Abstract
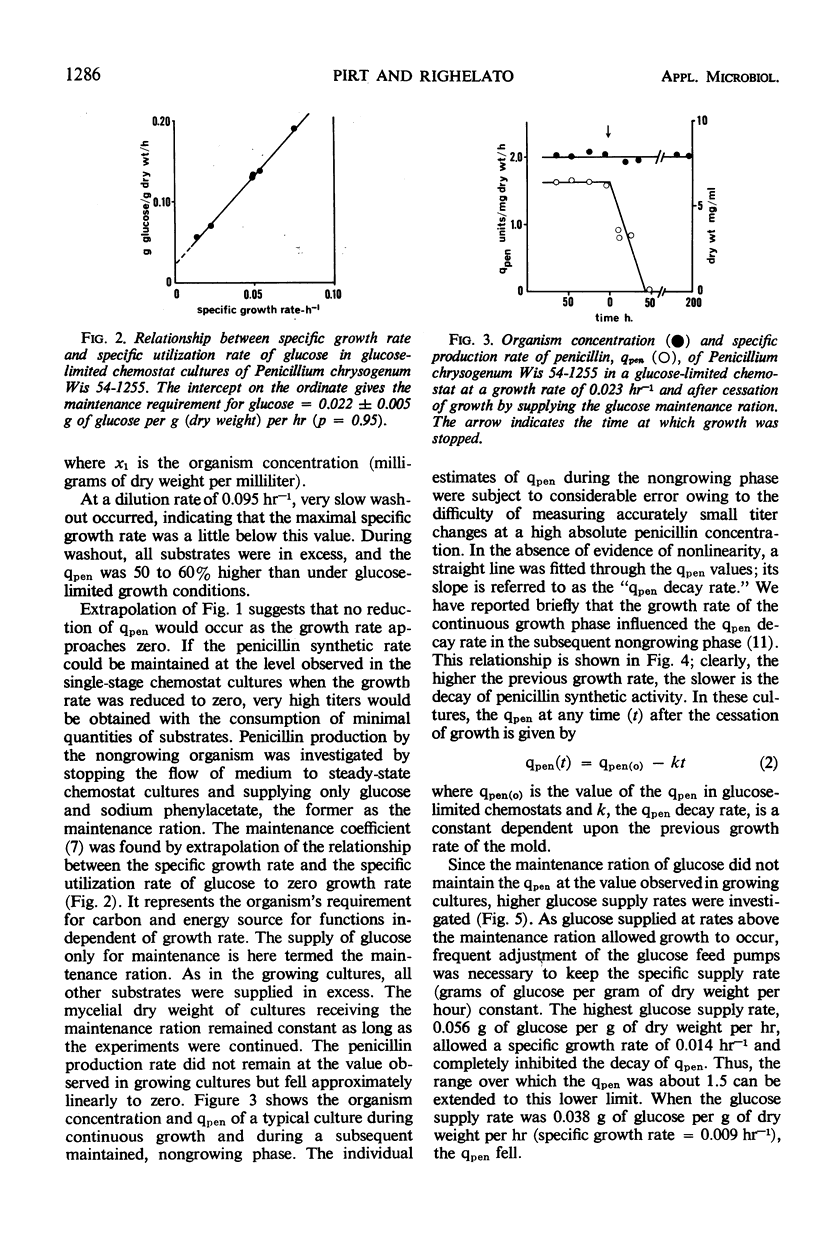
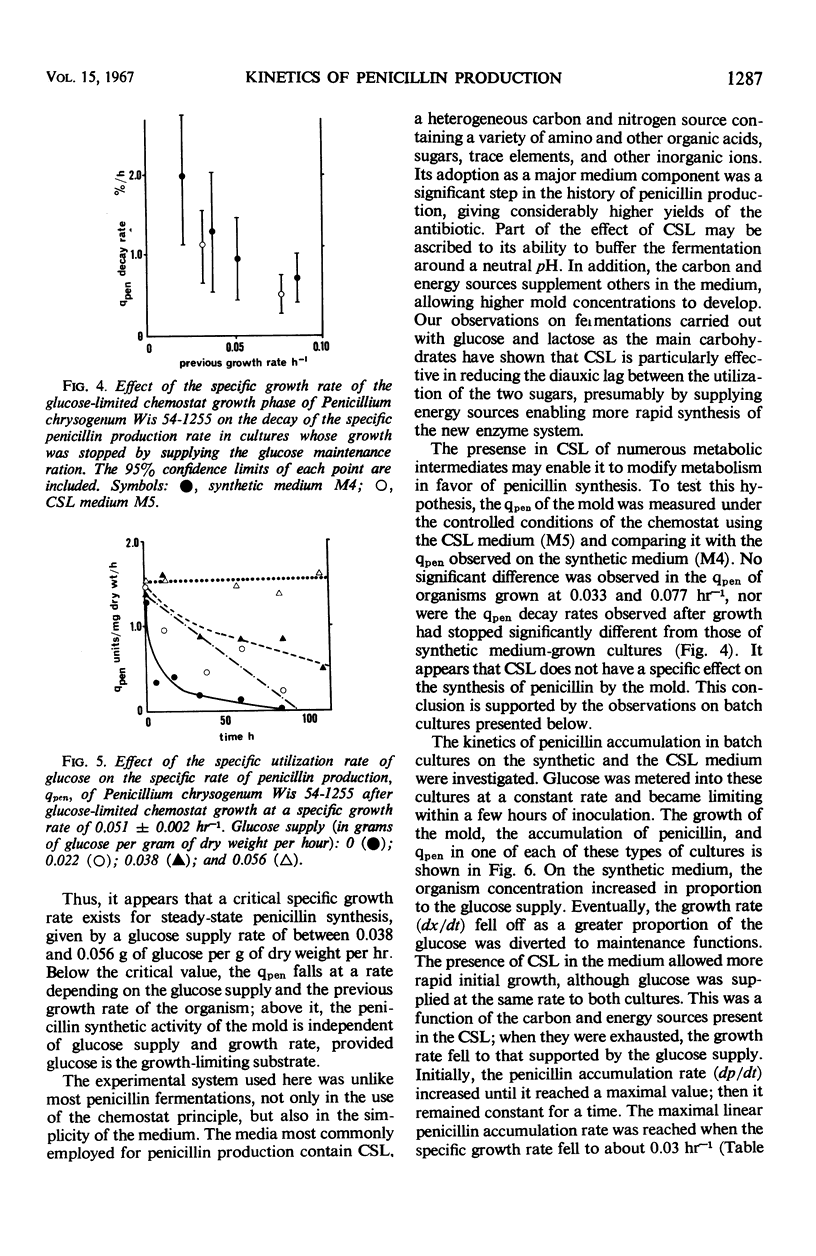
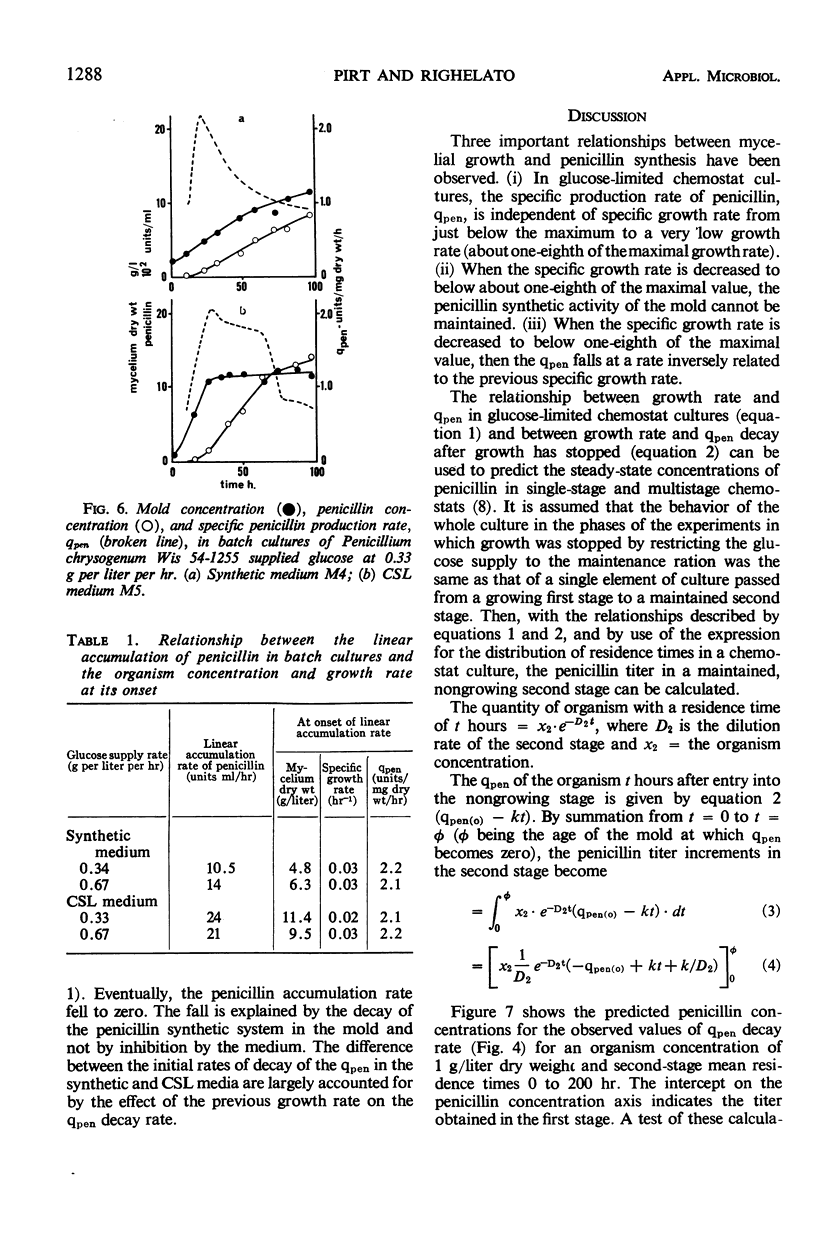
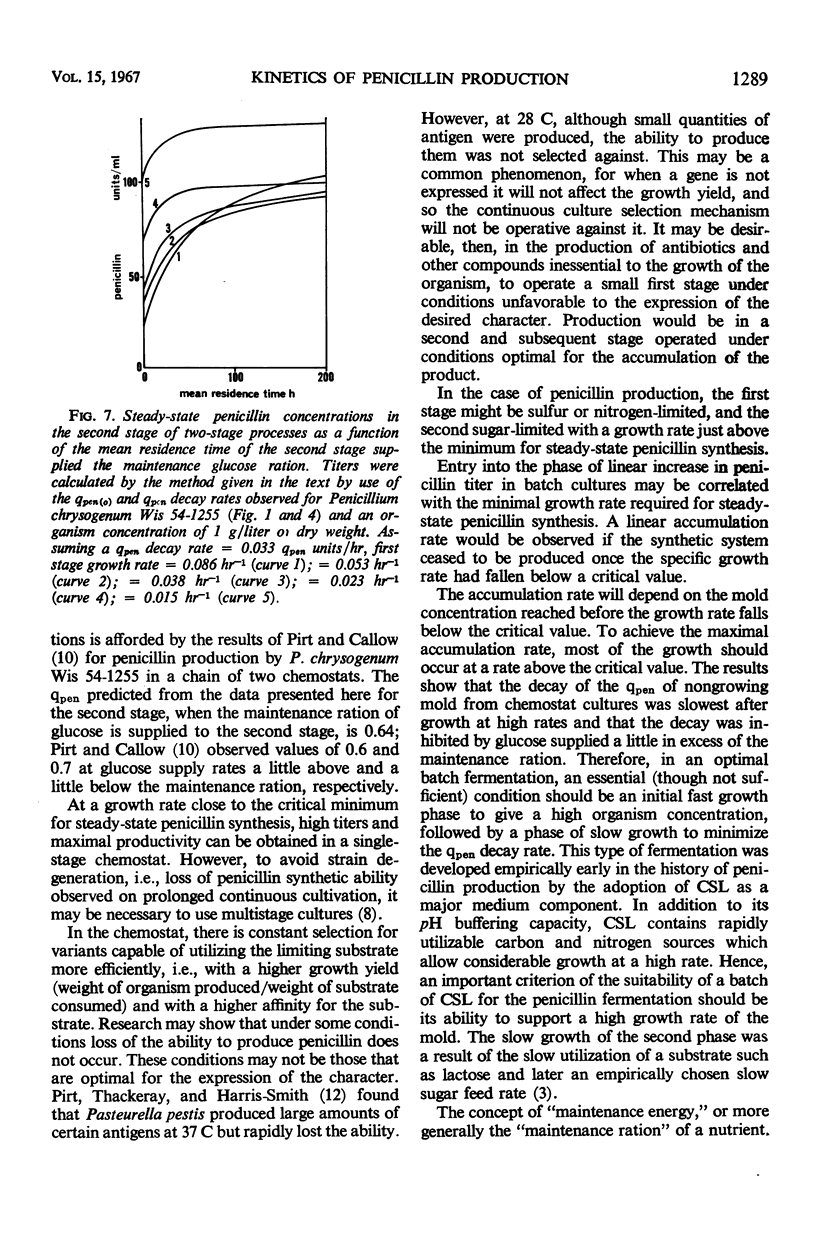
The kinetics of penicillin production by Penicillium chrysogenum Wis 54-1255 in a glucose-limited chemostat and in batch cultures are reported. The specific production rate of penicillin, qpen (units per milligram of dry weight per hour) was independent of specific growth rate over the range 0.014 to 0.086 hr-1. Growth was stopped by restricting the glucose supply to the “maintenance ration,” that is, the glucose requirement of the organism at zero growth rate with all other nutrients in excess. Under such conditions, the organism dry weight remained constant, but the qpen fell approximately linearly to zero at a rate inversely related to the previous growth rate. Glucose supplied in excess of the maintenance ration inhibited the decay of qpen. At a critical growth rate between 0.009 and 0.014 hr-1, the decay was completely inhibited. Quantitative expressions for the qpen of growing and nongrowing cultures were derived and used to predict the steady-state concentrations of penicillin accumulating in one- and two-stage continuous processes. A rational explanation of the kinetics of penicillin accumulation in batch cultures is given, relating the rate of penicillin synthesis to growth rate. It is concluded that an important role of corn steep liquor (CSL), a heterogeneous carbon and nitrogen source commonly used in penicillin production media, is the provision of substrates which allow a high concentration of mold to be reached before the growth rate falls below the critical value. CSL had no significant effect on qpen.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- MAXON W. D. Continuous fermentation; a discussion of its principles and applications. Appl Microbiol. 1955 Mar;3(2):110–120. doi: 10.1128/am.3.2.110-122.1955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moyer A. J., Coghill R. D. Penicillin: VIII. Production of Penicillin in Surface Cultures. J Bacteriol. 1946 Jan;51(1):57–78. [PMC free article] [PubMed] [Google Scholar]
- PIRT S. J., CALLOW D. S. The production of penicillin by continuous-flow fermentation. Sci Rep Ist Super Sanita. 1961 Jul;1:250–259. [PubMed] [Google Scholar]
- PIRT S. J., THACKERAY E. J., HARRIS-SMITH R. The influence of environment on antigen production by Pasteurella pestis studied by means of the continuous flow culture technique. J Gen Microbiol. 1961 May;25:119–130. doi: 10.1099/00221287-25-1-119. [DOI] [PubMed] [Google Scholar]
- Pirt S. J. The maintenance energy of bacteria in growing cultures. Proc R Soc Lond B Biol Sci. 1965 Oct 12;163(991):224–231. doi: 10.1098/rspb.1965.0069. [DOI] [PubMed] [Google Scholar]
- Righelato R. C., Pirt S. J. Improved control of organism concentration in continuous culture of filamentous micro-organisms. J Appl Bacteriol. 1967 Apr;30(1):246–250. doi: 10.1111/j.1365-2672.1967.tb00295.x. [DOI] [PubMed] [Google Scholar]
- Whiffen A. J., Savage G. M. The Relation of Natural Variation in Penicillium notatum to the Yield of Penicillin in Surface Culture. J Bacteriol. 1947 Feb;53(2):231–240. doi: 10.1128/jb.53.2.231-240.1947. [DOI] [PMC free article] [PubMed] [Google Scholar]


