Abstract
Lasik is a common surgery for treating anisometropia. In this study, we asked a specific question: what’s the effect of Lasik surgery on anisometropes’ sensory eye dominance ? Fifteen myopic anisometropes (mean age: 23 ± 6.9 years old; 6 females) participated in our experiment. We quantitatively measured participants’ sensory eye dominance before and after the Lasik surgery using a binocular phase combination paradigm. We found no significant change of sensory eye dominance within 16 weeks (measured between 8 to 96 days, for one or two repetitions) after the surgery (t(14) = −1.44, p = 0.17). A further following on eight patients showed that patients’ two eyes were much more balanced at 16 weeks or more (measured one or two times between 112 to 408 days) after the surgery (t(7) = −3.79, p = 0.007). Our results suggest that the benefit of Lasik surgery on anisometropes’ sensory eye dominance is not immediate, a long-term ‘adaptation’ period (16 weeks or more) is necessary to enable the surgery to be truly effective.
Introduction
Myopia is a common prevalent disorder, such a problem is particularly pronounced1 and affecting up to 90% of teenagers and young adults in China2. In some cases, patients’ two eyes have different degree of myopia, i.e, myopic anisometropia3, 4. Laser in-situ keratomileusis (LASIK) has become a popular method to treat anisometropia. Several postoperative evaluation studies have been conducted, which showed that LASIK surgery is safe and effective in reducing patients’ spherical equivalent manifest refraction and increasing their visual acuity5, but may increase the 3rd and higher order aberrations6 or there may be a risk of postoperative diplopia7.
On the other hand, anisometropia has been suggested to be an important cause of abnormal binocularity8, 9. Recently, Zhou et al.10 also showed that patients who are corrected for myopic anisometropia have abnormal sensory eye dominance, but that this improves during 16 or more weeks of refractive adaptation. Their results suggest that refractive adaptation may play a critical role in partially restoring the sensory eye balance in corrected anisometropes. In spite of many previous studies in anisometropia and binocularity, to our knowledge, there is no direct information on how sensory eye balance is affected after LASIK surgery. Therefore, we conducted a cohort study to quantitatively evaluate the postoperative effect of LASIK surgery on myopic anisometropes’ sensory eye dominance.
Results
In the experiment, we used a binocular phase combination paradigm to quantitatively access the sensory eye dominance of our participates. In particular, observers’ sensory eye dominance was quantified by the interocular contrast ratio that was needed when the two eyes were balanced in binocular phase combination (Fig. 1). Figure 2 shows individuals’ effective contrast ratio corresponding to their balance points before and after surgery. As is shown in Fig. 2, all the patients’ pre-surgery balance points (marked as open symbols) were lower than the ideal normal level (i.e., 1.0; marked as dashed line in each panel). This result is consistent with a recent report10, which suggests that anisometropes exhibit interocular sensory imbalances. Within 16 weeks after the surgery, the balance points (marked as black filled symbols) didn’t change much for most of the patients. A longer-term follow up in eight patients showed that patients’ balance points (marked as red filled symbols) were much closed to the ideal normal level, indicating improved interocular balance.
Figure 1.
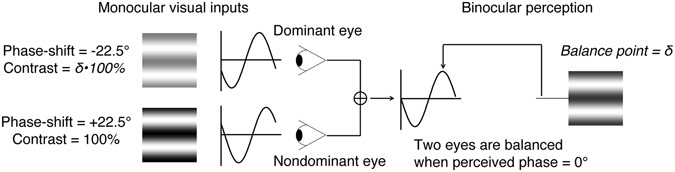
Illustration of the binocular phase combination paradigm. Two horizontal sine-wave gratings with equal and opposite phase-shifts of 22.5° (relative to the center of the screen) were dichoptically presented to the two eyes through the polarized glasses. The perceived phase of the cyclopean grating depends on the contribution of the two eyes in binocular phase combination. Sensory eye dominance is quantified by the interocular contrast difference that is needed to achieve a 0-degree of perceived phase, i.e., the balance point, where the two eyes are balanced in binocular phase combination.
Figure 2.
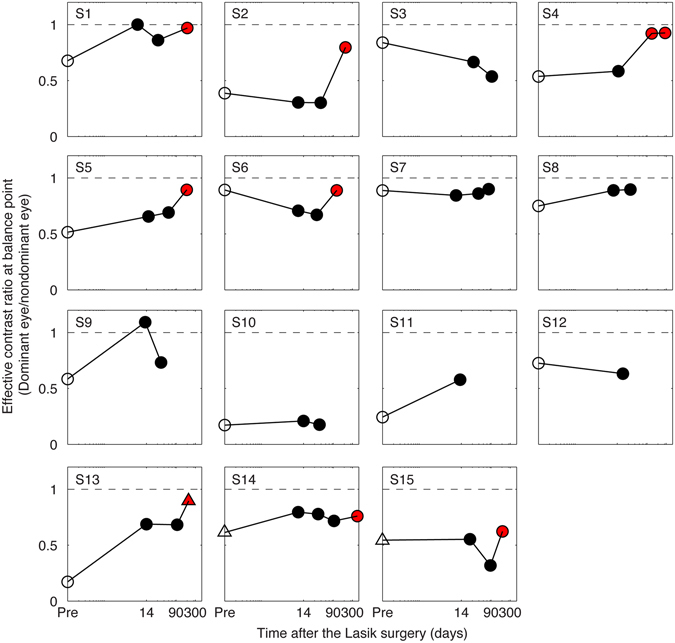
Patients’ sensory eye dominance before and after the surgery. Patients’ sensory eye dominance were quantified by individuals’ effective contrast ratio at balance point (i.e., the interocular contrast ratio that is needed when the two eyes are balanced in binocular phase combination) before and after surgery. The pre-surgery measurement is marked as open symbols; the post-surgery measurement that was measured within 16 weeks after the surgery is marked as black filled symbols and the post-surgery measurement that was measured at or longer than 16 weeks after the surgery is marked as red filled symbols. Except subjects S13, S14 and S15, who had a switch of eye dominance in one of the test sessions (marked as triangle symbols), most observers had consistent nondominant eye during this study.
To better show the change of balance points within 16 weeks after the surgery, we plotted individuals’ balance points before and after surgery in Fig. 3. It is obvious that the balance points within 16 weeks after the surgery were consistent with that of pre-surgery. A 2-tailed paired-samples t-test also show that there was no significant change of balance points within 16 weeks after the surgery: t(14) = −1.44, p = 0.17.
Figure 3.
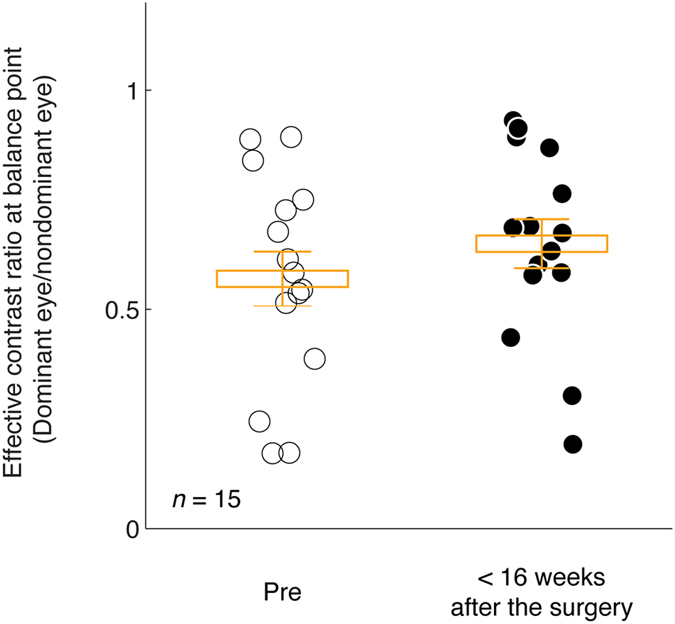
Sensory eye dominance before and within 16 weeks after the surgery. Each point represents the results of one subject. The average balance point and it’s standard error were marked using box and error bars for each time session.
To better illustrate the long-term effect of surgery on patients’ sensory eye dominance, we plotted patients’ balance points that were measured within 16 weeks after the surgery compared with those measured at 16 weeks and longer after the surgery in Fig. 4. Eight patients participated in this additional long-term following study. Most of the patients had an improved balance point at or longer than 16 weeks after the surgery. A 2-tailed paired-samples t-test also show that there was a significant change of balance points at these two time sessions: t(7) = −3.79, p = 0.007.
Figure 4.
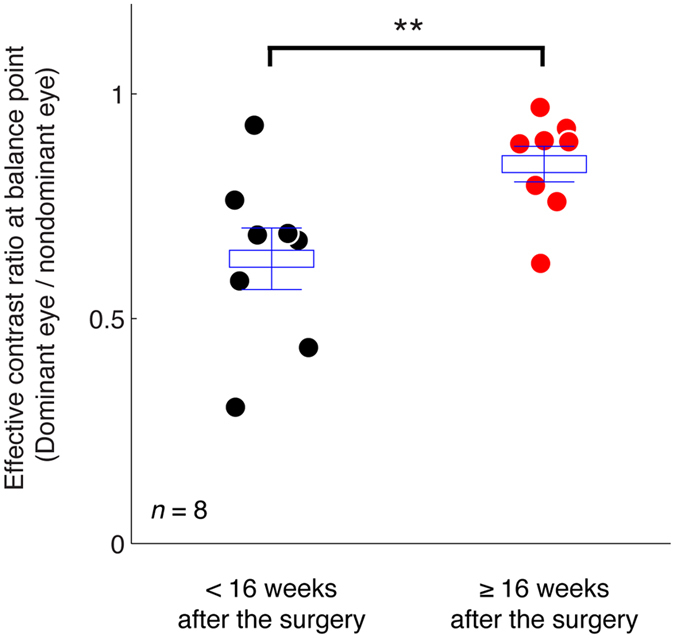
The long-term effect of surgery on patients’ sensory eye dominance. Each point represents the results of one subject. The average balance point and it’s standard error were marked using box and error bars for each time session. Eight patients participated in this study. **p < 0.01, 2-tailed paired-samples t-test.
Discussion
We show that LASIK surgery doesn’t produce an immediate effect on myopic anisometropes’ sensory eye dominance; the postoperative effect needs 16 weeks or more to be effective. It should be noted that while the first part of conclusions was derived from 15 myopic anisometropes’ results (Fig. 3), the second part of conclusions was derived from only 8 myopic anisometropes’ results (Fig. 4), as other patients didn’t participated in this long-term following due to personal reasons. We are therefore limited in what we can conclude about postoperative changes in all anisometropes. However, what we can conclude is that LASIK surgery doesn’t produce an immediate effect on myopic anisometropes’ sensory eye dominance. Nevertheless, our results are consistent with the recent report by Zhou et al.10, suggesting that a period of refractive adaptation (>16 weeks) is critical after the correction of anisometropia for improved interocular balance.
We noticed that, even after 16 or more weeks, there were still some patients who had abnormal sensory eye dominance (balance point less than 0.9). It is possible that in these cases that a longer period of refractive adaptation was required or alternatively the remaining eye imbalance may remain permanently after Lasik surgery. On the basis of our previous results on optically corrected anisometropes10 and what is known of refractive adaptation11, the latter conclusion is the more likely. Such a residual eye imbalance is more likely to be due to a neuronal change in ocular dominance than have a purely optical explanation as any interocular differences in higher order aberrations not corrected by or induced by Lasik surgery12 should involve mid to high spatial frequencies13 (>5 c/d) and not the very low spatial frequency (1 c/d) used for these measurements of eye balance.
In future studies, it will be interesting to see whether the residual imbalances in ocular dominance that remain after 16 weeks of refractive adaptation can recover after much longer duration of refractive adaptation, or whether they could be able to be rectified by a dichoptic training protocol that has proven successful in rebalancing the eyes of amblyopic adults. It involves playing dichoptic videogames14–16 or watching dichoptic movies17 where the contrast balance between the eyes is adjusted over time. Typically a 2–6 week period of 1 hr a day is sufficient to balance the eyes and provide better binocular vision and improved stereopsis.
Methods
Participants
Fifteen myopic anisometropes (mean age: 23 ± 6.9 years old; 6 females) participated in our experiment. They were recruited from the department of Ophthalmology of the First Affiliated Hospital of Anhui Medical University (Anhui, China). Anisometropia is defined as a 1.50D or larger interocular spherical difference. All participants have normal or corrected to normal visual acuity (LogMAR < 0.10) in the two eyes, no strabismus and no history of eye pathology before the study. Clinical details of patients before surgery are provided in Table 1. Individuals’ sensory eye dominance was assessed before and after the surgery at different time points. Observers wore their prescribed optical correction, if needed, for the data collection.
Table 1.
Clinical details of the participants.
| Subject# | Age (years old) | Gender | Right eye | Left eye | ||||
|---|---|---|---|---|---|---|---|---|
| Refractive errors | Visual acuity (LogMAR) | Refractive errors | Visual acuity (LogMAR) | |||||
| Uncorrected | Corrected | Uncorrected | Corrected | |||||
| S1 | 18 | Male | −4.25 | 0.70 | 0.00 | −1.75 | 0.52 | 0.00 |
| S2 | 19 | Male | −4.50/−1.25@10 | 1.10 | 0.00 | −9.50/−3.25@155 | 1.70 | 0.00 |
| S3 | 20 | Male | −3.75 | 0.92 | 0.00 | −5.75 | 1.10 | 0.00 |
| S4 | 18 | Male | −5.75/−1.25@15 | 1.22 | 0.00 | −3.75/−2.50@170 | 1.00 | 0.10 |
| S5 | 18 | Female | −5.25/−0.25@15 | 0.92 | 0.00 | −3.00/−0.50@10 | 0.92 | 0.00 |
| S6 | 27 | Male | −3.25/−1.00@180 | 0.92 | 0.00 | −5.50/−0.50@10 | 1.30 | 0.00 |
| S7 | 19 | Female | −1.00 | 0.70 | 0.00 | −7.00/−0.50@100 | 1.70 | 0.00 |
| S8 | 20 | Male | −4.00/−3.25@10 | 1.70 | 0.00 | −7.00/−2.75@5 | 1.70 | 0.00 |
| S9 | 32 | Female | −6.00/−1.75@170 | 1.00 | 0.10 | −11.00/−1.50@5 | 1.22 | 0.10 |
| S10 | 43 | Female | Plano | 0.10 | 0.10 | −3.50/−1.00@160 | 1.00 | 0.10 |
| S11 | 25 | Male | −1.25/−3.00@180 | 0.60 | 0.10 | −3.50/−4.00@170 | 0.60 | 0.10 |
| S12 | 23 | Male | −2.75/−1.00@105 | 0.92 | 0.00 | Plano | 0.00 | 0.00 |
| S13 | 25 | Female | −9.00/−0.50@90 | 1.52 | 0.00 | Plano | 0.10 | 0.10 |
| S14 | 20 | Female | −6.75/−1.75@150 | 1.40 | 0.00 | −4.25/−1.25@10 | 1.30 | 0.00 |
| S15 | 18 | Male | −5.50 | 1.00 | 0.00 | −9.00/−1.75@170 | 1.00 | 0.00 |
All subjects were naive as to the purpose of the experiment. A written informed consent was obtained from each of them after explanation of the nature and possible consequences of the study. This study complied with the Declaration of Helsinki and was approved by the Institutional Review Boards of Anhui Medical University, Wenzhou Medical University and McGill University.
Apparatus
All measurements were conducted on a PC computer running Matlab (MathWorks, Inc., Natick, MA) with PsychToolBox 3.0.9 extensions18, 19. The stimuli were presented on a gamma-corrected LG D2342PY 3D LED screen (LG Life Science, Korea) with a 1920 × 1080 resolution and a 60 Hz refresh rate. Subjects viewed the display dichoptically with polar glasses in a dimly lit room at a viewing distance of 136 cm. The background luminance was 46.2 cd/m2 on the screen and 18.8 cd/m2 through the polar glasses. A chin-forehead rest was used to minimize head movements during the experiment.
Lasik Surgery
All the patients underwent a series examination before femto-LASIK surgery. The examination included uncorrected distance visual acuity, slit-lamp microscopy, corneal topography, manifest and cycloplegic refraction, corrected distance visual acuity and fundus examination. All the surgeries were performed by the fifth author of this paper (Dr. Liao). During this research, WaveLight FS200 femtosecond laser was used to create superior hinged flaps, with 110 µm thickness and 8.5 mm diameter for the myopic eyes. After the flap was lifted, ablations were performed using Alcon WaveLight EX200 excimer laser (Fort Worth, TX, USA) with a 0.5-mm transition zone and 6.0-mm optical zone.
Design
We quantitatively accessed all the 15 anisometropes’ sensory eye dominance before the surgery and within 16 weeks after the surgery. The time for post-surgery measure ranged from 8 to 96 days for one or two measurement repetitions at the patients’ convenience. Eight patients participated in an additional, long-term follow up investigation at 16 weeks or longer after the surgery, ranged from 112 to 408 days (due to personal reasons, other patients didn’t participated in this long-term following).
A binocular phase combination paradigm20, 21 was used for measuring sensory eye dominance. The design was same as the one we used in previous studies22–24, in which observers were asked to dichoptically view two horizontal sine-wave gratings (spatial frequency: 1 cycle/°; size: 2° × 2°) having equal and opposite phase-shifts of 22.5° (relative to the center of the screen) through polarized glasses; the contrast of the nondominant eye was fixed at 100% and the contrast of the dominant eye was varied by an interocular contrast ratio δ = [0, 0.1, 0.2, 0.4, 0.8, 1.0] in different trials; the perceived phase of the grating in the cyclopean percept was measured as a function of the interocular contrast ratio. By this method, we were able to find a specific interocular contrast ratio where the perceived phase of the cyclopean grating was 0, indicating equal contribution of the two eyes to the binocular percept. This specific interocular contrast ratio is termed the “balance point” for binocular phase combination since the two eyes under these stimulus conditions are balanced in binocular viewing (Fig. 1). Similar to previous studies20–26, two configurations were used for each interocular contrast ratio in the measurement so that any potential positional bias will be cancelled out: in one configuration, the phase-shift was +22.5° in the nondominant eye and −22.5° in the dominant eye and in the other, the reverse. The perceived phase of the cyclopean grating at each interocular contrast ratio (δ) was quantified by half of the difference between the measured perceived phases in these two configurations. Different conditions (configurations and interocular contrast ratios) were randomized in different trials. The perceived phase and its standard error were calculated based on eight measurement repetitions.
Before the start of data collection, proper demonstrations of the task were provided by practice trials to ensure observers understood the task. The nondominant eye was selected based on individuals’ performance in the practice session: the eye that was less dominant in the binocular phase combination task when the two eye had same strength of input (i.e., 100% of contrast) was chosen as the nondominant eye and would have a constant contrast input of 100% during the test. Except subjects S13, S14 and S15, who had a switch of eye dominance in one of the test sessions (marked as triangle symbols in Fig. 2), most observers had consistent nondominant eye during this study.
Procedure
We used the same phase adjustment procedure as used by Huang et al.21 for measuring the perceived phase of the binocularly combined grating. In each trial, observers were asked firstly to align the stimuli from the two eyes; they were then instructed to adjust the position of a reference line to indicate the perceived phase of the binocularly combined grating. Surrounding the gratings, a high-contrast frame (width, 0.11°; length, 6°) with four white diagonal lines (width, 0.11°; length, 2.83°) was always presented during the test to help observers maintain fusion. The gratings had a period of 2 cycles corresponding to 180 pixels, thus the phase adjustment had a step size of 4 degrees of phase per pixel.
Curve fits
Individuals’ effective contrast ratio at balance point were calculated by fitting the perceived phase (φ) versus interocular contrast ratios (δ) functions (i.e., the PvR functions), with a modified contrast-gain control model from Huang et al.21:
| 1 |
In which, bp and γ are two free parameters. ‘bp’ represents the interocular contrast ratio when the two eyes make equally contributions to binocular combination (i.e., the balance point) and ‘γ’ represents a non-linear factor. Curve fitting was conducted in Matlab (MathWorks, Natick, MA) using the nonlinear least squares method to minimized ∑(φ theory − φ observed)2.
Acknowledgements
This work was supported by the National Natural Science Foundation of China grants NSFC 81300796 to LF and NSFC 81500754 to JZ, the Wenzhou Medical University grant QTJ16005 to JZ and Canadian Institutes of Health Research Grants MOP-53346, CCI-125686 and MT-10818, and an ERA-NET Neuron grant (JTC2015) to RFH.
Author Contributions
L.F., J.Z., and R.-F.H. conceived the experiments. H.L., Y.C., J.W., and R.L. performed the experiments. L.F., Y.W., J.Z., and R.-F.H. analyzed the data, interpreted the data and wrote the manuscript. All authors reviewed the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Lixia Feng, Email: lixiafeng@163.com.
Jiawei Zhou, Email: zhoujw@mail.eye.ac.cn.
References
- 1.Foster PJ, Jiang Y. Epidemiology of myopia. Eye. 2014;28:202–208. doi: 10.1038/eye.2013.280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dolgin E. The Myopia Boom. Nature. 2015;519:276–278. doi: 10.1038/519276a. [DOI] [PubMed] [Google Scholar]
- 3.Weale RA. On the age-related prevalence of anisometropia. Ophthalmic Res. 2002;34:389–392. doi: 10.1159/000067040. [DOI] [PubMed] [Google Scholar]
- 4.Vincent SJ, Collins MJ, Read SA, Carney LG. Myopic anisometropia: ocular characteristics and aetiological considerations. Clinical and Experimental Optometry. 2014;97:291–307. doi: 10.1111/cxo.12171. [DOI] [PubMed] [Google Scholar]
- 5.Rashad KM. Laser in situ keratomileusis for myopic anisometropia in children. Journal of refractive surgery. 1999;15:429–435. doi: 10.3928/1081-597X-19990701-07. [DOI] [PubMed] [Google Scholar]
- 6.Moreno-Barriuso E, et al. Ocular aberrations before and after myopic corneal refractive surgery: LASIK-induced changes measured with laser ray tracing. Invest Ophthalmol Vis Sci. 2001;42:1396–1403. [PubMed] [Google Scholar]
- 7.Valente P, et al. Refractive surgery in patients with high myopic anisometropia. Journal of refractive surgery. 2006;22:461–466. doi: 10.3928/1081-597X-20060501-07. [DOI] [PubMed] [Google Scholar]
- 8.Weakley DR. The association between nonstrabismic anisometropia, amblyopia, and subnormal binocularity. Ophthalmology. 2001;108:163–171. doi: 10.1016/S0161-6420(00)00425-5. [DOI] [PubMed] [Google Scholar]
- 9.Brooks SE, Johnson D, Fischer N. Anisometropia and Binocularity. Ophthalmology. 1996;103:1139–1143. doi: 10.1016/S0161-6420(96)30555-1. [DOI] [PubMed] [Google Scholar]
- 10.Zhou J, Feng L, Lin H, Hess RF. On the Maintenance of Normal Ocular Dominance and a Possible Mechanism Underlying Refractive Adaptation. Invest Ophthalmol Vis Sci. 2016;57:5181–5185. doi: 10.1167/iovs.16-19696. [DOI] [PubMed] [Google Scholar]
- 11.Stewart CE, Moseley MJ, Fielder AR, Stephens DA, Cooperative M. Refractive adaptation in amblyopia: quantification of effect and implications for practice. Br J Ophthalmol. 2004;88:1552–6. doi: 10.1136/bjo.2004.044214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Al-Zeraid, F. M. & Osuagwu, U. L. Induced Higher-order aberrations after Laser In Situ Keratomileusis (LASIK) Performed with Wavefront-Guided IntraLase Femtosecond Laser in moderate to high Astigmatism. Bmc Ophthalmol16, doi:10.1186/S12886-016-0205-5 (2016). [DOI] [PMC free article] [PubMed]
- 13.Williams D, et al. Visual benefit of correcting higher order aberrations of the eye. Journal of refractive surgery. 2000;16:S554–S559. doi: 10.3928/1081-597X-20000901-12. [DOI] [PubMed] [Google Scholar]
- 14.Li J, et al. Dichoptic training enables the adult amblyopic brain to learn. Curr Biol. 2013;23:R308–309. doi: 10.1016/j.cub.2013.01.059. [DOI] [PubMed] [Google Scholar]
- 15.Li S, et al. A binocular iPad treatment for amblyopic children. Eye. 2014;28:1246–1253. doi: 10.1038/eye.2014.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.To L, et al. A game platform for treatment of amblyopia. IEEE Transactions on Neural Systems and Rehabilitation Engineering. 2011;19:280–289. doi: 10.1109/TNSRE.2011.2115255. [DOI] [PubMed] [Google Scholar]
- 17.Li SL, et al. Dichoptic movie viewing treats childhood amblyopia. Journal of Aapos. 2015;19:401–405. doi: 10.1016/j.jaapos.2015.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pelli DG. The VideoToolbox software for visual psychophysics: Transforming numbers into movies. Spatial Vision. 1997;10:437–442. doi: 10.1163/156856897X00366. [DOI] [PubMed] [Google Scholar]
- 19.Brainard DH. The Psychophysics Toolbox. Spatial Vision. 1997;10:433–436. doi: 10.1163/156856897X00357. [DOI] [PubMed] [Google Scholar]
- 20.Ding J, Sperling G. A gain-control theory of binocular combination. Proc Natl Acad Sci USA. 2006;103:1141–1146. doi: 10.1073/pnas.0509629103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Huang CB, Zhou J, Lu ZL, Feng L, Zhou Y. Binocular combination in anisometropic amblyopia. J Vis. 2009;9(17):1–16. doi: 10.1167/9.3.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhou J, Huang P-C, Hess RF. Interocular suppression in amblyopia for global orientation processing. J Vis. 2013;13(19):1–14. doi: 10.1167/13.5.19. [DOI] [PubMed] [Google Scholar]
- 23.Zhou J, Jia W, Huang C-B, Hess RF. The effect of unilateral mean luminance on binocular combination in normal and amblyopic vision. Sci Rep. 2013;3(2012):1–7. doi: 10.1038/srep02012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Feng L, Zhou J, Chen L, Hess RF. Sensory eye balance in surgically corrected intermittent exotropes with normal stereopsis. Sci Rep. 2015;5:13075. doi: 10.1038/srep13075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhou J, Georgeson MA, Hess RF. Linear binocular combination of responses to contrast modulation: Contrast-weighted summation in first- and second-order vision. J Vis. 2014;14(24):1–19. doi: 10.1167/14.13.24. [DOI] [PubMed] [Google Scholar]
- 26.Zhou J, Liu R, Zhou Y, Hess RF. Binocular combination of second-order stimuli. PLoS One. 2014;9:e84632. doi: 10.1371/journal.pone.0084632. [DOI] [PMC free article] [PubMed] [Google Scholar]


