Abstract
Primary open-angle glaucoma (POAG) is a genetically, physiologically, and phenotypically complex neurodegenerative disorder. This study addressed the expanding collection of genes associated with POAG, referred to as the “POAGome.” We used bioinformatics tools to perform an extensive, systematic literature search and compiled 542 genes with confirmed associations with POAG and its related phenotypes (normal tension glaucoma, ocular hypertension, juvenile open-angle glaucoma, and primary congenital glaucoma). The genes were classified according to their associated ocular tissues and phenotypes, and functional annotation and pathway analyses were subsequently performed. Our study reveals that no single molecular pathway can encompass the pathophysiology of POAG. The analyses suggested that inflammation and senescence may play pivotal roles in both the development and perpetuation of the retinal ganglion cell degeneration seen in POAG. The TGF-β signaling pathway was repeatedly implicated in our analyses, suggesting that it may be an important contributor to the manifestation of POAG in the anterior and posterior segments of the globe. We propose a molecular model of POAG revolving around TGF-β signaling, which incorporates the roles of inflammation and senescence in this disease. Finally, we highlight emerging molecular therapies that show promise for treating POAG.
Keywords: Primary open-angle glaucoma (POAG), Neuroinflammation, Neurodegeneration, Gliosis, Senescence, Senescence-associated secretory phenotype (SASP), TGF-β, NFκB
1. Introduction to glaucoma
Glaucoma is comprised of a group of optic neuropathies that share certain biological and clinical characteristics including progressive loss of the RGCs that causes specific, irreversible changes to the optic disc and visual fields (Casson et al., 2012; Takamoto and Araie, 2014). Despite the disease’s pervasiveness, its pathogenesis is not completely understood. With the advancement of GWAS, family studies and functional investigation studies over the past few years, significant progress has been made in understanding the molecular basis and complexity of glaucoma. Current reviews of glaucoma genetics primarily based on findings of large GWAS and family studies have highlighted that dysregulation of pathways affecting lipid metabolism, inflammation, autoimmune processes, and oxidative stress may be some of the most important contributors to the development of glaucoma (Gemenetzi et al., 2012; Greco et al., 2016; Janssen et al., 2013). The pathogenesis of primary-open angle glaucoma (POAG), which is a subtype of glaucoma, involves a wide-range of pathological processes. There are hundreds of genes that may play significant roles in the identified biological processes, and it is becoming necessary to determine how they interact with one another in order to develop targeted therapies in the future.
In this study, we have attempted to establish a large compendium of genes that have been shown to be associated with the five POAG-related phenotypes in order to broadly survey the genetic complexity potentially contributing to POAG. We have performed several iterations of functional annotation and pathway analysis of POAG associated genes to further elucidate lesser known or researched pathways, biological processes, and molecular interactions potentially involved in the development of POAG. We are confident that the gene-glaucoma associations we present may help generate more hypotheses surrounding the genetics of glaucoma and, provide an impetus for the development of further studies to characterize the pathogenesis of this debilitating disease.
2. Background
2.1. Clinical, epidemiological considerations and risk factors for POAG
Glaucoma is one of the most prevalent causes of blindness worldwide, with estimates of over 60 million people affected by the disease (Quigley and Broman, 2006). POAG is a subset of glaucoma that is progressive in nature, and is characterized by the absence of any apparent obstruction of aqueous outflow through the TM with gonioscopy, but often with elevated IOP (Weinreb and Khaw, 2004). POAG accounts for about 70% of all glaucoma cases worldwide, and its prevalence has been increasing, likely due to increased awareness and improvements in diagnostic technology (Beidoe and Mousa, 2012). POAG can have a congenital onset, but the majority of cases occur in adults aged 35 years or older without a clear inheritance pattern (Weinreb and Khaw, 2004). Elevated IOP has been recognized as the most important risk factor for the development of POAG. However, many patients with glaucoma can present without elevated IOP, as in normal tension glaucoma (NTG) (Gemenetzi et al., 2012).
Closed-angle glaucoma is another glaucoma subtype, which is characterized by restricted angle depth that contributes to increased resistance to aqueous outflow and elevated IOP (Weinreb et al., 2014). As in OAG, most patients are asymptomatic and tend to present only after severe irreversible vision loss has occurred. A large minority of patients with closed-angle glaucoma present with acute primary angle-closure glaucoma, which is extremely painful and can result in a rapid loss of vision without an emergency intervention (Weinreb et al., 2014).
Family history and race are also significant risk factors for the development of POAG. Data from the Baltimore Eye Study suggests that patients with siblings or parents suffering from glaucoma are at significantly higher risk of having POAG, and that this risk is moderately amplified in African-American families (Charlson et al., 2015; Salowe et al., 2015; Tielsch et al., 1994). Studies have revealed that African-Americans have approximately three times higher age-adjusted prevalence of POAG than European-Americans in the United States (Friedman et al., 2004). The prevalence of OAG in patients of Hispanic or Latino descent are comparable to that of African-Americans (Varma et al., 2004). Gender is also an important, but comparatively minor risk factor for the development of glaucoma, with men 1.37 times more likely than women to have POAG (Rudnicka et al., 2006). Low socioeconomic status has not been shown to be a significant risk factor for the development of glaucoma, but it can compound the severity of the disease, as glaucoma patients of a lower socioeconomic status tend to present later with greater visual field deficits (Tsai et al., 2003).
2.2. Anatomy & related pathology in primary open-angle glaucoma
2.2.1. Anterior and posterior segments of the globe
Aspects of glaucoma’s pathology can manifest both within the anterior and posterior segments of the eye. The anterior segment is further subdivided into the anterior and posterior chambers. The anterior chamber is bounded by the cornea anteriorly and the iris posteriorly. The posterior chamber is bounded anteriorly by the iris, and posteriorly by the anterior vitreous face, and its major structures include the lens and the ciliary body. The posterior segment comprises the structures posterior to the suspensory ligaments including the vitreous, retina, and ONH. Fig. 1 displays the important anatomy involved in glaucoma and how pressure affects the eye’s delicate structures.
Fig. 1.

Schematic showing the anatomy of POAG. Sub-image A reflects the abnormally high pressure transmitted to the posterior segment of the globe containing the retina and optic disc where RGCs and their axons reside. Sub image B reflects aqueous outflow pathways.
2.2.1.1. Anterior segment: aqueous flow and intraocular pressure
IOP is primarily determined by the relative production and drainage of aqueous humor within the anterior segment of the eye. Aqueous humor is produced by the epithelial cells in the ciliary body and secreted posterior to the iris. Two outflow pathways have been described to explain the flow of aqueous humor produced in the ciliary body: the conventional, and non-conventional (or uveoscleral) pathways. In the conventional pathway, the aqueous humor flows from the posterior chamber, between the iris and lens to enter the anterior chamber (Fig. 1b). From there it flows through the TM, through the juxtacanalicular connective tissue, into Schlemm’s canal, and is absorbed into the episcleral venous system. In the uveoscleral outflow pathway, a fraction of the secreted aqueous humor passes through the ciliary muscle, suprachoroidal space, choroidal vessels, and lymphatic channels (Alm and Nilsson, 2009). Eyes exhibiting high-tension POAG exhibit increased levels of resistance in the aqueous outflow (Johnson, 2006). Eyes with POAG have been found to demonstrate increased hyalinization, thickening, and stiffening of the TM, as well as increased signs of oxidative stress and apoptosis. These changes are thought to inhibit aqueous humor outflow (Janssen et al., 2013).
IOP is known to correlate positively with disease severity, and a decrease in IOP has been shown to reduce progression of POAG (AGIS Investigators, 2000). Furthermore, IOP seems to also play a role in the development of NTG, as patients diagnosed with NTG still realize a clinical benefit when their “normal” IOP is lowered (Collaborative Normal-Tension Glaucoma Study Group, 1998). An understanding of the anatomical determinants of IOP is therefore important to the understanding of POAG.
2.2.1.2. Posterior segment: retina & optic nerve
The retina is an incredibly complex component which contains the retinal pigment epithelium at the sclerad margin, and a neurosensory tissue which functions to convert photons into electrical impulses (Wyszecki and Stiles, 1982). The neurosensory retina contains 5 basic types of neurons, including, in order from outermost to innermost, rods and cones (photoreceptors), horizontal cells, bipolar cells, amacrine cells, RGCs and the nerve fiber layer (Purves et al., 2004). Visual information sent from the retina is delivered via the RGCs (Wyszecki and Stiles, 1982). The RGC axons are supported by glia in the optic nerve head and optic nerve, including oligodendrocytes and astrocytes, which provide important nutritional, structural and immunological support for the RGCs (Butt et al., 2004). Glia located in the inner retina, including astrocytes and Müller cells, provide similar supportive functions to RGC cell bodies (Johnson and Morrison, 2009). Additionally, retinal and optic nerve microglia, which are specialized, resident macrophages, play an important immunological role, and can affect RGC survival by secreting cytokines and neurotrophins (Butt et al., 2004; Perry and Gordon, 1991).
One of the most important diagnostic factors in glaucoma is an assessment of the ONH. The optic nerve is comprised of the axons of the RGC and surrounding glia. A central “cup” is formed that represents a depression as the axons bend at 90-degree angles to exit the eye. Increased IOP, in combination with other factors, such as inflammation from reactive glia, or impaired axonal transport of neurotrophins, damages the RGC axons at the level of the ONH and may lead to axonal degeneration (Soto and Howell, 2014; Vrabec and Levin, 2007). RGC death consequently decreases the size of the axonal rim, and increases the size of the optic cup relative to the disc, called “cupping.” Neuronal death is ultimately the cause of late-stage visual symptoms in glaucoma, and assessing various ONH parameters such as neuroretinal rim thickness, disk diameter, retinal nerve fiber layer health, cup-to-disc ratio, are some of the most commonly employed tools for monitoring optic nerve health (Spaeth et al., 2002).
2.2.1.3. Lamina cribrosa
As the optic nerve exits the eye, it passes through the lamina cribrosa, a sieve-like tissue comprised of LC cells and ECM principally composed of collagen fibers. The LC is sensitive to changes in IOP, and may be the primary site of RGC axonal damage in POAG (Crawford Downs et al., 2011; Howell et al., 2007). It has been hypothesized that with an increase in IOP, the LC deforms, stretching and compressing the encased optic nerve fibers. The LC also exhibits molecular remodeling and pathological changes in the supporting ECM, which may be in reaction to mechanical stretch from elevated IOP, hypoxia, altered TGF-β expression, or other factors that further perturb the environment of glia and RGC axons; although how this relates to axonal damage has not been proven (Pena et al., 2001).
2.3. The complexity of POAG’s genetics, pathogenesis and presentation
Many genome-glaucoma association studies performed in the last decade have revealed several genes that appear to play an important role in the pathogenesis of glaucoma (Janssen et al., 2013). Functional studies have attempted to uncover some of the important molecular mechanisms implicated in the development of POAG (Iglesias et al., 2014, 2015; Janssen et al., 2013). While there are several genes associated with early and severe forms of OAG that appear to be caused by a Mendelian inheritance pattern, it has largely been found that POAG is an extremely genetically complex disease, with many genes contributing to its development. Therefore, it is hypothesized that different variants cause alterations in RGCs, making them more or less susceptible to cell death (Dietz et al., 2014; Heijl et al., 2002; Kass et al., 2002; Li et al., 2007) It is also speculated that certain variants compromise the homeostasis of a patient’s axon-supporting ONH glia, leading to neuroinflammation or a bottleneck of the axonal transport systems at the ONH (Johnson and Morrison, 2009; Pasutto et al., 2009). Additionally, within the anterior compartment, pathological variants could conceivably contribute to TM cells producing disorganized ECM components, contributing to ocular hypertension (OHT) (Libby et al., 2005; Vranka et al., 2015).
The etiology of POAG remains unclear. The clinical definition of POAG has been a matter of persistent debate, such that there are many clinically unique phenotypes that may be considered as variations of the same disease process, or perhaps entirely different processes leading to related clinical endpoints. These phenotypes include, high tension glaucoma (HTG), NTG, juvenile open-angle glaucoma (JOAG), primary congenital glaucoma (PCG), OHT, and possibly others which have not yet been well characterized.
2.3.1. POAG phenotypes and their ambiguity within the literature
HTG, which is often simply referred to as POAG, is characterized by the presence of RGC loss in the setting of OHT (IOP > 21 mmHg). The delineation of “POAG,” “HTG,” and “NTG” in the literature is often unclear. For example, NTG can be considered to be specific “POAG” phenotype, or may be strictly differentiated from “POAG,” when the authors require that “POAG” involves elevated IOP (i.e. a synonym for HTG). In this review, we accommodated the ambiguity found within the literature by deeming the phenotype in question as “POAG” when the study explicitly described HTG or did not include IOP as a defining feature, making the delineation of HTG and NTG impossible.
NTG is a condition where the patient exhibits a normal IOP (10–21 mmHg), and also has clinical signs of optic nerve damage or visual field loss (Kanski et al., 2011). Except for increased IOP, the risk factors which increase the likelihood of progression for POAG, also contribute to NTG (Kanski et al., 2011). However, the Collaborative Normal Tension Glaucoma Study concluded that reduction of IOP reduced the progression of NTG (Collaborative Normal-Tension Glaucoma Study Group, 1998). Therefore, it seems that the optic nerves of patients suffering from NTG have a lower threshold for the development of pressure-induced RGC damage, despite the absence of elevated IOP (Kanski et al., 2011; Mallick et al., 2016; Shields, 2008).
JOAG is characterized by the presence of POAG in young patients. Typically, patients with disease onset at 40 years or older are excluded from the diagnosis. PCG is found in infants that also presents in a diverse number of ways. It could be argued that PCG should not be included as a POAG phenotype, given that the disease is generally characterized by the presence of severe developmental abnormalities of TM, referred to as trabeculodysgenesis. Some consider PCG to be a form of secondary OAG, however, it is included in this study, as there are a considerable number of family-based studies that have revealed mutations in CYP1B1 in families affected by PCG that also manifest as adult-onset POAG within the same family (Khan et al., 2011a; Vasiliou and Gonzalez, 2008).
OHT is defined by the presence of elevated IOP without any optic nerve or visual field changes. Approximately 4–7% of U.S. patients greater than 40 years old have elevated IOP, without any signs of glaucomatous optic neuropathy (Kass et al., 2002). However, these patients are at an elevated risk for developing POAG, and do have evidence of RGC loss (Gyatsho et al., 2008).
3. Literature search for gene-POAG associations
Since genome-sequencing technology has become increasingly available and efficient, the number of genes shown to be associated with POAG has grown rapidly over two decades (Rao et al., 2011; Takamoto and Araie, 2014). Therefore, we adopted a bioinformatics-based approach to compile our collection of genes in order to understand their function and role in pathways associated with POAG. We used DisGeNET, a Integrative Biomedical Informatics Group database, to guide our literature review. DisGeNET compiles gene-disease associations from various sources including primary research articles using a text-mining algorithm, as well as curated databases such as UniProt and The Comparative Toxicogenomics Database (CTD) (Bauer-Mehren et al., 2010, 2011; Pinero et al., 2015). The search terms used in DisGeNET were Unified Medical Language System (UMLS) codes. The following codes were used in our literature search: (1) “glaucoma” (umls:C0017601), (2) “glaucoma, open-angle” (3) “primary open angle glaucoma” (umls:C0339573), (4) “low tension glaucoma” (umls:C0152136), (5) “ocular hypertension” (umls:C0028840), (6) “glaucoma of childhood” (umls:C2981140) for JOAG, and (7) primary congenital glaucoma (umls:C1533041).
There are databases similar to DisGeNET, most notably Mala-Cards Human Disease Database, which not only reports gene-disease associations, but also provides summaries of important therapies and clinical trials for each disease (Rappaport et al., 2013). However, the primary source retrieval of MalaCards was more limited, and therefore proved less useful for the purposes of this study. For instance, a search for “primary open-angle glaucoma” in Malacards retrieved 33 potentially associated genes, whereas DisGeNET retrieved 168 genes. Additionally, as an example, OPTN was reported to be associated with POAG by both of the databases, however, Malacards retrieved 12 supporting manuscripts while DisGeNET retrieved 44 manuscripts.
We reviewed each of the manuscripts DisGeNET cited as having a gene association with one of the seven terms used. Many of the associations that DisGeNET cited were false, since the text-mining algorithm does not consistently differentiate between phrases such as “MYOC was found to be associated with POAG” and “MYOC was not found to be associated with POAG.” There were also true associations that DisGeNET failed to report, especially if a table or figure within a paper cited a significant association that was not repeated in the body of the paper. Additionally, some important POAG GWAS were excluded by DisGeNET, primarily because many of the GWAS were not studying POAG itself, but important POAG endophenotypes and risk factors, such as CCT, CDR, and other optic disc parameters. In order to make sure we were including the relevant GWAS studies we surveyed literature reviews on POAG GWAS and the comprehensive GWAS database, GWAS Central GWAS Central at www.gwascentral.org (Abu-Amero et al., 2015; Beck et al., 2014; Janssen et al., 2013). Therefore we added 12 GWAS to the 486 papers selected by DisGeNET which added 17 additional genes to our analyses (Fig. 2). Genes were reported as associated with “POAG” in our study if the study investigating the association looked at patients diagnosed with POAG with IOP ≥21 mmHg, age ≥40 years old at diagnosis, or if the IOP or age of the patient population was not clearly described. Genes associated with glaucoma from functional studies were included if the authors studied ocular tissues, protein, or nucleic acid derived from patients with POAG or animal models exhibiting both elevated IOP and optic nerve damage. Genes were reported as associated with “NTG” if the study investigated patients with IOP <21 mmHg at diagnosis. Genes cited in functional studies were deemed associated with NTG if the experimental model comprised patients or animals with optic nerve damage without an elevated IOP. Genes were reported as associated with “JOAG” if the study investigated patients with POAG and the ages of the patients at diagnosis were <40 years old. Genes were associated doubly with NTG and JOAG if the patients were less than 40 years old and had an IOP <21 mmHg at diagnosis. Genes were reported as associated with “OHT” if the study investigated patients or another model system with IOP ≥21 and there was no evidence of optic nerve or visual field abnormalities.
Fig. 2.
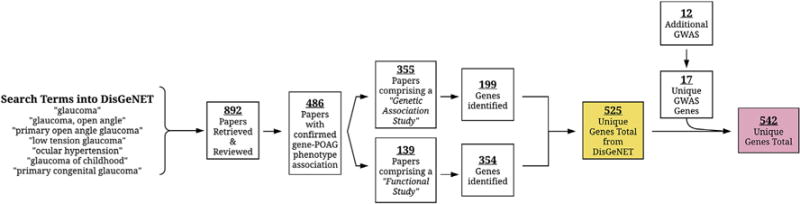
Flowchart summarizing the findings of our comprehensive literature review of 904 studies (892 from DisGeNET and 12 from additional GWAS). 542 genes were confirmed to be associated with POAG and related phenotypes.
The GWAS has emerged as popular tool used to associate genomic SNPs with disease risk. However, despite the incredible value of GWAS, only a limited number of extant molecular therapies target the gene products of genes discovered in such studies. This fact highlights the importance of expanding molecular networks beyond GWAS hits to search for other important regulatory interactions (Cao and Moult, 2014). Therefore, as a general guiding principle for this study, we decided to err on the side of inclusiveness to establish this compendium of genes. We felt this approach would help us compile a more comprehensive collection of genes. This would better to serve as a means to generate more hypotheses by highlighting potentially important, yet understudied loci and genes. We did not include synoptic reviews, or studies looking at secondary forms of OAG, such as pseudoexfoliation glaucoma, or pigment dispersion glaucoma. We did include associations confirmed within meta-analyses. Additionally, studies citing previously discovered gene-glaucoma associations were not included unless the association was confirmed with independent experiments. Additionally, since many different study types were included in this review, we did not set our own thresholds for statistical significance. Instead, we relied upon the individual studies’ thresholds for significance when deeming “true” gene-glaucoma associations. We also recorded the implicated patient populations and/or animal models investigated from each paper, and classified the studies into two large groups of study types, genetic association studies, and functional studies. We defined genetic association studies as those that identified genomic alterations associated with one of the above phenotypes, such as SNPs, microsatellite markers, and CNVs, and meta-analyses summarizing these findings. Additionally, we defined functional studies as those comprising microarray studies, those which illustrated that the expression of the genes of interest were affected by experimental conditions related to POAG, or that variations in gene expression affected ocular tissues related to POAG in a significant way.
4. Pathway analysis and functional annotation
Pathway analysis and functional annotation were performed using ConsensusPathDB (CPDB) and Ingenuity® Pathway Analysis (IPA®, QIAGEN Redwood City, www.qiagen.com/ingenuity) (Kamburov et al., 2009, 2011). The gene lists we wished to analyze according to ocular tissues or POAG-phenotypes were entered into CPDB’s overrepresentation analysis tool and IPA’s core analysis program. In CPDB we specifically searched for enriched sets of genes in pathway-based sets and gene-ontology (GO) categories. The similar pathway with the larger p-value was discarded from the association list. For IPA core analysis we limited our analysis to humans alone.
We reviewed 892 papers retrieved from DisGeNET published between 1988 and May 2016. After considering all functional, genetic-association studies (including 12 GWAS studies that DisGeNET overlooked) we selected 542 genes with cited associations to POAG and the related phenotypes (Supplementary Tables 1 and 2). Supplementary Table 3 includes all of the studies we reviewed from DisGeNET, including those excluded from our pathway analyses. A flowchart summarizing our review process is presented in Fig. 2, and Fig. 3 shows the number of genes associated with our defined POAG-related phenotypes. 295 (54.8%) genes were found to be associated with the POAG phenotype alone. This is not surprising given that many studies, including this one, broadly define “POAG.” 109 (20.3%) genes were associated with OHT alone, most of which derived from functional studies using OHT models to study variation in gene-expression in glaucoma. We attempted to determine the ethnic background of the patient populations studied for each genetic association and found a significant gene-POAG phenotype association (Fig. 4). Many different specific ethnicities were studied within the genetic-association studies we found citing a gene-glaucoma association. We grouped many of the specific ethnicities to reflect the United Nations geoscheme in order to highlight which regions of the globe are over- or underrepresented in glaucoma-genetics research. According to our findings, Europeans are greatly over-represented, while South American and African populations are under-represented, despite their elevated risk for the disease.
Fig. 3.
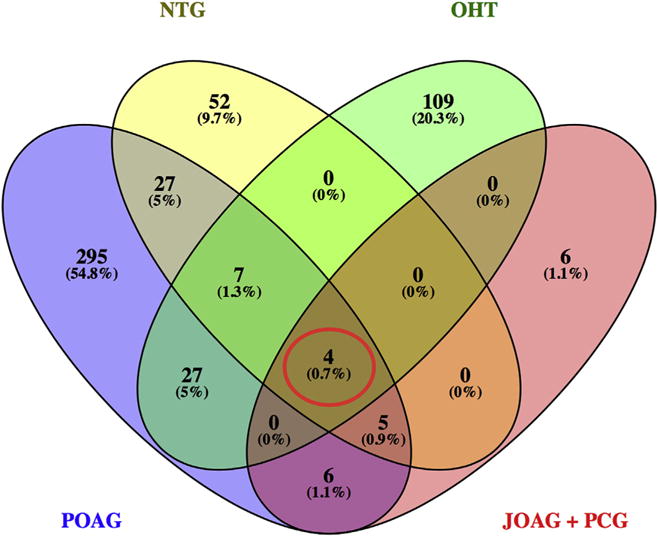
Venn diagram summarizing POAG-associated genes from 891 studies. Among the 522 genes found to be associated with a POAG phenotype, 4 genes (MYOC, CYP1B1, OPTN, WDR36–red circle) were found to be associated with all POAG phenotypes. [POAG = Primary Open Angle Glaucoma, NTG = Normal Tension Glaucoma, OHT = Ocular Hypertension, JOAG = Juvenile Open Angle Glaucoma, PCG = Primary Congenital Glaucoma].
Fig. 4.
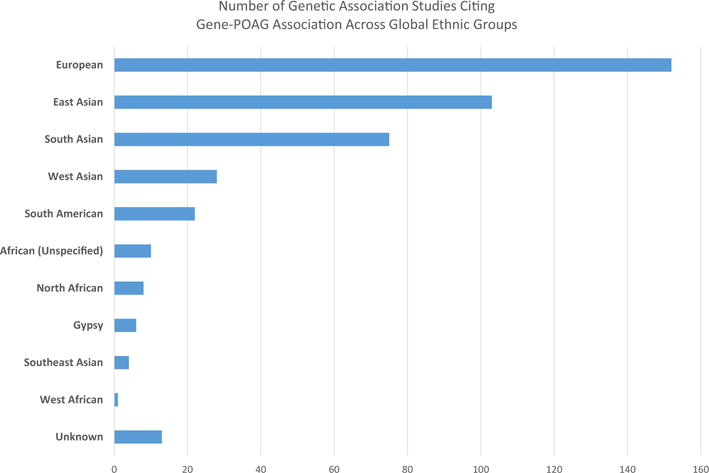
This summarizes the number of studies with a confirmed gene-glaucoma association with subjects from several different ethnic populations. We binned ethnic groups roughly according to the United Nations geoscheme. The vast majority of studies were performed in European, Chinese, Japanese, and Indian populations.
In summary, the structure of this review is such that we used DisGeNET (and additional GWAS) to guide our collection of “POAG genes” which were subsequently used to conduct pathway and network analyses with CPDB and IPA. These pathways and networks were viewed as “results” which we then interpreted and discussed through the lens of additional papers surrounding the molecular biology of glaucoma and neurodegeneration, which may have been excluded by DisGeNET.
4.1. Ocular tissue-specific pathway analyses
4.1.1. Lamina cribrosa gene analyses
Sixty-nine genes associated with a POAG-related phenotype were identified in studies using LC cells or tissue. The gene input lists for tissue based analyses are presented in Supplementary Table 4. Three of the 10 most implicated pathways according to CPDB analysis are shown in Table 1. Most of these pathways are related to ECM organization and ECM fiber formation. This finding is expected, given that 59 (85.5%) of these genes were identified in a microarray study performed by Kirwan et al., where their pathway analysis revealed significant overrepresentation of ECM-related genes when comparing gene expression between LC cells from healthy controls and POAG patients (Kirwan et al., 2009). When the 10 genes that were not identified in this microarray study were analyzed (Supplementary Table 4), “ECM organization” was still significantly overrepresented, along with several pathways closely related to the canonical WNT signaling pathway. This seems to confirm that ECM organization within the ONH is an important role of the LC, and suggests that its perturbation may contribute to the pathogenesis of POAG. Indeed, the study performed by Kirwan et al stated that LC cells from patients suffering from POAG tend to produce a pro-fibrotic milieu, perhaps detrimentally affecting the RGC axons passing through the sieve-like LC (Kirwan et al., 2009).
Table 1.
This table summarizes the pathway analysis results from CPDB for the genes found to be potentially important in the biology of the lamina cribrosa. The CPDB analysis for all of the genes discovered, and those discovered outside of the microarray study by Kirwan et al. are included. The upstream regulators from the IPA analysis of the 69 gene set is also represented.
| Lamina Cribrosa CPDB Pathways (All Genes Included) | |||
|---|---|---|---|
|
| |||
| Pathway | Pathway Source | Pathway-Input Gene Overlap | p-value |
| Extracellular matrix organization | Reactome | TGFB1, TGFB2, DMD, COL11A1, VCAN, MFAP4, FBLN2, FBLN1, LOX, DCN, PLOD2, THBS1, COL18A1, SPARC, P3H2, EFEMP1, ITGA4, COL1A1, COL5A1, BMP4, BMP1, HAPLN1, COL13A1, MMP9, MMP2 | 4.29e-15 |
| Collagen formation | Reactome | COL11A1, BMP1, COL18A1, COL1A1, PLOD2, COL13A1, P3H2, MMP9, LOX, COL5A1 | 7.19e-11 |
| Molecules associated with elastic fibres | Reactome | TGFB1, TGFB2, BMP4, EFEMP1, MFAP4, FBLN2, FBLN1 | 3.73e-08 |
|
| |||
| Lamina Cribrosa CPDB Pathways (Kirwan et al Microarray Excluded) | |||
|
| |||
| Pathway | Pathway Source | Pathway-Input Gene Overlap | p-value |
|
| |||
| Extracellular matrix organization | Reactome | TGFB2, BMP4, VCAN, ITGA4, MMP9, MMP2 | 2.37e-07 |
| Proteoglycans in cancer - Homo sapiens (human) | Kegg | FZD2, MMP9, MMP2, TGFB2, FZD7 | 2.19e-06 |
| Pathways in cancer - Homo sapiens (human) | Kegg | FZD2, BMP4, FZD7, TGFB2, MMP9, MMP2 | 2.70e-06 |
|
| |||
| Lamina Cribrosa Top Upstream Regulators (IPA Core Analysis of All Genes) | |||
|
| |||
| Regulator | Target Genes from Input List | p-value | |
|
| |||
| TGFB1 | ACTA2, BMP1, CDH2, COL18A1, COL1A1, COL5A1, EDN1, ITGA4, MMP2, MMP9, PLOD2, SPARC, SPOCK1, TGFB1, TGFBI, THBS1 | 4.29e-15 | |
| CTNNB1 | APOD, BMP4, CDH2, EDN1, IFITM1, LBH, LMO2, MMP2, MMP9, VCAN | 7.19e-11 | |
IPA analysis with all LC genes did not explicitly highlight ECM organization as a top overrepresented pathway. However, as shown in Table 1, TGF-β1 and β-catenin were found to be the two highest scoring “Upstream Regulators” to the set of input genes. Both molecules were shown to be tightly related to ECM organization in different ocular tissues. It is reported that β-catenin stimulation effectively inhibited TGF-β2-induced overexpression of laminin, collagen I and collagen IV proteins in TM cells (Villarreal et al., 2014). Supplementary Fig. 1 reveals the genes found in studies evaluating LC that are regulated by TGF-β1 and β-catenin.
4.1.2. Optic nerve tissue gene analyses
In 10 studies focusing on optic nerve tissue, excluding isolated ONH astrocytes, 62 genes were found to be possibly implicated in the pathogenesis of POAG (Supplementary Table 4). As shown in Supplementary Table 5, CPDB pathway analysis revealed that “cytokine-cytokine receptor interaction” was the most significantly overrepresented pathway, with 24 of the 62 genes from our input list represented in this pathway. Members of the tumor necrosis factor cytokine/receptor superfamily made up 5 of these 24 input genes, specifically, TNF, LTBR, TNFRSF9, TNFRSF11B, and TNFRSF13B. Increased expression of these genes in the optic nerve was associated with greater RGC death in rodent glaucoma models (Howell et al., 2011; Roh et al., 2012).
The IPA results agreed with CPDB, in that TNF and inflammatory processes were important players in the optic nerve gene set. The top 5 overrepresented canonical pathways from IPA represented inflammatory and immune responses including “Granulocyte Adhesion and Diapedesis,” and “Pathogenesis of Multiple Sclerosis” (Supplementary Table 5). The top associated network function was “Cell-To-Cell Signaling and Interaction, Cellular Movement, Immune Cell Trafficking.” This network reveals that TNF does indeed appear to play a central role in this gene set (Supplementary Fig. 2).
4.1.3. Isolated ONH astrocyte gene analyses
Fifteen genes were implicated in the pathogenesis of POAG from studies using isolated ONH astrocytes (Supplementary Table 4). The most significantly overrepresented pathways according to CPDB include “focal adhesion,” “cytokine-cytokine receptor interaction,” “ECM organization,” and “Signaling by VEGF” (Table 2). Interestingly, TGFB2 was included among the list of candidate genes involved in “cytokine-cytokine receptor interaction” and “ECM organization” (Miao et al., 2010; Zode et al., 2009). IPA for the ONH astrocyte gene set confirmed TGF-β signaling as a significantly overrepresented biological process, and showed that targets of the transcription factors, SP1 and ZNF652, were significantly over-represented in our gene set (Table 2). SP1 is required for the induction of certain TGF-β-regulated events in cancer, including angiogenesis and cell survival, and is overexpressed in cortical neurons experiencing oxidative stress (Ellenrieder, 2008; Jungert et al., 2007; Ryu et al., 2003; Solomon et al., 2008). ZNF652 represses the transcription of TGFB2, TGFBR2, and SMAD2, potentially playing a key role in the TGF-β signaling cascade (Kumar et al., 2011; Neilsen et al., 2013). IPA also revealed “Cardiovascular System Development and Function, Cellular Movement, Cellular Development” as the most highly associated network function for the ONH astrocyte gene set. Fig. 5 reveals that many of the molecules from the ONH astrocyte gene set act directly or indirectly act on the NFκB complex, which has been shown to play an important role in gliosis and subsequent neurodegeneration in Parkinson’s disease and Alzheimer’s disease again suggesting that inflammation and the stress response may be important players within the ONH astrocytes during the pathogenesis of POAG (Blesa and Przedborski, 2014; Hayden and Ghosh, 2012; Salminen et al., 2009).
Table 2.
This table summarizes the results of CPDB’s pathway analysis of the 15 genes potentially important in the biology of ONH astrocytes in functional studies investigating POAG. Additionally, the top 4 most significantly associated upstream regulators from IPA’s analysis of the same gene set is presenteddall of which have regulatory functions for the TGF-β2 signaling cascade.
| Optic Nerve Head Astrocyte Top CPDB Pathways | |||
|---|---|---|---|
|
| |||
| Pathway | Pathway Source | Pathway-Input Gene Overlap | p-value |
| Focal Adhesion | Wikipathways | PDGFA, EGFR, MYLK, ITGA6, VEGFC, FLT1 | 9.73e-08 |
| Cytokine-cytokine receptor interaction - Homo sapiens (human) | KEGG | PDGFA, TGFB2, TGFBR2, EGFR, VEGFC, FLT1 | 6.95e-07 |
| PI3K-Akt signaling pathway - Homo sapiens (human) | KEGG | PDGFA, EGFR, ITGA6, VEGFC, ANGPT1, FLT1 | 3.30e-06 |
| Extracellular matrix organization | Reactome | BMP7, TGFB1, CD44, FN1, SDC1, CD47, SDC4, SPP1, ITGA6, COL5A2, AGRN, COL1A2, DAG1, COL4A1, COL1A1, ITGB1, COL3A1, LAMC1, SDC2 | 1.71e-05 |
| Signaling by VEGF | Reactome | PDGFA, ANGPT1, EGFR, VEGFC, FLT1 | 2.04e-05 |
|
| |||
| Optic Nerve Head Astrocyte Top Upstream Regulators (IPA Core Analysis) | |||
|
| |||
| Regulator | Target Genes from Input List | p-value | |
|
| |||
| ZNF652 | EGFR, TGFB2, TGFBR2 | 1.09e-08 | |
| SP1 | BMP4, EGFR, FLT1, GJA1, MYLK, PDGFA, TGFB2, TGFBR2 | 1.27e-08 | |
| Estrogen receptor | EGFR, PDGFA, TGFB2, TGFBR2, VEGFC | 1.99e-07 | |
| TGFB1 | COL18A1, ELN, FLT1, ITGA6, PDGFA, TGFB2, TGFBR2 | 1.97e-06 | |
Fig. 5.
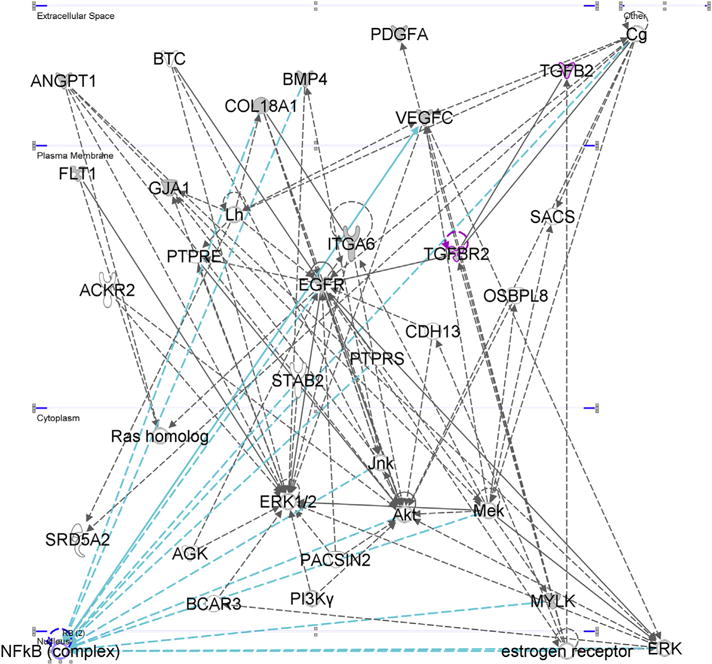
Top associated network function for ONH astrocyte genes: “Cardiovascular System Development and Function, Cellular Movement, Cellular Development.” Many of the genes converge onto the NFκB complex (within the nucleus) suggesting that inflammation and cell survival are important biological processes within ONH astrocytes during POAG’s pathogenesis.
4.1.4. Retina & RGC gene analyses
Twenty-one functional studies focused on retina tissue and retinal progenitor cells, but not isolated RGCs. 47 genes were associated with model systems exhibiting OHT, POAG, or NTG phenotypes (Supplementary Table 4). The most strongly associated pathways with this gene set appeared to fall within a wider theme of cell-cycle regulation, with “IL6-mediated signaling events,” “Oncostatin M Signaling Pathway,” “Cellular Senescence,” and “Apoptosis Modulation and Signaling” (Table 3). Many of the genes involved in these pathways, like JUN, FOS, CDKN2A, and TP53, were upregulated in experimental rodent models of glaucoma (induced OHT, optic nerve transection), and likely contribute to glaucoma’s characteristic optic nerve atrophy by inducing apoptosis in RGCs or the supporting retinal glia (He et al., 2013; Yang et al., 2007).
Table 3.
This table summarizes the results of CPDB’s pathway analysis of the 21 genes potentially important in the biology of retinal tissue and isolated RGCs in functional studies investigating POAG. Additionally, the top 2 most significantly associated upstream regulators from IPA’s analysis of the same gene set is presented—all of which have regulatory function for TNF-α signaling and cellular senescence.
| Retina Tissue Top CPDB Pathways (Excluding Studies Investigating RGCs Alone) | |||
|---|---|---|---|
|
| |||
| Pathway (Rank) | Pathway Source | Pathway-Input Gene Overlap | p-value |
| IL6-mediated signaling events | PID | CEBPB, JUNB, STAT3, STAT1, TIMP1, CEBPD, FOS,JUN | 5.47e-12 |
| Oncostatin M Signaling Pathway | Wikipathways | CEBPB, TP53, STAT1, FOS, CDKN1B, JUNB, VEGFA, STAT3 | 8.36e-11 |
| AP-1 transcription factor network | PID | CDKN2A, TP53, TIMP1, FOS, CDKN1B, JUNB, JUN, ATF3 | 1.54e-10 |
| Cellular responses to stress | Reactome | CDKN2B, CEBPB, CDKN2A, TP53, HSPB1, SOD1, CDKN1B, FOS, JUN, TXN2, TXN | 6.57e-08 |
| Cellular Senescence | Reactome | CDKN2B, TXN, CDKN2A, TP53, CEBPB, FOS, CDKN1B, JUN | 2.83e-07 |
|
| |||
| Retinal Ganglion Cell Top CPDB Pathways | |||
|
| |||
| Pathway | Pathway Source | Pathway-Input Gene Overlap | p-value |
| SHP2 signaling | PID | TGFB2, BMP4, VCAN, ITGA4, MMP9, MMP2 | 1.68e-12 |
| BDNF signaling pathway | Wikipathways | FZD2, MMP9, MMP2, TGFB2, FZD7 | 2.18e-12 |
| Neurotrophic factor-mediated Trk receptor signaling | PID | FZD2, BMP4, FZD7, TGFB2, MMP9, MMP2 | 2.33e-10 |
| p75(NTR)-mediated signaling | PID | NGF, LINGO1, XIAP, NTRK1, BAD, NGFR, BDNF | 4.99e-10 |
| Apoptosis - Homo sapiens (human) | KEGG | NGF, BAD, NTRK1, XIAP, TNF, PPP3CA | 8.82e-08 |
|
| |||
| Retinal Ganglion Cell Top Upstream Regulators (IPA Core Analysis) | |||
|
| |||
| Regulator | Target Genes from Input List | p-value | |
|
| |||
| Ap1 | EDN1, NGF, SNCG, TNF | 2.14e-07 | |
| NFkB (complex) | BAD, BECN1, EDN1, EDNRB, TNF, XIAP | 1.16e-06 | |
We identified 24 studies citing associations between 32 genes and POAG phenotypes focusing on genetic alterations specifically seen in RGCs (Supplementary Table 4). Some of the pathways included “SHP2 signaling,” “BDNF signaling pathway,” “Apoptosis,” and “p75NTR regulates axonogenesis”, suggesting broader themes involving neuronal survival and development (Table 3). While the SHP2 gene itself was not within our RGC gene set, “SHP2 signaling” was the most significantly overrepresented pathway. Cai et al. reported that Shp2 ablation in mice triggered apoptosis in all retinal cell types, which could be reversed by constitutively activating Kras signaling, revealing that the Ras-MAPK signaling pathway may play an important role in RGC survival (Cai et al., 2011). This agrees with our pathway analysis as the “Ras signaling pathway” and “MAPK signaling pathway” were the 10 t h and 12 t h most overrepresented pathways in our gene set, respectively.
IPA of the RGC genes largely agreed with CPDB’s outputs (Table 3). “Axonal Guidance Signaling” and “Neurotrophin/TRK Signaling” were the 1st and 3rd top associated canonical pathways in the RGC gene input list, respectively. The top 5 associated diseases and biological functions included “neurological disease,” “connective tissue disorders,” and “inflammatory disease.” Additionally, the most strongly associated upstream regulators, were AP1 and NFκB, important inflammatory regulators. The top associated network for this gene set was entitled “Cell Death and Survival, Nervous System Development and Function, Tissue Morphology” (Fig. 6).
Fig. 6.

“Cell Death and Survival, Nervous System Development and Function, Tissue Morphology.” This was the top most associated network according to IPA core analysis of the RGC gene input set. Molecules within the ERK and MAP families seem to play an important role, as do the Ap1 and Creb transcription factors. The ERK1/2 family is highlighted above with the blue arrows highlighting interactions with its immediate neighbors, including VEGFA, TNF, NGF, and NRTK2.
4.1.5. Trabecular meshwork gene analyses
Forty-nine studies were identified which cited associations with 139 genes involved in TM physiology (Supplementary Table 4). The top overrepresented pathways within this gene set include “AP-1 transcription factor network,” “Senescence and Autophagy in Cancer,” “TGF Beta Signaling Pathway,” “Copper Homeostasis,” and “Extracellular matrix organization” (Table 4). This collection of pathways suggests that ECM regulation and cell-cycle regulation are important processes within the TM in POAG-related phenotypes such as OHT and HTG. It is important to note that 79 (56.8%) of these POAG-associated genes were exclusively found in a microarray study comparing gene expression profiles between steroid-treated and untreated TM cells in an attempt to model OHT conditions (Supplementary Table 3) (Fan et al., 2008). We repeated pathway analysis without these 79 genes to avoid study bias and discovered that the “TGF Beta signaling pathway,” “extracellular matrix organization,” and “Senescence and Autophagy in Cancer” remained in the top 10 most overrepresented pathways.
Table 4.
This table summarizes the results of CPDB’s pathway analysis of the 139 genes potentially important in the biology of trabecular meshwork tissue and cells in functional studies investigating POAG phenotypes. Additionally, 4 of the most significantly associated upstream regulators from IPA’s analysis of the same gene set is presented—all of which have regulatory functions for cellular senescence.
| Trabecular Meshwork Top CPDB Pathways (All Genes Included) | |||
|---|---|---|---|
|
| |||
| Pathway (Rank) | Pathway Source | Pathway-Input Gene Overlap | p-value |
| AP-1 transcription factor network | PID | CCL2, TGFB1, EGR1, MT2A, FOS, JUNB, TCF7L2, CTNNB1, PLAU, MMP9, GJA1, MMP1 | 1.11e-11 |
| Senescence and Autophagy in Cancer | Wikipathways | TGFB1, IRF7, BMP2, FN1, INHBA, CD44, PLAU, GSK3B, SMAD3, IGFBP3, IL1B, THBS1 | 1.48e-09 |
| TGF Beta Signaling Pathway | Wikipathways | TGFB1, BMP4, FOS, SMAD2, THBS1, CTNNB1, INHBA, FST, SMAD3 | 7.21e-09 |
| Copper homeostasis | Wikipathways | MT1X, MT2A, GSK3B, MT1L, MT1H, MT1E, MT1F, MT1G | 1.33e-07 |
| Extracellular matrix organization | Reactome | TGFB1, TGFB2, BMP4, LAMA4, BMP2, FN1, CD44, P4HB, THBS1, COL13A1, MMP9, PCOLCE2, MMP2, FBLN5, MMP1 | 1.78e-07 |
|
| |||
| Trabecular Meshwork Top Upstream Regulators (IPA Core Analysis) | |||
|
| |||
| Regulator | Target Genes from Input List | p-value | |
|
| |||
| TNF | BMP2, CASP4, CAV1, CCL2, CD44, CDH11, CDH2, CYP1B1, CYP27A1, EGR1, ENPP2, FAS, FN1, FOS, FST, IER3, IGFBP2, IGFBP3, IL1B, INHBA, JUNB, MGP, MMP1, MMP2, MMP9, MT1L, PLAU, PPIF, PTGFR, PTGS2, SAA1, SFRP1, SNCG, SOAT1, TFRC, TGFB1, TGM2, THBS2, TJP1, TSC22D3 | 9.21e-26 | |
| TGFB1 | ACTA2, CASP4, CAV1, CCL2, CD44, CDH11, CDH2, CTGF, EDNRA, ELK3, FAS, FBLN5, FN1, FOS, IGFBP2, IGFBP3, IL1B, INHBA,JUNB, MMP1, MMP2, MMP9, PLAU, PTGS2, SCD, SFRP1, SMAD3, SPOCK1, TGFB1, TGFB2, TGFBI, TGM2, THBS1 | 8.69e-24 | |
| TP53 | IER3, IGFBP2, IGFBP3, IL1B, INHBA, IRF7, JUNB, MMP2, MMP9, MT1L, MYO6, P4HB, PALLD, PLK2, PRDX6, PTGS2, RAB8A, SEMA6A, SFRP1, SFRP2, TCF7L2, TGFB1, TGFB2, TGFBI, TGM2, THBS1, TJP1, TSC22D3, VCAN | 1.85e-23 | |
| P38 MAPK | CCL2, CD44, CTGF, DDIT3, EGR1, FAS, FN1, FOS, FST, HSPA5, IER3, IL1B, INHBA, IRF7, JUNB, MMP1, MMP2, MMP9, PTGS2, S100A12, TGFB1, THBS1 | 9.89e-22 | |
The top upstream regulators identified for this gene set included TNF, TGFB1, TP53, and P38 MAPK, and the top associated network was “Tissue Development, Cardiovascular System Development and Function, Organismal Development” (Fig. 7). When the genes discovered only in the Fan et al. microarray study were removed, TGFB1, P38 MAPK, TNF, and NFκB were the top upstream regulators and “Cellular Movement, Cell Death and Survival, Cellular Development,” became the most associated pathway. The above findings highlighted the importance of the TGF-β signaling cascade and its association with ECM regulation in the TM. Several of the studies we reviewed described various pathological effects TGF-β has on TM cells. For instance, treating TM cells from healthy donors with TGF-β2 resulted in a phenotype similar to that seen TM cells derived from patients with POAG, such as increased ROS production, elevated ECM component expression, and higher rates of cellular senescence (Fatma et al., 2009; Yu et al., 2010). It is thought that persistent ECM turnover and remodeling is important for the proper functioning of the TM and for maintaining adequately low resistance to aqueous outflow (Acott and Kelley, 2008; Vranka et al., 2015). Therefore, we infer that perturbation of balance of ECM biogenesis, such as TGF-β-induced TM cell senescence or ECM overexpression, could result in the development of OHT, eventually leading to glaucoma.
Fig. 7.

“Tissue Development, Cardiovascular System Development and Function, Organismal Development.” This was the top most associated network according to IPA core analysis of the TM gene input set which included the microarray study by Fan et al. The TGF-β signaling pathway played an important role in this network.
4.2. Phenotype-specific pathway analysis
4.2.1. Primary open-angle glaucoma gene analyses
Several studies pursuing functional gene interactions in POAG limited the number of genes they analyzed to a select few following a stricter exclusion criteria. Most notably, in the study by Janssen et al, 2013 over 60 genes were deemed to be likely POAG candidates, on which IPA core analysis was performed. The top four networks produced by IPA were entitled: (1) “Small molecule biochemistry, molecular transport, cardiovascular system development and function,” (2) “Cell morphology, lipid metabolism, molecular transport,” (3) “Tissue morphology, visual system development and function, embryonic development,: (4) “Ophthalmic disease, cell cycle, connective tissue development and function ” (Janssen et al., 2013). To draw comparisons with our data, we performed CPDB pathway analysis on this gene set (Supplementary Table 6) and found that the most significantly overrepresented pathways were “Nuclear receptors Meta-pathway,” “MAPK signaling,” “Legionellosis,” “NFκB signaling pathway,” and “Spinal cord injury” (Supplementary Table 7). Additionally, the top upstream regulators of their gene set included LDL, estrogen receptor, IL18, and SP1 (Supplementary Table 7). Overall these findings agree with many of our findings, suggesting molecular networks associated with inflammation and cell cycle regulation are important in the pathogenesis of POAG.
Before investigating larger POAG candidate gene networks, we also performed analyses on more exclusive gene sets with greater numbers of genes associated to the disease. We selected 51 genes associated with POAG, NTG, OHT and related endophenotypes (CCT, CDR, disc size) that had been implicated in GWAS (Supplementary Table 6). The most strongly associated pathways for this gene set using CPDB pathway analysis included the “glucocorticoid receptor pathway,” “RXR and RAR heterodimerization with other nuclear receptor,” “TGF beta signaling pathway,” and “NRAGE signals death through JNK” (Supplementary Table 8) Additionally the top upstream regulators included NPC1 (NPC Intracellular Cholesterol Transporter 1), estrogen receptor, and NR1H (also known as an LXR for liver X receptor).
The top associated network constructed by IPA was entitled “Lipid Metabolism, Molecular Transport, Small Molecule Biochemistry” (Supplementary Fig. 3). Again, these findings suggest that inflammation and cell cycle, cholesterol metabolism and steroid hormone signaling may play important roles in glaucoma. The RXRA gene, which was included in several of CPDB’s pathways, interacts with the upstream regulator NR1H (or LXR) to regulate lipid metabolism, inflammation, and macrophage activation (Costet et al., 2000; Korf et al., 2009). RXRA was included in the gene input list as several GWAS have identified SNPs within the 9q34.2-q34.3 region between genes RXRA and COL5A1 (Cornes et al., 2012; Hoehn et al., 2012; Vithana et al., 2011). While this association does not provide sure evidence that either of these genes play a role in the development of POAG, it is interesting to note that within IPA network one of this gene set, RXRA is shown to induce expression of ABCA1 when it dimerizes with LXRα in cholesterol-loaded macrophages (Supplementary Fig. 3) (Costet et al., 2000). RXRA also triggers the expression of TNF-α, another important POAG locus when it dimerizes with LXR in response to elevated cholesterol levels (Landis et al., 2002; Zhang et al., 2002). Finally, we were intrigued to find that NRAGE signaling has been implicated in RGC apoptosis (Lebrun-Julien et al., 2010). Specifically, Müller cells in mice null for NRAGE produce significantly less TNF-α, leading to reduced RGC death (Lebrun-Julien et al., 2010).
Next we only analysed those genes that have been found to have an association with a POAG phenotype (including NTG, HTG, OHT, PCG, JOAG and related endophenotypes) in at least one functional study and one genetic-association study. This gene set consists of 30 genes including well known POAG loci such as CDKN2B, OPTN, MYOC, and CYP1B1 as well as lesser studied genes such as CDH11 and BMP7 (Supplementary Table 6). The top scoring pathways from CPDB included “Spinal Cord Injury,” “AP-1 transcription factor network,” “Activation of Matrix Metalloproteinases,” “Extracellular Matrix Organization,” and “Oncogene Induced Senescence” (Supplementary Table 9). “Activation of Matrix Metalloproteinases” and “Extracellular Matrix Organization” were primarily built upon genes which have not been implicated in GWAS, including MMP1, MMP2, MMP9, and COL18A1. However, several candidate gene studies and functional studies have shown that they may play a role in tissue remodeling in both the TM and ONH (Supplementary Tables 1 and 2) (Guo et al., 2012; Majsterek et al., 2011; Markiewicz et al., 2013; Rudzinski et al., 2008; Wiggs et al., 2013). Interestingly, the top associated diseases to this gene set were POAG and osteoarthritis, and the two top upstream regulators for this gene input list were TERT (telomerase reverse transcriptase) and TGFB1 (Supplementary Table 9). Dysregulation of TERT (telomerase reverse transcriptase) has been shown to be involved in the pathogenesis of chronic inflammatory states, and also has an important role in regulating apoptosis and cellular senescence (Cao et al., 2002; Kordinas et al., 2016). Limited research has been performed to determine the specific role of TERT in neurodegenerative diseases, however, there is some evidence that the absence of TERT in microglia and hippocampal neurons may increase the risk of the development of tauopathies, like Alzheimer’s disease (Spilsbury et al., 2015). The highest scoring IPA network for this gene set, “Cellular Development, Connective Tissue Development and Function, Tissue Development,” (Supplementary Fig. 4) revealed important interactions between inflammatory regulators, such as TNF-α, and IL1B with the PI3K complex, through which these cytokines activate the NFκВ complex (Reddy et al., 1997, 2000).
We also combined the GWAS and functional-genetic association study overlap gene sets, expanding the input set to 75 genes, and performed IPA and CPDB pathway analysis (Supplementary Table 6). Mostly, the results were very similar to the GWAS and functional-genetic association overlap analyses. According to IPA, the most highly associated diseases to this gene set were glaucoma, connective/soft tissue tumors, calcific aortic valve stenosis, diabetes mellitus, and osteoarthritis further suggesting that the genes were important in ECM organization, cell cycle regulation, and inflammation. The most highly associated pathways from CPDB also included “Validated transcriptional targets of AP1 family members Fra1 and Fra2,” “Spinal cord injury,” and “Activation of Matrix Metalloproteinases” (Supplementary Table 10). The Fra1 and Fra2 transcription factors are important regulators of ECM remodeling, fibrosis, and inflammation (Karreth et al., 2004; Rajasekaran et al., 2013). The top upstream regulators for this gene input list included TERT, TGFB1, and the estrogen receptor (Supplementary Table 10). In the animal model studies of spinal cord injury it is reported that administration of estrogen results in the upregulation of anti-inflammatory cytokines, and the down-regulation of pro-inflammatory cytokines, including IL-1β and TNF-α (Samantaray et al., 2011; Shivers et al., 2015). We find these findings interesting since these models are similar to the optic nerve transection and crush models of glaucoma, and epidemiological evidence suggests that estrogen shows therapeutic potential for POAG in the future (Dewundara et al., 2016). The highest scoring IPA network for this gene set was “DNA Replication, Recombination and Repair, Cell Cycle, Cellular Development” (Supplementary Fig. 5). This network portrayed significant molecular convergence onto the NFκB complex and TP53 which interacted with important POAG GWAS loci involved with cell cycle regulation, senescence, and inflammation such as TGFBR3, CDKN2B, CDKN2B-AS1, and OPTN.
To cast a wider “gene net” and highlight other potentially implicated molecular networks and pathways that may have been overlooked with our stricter inclusion criteria, we also performed pathway analyses on 360 genes thought to be associated with the POAG phenotype from a total of 331 genetic-association and functional studies (Supplementary Table 6). The most over-represented pathway according to CPDB was “extracellular matrix organization.” Other highly overrepresented pathways included “cytokine-cytokine receptor interactions,” “Senescence and Autophagy in Cancer,” and “Spinal Cord Injury” (Table 5).
Table 5.
This table summarizes the results of CPDB’s pathway analysis and IPA’s core analysis of the 353 genes potentially important in development of POAG from all study types.
| Primary Open-Angle Glaucoma CPDB Pathways (All Genes Included) | |||
|---|---|---|---|
|
| |||
| Pathway (Rank) | Pathway Source | Pathway-Input Gene Overlap | p-value |
| Extracellular matrix organization | Reactome | COL15A1, MMP1, FBLN5, ITGA4, DCN, TIMP1, DAG1, FBLN1, SDC1, BMP2, FBLN2, COL1A2, BMP1, P3H2, EFEMP1, THBS1, COL1A1, COL11A1, BMP4, HAPLN1, COL4A1, LOXL1, LTBP2, COL3A1, JAM2, FN1, P4HB, MMP9, TGFB1, COL13A1, CD47, FGF2, SPP1, MFAP4, PDGFA, ITGA6, MMP2, TGFB2, SPARC, PLOD2, CDH1, DMD, VCAN, BMP7, SDC2, PCOLCE2, COL8A2, COL18A1, A2M, CD44, ITGA2B, COL5A2, AGRN, LOX, ITGA2, SDC4, LAMC1, ITGB1, COL5A1 | 3.17e-37 |
| Cytokine-cytokine receptor interaction - Homo sapiens (human) | KEGG | IL13RA1, TNFRSF9, PDGFA, EPO, IL20RB, IL18RAP, TNFRSF11B, BMP2, BMP7, LEPR, IL1R2, NGFR, CCL3, CCR1, CCL4, CCL5, CCR4, CXCL11, CCR5, INHBA, CNTF, VEGFA, VEGFC, LTBR, TGFB2, TNFRSF1A, TGFBR2, FAS, FLT1, OSM, CSF1, CSF1R, CSF2RB, EGFR, IL6, IL2RG, TNFRSF13B, TNF, OSMR, IL1A, IL1B, TGFB1, IL3RA | 3.16e-21 |
| Senescence and Autophagy in Cancer | Wikipathways | IGF1, IL1B, IL1A, THBS1, MAP1LC3A, IL6, MAP2K1, PLAT, SPARC, TP53, BMP2, JUN, CDKN1B, CDKN1A, CD44, CDKN2A, SMAD3, CCL3, BECN1, INHBA, COL1A1, COL3A1, FN1, TGFB1 | 9.05e-16 |
| Spinal Cord Injury | Wikipathways | PTGS2, IL1B, IL1A, C1QB, EGFR, TNF, APEX1, IL6, NGFR, ANXA1, BDNF, TP53, TLR4, CDKN1B, CD47, COL4A1, GJA1, FOS, VCAN, NOS2, MMP9, PPP3CA, MBP, TGFB1 | 1.00e-14 |
|
| |||
| Top Upstream Regulators of Primary Open-Angle Glaucoma (IPA Core Analysis of All Genes) | |||
|
| |||
| Regulator | Target Genes from Input List | p-value | |
|
| |||
| TNF | ACTA2, ADM, ALDH2, APOE, BMP2, CCL3, CCL4, CCL5, CCR4, CD44, CD47, CDH1, CDH11, CDH2, CDKN1A, CFD, COL1A2, CSF1, CXCL11, EDN1, EGFR, EPO, FAS, FLT1, FN1, FOS, FST, IL1A, IL1B, IL6, INHBA, ITGA4,JUN, LCN2, LTBP2, MMP1, MMP2, MMP9, MYLK, NFKB1, NGF, NOS2, OSMR, PLOD2, PTGS2, RFTN1, RXRA, SAA1, SDC4, SELE, SOD2, STAT1, TAP1, TDRD7, TGFB1, TGFBR2, TIMP1, TIMP3, TJP1, TLR2, TLR4, TM4SF1, TNF, TNFRSF11B, TP53, VEGFA, VEGFC, XIAP | 3.52e-42 | |
| TGFB1 | ACTA2, AKR1C1/AKR1C2, BMP1, BMP7, CCL5, CD44, CDH1, CDH11, CDH2, CDKN1A, CDKN1B, CDKN2B, COL18A1, COL1A1, COL1A2, COL3A1, COL5A1, CSF1R, CTGF, CTNNB1, EDN1, ESR2, FAS, FGF2, FLT1, FN1, FOS, IGF1, IL6, ITGA2, ITGA4, ITGA6, ITGB1, JUN, JUNB, LTBP2, MMP1, MMP2, MMP9, PDGFA, PLOD2, PTGS2, SDC1, SMAD3, SPARC, SPOCK1, TGFB1, TGFBI, TGFBR2, TGM2, THBS1, TIMP1, TNF, TP53, VEGFA | 6.07e-38 | |
| SP1 | ATF3, BMP4, CAV1, CDH1, CDH2, CDKN1A, CDKN1B, CDKN2A, CDKN2B, CEBPD, COL1A1, CR2, EGFR, ESR1, FGF2, FLT1, FN1, GJA1, HK2, HSD17B1, HSPA5, ITGA2, JUN, MMP2, NF1, NFKB1, NGFR, NOS3, NTF4, NTRK1, OGG1, PTGS2, PTN, SMAD3, SNCG, SOD2, SPP1, STAT1, TFPI2, TGFB1, TGFB2, TGFBR2, TLR2, TNF, TP53, VEGFA | 1.4e-32 | |
| NFκB (complex) | BAD, BECN1, BMP2, CCL3, CCL4, CCL5, CD44, CDKN1A, CLU, CTNNB1, CXCL11, CYP3A4, EDN1, EDNRB, FAS, FTH1, IL1B, IL6, ITGB1, JUNB, LCN2, MMP1, MMP2, MMP9, NFKB1, NOS2, PTGS2, RFTN1, SDC4, SENP1, SMAD3, SOD2, TAP1, TFPI2, TGFB1, TGM2, TNF, TNFRSF1A, TP53, VEGFC, XIAP | 1.3e-28 | |
| ERBB2 | BMP1, BMP7, CDH11, CDH2, CDKN1A, CDKN1B, CDKN2B, COL18A1, COL1A1, COL5A1, CSF1R, CTGF, EDN1, EGFR, FN1, FOS, IL1A, IL6, ITGA2, ITGB1, JUN, JUNB, LCN2, LTBP2, MMP1, PTGS2, SDC1, SMAD3, SPOCK1, THBS1, TNF, TP53, VEGFA, VEGFC | 3.45e-25 | |
IPA analysis of all 360 genes revealed that TNF, TGFB1, SP1, the NFκB complex, and ERBB2 (also known as HER-2/neu) were the top 5 upstream regulators (Table 5). Additionally, various neoplasms, connective tissue disorders, inflammatory diseases, and dementias (including Alzheimer’s disease) ranked among the most strongly associated diseases according to IPA analysis. Similar to our more exclusive analyses, these results also appear to suggest that, ECM organization and inflammatory cytokine signaling might be important processes in POAG’s pathophysiology. However, the top associated network for this gene set, entitled “Cell Cycle, Skeletal and Muscular System Development and Function, Cellular Development” (Fig. 8) highlighted connections well known POAG loci have with molecules which have not been as closely studied within the realm of glaucoma research. For instance, this network shows how TGF-β signaling promotes the expression of the proteoglycan versican (VCAN) and the enzyme encoded by PLOD2, a membrane-bound enzyme responsible for catalyzing collagen crosslinking during fibrosis (van der Slot et al., 2003). VCAN was included in the gene input set since two separate functional studies have shown that its expression is significantly upregulated in LC cells from POAG patients compared to LC cells from healthy controls (Kirwan et al., 2009; Luo et al., 2013). PLOD2 was also included since it was found to be upregulated in LC cells from patients with POAG (Luo et al., 2013). Interestingly, both VCAN and PLOD2 have been found to be associated with ocular disorders like Wagner syndrome (Brezin et al., 2011; Kloeckener-Gruissem et al., 2006; Rothschild et al., 2013) and Axenfeld-Rieger syndrome (Holmberg et al., 2004; Weng et al., 2008) respectively, other than POAG. VCAN was of particular interest to us as there is a growing body of evidence that its upregulation promotes an inflammatory response and may play a role in the pathogenesis of neurodegenerative processes (Zhang et al., 2011, 2012) VCAN can promote inflammation and senescence in a number of ways, such as activating monocytes, promoting the expression of TNF-α, and concentrating TGF-β levels in the extracellular space (Choocheep et al., 2010; Kim et al., 2009; Zhang et al., 2012). Overall, we found including the lesser known genes with cited associations to POAG yielded extremely interesting results which may influence our future research interests in unraveling the relationship between optic nerve degeneration and inflammation.
Fig. 8.

“Cell Cycle, Skeletal and Muscular System Development and Function, Cellular Development.” This was the top most associated network according to IPA core analysis of the POAG gene input set which included all 360 genes from functional and genetic association studies investigating the POAG phenotype.
4.2.2. Normal tension glaucoma gene analyses
We analyzed 65 genes that had been associated with either POAG or NTG in a GWAS or in both a functional study and genetic association study (Supplementary Table 6). Had NTG been considered alone, this gene set would have been too limited to retrieve significant results from CPDB or IPA. The top pathways from CPDB included “Proteoglycans in cancer,” “Spinal cord injury,” “Cell cycle: g1/s checkpoint,” and “RXR and RAR heterodimerization” (Supplementary Table 11). IPA’s top upstream regulators included PROC (protein C), the estrogen receptor, and TERT (Supplementary Table 11). Protein C is best known for its role in anticoagulation, but also serves an anti-inflammatory role and can reduce apoptosis in response to ischemia and inflammation in endothelial cells (Esmon, 2003). IPA’s top associated diseases and bio-functions included “Steroid metabolism,” “Familial glaucoma,” and “Formation of the eye” and the top scoring IPA network was entitled “Respiratory System Development and Function, Tissue Morphology, Lipid Metabolism” (Supplementary Fig. 6). This network included several top POAG genes including CARD10, OPTN. TNF, ABCA1, APOE, and TLR4. There was significant convergence upon TNF-α, Ap1, ERK (Extracellular Signal-Regulated Kinase), and IL12 within this network. Persistent activation of ERK may contribute to neurodegeneration, and ERK pathway inhibition has been associated with lower rates of RGC death (Colucci-D’Amato et al., 2003; Luo et al., 2007; Subramaniam et al., 2004) Mice deficient in an IL12 subunit have been shown to be resistant to the development of autoimmune responses to joint and retinal antigens (Teng et al., 2015). Our analysis suggests that many of these genes have roles in apoptosis, inflammatory responses, and cell cycle regulation.
We expanded our NTG analyses to include all 95 genes with associations to the NTG phenotype in all of the reviewed functional and genetic-association studies (Supplementary Table 6). CPDB analysis revealed “MAPK Signaling,” “Fc epsilon RI signaling pathway,” “Pathways in Cancer,” and “estrogen metabolism” to be the most overrepresented pathways (Table 6). GO analysis revealed significant associations of NTG with processes involved in nervous system development and the cell cycle including: “system development,” “cell differentiation,” “neurogenesis,” “nervous system development,” and “neuron differentiation.” ECM organization and inflammatory processes, which were highly represented in the POAG phenotype appeared to be less important in NTG.
IPA agreed with CPDB’s analysis. Fc epsilon RI signaling was also represented in IPA’s top 5 most associated canonical pathways. FcεRI is a mast cell receptor for the Fc component of IgE. Optic nerves subject to traumatic, vaso-occlusive, or inflammatory processes have significantly higher optic nerve mast cell levels compared to healthy control optic nerves (Levin et al., 1993). More recently it has been discovered that elevated IgE titers in relation to cockroach and cat allergens are associated with a significantly higher risk of glaucoma (angle status was not specified) (Tseng et al., 2015). The top 5 most implicated upstream regulators were: NPC1, a cholesterol trafficking regulator; ESR1, an estrogen receptor; CDKN2B-AS1, a long non-coding RNA; CBX8, a polycomb group protein; and DIAPH1, an actin polymerization and neurite outgrowth regulator (Table 6). This diverse set of upstream regulators suggests that there is a diverse and complex array of biological processes implicated in the development of NTG.
Table 6.
This table summarizes the results of CPDB’s pathway analysis of the 95 genes potentially important in development of NTG from all study types. 5 of top upstream regulators for all genes are shown for this same gene set, which suggest cholesterol regulation, estrogen signaling, inflammation, and cell cycle regulation play a role in NTG’s pathogenesis.
| Normal Tension Glaucoma CPDB Pathways (All Genes Included) | |||
|---|---|---|---|
|
| |||
| Pathway (Rank) | Pathway Source | Pathway-Input Gene Overlap | p-value |
| MAPK signaling pathway - Homo sapiens (human) | KEGG | NTF4, RAC3, SOS1, MECOM, CACNG4, CACNA1C, GADD45G, MEF2C, PRKCA, CD14, TP53, PLA2G4B, MAPKAPK3, CACNG5, JMJD7-PLA2G4B | 2.24e-10 |
| Tryptophan metabolism - Homo sapiens (human) | KEGG | CYP1A1, CYP1A2, CYP1B1, ALDH3A2, ECHS1, IL4I1, EHHADH | 1.03e-08 |
| Fc epsilon RI signaling pathway - Homo sapiens (human) | KEGG | RAC3, SOS1, PRKCA, VAV3, VAV2, FYN, JMJD7-PLA2G4B, PLA2G4B | 2.84e-08 |
| Pathways in cancer - Homo sapiens (human) | KEGG | CDKN2B, CDKN2A, TP53, RAC3, SOS1, MECOM, WNT10A, WNT6, WNT4, PRKCA, WNT8B, EDNRA, AGTR1, EDNRB, HHIP | 9.47e-08 |
| Estrogen metabolism | Wikipathways | CYP1A1, CYP1A2, CYP1B1, COMT, STS | 1.23e-07 |
|
| |||
| Normal Tension Glaucoma Top Upstream Regulators (IPA Core Analysis) | |||
|
| |||
| Regulator | Target Genes from Input List | p-value | |
|
| |||
| NPC1 | ABCA1, CAV1, CAV2, TLR4 | 4.39e-08 | |
| ESR1 | CAV1, CAV2, CDKN2A, COMT, CYP1A1, CYP1B1, EDN1, TP53, VAV3 | 1.89e-05 | |
| CBX8 | CDKN2A, CDKN2B | 1.92e-05 | |
| CDKN2B-AS1 | CDKN2A, CDKN2B | 1.92e-05 | |
| DIAPH1 | CDKN2A, CDKN2B | 1.92e-05 | |
The two most associated networks from IPA also reflected this molecular complexity. Network 1 was entitled “Lipid Metabolism, Small Molecule Biochemistry, Vitamin and Mineral Metabolism” (Supplementary Fig. 7). This network included 5 genes which had been previously cited as major POAG-loci: APOE, CAV1, CAV2, OPA1, and CYP1B1 (Janssen et al., 2013). Network 1 appears to reveal a central role of TP53, which is understandable since the upregulation of TP53 has been associated with increased RGC death (Nickells, 1999). Additionally, there is a high degree of overlap between several of the molecules within this network, and important contributors in the pathogenesis of Alzheimer’s disease.
The APOE and ABCA1 genes have both been associated with Alzheimer’s disease and POAG, and are shown to interact with one another in Network 1 (Ananth et al., 2014; Liu et al., 2013). Ingenuity highlighted several studies which indicate that ABCA1 is needed for the proper metabolism of ApoE in the brain and preventing plaque formation and neurodegeneration (Hirsch-Reinshagen et al., 2004; Wahrle et al., 2004). The role of APOE in the pathogenesis of Alzheimer’s disease is well established. The ε4 allele of APOE is the primary genetic risk factor for Alzheimer’s disease (Liu et al., 2013). The ε4 allele has also been associated with increased odds for developing POAG in certain ethnic groups, most notably in individuals of East-Asian descent, which is interesting given that Japanese patients harboring the ε4/ε4 genotype appear to be at the greatest risk for developing Alzheimer’s disease when compared to individuals of African, Hispanic, or European descent with the same genotype (Farrer et al., 1997; Liao et al., 2014; Wang et al., 2014). The role of the ε4 allele in the development of POAG is not established in POAG, however as we can see in the Network 1, that the molecular similarities between these two diseases go beyond APOE.
ABCA1 encodes for a cholesterol efflux transporter, which has been shown to play a role in Alzheimer’s disease and ocular diseases, including HTG and NTG (Ananth et al., 2014; Chen et al., 2014b; Liu et al., 2013). ABCA1 maintains cellular cholesterol homeostasis, by transferring cholesterol from cells (including neurons and glia) to the HDL molecule (Liu et al., 2013; Phillips, 2014). ABCA1 lipidates the APOE peptide, forming the APOE lipoprotein, which in turn plays a role in clearing amyloid-β from neuronal tissues (Liu et al., 2013). Variants near ABCA1 have been shown to be significantly associated with the incidence of HTG, NTG, and OHT (Chen et al., 2014b; Hysi et al., 2014). Additionally, ABCA1 is expressed throughout the eye, including in RGCs, but to our knowledge no functional studies have been performed to determine how ABCA1 expression is directly related to the health of the optic nerve (Ananth et al., 2014; Gharahkhani et al., 2014). However, it has been shown that reduced expression of ABCA1 leads to accumulation of retinal cholesterol, and is associated with a pro-inflammatory state, contributing to the development of dry AMD in rodent models (Ananth et al., 2014). This lends credence to the idea that disruption of ABCA1 functionality could contribute to optic nerve inflammation and subsequent RGC death.
CAV1 and CAV2 were also included in Network 1 of our NTG gene set analysis. CAV1 codes for Caveolin-1, which is an integral membrane protein that binds cholesterol and helps form lipid rafts. CAV2 encodes for Caveolin-2, which is a protein associated with CAV1 intracellularly (Williams and Lisanti, 2004). Variants within the CAV1/CAV2 region have been associated with NTG and HTG in various ethnic populations (Loomis et al., 2014; Thorleifsson et al., 2010). Functional studies have shown that silencing of CAV1 and CAV2 results in improved outflow through the TM (Aga et al., 2014). The caveolins appear to play an important role in glial cells, particularly astrocytes and microglia (Parat and Riggins, 2012). In astrocytes, CAV1 contributes to Aβ-induced cellular processes, including cholesterol efflux (Parat and Riggins, 2012). Additionally, Cav1 knockout mice have significantly higher rates of microgliosis and astrogliosis and more rapid onset of neurodegenerative processes (Head et al., 2010; Niesman et al., 2014).
Network 2 from IPA analysis of the NTG gene set was entitled “Free Radical Scavenging, Glomerular Injury, Organismal Injury and Abnormalities” (Fig. 9). This network included 6 major POAG-loci: WDR36, CDKN2B, CDKN2A, CDKN2B-AS1, TLR4, and OPTN (Janssen et al., 2013). One of the primary reasons we chose to highlight this pathway, was that it was one of the few to depict the interaction between the 3 major genes within the 9p21 chromosomal region, CDKN2A, CDKN2B, and CDKN2B-AS1. These genes have been consistently shown to be associated with the development of glaucoma in several GWAS and genotyping studies (Janssen et al., 2013; Ng et al., 2014). CDKN2B-AS1 encodes a long non-coding RNA (termed CDKN2B-AS1 or ANRIL) that is transcribed in the antisense direction of the CDKN2A-CDKN2B gene cluster, which encodes p16 (CDKN2A) and p15 (CDKN2B) proteins (Holdt et al., 2013; Jarinova et al., 2009; Visel et al., 2010). All the three genes have been found to be expressed within most of the ocular tissues involved in the pathogenesis of glaucoma, including the TM, ciliary body epithelial cells, retina, and optic nerve (Burdon et al., 2012; Wagner et al., 2013).
Fig. 9.
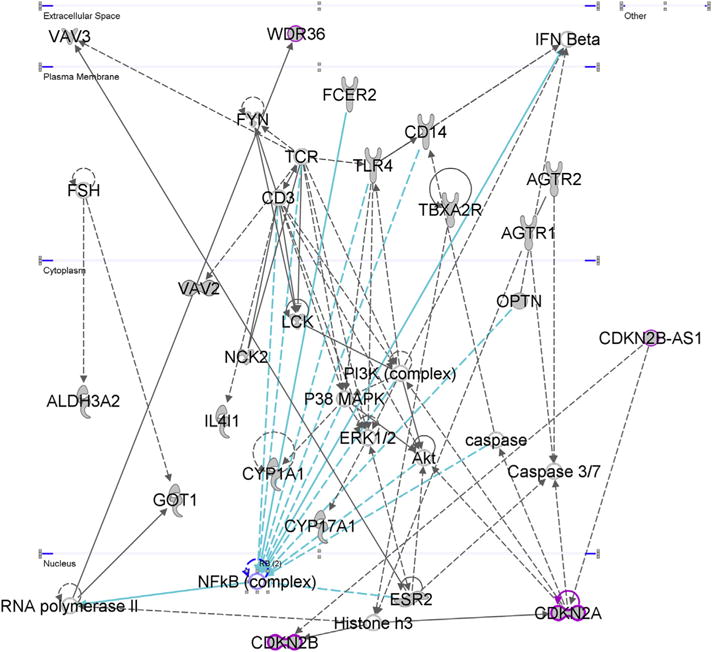
“Free Radical Scavenging, Glomerular Injury, Organismal Injury and Abnormalities.” This was the second most associated network according to IPA core analysis of the NTG gene input set which included all 95 genes from functional and genetic association studies investigating the NTG phenotype. We highlighted CDKN2A, CDKN2B, and CDKN2B-AS1 in magenta, all of which have been highlighted in POAG GWAS’. Other important POAG GWAS loci in this network include TLR4, WDR36, and OPTN.
Functional studies on CDKN2B-AS1 have focused on its potential role in cancer and atherosclerosis, which have revealed important regulatory functions of CDKN2B-AS1 on CDKN2B and CDKN2A. Yu et al found that CDKN2B-AS1 overexpression caused epigenetic silencing of CDKN2B in acute lymphoblastic leukemia and acute myeloid leukemia leukocytes (Yu et al., 2008). In coronary artery disease, Jarinova et al. discovered that patients with higher levels of CDKN2B-AS1 isoforms expressed significantly lower levels of the tumor suppressors CDKN2B and CDKN2A, confirming a reciprocal cis-acting regulatory relationship between these 3 genes (Jarinova et al., 2009). Chen et al. found that CDKN2B-AS1 appears to play an important role in TGFβ1 signaling, such that it likely inhibits TGFβ1-induced cell cycle inhibition by blocking SMAD signaling and contributing to the downregulation of CDKN2B (Chen et al., 2014a). This supports the idea that neuroinflammation is contributing to RGC or glial death and senescence during the pathogenesis of glaucoma.
NFκB also appears to have an important role in this pathway. TLR4 and OPTN are genes implicated in POAG by various genetic association studies, and shown as direct neighbors to the NFκB complex in Network 2 (Janssen et al., 2013). Over a decade ago, Rezaie et al. published their findings that several SNPs within OPTN were associated with NTG (Rezaie et al., 2002). OPTN plays a significant role in a negative feedback loop for TNF-α-induced NFκB activation. Therefore, a mutation hindering OPTN’s functionality could result in relatively uncontrolled TNF-α and NFκB-mediated gliosis and neuronal cell death (Ying and Yue, 2012). In 2013, Minegishi et al. discovered that transgenic mice harboring the E50K OPTN mutation developed NTG associated with marked retinal gliosis (Minegishi et al., 2013).
TLR4 is a toll-like receptor, which has been found to be potentially implicated in the development of POAG in several genetic-association studies across different ethnic populations (Janssen et al., 2013). TLR4 plays a role in activating the innate immune system in response to pathogen-associated molecular patterns (PAMPs), like lipopolysaccharide (LPS), or damage-associated molecular patterns (DAMPs) such as HMGB1 (a chromosomal protein released by cells during a stress response). Secreted HMGB1 is highly pro-inflammatory and has been associated with RGC death (Forte et al., 2014; Schallenberg and Thanos, 2009). Activation of TLR4 results in the activation of the NFκB complex, which promotes gliosis and the production of a pro-inflammatory cytokine milieu (Kigerl et al., 2007).
4.2.3. Ocular hypertension gene analyses
CPDB pathway and IPA analysis on the 30 genes associated with OHT or IOP in genetic association studies alone (Supplementary Table 6) showed that the most overrepresented pathways included “corticosteroids and cardioprotection,” and “visceral fat deposits and the metabolic syndrome” (Supplementary Table 12). Of note, some of the most prominent glaucoma GWAS hits were represented in the process “regulation of anatomical structure morphogenesis,” including MYOC, CYP1B1, WDR36, and GAS7. IPA analysis using the genes associated with OHT or IOP in genetic-association studies, showed the top associated network as “Skeletal and Muscular System Development and Function, Hereditary Disorder, Ophthalmic Disease” (Supplementary Fig. 8). The top POAG GWAS genes in this pathway included: CAV1, CAV2, MYOC, CYP1B1, and TNF (Janssen et al., 2013). An interaction we found particularly interesting was that between TNF (TNF-α), ADRB2 (β2 adrenoreceptor), and CYP1B1 (Cytochrome P450 Family 1 Subfamily B Member 1). CYP1B1 is important in xenobiotic and estradiol metabolism, and has recently been found to be a regulator of cell proliferation in cancer cells (Kwon et al., 2016). It has also been noted that in astrocytes, β2-adrenergic signaling inhibits TNF-α-mediated activation of NFκB, which reduces astrogliosis (Laureys et al., 2014). Since TM cells have physiologic similarities to smooth muscle cells, such as relaxing in response to β-agonists, it is possible that chronic exposure to TNF-α and TGF-β may reduce TM cell responses to endogenous catecholamines, contributing to OHT (Wiederholt et al., 2000). Overall, these results suggest that TGF-β and TNF-α signaling contribute to physiological disruptions in both the trabecular meshwork and optic nerve.
Expanding our inclusion criteria, we found thirty-nine genetic association and functional studies citing an association between 147 different genes and the OHT phenotype (Supplementary Table 6). When all 147 genes were included in CPDB, the most overrepresented pathways included “AP-1 transcription factor network,” “Spinal cord injury,” “TNF signaling pathway,” and “Rheumatoid arthritis” (Supplementary Table 12). IPA analysis of these genes revealed that the most significantly associated upstream regulators included the NFκB complex, COL18A1, and TNF (Supplementary Table 12). Additionally, the most highly associated disorders with this gene set were “neurological disease” and “skeletal and muscle disorders,” which seemed to partially coincide with CPDB’s results. The top associated network for this gene set was “Organismal Injury and Abnormalities, Reproductive System Disease, Cardiovascular System Development and Function” (Supplementary Fig. 9). This network included the major POAG GWAS hits CAV1 and CAV2 and involved a diverse set of genes involved in a myriad of different physiological functions. CAV1 is needed for the mechanosensitive activation of Akt in vascular smooth muscle cells in response to increased tensile stretch experienced during essential hypertension (Kaimoto et al., 2010) (Xue et al., 2007). In TM cells, elevated OHT also activates Akt, which in turn activates matrix metalloproteinases (MMPs). The Akt-MMP2 interaction is represented in the IPA network (Supplementary Fig. 8). MMPs play a central role in ECM turnover, which reduces resistance to aqueous outflow and can serve as a counter-mechanism to IOP elevation (Zhavoronkov et al., 2016). In light of above studies, it is likely that caveolins and the PI3K-Akt pathway plays a pivotal role in TM physiology and contribute to the pathogenesis of POAG and OHT.
Among the 147 OHT genes, 81 genes were discovered in a microarray study comparing gene expression between steroid-treated and untreated TM cells to study OHT and POAG (Fan et al., 2008). We repeated the analysis after removing the 81 genes (Supplementary Table 6). The top scoring network from IPA was “Hereditary Disorder, Ophthalmic Disease, Organismal Injury and Abnormalities” (Supplementary Fig. 10). The major POAG GWAS loci included in this network were: CYP1B1, MYOC, OPTN, CAV1, CAV2, and WDR36. The network tended to converge upon the NFκB complex, but we found several of the interactions between the “estrogen receptor” and other molecules in the network to be interesting, especially given that elevated estrogen levels in women have been associated with lower IOP (Wei et al., 2012). Upon investigating interactions between estrogen and TGF-β, we found that estrogen can block the TGF-β signaling pathway through ERβ’s inhibitory interaction with SMAD3, potentially limiting cellular senescence and apoptosis (Matsuda et al., 2001). The network also reveals that estrogen receptors have a regulatory function on CAV1 and CAV2. While studying this interaction, we found that in neuronal cells, overexpression of ERα (encoded by ESR1) silences CAV1 expression, and CAV1 expression is restored when ERα and ERβ are both over-expressed. This suggests that ERβ has an inhibitory effect on ERα-dependent CAV1 expression (Low and Nicholson, 2015). When we consider interactions between estrogen, TGF-β and CAV1 signaling along with the population-based studies, it appears that the estrogen pathway may play an important role in the regulation aqueous outflow and the development of OHT.
4.2.4. Juvenile open-angle glaucoma gene analyses
We found 62 studies reporting significant associations between 16 genes and the JOAG phenotype (Supplementary Table 6). CPDB analysis revealed that “estrogen metabolism,” and “metabolism of xenobiotics by cytochrome P450” as the most highly overrepresented pathways (Supplementary Table 13). The top associated network from IPA analysis of this gene set was “Cellular Movement, Cell Signaling, Small Molecule Biochemistry” (Supplementary Fig. 11). The major POAG GWAS genes within this network included TNF, MYOC, WDR36, APOE, CYP1B1 and NTF4. Like other networks we have presented, there was significant convergence onto TNF-α. However, we found a set of interactions with MYOC to be of particular interest and wanted to study them further since MYOC had the greatest number of studies (36) associating it with JOAG. In this network, MYOC is shown to interact with PXDN, which was included in our input gene list, and NEFM (neurofilament, medium polypeptide) and CD44, which were not. All the three genes have been shown to be upregulated in response to MYOC overexpression in HEK293 cells (Joe et al., 2014). NEFM is thought to be important in neural processes and the radial outgrowth of axons during development (Yuan et al., 2012). While there have not been any SNPs within NEFM associated with POAG phenotypes, a mutation in NEFM has been associated with early onset Parkinson’s disease (Lavedan et al., 2002). CD44 encodes a group of cell surface proteins that have a variety of functions, including the regulation of cell motility, differentiation, and survival. These cellular processes play roles in axonal growth and guidance, organ development, and regulation of the immune response (Ponta et al., 2003). While CD44 was not associated with JOAG specifically, it has been shown to be upregulated in a murine models of glaucoma and OHT (Howell et al., 2011; Giovingo et al., 2013).
Peroxidasin is an enzyme secreted into the ECM that may play an important role in the organization of the fibronectin matrix (Peterfi et al., 2009). Myofibroblasts, which share features with TM cells, secrete peroxidasin when activated by TGF-β (Peterfi et al., 2009). Additionally, children from a Cambodian family who developed severe OHT and glaucoma harbored a truncating mutation within exon 10 of the PXDN gene (Khan et al., 2011b; Sia et al., 2010). The authors hypothesized that peroxidasin plays an important protective, antioxidant role within the ECM of anterior chamber tissues which may help prevent OHT (Khan et al., 2011b). Additionally, mice with Pxdn mutations developed severe anterior segment dysgenesis (Yan et al., 2014). The above findings suggest that patients suffering from JOAG secondary to mutations in PXDN or its regulator MYOC may suffer from ECM disorganization compounded by relatively uncontrolled oxidative damage within the anterior chamber of their eyes.
4.2.5. Primary congenital glaucoma gene analyses
Seventy-six studies cited significant associations between 7 different genes/loci and the PCG phenotype (Supplementary Table 6). These genes included CYP1B1, VAV2, VAV3, LTBP2, FOXC1, and loci GLC3C and GLC3B. VAV2 and VAV3 encode guanine nucleotide exchange factors, that control signaling cascades involved in cell morphogenesis, migration, and adhesion (Fujikawa et al., 2010). Vav2/Vav3 deficient mice develop a phenotype similar to PCG, and intronic SNPs within VAV2 and VAV3 were found to be significantly associated with POAG in a Japanese population (Fujikawa et al., 2010). When VAV2 and VAV3 were included in the CPDB analysis, they biased the results such that the two most significantly overrepresented pathways included “VEGFR2 mediated vascular permeability” and “EPH-ephrin mediated repulsion of cells,” which solely included the VAV2 and VAV3 genes. Supplementary Table 14 summarizes the results of these analyses and reveals the noted bias. Since only one study cited the VAV2/3-PCG association, we repeated the analysis without them. Interestingly, there were no pathways found to be significantly overrepresented when VAV2 and VAV3 were removed from the input list for CPDB, however GO analysis revealed that “collagen fibril organization,” and “regulation of anatomical structure morphogenesis,” were significantly enriched biological processes. CYP1B1 and FOXC1 were the genes involved in collagen fibril association (Supplementary Table 14). These results are consistent with the fact that PCG arises from trabeculodysgenesis, a developmental malformation of the TM, and that ECM organization and regulation has been clearly shown to be an important process in the maintenance of healthy TM cells.
IPA core analysis of the gene sets with and without VAV2 and VAV3 did not differ nearly as much as CPDB’s analysis of the same sets. For both gene sets, the top most associated upstream regulator was identified as HDAC6, encoding histone deacetylase 6, an enzyme which regulates gene expression by modifying chromatin structure (Supplementary Table 14; Li et al., 2013). HDAC6 appears to have important roles in inflammation and neurodegeneration, and there is evidence that its proper functioning is important in the clearing of misfolded protein aggregates through autophagy (Simoes-Pires et al., 2013).
The top associated diseases and biological functions for both of these gene sets were “familial glaucoma,” “juvenile-onset primary open angle glaucoma,” and “primary congenital glaucoma.” When VAV2/3 were included in the analysis, the top associated network was “Cellular Movement, Cancer, Organismal Injury and Abnormalities” (Supplementary Fig. 12). In this network, both VAV2 and VAV3 are shown to interact with TCR (T-cell receptor) and RHOG (Ras homolog family member G). Both VAV2 and VAV3 activated RHOG, which has been shown to be an important regulator of neurite outgrowth, a process that has is hindered in some POAG patients (Katoh et al., 2000; Zimering et al., 2013). VAV3 has been shown to stimulate IL-2 expression, making these proteins potentially important inflammatory mediators (Zakaria et al., 2004).
When VAV2 and VAV3 were removed from the gene input list, the most associated network was “Cancer, Organismal Injury and Abnormalities, Cellular Function and Maintenance,” shown in Supplementary Fig. 13. Not surprisingly, MYOC and CYP1B1 appeared to be central players in this network, however, these genes did not interact with each other and did not share any direct interaction neighbors. The interaction between CYP1B1, an important POAG/PCG locus, and BRCA1, one of the most prominent cancer-related genes, is of interest. IPA based this interaction on a study by Kang et al., which revealed that BRCA1 is recruited to the promoter of CYP1B1 following human breast cancer cell exposure to xenobiotics such as tetrachlorodibenzodioxin (TCDD) (Kang et al., 2006). However, there is evidence that the BRCA1-CYP1B1 interaction is likely important for the metabolism of endogenous hormones, such as estrogen (Vasiliou and Gonzalez, 2008). CYP1B1 metabolizes sex hormones, and it is thought that the levels of these hormones and their metabolites may have an important role in protecting the anterior segment tissues from oxidative stress (Ganatra et al., 2013; Jansson et al., 2001). Estrogen has also been shown to be important for normal retinal development in zebra-fish, and may have a protective effect on the development of OHT and POAG (Hamad et al., 2007).
This network also revealed an interaction between MYOC and PGR, which encodes the progesterone receptor. MYOC overexpression induces upregulated expression of PGR (Joe et al., 2014). Like estrogen, progesterone has also been shown to play an important role in neural development, with high levels of PGR expression in the neurons and radial glial cells of developing zebrafish (Diotel et al., 2011). The progesterone hormone may actually be produced by certain glial cells and have an important neuroprotective role, especially in age-related neurodegenerative disorders like Alzheimer’s disease (Diotel et al., 2011; Schumacher et al., 2012; Singh and Su, 2013). Additionally, animal models have demonstrated that progesterone can help limit the progression of TBI and ischemic cerebral injuries by dampening neuroinflammation and oxidative stress (Singh and Su, 2013; Webster et al., 2015). However, it is not entirely clear how progesterone affects the pathogenesis of glaucoma. It has been reported that estrogen and combination progesterone-estrogen replacement therapy in post-menopausal women significantly reduces the risk of developing POAG, with women taking combination oral contraceptives realizing the greatest benefit (Newman-Casey et al., 2014). However, others have reported that women who reported taking oral contraceptives for three years or more had an elevated risk for POAG and OHT (Wang et al., 2016). Animal studies have shown that topical and endogenous progesterones cause mild elevations in IOP (Knepper et al., 1985; Ofri et al., 1999). Overall, it is clear that progesterone’s impact on POAG pathogenesis is incredibly complex and needs further research to fully understand its role.
5. Interpretations: the sinister roles of inflammation and senescence in POAG
After surveying our findings, it is apparent that inflammation and its dysregulation seems to be playing an extremely important role in the development of OHT and glaucoma. Specifically, it appears that TNF-α, TGF-β, and NFκB signaling are consistently identified in the networks and biological processes elucidated by the pathway analysis and functional annotation performed on our various gene sets. Neuroinflammation and its role in neurodegenerative processes has been studied and discussed extensively during the last two decades (Neufeld and Liu, 2003). Inflammatory processes have been implicated in the development of POAG for many years, but the role of inflammation in this disease is remarkably complex and multifaceted, making successful intervention frustratingly elusive (Janssen et al., 2013; Kwon et al., 2009; Mowatt and McIntosh, 2013; Neufeld and Liu, 2003).
Currently the only recognized modifiable risk factor for POAG is IOP, and lowering IOP has been shown to be protective even in NTG (Neufeld and Liu, 2003). This implies that there are different degrees of RGC susceptibility amongst individuals, and that elevated IOP is an important inciting factor in the initiation of the pathogenesis of glaucoma. However, it is becoming increasingly apparent that elevated IOP is just one of many possible POAG-instigating insults, and we believe that our study has helped shed more light on this proposition. We have shown that the pathophysiology of POAG shares many similarities with other neurological diseases which have clear inciting factors, like stroke and TBI; but also closely parallels the development of more insidious but equally devastating conditions, such as Alzheimer’s disease and multiple sclerosis.
From our review of the literature, it is apparent that POAG represents a perpetual dysregulation of the retinal and optic nerve microenvironments, triggered by an activating stimulus that may or may not be perpetually present. Outside of elevated IOP, these insults likely include hypoxia and oxidative damage, metabolic derangements, alterations in hormonal homeostasis, and perhaps most importantly, inflammation. We highlight the significance of inflammation in the pathogenesis of POAG primarily because it appears to be an important pathological player within the anterior and posterior compartments of the globe, regardless of what the initial insult was, especially in adult-onset disease. Additionally, it is more apparent that cellular senescence may be a chief offender in the maintenance of a pro-inflammatory milieu within a wide variety of aging tissues, including the CNS (Chinta et al., 2015).
Cellular senescence has been linked to many age-related diseases throughout the last two decades, such as atherosclerosis, but it has only been studied recently in the context of chronic neurodegenerative diseases (Chinta et al., 2015). The characterization of the senescence-associated secretory phenotype (SASP) in the last ten years has revealed that aging tissues are chronically inflamed, which limits their ability to heal or regenerate (Young and Narita, 2009). In senescent cells, the SASP is characterized by the secretion of pro-inflammatory cytokines, including IL-1, IL-6, IL-8, and TNF-α (Chen et al., 2016; Chinta et al., 2015; Hubackova et al., 2012). Cells expressing the SASP also undergo activation of the AP-1/c-JUN, JAK/STAT, TGFβ/SMAD, and IL1/NFκB signaling pathways, which tend to further promote senescence and inflammation (Chen et al., 2016; Freund et al., 2010; Hubackova et al., 2012). Therefore, it appears that there is a destructive positive-feedback loop characterized by senescence-induced inflammation and inflammation-induced senescence (Chen et al., 2016). This has several unfortunate consequences for neural tissue, such as the optic nerve. First, the pro-inflammatory state can attract destructive leukocytes, result in demyelination, and damage supporting vasculature (Chinta et al., 2015). Second, it stifles the population of neurosupportive glia, such as astrocytes and retinal Müller glia, by impairing their function and halting their entry into the cell cycle (Bhat et al., 2012; Lindholm et al., 1992). And third, senescence causes cells to become more prone to TNF-α-induced apoptosis due to up regulation of pro-apoptotic proteins, such as FAS receptor, inevitably leading to degeneration of already inflamed (i.e. TNF-α-saturated) tissues (Jeon and Boo, 2013).
It is also important to note that glutamate-induced neurotoxicity may be playing an important role in RGC death in glaucoma, and may be closely tied to retinal and optic nerve inflammation and senescence. High glutamate levels trigger excess influx of calcium and can trigger apoptosis in the neurons and glia (Mowatt and McIntosh, 2013). During a neuroinflammatory state, degenerating RGCs release excess glutamate, and dysfunctioning Müller cells and astrocytes have an impaired ability to remove glutamate from the extracellular space, leading to retinal and optic nerve degeneration (Mowatt and McIntosh, 2013).
The senescent-inflammatory phenotype might also play an important role in the development of JOAG and PCG. For JOAG, our pathway analyses revealed that dysregulation of molecules such as myocilin (MYOC) or peroxidasin (PXDN) can lead to fibronectin disorganization and oxidative stress, both of which can lead to inflammatory cytokine release, senescence, and apoptosis (Austin et al., 2009; Joe and Tomarev, 2010). For PCG, our pathway analyses revealed the importance of CYP1B1, and its relation to estrogen and progesterone regulation. Interestingly, estrogen appears to inhibit senescence in endothelial progenitor cells, while progesterone can promote senescence in ovarian cancer cells–suggesting the balance of these sex hormones may have important roles in tissue inflammation and aging (Diep et al., 2013; Imanishi et al., 2005).
Therefore, the senescent-inflammatory phenotype of POAG incorporates many of the pathological mechanisms highlighted by our pathway analyses for each of the investigated phenotypes. What is particularly difficult about this finding is that stress-induced senescence is a self-perpetuating process and can be initiated by practically anything that triggers the development of a pro-inflammatory cytokine milieu, a non-specific response to a host of different stressors. These stressors include almost all of the insults we have discussed in this review, including excess or irregular ECM protein expression, cholesterol dysregulation, hormonal and metabolic disturbances, oxidative stress, and misfolded proteins. Table 7 displays some examples of insults with the POAG-associated genes related to them.
Table 7.
Relationships between the various insults leading to the inflammatory-senescent phenotype and genes implicated in POAG which may contribute to the development, amplification, or modification of these insults. Each of these genes can be found in either Supplementary Table 1 or 2, where the source manuscript for the glaucoma-gene association was confirmed.
| Insults Triggering Inflammation & Senescence | Relationship of Insult to Inflammation & Senescence | Select POAG-Associated Genes Contributing to Insult | References |
|---|---|---|---|
| Irregular ECM Organization | - Degraded ECM proteins serve as “alarmins” or “DAMPs” which bind TLRs. - MMPs from inflammatory cells amplify ECM protein degradation & DAMP production. - ECM proteins are released as SASP factors in senescent cells. |
TGFB1, TGFB2, MMP1, MMP2, MMP9, FN1, COL11A1, COL18A1, CD44, BMP1, BMP4 | (Coppe et al., 2010; Gaudet and Popovich, 2014) |
| Cholesterol Dysregulation | - LDLR knockout mice have elevated microgliosis and astrogliosis and excess expression of proinflammatory cytokines. - Oxidized LDL promotes vascular senescence. |
ABCA1, APOE, CAV1, CAV2, GHRL, GHSR, ESR1, ESR2 | (Fyhrquist et al., 2013; Thirumangalakudi et al., 2008) |
| Hormonal Disturbances | - Treating astrocytes with high dose E2 reduces LPS-induced activation of NFκB. - Microglia treated with E2 express less TNF-α when exposed to LPS. - Estrogen replacement can reverse neuronal senescence as marked by dendritic spine restoration in oophorectormized rats. |
ESR1, ESR2, CYP1B1, COMT, AKR1C4, AKR1D1, CYP17A1, CYP1A1, CYP1A2, CYP3A4, HSD17B1, STS | (Dimayuga et al., 2005; Dodel et al., 1999; Hof and Morrison, 2004) |
| Oxidative Damage | - Oxidative stress activates inflammatory regulators like NFκB and AP-1. - Activated microglia contribute to oxidative damage in neuroinflammatory processes. - Oxidative stress triggers premature cellular senescence, hydrogen peroxide is used to induce senescence for in vitro models. |
GSTM1, GSTT1, GSTP1, MTHFR, NOS2, NOS3 | (Chen et al., 2007; Reuter et al., 2010) |
| Neurotrophin Imbalance | - Neurotrophins attenuate inflammation in the CNS and various other tissues. - Neurotrophins inhibit the development of senescence in cortical neurons. |
BDNF, NGF, NTF4, NGFR, NTRK1, NTRK2 | (Sato et al., 2001; Wei and Jonakait, 1999) |
| Misfolded Proteins | - Misfolded protein accumulation can trigger the inflammasome and production of pro-inflammatory cytokines. - The unfolded protein response (UPR) may be a driver or consequence of cellular senescence. |
MYOC, OPTN, SNCG | (Martinon and Aksentijevich, 2015; Pluquet et al., 2015) |
5.1. TGF-β signaling as a model of the senescent-inflammatory phenotype in POAG
Our pathway analyses repeatedly highlighted the senescence-related AP-1/c-JUN, TGFβ/SMAD, and IL1/NFκB signaling cascades as important participants within our input gene sets, suggesting cellular senescence may be an important contributor to the development of POAG phenotypes. We find the TGFβ/SMAD pathway to be of particular interest, since functional studies have shown that TGF-β signaling is an important factor in the development of pathological changes associated with POAG in both the anterior and posterior segments of the eye (Fatma et al., 2009; Fuchshofer and Tamm, 2012). Additionally, TGF-β has been shown to trigger ROS accumulation and to activate the NFκB pathway by promoting significant expression of proinflammatory cytokines, such as IL-8 (Hubackova et al., 2012; Salminen et al., 2012).
TGF-β2 has received particular attention in ocular research, since it found to be elevated in the aqueous humor of patients with POAG, but not in other forms of glaucoma (Fuchshofer and Tamm, 2012). TM cells treated with TGF-β1 and TGF-β2 exhibit markedly increased ROS production and senescence (Fatma et al., 2009; Li et al., 1996). The TGF-β signaling pathway is also intriguing when considering POAG pathology because several genes encoding some of its important downstream effectors are some of the most important POAG GWAS hits, specifically CDKN2A, CDKN2B, and CDKN2B-AS1 (Burdon et al., 2011; Janssen et al., 2013). We previously mentioned that CDKN2B-AS1 appears to play a regulatory role in TGFβ/SMAD signaling by inhibiting SMAD-induced expression of CDKN2B (Fig. 10) (Chen et al., 2014a). Elevated levels of CDKN2B trigger senescence, and have been shown to do so in a variety of cell types, including human glioma cells (Fuxe et al., 2000). Interestingly, SNPs at rs4977756 within CDKN2B-AS1 have been associated with increased risk of glaucoma (excess RGC death) when patients are homozygous for the A allele, and with increased of high-grade gliomas (impaired glial death) when patients are homozygous for the G allele (Burdon et al., 2011). This would support the hypothesis that this region plays a role in regulation of cell senescence and apoptosis, possibly by affecting the expression levels or functionality of CDKN2B-AS1 transcripts (Fig. 10).
Fig. 10.
TGF-β and the senescent-inflammatory phenotype in action during POAG. In panel A of the figure we see important cells of the posterior (astrocytes and microglia) and anterior segments (TM cells) of the globe. In healthy eyes, these cells are in a quiescent state. However, an initial insult, such as elevated IOP, triggers inflammatory (or gliotic) responses from these cells causing the release of inflammatory cytokines, including TGF-β. In panel B we see TGF-β interaction with its receptor on the surface of a non-descript cell, which could represent other glia, TM cells, or even RGCs. This triggers SMAD activation which eventually results in the transcription of other inflammatory cytokines as well as senescence-inducing molecules such as CDKN2B. Senescence also contributes to the inflammatory milieu through the SASP. CDKN2B-AS1 may modulate inflammation and senescence by inhibitory interactions with the SMAD complex or CDKN2B directly–however the exact mechanism of action of CDKN2B-AS1 is still unclear. SNPs in the 9p21 region which are associated with POAG may impair the function or expression of CDKN2B-AS1 thus contributing to development and progression of the senescent-inflammatory phenotype.
6. Future directions and conclusions: targeted therapy for POAG and combating the senescent-inflammatory phenotype
POAG is an enormously complex disease, with various tissue types, molecular pathways, and genes playing important, interrelated roles in its pathogenesis. And while lowering IOP should undoubtedly remain an important therapeutic and preventative modality, future treatment regimens should reflect the multifaceted nature of this disease. The field is making promising strides in the development of molecular and targeted therapies. Providing neurotropic sustenance to RGCs has great therapeutic potential in glaucoma. For instance, BDNF has been shown to prevent RGC death in certain models of glaucoma (Mey and Thanos, 1993). However, studies to date have shown that multiple doses of BDNF need to be delivered into the vitreous in order to achieve a therapeutic benefit (Domenici et al., 2014; Galindo-Romero et al., 2013). Interestingly, mesenchymal stem cells (MSCs) have been shown to secrete a variety of neurotrophic factors, including BDNF and NGF, and to prevent RGC death in vitro (Mead and Scheven, 2015). MSCs injected intravitreally can survive up to 21 days, and have been shown to prevent RGC death and promote axonogenesis in rodents with experimental glaucoma or optic nerve crush (Mead et al., 2013; Mead and Scheven, 2015).
Modulation of the senescent-inflammatory phenotype, and its plethora of mediators, is another promising therapeutic strategy for POAG. We had previously mentioned that etanercept, an anti-TNF-α agent prevented RGC death in rats with OHT, seems quite promising (Roh et al., 2012). A phase II clinical trial of etanercept in patients with Alzheimer’s disease demonstrated modest improvement in neuropsychiatric measures, but no significant improvement in measures of cognition (Butchart et al., 2015). Clinical trials in humans investigating the efficacy of intravitreal etanercept in glaucoma have yet to be performed.
NFκB revealed itself to be an important central regulator in many of the processes contributing to POAG’s pathogenesis. Inhibitors of NFκB have yet to be investigated within the specific context of glaucoma, but have been shown to have promising anti-inflammatory effects, including the attenuation of neuroinflammatory processes in animal models (Lei et al., 2009; Rummel et al., 2011). Specifically, mice given parthenolide, a plant extract which inhibits IκB kinase (an upstream activator of NFκB) experience less systemic and glial inflammatory cytokine production (Mathema et al., 2012; Rummel et al., 2011). NFκB/IκB inhibitors have also been shown to inhibit the development and progression of inflammatory diseases including atherosclerosis and psoriasis in animal models, making these potentially effective therapies for a wide range of chronic inflammatory conditions (Cuaz-Perolin et al., 2008; Wang et al., 2009). However, the effect of these NFκB inhibitors on nervous tissue remains unclear and will require further research to elucidate any adverse side effects (Lei et al., 2009; Zanotto-Filho et al., 2011).
Targeting the TGF-β signaling pathway is another promising therapeutic target for POAG. As discussed previously, TGF-β appears to be playing an important role in the development of OHT by inducing ECM protein deposition and the senescent-inflammatory phenotype within the TM. Additionally, it is plausible that overactive TGF-β signaling in the retina and ONH is contributing to the development of the senescent-inflammatory phenotype within these tissues, contributing to RGC death in a variety of different ways. Therefore, the inhibition of TGF-β signaling may attenuate the development and maintenance of the pathological processes occurring in each of the tissues and cell types implicated in POAG. The anti-TGF-β monoclonal antibody, fresolimumab (Genzyme, Cambridge, MA, USA), targets three TGF-β isoforms, and has been studied in various diseases (Buijs et al., 2012; Rice et al., 2015; Trachtman et al., 2011). Rice et al. report that patients with systemic sclerosis who received fresolimumab exhibited significant reduction of expression of THBS1 (thrombospondin-1), SERPINE1 (plasminogen activator inhibitor-1), and CTGF (connective tissue growth factor) biomarkers when compared to the pre-treatment levels (Rice et al., 2015). These patients also exhibited significant clinical improvement as determined by a clinical measure of skin fibrosis (Rice et al., 2015). Interestingly, TM and LC cells from patients with POAG express significantly higher levels of THBS1 (Supplementary Table 2) (Fatma et al., 2009; Kirwan et al., 2009). Additionally, overexpression of CTGF in rats caused an excess deposition of fibronectin and collagen IV in the TM, causing an increase in resistance to aqueous outflow (Supplementary Table 2) (Su et al., 2013). Therefore, it is conceivable that fresolimumab could attenuate the development of OHT, and quite possibly inhibit the development of the senescent-inflammatory phenotype throughout the eye.
In conclusion, POAG is an incredibly complex neurological disease as evidenced by the many genes implicated in its pathogenesis and in its clinical presentation. It is clear from our study that inflammation and senescence play pivotal roles in the development and progression of this devastating disease. Apart from the administration of IOP lowering drugs, there seems to be ample opportunity to develop novel and effective therapeutic interventions to tackle the neuroinflammation, tissue remodeling, and cell death that ultimately result in blindness. Meta-analysis of several published and ongoing large GWAS studies in different ethnic populations, especially in African Americans (POAAGG study) (Charlson et al., 2015), will help us understand the genetic and epistasis mechanisms that are responsible for causing this debilitating disease.
Supplementary Material
Acknowledgments
The authors would like to thank Maureen G. Maguire, PhD and Anita S. Bowman, MS for their helpful input regarding the analysis methods used in this study. We would also like to thank Mary A. Leonard of the Biomedical Art and Design office at the University of Pennsylvania for her wonderful illustrations.
Financial support
This work was supported by the National Eye Institute, Bethesda, Maryland (grant #1RO1EY023557-01) and the Department of Ophthalmology at the Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA. Funds also come from the F. M. Kirby Foundation, Research to Prevent Blindness, The Paul and Evanina Bell Mackall Foundation Trust, and the National Eye Institute, National Institutes of Health, Department of Health and Human Services, under eyeGENETM and contract Nos. HHSN260220700001C and HHSN263201200001C. The sponsor or funding organization had no role in the design or conduct of this research.
Abbreviations
- CCT
Central Corneal Thickness
- CDR
Cup-to-Disc Ratio
- CNS
Central Nervous System
- ECM
Extracellular Matrix
- GO
Gene-Ontology
- HTG
High Tension Glaucoma
- IOP
Intraocular Pressure
- JOAG
Juvenile Open-Angle Glaucoma
- LC
Lamina Cribrosa
- NTG
Normal Tension Glaucoma
- OAG
Open-Angle Glaucoma
- OHT
Ocular Hypertension
- ONH
Optic Nerve Head
- PCG
Primary Congenital Glaucoma
- POAG
Primary Open-Angle Glaucoma
- RGC
Retinal Ganglion Cell
- ROS
Reactive Oxygen Species
- TBI
Traumatic Brain Injury
- TM
Trabecular Meshwork
- GWAS
Genome Wide Association Study
Footnotes
Appendix A. Supplementary data
Supplementary data related to this article can be found at http://dx.doi.org/10.1016/j.preteyeres.2017.02.001.
References
- Abu-Amero K, Kondkar AA, Chalam KV. An updated review on the genetics of primary open angle glaucoma. Int J Mol Sci. 2015;16:28886–28911. doi: 10.3390/ijms161226135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Acott TS, Kelley MJ. Extracellular matrix in the trabecular meshwork. Exp Eye Res. 2008;86:543–561. doi: 10.1016/j.exer.2008.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aga M, Bradley JM, Wanchu R, Yang Y-f, Acott TS, Keller KE. Differential effects of caveolin-1 and -2 knockdown on aqueous outflow and altered extracellular matrix turnover in caveolin-silenced trabecular meshwork cells. Invest Ophthalmol Vis Sci. 2014;55:5497–5509. doi: 10.1167/iovs.14-14519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- AGIS Investigators. The advanced glaucoma intervention study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration The AGIS investigators. Am J Ophthalmol. 2000;130:429–440. doi: 10.1016/s0002-9394(00)00538-9. [DOI] [PubMed] [Google Scholar]
- Alm A, Nilsson SF. Uveoscleral outflow–a review. Exp Eye Res. 2009;88:760–768. doi: 10.1016/j.exer.2008.12.012. [DOI] [PubMed] [Google Scholar]
- Ananth S, Gnana-Prakasam JP, Bhutia YD, Veeranan-Karmegam R, Martin PM, Smith SB, Ganapathy V. Regulation of the cholesterol efflux transporters ABCA1 and ABCG1 in retina in hemochromatosis and by the endogenous siderophore 2,5-dihydroxybenzoic acid. Biochim Biophys Acta. 2014;1842:603–612. doi: 10.1016/j.bbadis.2014.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Austin BA, Liu B, Li Z, Nussenblatt RB. Biologically active fibronectin fragments stimulate release of MCP-1 and catabolic cytokines from murine retinal pigment epithelium. Invest Ophthalmol Vis Sci. 2009;50:2896–2902. doi: 10.1167/iovs.08-2495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauer-Mehren A, Bundschus M, Rautschka M, Mayer MA, Sanz F, Furlong LI. Gene-disease network analysis reveals functional modules in mendelian, complex and environmental diseases. PLoS One. 2011;6:e20284. doi: 10.1371/journal.pone.0020284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauer-Mehren A, Rautschka M, Sanz F, Furlong LI. DisGeNET: a Cytoscape plugin to visualize, integrate, search and analyze gene-disease networks. Bioinformatics. 2010;26:2924–2926. doi: 10.1093/bioinformatics/btq538. [DOI] [PubMed] [Google Scholar]
- Beck T, Hastings RK, Gollapudi S, Free RC, Brookes AJ. GWAS Central: a comprehensive resource for the comparison and interrogation of genome-wide association studies. Eur J Hum Genet. 2014;22:949–952. doi: 10.1038/ejhg.2013.274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beidoe G, Mousa SA. Current primary open-angle glaucoma treatments and future directions. Clin Ophthalmol. 2012;6:1699–1707. doi: 10.2147/OPTH.S32933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhat R, Crowe EP, Bitto A, Moh M, Katsetos CD, Garcia FU, Johnson FB, Trojanowski JQ, Sell C, Torres C. Astrocyte senescence as a component of Alzheimer’s disease. PLoS One. 2012;7:e45069. doi: 10.1371/journal.pone.0045069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blesa J, Przedborski S. Parkinson’s disease: animal models and dopaminergic cell vulnerability. Front Neuroanat. 2014;8:155. doi: 10.3389/fnana.2014.00155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brezin AP, Nedelec B, Barjol A, Rothschild PR, Delpech M, Valleix S. A new VCAN/versican splice acceptor site mutation in a French Wagner family associated with vascular and inflammatory ocular features. Mol Vis. 2011;17:1669–1678. [PMC free article] [PubMed] [Google Scholar]
- Buijs JT, Stayrook KR, Guise TA. The role of TGF-beta in bone metastasis: novel therapeutic perspectives. Bonekey Rep. 2012;1:96. doi: 10.1038/bonekey.2012.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burdon KP, Crawford A, Casson RJ, Hewitt AW, Landers J, Danoy P, Mackey DA, Mitchell P, Healey PR, Craig JE. Glaucoma risk alleles at CDKN2B-AS1 are associated with lower intraocular pressure, normal-tension glaucoma, and advanced glaucoma. Ophthalmology. 2012;119:1539–1545. doi: 10.1016/j.ophtha.2012.02.004. [DOI] [PubMed] [Google Scholar]
- Burdon KP, Macgregor S, Hewitt AW, Sharma S, Chidlow G, Mills RA, Danoy P, Casson R, Viswanathan AC, Liu JZ, Landers J, Henders AK, Wood J, Souzeau E, Crawford A, Leo P, Wang JJ, Rochtchina E, Nyholt DR, Martin NG, Montgomery GW, Mitchell P, Brown MA, Mackey DA, Craig JE. Genome-wide association study identifies susceptibility loci for open angle glaucoma at TMCO1 and CDKN2B-AS1. Nat Genet. 2011;43:574–578. doi: 10.1038/ng.824. [DOI] [PubMed] [Google Scholar]
- Butchart J, Brook L, Hopkins V, Teeling J, Puntener U, Culliford D, Sharples R, Sharif S, McFarlane B, Raybould R, Thomas R, Passmore P, Perry VH, Holmes C. Etanercept in Alzheimer disease: a randomized, placebo-controlled, double-blind, phase 2 trial. Neurology. 2015;84:2161–2168. doi: 10.1212/WNL.0000000000001617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butt AM, Pugh M, Hubbard P, James G. Functions of optic nerve glia: axoglial signalling in physiology and pathology. Eye Lond.d) 2004;18:1110–1121. doi: 10.1038/sj.eye.6701595. [DOI] [PubMed] [Google Scholar]
- Cai Z, Simons DL, Fu XY, Feng GS, Wu SM, Zhang X. Loss of Shp2-mediated mitogen-activated protein kinase signaling in Muller glial cells results in retinal degeneration. Mol Cell Biol. 2011;31:2973–2983. doi: 10.1128/MCB.05054-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao C, Moult J. GWAS and drug targets. BMC Genomics. 2014;15(Suppl. 4):S5. doi: 10.1186/1471-2164-15-S4-S5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao Y, Li H, Deb S, Liu JP. TERT regulates cell survival independent of telomerase enzymatic activity. Oncogene. 2002;21:3130–3138. doi: 10.1038/sj.onc.1205419. [DOI] [PubMed] [Google Scholar]
- Casson RJ, Chidlow G, Wood JP, Crowston JG, Goldberg I. Definition of glaucoma: clinical and experimental concepts. Clin Exp Ophthalmol. 2012;40:341–349. doi: 10.1111/j.1442-9071.2012.02773.x. [DOI] [PubMed] [Google Scholar]
- Charlson ES, Sankar PS, Miller-Ellis E, Regina M, Fertig R, Salinas J, Pistilli M, Salowe RJ, Rhodes AL, Merritt WT, 3rd, Chua M, Trachtman BT, Gudiseva HV, Collins DW, Chavali VR, Nichols C, Henderer J, Ying GS, Varma R, Jorgenson E, O’Brien JM. The primary open-angle african american glaucoma genetics study: baseline demographics. Ophthalmology. 2015;122:711–720. doi: 10.1016/j.ophtha.2014.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen D, Zhang Z, Mao C, Zhou Y, Yu L, Yin Y, Wu S, Mou X, Zhu Y. ANRIL inhibits p15(INK4b) through the TGFbeta1 signaling pathway in human esophageal squamous cell carcinoma. Cell Immunol. 2014a;289:91–96. doi: 10.1016/j.cellimm.2014.03.015. [DOI] [PubMed] [Google Scholar]
- Chen JH, Ozanne SE, Hales CN. Methods of cellular senescence induction using oxidative stress. Methods Mol Biol. 2007;371:179–189. doi: 10.1007/978-1-59745-361-5_14. [DOI] [PubMed] [Google Scholar]
- Chen L, Peng Z, Meng Q, Mongan M, Wang J, Sartor M, Chen J, Niu L, Medvedovic M, Kao W, Xia Y. Loss of IkappaB kinase beta promotes myofibroblast transformation and senescence through activation of the ROS-TGFbeta autocrine loop. Protein Cell. 2016;7:338–350. doi: 10.1007/s13238-015-0241-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y, Lin Y, Vithana EN, Jia L, Zuo X, Wong TY, Chen LJ, Zhu X, Tam POS, Gong B, Qian S, Li Z, Liu X, Mani B, Luo Q, Guzman C, Leung CKS, Li X, Cao W, Yang Q, Tham CCY, Cheng Y, Zhang X, Wang N, Aung T, Khor CC, Pang CP, Sun X, Yang Z. Common variants near ABCA1 and in PMM2 are associated with primary open-angle glaucoma. Nat Genet. 2014b;46:1115–1119. doi: 10.1038/ng.3078. [DOI] [PubMed] [Google Scholar]
- Chinta SJ, Woods G, Rane A, Demaria M, Campisi J, Andersen JK. Cellular senescence and the aging brain. Exp Gerontol. 2015;68:3–7. doi: 10.1016/j.exger.2014.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choocheep K, Hatano S, Takagi H, Watanabe H, Kimata K, Kongtawelert P, Watanabe H. Versican facilitates chondrocyte differentiation and regulates joint morphogenesis. J Biol Chem. 2010;285:21114–21125. doi: 10.1074/jbc.M109.096479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collaborative Normal-Tension Glaucoma Study Group. The effectiveness of intraocular pressure reduction in the treatment of normal-tension glaucoma. Collaborative normal-tension glaucoma dtudy group. Am J Ophthalmol. 1998;126:498–505. doi: 10.1016/s0002-9394(98)00272-4. [DOI] [PubMed] [Google Scholar]
- Colucci-D’Amato L, Perrone-Capano C, di Porzio U. Chronic activation of ERK and neurodegenerative diseases. Bioessays. 2003;25:1085–1095. doi: 10.1002/bies.10355. [DOI] [PubMed] [Google Scholar]
- Coppe JP, Desprez PY, Krtolica A, Campisi J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol. 2010;5:99–118. doi: 10.1146/annurev-pathol-121808-102144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornes BK, Khor CC, Nongpiur ME, Xu L, Tay WT, Zheng Y, Lavanya R, Li Y, Wu R, Sim X, Wang YX, Chen P, Teo YY, Chia KS, Seielstad M, Liu J, Hibberd ML, Cheng CY, Saw SM, Tai ES, Jonas JB, Vithana EN, Wong TY, Aung T. Identification of four novel variants that influence central corneal thickness in multi-ethnic Asian populations. Hum Mol Genet. 2012;21:437–445. doi: 10.1093/hmg/ddr463. [DOI] [PubMed] [Google Scholar]
- Costet P, Luo Y, Wang N, Tall AR. Sterol-dependent transactivation of the ABC1 promoter by the liver X receptor/retinoid X receptor. J Biol Chem. 2000;275:28240–28245. doi: 10.1074/jbc.M003337200. [DOI] [PubMed] [Google Scholar]
- Crawford Downs J, Roberts MD, Sigal IA. Glaucomatous cupping of the lamina cribrosa: a review of the evidence for active progressive remodeling as a mechanism. Exp Eye Res. 2011;93:133–140. doi: 10.1016/j.exer.2010.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuaz-Perolin C, Billiet L, Bauge E, Copin C, Scott-Algara D, Genze F, Buchele B, Syrovets T, Simmet T, Rouis M. Antiinflammatory and antiatherogenic effects of the NF-kappaB inhibitor acetyl-11-keto-beta-boswellic acid in LPS-challenged ApoE−/− mice. Arterioscler Thromb Vasc Biol. 2008;28:272–277. doi: 10.1161/ATVBAHA.107.155606. [DOI] [PubMed] [Google Scholar]
- Dewundara SS, Wiggs JL, Sullivan DA, Pasquale LR. Is estrogen a therapeutic target for glaucoma? Semin Ophthalmol. 2016;31:140–146. doi: 10.3109/08820538.2015.1114845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diep CH, Charles NJ, Gilks CB, Kalloger SE, Argenta PA, Lange CA. Progesterone receptors induce FOXO1-dependent senescence in ovarian cancer cells. Cell Cycle. 2013;12:1433–1449. doi: 10.4161/cc.24550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dietz JA, Maes ME, Huang S, Yandell BS, Schlamp CL, Montgomery AD, Allingham RR, Hauser MA, Nickells RW. Spink2 modulates apoptotic susceptibility and is a candidate gene in the Rgcs1 QTL that affects retinal ganglion cell death after optic nerve damage. PLoS One. 2014;9:e93564. doi: 10.1371/journal.pone.0093564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dimayuga FO, Reed JL, Carnero GA, Wang C, Dimayuga ER, Dimayuga VM, Perger A, Wilson ME, Keller JN, Bruce-Keller AJ. Estrogen and brain inflammation: effects on microglial expression of MHC, costimulatory molecules and cytokines. J Neuroimmunol. 2005;161:123–136. doi: 10.1016/j.jneuroim.2004.12.016. [DOI] [PubMed] [Google Scholar]
- Diotel N, Servili A, Gueguen MM, Mironov S, Pellegrini E, Vaillant C, Zhu Y, Kah O, Anglade I. Nuclear progesterone receptors are up-regulated by estrogens in neurons and radial glial progenitors in the brain of zebrafish. PLoS One. 2011;6:e28375. doi: 10.1371/journal.pone.0028375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dodel RC, Du Y, Bales KR, Gao F, Paul SM. Sodium salicylate and 17beta-estradiol attenuate nuclear transcription factor NF-kappaB translocation in cultured rat astroglial cultures following exposure to amyloid A beta(1–40) and lipopolysaccharides. J Neurochem. 1999;73:1453–1460. doi: 10.1046/j.1471-4159.1999.0731453.x. [DOI] [PubMed] [Google Scholar]
- Domenici L, Origlia N, Falsini B, Cerri E, Barloscio D, Fabiani C, Sanso M, Giovannini L. Rescue of retinal function by BDNF in a mouse model of glaucoma. PLoS One. 2014;9:e115579. doi: 10.1371/journal.pone.0115579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellenrieder V. TGFbeta regulated gene expression by Smads and Sp1/KLF-like transcription factors in cancer. Anticancer Res. 2008;28:1531–1539. [PubMed] [Google Scholar]
- Esmon CT. The protein C pathway. Chest. 2003;124:26S–32S. doi: 10.1378/chest.124.3_suppl.26s. [DOI] [PubMed] [Google Scholar]
- Fan BJ, Wang DY, Tham CCY, Lam DSC, Pang CP. Gene expression profiles of human trabecular meshwork cells induced by triamcinolone and dexamethasone. Invest Ophthalmol Vis Sci. 2008;49:1886–1897. doi: 10.1167/iovs.07-0414. [DOI] [PubMed] [Google Scholar]
- Farrer LA, Cupples LA, Haines JL, Hyman B, Kukull WA, Mayeux R, Myers RH, Pericak-Vance MA, Risch N, van Duijn CM. Effects of age, sex, and ethnicity on the association between apolipoprotein E genotype and Alzheimer disease. A meta-analysis APOE and Alzheimer disease meta analysis consortium. JAMA. 1997;278:1349–1356. [PubMed] [Google Scholar]
- Fatma N, Kubo E, Toris CB, Stamer WD, Camras CB, Singh DP. PRDX6 attenuates oxidative stress- and TGFbeta-induced abnormalities of human trabecular meshwork cells. Free Radic Res. 2009;43:783–795. doi: 10.1080/10715760903062887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forte S, Grybauskas A, Kuprys PV, Wanderling C, Walker L, Velagapudi C, Samples JR, Johnson A, Zaparackas Z, Knepper PA. Platelet Toll-like receptor 4 expression in POAG: the innate immune system. Invest Ophthalmol Vis Sci. 2014;55:4523–4523. [Google Scholar]
- Freund A, Orjalo AV, Desprez PY, Campisi J. Inflammatory networks during cellular senescence: causes and consequences. Trends Mol Med. 2010;16:238–246. doi: 10.1016/j.molmed.2010.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedman DS, Wolfs RC, O’Colmain BJ, Klein BE, Taylor HR, West S, Leske MC, Mitchell P, Congdon N, Kempen J, Eye Diseases Prevalence Research, G. Prevalence of open-angle glaucoma among adults in the United States. Arch Ophthalmol. 2004;122:532–538. doi: 10.1001/archopht.122.4.532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuchshofer R, Tamm ER. The role of TGF-beta in the pathogenesis of primary open-angle glaucoma. Cell Tissue Res. 2012;347:279–290. doi: 10.1007/s00441-011-1274-7. [DOI] [PubMed] [Google Scholar]
- Fujikawa K, Iwata T, Inoue K, Akahori M, Kadotani H, Fukaya M, Watanabe M, Chang Q, Barnett EM, Swat W. VAV2 and VAV3 as candidate disease genes for spontaneous glaucoma in mice and humans. PLoS One. 2010;5:e9050. doi: 10.1371/journal.pone.0009050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuxe J, Akusjarvi G, Goike HM, Roos G, Collins VP, Pettersson RF. Adenovirus-mediated overexpression of p15INK4B inhibits human glioma cell growth, induces replicative senescence, and inhibits telomerase activity similarly to p16INK4A. Cell Growth Differ. 2000;11:373–384. [PubMed] [Google Scholar]
- Fyhrquist F, Saijonmaa O, Strandberg T. The roles of senescence and telomere shortening in cardiovascular disease. Nat Rev Cardiol. 2013;10:274–283. doi: 10.1038/nrcardio.2013.30. [DOI] [PubMed] [Google Scholar]
- Galindo-Romero C, Valiente-Soriano FJ, Jimenez-Lopez M, Garcia-Ayuso D, Villegas-Perez MP, Vidal-Sanz M, Agudo-Barriuso M. Effect of brain-derived neurotrophic factor on mouse axotomized retinal ganglion cells and phagocytic microglia. Invest Ophthalmol Vis Sci. 2013;54:974–985. doi: 10.1167/iovs.12-11207. [DOI] [PubMed] [Google Scholar]
- Ganatra DA, Johar KS, Parmar TJ, Patel AR, Rajkumar S, Arora AI, Kayastha FB, Pal AK, Gajjar DU, Vasavada AR. Estrogen mediated protection of cytoskeleton against oxidative stress. Indian J Med Res. 2013;137:117–124. [PMC free article] [PubMed] [Google Scholar]
- Gaudet AD, Popovich PG. Extracellular matrix regulation of inflammation in the healthy and injured spinal cord. Exp Neurol. 2014;258:24–34. doi: 10.1016/j.expneurol.2013.11.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gemenetzi M, Yang Y, Lotery AJ. Current concepts on primary open-angle glaucoma genetics: a contribution to disease pathophysiology and future treatment. Eye Lond.d) 2012;26:355–369. doi: 10.1038/eye.2011.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gharahkhani P, Burdon KP, Fogarty R, Sharma S, Hewitt AW, Martin S, Law MH, Cremin K, Bailey JNC, Loomis SJ, Pasquale LR, Haines JL, Hauser MA, Viswanathan AC, McGuffin P, Topouzis F, Foster PJ, Graham SL, Casson RJ, Chehade M, White AJ, Zhou T, Souzeau E, Landers J, Fitzgerald JT, Klebe S, Ruddle JB, Goldberg I, Healey PR, Wellcome Trust Case Control, C., Consortium, N. Mills RA, Wang JJ, Montgomery GW, Martin NG, Radford-Smith G, Whiteman DC, Brown MA, Wiggs JL, Mackey DA, Mitchell P, MacGregor S, Craig JE. Common variants near ABCA1, AFAP1 and GMDS confer risk of primary open-angle glaucoma. Nat Genet. 2014;46:1120–1125. doi: 10.1038/ng.3079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giovingo M, Nolan M, McCarty R, Pang IH, Clark AF, Beverley RM, Schwartz S, Stamer WD, Walker L, Grybauskas A, Skuran K, Kuprys PV, Yue BY, Knepper PA. sCD44 overexpression increases intraocular pressure and aqueous outflow resistance. Mol Vis. 2013;19:2151–2164. [PMC free article] [PubMed] [Google Scholar]
- Greco A, Rizzo MI, De Virgilio A, Gallo A, Fusconi M, de Vincentiis M. Emerging concepts in glaucoma and review of the literature. Am J Med. 2016 Sep;129(9):1000.e7–1000.e13. doi: 10.1016/j.amjmed.2016.03.038. [DOI] [PubMed] [Google Scholar]
- Guo MS, Wu YY, Liang ZB. Hyaluronic acid increases MMP-2 and MMP-9 expressions in cultured trabecular meshwork cells from patients with primary open-angle glaucoma. Mol Vis. 2012;18:1175–1181. [PMC free article] [PubMed] [Google Scholar]
- Gyatsho J, Kaushik S, Gupta A, Pandav SS, Ram J. Retinal nerve fiber layer thickness in normal, ocular hypertensive, and glaucomatous Indian eyes: an optical coherence tomography study. J Glaucoma. 2008;17:122–127. doi: 10.1097/IJG.0b013e31814b9817. [DOI] [PubMed] [Google Scholar]
- Hamad A, Kluk M, Fox J, Park M, Turner JE. The effects of aromatase inhibitors and selective estrogen receptor modulators on eye development in the zebrafish (Danio rerio) Curr Eye Res. 2007;32:819–827. doi: 10.1080/02713680701573712. [DOI] [PubMed] [Google Scholar]
- Hayden MS, Ghosh S. NF-kappaB, the first quarter-century: remarkable progress and outstanding questions. Genes Dev. 2012;26:203–234. doi: 10.1101/gad.183434.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He S, Minton AZ, Ma HY, Stankowska DL, Sun X, Krishnamoorthy RR. Involvement of AP-1 and C/EBPβ in upregulation of endothelin B (ETB) receptor expression in a rodent model of glaucoma. PLoS One. 2013;8:e79183. doi: 10.1371/journal.pone.0079183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Head BP, Peart JN, Panneerselvam M, Yokoyama T, Pearn ML, Niesman IR, Bonds JA, Schilling JM, Miyanohara A, Headrick J, Ali SS, Roth DM, Patel PM, Patel HH. Loss of caveolin-1 accelerates neurodegeneration and aging. PLoS One. 2010;5:e15697. doi: 10.1371/journal.pone.0015697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heijl A, Leske MC, Bengtsson B, Hyman L, Bengtsson B, Hussein M, Early manifest glaucoma trial, G Reduction of intraocular pressure and glaucoma progression: results from the early manifest glaucoma trial. Arch Ophthalmol. 2002;120:1268–1279. doi: 10.1001/archopht.120.10.1268. [DOI] [PubMed] [Google Scholar]
- Hirsch-Reinshagen V, Zhou S, Burgess BL, Bernier L, McIsaac SA, Chan JY, Tansley GH, Cohn JS, Hayden MR, Wellington CL. Deficiency of ABCA1 impairs apolipoprotein E metabolism in brain. J Biol Chem. 2004;279:41197–41207. doi: 10.1074/jbc.M407962200. [DOI] [PubMed] [Google Scholar]
- Hoehn R, Zeller T, Verhoeven VJ, Grus F, Adler M, Wolfs RC, Uitterlinden AG, Castagne R, Schillert A, Klaver CC, Pfeiffer N, Mirshahi A. Population-based meta-analysis in Caucasians confirms association with COL5A1 and ZNF469 but not COL8A2 with central corneal thickness. Hum Genet. 2012;131:1783–1793. doi: 10.1007/s00439-012-1201-3. [DOI] [PubMed] [Google Scholar]
- Hof PR, Morrison JH. The aging brain: morphomolecular senescence of cortical circuits. Trends Neurosci. 2004;27:607–613. doi: 10.1016/j.tins.2004.07.013. [DOI] [PubMed] [Google Scholar]
- Holdt LM, Hoffmann S, Sass K, Langenberger D, Scholz M, Krohn K, Finstermeier K, Stahringer A, Wilfert W, Beutner F, Gielen S, Schuler G, Gabel G, Bergert H, Bechmann I, Stadler PF, Thiery J, Teupser D. Alu elements in ANRIL non-coding RNA at chromosome 9p21 modulate atherogenic cell functions through trans-regulation of gene networks. PLoS Genet. 2013;9:e1003588. doi: 10.1371/journal.pgen.1003588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmberg J, Liu CY, Hjalt TA. PITX2 gain-of-function in Rieger syndrome eye model. Am J Pathol. 2004;165:1633–1641. doi: 10.1016/S0002-9440(10)63420-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howell GR, Libby RT, Jakobs TC, Smith RS, Phalan FC, Barter JW, Barbay JM, Marchant JK, Mahesh N, Porciatti V, Whitmore AV, Masland RH, John SW. Axons of retinal ganglion cells are insulted in the optic nerve early in DBA/2J glaucoma. J Cell Biol. 2007;179:1523–1537. doi: 10.1083/jcb.200706181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howell GR, Macalinao DG, Sousa GL, Walden M, Soto I, Kneeland SC, Barbay JM, King BL, Marchant JK, Hibbs M, Stevens B, Barres BA, Clark AF, Libby RT, John SWM. Molecular clustering identifies complement and endothelin induction as early events in a mouse model of glaucoma. J Clin Invest. 2011;121:1429–1444. doi: 10.1172/JCI44646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hubackova S, Krejcikova K, Bartek J, Hodny Z. IL1- and TGFbeta-Nox4 signaling, oxidative stress and DNA damage response are shared features of replicative, oncogene-induced, and drug-induced paracrine ‘bystander senescence’. Aging (Albany NY) 2012;4:932–951. doi: 10.18632/aging.100520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hysi PG, Cheng CY, Springelkamp H, Macgregor S, Bailey JNC, Wojciechowski R, Vitart V, Nag A, Hewitt AW, Höhn R, Venturini C, Mirshahi A, Ramdas WD, Thorleifsson G, Vithana E, Khor CC, Stefansson AB, Liao J, Haines JL, Amin N, Wang YX, Wild PS, Ozel AB, Li JZ, Fleck BW, Zeller T, Staffieri SE, Teo YY, Cuellar-Partida G, Luo X, Allingham RR, Richards JE, Senft A, Karssen LC, Zheng Y, Bellenguez C, Xu L, Iglesias AI, Wilson JF, Kang JH, van Leeuwen EM, Jonsson V, Thorsteinsdottir U, Despriet DDG, Ennis S, Moroi SE, Martin NG, Jansonius NM, Yazar S, Tai ES, Amouyel P, Kirwan J, van Koolwijk LME, Hauser MA, Jonasson F, Leo P, Loomis SJ, Fogarty R, Rivadeneira F, Kearns L, Lackner KJ, de Jong PTVM, Simpson CL, Pennell CE, Oostra BA, Uitterlinden AG, Saw SM, Lotery AJ, Bailey-Wilson JE, Hofman A, Vingerling JR, Maubaret C, Pfeiffer N, Wolfs RCW, Lemij HG, Young TL, Pasquale LR, Delcourt C, Spector TD, Klaver CCW, Small KS, Burdon KP, Stefansson K, Wong TY, BG Group, N Consortium, Wellcome Trust Case Control, C. Viswanathan A, Mackey DA, Craig JE, Wiggs JL, van Duijn CM, Hammond CJ, Aung T. Genome-wide analysis of multi-ancestry cohorts identifies new loci influencing intraocular pressure and susceptibility to glaucoma. Nat Genet. 2014;46:1126–1130. doi: 10.1038/ng.3087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iglesias AI, Springelkamp H, Ramdas WD, Klaver CC, Willemsen R, van Duijn CM. Genes, pathways, and animal models in primary open-angle glaucoma. Eye Lond.d) 2015;29:1285–1298. doi: 10.1038/eye.2015.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iglesias AI, Springelkamp H, van der Linde H, Severijnen LA, Amin N, Oostra B, Kockx CEM, van den Hout MCGN, van Ijcken WFJ, Hofman A, Uitterlinden AG, Verdijk RM, Klaver CCW, Willemsen R, van Duijn CM. Exome sequencing and functional analyses suggest that SIX6 is a gene involved in an altered proliferation-differentiation balance early in life and optic nerve degeneration at old age. Hum Mol Genet. 2014;23:1320–1332. doi: 10.1093/hmg/ddt522. [DOI] [PubMed] [Google Scholar]
- Imanishi T, Kobayashi K, Hano T, Nishio I. Effect of estrogen on differentiation and senescence in endothelial progenitor cells derived from bone marrow in spontaneously hypertensive rats. Hypertens Res. 2005;28:763–772. doi: 10.1291/hypres.28.763. [DOI] [PubMed] [Google Scholar]
- Janssen SF, Gorgels TG, Ramdas WD, Klaver CC, van Duijn CM, Jansonius NM, Bergen AA. The vast complexity of primary open angle glaucoma: disease genes, risks, molecular mechanisms and pathobiology. Prog Retin Eye Res. 2013;37:31–67. doi: 10.1016/j.preteyeres.2013.09.001. [DOI] [PubMed] [Google Scholar]
- Jansson I, Stoilov I, Sarfarazi M, Schenkman JB. Effect of two mutations of human CYP1B1, G61E and R469W, on stability and endogenous steroid substrate metabolism. Pharmacogenetics. 2001;11:793–801. doi: 10.1097/00008571-200112000-00007. [DOI] [PubMed] [Google Scholar]
- Jarinova O, Stewart AF, Roberts R, Wells G, Lau P, Naing T, Buerki C, McLean BW, Cook RC, Parker JS, McPherson R. Functional analysis of the chromosome 9p21.3 coronary artery disease risk locus. Arterioscler Thromb Vasc Biol. 2009;29:1671–1677. doi: 10.1161/ATVBAHA.109.189522. [DOI] [PubMed] [Google Scholar]
- Jeon H, Boo YC. Senescent endothelial cells are prone to TNF-alpha-induced cell death due to expression of FAS receptor. Biochem Biophys Res Commun. 2013;438:277–282. doi: 10.1016/j.bbrc.2013.07.052. [DOI] [PubMed] [Google Scholar]
- Joe MK, Kwon HS, Cojocaru R, Tomarev SI. Myocilin regulates cell proliferation and survival. J Biol Chem. 2014;289:10155–10167. doi: 10.1074/jbc.M113.547091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joe MK, Tomarev SI. Expression of myocilin mutants sensitizes cells to oxidative stress-induced apoptosis: implication for glaucoma pathogenesis. Am J Pathol. 2010;176:2880–2890. doi: 10.2353/ajpath.2010.090853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson EC, Morrison JC. Friend or foe? Resolving the impact of glial responses in glaucoma. J Glaucoma. 2009;18:341–353. doi: 10.1097/IJG.0b013e31818c6ef6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson M. What controls aqueous humour outflow resistance? Exp Eye Res. 2006;82:545–557. doi: 10.1016/j.exer.2005.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jungert K, Buck A, von Wichert G, Adler G, Konig A, Buchholz M, Gress TM, Ellenrieder V. Sp1 is required for transforming growth factor-beta-induced mesenchymal transition and migration in pancreatic cancer cells. Cancer Res. 2007;67:1563–1570. doi: 10.1158/0008-5472.CAN-06-1670. [DOI] [PubMed] [Google Scholar]
- Kaimoto T, Yasuda O, Ohishi M, Mogi M, Takemura Y, Suhara T, Ogihara T, Fukuo K, Rakugi H. Nifedipine inhibits vascular smooth muscle cell dedifferentiation via downregulation of Akt signaling. Hypertension. 2010;56:247–252. doi: 10.1161/HYPERTENSIONAHA.110.149781. [DOI] [PubMed] [Google Scholar]
- Kamburov A, Pentchev K, Galicka H, Wierling C, Lehrach H, Herwig R. ConsensusPathDB: toward a more complete picture of cell biology. Nucleic Acids Res. 2011;39:D712–D717. doi: 10.1093/nar/gkq1156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamburov A, Wierling C, Lehrach H, Herwig R. ConsensusPathDB–a database for integrating human functional interaction networks. Nucleic Acids Res. 2009;37:D623–D628. doi: 10.1093/nar/gkn698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang HJ, Kim HJ, Kim SK, Barouki R, Cho CH, Khanna KK, Rosen EM, Bae I. BRCA1 modulates xenobiotic stress-inducible gene expression by interacting with ARNT in human breast cancer cells. J Biol Chem. 2006;281:14654–14662. doi: 10.1074/jbc.M601613200. [DOI] [PubMed] [Google Scholar]
- Kanski JJ, Bowling B, Nischal KK, Pearson A. Ch 10 Glaucoma, Clinical Ophthalmology: a Systematic Approach. seventh. Elsevier/Saunders; Edinburgh, New York: 2011. p. 1. online resource (ix, 909 pp.) [Google Scholar]
- Karreth F, Hoebertz A, Scheuch H, Eferl R, Wagner EF. The AP1 transcription factor Fra2 is required for efficient cartilage development. Development. 2004;131:5717–5725. doi: 10.1242/dev.01414. [DOI] [PubMed] [Google Scholar]
- Kass MA, Heuer DK, Higginbotham EJ, Johnson CA, Keltner JL, Miller JP, Parrish RK, 2nd, Wilson MR, Gordon MO. The Ocular Hypertension Treatment Study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma. Arch Ophthalmol. 2002;120:701–713. doi: 10.1001/archopht.120.6.701. discussion 829–730. [DOI] [PubMed] [Google Scholar]
- Katoh H, Yasui H, Yamaguchi Y, Aoki J, Fujita H, Mori K, Negishi M. Small GTPase RhoG is a key regulator for neurite outgrowth in PC12 cells. Mol Cell Biol. 2000;20:7378–7387. doi: 10.1128/mcb.20.19.7378-7387.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan AO, Al-Abdi L, Mohamed JY, Aldahmesh MA, Alkuraya FS. Familial juvenile glaucoma with underlying homozygous pG61E CYP1B1 mutations. J AAPOS. 2011a;15:198–199. doi: 10.1016/j.jaapos.2011.01.156. [DOI] [PubMed] [Google Scholar]
- Khan K, Rudkin A, Parry DA, Burdon KP, McKibbin M, Logan CV, Abdelhamed ZIA, Muecke JS, Fernandez-Fuentes N, Laurie KJ, Shires M, Fogarty R, Carr IM, Poulter JA, Morgan JE, Mohamed MD, Jafri H, Raashid Y, Meng N, Piseth H, Toomes C, Casson RJ, Taylor GR, Hammerton M, Sheridan E, Johnson CA, Inglehearn CF, Craig JE, Ali M. Homozygous mutations in PXDN cause congenital cataract, corneal opacity, and developmental glaucoma. Am J Hum Genet. 2011b;89:464–473. doi: 10.1016/j.ajhg.2011.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kigerl KA, Lai W, Rivest S, Hart RP, Satoskar AR, Popovich PG. Toll-like receptor (TLR)-2 and TLR-4 regulate inflammation, gliosis, and myelin sparing after spinal cord injury. J Neurochem. 2007;102:37–50. doi: 10.1111/j.1471-4159.2007.04524.x. [DOI] [PubMed] [Google Scholar]
- Kim S, Takahashi H, Lin WW, Descargues P, Grivennikov S, Kim Y, Luo JL, Karin M. Carcinoma-produced factors activate myeloid cells through TLR2 to stimulate metastasis. Nature. 2009;457:102–106. doi: 10.1038/nature07623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirwan RP, Wordinger RJ, Clark AF, O’Brien CJ. Differential global and extra-cellular matrix focused gene expression patterns between normal and glaucomatous human lamina cribrosa cells. Mol Vis. 2009;15:76–88. [PMC free article] [PubMed] [Google Scholar]
- Kloeckener-Gruissem B, Bartholdi D, Abdou MT, Zimmermann DR, Berger W. Identification of the genetic defect in the original Wagner syndrome family. Mol Vis. 2006;12:350–355. [PubMed] [Google Scholar]
- Knepper PA, Collins JA, Frederick R. Effects of dexamethasone, progesterone, and testosterone on IOP and GAGs in the rabbit eye. Invest Ophthalmol Vis Sci. 1985;26:1093–1100. [PubMed] [Google Scholar]
- Kordinas V, Ioannidis A, Chatzipanagiotou S. The telomere/telomerase system in chronic inflammatory diseases. Cause or effect? Genes (Basel) 2016;7 doi: 10.3390/genes7090060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korf H, Vander Beken S, Romano M, Steffensen KR, Stijlemans B, Gustafsson JA, Grooten J, Huygen K. Liver X receptors contribute to the protective immune response against Mycobacterium tuberculosis in mice. J Clin Invest. 2009;119:1626–1637. doi: 10.1172/JCI35288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar R, Selth LA, Schulz RB, Tay BS, Neilsen PM, Callen DF. Genome-wide mapping of ZNF652 promoter binding sites in breast cancer cells. J Cell Biochem. 2011;112:2742–2747. doi: 10.1002/jcb.23214. [DOI] [PubMed] [Google Scholar]
- Kwon YH, Fingert JH, Kuehn MH, Alward WL. Primary open-angle glaucoma. N Engl J Med. 2009;360:1113–1124. doi: 10.1056/NEJMra0804630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon YJ, Baek HS, Ye DJ, Shin S, Kim D, Chun YJ. CYP1B1 enhances cell proliferation and metastasis through induction of EMT and activation of wnt/beta-catenin signaling via Sp1 upregulation. PLoS One. 2016;11:e0151598. doi: 10.1371/journal.pone.0151598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landis MS, Patel HV, Capone JP. Oxysterol activators of liver X receptor and 9-cis-retinoic acid promote sequential steps in the synthesis and secretion of tumor necrosis factor-alpha from human monocytes. J Biol Chem. 2002;277:4713–4721. doi: 10.1074/jbc.M108807200. [DOI] [PubMed] [Google Scholar]
- Laureys G, Gerlo S, Spooren A, Demol F, De Keyser J, Aerts JL. beta(2)-adrenergic agonists modulate TNF-alpha induced astrocytic inflammatory gene expression and brain inflammatory cell populations. J Neuroinflamm. 2014;11:21. doi: 10.1186/1742-2094-11-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavedan C, Buchholtz S, Nussbaum RL, Albin RL, Polymeropoulos MH. A mutation in the human neurofilament M gene in Parkinson’s disease that suggests a role for the cytoskeleton in neuronal degeneration. Neurosci Lett. 2002;322:57–61. doi: 10.1016/s0304-3940(01)02513-7. [DOI] [PubMed] [Google Scholar]
- Lebrun-Julien F, Bertrand MJ, De Backer O, Stellwagen D, Morales CR, Di Polo A, Barker PA. ProNGF induces TNFalpha-dependent death of retinal ganglion cells through a p75NTR non-cell-autonomous signaling pathway. Proc Natl Acad Sci U S A. 2010;107:3817–3822. doi: 10.1073/pnas.0909276107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lei ZN, Zeng SL, Wang L, Zhu JB, Li T. Parthenolide inhibits neuroinflammation and promotes neurogenesis in the ischemic striatum following transient middle cerebral artery occlusion in the adult rat brain. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2009;25:994–997. [PubMed] [Google Scholar]
- Levin LA, Albert DM, Johnson D. Mast cells in human optic nerve. Invest Ophthalmol Vis Sci. 1993;34:3147–3153. [PubMed] [Google Scholar]
- Li J, Tripathi BJ, Chalam KV, Tripathi RC. Transforming growth factor-beta 1 and -beta 2 positively regulate TGF-beta 1 mRNA expression in trabecular cells. Invest Ophthalmol Vis Sci. 1996;37:2778–2782. [PubMed] [Google Scholar]
- Li Y, Semaan SJ, Schlamp CL, Nickells RW. Dominant inheritance of retinal ganglion cell resistance to optic nerve crush in mice. BMC Neurosci. 2007;8:19. doi: 10.1186/1471-2202-8-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Shin D, Kwon SH. Histone deacetylase 6 plays a role as a distinct regulator of diverse cellular processes. FEBS J. 2013;280:775–793. doi: 10.1111/febs.12079. [DOI] [PubMed] [Google Scholar]
- Liao R, Ye M, Xu X. An updated meta-analysis: apolipoprotein E genotypes and risk of primary open-angle glaucoma. Mol Vis. 2014;20:1025–1036. [PMC free article] [PubMed] [Google Scholar]
- Libby RT, Gould DB, Anderson MG, John SW. Complex genetics of glaucoma susceptibility. Annu Rev Genomics Hum Genet. 2005;6:15–44. doi: 10.1146/annurev.genom.6.080604.162209. [DOI] [PubMed] [Google Scholar]
- Lindholm D, Castren E, Kiefer R, Zafra F, Thoenen H. Transforming growth factor-beta 1 in the rat brain: increase after injury and inhibition of astrocyte proliferation. J Cell Biol. 1992;117:395–400. doi: 10.1083/jcb.117.2.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu CC, Kanekiyo T, Xu H, Bu G. Apolipoprotein E and Alzheimer disease: risk, mechanisms and therapy. Nat Rev Neurol. 2013;9:106–118. doi: 10.1038/nrneurol.2012.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loomis SJ, Kang JH, Weinreb RN, Yaspan BL, Cooke Bailey JN, Gaasterland D, Gaasterland T, Lee RK, Lichter PR, Budenz DL, Liu Y, Realini T, Friedman DS, McCarty CA, Moroi SE, Olson L, Schuman JS, Singh K, Vollrath D, Wollstein G, Zack DJ, Brilliant M, Sit AJ, Christen WG, Fingert J, Kraft P, Zhang K, Allingham RR, Pericak-Vance MA, Richards JE, Hauser MA, Haines JL, Pasquale LR, Wiggs JL. Association of CAV1/CAV2 genomic variants with primary open-angle glaucoma overall and by gender and pattern of visual field loss. Ophthalmology. 2014;121:508–516. doi: 10.1016/j.ophtha.2013.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Low JY, Nicholson HD. Epigenetic modifications of caveolae associated proteins in health and disease. BMC Genet. 2015;16:71. doi: 10.1186/s12863-015-0231-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo D, Liu K, Zhu B, Xu X. Expression profiling in glaucomatous human lamina cribrosa cells based on graph-clustering approach. Curr Eye Res. 2013;38:767–773. doi: 10.3109/02713683.2013.770039. [DOI] [PubMed] [Google Scholar]
- Luo JM, Cen LP, Zhang XM, Chiang SW, Huang Y, Lin D, Fan YM, van Rooijen N, Lam DS, Pang CP, Cui Q. PI3K/akt, JAK/STAT and MEK/ERK pathway inhibition protects retinal ganglion cells via different mechanisms after optic nerve injury. Eur J Neurosci. 2007;26:828–842. doi: 10.1111/j.1460-9568.2007.05718.x. [DOI] [PubMed] [Google Scholar]
- Majsterek I, Markiewicz L, Przybylowska K, Gacek M, Kurowska AK, Kaminska A, Szaflik J, Szaflik JP. Association of MMP1-1607 1G/2G and TIMP1 372 T/C gene polymorphisms with risk of primary open angle glaucoma in a Polish population. Med Sci Monit. 2011;17:CR417–421. doi: 10.12659/MSM.881854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mallick J, Devi L, Malik PK, Mallick J. Update on normal tension glaucoma. J Ophthalmic Vis Res. 2016;11:204–208. doi: 10.4103/2008-322X.183914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markiewicz L, Majsterek I, Przybylowska K, Dziki L, Waszczyk M, Gacek M, Kaminska A, Szaflik J, Szaflik JP. Gene polymorphisms of the MMP1, MMP9, MMP12, IL-1β and TIMP1 and the risk of primary open-angle glaucoma. Acta Ophthalmol. 2013;91:e516–523. doi: 10.1111/aos.12149. [DOI] [PubMed] [Google Scholar]
- Martinon F, Aksentijevich I. New players driving inflammation in monogenic autoinflammatory diseases. Nat Rev Rheumatol. 2015;11:11–20. doi: 10.1038/nrrheum.2014.158. [DOI] [PubMed] [Google Scholar]
- Mathema VB, Koh YS, Thakuri BC, Sillanpaa M. Parthenolide, a sesquiterpene lactone, expresses multiple anti-cancer and anti-inflammatory activities. Inflammation. 2012;35:560–565. doi: 10.1007/s10753-011-9346-0. [DOI] [PubMed] [Google Scholar]
- Matsuda T, Yamamoto T, Muraguchi A, Saatcioglu F. Cross-talk between transforming growth factor-beta and estrogen receptor signaling through Smad3. J Biol Chem. 2001;276:42908–42914. doi: 10.1074/jbc.M105316200. [DOI] [PubMed] [Google Scholar]
- Mead B, Logan A, Berry M, Leadbeater W, Scheven BA. Intravitreally transplanted dental pulp stem cells promote neuroprotection and axon regeneration of retinal ganglion cells after optic nerve injury. Invest Ophthalmol Vis Sci. 2013;54:7544–7556. doi: 10.1167/iovs.13-13045. [DOI] [PubMed] [Google Scholar]
- Mead B, Scheven BA. Mesenchymal stem cell therapy for retinal ganglion cell neuroprotection and axon regeneration. Neural Regen Res. 2015;10:371–373. doi: 10.4103/1673-5374.153681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mey J, Thanos S. Intravitreal injections of neurotrophic factors support the survival of axotomized retinal ganglion cells in adult rats in vivo. Brain Res. 1993;602:304–317. doi: 10.1016/0006-8993(93)90695-j. [DOI] [PubMed] [Google Scholar]
- Miao H, Crabb AW, Hernandez MR, Lukas TJ. Modulation of factors affecting optic nerve head astrocyte migration. Invest Ophthalmol Vis Sci. 2010;51:4096–4103. doi: 10.1167/iovs.10-5177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minegishi Y, Iejima D, Kobayashi H, Chi ZL, Kawase K, Yamamoto T, Seki T, Yuasa S, Fukuda K, Iwata T. Enhanced optineurin E50K-TBK1 interaction evokes protein insolubility and initiates familial primary open-angle glaucoma. Hum Mol Genet. 2013;22:3559–3567. doi: 10.1093/hmg/ddt210. [DOI] [PubMed] [Google Scholar]
- Mowatt L, McIntosh M. Strategies for neuroprotection in glaucoma. In: Rumelt S, editor. Glaucoma - Basic and Clinical Aspects. InTech; 2013. pp. 203–226. [Google Scholar]
- Neilsen PM, Noll JE, Mattiske S, Bracken CP, Gregory PA, Schulz RB, Lim SP, Kumar R, Suetani RJ, Goodall GJ, Callen DF. Mutant p53 drives invasion in breast tumors through up-regulation of miR-155. Oncogene. 2013;32:2992–3000. doi: 10.1038/onc.2012.305. [DOI] [PubMed] [Google Scholar]
- Neufeld AH, Liu B. Glaucomatous optic neuropathy: when glia misbehave. Neuroscientist. 2003;9:485–495. doi: 10.1177/1073858403253460. [DOI] [PubMed] [Google Scholar]
- Newman-Casey PA, Talwar N, Nan B, Musch DC, Pasquale LR, Stein JD. The potential association between postmenopausal hormone use and primary open-angle glaucoma. JAMA Ophthalmol. 2014;132:298–303. doi: 10.1001/jamaophthalmol.2013.7618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng SK, Casson RJ, Burdon KP, Craig JE. Chromosome 9p21 primary open-angle glaucoma susceptibility locus: a review. Clin Exp Ophthalmol. 2014;42:25–32. doi: 10.1111/ceo.12234. [DOI] [PubMed] [Google Scholar]
- Nickells RW. Apoptosis of retinal ganglion cells in glaucoma: an update of the molecular pathways involved in cell death. Surv Ophthalmol. 1999;43(Suppl. 1):S151–S161. doi: 10.1016/s0039-6257(99)00029-6. [DOI] [PubMed] [Google Scholar]
- Niesman IR, Schilling JM, Shapiro LA, Kellerhals SE, Bonds JA, Kleschevnikov AM, Cui W, Voong A, Krajewski S, Ali SS, Roth DM, Patel HH, Patel PM, Head BP. Traumatic brain injury enhances neuroinflammation and lesion volume in caveolin deficient mice. J Neuroinflamm. 2014;11:39. doi: 10.1186/1742-2094-11-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ofri R, Shore LS, Kass PH, Horowitz IH. The effect of elevated progesterone levels on intraocular pressure in lions (Panthera leo) Res Vet Sci. 1999;67:121–123. doi: 10.1053/rvsc.1998.0300. [DOI] [PubMed] [Google Scholar]
- Parat MO, Riggins GJ. Caveolin-1, caveolae, and glioblastoma. Neuro Oncol. 2012;14:679–688. doi: 10.1093/neuonc/nos079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasutto F, Matsumoto T, Mardin CY, Sticht H, Brandstätter JH, Michels-Rautenstrauss K, Weisschuh N, Gramer E, Ramdas WD, van Koolwijk LME, Klaver CCW, Vingerling JR, Weber BHF, Kruse FE, Rautenstrauss B, Barde YA, Reis A. Heterozygous NTF4 mutations impairing neurotrophin-4 signaling in patients with primary open-angle glaucoma. Am J Hum Genet. 2009;85:447–456. doi: 10.1016/j.ajhg.2009.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pena JD, Agapova O, Gabelt BT, Levin LA, Lucarelli MJ, Kaufman PL, Hernandez MR. Increased elastin expression in astrocytes of the lamina cribrosa in response to elevated intraocular pressure. Invest Ophthalmol Vis Sci. 2001;42:2303–2314. [PubMed] [Google Scholar]
- Perry VH, Gordon S. Macrophages and the nervous system. Int Rev Cytol. 1991;125:203–244. doi: 10.1016/s0074-7696(08)61220-6. [DOI] [PubMed] [Google Scholar]
- Peterfi Z, Donko A, Orient A, Sum A, Prokai A, Molnar B, Vereb Z, Rajnavolgyi E, Kovacs KJ, Muller V, Szabo AJ, Geiszt M. Peroxidasin is secreted and incorporated into the extracellular matrix of myofibroblasts and fibrotic kidney. Am J Pathol. 2009;175:725–735. doi: 10.2353/ajpath.2009.080693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phillips MC. Molecular mechanisms of cellular cholesterol efflux. J Biol Chem. 2014;289:24020–24029. doi: 10.1074/jbc.R114.583658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinero J, Queralt-Rosinach N, Bravo A, Deu-Pons J, Bauer-Mehren A, Baron M, Sanz F, Furlong LI. DisGeNET: a Discovery Platform for the Dynamical Exploration of Human Diseases and Their Genes. Database (Oxford) 2015;2015:bav028. doi: 10.1093/database/bav028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pluquet O, Pourtier A, Abbadie C. The unfolded protein response and cellular senescence. A review in the theme: cellular mechanisms of endoplasmic reticulum stress signaling in health and disease. Am J Physiol Cell Physiol. 2015;308:C415–C425. doi: 10.1152/ajpcell.00334.2014. [DOI] [PubMed] [Google Scholar]
- Ponta H, Sherman L, Herrlich PA. CD44: from adhesion molecules to signalling regulators. Nat Rev Mol Cell Biol. 2003;4:33–45. doi: 10.1038/nrm1004. [DOI] [PubMed] [Google Scholar]
- Purves D, Augustine GJ, Fitzpatrick D, Katz LC, LaMantia A-S, McNamara JO, Williams SM. Neuroscience. In: Purves D, Augustine GJ, Fitzpatrick D, Katz LC, LaMantia A-S, McNamara JO, Williams SM, editors. Neuroscience. third. Sinauer Associates, Inc; 2004. pp. 229–258. [Google Scholar]
- Quigley HA, Broman AT. The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol. 2006;90:262–267. doi: 10.1136/bjo.2005.081224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajasekaran S, Reddy NM, Zhang W, Reddy SP. Expression profiling of genes regulated by Fra-1/AP-1 transcription factor during bleomycin-induced pulmonary fibrosis. BMC Genomics. 2013;14:381. doi: 10.1186/1471-2164-14-381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao KN, Nagireddy S, Chakrabarti S. Complex genetic mechanisms in glaucoma: an overview. Indian J Ophthalmol. 2011;59(Suppl. l):S31–S42. doi: 10.4103/0301-4738.73685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rappaport N, Nativ N, Stelzer G, Twik M, Guan-Golan Y, Stein TI, Bahir I, Belinky F, Morrey CP, Safran M, Lancet D. MalaCards: an Integrated Compendium for Diseases and Their Annotation. Database (Oxford) 2013;2013:bat018. doi: 10.1093/database/bat018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddy SA, Huang JH, Liao WS. Phosphatidylinositol 3-kinase in interleukin 1 signaling. Physical interaction with the interleukin 1 receptor and requirement in NFkappaB and AP-1 activation. J Biol Chem. 1997;272:29167–29173. doi: 10.1074/jbc.272.46.29167. [DOI] [PubMed] [Google Scholar]
- Reddy SA, Huang JH, Liao WS. Phosphatidylinositol 3-kinase as a mediator of TNF-induced NF-kappa B activation. J Immunol. 2000;164:1355–1363. doi: 10.4049/jimmunol.164.3.1355. [DOI] [PubMed] [Google Scholar]
- Reuter S, Gupta SC, Chaturvedi MM, Aggarwal BB. Oxidative stress, inflammation, and cancer: how are they linked? Free Radic Biol Med. 2010;49:1603–1616. doi: 10.1016/j.freeradbiomed.2010.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rezaie T, Child A, Hitchings R, Brice G, Miller L, Coca-Prados M, Héon E, Krupin T, Ritch R, Kreutzer D, Crick RP, Sarfarazi M. Adult-onset primary open-angle glaucoma caused by mutations in optineurin. Science. 2002;295:1077–1079. doi: 10.1126/science.1066901. [DOI] [PubMed] [Google Scholar]
- Rice LM, Padilla CM, McLaughlin SR, Mathes A, Ziemek J, Goummih S, Nakerakanti S, York M, Farina G, Whitfield ML, Spiera RF, Christmann RB, Gordon JK, Weinberg J, Simms RW, Lafyatis R. Fresolimumab treatment decreases biomarkers and improves clinical symptoms in systemic sclerosis patients. J Clin Invest. 2015;125:2795–2807. doi: 10.1172/JCI77958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roh M, Zhang Y, Murakami Y, Thanos A, Lee SC, Vavvas DG, Benowitz LI, Miller JW. Etanercept, a widely used inhibitor of tumor necrosis factor-α (TNF-α), prevents retinal ganglion cell loss in a rat model of glaucoma. PLoS One. 2012;7:e40065. doi: 10.1371/journal.pone.0040065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothschild PR, Brezin AP, Nedelec B, Burin des Roziers C, Ghiotti T, Orhant L, Boimard M, Valleix S. A family with Wagner syndrome with uveitis and a new versican mutation. Mol Vis. 2013;19:2040–2049. [PMC free article] [PubMed] [Google Scholar]
- Rudnicka AR, Mt-Isa S, Owen CG, Cook DG, Ashby D. Variations in primary open-angle glaucoma prevalence by age, gender, and race: a Bayesian meta-analysis. Invest Ophthalmol Vis Sci. 2006;47:4254–4261. doi: 10.1167/iovs.06-0299. [DOI] [PubMed] [Google Scholar]
- Rudzinski MN, Chen L, Hernandez MR. Antiangiogenic characteristics of astrocytes from optic nerve heads with primary open-angle glaucoma. Arch Ophthalmol. 2008;126:679–685. doi: 10.1001/archopht.126.5.679. [DOI] [PubMed] [Google Scholar]
- Rummel C, Gerstberger R, Roth J, Hubschle T. Parthenolide attenuates LPS-induced fever, circulating cytokines and markers of brain inflammation in rats. Cytokine. 2011;56:739–748. doi: 10.1016/j.cyto.2011.09.022. [DOI] [PubMed] [Google Scholar]
- Ryu H, Lee J, Zaman K, Kubilis J, Ferrante RJ, Ross BD, Neve R, Ratan RR. Sp1 and Sp3 are oxidative stress-inducible, antideath transcription factors in cortical neurons. J Neurosci. 2003;23:3597–3606. doi: 10.1523/JNEUROSCI.23-09-03597.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salminen A, Kauppinen A, Kaarniranta K. Emerging role of NF-kappaB signaling in the induction of senescence-associated secretory phenotype (SASP) Cell Signal. 2012;24:835–845. doi: 10.1016/j.cellsig.2011.12.006. [DOI] [PubMed] [Google Scholar]
- Salminen A, Kauppinen A, Suuronen T, Kaarniranta K, Ojala J. ER stress in Alzheimer’s disease: a novel neuronal trigger for inflammation and Alzheimer’s pathology. J Neuroinflamm. 2009;6:41. doi: 10.1186/1742-2094-6-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salowe R, Salinas J, Farbman NH, Mohammed A, Warren JZ, Rhodes A, Brucker A, Regina M, Miller-Ellis E, Sankar PS, Lehman A, O’Brien JM. Primary open-angle glaucoma in individuals of african descent: a review of risk factors. J Clin Exp Ophthalmol. 2015;6 doi: 10.4172/2155-9570.1000450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samantaray S, Smith JA, Das A, Matzelle DD, Varma AK, Ray SK, Banik NL. Low dose estrogen prevents neuronal degeneration and microglial reactivity in an acute model of spinal cord injury: effect of dosing, route of administration, and therapy delay. Neurochem Res. 2011;36:1809–1816. doi: 10.1007/s11064-011-0498-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato T, Wilson TS, Hughes LF, Konrad HR, Nakayama M, Helfert RH. Age-related changes in levels of tyrosine kinase B receptor and fibroblast growth factor receptor 2 in the rat inferior colliculus: implications for neural senescence. Neuroscience. 2001;103:695–702. doi: 10.1016/s0306-4522(01)00022-7. [DOI] [PubMed] [Google Scholar]
- Schallenberg M, Thanos S. Non-histone HMGB1 is a transcriptional factor involved in degeneration of injured RGCs. Invest Ophthalmol Vis Sci. 2009;50:135–135. [Google Scholar]
- Schumacher M, Hussain R, Gago N, Oudinet JP, Mattern C, Ghoumari AM. Progesterone synthesis in the nervous system: implications for myeli-nation and myelin repair. Front Neurosci. 2012;6:10. doi: 10.3389/fnins.2012.00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shields MB. Normal-tension glaucoma: is it different from primary open-angle glaucoma? Curr Opin Ophthalmol. 2008;19:85–88. doi: 10.1097/ICU.0b013e3282f3919b. [DOI] [PubMed] [Google Scholar]
- Shivers KY, Amador N, Abrams L, Hunter D, Jenab S, Quinones-Jenab V. Estrogen alters baseline and inflammatory-induced cytokine levels independent from hypothalamic-pituitary-adrenal axis activity. Cytokine. 2015;72:121–129. doi: 10.1016/j.cyto.2015.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sia DI, Muecke J, Hammerton M, Ngy M, Kong A, Morse A, Holmes M, Piseth H, Hamilton C, Selva D. A survey of visual impairment and blindness in children attending four schools for the blind in Cambodia. Ophthalmic Epidemiol. 2010;17:225–233. doi: 10.3109/09286586.2010.489250. [DOI] [PubMed] [Google Scholar]
- Simoes-Pires C, Zwick V, Nurisso A, Schenker E, Carrupt PA, Cuendet M. HDAC6 as a target for neurodegenerative diseases: what makes it different from the other HDACs? Mol Neurodegener. 2013;8:7. doi: 10.1186/1750-1326-8-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh M, Su C. Progesterone and neuroprotection. Horm Behav. 2013;63:284–290. doi: 10.1016/j.yhbeh.2012.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solomon SS, Majumdar G, Martinez-Hernandez A, Raghow R. A critical role of Sp1 transcription factor in regulating gene expression in response to insulin and other hormones. Life Sci. 2008;83:305–312. doi: 10.1016/j.lfs.2008.06.024. [DOI] [PubMed] [Google Scholar]
- Soto I, Howell GR. The complex role of neuroinflammation in glaucoma. Cold Spring Harb Perspect Med. 2014;4 doi: 10.1101/cshperspect.a017269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spaeth GL, Henderer J, Liu C, Kesen M, Altangerel U, Bayer A, Katz LJ, Myers J, Rhee D, Steinmann W. The disc damage likelihood scale: reproducibility of a new method of estimating the amount of optic nerve damage caused by glaucoma. Trans Am Ophthalmol Soc. 2002;100:181–185. discussion 185–186. [PMC free article] [PubMed] [Google Scholar]
- Spilsbury A, Miwa S, Attems J, Saretzki G. The role of telomerase protein TERT in Alzheimer’s disease and in tau-related pathology in vitro. J Neurosci. 2015;35:1659–1674. doi: 10.1523/JNEUROSCI.2925-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su Y, Cheng J, Liu H, Wang F, Zhao S. Adenovirus conducted connective tissue growth factor on extracellular matrix in trabecular meshwork and its role on aqueous humor outflow facility. Mol Biol Rep. 2013;40:6091–6096. doi: 10.1007/s11033-013-2720-2. [DOI] [PubMed] [Google Scholar]
- Subramaniam S, Zirrgiebel U, von Bohlen Und Halbach O, Strelau J, Laliberte C, Kaplan DR, Unsicker K. ERK activation promotes neuronal degeneration predominantly through plasma membrane damage and independently of caspase-3. J Cell Biol. 2004;165:357–369. doi: 10.1083/jcb.200403028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takamoto M, Araie M. Genetics of primary open angle glaucoma. Jpn J Ophthalmol. 2014;58:1–15. doi: 10.1007/s10384-013-0286-0. [DOI] [PubMed] [Google Scholar]
- Teng MW, Bowman EP, McElwee JJ, Smyth MJ, Casanova JL, Cooper AM, Cua DJ. IL-12 and IL-23 cytokines: from discovery to targeted therapies for immune-mediated inflammatory diseases. Nat Med. 2015;21:719–729. doi: 10.1038/nm.3895. [DOI] [PubMed] [Google Scholar]
- Thirumangalakudi L, Prakasam A, Zhang R, Bimonte-Nelson H, Sambamurti K, Kindy MS, Bhat NR. High cholesterol-induced neuroinflammation and amyloid precursor protein processing correlate with loss of working memory in mice. J Neurochem. 2008;106:475–485. doi: 10.1111/j.1471-4159.2008.05415.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorleifsson G, Walters GB, Hewitt AW, Masson G, Helgason A, DeWan A, Sigurdsson A, Jonasdottir A, Gudjonsson SA, Magnusson KP, Stefansson H, Lam DSC, Tam POS, Gudmundsdottir GJ, Southgate L, Burdon KP, Gottfredsdottir MS, Aldred MA, Mitchell P, St Clair D, Collier DA, Tang N, Sveinsson O, Macgregor S, Martin NG, Cree AJ, Gibson J, Macleod A, Jacob A, Ennis S, Young TL, Chan JCN, Karwatowski WSS, Hammond CJ, Thordarson K, Zhang M, Wadelius C, Lotery AJ, Trembath RC, Pang CP, Hoh J, Craig JE, Kong A, Mackey DA, Jonasson F, Thorsteinsdottir U, Stefansson K. Common variants near CAV1 and CAV2 are associated with primary open-angle glaucoma. Nat Genet. 2010;42:906–909. doi: 10.1038/ng.661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tielsch JM, Katz J, Sommer A, Quigley HA, Javitt JC. Family history and risk of primary open angle glaucoma. The Baltimore eye survey. Arch Ophthalmol. 1994;112:69–73. doi: 10.1001/archopht.1994.01090130079022. [DOI] [PubMed] [Google Scholar]
- Trachtman H, Fervenza FC, Gipson DS, Heering P, Jayne DR, Peters H, Rota S, Remuzzi G, Rump LC, Sellin LK, Heaton JP, Streisand JB, Hard ML, Ledbetter SR, Vincenti F. A phase 1, single-dose study of fresolimumab, an anti-TGF-beta antibody, in treatment-resistant primary focal segmental glomerulosclerosis. Kidney Int. 2011;79:1236–1243. doi: 10.1038/ki.2011.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai JC, McClure CA, Ramos SE, Schlundt DG, Pichert JW. Compliance barriers in glaucoma: a systematic classification. J Glaucoma. 2003;12:393–398. doi: 10.1097/00061198-200310000-00001. [DOI] [PubMed] [Google Scholar]
- Tseng VL, Lee GY, Shaikh Y, Yu F, Coleman AL. The association between glaucoma and immunoglobulin E antibody response to indoor allergens. Am J Ophthalmol. 2015;159:986–993 e981. doi: 10.1016/j.ajo.2015.02.007. [DOI] [PubMed] [Google Scholar]
- van der Slot AJ, Zuurmond AM, Bardoel AF, Wijmenga C, Pruijs HE, Sillence DO, Brinckmann J, Abraham DJ, Black CM, Verzijl N, DeGroot J, Hanemaaijer R, TeKoppele JM, Huizinga TW, Bank RA. Identification of PLOD2 as telopeptide lysyl hydroxylase, an important enzyme in fibrosis. J Biol Chem. 2003;278:40967–40972. doi: 10.1074/jbc.M307380200. [DOI] [PubMed] [Google Scholar]
- Varma R, Ying-Lai M, Francis BA, Nguyen BB, Deneen J, Wilson MR, Azen SP, Los Angeles Latino Eye Study, G. Prevalence of open-angle glaucoma and ocular hypertension in Latinos: the Los Angeles Latino eye study. Ophthalmology. 2004;111:1439–1448. doi: 10.1016/j.ophtha.2004.01.025. [DOI] [PubMed] [Google Scholar]
- Vasiliou V, Gonzalez FJ. Role of CYP1B1 in glaucoma. Annu Rev Pharmacol Toxicol. 2008;48:333–358. doi: 10.1146/annurev.pharmtox.48.061807.154729. [DOI] [PubMed] [Google Scholar]
- Villarreal G, Jr, Chatterjee A, Oh SS, Oh DJ, Kang MH, Rhee DJ. Canonical wnt signaling regulates extracellular matrix expression in the trabecular meshwork. Invest Ophthalmol Vis Sci. 2014;55:7433–7440. doi: 10.1167/iovs.13-12652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Visel A, Zhu Y, May D, Afzal V, Gong E, Attanasio C, Blow MJ, Cohen JC, Rubin EM, Pennacchio LA. Targeted deletion of the 9p21 non-coding coronary artery disease risk interval in mice. Nature. 2010;464:409–412. doi: 10.1038/nature08801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vithana EN, Aung T, Khor CC, Cornes BK, Tay WT, Sim X, Lavanya R, Wu R, Zheng Y, Hibberd ML, Chia KS, Seielstad M, Goh LK, Saw SM, Tai ES, Wong TY. Collagen-related genes influence the glaucoma risk factor, central corneal thickness. Hum Mol Genet. 2011;20:649–658. doi: 10.1093/hmg/ddq511. [DOI] [PubMed] [Google Scholar]
- Vrabec JP, Levin LA. The neurobiology of cell death in glaucoma. Eye Lond.d) 2007;21(Suppl. 1):S11–S14. doi: 10.1038/sj.eye.6702880. [DOI] [PubMed] [Google Scholar]
- Vranka JA, Kelley MJ, Acott TS, Keller KE. Extracellular matrix in the trabecular meshwork: intraocular pressure regulation and dysregulation in glaucoma. Exp Eye Res. 2015;133:112–125. doi: 10.1016/j.exer.2014.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner AH, Anand VN, Wang WH, Chatterton JE, Sun D, Shepard AR, Jacobson N, Pang IH, Deluca AP, Casavant TL, Scheetz TE, Mullins RF, Braun TA, Clark AF. Exon-level expression profiling of ocular tissues. Exp Eye Res. 2013;111:105–111. doi: 10.1016/j.exer.2013.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wahrle SE, Jiang H, Parsadanian M, Legleiter J, Han X, Fryer JD, Kowalewski T, Holtzman DM. ABCA1 is required for normal central nervous system ApoE levels and for lipidation of astrocyte-secreted apoE. J Biol Chem. 2004;279:40987–40993. doi: 10.1074/jbc.M407963200. [DOI] [PubMed] [Google Scholar]
- Wang H, Syrovets T, Kess D, Buchele B, Hainzl H, Lunov O, Weiss JM, Scharffetter-Kochanek K, Simmet T. Targeting NF-kappa B with a natural triterpenoid alleviates skin inflammation in a mouse model of psoriasis. J Immunol. 2009;183:4755–4763. doi: 10.4049/jimmunol.0900521. [DOI] [PubMed] [Google Scholar]
- Wang Y, Zhou YF, Zhao BY, Gu ZY, Li SL. Apolipoprotein E gene ε4ε4 is associated with elevated risk of primary open angle glaucoma in Asians: a meta-analysis. BMC Med Genet. 2014;15:60. doi: 10.1186/1471-2350-15-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang YE, Kakigi C, Barbosa D, Porco T, Chen R, Wang S, Li Y, Singh K, Pasquale LR, Lin SC. Oral contraceptive use and prevalence of self-reported glaucoma or ocular hypertension in the United States. Ophthalmology. 2016;123:729–736. doi: 10.1016/j.ophtha.2015.11.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webster KM, Wright DK, Sun M, Semple BD, Ozturk E, Stein DG, O’Brien TJ, Shultz SR. Progesterone treatment reduces neuroinflammation, oxida-tive stress and brain damage and improves long-term outcomes in a rat model of repeated mild traumatic brain injury. J Neuroinflamm. 2015;12:238. doi: 10.1186/s12974-015-0457-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei R, Jonakait GM. Neurotrophins and the anti-inflammatory agents interleukin-4 (IL-4), IL-10, IL-11 and transforming growth factor-beta1 (TGF-beta1) down-regulate T cell costimulatory molecules B7 and CD40 on cultured rat microglia. J Neuroimmunol. 1999;95:8–18. doi: 10.1016/s0165-5728(98)00248-3. [DOI] [PubMed] [Google Scholar]
- Wei X, Cai SP, Zhang X, Li X, Chen X, Liu X. Is low dose of estrogen beneficial for prevention of glaucoma? Med Hypotheses. 2012;79:377–380. doi: 10.1016/j.mehy.2012.05.041. [DOI] [PubMed] [Google Scholar]
- Weinreb RN, Aung T, Medeiros FA. The pathophysiology and treatment of glaucoma: a review. JAMA. 2014;311:1901–1911. doi: 10.1001/jama.2014.3192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinreb RN, Khaw PT. Primary open-angle glaucoma. Lancet. 2004;363:1711–1720. doi: 10.1016/S0140-6736(04)16257-0. [DOI] [PubMed] [Google Scholar]
- Weng J, Luo J, Cheng X, Jin C, Zhou X, Qu J, Tu L, Ai D, Li D, Wang J, Martin JF, Amendt BA, Liu M. Deletion of G protein-coupled receptor 48 leads to ocular anterior segment dysgenesis (ASD) through down-regulation of Pitx2. Proc Natl Acad Sci U S A. 2008;105:6081–6086. doi: 10.1073/pnas.0708257105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiederholt M, Thieme H, Stumpff F. The regulation of trabecular meshwork and ciliary muscle contractility. Prog Retin Eye Res. 2000;19:271–295. doi: 10.1016/s1350-9462(99)00015-4. [DOI] [PubMed] [Google Scholar]
- Wiggs JL, Howell GR, Linkroum K, Abdrabou W, Hodges E, Braine CE, Pasquale LR, Hannon GJ, Haines JL, John SWM. Variations in COL15A1 and COL18A1 influence age of onset of primary open angle glaucoma. Clin Genet. 2013;84:167–174. doi: 10.1111/cge.12176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams TM, Lisanti MP. The caveolin proteins. Genome Biol. 2004;5:214. doi: 10.1186/gb-2004-5-3-214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wyszecki G, Stiles WS. Color science. Wiley; N.Y.: 1982. pp. 85–88. [Google Scholar]
- Xue W, Comes N, Borras T. Presence of an established calcification marker in trabecular meshwork tissue of glaucoma donors. Invest Ophthalmol Vis Sci. 2007;48:3184–3194. doi: 10.1167/iovs.06-1403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan X, Sabrautzki S, Horsch M, Fuchs H, Gailus-Durner V, Beckers J, Hrabe de Angelis M, Graw J. Peroxidasin is essential for eye development in the mouse. Hum Mol Genet. 2014;23:5597–5614. doi: 10.1093/hmg/ddu274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Z, Quigley HA, Pease ME, Yang Y, Qian J, Valenta D, Zack DJ. Changes in gene expression in experimental glaucoma and optic nerve transection: the equilibrium between protective and detrimental mechanisms. Invest Ophthalmol Vis Sci. 2007;48:5539–5548. doi: 10.1167/iovs.07-0542. [DOI] [PubMed] [Google Scholar]
- Ying H, Yue BY. Cellular and molecular biology of optineurin. Int Rev Cell Mol Biol. 2012;294:223–258. doi: 10.1016/B978-0-12-394305-7.00005-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young AR, Narita M. SASP reflects senescence. EMBO Rep. 2009;10:228–230. doi: 10.1038/embor.2009.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu AL, Birke K, Moriniere J, Welge-Lussen U. TGF-{beta}2 induces senescence-associated changes in human trabecular meshwork cells. Invest Ophthalmol Vis Sci. 2010;51:5718–5723. doi: 10.1167/iovs.10-5679. [DOI] [PubMed] [Google Scholar]
- Yu W, Gius D, Onyango P, Muldoon-Jacobs K, Karp J, Feinberg AP, Cui H. Epigenetic silencing of tumour suppressor gene p15 by its antisense RNA. Nature. 2008;451:202–206. doi: 10.1038/nature06468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan A, Rao MV, Veeranna, Nixon RA. Neurofilaments at a glance. J Cell Sci. 2012;125:3257–3263. doi: 10.1242/jcs.104729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zakaria S, Gomez TS, Savoy DN, McAdam S, Turner M, Abraham RT, Billadeau DD. Differential regulation of TCR-mediated gene transcription by Vav family members. J Exp Med. 2004;199:429–434. doi: 10.1084/jem.20031228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zanotto-Filho A, Braganhol E, Schroder R, de Souza LH, Dalmolin RJ, Pasquali MA, Gelain DP, Battastini AM, Moreira JC. NFkappaB inhibitors induce cell death in glioblastomas. Biochem Pharmacol. 2011;81:412–424. doi: 10.1016/j.bcp.2010.10.014. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Beyer TP, Bramlett KS, Yao S, Burris TP, Schmidt RJ, Eacho PI, Cao G. Liver X receptor and retinoic X receptor mediated ABCA1 regulation and cholesterol efflux in macrophage cells-messenger RNA measured by branched DNA technology. Mol Genet Metab. 2002;77:150–158. doi: 10.1016/s1096-7192(02)00111-7. [DOI] [PubMed] [Google Scholar]
- Zhang Z, Miao L, Wang L. Inflammation amplification by Versican: the first mediator. Int J Mol Sci. 2012;13:6873–6882. doi: 10.3390/ijms13066873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang ZW, Zhang JP, Zhou TT, Feng WH, Jiao BH. Does the expression of versican isoforms contribute to the pathogenesis of neurodegenerative diseases? Arch Med Res. 2011;42:258–260. doi: 10.1016/j.arcmed.2011.04.010. [DOI] [PubMed] [Google Scholar]
- Zhavoronkov A, Kanherkar RR, Izumchenko E, Teka M, Cantor C, Manaye K, Sidransky D, West MD, Makarev E, Csoka AB. Pro-fibrotic pathway activation in trabecular meshwork and lamina cribrosa is the main driving force of glaucoma. Cell Cycle. 2016;15:1643–1652. doi: 10.1080/15384101.2016.1170261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimering MB, Moritz TE, Donnelly RJ. Anti-neurotrophic effects from autoantibodies in adult diabetes having primary open angle glaucoma or dementia. Front Endocrinol (Lausanne) 2013;4:58. doi: 10.3389/fendo.2013.00058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zode GS, Clark AF, Wordinger RJ. Bone morphogenetic protein 4 inhibits TGF-beta2 stimulation of extracellular matrix proteins in optic nerve head cells: role of gremlin in ECM modulation. Glia. 2009;57:755–766. doi: 10.1002/glia.20803. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


