Abstract
This study aimed to assess the effect of intra-habenular injection of morphine on acute trigeminal pain in rats. Also here, we examined the involvement of raphe nucleus opioid and 5HT3 receptors on the antinociceptive activity of intra habenular morphine to explore the possibility of existence of descending antinociceptive relay between the habenula and raphe nucleus. The numbers of eye wiping response elicited by applying a drop (40 μL) of NaCl (5 M) solution on the corneal surface were taken as an index of acute trigeminal nociception. Intra habenular microinjection of morphine at a dose of 2 μg was without effect, whereas at doses of 5 and 8 μg significantly produced antinociception. Microinjection of naltrexone (4 μg) and ondansetron (1 μg) into the dorsal raphe nucleus prior to intra-habenular saline did not produce any significant effect on corneal pain perception. Pretreatment of the raphe nucleus with ondansetron but not naltrexone prevented intra habenular morphine (8 μg) induced antinociception. Also, intra habenular injection of lidocaine (2%, 0.5 μL reduced corneal pain response. Moreover, intra-habenular microinjection of L-glutamic acid (1 and 2 μg/site) did not produce any analgesic activity in this model of pain. In conclusion, the present results suggest that the activation of the habenular μ opioid receptor by microinjection of morphine or inhibition of habenular neurons by microinjection of lidocaine produced an analgesic effect in the acute trigeminal model of pain in rats. The analgesic effect of intra habenular morphine was blocked by intra-dorsal raphe injection of serotonin 5-HT3 antagonist.
Keywords: Lateral habenula, Morphine, Ondansetrone, Dorsal raphe, Acute trigeminal pain, Rats
INTRODUCTION
The habenular complexes (include the medial and the lateral nuclei) are small bilateral epithalamic structures that modulate a wide range of physiological behaviors involving nociception, depression, stress, endocrine, reward function and addiction (1,2).
A wealth of evidence suggests a key role for the lateral habenula in central modulation of acute and chronic models of pain in the animals and human (3,4). It has been shown that the electrical stimulation of the habenula induces an antinociceptive effect in the formalin test (5).
Microinjection of morphine into the lateral habenula produced analgesia in the formalin test (6). Moreover, it is well established that μ opioid receptors are dense in the habenula but delta and kappa receptors have less density in this region (7). Also, morphine is exhibited inhibition and facilitation of excitatory synaptic transmission in the habenular nucleus neurons of rat (8).
Depending on the intensity of the noxious stimuli (but not non-noxious stimuli) lateral habenular neurons are responsive to peripheral nociceptive stimulation in excitatory or inhibitory patterns (9).
Lateral habenula received pain inputs from direct (via trigeminal nucleus and lamina I of the dorsal horn) and indirect (via structures such as the lateral hypothalamus) afferents (4,10,11). It has been shown that the lateral habenula (LHb) sends its projections to the dorsal and median raphe nuclei, ventral tegmental area, substantia nigra pars compacta and periaqueductal gray (12,13,14). Dorsal raphe nucleus is one of the important nucleus in pain modulation. This nucleus is involved in the pain perception through serotoninergic, opioidergic and GABAergic system (15). Dorsal raphe sends some afferents directly into the spinal trigeminal nucleus and spinal cord to contribute in descending serotoninergic inhibitory pain pathway (15,16).
The connection between habenula and midbrain regions such as the PAG and dorsal raphe seems to be important in the habenular modulation of nociception. Therefore the present study was aimed to investigate the effect of morphine, L-glutamic acid and lidocaine (for pharmacological blockage of the habenula) microinjection into the lateral habenula in the acute trigeminal model of pain. The possible existence of a descending pain modulatory pathway from the lateral habenula to the dorsal raphe, which is may be involved in the intra-habenular morphine induced analgesia was determined by blocking of μ opioid and 5HT3 serotonin receptors in the dorsal raphe by microinjection of opioid receptor antagonist naltrexone and 5HT3 serotonin receptor antagonist ondansetron before intra habenular injection of morphine.
MATERIALS AND METHODS
Animals
Adult male Wistar rats, weighing 250-280 g were used in this study. They were randomly housed in polyethylene cages with ad libitum access to food and water in a room with controlled temperature (22 ± 1 °C) and under a 12 h light–dark cycle (lights on from 07:00 h). Six rats were used in each test group. All experiments were performed between 11:00 h and 15:00 h. All research and animal care procedures were approved by the Veterinary Ethics Committee of the Faculty of Veterinary Medicine (University of Tabriz) (Registration No. D/2013.31) and were performed in accordance with the current guidelines for the care of laboratory animals and the ethical guidelines for investigations of experimental pain in conscious animals (17).
Drugs and chemicals
Morphine sulfate was purchased from TEMAD Co. (Tehran, Iran). Naltrexone hydrochloride and ondansetron hydrochloride were purchased from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA). NaCl and L-glutamic acid were purchased from Merck Chemicals Co. (Darmstadt, Germany). All drugs and chemicals were dissolved in physiological saline, and just only NaCl dissolved in distilled water.
Surgical procedure
The rats were bilaterally implanted with two guide cannulas into the habenular nucleus. In addition, all rats concurrently were implanted with a guide cannula into the dorsal raphe nucleus. Briefly, rats were anesthetized with a mixture of ketamine (80 mg/kg, Alfasan, Woerden, Holland) and xylazine (10 mg/kg, Alfasan, Woerden, Holland) injected intraperitoneally (i.p.), and then placed in a stereotaxic apparatus (Stoelting, Wood Lane, IL, USA). The scalp was incised, and the skull was leveled off around the bregma. A 23-gauge, 15 mm stainless steel guide cannula was introduced at the following coordinates: habenular complex: AP: -3.3, L: 0.7, V: 4.8 and dorsal raphe: AP: -7.8, L: 0, V: 6.4 with respect to bregma at a 0 angle. The cannulas were then fixed to the skull using three screws and dental acrylic (SR Triplex Cold, Ivoclar Vivadent AG, Liechtenstein). A 15-mm stylet was inserted on each cannula to keep them patent prior to injection. At least 10 days was allowed for recovery from the surgery (18).
Injection protocol
For intra-habenular or dorsal raphe microinjections of chemicals a 30 gauge, 15 mm injection needle was attached to a 30 cm PE10 polyethylene tube fitted to a 1 μΕ Hamilton syringe. After stylet removal, the rats were placed on a wooden plate for a period of 15 min for habituation, thereafter the injection needle was inserted into the guide cannula. The volume of the drug solution to be injected into the each site was 0.5 μL and the injection was slowly made over a period of 1 min. The injection needle was left in place for a further 1 min after completion of injection to facilitate diffusion of the drug. Intra-dorsal raphe microinjection of saline, naltrexone (4 μg) and ondansetrone (1 μg) were performed 5 min before intra-habenular injection of chemicals. The possible analgesic activity of morphine (2, 5 and 8 μg/site), l-glutamic acid (1 and 2 μg/site) and lidocaine (2%) were evaluated 5, 15, 30 and 45 min after intra-habenular injection. Morphine doses were chosen from previous studies because these doses effectively prevent from nociception in the different models of pain (19,20). The glutamate, naltrexone and ondansetron doses were chosen based on the previous studies (21,22,23).
Cannula verification
Cannula tip placement determined at the end of the experiment. Briefly at the end of the study, rats euthanized with the high dose of ether and then methylene blue dye (0.5 μL) was injected into each nucleus. The brains were removed and placed in the 10% formalin at least for 7 days. After fixation, the brains were cut coronally, and examined under a stereo-microscope for identification of the injection site to localize in diagrams from the rat brain atlas (Fig. 1) (18). Only data obtained from animals with the correct injection site were contributed in the statistical analysis.
Fig. 1.
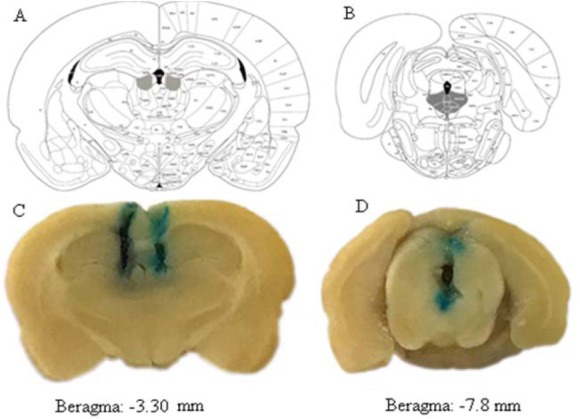
Schematic illustration of coronal sections of the rat brain showing the location of (A) the lateral habenula and (B) dorsal raphe from the atlas of Paxinos and Watson (indicated by gray color). Location of the injection cannulas tip in the lateral (C) habenula and (D) dorsal raphe in this study.
Acute trigeminal test
Each rat was placed on a 50 × 50 × 1 cm wooden table and after a 15 min habituation period, one drop (40 μL) of NaCl 5 M solution was topically applied on the surface of the cornea using a pipette (Transferpette® S 10-100 μL Brand CO, Germany). After topical application of NaCl 5 M solution, rats always wiped with the forepaw and sometimes rapidly scratched the eye with the hind paw. The numbers of eye wipes performed with ipsilateral forepaw were counted for a period of 30 s. Also, each burst of hind paw scratches was counted as one wipe (24,25). The test was performed pre-drug and post-drug administration at the same eye of the same animal with minimum 30 min interval. The effect of drugs from the maximal possible effect (%MPE) was calculated for eye-wipes according to the following formula: %MPE = 100× post drug wipes count - pre drug wipes count / (0 - pre drug wipes count)
Statistical analysis
Statistical differences were determined by two-way analysis of variance (ANOVA) with repeated measures, followed by Bonferroni post hoc test using GraphPad Prism® software version 5 (GraphPad Prism Software, Inc., USA). In figures, all values are expressed as Mean ± SEM. A value of P < 0.05 was considered statistically significant.
RESULTS
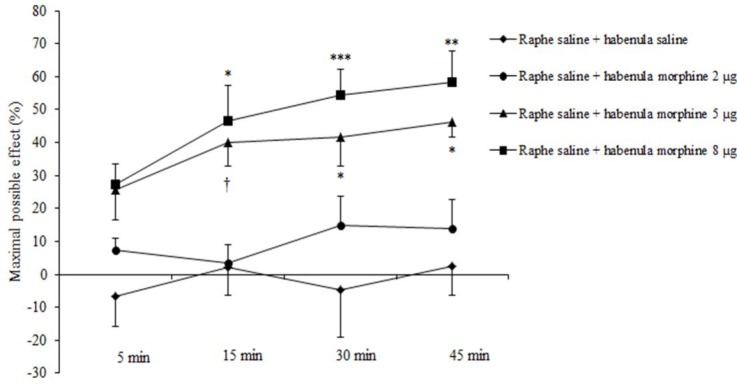
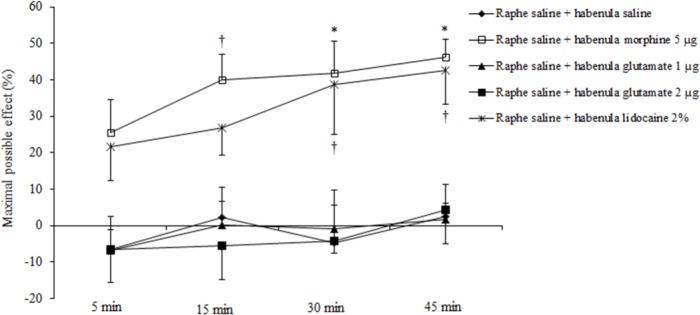
Morphine at a dose of 2 μg was without effect, whereas at a dose of 5 μg significantly produced an antinociceptive effect 15 min (39.93 ± 7.16%, P < 0.05), 30 min (41.78 ± 8.90%, P < 0.01), and 45 min (46.35 ± 4.81%, P < 0.01) after intra-habenular injection. Also, morphine at a dose of 8 μg/site, significantly produced an antinociceptive effect 15 min (46.42 ± 10.74%, P < 0.01), 30 min (54.30 ± 7.85%, P < 0.0001), and 45 min (58.42 ± 9.44%, P < 0.001) after intra-habenular injection (treatment effect: F (3, 80) = 29.35, P < 0.0001; time effect: F (3, 80) = 2.80, P = 0. 0455; treatment and time interaction effect: F (9, 80) = 0.55, P = 0.8352; Fig. 2). Lidocaine (2%, 0.5 μL) significantly reduced corneal pain response in 30 min (38.66 ± 13.52%, P < 0.05) and 45 min (42.66 ± 9.23%, P < 0.05) after intra-habenular injection. Moreover, intra-habenular microinjection of L-glutamic acid (1 and 2 μg/site) did not produce any analgesic activity in this model of pain (treatment effect: F (3, 76) = 15.33, P < 0.0001; time effect: F (3, 76) = 1.33, P = 0. 2707; treatment and time interaction effect: F (9, 76) = 0.23, P = 0.9887; Fig. 3).
Fig. 2.
Effect of intra-habenular microinjection of morphine after intra-dorsal raphe microinjection of saline (0.5 μL/site) on corneal pain response induced by NaCl 5 M solution applied to the corneal surface in rats. Maximal possible effect (MPE%) is considered as an index for comparison between the results of two tests (for calculation of % MPE see the text). The values are expressed as mean ± SEM (n = 6 per group). The data are compared with two-way analysis of variance (ANOVA) followed by bonferroni post hoc test; ***P < 0.0001, **P < 0.001, *P < 0.01 and †P < 0.05 as compared to normal saline treated group. Raphe, dorsal raphe; habenula, lateral habenula.
Fig. 3.
Effect of intra-habenular microinjection of l-glutamic acid and lidocaine after intra-dorsal raphe microinjection of saline (0.5 μL/site) on corneal pain response induced by NaCl 5 M solution applied to the corneal surface in rats. Maximal possible effect (MPE%) is considered as an index for comparison between the results of two tests (for calculation of % MPE see the text). The values are expressed as mean ± SEM (n = 6 per group). The data are compared with two-way analysis of variance (ANOVA) followed by bonferroni post hoc test; *P < 0.01 and †P < 0.05 as compared to normal saline treated group. Raphe, dorsal raphe; habenula, lateral habenula; glutamate, L-glutamic acid.
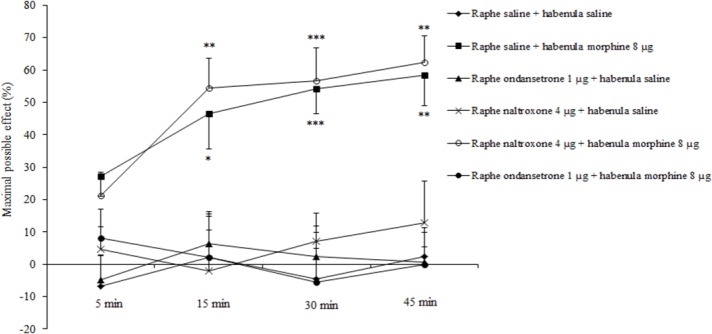
Microinjection of naltrexone (4 μg) and ondansetron (1 μg) into the dorsal raphe nucleus prior to intra-habenular saline did not produce any significant effect on corneal pain perception. Intra-dorsal raphe injection of ondansetron (1 μg) prior to intra-habenular morphine (8 μg) completely block morphine induced analgesia in the acute corneal pain response.
Intra-dorsal raphe injection of naltrexone (4 μg) prior to intra-habenular morphine (8 μg) did not alter morphine induced analgesia 15 min (54.34 ± 9.35%, P < 0.001), 30 min (56.70 ± 10.07%, P < 0.0001), and 45 min (62.30 ± 8.34%, P < 0.001) after intra- habenular injection of morphine (treatment effect: F (4, 100) = 17.43, P < 0.0001; time effect: F (3, 100) = 1.06, P = 0. 3711; treatment and time interaction effect: F (12, 100) = 0.84, P = 0.6109; Fig. 4).
Fig. 4.
Effect of intra-dorsal raphe microinjection of naltrexone and ondansetron before intra-habenular injection of saline (0.5 μL/site) and morphine on corneal pain response induced by NaCl 5 M solution applied to the corneal surface in rats. The values are expressed as mean ± SEM (n = 6 per group). The data are compared with two-way analysis of variance (ANOVA) followed by bonferroni post hoc test; ***P < 0.0001, **P < 0.001 and *P < 0.01 as compared to normal saline treated group. Raphe, dorsal raphe; habenula, lateral habenula.
DISCUSSION
In the present study intra-habenular injection of l-glutamic acid did not alter eye wiping responses, but microinjection of morphine and lidocaine into the habenular complex produced significant analgesia in acute trigeminal model of pain in rats. More importantly, analgesia produced by intra habenular injection of morphine, blocked by intra dorsal raphe injection of ondansetron but μ-opioid receptor antagonist naltrexone did not prevent the analgesic effect of intra-habenular morphine. These results probably indicate that activation of 5-HT3 serotonin receptor in the dorsal raphe nucleus following morphine microinjection into the habenula, could cause analgesia in the acute trigeminal model of pain in rats. Habenula receives direct pain signals from trigeminal nucleus and lamina I of the dorsal horn and indirectly receive signals from other brain structures that contribute in pain processing like lateral hypothalamus, limbic forebrain structures and nucleus accumbens (4,10,11). Projections from habenula are terminated on the some well-known structures that involved in pain processing like periaqueductal gray and dorsal raphe nuclei (4,12). These anatomical evidences are supported by some other electrophysiological (9), behavioral (6) and immunohistological (26) evidences that confirming pain processing role of the habenular complex in the brain. Moreover, habenula is one of the few regions in the brain that play an important role in the regulation of both dopamine and serotonin systems (2).
It has been reported that stimulation of lateral habenula can modulate dorsal raphe neuronal activity (27). Lidocaine injection into the habenular complex or its lesion could improve depressive signs via increasing of serotonin level in dorsal raphe nucleus of depressed rats (28). Also, the increase in serotonin release is reported in the striatum after electrical stimulation of lateral habenula and this effect blocked by microinjection of the non-selective N-methyl-D-aspartate receptor coupled glycine B receptor NMDA/GLYB receptor antagonist kynurenic acid into the dorsal raphe nucleus (29,30). Furthermore, extracellular level of serotonin in the dorsal raphe significantly decreased following intra-habenular microinjection of l- glutamic acid in normal rats (28). On the other hand, in the behavioral study, it was observed that activation of the lateral habenula by microinjection of l-glutamic acid decreased tail flick latency in rats (31). However, in this study, we found that microinjection of L- glutamic acid into the habenular complex did not produce any effect on hypertonic saline induced eye wiping response.
Analgesia produced by microinjection of morphine into the habenular complex has been reported previously in the formalin and the tail flick test (6,32). Habenula is one of the brain sites with high levels of μ opioid receptor expression (7). Microinjection of morphine into the lateral habenula induced both inhibition and facilitation of glutamatergic synaptic transmission (8). There are some reports indicate that habenulo-raphe projection is excitatory and synapses on GABA interneurons, which in turn inhibit serotonergic projections and reduced serotonin level, particularly in the dorsal raphe (33,34,35). Our results suggest that inhibition, but not activation of habenular complex by morphine (like lidocaine effect) may lead to decrease of glutamate release from habenulo-raphe excitatory projection thus decrease GABAergic interneurons activity and subsequently increase serotonin release from dorsal raphe serotonin-containing neurons.
It is well established that serotonin is one of the key neurotransmitters in the descending pain inhibitory pathways (36) that also involved in morphine-induced antinociception (37). Serotonin 5-HT3 receptors have both proand antinociceptive role depending on their expression site (peripheral or central). The existence of 5-HT3 receptors in dorsal raphe has been demonstrated by Laporte, et al. (38). Serotonin 5-HT3 receptors have an undeniable role in the antinociceptive activity of serotonin in the central nervous system (39,40,41). The analgesic effect of intrathecal injection of serotonin and 1-(m-chlorophenyl)-bi-guanide (5-HT3 receptor agonist) inhibited by intrathecal pretreatment of rats with tropisetron and granisetron (5-HT3 receptor antagonists) (41).
CONCLUSION
In conclusion, the present results suggest that the activation of the habenular μ opioid receptor by microinjection of morphine or inhibition of habenular neurons by microinjection of lidocaine produced an analgesic effect in the acute trigeminal model of pain in rats. The analgesic effect of intra habenular morphine was blocked by intradorsal raphe injection of serotonin 5-HT3 antagonist.
ACKNOWLEDGMENTS
This paper is published as part of a research project supported by the University of Tabriz Research Affairs Office.
REFERENCES
- 1.Amat J, Sparks PD, Matus-Amat P, Griggs J, Watkins LR, Maier SF. The role of the habenular complex in the elevation of dorsal raphe nucleus serotonin and the changes in the behavioral responses produced by uncontrollable stress. Brain Res. 2001;917(1):118–126. doi: 10.1016/s0006-8993(01)02934-1. [DOI] [PubMed] [Google Scholar]
- 2.Hikosaka O. The habenula: from stress evasion to value-based decisionmaking. Nat Rev Neurosci. 2010;11(7):503–513. doi: 10.1038/nrn2866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dafny N, Dong WQ, Prieto-Gomez C, Reyes- Vazquez C, Stanford J, Qiao JT. Lateral hypothalamus: site involved in pain modulation. Neuroscience. 1996;70(2):449–460. doi: 10.1016/0306-4522(95)00358-4. [DOI] [PubMed] [Google Scholar]
- 4.Shelton L, Becerra L, Borsook D. Unmasking the mysteries of the habenula in pain and analgesia. Prog Neurobiol. 2012;96(2):208–219. doi: 10.1016/j.pneurobio.2012.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cohen SR, Melzack R. Habenular stimulation produces analgesia in the formalin test. Neurosci Lett. 1986;70(1):165–169. doi: 10.1016/0304-3940(86)90457-x. [DOI] [PubMed] [Google Scholar]
- 6.Cohen SR, Melzack R. Morphine injected into the habenula and dorsal posteromedial thalamus produces analgesia in the formalin test. Brain Res. 1985;359(1-2):131–139. doi: 10.1016/0006-8993(85)91420-9. [DOI] [PubMed] [Google Scholar]
- 7.Mansour A, Khachaturian H, Lewis ME, Akil H, Watson SJ. Autoradiographic differentiation of mu, delta, and kappa opioid receptors in the rat forebrain and midbrain. J Neurosci. 1987;7(8):2445–2464. [PMC free article] [PubMed] [Google Scholar]
- 8.Hashimoto K, Amano T, Sakai N, Suzuki T, Narita M. Cell-dependent physiological synaptic action of morphine in the rat habenular nucleus: morphine both inhibits and facilitates excitatory synaptic transmission. Neurosci Lett. 2009;451(3):270–273. doi: 10.1016/j.neulet.2009.01.009. [DOI] [PubMed] [Google Scholar]
- 9.Gao DM, Hoffman D, Benabid AL. Simultaneous recording of spontaneous activities and nociceptive responses from neurons in the pars compacta of substantia nigra and in the lateral habenula. Eur J Neurosci. 1996;8(7):1474–1478. doi: 10.1111/j.1460-9568.1996.tb01609.x. [DOI] [PubMed] [Google Scholar]
- 10.Goto M, Canteras NS, Burns G, Swanson LW. Projections from the subfornical region of the lateral hypothalamic area. J Comp Neurol. 2005;493(3):412–438. doi: 10.1002/cne.20764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Craig AD. Distribution of trigeminothalamic and spinothalamic lamina I terminations in the cat. Somatosens Mot Res. 2003;20(3-4):209–222. doi: 10.1080/08990220310001623013. [DOI] [PubMed] [Google Scholar]
- 12.Ferraro G, Montalbano ME, Sardo P, La Grutta V. Lateral habenular influence on dorsal raphe neurons. Brain Res Bull. 1996;41(1):47–52. doi: 10.1016/0361-9230(96)00170-0. [DOI] [PubMed] [Google Scholar]
- 13.Alenina N, Bashammakh S, Bader M. Specification and differentiation of serotonergic neurons. Stem Cell Rev. 2006;2(1):5–10. doi: 10.1007/s12015-006-0002-2. [DOI] [PubMed] [Google Scholar]
- 14.Bianco IH, Wilson SW. The habenular nuclei: a conserved asymmetric relay station in the vertebrate brain. Philos Trans R Soc Lond B Biol Sci. 2009;364(1519):1005–1020. doi: 10.1098/rstb.2008.0213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wang QP, Nakai Y. The dorsal raphe: an important nucleus in pain modulation. Brain Res Bull. 1994;34(6):575–585. doi: 10.1016/0361-9230(94)90143-0. [DOI] [PubMed] [Google Scholar]
- 16.Beitz AJ, Clements JR, Ecklund LJ, Mullett MM. The nuclei of origin of brainstem enkephalin and cholecystokinin projections to the spinal trigeminal nucleus of the rat. Neuroscience. 1987;20(2):409–425. doi: 10.1016/0306-4522(87)90101-1. [DOI] [PubMed] [Google Scholar]
- 17.Zimmermann M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain. 1983;16(2):109–110. doi: 10.1016/0304-3959(83)90201-4. [DOI] [PubMed] [Google Scholar]
- 18.Paxinos G, Watson C. The rat brain in stereotaxic coordinates. 6th ed. New York, USA: Elsevier; 2007. pp. 61–110. [Google Scholar]
- 19.Erfanparast A, Tamaddonfard E, Farshid AA, Khalilzadeh E. Antinociceptive effect of morphine microinjections into the dorsal hippocampus in the formalin-induced orofacial pain in rats. Veterinary Research Forum. 2010;1:83–89. [Google Scholar]
- 20.Haghparast A, Ahmad-Molaei L, Alizadeh A, Azizi P. Blockade of opioid receptors located in the rat nucleus cuneiformis reduced the antinociceptive responses of local but not systemic administration of morphine in formalin test. Basic and Clinical Nurosciense. 2010;2:13–19. [Google Scholar]
- 21.Miranda MI, Ferreira G, Ramírez-Lugo L, Bermúdez-Rattoni F. Glutamatergic activity in the amygdala signals visceral input during taste memory formation. Proc Natl Acad Sci USA. 2002;99(17):11417–11422. doi: 10.1073/pnas.182200499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Helmstetter FJ, Landeira-Fernandez J. Conditional hypoalgesia is attenuated by Naltrexone applied to the periaqueductal gray. Brain Res. 1999;537(1- 2):88–92. doi: 10.1016/0006-8993(90)90343-a. [DOI] [PubMed] [Google Scholar]
- 23.Ohmura Y, Yoshida T, Konno K, Minami M, Watanabe M, Yoshioka M. Serotonin 5-HT7 receptor in the ventral hippocampus modulates the retrieval of fear memory and stress-induced defecation. Int J Neuropsychopharmacol. 2015;19(6):1–12. doi: 10.1093/ijnp/pyv131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Farazifard R, Safapour F, Sheibani V, Javan M. Eye wiping test: a sensitive animal model for acute trigeminal pain studies. Brain Res Brain Res Protoc. 2005;16(1-3):44–49. doi: 10.1016/j.brainresprot.2005.10.003. [DOI] [PubMed] [Google Scholar]
- 25.Khalilzadeh E, Hazrati R, Vafaei Saiah G. Effects of topical and systemic administration of Eugenia caryophyllata buds essential oil on corneal anesthesia and analgesia. Res Pharm Sci. 2016;11(4):293–302. doi: 10.4103/1735-5362.189297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lehner M, Taracha E, Skorzewska A, Wislowska A, Zienowicz M, Maciejak P, et al. Sensitivity to pain and c-Fos expression in brain structures in rats. Neurosci Lett. 2004;370(1):74–79. doi: 10.1016/j.neulet.2004.07.089. [DOI] [PubMed] [Google Scholar]
- 27.Wang RY, Aghajanian GK. Physiological evidence for habenula as major link between forebrain and midbrain raphe. Science. 1977;197(4298):89–91. doi: 10.1126/science.194312. [DOI] [PubMed] [Google Scholar]
- 28.Yang LM, Hu B, Xia YH, Zhang BL, Zhao H. Lateral habenula lesions improve the behavioral response in depressed rats via increasing the serotonin level in dorsal raphe nucleus. Behav Brain Res. 2008;188(1):84–90. doi: 10.1016/j.bbr.2007.10.022. [DOI] [PubMed] [Google Scholar]
- 29.Kalen P, Strecker RE, Rosengren E, Bjorklund A. Regulation of striatal serotonin release by the lateral habenula-dorsal raphe pathway in the rat as demonstrated by in vivo microdialysis: role of excitatory amino acids and GABA. Brain Res. 1989;492(1-2):187–202. doi: 10.1016/0006-8993(89)90901-3. [DOI] [PubMed] [Google Scholar]
- 30.Waselus M, Galvez JP, Valentino RJ, Van Bockstaele E J. Differential projections of dorsal raphe nucleus neurons to the lateral septum and striatum. J Chem Neuroanat. 2006;31(4):233–242. doi: 10.1016/j.jchemneu.2006.01.007. [DOI] [PubMed] [Google Scholar]
- 31.Zhao H, Wang S. Different effects of l-glutamate microinjection into medial or lateral habenular nucleus on pain threshold. Sheng Li Xue Bao. 1995;47(3):292–296. [PubMed] [Google Scholar]
- 32.Cohen SR, Melzack R. The habenula and pain: repeated electrical stimulation produces prolonged analgesia but lesions have no effect on formalin pain or morphine analgesia. Behav Brain Res. 1993;54(2):171–178. doi: 10.1016/0166-4328(93)90076-3. [DOI] [PubMed] [Google Scholar]
- 33.Ferraro G, Montalbano ME, Sardo P, La Grutta V. Lateral habenula and hippocampus: a complex interaction raphe cells mediated. J Neural Transm. 1997;104(6-7):615–631. doi: 10.1007/BF01291880. [DOI] [PubMed] [Google Scholar]
- 34.Nishikawa T, Scatton B. Inhibitory influence of GABA on central serotonergic transmission. Involvement of the habenulo-raphe pathways in the GABAergic inhibition of ascending cerebral serotonergic neurons. Brain Res. 1985;331:81–90. doi: 10.1016/0006-8993(85)90717-6. [DOI] [PubMed] [Google Scholar]
- 35.Sabatino M, Ferraro G, La Grutta V. Relay stations and neurotransmitters between the palidal region and the hippocampus. Electroencephalogr Clin Neurophysiol. 1991;78(4):302–310. doi: 10.1016/0013-4694(91)90185-7. [DOI] [PubMed] [Google Scholar]
- 36.Millan MJ. The role of descending noradrenergic and serotoninergic pathways in the modulation of nociception: focus on receptor multiplicity, Handbook of experimental pharmacology. In: Dickenson AH, Besson JM, editors. The pharmacology of pain. Vol. 130. Heidelberg: Springer; 1997. pp. 385–446. [Google Scholar]
- 37.Wigdor S, Wilcox GL. Central and systemic morphine-induced antinociception in mice: contribution of descending serotonergic and noradrenergic pathways. J Pharmacol Exp Ther. 1987;242(1):90–95. [PubMed] [Google Scholar]
- 38.Laporte AM, Koscielniak T, Ponchant M, Vergé D, Hamon M, Gozlan H. Quantitative autoradiographic mapping of 5-HT3 receptors in the rat CNS using [125I]iodo-zacopride and [3H]zacopride as radioligands. Synapse. 1992;10(4):271–281. doi: 10.1002/syn.890100402. [DOI] [PubMed] [Google Scholar]
- 39.Glaum SR, Proudfit HK, Anderson EG. Reversal of the antinociceptive effects of intrathecally administered serotonin in the rat by a selective 5HT3 receptor antagonist. Neurosci Lett. 1988;95(1-3):313–317. doi: 10.1016/0304-3940(88)90677-5. [DOI] [PubMed] [Google Scholar]
- 40.Glaum SR, Proudfit HK, Anderson EG. 5HT3 receptors modulate spinal nociceptive reflexes. Brain Res. 1990;510(1):12–16. doi: 10.1016/0006-8993(90)90721-m. [DOI] [PubMed] [Google Scholar]
- 41.Bardin L, Jourdan D, Alloui A, Lavarenne J, Eschalier A. Differential influence of two serotonin 5-HT3 receptor antagonists on spinal serotonin- induced analgesia in rats. Brain Res. 1997;765(2):267–272. doi: 10.1016/s0006-8993(97)00566-0. [DOI] [PubMed] [Google Scholar]