Abstract
Gnotobiotic piglets inoculated with Escherichia coli O157:H7, its luxS mutant derivative, or nonpathogenic E. coli were evaluated for attaching and effacing lesions. Although no differences in clinical symptoms were seen between pigs inoculated with the parent and those inoculated with the luxS mutant, the luxS mutant-inoculated pigs had a lower frequency of attaching and effacing lesions in the spiral colon than parent strain-inoculated pigs.
Quorum sensing (QS) is a bacterial cell-to-cell signaling mechanism used by a wide variety of bacterial species for recognition of the surrounding microbial community. The luxS QS system is responsible for production of a signaling compound called autoinducer 2 (AI-2) that can control gene expression in a wide variety of gram-positive and gram-negative species (10). In Escherichia coli O157:H7, the LuxS enzyme contributes to the production of another signaling compound, AI-3, which is involved in the QS regulation of some enterohemorrhagic E. coli (EHEC) virulence genes (11, 13). The AI-3 signaling compound is also produced by the human resident intestinal flora and hypothesized to be involved in signaling to EHEC that it has reached the large intestine of the host (4, 13). This QS signaling cascade activates transcription of the genes within the locus of enterocyte effacement (LEE) pathogenicity island, which encode a type III secretion system, bacterial effectors, and the bacterial adhesin intimin, essential for formation of the attaching and effacing (A/E) lesions in enterocytes (6, 11, 13). A luxS mutation in EHEC results in decreased transcription of the LEE region genes and a deficiency in type III secretion in broth cultures (11). Due to the influence of LuxS on the synthesis of AI-3, which modulates EHEC colonization factors, it is hypothesized that LuxS facilitates intestinal colonization of gnotobiotic pigs by E. coli O157:H7. The objective of this study was to determine if mutation of the luxS gene would impair the ability of E. coli O157:H7 to colonize gnotobiotic piglets.
Germfree piglets, 24 h old, were orally inoculated with 105 CFU of bacteria. Eleven pigs from four litters were inoculated with the parent strain, a spontaneous streptomycin- and nalidixic acid-resistant derivative of E. coli O157:H7 86-24 (14). Fourteen pigs from five litters were inoculated with strain VS97, a luxS mutant derived from the parent strain by methods described by Sperandio et al. (12). Six pigs from two litters were inoculated with nonpathogenic E. coli strain 123 (NPE 123) (7) to serve as controls.
Strains were grown as overnight cultures in tryptic soy broth with appropriate antibiotics. The parent strain was grown with 50 μg of streptomycin/ml and 10 μg of nalidixic acid/ml, the luxS mutant was grown with 10 μg of tetracycline/ml, and NPE 123 was grown without antibiotics. Inocula were prepared as previously described (1, 2, 9).
Pigs inoculated with the parent strain and VS97 were necropsied when neurologic signs were demonstrated or at 7 days postinoculation (p.i.). Control pigs were necropsied at 4 days p.i., the average time of onset of clinical signs for pigs inoculated with the pathogenic strains. At necropsy, sections of ilea, ceca, spiral colons, cerebrums, cerebellums, and brain stems were collected in neutral buffered formalin for histopathology, processed, and stained with hematoxylin and eosin. A specific horseradish peroxidase immunohistochemical stain with the primary antibody targeting the O157 antigen was used for the intestinal sections (3). Cross-sections from each intestinal segment were examined microscopically in a blind fashion and scored for A/E lesions with intimately associated bacteria as a measure of colonization (5, 8). Multiple sections of spiral colons from five pigs that were inoculated with the parent strain and the luxS mutant were prepared and evaluated by transmission electron microscopy.
Contingency analysis for ordinal data, followed by pairwise testing, was done for the A/E scores. JMP (version 5.0.1a; SAS Institute, Inc.) was the statistical program utilized for analysis; a P value of <0.05 was used as the level of significance for all evaluations.
Pigs from groups inoculated with the parent strain or its luxS mutant derivative showed clinical signs of ataxia, head-pressing, and/or recumbency due to the systemic effects of Shiga toxins (15). Eleven of 14 pigs inoculated with the luxS mutant developed neurologic signs between 3.5 and 7 days p.i. Nine of 11 pigs inoculated with the parent strain developed neurologic signs between 3 and 7 days p.i. No clinical signs were observed in the control pigs necropsied at 4 days p.i.
Upon necropsy, 10 of 14 luxS mutant-inoculated pigs and all 11 parent strain-inoculated pigs had mesocolonic edema. No gross enteric lesions were present in the NPE 123-inoculated control pigs.
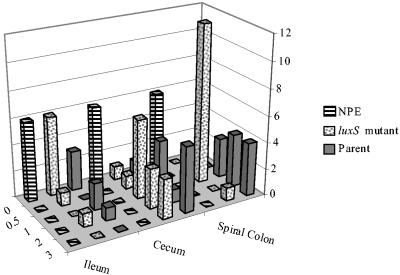
Histologically, the A/E lesions in the ilea, ceca, and spiral colons from the luxS mutant-inoculated pigs were qualitatively similar to lesions from the pigs inoculated with the parent strain. The distributions of the A/E lesion scores are summarized in Fig. 1. The parent strain-inoculated group had significantly higher (P = 0.0025) A/E scores (median, 2; range, 1 to 3) for the spiral colon than did the luxS mutant-inoculated pigs (median, 1; range, 0 to 3). For the spiral colon sections, both the parent strain- and the luxS mutant-inoculated pigs had significantly higher (P < 0.0001) A/E lesion scores than did the NPE 123-inoculated pigs. There was no significant difference (P > 0.5) between the cecal A/E lesion scores for the parent strain (median, 2; range, 0 to 3) and those for the luxS mutant-inoculated pigs (median, 1; range, 0 to 3); these two groups had significantly higher cecal A/E lesion scores than did the NPE 123-inoculated pigs (P < 0.002). There were no A/E lesions in any intestinal segments from the pigs inoculated with NPE 123. There were no significant differences in A/E lesion scores for the ileal sections from pigs inoculated with any of the strains. Classical A/E lesions (8) were identified with electron microscopy in the spiral colons from all the parent strain- and luxS mutant-inoculated pigs examined. Microhemorrhages and necrotic vessels were detected in the brains of pigs inoculated with the parent strain or its luxS derivative; results are shown in Table 1. There was no statistically significant difference between the scores of these two groups of pigs for neurological lesions. Pigs inoculated with NPE 123 had no neurologic lesions.
FIG. 1.
A/E lesion scores in tissues of gnotobiotic piglets inoculated with nonpathogenic E. coli (NPE), E. coli O157:H7 strain VS97 (luxS mutant), or 86-24 (Parent). The A/E lesion scores of the formalin-fixed intestinal sections were as follows: 0, no A/E lesions with discernable associated bacteria visible throughout the sections; 0.5, infrequent A/E lesions identified only with the immunohistochemical stain; 1, discernable lesions on fewer than 10% of the enterocytes of the sections; 2, discernable lesions on more than 10% but fewer than 50% of the enterocytes; 3, lesions present on 50% or more of the enterocytes. Contingency analysis detected statistically significant differences between the scores for the luxS mutant-inoculated tissues and those for the parent-inoculated tissues from the spiral colons (P < 0.03).
TABLE 1.
Clinical and microscopic findings for gnotobiotic piglets inoculated with E. coli O157:H7 strain 86-24, VS97, or NPE 123
| Strain used for inoculation | No. of samples positive for indicated finding/no. of pigs inoculated
|
|||||
|---|---|---|---|---|---|---|
| Clinical signs | Tissue colonization
|
Brain histopathology
|
||||
| Ileum | Cecum | Spiral colon | Microhemorrhage | Vessel necrosis | ||
| Parent | 9/11 | 3/6 | 10/11 | 11/11 | 2/11 | 8/11 |
| luxS mutant | 11/14 | 2/6 | 13/14 | 14/14 | 6/14 | 9/14 |
| NPE 123 | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 |
We hypothesized that LuxS was a critical enzyme in the production of autoinducer signals that are important for the expression of virulence factors, such as those required for A/E lesion production by E. coli O157:H7 in vitro (11, 12). This study demonstrated that A/E lesions in spiral colons occurred less frequently in gnotobiotic piglets inoculated with the luxS mutant strain than in those inoculated with the parent strain. There were no significant differences in the frequencies of lesions in the ceca and ilea. Mutation in luxS did not eliminate the ability of E. coli O157:H7 to produce A/E lesions or to cause clinical disease in piglets due to the effects of Shiga toxin. These results were consistent with a recent report by Sperandio et al. (13) demonstrating that the eukaryotic hormones epinephrine and norepinephrine activate the type III secretion system, LEE transcription, and, consequently, A/E lesion formation by the luxS mutant. Therefore, the host's epinephrine and norepinephrine may be substituting for the AI-3 bacterial signaling (13). In this study, the presence of these hormones or other host factors in the intestines of the gnotobiotic piglets presumably may have signaled to the bacteria to express the LEE region genes for the development of A/E lesions; therefore, compelling differences were not demonstrated in vivo as they were in vitro with the luxS mutant of E. coli O157:H7.
Acknowledgments
This work was supported by NIH grant 1R01DK58957, awarded to J.B.K.
We thank Lawayne Nusz and the NADC animal caretaker, Roger Spaete, for their assistance; Judy Stasko for transmission electron microscopy assistance; Richard Evans for statistical consultation; and, especially, Sheridan Booher for technical assistance in bacteriology and animal handling.
Editor: J. B. Bliska
REFERENCES
- 1.Booher, S. L., N. A. Cornick, and H. W. Moon. 2002. Persistence of Escherichia coli O157:H7 in experimentally infected swine. Vet. Microbiol. 89:69-81. [DOI] [PubMed] [Google Scholar]
- 2.Cornick, N. A., S. L. Booher, T. A. Casey, and H. W. Moon. 2000. Persistent colonization of sheep by Escherichia coli O157:H7 and other E. coli pathotypes. Appl. Environ. Microbiol. 66:4926-4934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dean-Nystrom, E. A., B. T. Bosworth, W. C. Cray, Jr., and H. W. Moon. 1997. Pathogenicity of Escherichia coli O157:H7 in the intestines of neonatal calves. Infect. Immun. 65:1842-1848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Falcao, J. P., F. Sharp, and V. Sperandio. 2004. Cell-to-cell signaling in intestinal pathogens. Curr. Issues Intest. Microbiol. 5:9-17. [PubMed] [Google Scholar]
- 5.Jordan, D. M., N. Cornick, A. G. Torres, E. A. Dean-Nystrom, J. B. Kaper, and H. W. Moon. 2004. Long polar fimbriae contribute to colonization by Escherichia coli O157:H7 in vivo. Infect. Immun. 72:6168-6171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.McDaniel, T. K., K. G. Jarvis, M. S. Donnenberg, and J. B. Kaper. 1995. A genetic locus of enterocyte effacement conserved among diverse enterobacterial pathogens. Proc. Natl. Acad. Sci. USA 92:1664-1668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Moon, H. W., and S. C. Whipp. 1971. Systems for testing the enteropathogenicity of Escherichia coli. Ann. N. Y. Acad. Sci. 176:197-211. [Google Scholar]
- 8.Moon, H. W., S. C. Whipp, R. A. Argenzio, M. M. Levine, and R. A. Giannella. 1983. Attaching and effacing activities of rabbit and human enteropathogenic Escherichia coli in pig and rabbit intestines. Infect. Immun. 41:1340-1351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sarmiento, J. I., T. A. Casey, and H. W. Moon. 1988. Postweaning diarrhea in swine: experimental model of enterotoxigenic Escherichia coli infection. Am. J. Vet. Res. 49:1154-1159. [PubMed] [Google Scholar]
- 10.Schauder, S., K. Shokat, M. G. Surette, and B. L. Bassler. 2001. The LuxS family of bacterial autoinducers: biosynthesis of a novel quorum-sensing signal molecule. Mol. Microbiol. 41:463-476. [DOI] [PubMed] [Google Scholar]
- 11.Sperandio, V., J. L. Mellies, W. Nguyen, S. Shin, and J. B. Kaper. 1999. Quorum sensing controls expression of the type III secretion gene transcription and protein secretion in enterohemorrhagic and enteropathogenic Escherichia coli. Proc. Natl. Acad. Sci. USA 96:15196-15201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sperandio, V., A. G. Torres, J. A. Girón, and J. B. Kaper. 2001. Quorum sensing is a global regulatory mechanism in enterohemorrhagic Escherichia coli O157:H7. J. Bacteriol. 183:5187-5197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sperandio, V., A. G. Torres, B. Jarvis, J. P. Nataro, and J. B. Kaper. 2003. Bacteria-host communication: the language of hormones. Proc. Natl. Acad. Sci. USA 100:8951-8956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tarr, P. I., M. A. Neill, C. R. Clausen, J. W. Newland, R. J. Neill, and S. L. Moseley. 1989. Genotypic variation in pathogenic Escherichia coli O157:H7 isolated from patients in Washington, 1984-1987. J. Infect. Dis. 159:344-347. [DOI] [PubMed] [Google Scholar]
- 15.Tzipori, S., H. Karch, K. I. Wachsmuth, R. M. Robins-Browne, A. D. O'Brien, H. Lior, M. L. Cohen, J. Smithers, and M. M. Levine. 1987. Role of a 60-megadalton plasmid and Shiga-like toxins in the pathogenesis of infection caused by enterohemorrhagic Escherichia coli O157:H7 in gnotobiotic piglets. Infect. Immun. 55:3117-3125. [DOI] [PMC free article] [PubMed] [Google Scholar]