Abstract
Posterior reversible encephalopathy syndrome has been associated with hypertension, preeclampsia, cancer chemotherapy, and drugs of abuse, such as amphetamine and methamphetamine. We report a young man who suddenly developed severe headache, disorientation, and aphasia following ingestion of kratom and Adderall. Computed tomography and magnetic resonance imaging of his head revealed foci of vasogenic edema in the posterior occipital lobes, frontal lobes, and brainstem. In addition, he had a small area of hemorrhage in the left posterior occipital lobe. Lumbar puncture revealed an increased number of red blood cells but no other abnormalities. His initial blood pressure was elevated but returned to normal during hospitalization. This case suggests that kratom can cause posterior reversible encephalopathy syndrome and needs to be considered when patients present to emergency centers with headaches, confusion, and visual disturbances.
Mitragyna speciosa (kratom) is a tropical evergreen tree native to Southeast Asia. It is a member of the Rubiaceae family (Ulbricht) and contains the psychoactive compounds mitragynine (C23H30N2O4, molecular weight 398.49) and 7-hydoxymitragynine (7-HMG) (Figure 1) (1). Kratom leaves have traditionally been used in Southeast Asia to treat chronic pain or opioid withdrawal, although no scientific evidence supports this use. Over the past years, kratom plant use has increased in the United States due to its psychoactive and opioid-like activity, and the leaves are used for recreation and for self-medicated opioid withdrawal treatment. We report a patient with posterior reversible leukoencephalopathy syndrome (PRES) possibly related to kratom.
Figure 1.
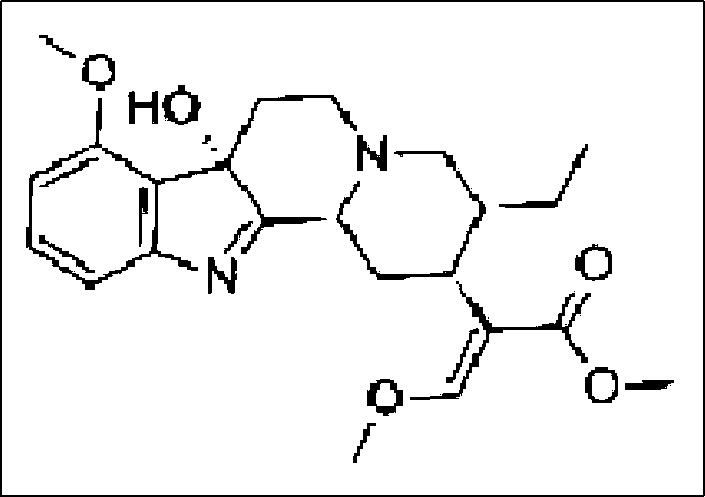
7-hydroxymitragynine (7-HMG).
CASE PRESENTATION
A 22-year-old man presented with a severe headache (“absolute worse pounding”) that had started earlier that morning. His headache was described as throbbing (10/10), mostly in the back of his head, and unrelieved by acetaminophen. He also reported mild confusion, dizziness, and disorientation. The patient was prescribed fluoxetine for depression and quetiapine for insomnia. He occasionally misused dextroamphetamine and marijuana. His blood pressure was 180/105 mm Hg, heart rate 53 beats per minute, and respiratory rate 16 breaths per minute. He was alert and oriented but uncomfortable. His Glasgow coma scale score was 15. His white blood cell count was 12.8 k/µL, hemoglobin 17.1 g/dL, and platelet count 247 k/µL. The serum sodium was 136 mEq/L, potassium 3.6 mEq/L, chloride 101 mEq/L, blood urea nitrogen 12 mg/dL, and creatinine 0.9 mg/dL. The prothrombin time was 11.6 sec. A computed tomography (CT) scan of the head revealed a possible occipital intraparenchymal hemorrhage. A lumbar puncture was performed, and the cerebrospinal fluid had 1500 red blood cells/mm3, 0 white blood cells/mm3, glucose 65 mg/dL, protein 21 mg/dL, and a negative Gram stain.
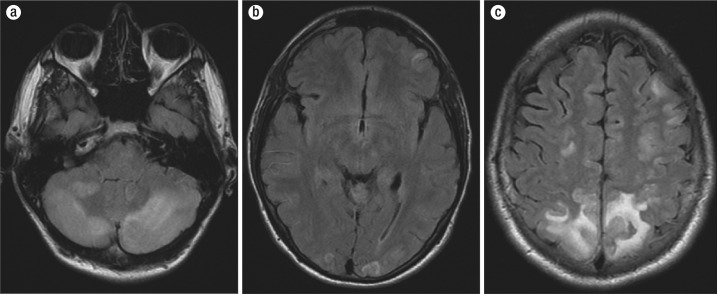
The patient was then transferred to our facility; during his initial interview he was confused, disoriented to place and time, and aphasic. He had photophobia. His systolic pressure varied from 160 mm Hg to 100 mm Hg. His pupils were equal, round, and reactive to light. Motor examination and coordination were normal. His Glasgow coma scale score was 14. His drug screen was positive for amphetamine, benzodiazepine (lorazepam given in local emergency room), cannabinoids, and opiates (morphine given in local emergency room). His urine was not tested for kratom. A repeat CT scan of the head revealed bilateral low-density changes consistent with edema in both cerebellar hemispheres, in both occipital lobes, and in both parietal lobes consistent with PRES and a left occipitoparietal intraparenchymal bleed (Figure 2). The patient was started on nicardipine. Magnetic resonance imaging (MRI) of his head with gadolinium contrast showed multifocal areas of abnormal T2 fluid-attenuated inversion recovery signal; gyriform enhancement; restricted diffusion in the superior parietal lobes, both occipital lobes, and both cerebellar hemispheres; and minimal hemorrhage in the left superior parietal lobe consistent with atypical PRES (Figure 3). The patient eventually reported that prior to admission he had abused kratom (amount unknown) and had taken six tablets of dextroamphetamine; he was unwilling to provide more detail about the use of these drugs. He was hospitalized for 5 days. His headache steadily improved, and a CT scan on the day before discharge was unchanged. On the day of discharge, his blood pressure was within normal limits without treatment, and his neurological examination was within normal limits.
Figure 2.
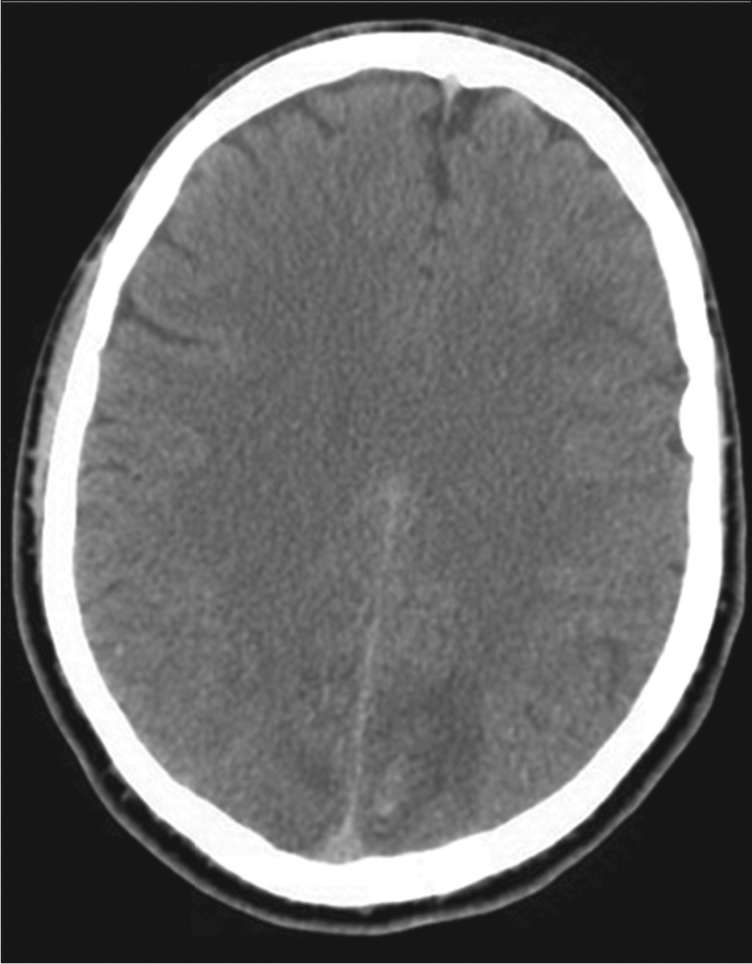
A CT scan of the head reveals low attenuation areas in both occipital lobes, more so on the left, and punctate hemorrhages in the left occipital lobe. This result is consistent with vasogenic edema.
Figure 3.
MRI T2 fluid-attenuated inversion recovery images of (a) the cerebellum, (b) occipital regions, and (c) frontoparietal lobes.
DISCUSSION
Although kratom has been used as an herbal remedy for many years in Southeast Asia, published investigations of it have been limited to case reports and small trials. Kratom's toxic effects include dry mouth, changes in urination, nausea, vomiting, and weight loss. Serious complications, such as seizures and coma, have been reported. The effects of kratom are dose dependent. Small doses produce cocaine-like effects; larger doses cause morphine-like sedative effects. Kratom users report increased work capacity, alertness, sociability, and sometimes increased sexual desire (2). Mitragynine and 7-hydroxymitragynine are the two alkaloids responsible for the effects of kratom and act as full agonists at µ-subtype opioid receptors (Figure 1). Pharmacological activities are mainly mediated via opioid receptors and neuronal Ca++ channels with a dominant effect at neuromuscular junctions and not in skeletal muscles or somatic nerves (2). In one small trial, pharmacokinetics demonstrated a terminal half-life of about 1 day (3).
PRES is a distinct clinical syndrome with characteristic radiographic abnormalities on CT and MRI. It has developed in patients with hypertensive encephalopathy, eclampsia, and disorders treated with cytotoxic or immunosuppressive drugs. The initial clinical series reported in 1996 included 13 women and 2 men with ages ranging from 15 to 62 years (4). Twelve of these patients had hypertension (systolic blood pressure range 142 to 200 mm Hg), eight had elevated creatinines (range 1.5 to 16.1 mg/dL), three were taking cyclosporine, and three were taking tacrolimus. CT and MRI showed extensive white matter changes consistent with edema in the posterior regions of both cerebral hemispheres. Although the pathogenesis of PRES is not fully understood, abnormal cerebral blood flow regulation likely contributes to its development (5). As the arterial blood pressure rises, this pressure change could cause blood-brain barrier disruption and extravasation of fluid into the brain parenchyma, producing vasogenic edema. PRES has been reported after intrathecal morphine (6). The interaction of kratom with µ-subtype opioid receptors could cause similar effects leading to PRES. Reversible cerebral vasoconstriction syndrome presents with sudden-onset “thunder clap headache” due to vasospasm of cerebral arteries. The diagnosis is clinical and supported with cerebral angiography, which was not performed in this patient.
Our patient also ingested dextroamphetamine. Dextroamphetamine increases dopamine release into the presynaptic neuron and increases the release of these monoamines into the extraneuronal space. These effects can increase blood pressure (4). The patient's blood pressure was elevated upon presentation to his local emergency department, and this likely contributed to the development of his PRES (5, 7). It is possible that kratom interacted with amphetamines to raise his blood pressure or caused direct neurotoxicity and produced PRES. Many individuals, including this patient, reporting adverse effects associated with kratom have also used other drugs (8). Consequently, the pathogenesis of adverse effects and toxicity likely involves individual drug effects, synergistic drug effects, and possible adulterants in unregulated products like kratom. Fluoxetine and quetiapine have not been associated with PRES.
Three to five million Americans use kratom, with estimated sales figures >$1 billion per year. Anwar et al reported that US Poison Control Centers received 660 calls about exposure to kratom between January 2010 and December 2015 (8). Many of these individuals had taken kratom with other substances, such as ethanol, benzodiazepines, narcotics, and acetaminophen. Users were mostly males (71.7%) with a median age of 28 years (range 2 months to 69 years). Symptoms and signs included tachycardia (25.0%), agitation or irritability (23.8%), drowsiness (19.4%), nausea (14.7%), and hypertension (11.7%). Kratom use has been associated with seizures, coma, and several deaths (9). The Drug Enforcement Administration opened an official public comment period which ended on December 1, 2016, for people to share their experiences using kratom as a medical treatment and has asked the US Food and Drug Administration to speed up scientific research.
References
- 1.Warner ML, Kaufman NC, Grundmann O. The pharmacology and toxicology of kratom: from traditional herb to drug of abuse. Int J Legal Med. 2016;130(1):127–138. doi: 10.1007/s00414-015-1279-y. [DOI] [PubMed] [Google Scholar]
- 2.Suhaimi FW, Yusoff NH, Hassan R, Mansor SM, Navaratnam V, Muller CP, Hassan Z. Neurobiology of kratom and its main alkaloid mitragynine. Brain Res Bull. 2016;126(Pt 1):29–40. doi: 10.1016/j.brainresbull.2016.03.015. [DOI] [PubMed] [Google Scholar]
- 3.Trakulsrichai S, Sathirakul K, Auparakkitanon S, Krongvorakul J, Sueajai J, Sukasem C, Wananukul W. Pharmacokinetics of mitragynine in man. Drug Des Devel Ther. 2015;9:2421–2429. doi: 10.2147/DDDT.S79658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Briars L, Todd T. A review of pharmacological management of attention-deficit/hyperactivity disorder. J Pediatr Pharmacol Ther. 2016;21(3):192–206. doi: 10.5863/1551-6776-21.3.192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hinchey J, Chaves C, Appignani B, Breen J, Pao L, Wang A, Pessin MS, Lamy C, Mas JL, Caplan LR. A reversible posterior leukoencephalopathy syndrome. N Engl J Med. 1996;334(8):494–500. doi: 10.1056/NEJM199602223340803. [DOI] [PubMed] [Google Scholar]
- 6.Eran A, Barak M. Posterior reversible encephalopathy syndrome after combined general and spinal anesthesia with intrathecal morphine. Anesth Analg. 2009;108(2):609–612. doi: 10.1213/ane.0b013e31818f635e. [DOI] [PubMed] [Google Scholar]
- 7.Schwartz RB, Jones KM, Kalina P, Bajakian RL, Mantello MT, Garada B, Holman BL. Hypertensive encephalopathy: findings on CT, MR imaging, and SPECT imaging in 14 cases. AJR Am J Roentgenol. 1992;159(2):379–383. doi: 10.2214/ajr.159.2.1632361. [DOI] [PubMed] [Google Scholar]
- 8.Anwar M, Law R, Schier J. Notes from the field: Kratom (Mitragyna speciosa) exposures reported to poison centers—United States, 2010–2015. MMWR Morb Mortal Wkly Rep. 2016;65(29):748–749. doi: 10.15585/mmwr.mm6529a4. [DOI] [PubMed] [Google Scholar]
- 9.Nelsen JL, Lapoint J, Hodgman MJ, Aldous KM. Seizure and coma following kratom (Mitragynina speciosa Korth) exposure. J Med Toxicol. 2010;6(4):424–426. doi: 10.1007/s13181-010-0079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]