) are the most commonly detected mutations in the FLT3 gene.(1, 2) These mutations confer ligand-independent constitutive activation of the FLT3 kinase and its downstream signaling pathway which stimulates AML cell proliferation.(3) Patients with FLT3+ AML share clinical, cytogenetic, and molecular common features at diagnosis, typically presenting with high white blood cell counts (WBC), normal cytogenetics, presence of the nucleophosmin (NPM1) gene mutation, and FAB subtypes M4 and M5.(1) However, the prognosis of patients with FLT3+ AML is poor mainly due to frequent and early relapse in both adult and pediatric populations.(4–10)
Allogeneic hematopoietic cell transplantation (HCT) for FLT3+ AML from sibling or unrelated donors (URD) has been most often reported in first complete remission (CR1) given the poor prognosis of disease.(11–19) Leukemia-free survival (LFS) at 2 years post HCT approximates 50–60% in most studies; (8, 13, 20, 21) although it ranges widely from 20%(5, 15) to 70%(22). Umbilical cord blood (UCB) HCT has increasingly been used for patients when suitable HLA matched donors are unavailable and when proceeding to transplantation is urgent(23–29), potentially as in FLT3+ AML. The outcomes of UCB HCT are reportedly similar to sibling or URD HCT for various diseases.(24, 30, 31) The outcomes of patients with FLT3+ AML after UCB HCT are largely unknown except for a recent University of Minnesota report.(32)
In this large retrospective study, we compared the efficacy of UCB HCT with matched sibling and URD grafts in FLT3+ AML using data from 3 large international observational registries. We hypothesized that relapse and LFS after UCB HCT would be similar to sibling or URD HCT.
Patients and Methods
Data Collection
The data on sibling and URD HCT were obtained solely from the Center for International Blood and Marrow Transplant Research (CIBMTR), a voluntary network of more than 450 transplant centers worldwide that report data on consecutive HCTs. Patient, disease and HCT characteristics and outcome data are reported on standardized forms submitted at the time of HCT (baseline) and at 100 days, 6 months and annually thereafter. Data on UCB HCT were obtained from the CIBMTR, Eurocord, and the European Group for Blood and Marrow Transplantation (EBMT). All patients provided written informed consent for research. The Institutional Review Board of the National Marrow Donor Program and Eurocord approved this study.
Inclusion Criteria
Included are adult FLT3+ AML patients (aged ≥18 years) who received UCB HCT (single or double unit), sibling or URD HCT in first or second complete remission (CR1 or CR2) between 2007 and 2012 as data on FLT3 mutation status was incompletely reported in prior years. The presence of FLT3+ mutation was reported by the transplant center. Assay method and quantitative data are not available. Previous HCT, ex vivo manipulated UCB, UCB combined with another source of stem cells, and haploidentical donor HCTs were excluded. There were no exclusions regarding conditioning regimen, alemtuzumab or anti-thymocyte globulin (ATG) use or regimen intensity.
Definitions
Cytogenetic data (G-banding and/or FISH analyses) at diagnosis were classified according to the Southwest Oncology Group (SWOG)/European Leukemia Net (ELN).(33, 34) LFS and CR were defined according to the International Working Group criteria.(35) Conditioning regimen intensity was based on the report of Bacigalupo et al.(36)
Endpoints
Relapse, the primary endpoint, was defined as morphological recurrence of disease, and non-relapse mortality (NRM) was considered a competing risk. Molecular (FLT3 mutation) evidence of leukemia as well as tyrosine kinase use before or after HCT was not considered for relapse or measures of minimal residual disease as these data were not available. Secondary endpoints included LFS, NRM, and overall survival (OS). Relapse or death from any cause was considered an event for LFS–the opposite of treatment failure. NRM was defined as death in remission, and disease relapse was considered a competing risk. Neutrophil recovery was defined as achieving an absolute neutrophil count of ≥0.5 × 109/L for the first of 3 measurements. Platelet recovery was defined as achieving platelets ≥20 × 109/L, unsupported by platelet transfusion for 7 days. Grade II-IV acute and chronic graft-versus-host disease (GVHD) were graded using standard criteria.(37, 38) For neutrophil recovery, platelet recovery, acute and chronic GVHD, death without specific event was considered a competing risk. Study subjects were right-censored if corresponding event was not observed at end of study.
Statistical Analysis
Patient-, disease-, and transplant-related variables for donor types were compared using chi-square statistics for categorical variables and the Kruskal-Wallis test for continuous variables. Probabilities for relapse, NRM and GVHD were calculated using the cumulative incidence (CI) estimator to accommodate competing risks. Kaplan-Meier estimates were used to calculate the probability of LFS and OS. The composite endpoint of GvHD-free (no grade III/IV acute GVHD and no chronic GVHD), relapse-free survival (GFRS) point estimates are provided using unadjusted Kaplan-Meier estimates. Time to event endpoints were measured from the date of HCT. The Cox proportional hazards regression model was used to identify risk factors associated with acute and chronic GVHD, relapse, NRM, LFS (treatment failure) and OS (overall mortality). As the primary variable of interest was donor type (UCB vs. HLA-matched sibling vs. URD), this variable was included in all steps of model building regardless of level of significance. For other variables a forward selection method was used to build the regression models. Variables tested included: age (18–29 vs. 30–49 vs. 50–69), gender (male vs. female), performance score (90–100 vs. <90), white blood cell count (WBC) at diagnosis (<10 vs. 11–50 × 109/L vs. >50 × 109/L), cytogenetic risk group (favorable/intermediate vs. adverse), time from diagnosis to CR1 (<5 vs. 5–8 vs. >8 weeks), and disease status at HCT (CR1 vs. CR2). None of the variables violated the assumptions of proportionality. Variables which were statistically significant with p-value ≤0.05 were retained in the final models. There were no first order interactions between the main effect (donor type) and variables in the final multivariate models. Adjusted probabilities of LFS and survival, and adjusted cumulative incidence functions of NRM, relapse and acute and chronic GVHD were calculated using the multivariate models, stratified on type of transplant and weighted by the pooled sample proportion value for each prognostic factor.(39, 40) These adjusted probabilities estimate likelihood of outcomes in populations with similar prognostic factors. All analyses were done using the statistical package SAS version 9.3 (Cary, NC).
Results
A total of 284 FLT3+ AML patients received HCT. Their clinical and treatment characteristics are shown in Table 1. One hundred and twenty-six patients received unrelated UCB (76 (60%) double units UCB grafts), 91 patients received peripheral blood (n=73) or bone marrow (n=18) from adult URD donors and 67 patients received peripheral blood (n=64) or bone marrow (n=3) from HLA-matched siblings. The median ages of the three graft type groups (UCB, sibling, URD) ranged from 41 to 48 years. Approximately 80% of HCTs occurred in CR1 and the most common conventional cytogenetic risk was intermediate (i.e., normal karyotype) in all treatment groups. The median time to achieve CR1 was 5 weeks for the adult donor HCT and 6 weeks for UCB HCT. Among patients transplanted in CR1, approximately half of sibling HCT recipients received their HCT less than 12 weeks from achieving CR1 (median time to HCT 11 weeks). In contrast, only 20% of UCB and URD recipients received their HCT within 12 weeks from CR1 (median time to HCT 17 and 16 weeks, respectively). Most recipients of sibling and URD HCT received myeloablative-conditioning regimen (MAC) while a third of UCB recipients received a reduced intensity-conditioning (RIC) regimen. Although most patients received calcineurin inhibitor containing GVHD prophylaxis, mycophenolate was the predominant second agent for UCB HCT vs. methotrexate for sibling and URD HCT. The median follow-up of survivors in each of the treatment groups was 3 years.
Table 1.
Patients characteristics
| Variable | HLA-matched sibling | Unrelated Donor | Umbilical Cord Blood | P-value |
|---|---|---|---|---|
| Number | 67 | 91 | 126 | |
| Gender | 0.91 | |||
| Male | 31 (46) | 39 (43) | 55 (44) | |
| Female | 36 (54) | 52 (57) | 71 (56) | |
| Age, years | 0.02 | |||
| Median (range) | 48 (18–59) | 43 (19–60) | 41 (18–67) | 0.002 |
| 18 – 29 | 5 (7) | 21 (23) | 34 (27) | |
| 30 – 49 | 31 (46) | 39 (43) | 58 (46) | |
| 50 – 69 | 31 (36) | 31 (34) | 34 (27) | |
| Performance score | 0.04 | |||
| < 90 | 21 (31) | 28 (31) | 22 (17) | |
| 90 – 100 | 42 (63) | 60 (66) | 101 (80) | |
| Not reported | 4 (6) | 3 (3) | 3 (2) | |
| WBC, diagnosis | <0.001 | |||
| ≤10 × 109/L | 18 (27) | 21 (23) | 22 (17) | |
| 11 – 50 × 109/L | 26 (39) | 31 (34) | 29 (23) | |
| > 50 × 109/L | 22 (33) | 34 (37) | 41 (33) | |
| Not reported | 1 (1) | 5 (5) | 34 (27) | |
| Cytogenetic risk | 0.65 | |||
| Favorable | 2 (3) | 3 (3) | 5 (4) | |
| Intermediate | 55 (82) | 73 (80) | 106 (84) | |
| Poor | 9 (13) | 11 (12) | 8 (6) | |
| Missing | 1 (1) | 4 (4) | 7 (6) | |
| Recipient CMV | 0.30 | |||
| Negative | 25 (37) | 35 (38) | 39 (31) | |
| Positive | 42 (63) | 54 (59) | 84 (67) | |
| Missing | 0 | 2 (2) | 3 (2) | |
| Time to CR1, weeks | 0.02 | |||
| ≤ 5 | 36 (54) | 44 (48) | 37 (29) | |
| 6 – 8 | 18 (27) | 24 (26) | 39 (31) | |
| > 8 | 8 (12) | 16 (18) | 36 (29) | |
| Not reported | 5 (7) | 7 (8) | 14 (11) | |
| Disease status, HCT | 0.82 | |||
| CR1 | 52 (78) | 73 (80) | 97 (77) | |
| CR2 | 15 (22) | 18 (20) | 29 (23) | |
| Duration of CR1 | 0.21 | |||
| <12 months | 12 (80) | 11 (61) | 17 (59) | |
| ≥12 months | 2 (13) | 5 (28) | 3 (10) | |
| Missing | 1 (7) | 2 (11) | 9 (31) | |
| Conditioning regimen | ||||
| Myeloablative | ||||
| TBI+Cy±other | 37 (55) | 35 (38) | 45 (36) | |
| TBI+other | 2 (3) | 0 | 5 (4) | |
| Bu+Cy/other | 22 (33) | 42 (46) | 33 (26) | |
| Other* | 0 | 0 | 3 (2) | |
| Reduced Intensity | ||||
| Bu+Flu | 4 (6) | 8 (9) | 0 | |
| TBI 200cGy ±Flu±other | 0 | 3 (3) | 40 (32) | |
| Other* | 2 (3) | 3 (3) | 0 | |
| Graft type | ||||
| Bone marrow | 3 (4) | 18 (20) | ||
| Peripheral blood | 64 (96) | 73 (80) | ||
| Umbilical cord blood | ||||
| Single | 50 (40) | |||
| Double | 76 (60) | |||
| GVHD prophylaxis | ||||
| CsA/Tac+MMF | 10 (15) | 17 (19) | 97 (77) | |
| CsA/Tac+MTX | 42 (63) | 69 (76) | 1 (<1) | |
| CsA/Tac+other | 15 (22) | 2 (2) | 21 (17) | |
| Other** | 0 | 3 (3) | 7 (6) | |
| Transplant period | 0.68 | |||
| 2007 – 2009 | 33 (49) | 47 (52) | 57 (45) | |
| 2010 – 2012 | 34 (51) | 44 (48) | 69 (55) | |
| Follow up, median (range), months | 37 (13 – 61) | 37 (12 – 65) | 37 (6 – 84) |
Abbreviations: WBC = white blood cell count; CMV=cytomegalovirus; CR = complete remission; HCT = hematopoietic cell transplant; TBI = total body irradiation; Cy = cyclophosphamide; CsA = cyclosporine; Tac = tacrolimus
MAC other: Flu+Mel+Thio+ATG, n=3 and RIC other: Bu+Clo, n=1; Flu+Mel, n=1; TLI+ATG, n=3
Other GVHD prophylaxis: MTX, n=2; Unknown, n=9
Relapse and Leukemia-free Survival (LFS)
The primary outcome of interest was relapse after HCT. After adjusting for the effects of white blood cell count at diagnosis and disease status at the time HCT, there were no significant differences in relapse risks between UCB or HLA-matched siblings or URD donors (Table 2), and no difference between UCB HCT compared with URD (HR 1.05, 95% CI 0.65 – 1.69, p=0.84). The 3-year probabilities of relapse, adjusted for white blood cell count and remission status were: HLA-matched sibling 44% (95% CI 31–55); UCB 33% (95% CI 25–42) and URD 33% (95% CI 24–42), p >0.72 (Figure 1, Supplemental Table 1). Pairwise comparisons between each donor type were not significant (all p>0.16) (Supplemental Table 1). Relapse risks were higher in patients with WBC >50 × 109/L at diagnosis compared with ≤10 ×109/L (HR 2.72, 95% CI 1.52 – 4.86, p=0.0007) and in those receiving HCT in CR2 compared with CR1 (HR 1.83, 95% CI 1.17 – 2.84, p=0.008).
Table 2.
Multivariate analysis using proportional hazards model for relapse, NRM, LFS, and OS
| Relapse | Non-Relapse Mortality | Leukemia-free Survival | Overall Survival | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variables | N | RR | 95% CI | p-value | RR | 95% CI | p-value | RR | 95% CI | p-value | RR | 95% CI | p-value |
| Graft source / donor | 0.72 | 0.01 | 0.42 | 0.26 | |||||||||
| BM/PB sibling | 67 | 1 | 1 | 1 | 1 | ||||||||
| BM/PB unrelated | 91 | 0.81 | 0.49–1.36 | 0.43 | 1.49 | 0.65–3.38 | 0.34 | 0.94 | 0.61–1.45 | 0.78 | 1.09 | 0.69–1.70 | 0.72 |
| UCB | 126 | 0.86 | 0.52–1.42 | 0.54 | 2.83 | 1.33–6.04 | 0.007 | 1.19 | 0.79–1.80 | 0.40 | 1.36 | 0.9–2.06 | 0.14 |
| Time From Diagnosis to CR1 | 0.056 | ||||||||||||
| <5 weeks | 117 | 1 | |||||||||||
| 5–8 weeks | 81 | 0.37 | 0.18–0.75 | 0.0063 | |||||||||
| ≥8 weeks | 60 | 0.74 | 0.38–1.41 | 0.36 | |||||||||
| Missing | 26 | 0.66 | 0.25–1.70 | 0.38 | |||||||||
| WBC at Diagnosis | 0.001 | 0.0031 | |||||||||||
| ≤10 | 61 | 1 | 1 | ||||||||||
| 10–50 | 86 | 1.48 | 0.78–2.79 | 0.23 | 1.44 | 0.89–2.33 | 0.14 | ||||||
| >50 | 97 | 2.72 | 1.52–4.86 | 0.0007 | 2.16 | 1.37–3.40 | 0.0009 | ||||||
| Missing | 40 | 1.18 | 0.52–2.66 | 0.69 | 1.22 | 0.67–2.21 | 0.52 | ||||||
| Disease status prior to HCT | |||||||||||||
| CR1 | 222 | 1 | 1 | 1 | |||||||||
| CR2 | 62 | 1.83 | 1.17–2.84 | 0.0076 | 1.67 | 1.17–2.39 | 0.0052 | 1.55 | 1.08–2.22 | 0.019 | |||
Figure 1.
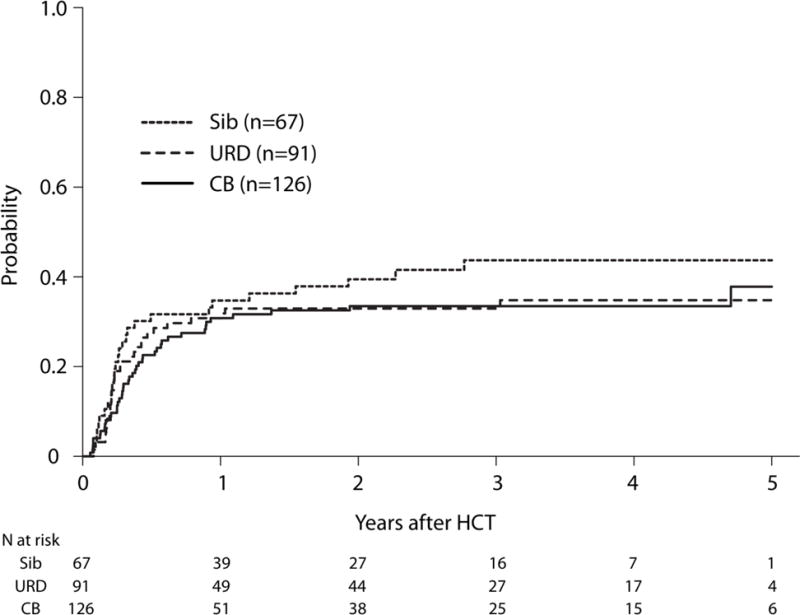
Adjusted Cumulative Incidence of Relapse by Donor Type. The 3-year probabilities of relapse, adjusted for white blood cell count and remission status, were 44% (95% CI 31–55), 33% (95% CI 25–42) and 33% (95% CI 24–42) after HLA-matched sibling, UCB and URD HCTs, respectively, p=0.72.
After adjusting for the effects of WBC at diagnosis and disease status at the time HCT, the risk of treatment failure (relapse or death; inverse of LFS) was similar after UCB and URD HCTs as compared with HLA-matched sibling HCT (Table 2). Similarly, there was no significant difference in the risk of treatment failure after UCB compared with URD HCT (HR 1.27, 95% CI 0.87 – 1.85, p=0.21). The 3-year probabilities of LFS, after adjusting for WBC and disease status were 43% (95% CI 30–54), 39% (95% CI 30–47) and 50% (95% CI 40–60) after HLA-matched sibling, UCB and URD HCTs, respectively, p=0.42 (Figure 2, Supplemental Table 1). The risk of treatment failure was greater in patients with WBC>50 × 109/L at diagnosis (HR 2.16, 95% CI 1.37 – 3.40, p=0.0009) and for patients receiving HCTs in CR2 (HR 1.67, 95% CI 1.17 – 2.39, p=0.005).
Figure 2.
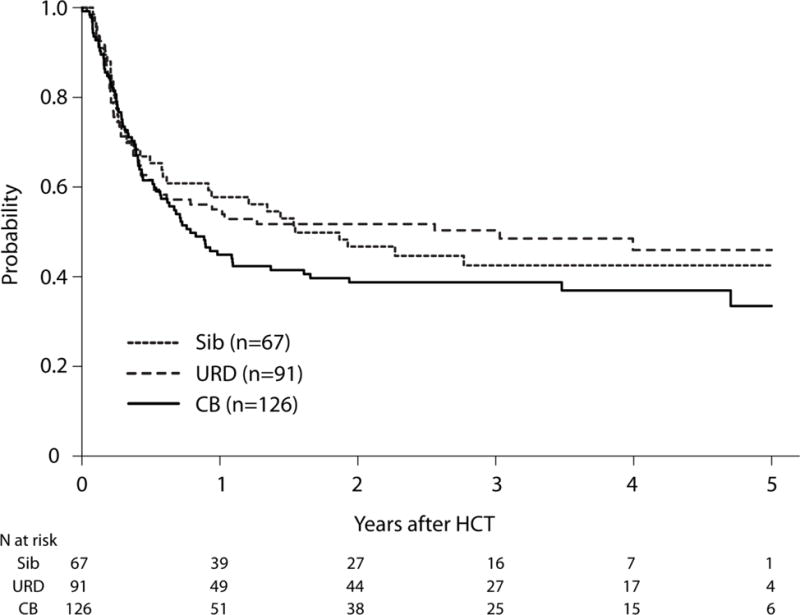
Adjusted Leukemia-free Survival by Donor Type. The 3-year probabilities of LFS, after adjusting for WBC and disease status were 43% (95% CI 30–54), 39% (95% CI 30–47) and 50% (95% CI 40–60) after HLA-matched sibling, UCB and URD HCTs, respectively, (p=0.42).
Non-relapse Mortality (NRM) and Overall Survival (OS)
Compared with HLA-matched sibling HCT, NRM risks were higher after UCB HCT, but not after URD HCT (Table 2). NRM risks were marginally, but not significantly higher after UCB HCT compared with URD HCT (HR 1.72, 95% CI 0.95 – 3.12, p=0.07). The 3-year probabilities of NRM were 14% (95% CI 7–23), 28% (95% CI 20 – 36) and 17% (95% CI 10–25) after HLA-matched sibling, UCB and URD HCTs, respectively (Figure 3, Supplemental Table 1). However, there were no significant differences in risks of overall mortality between the three donor types (Table 2). The 3-year probabilities of OS, adjusted for disease status were 46% (95% CI 33–59), 43% (95% CI 34–52) and 50% (95% CI 39–60) after HLA-matched sibling, UCB and URD HCTs, respectively, p=0.26 (Figure 4). The most common cause of death was disease relapse for each treatment group (Supplemental Table 2). Overall mortality was higher for HCTs in CR2 (1.55, 95% CI 1.08 – 2.22, p=0.02) (Table 2).
Figure 3.
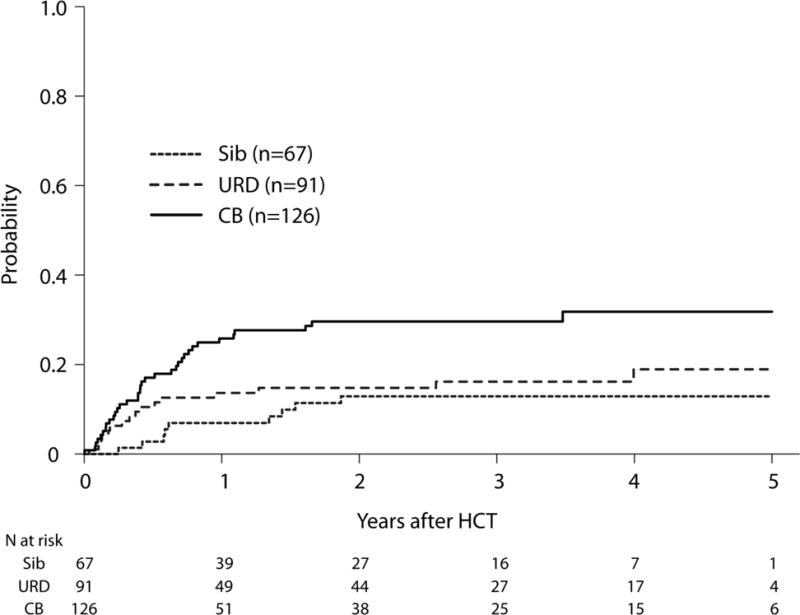
Adjusted Cumulative Incidence of Non-relapse Mortality (NRM) by Graft Source. The 3-year probabilities of NRM, after adjusting for time from diagnosis to CR1 were 14% (95% CI 7–23), 28% (95% CI 20 – 36) and 17% (95% CI 10–25) after HLA-matched sibling, UCB and URD HCTs, respectively. Pairwise comparisons: UCB vs. URD, p=0.07; UCB vs. sibling, p=0.02; URD vs. sibling, p=0.47.
Figure 4.
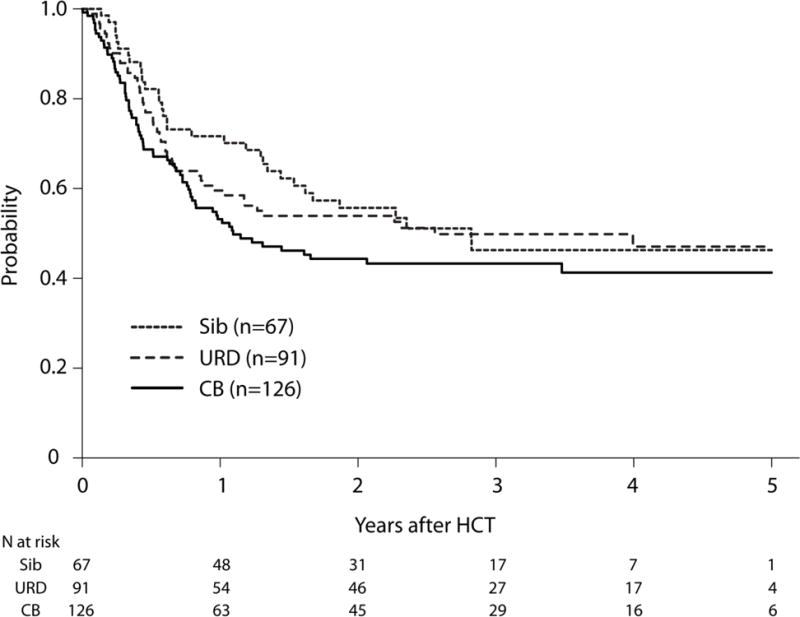
Adjusted Overall Survival by Donor Type. The 3-year probabilities of OS, adjusted for disease status were 46% (95% CI 33–59), 43% (95% CI 34–52) and 50% (95% CI 39–60) after HLA-matched sibling, UCB and URD HCTs, respectively, p=0.26.
Hematopoietic Recovery and Graft versus Host Disease (GVHD)
The median time to reach neutrophil engraftment was longer for UCB HCT (day +22) compared with HLA-matched sibling and URD HCTs (day +14 for each), (p<0.001). However, by day +60, there was no significant difference in engraftment among the 3 groups. Compared with HLA-matched sibling HCT, grade II-IV acute GVHD risks were higher after URD HCT (HR 1.85, 95% CI 1.08 – 3.15, p=0.02), but were not statistically different after UCB HCT (HR 1.66, 95% CI 0.99 – 2.78, p=0.06). The day-100 probabilities of acute GVHD were 27% (95% CI 17–38), 42% (95% CI 33–50) and 45% (95% CI 35–56) after HLA-matched sibling, UCB and URD HCTs, respectively. In contrast, chronic GVHD risks were significantly lower after UCB HCT compared with HLA-matched sibling (HR 0.59, 95% CI 0.37 – 0.94, p=0.03) or URD HCT (HR 0.50, 95% CI 0.32 – 0.76, p=0.001). Chronic GVHD risks were similar after HLA-matched sibling or URD HCT (HR 1.19, 95% CI 0.77 – 1.83, p=0.44). The 3-year probabilities of chronic GVHD were 62% (95% CI 49–75), 32% (95% CI 24–41) and 60% (95% CI 49–70) after HLA-matched sibling, UCB and URD HCTs, respectively.
GVHD-free, relapse free survival (GFRS) at year 1 and 3 years was slightly, but not significantly higher in UCB HCT (26%, 95%CI 18–34 and 20%, 95%CI 14–28) vs. sibling HCT (16%, 95%CI 9–26 and 5%, 95%CI 1–12), or URD HCT (16%, 95%CI 10–25 and 8%, 95%CI 3–15), p=0.12.
Prognostic Factors in UCB HCT
In the cohort of UCB HCTs, the 2-year relapse risk was significantly lower in patients receiving MAC compared with RIC (25%, 95%CI 16–35% vs. 45%, 95%CI 30–61, p=0.03). In contrast, NRM risk at 2 years was significantly higher in patients receiving MAC compared with RIC (37%, 95%CI 16–35 vs. 13%, 95%CI 4–25%, p=0.009). This resulted in similar 2-year LFS for MAC (38%, 95%CI 27–49) and RIC (42%, 95%CI 27–58, p=0.65) UCB HCTs and similar 2-years OS for MAC (40%, 95%CI 30–51 and RIC (52%, 95%CI 37–67, p=0.21). The number of UCB units infused (i.e., single vs. double) had no impact on any reported outcomes.
Discussion
In this study, we found that FLT3+ AML patients receiving UCB HCT had no statistically significant difference in relapse and LFS rates compared with HLA-matched sibling or URD HCTs. This is concordant with prior studies comparing UCB with other graft sources. (24, 30, 41) In our study, despite a greater proportion of UCB recipients receiving RIC than MAC (32% vs. Sibling 9% and MUD 15%), which is associated with higher relapse rates in patients with AML, with or without FLT3 mutation, (19, 42) the adjusted risks of relapse and treatment failure were similar for the 3 groups. UCB HCT patients also had a longer duration to reach CR1. Therefore, our data support an inference that there is graft-vs-leukemia (GVL) effect after single or double UCB HCT(24, 43), even for FLT3+ AML and its attendant high risk of relapse. Consistent with our large multicenter study, a single center study on 66 AML patients (22 FLT3+ and 44 FLT3-) receiving UCB HCT showed that the negative effect of FLT3+ AML was overcome by UCB HCT.(32) Two-year relapse rate was for FLT3+ AML 29% whereas 36% for FLT-AML, which led to LFS: 48% vs. 37%, and OS: 47% vs. 42% in FLT3+ AML vs. FLT3-AML, respectively). As the high risk nature of FLT3+ AML and the need for aggressive consolidation with allogeneic HCT is well recognized, our observation has major clinical implications for FLT3+ AML patients in CR after initial therapy. Non-HCT consolidative chemotherapies may lead to increased FLT3 ligand plasma levels and thus resistance to further therapies.(44) Our data demonstrates that performing allogeneic HCT as consolidation in CR1 yields favorable outcomes given poorer survival for HCT during CR2.
The higher NRM after UCB HCT may be attributed to slower hematopoietic recovery and subsequent infections. (24, 45, 46) Some UCB reports suggest that acute GVHD risks are similar or lower than after HLA-matched sibling HCT or URD HCT.(24, 47, 48) The notably lower risks of chronic GVHD offset these early complications in UCB HCT.(24, 49–51) In this study, while the incidence of acute GVHD after UCB was not significantly different than other graft types; however, chronic GVHD was significantly less frequent after UCB HCT. Although there was no statistically significant difference observed, OS was slightly lower and GRFS was slightly better for UCB HCT compared with other donor grafts.
We had insufficient data available to separately analyze FLT3/ITD+ or FLT3/TKD+ AML or the FLT3-mutant allelic burden (5, 9, 52, 53) and had only incomplete data on NPM1 mutations. In the literature, FLT3/ITD mutation is frequently associated with poor prognosis; this is less certain for the FLT3/TKD mutation.(4, 15) While the co-existence of NPM1 mutations in patients with FL3/ITD+ AML may influence the risk of relapse, (21, 54) a recent study from MD Anderson showed that allogeneic HCT remained statistically significant with improved RFS and OS independent of FLT3/ITD allelic ratio and NPM1 mutation status in multivariate regression models.(55) This might not be true for RIC allogenic HCT (56). In our study, we were unable address this controversial issue. Interestingly, we observed that high WBC (>50 × 109/L) at diagnosis was found to be associated with a higher relapse risk(57) and it may be correlated with the FLT3/ITD allelic ratio.(1, 5, 57, 58) As expected, HCT during CR2 was also associated with significantly increase relapse and with inferior LFS and OS. Another potential limitation of the study is that the available data from the three international registries had differing numbers of cases using each graft type. While referral patterns and graft choices and the resultant influence on outcomes might differ in the cases reported from each registry we could not directly probe this possibility with available data. We could not evaluate HCT-comorbidity index, shown to be associated with NRM and OS, (59) due to insufficient data.
These data support the use of UCB as a donor graft for patients with FLT3+ AML who lack a readily available HLA matched sibling donor. Our data also suggest that delay to HCT in these patients with an expectedly short CR1 adversely affects outcomes, possibly further favoring the more quickly available UCB. Studies on the use of partially matched related donors, as yet another rapidly available donor type are warranted. Additionally, following any donor HCT, post-transplant maintenance with FLT3 inhibitors seems promising because 30–40% of patients still relapse after allogeneic HCT regardless of graft type.(60)
Supplementary Material
Acknowledgments
CIBMTR Support
The CIBMTR is supported by Public Health Service Grant/Cooperative Agreement 5U24-CA076518 from the National Cancer Institute (NCI), the National Heart, Lung and Blood Institute (NHLBI) and the National Institute of Allergy and Infectious Diseases (NIAID); a Grant/Cooperative Agreement 5U10HL069294 from NHLBI and NCI; a contract HHSH250201200016C with Health Resources and Services Administration (HRSA/DHHS); two Grants N00014-15-1-0848 and N00014-16-1-2020 from the Office of Naval Research; and grants from Alexion; *Amgen, Inc.; Anonymous donation to the Medical College of Wisconsin; Astellas Pharma US; AstraZeneca; Be the Match Foundation; *Bluebird Bio, Inc.; *Bristol Myers Squibb Oncology; *Celgene Corporation; Cellular Dynamics International, Inc.; *Chimerix, Inc.; Fred Hutchinson Cancer Research Center; Gamida Cell Ltd.; Genentech, Inc.; Genzyme Corporation; *Gilead Sciences, Inc.; Health Research, Inc. Roswell Park Cancer Institute; HistoGenetics, Inc.; Incyte Corporation; Janssen Scientific Affairs, LLC; *Jazz Pharmaceuticals, Inc.; Jeff Gordon Children’s Foundation; The Leukemia & Lymphoma Society; Medac, GmbH; MedImmune; The Medical College of Wisconsin; *Merck & Co, Inc.; Mesoblast; MesoScale Diagnostics, Inc.; *Miltenyi Biotec, Inc.; National Marrow Donor Program; Neovii Biotech NA, Inc.; Novartis Pharmaceuticals Corporation; Onyx Pharmaceuticals; Optum Healthcare Solutions, Inc.; Otsuka America Pharmaceutical, Inc.; Otsuka Pharmaceutical Co, Ltd. – Japan; PCORI; Perkin Elmer, Inc.; Pfizer, Inc; *Sanofi US; *Seattle Genetics; *Spectrum Pharmaceuticals, Inc.; St. Baldrick’s Foundation; *Sunesis Pharmaceuticals, Inc.; Swedish Orphan Biovitrum, Inc.; Takeda Oncology; Telomere Diagnostics, Inc.; University of Minnesota; and *Wellpoint, Inc. The views expressed in this article do not reflect the official policy or position of the National Institute of Health, the Department of the Navy, the Department of Defense, Health Resources and Services Administration (HRSA) or any other agency of the U.S. Government.
*Corporate Members
Footnotes
Additional Contributing coauthors: Ravi Vij, Bipin N Savani, James Foran, Ulrike Bacher, Ann Jakubowski, Betty Hamilton, Michael Grunwald, Saar Gill, Mark Hertzberg, Samer Al-Homsi, Ghada Abusin, Bruce Camitta, Mitchell Cairo, Richard Olsson, Ran Reshef, Ayman Saad, Sahara Chhabra, David Rizzieri, Miguel A.Diaz, Mohammed Mir, Cesar Freytes, Rammurti Kamble, Zachariah Defilipp, Taiga Nishihori, Marlise Luskin Sachiko Seo, Mona Wirk, Patrick Stiff, Gary Schiller, Muthalagu Ramanathan, Mahmoud Aljurf, Sid Ganguly, Veronika Bachanova, Nelson Chao, Peter Wiernik, Selina Luger, Mary Laughin, Jean Khoury, Saber Wael, Robert P. Gale, Mark Litzow, Hillard Lazarus
COI: The authors report no conflicts of interest in the analysis or report of the data.
References
- 1.Thiede C, Steudel C, Mohr B, Schaich M, Schakel U, Platzbecker U, et al. Analysis of FLT3-activating mutations in 979 patients with acute myelogenous leukemia: association with FAB subtypes and identification of subgroups with poor prognosis. Blood. 2002;99(12):4326–35. doi: 10.1182/blood.v99.12.4326. [DOI] [PubMed] [Google Scholar]
- 2.Yamamoto Y, Kiyoi H, Nakano Y, Suzuki R, Kodera Y, Miyawaki S, et al. Activating mutation of D835 within the activation loop of FLT3 in human hematologic malignancies. Blood. 2001;97(8):2434–9. doi: 10.1182/blood.v97.8.2434. [DOI] [PubMed] [Google Scholar]
- 3.Gilliland DG, Griffin JD. The roles of FLT3 in hematopoiesis and leukemia. Blood. 2002;100(5):1532–42. doi: 10.1182/blood-2002-02-0492. [DOI] [PubMed] [Google Scholar]
- 4.Kottaridis PD, Gale RE, Frew ME, Harrison G, Langabeer SE, Belton AA, et al. The presence of a FLT3 internal tandem duplication in patients with acute myeloid leukemia (AML) adds important prognostic information to cytogenetic risk group and response to the first cycle of chemotherapy: analysis of 854 patients from the United Kingdom Medical Research Council AML 10 and 12 trials. Blood. 2001;98(6):1752–9. doi: 10.1182/blood.v98.6.1752. [DOI] [PubMed] [Google Scholar]
- 5.Gale RE, Green C, Allen C, Mead AJ, Burnett AK, Hills RK, et al. The impact of FLT3 internal tandem duplication mutant level, number, size, and interaction with NPM1 mutations in a large cohort of young adult patients with acute myeloid leukemia. Blood. 2008;111(5):2776–84. doi: 10.1182/blood-2007-08-109090. [DOI] [PubMed] [Google Scholar]
- 6.Meshinchi S, Alonzo TA, Stirewalt DL, Zwaan M, Zimmerman M, Reinhardt D, et al. Clinical implications of FLT3 mutations in pediatric AML. Blood. 2006;108(12):3654–61. doi: 10.1182/blood-2006-03-009233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Whitman SP, Ruppert AS, Radmacher MD, Mrozek K, Paschka P, Langer C, et al. FLT3 D835/I836 mutations are associated with poor disease-free survival and a distinct gene-expression signature among younger adults with de novo cytogenetically normal acute myeloid leukemia lacking FLT3 internal tandem duplications. Blood. 2008;111(3):1552–9. doi: 10.1182/blood-2007-08-107946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Brunet S, Labopin M, Esteve J, Cornelissen J, Socie G, Iori AP, et al. Impact of FLT3 internal tandem duplication on the outcome of related and unrelated hematopoietic transplantation for adult acute myeloid leukemia in first remission: a retrospective analysis. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2012;30(7):735–41. doi: 10.1200/JCO.2011.36.9868. [DOI] [PubMed] [Google Scholar]
- 9.Linch DC, Hills RK, Burnett AK, Khwaja A, Gale RE. Impact of FLT3(ITD) mutant allele level on relapse risk in intermediate-risk acute myeloid leukemia. Blood. 2014;124(2):273–6. doi: 10.1182/blood-2014-02-554667. [DOI] [PubMed] [Google Scholar]
- 10.Whitman SP, Maharry K, Radmacher MD, Becker H, Mrozek K, Margeson D, et al. FLT3 internal tandem duplication associates with adverse outcome and gene- and microRNA-expression signatures in patients 60 years of age or older with primary cytogenetically normal acute myeloid leukemia: a Cancer and Leukemia Group B study. Blood. 2010;116(18):3622–6. doi: 10.1182/blood-2010-05-283648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gale RE, Hills R, Kottaridis PD, Srirangan S, Wheatley K, Burnett AK, et al. No evidence that FLT3 status should be considered as an indicator for transplantation in acute myeloid leukemia (AML): an analysis of 1135 patients, excluding acute promyelocytic leukemia, from the UK MRC AML 10 and 12 trials. Blood. 2005;106(10):3658–65. doi: 10.1182/blood-2005-03-1323. [DOI] [PubMed] [Google Scholar]
- 12.Meshinchi S, Arceci RJ, Sanders JE, Smith FO, Woods WB, Radich JR, et al. Role of allogeneic stem cell transplantation in FLT3/ITD-positive AML. Blood. 2006;108(1):400. doi: 10.1182/blood-2005-12-4938. [DOI] [PubMed] [Google Scholar]
- 13.Bornhauser M, Illmer T, Schaich M, Soucek S, Ehninger G, Thiede C. Improved outcome after stem-cell transplantation in FLT3/ITD-positive AML. Blood. 2007;109(5):2264–5. doi: 10.1182/blood-2006-09-047225. [DOI] [PubMed] [Google Scholar]
- 14.DeZern AE, Sung A, Kim S, Tsai HL, Kowalski J, Smith BD, et al. Patients with FLT3/ITD AML May Benefit From Allogeneic Transplant In First Remission: Outcomes From a Consecutive Series of Patients at a Single Institution. Blood. 2010;116(21):896–7. [Google Scholar]
- 15.Sengsayadeth SM, Jagasia M, Engelhardt BG, Kassim A, Strickland SA, Goodman S, et al. Allogeneic Stem Cell Transplantation for Flt3/Itd Positive Acute Myeloid Leukemia in CR1 Implication for Personalized Therapy to Prevent Early Relapses. Biol Blood Marrow Tr. 2012;18(2):S332–S3. [Google Scholar]
- 16.Kayser S, Dohner K, Krauter J, Kohne C, Horst HA, Held G, et al. Allogeneic transplantation from matched related and unrelated donors in first complete remission in younger adult AML patients with FLT3 internal tandem duplications. Bone Marrow Transpl. 2011;46:S40–S1. [Google Scholar]
- 17.Hemmati P, Terwey TH, Vuong LG, le Coutre PD, Dorken B, Arnold R. Allogeneic Stem Cell Transplantation For Cytogenetically Normal Acute Myeloid Leukemia: Impact Of FLT3 and NPM1 Mutational Status. Blood. 2013;122(21) [Google Scholar]
- 18.Liegel J, Courville E, Sachs Z, Ustun C. Use of sorafenib for post-transplant relapse in FLT3/ITD-positive acute myelogenous leukemia: maturation induction and cytotoxic effect. Haematologica. 2014;99(11):e222–4. doi: 10.3324/haematol.2014.109975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Deol A, Sengsayadeth S, Ahn K, Wang H, Aljurf M, Antin J, et al. ADoes FLT3 Mutation Impact Survival after Hematopoietic Cell Transplant for AML? A CIBMTR Analysis Cancer. 2016 doi: 10.1002/cncr.30140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Laboure G, Dulucq S, Vigouroux S, Lippert E, Pigneux A, Tabrizi R, et al. Potent Graft-Versus-Leukemia Effect After Reduced Intensity (RIC) Allogeneic Stem-Cell Transplantation (ASCT) As Post-Remission Therapy for Intermediate-Risk De-Novo Acute Myeloid Leukemia (AML) with FLT3-ITD Genotype or Wild-Type (WT) NPM1 and CEBPA without FLT3-ITD. Blood. 2011;118(21):1766. [Google Scholar]
- 21.Schmid C, Labopin M, Socie G, Daguindau E, Volin L, Huynh A, et al. Outcome of patients with distinct molecular genotypes and cytogenetically normal AML after allogeneic transplantation. Blood. 2015;126(17):2062–9. doi: 10.1182/blood-2015-06-651562. [DOI] [PubMed] [Google Scholar]
- 22.DeZern AE, Sung A, Kim S, Smith BD, Karp JE, Gore SD, et al. Role of Allogeneic Transplantation for FLT3/ITD Acute Myeloid Leukemia: Outcomes from 133 Consecutive Newly Diagnosed Patients from a Single Institution. Biol Blood Marrow Tr. 2011;17(9):1404–9. doi: 10.1016/j.bbmt.2011.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bejanyan N, Oran B, Shanley R, Warlick E, Ustun C, Vercellotti G, et al. Clinical outcomes of AML patients relapsing after matched-related donor and umbilical cord blood transplantation. Bone Marrow Transplant. 2014;49(8):1029–35. doi: 10.1038/bmt.2014.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brunstein CG, Gutman JA, Weisdorf DJ, Woolfrey AE, Defor TE, Gooley TA, et al. Allogeneic hematopoietic cell transplantation for hematologic malignancy: relative risks and benefits of double umbilical cord blood. Blood. 2010;116(22):4693–9. doi: 10.1182/blood-2010-05-285304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ruggeri A, Labopin M, Sormani MP, Sanz G, Sanz J, Volt F, et al. Engraftment kinetics and graft failure after single umbilical cord blood transplantation using a myeloablative conditioning regimen. Haematologica. 2014;99(9):1509–15. doi: 10.3324/haematol.2014.109280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rocha V, Cornish J, Sievers EL, Filipovich A, Locatelli F, Peters C, et al. Comparison of outcomes of unrelated bone marrow and umbilical cord blood transplants in children with acute leukemia. Blood. 2001;97(10):2962–71. doi: 10.1182/blood.v97.10.2962. [DOI] [PubMed] [Google Scholar]
- 27.Eapen M, Klein JP, Ruggeri A, Spellman S, Lee SJ, Anasetti C, et al. Impact of allele-level HLA matching on outcomes after myeloablative single unit umbilical cord blood transplantation for hematologic malignancy. Blood. 2014;123(1):133–40. doi: 10.1182/blood-2013-05-506253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Barker JN, Weisdorf DJ, Wagner JE. Creation of a double chimera after the transplantation of umbilical-cord blood from two partially matched unrelated donors. N Engl J Med. 2001;344(24):1870–1. doi: 10.1056/NEJM200106143442417. [DOI] [PubMed] [Google Scholar]
- 29.Sandhu KS, Brunstein C, DeFor T, Bejanyan N, Arora M, Warlick E, et al. Umbilical Cord Blood Transplantation Outcomes in Acute Myelogenous Leukemia/Myelodysplastic Syndrome Patients Aged >/=70 Years. Biol Blood Marrow Transplant. 2015 doi: 10.1016/j.bbmt.2015.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Weisdorf D, Eapen M, Ruggeri A, Zhang MJ, Zhong X, Brunstein C, et al. Alternative Donor Transplantation for Older Patients with Acute Myeloid Leukemia in First Complete Remission: A Center for International Blood and Marrow Transplant Research-Eurocord Analysis. Biol Blood Marrow Tr. 2014;20(6):816–22. doi: 10.1016/j.bbmt.2014.02.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.de Latour RP, Brunstein CG, Porcher R, Chevallier P, Robin M, Warlick E, et al. Similar Overall Survival Using Sibling, Unrelated Donor, and Cord Blood Grafts after Reduced-Intensity Conditioning for Older Patients with Acute Myelogenous Leukemia. Biol Blood Marrow Tr. 2013;19(9):1355–60. doi: 10.1016/j.bbmt.2013.06.006. [DOI] [PubMed] [Google Scholar]
- 32.Eckfeldt CE, Randall N, Shanley RM, Yohe S, Bejanyan N, Dolan M, et al. Umbilical cord blood transplantation is a suitable option for consolidation of acute myeloid leukemia with FLT3-ITD. Haematologica. 2016 doi: 10.3324/haematol.2016.143628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Slovak ML, Kopecky KJ, Cassileth PA, Harrington DH, Theil KS, Mohamed A, et al. Karyotypic analysis predicts outcome of preremission and postremission therapy in adult acute myeloid leukemia: a Southwest Oncology Group/Eastern Cooperative Oncology Group Study. Blood. 2000;96(13):4075–83. [PubMed] [Google Scholar]
- 34.Mrozek K, Marcucci G, Nicolet D, Maharry KS, Becker H, Whitman SP, et al. Prognostic significance of the European LeukemiaNet standardized system for reporting cytogenetic and molecular alterations in adults with acute myeloid leukemia. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2012;30(36):4515–23. doi: 10.1200/JCO.2012.43.4738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cheson BD, Bennett JM, Kopecky KJ, Buchner T, Willman CL, Estey EH, et al. Revised recommendations of the International Working Group for Diagnosis, Standardization of Response Criteria, Treatment Outcomes, and Reporting Standards for Therapeutic Trials in Acute Myeloid Leukemia. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2003;21(24):4642–9. doi: 10.1200/JCO.2003.04.036. [DOI] [PubMed] [Google Scholar]
- 36.Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V, et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant. 2009;15(12):1628–33. doi: 10.1016/j.bbmt.2009.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant. 1995;15(6):825–8. [PubMed] [Google Scholar]
- 38.Flowers ME, Kansu E, Sullivan KM. Pathophysiology and treatment of graft-versus-host disease. Hematol Oncol Clin North Am. 1999;13(5):1091–112. viii–ix. doi: 10.1016/s0889-8588(05)70111-8. [DOI] [PubMed] [Google Scholar]
- 39.Zhang X, Zhang MJ. SAS macros for estimation of direct adjusted cumulative incidence curves under proportional subdistribution hazards models. Comput Methods Programs Biomed. 2011;101(1):87–93. doi: 10.1016/j.cmpb.2010.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhang X, Loberiza FR, Klein JP, Zhang MJ. A SAS macro for estimation of direct adjusted survival curves based on a stratified Cox regression model. Comput Methods Programs Biomed. 2007;88(2):95–101. doi: 10.1016/j.cmpb.2007.07.010. [DOI] [PubMed] [Google Scholar]
- 41.de Latour RP, Brunstein C, Porcher R, Chevallier P, Robin M, Warlick E, et al. Equivalent outcome between siblings, unrelated donors and cord blood after reduced intensity allogeneic haematopoietic cell transplantation for patients older than 50 years with acute myeloid leukaemia in complete remission. Bone Marrow Transpl. 2013;48:S379–S80. [Google Scholar]
- 42.Ustun C, Courville E, DeFor T, Dolan M, Randall N, Yohe S, et al. Myeloablative, but not Reduced-Intensity, Conditioning Overcomes the Negative Effect of Flow-Cytometric Evidence of Leukemia in Acute Myeloid Leukemia. Biol Blood Marrow Transplant. 2015 doi: 10.1016/j.bbmt.2015.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ponce DM, Hilden P, Devlin SM, Maloy M, Lubin M, Castro-Malaspina H, et al. High Disease-Free Survival with Enhanced Protection against Relapse after Double-Unit Cord Blood Transplantation When Compared with T Cell-Depleted Unrelated Donor Transplantation in Patients with Acute Leukemia and Chronic Myelogenous Leukemia. Biol Blood Marrow Transplant. 2015;21(11):1985–93. doi: 10.1016/j.bbmt.2015.07.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sato T, Yang X, Knapper S, White P, Smith BD, Galkin S, et al. FLT3 ligand impedes the efficacy of FLT3 inhibitors in vitro and in vivo. Blood. 2011;117(12):3286–93. doi: 10.1182/blood-2010-01-266742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rocha V, Gluckman E, Eurocord-Netcord r, European B, Marrow Transplant g Improving outcomes of cord blood transplantation: HLA matching, cell dose and other graft- and transplantation-related factors. Br J Haematol. 2009;147(2):262–74. doi: 10.1111/j.1365-2141.2009.07883.x. [DOI] [PubMed] [Google Scholar]
- 46.Eapen M, Rubinstein P, Zhang MJ, Stevens C, Kurtzberg J, Scaradavou A, et al. Outcomes of transplantation of unrelated donor umbilical cord blood and bone marrow in children with acute leukaemia: a comparison study. Lancet. 2007;369(9577):1947–54. doi: 10.1016/S0140-6736(07)60915-5. [DOI] [PubMed] [Google Scholar]
- 47.Rocha V, Labopin M, Sanz G, Arcese W, Schwerdtfeger R, Bosi A, et al. Transplants of umbilical-cord blood or bone marrow from unrelated donors in adults with acute leukemia. N Engl J Med. 2004;351(22):2276–85. doi: 10.1056/NEJMoa041469. [DOI] [PubMed] [Google Scholar]
- 48.Grewal SS, Barker JN, Davies SM, Wagner JE. Unrelated donor hematopoietic cell transplantation: marrow or umbilical cord blood? Blood. 2003;101(11):4233–44. doi: 10.1182/blood-2002-08-2510. [DOI] [PubMed] [Google Scholar]
- 49.Sugimoto K, Narimatsu H, Kawase T, Iida H, Watanabe M, Kohno A, et al. Clinical characteristics of chronic graft-versus-host disease following umbilical cord blood transplantation for adults. Bone Marrow Transplant. 2008;41(8):729–36. doi: 10.1038/sj.bmt.1705959. [DOI] [PubMed] [Google Scholar]
- 50.Lazaryan A, Weisdorf DJ, DeFor T, Brunstein CG, MacMillan ML, Bejanyan N, et al. Risk Factors for Acute and Chronic Graft-versus-Host Disease after Allogeneic Hematopoietic Cell Transplantation with Umbilical Cord Blood and Matched Sibling Donors. Biol Blood Marrow Transplant. 2015 doi: 10.1016/j.bbmt.2015.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Narimatsu H, Miyakoshi S, Yamaguchi T, Kami M, Matsumura T, Yuji K, et al. Chronic graft-versus-host disease following umbilical cord blood transplantation: retrospective survey involving 1072 patients in Japan. Blood. 2008;112(6):2579–82. doi: 10.1182/blood-2007-11-118893. [DOI] [PubMed] [Google Scholar]
- 52.Pratz KW, Sato T, Murphy KM, Stine A, Rajkhowa T, Levis M. FLT3-mutant allelic burden and clinical status are predictive of response to FLT3 inhibitors in AML. Blood. 2010;115(7):1425–32. doi: 10.1182/blood-2009-09-242859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Schlenk RF, Kayser S, Bullinger L, Kobbe G, Casper J, Ringhoffer M, et al. Differential impact of allelic ratio and insertion site in FLT3-ITD-positive AML with respect to allogeneic transplantation. Blood. 2014;124(23):3441–9. doi: 10.1182/blood-2014-05-578070. [DOI] [PubMed] [Google Scholar]
- 54.Pratcorona M, Brunet S, Nomdedeu J, Ribera JM, Tormo M, Duarte R, et al. Favorable outcome of patients with acute myeloid leukemia harboring a low-allelic burden FLT3-ITD mutation and concomitant NPM1 mutation: relevance to post-remission therapy. Blood. 2013;121(14):2734–8. doi: 10.1182/blood-2012-06-431122. [DOI] [PubMed] [Google Scholar]
- 55.Oran B, Cortes J, Beitinjaneh A, Chen HC, de Lima M, Patel K, et al. Allogeneic Transplantation in First Remission Improves Outcomes Irrespective of FLT3-ITD Allelic Ratio in FLT3-ITD-Positive Acute Myelogenous Leukemia. Biol Blood Marrow Transplant. 2016;22(7):1218–26. doi: 10.1016/j.bbmt.2016.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Versluis J, In‘t Hout FE, Devillier R, van Putten WL, Manz MG, Vekemans MC, et al. Comparative value of post-remission treatment in cytogenetically normal AML subclassified by NPM1 and FLT3-ITD allelic ratio. Leukemia. 2016 Jul 15; doi: 10.1038/leu.2016.183. [DOI] [PubMed] [Google Scholar]
- 57.de Jonge HJ, Valk PJ, de Bont ES, Schuringa JJ, Ossenkoppele G, Vellenga E, et al. Prognostic impact of white blood cell count in intermediate risk acute myeloid leukemia: relevance of mutated NPM1 and FLT3-ITD. Haematologica. 2011;96(9):1310–7. doi: 10.3324/haematol.2011.040592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Whitman SP, Archer KJ, Feng L, Baldus C, Becknell B, Carlson BD, et al. Absence of the wild-type allele predicts poor prognosis in adult de novo acute myeloid leukemia with normal cytogenetics and the internal tandem duplication of FLT3: a cancer and leukemia group B study. Cancer Res. 2001;61(19):7233–9. [PubMed] [Google Scholar]
- 59.Sorror ML, Giralt S, Sandmaier BM, De Lima M, Shahjahan M, Maloney DG, et al. Hematopoietic cell transplantation specific comorbidity index as an outcome predictor for patients with acute myeloid leukemia in first remission: combined FHCRC and MDACC experiences. Blood. 2007 Dec 15;110(13):4606–4613. doi: 10.1182/blood-2007-06-096966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Chen YB, Li S, Lane AA, Connolly C, Del Rio C, Valles B, et al. Phase I trial of maintenance sorafenib after allogeneic hematopoietic stem cell transplantation for fms-like tyrosine kinase 3 internal tandem duplication acute myeloid leukemia. Biol Blood Marrow Transplant. 2014;20(12):2042–8. doi: 10.1016/j.bbmt.2014.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


